Abstract
Candida albicans is a significant cause of disease in immunocompromised humans. Because the number of people infected by fungal pathogens is increasing, strategies are being developed to target RNAs in fungi. This work shows that oligonucleotides can serve as therapeutics against C. albicans. In particular, oligonucleotides are taken up from cell culture medium in an energy-dependent process. After uptake, oligonucleotides, including RNA, remain mostly intact after 12 h in culture. For culture conditions designed for mammalian cells, intracellular concentrations of oligonucleotides in C. albicans exceed those in COS-7 mammalian cells, suggesting that uptake can provide selective targeting of fungi over human cells. A 19-mer 2′OMe (oligonucleotide with a 2′-O-methyl backbone) hairpin is described that inhibits growth of a C. albicans strain at pH < 4.0. This pH is easily tolerated in some parts of the body subject to C. albicans infections. In vivo dimethyl sulfate modification of ribosomal RNA and the decreased rate of protein synthesis suggest that this hairpin's activity may be due to targeting the ribosome in a way that does not depend on base pairing. Addition of anti-C. albicans oligonucleotides to COS-7 mammalian cells has no effect on cell growth. Evidently, oligonucleotides can selectively serve as therapeutics toward C. albicans and, presumably, other pathogens. Information from genome sequencing and functional genomics studies on C. albicans and other pathogens should allow rapid design and testing of other approaches for oligonucleotide therapies.
The fungus Candida albicans infects humans and is particularly pernicious for immunocompromised hosts, such as cancer and AIDS patients. Because the number of humans infected by fungi and the occurrence of resistant strains are increasing, finding new compounds to treat these infections is important (1). In vitro results suggest that oligonucleotides are promising compounds for targeting RNA in fungal pathogens (2, 3).
Oligonucleotides have several advantages for targeting RNA. Pairing rules between an oligonucleotide and RNA facilitate rational design. Gene expression can be reduced by RNase H cleavage of target RNA (4) or by blocking mRNA translation (5). Oligonucleotides can also serve as suicide inhibitors (2, 3) or misfold RNAs (6). Nucleic acids are easily synthesized (7), which allows rapid testing of oligonucleotides of different composition. Properties such as higher affinity and nuclease stability can be programmed into oligonucleotides by modifying the backbone, sugar, or base (8).
The use of oligonucleotides in cell culture poses several challenges. For example, uptake into mammalian cells is not efficient and oligonucleotides are often degraded after entering cells (4). Transfection agents, small molecules, and peptides have been used to circumvent these problems in mammalian cells (9). Little is known, however, about oligonucleotide uptake into other organisms, such as fungi. Cell membranes and metabolism differ substantially between fungi and mammalian cells. Thus, uptake and metabolism of oligonucleotides may differ.
This paper reports that oligonucleotides are readily taken into C. albicans in an energy-dependent manner, producing intracellular concentrations as much as 100-fold higher than the concentrations in the media. Surprisingly, oligonucleotides, including RNA, are not significantly degraded in C. albicans after incubations of 12 h. Also, the intracellular concentration due to oligonucleotide uptake by COS-7 mammalian cells is at least 10-fold lower than for C. albicans. Thus, uptake may provide selectivity for targeting fungi with oligonucleotides.
An oligonucleotide is described that is a mimic of a ribosomal RNA (rRNA) hairpin. This oligonucleotide inhibits growth of a C. albicans strain below pH 4.0. In vivo dimethyl sulfate (DMS) modification of rRNA and rates of protein synthesis suggest that the inhibitory effect may be due to targeting of the ribosome. These results lay the foundation for development of oligonucleotide therapeutics toward C. albicans and other fungi by exploiting the information from genome sequencing and functional genomics projects (10).
Materials and Methods
General Procedures.
Oligonucleotides were synthesized, purified as described (11), and sterilized by filtration. Concentrations were determined from absorbance at 80°C (12). C. albicans strains were clinical isolates from the University of Rochester Medical Center.
Media were supplemented with 10 μg/ml ampicillin and sterilized by filtration after the pH was adjusted by addition of HCl. Cultures were grown in 15-ml vials. After the addition of oligonucleotide and several hours of cell growth, the pH of the media did not change. Miconazole was dissolved in DMSO. Cultures were grown at 37°C with vigorous shaking. Optical melting experiments were performed as described (13).
Oligonucleotide Uptake and Stability.
C. albicans strains were grown overnight in yeast extract/peptone/dextrose (YPD) medium and placed into yeast extract nitrogen base (YNB) medium at an initial absorbance of 0.01 at 540 nm (OD540) with 1 μM unlabeled oligonucleotide and trace 5′ end 32P-labeled oligonucleotide. Cultures were incubated for ≈6 h. The number of cells in culture was determined from serial dilutions with sterile water and plating onto YPD plates; the number of cells determined by this method is approximately equal to values from hemocytometry. Cells were harvested by centrifugation and washed with sterile water or 0.05% Triton X-100 three times; with either treatment, little radioactivity was washed from the cells. The amount of cell-associated radioactivity was determined by scintillation counting. For calculation of intracellular concentration, a cell diameter of 4 μm was assumed.
To determine whether oligonucleotides are stable once they enter C. albicans, cultures were grown with 1 μM oligonucleotide for 12 h. Cells were harvested and washed to remove radioactivity not cell associated. Total nucleic acid was isolated using 1 ml of Triazol (GIBCO) and by vortexing cells with 100 μl of glass beads (Sigma, 4.25–6 μm). The nucleic acid pellet was separated by denaturing 20% PAGE.
Rate and energy dependence of uptake experiments were run in 20% YNB medium diluted with PBS at pH 2.5 with or without 0.5% NaN3. The starting OD540 was 1.0. Energy-dependence experiments were incubated for 1 h.
Inhibition of Cell Growth.
Cultures were grown in the presence or absence of oligonucleotide in YNB medium with a starting OD540 of 0.01 at various pH. When cultures without oligonucleotide reached ≈0.6 OD540, the OD540 in each culture was measured. Experiments were run at least in duplicate. For 2′OMe (oligonucleotide with a 2′-O-methyl backbone) HP1 (hairpin mimic of LSU rRNA sequence) and 2′OMe Rev HP1 (Fig. 1), several different syntheses were tested and differences in the IC50 of ≈2-fold were observed between batches.
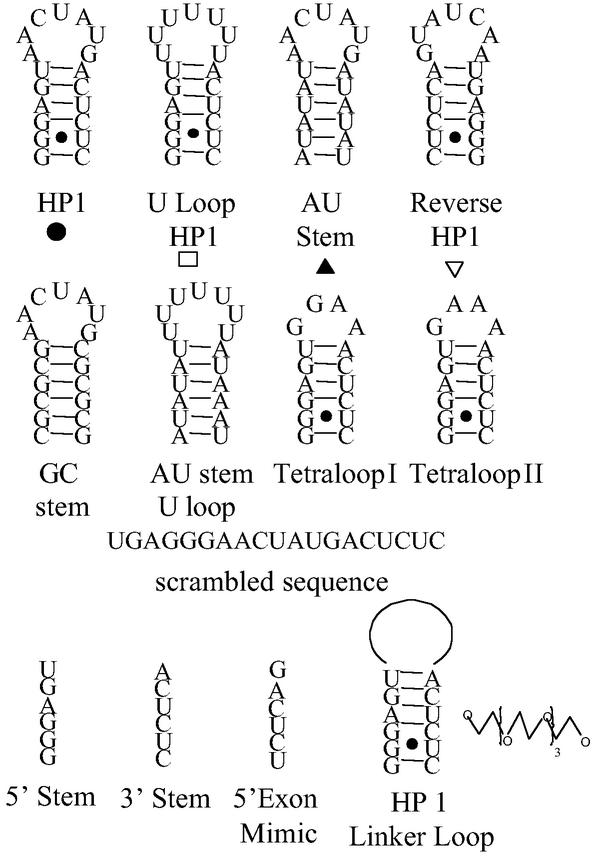
Figure 1.
Oligonucleotides used in this study. HP1 is a mimic of a rRNA sequence in C. albicans that forms quaternary and tertiary contacts in both Escherichia coli (19, 28) and Thermus thermophilus (25) ribosomes.
COS-7 Cell Growth and Oligonucleotide Uptake.
Oligonucleotides were added to cultures containing ≈2,000 cells in each well of either a 96- or 12-well plate. Cultures were grown in a 5% CO2 atmosphere in Ham's F12 medium with 10% FBS for at least 7 days. Growth was quantified by measuring ODs with a microplate reader or by removing cells from the well by digestion with proteinase and measuring OD660. In each assay, medium and oligonucleotide were replaced every 24–36 h.
Uptake of radioactive oligonucleotide into COS-7 cells was monitored using trace 5′ end-labeled oligonucleotide with 1 μM cold oligonucleotide and ≈3 × 106 cells grown in a T-25 cell culture dish in F12 medium with 10% FBS. Cultures were grown at 37°C and washed with 5 ml of F12 medium by gently shaking the cultures for 2 min. Washes were continued until little or no radioactivity was removed (approximately six washes). The number of cells in culture was determined by removing the cells from culture dishes by incubation with proteases and counting by hemocytometry. Cell-associated radioactivity was measured by scintillation counting and the volume of COS-7 cells calculated by assuming a cell diameter of 20 μm. Experiments to compare uptake into C. albicans and COS-7 cells were run in the same medium.
In Vivo DMS Modification.
Cells were placed into YNB (pH 2.5) medium at an OD540 of 0.4 along with 0, 1, 10, and 100 μM 2′OMe HP1 or 100 μM 2′OMe Rev HP1 in a total volume of 300 μl. Cultures were grown for 3 h, then a 1% volume of DMS was added and the cultures incubated for another 30 min. Cells were isolated by centrifugation, and total RNA was isolated by adding 1 ml of Triazol (GIBCO) and 100 μl of glass beads, following GIBCO's protocol. After reduction with NaBH3 and cleavage with aniline acetate (14), RT was used to detect positions of modification. RT reactions were performed at 42°C for 2 h with 4 μg of total RNA for DMS modification lanes and 10 μg of total RNA for sequencing lanes. Enhancements in modification are reported if modification increases as a function of oligonucleotide concentration and is ≈20% greater than cultures grown with 100 μM 2′OMe HP1 rather than 2′OMe Rev HP1. Each band was quantified with a phosphorimager and normalized relative to an RT stop, to adjust for loading.
Rate of Protein Synthesis.
C. albicans at 0.1 OD540 in 300 μl of pH 2.5 YNB medium with 0, 1, 10, or 100 μM of either 2′OMe HP1 or 2′OMe Rev HP1 were grown at 37°C for ≈2 h. Cells were harvested by centrifugation and washed with sterile water. Cultures were diluted to 0.1 OD540 in pH 2.5 YNB medium without methionine (15) and the same concentration of oligonucleotide added as the first incubation, along with trace [35S]methionine. The 300-μl culture was incubated for 5–10 min and placed on ice. Cells were lysed and radioactivity was measured (16).
Results
Oligonucleotide Uptake.
To determine whether the 5′ phosphate on the radioactive oligonucleotide affects uptake, increasing amounts of unlabeled d(GCCTCT) were added to cultures with the same amount of 5′ end-labeled oligonucleotide and cell-associated radioactivity was determined. The amount of cell-associated radioactivity decreases as the amount of unlabeled d(GCCTCT) increases, suggesting that uptake is not affected by the 5′ phosphate (see Fig. 6, which is published as supporting information on the PNAS web site, www.pnas.org).
Uptake of oligonucleotides of various length and backbone into C. albicans was measured (Table 1). For all sequences studied, oligonucleotides were internalized to give an ≈10-fold or higher intra- than extracellular concentration. Uptake depends on structure because dT10–40 all entered C. albicans better than the DNA hairpin (Table 1). Confocal microscopy shows that fluorescently labeled d(GCCTCT) is present throughout the cells (see Fig. 7, which is published as supporting information on the PNAS web site). Experiments with Saccharomyces cerevisiae also show that dT10–40 are taken up with a higher intra- than extracellular concentration (see Table 4, which is published as supporting information on the PNAS web site).
Table 1.
Uptake of oligonucleotides into C. albicans strain R099 in YNB pH 2.5 medium with 1 μM oligonucleotide added to culture
| Sequence | Intracellular concentration, μM
|
||
|---|---|---|---|
| Initial OD540 of 0.01 | Initial OD540 of 1.0
|
||
| − NaN3 | + NaN3 | ||
| 2′OMe HP1 | 160 ± 70* | 120 ± 53 | 1.5 ± 0.1 |
| 2′OMe Rev HP1 | 140 ± 30 | ||
| DNA HP1 | 30 ± 12 | 22 ± 5 | 0.5 ± 0.1 |
| RNA HP1 | 21 ± 8 | 25 ± 7 | 1.5 ± 0.7 |
| d(GCCTCT) | 16 ± 8 | ||
| dT10 | 200 ± 50 | ||
| dT20 | 600 ± 150 | ||
| dT30 | 500 ± 80 | ||
| dT40 | 650 ± 104 | ||
Intracellular concentrations of 2′OMe HP1 at pH 2, 3, 3.5, 4, and 4.5 are 180 ± 60, 100 ± 32, 56 ± 20, 34 ± 7, and 38 ± 10 μM, respectively, when the initial OD540 is 0.01.
The studies suggest active transport across the cell membrane. To test for energy-dependent uptake, NaN3, a metabolic inhibitor, was added to cultures; if uptake is energy dependent, then NaN3 should reduce uptake (17). For each backbone tested, NaN3 reduces uptake >10-fold (Table 1).
The rate of uptake was determined for 2′OMe HP1. Uptake has a half-life of ≈10 min and saturates at ≈100 min, with the intracellular concentration reaching ≈400 μM (see Fig. 8, which is published as supporting information on the PNAS web site).
Uptake of 2′OMe HP1 was measured for COS-7 cells and C. albicans in F12 medium with 10% FBS at 37°C. C. albicans was suspended in culture medium and COS-7 cells were grown in a T-25 cell culture dish. For the COS-7 cells, internal concentration was 10 and 8 nM after 20-min and 1-h incubations, respectively, and 3.6 μM after 6 h. For C. albicans, internal concentration was 38 μM after a 20-min incubation.
Oligonucleotide Stability.
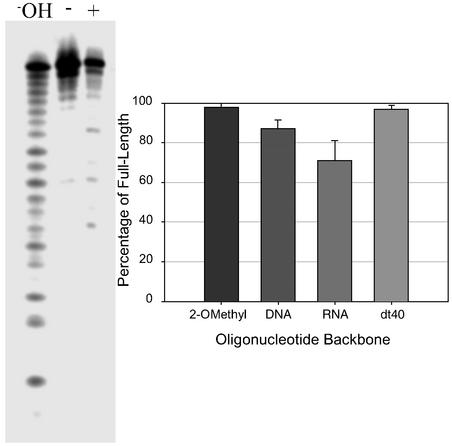
DNA and 2′OMe HP1 hairpins and dT40 harvested from inside C. albicans after 12-h incubations were >80% intact (Fig. 2). The stability of dT40 indicates that chemical stability does not require a folded structure. RNA HP1 also remained 60% intact, which is surprising because endogenous RNAs are degraded in yeast (18) and, presumably, C. albicans.
Figure 2.
Oligonucleotide stability in C. albicans after growth for 12 h. (Left) An autoradiogram shows typical results for HP1 with RNA backbone; +, oligonucleotide harvested from cells; −, oligonucleotide before exposure to cells; −OH, hydrolysis ladder. (Right) Results with other oligonucleotides.
Inhibition of Cell Growth.
Mimics of the 3′ end of the 5′ exon inhibit in vitro self-splicing of the C. albicans group I intron (2). These mimics were tested in culture with C. albicans. Under all conditions tested, growth was unaffected for strains that harbor a group I intron in their LSU rRNA precursor. At pH 2.5, however, growth of a strain without a group I intron (R099) was slowed by the 2′OMe 5′ exon mimic, m(GACUCU), with an extracellular IC50 of ≈50 μM (Table 2). This hexamer is found in the stem of a hairpin in 25S rRNA (Fig. 1; ref. 19), which suggests that a mimic of the entire hairpin might be a more effective inhibitor.
Table 2.
IC50s (μM) for inhibition of C. albicans cell growth in YNB pH 2.5 medium by oligonucleotides
| Sequence | Backbone
|
|
|---|---|---|
| 2′OMe | DNA | |
| HP1* | 2.0 ± 1.5 | 18 ± 2 |
| Rev HP1 | 17 ± 6 | >100 |
| Comp HP1† | 1.5 ± 1 | ≈55 |
| Duplex‡ | 1 ± 0.5 | |
| AU stem | 11 ± 2 | 40 ± 8 |
| GC stem | 30 ± 10 | |
| HP1 −2§ | 5 ± 3 | |
| Rev HP1 −2§ | 15 ± 5 | |
| Tetraloop I | 6 ± 4 | |
| Tetraloop II | 5 ± 3 | |
| U loop | 6.6 ± 1 | 29 ± 4 |
| Linker HP1¶ | 10 ± 2 | |
| Rev linker HP1¶ | 36 ± 5 | |
| AU stem and U loop | >100 | |
| Scrambled sequence | >100 | |
| N18C‖ | >100 | |
| GACUCU | ≈50 | |
| ACUCUC | ≈50 | |
| GGGAGU | ≈50 | |
RNA HP1 inhibits with an IC50 of 15 ± 5 μM.
Comp HP1 is an oligonucleotide that is completely complementary to HP1.
Duplex is the HP1-Comp HP1 oligonucleotides after they are heated at 80°C for 5 min and cooled to room temperature by placing the vial on a bench top in pH 2.5 YNB media to allow duplex formation.
HP1 −2 is an oligonucleotide that does not have the terminal 2 bp in HP1.
Linker refers to an oligonucleotide where the loop nucleotides in the hairpin are replaced with two C18 spacers.
N18C is a mixture of randomized oligonucleotides with a C at the 3′ end.
As shown in Table 2, the 2′OMe hairpin, HP1, is a 25-fold more potent inhibitor of cell growth than m(GACUCU). Other sequences were studied to determine the sequence dependence of inhibition; all but one change increased the IC50 (Table 2, Fig. 3). The exception is a sequence that is completely complementary to HP1.
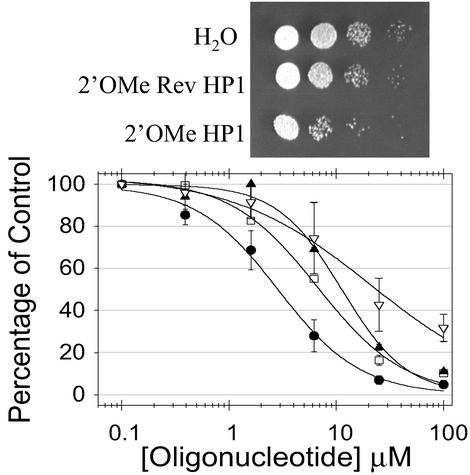
Figure 3.
Inhibition of cell growth with oligonucleotides. (Upper) Serial dilutions of cultures incubated with 25 μM of oligonucleotides spotted onto a yeast extract/peptone/dextrose (YPD) plate. (Lower) Plots of data for various 2′OMe oligonucleotides: ●, HP1; □, U loop; ▴, AU stem; ▿, Rev HP1.
Oligonucleotides with other backbones were tested. Results with DNA mirror the sequence dependence of 2′OMe oligonucleotides, with DNA HP1 an ≈10-fold less effective inhibitor of growth (Table 2). RNA HP1 has an IC50 of 15 ± 5 μM. Thus, it is ≈7-fold less effective than 2′OMe HP1, but still retains activity, giving further evidence that RNA oligonucleotides are not rapidly degraded in C. albicans.
Effects of pH.
Studies of 2′OMe HP1 as a function of pH show that uptake and inhibition of cell growth are best at low pH (see Figs. 9 and 10, which are published as supporting information on the PNAS web site, and Table 1). Evidently, uptake may be one reason that inhibition is optimal at low pH.
Growth rates at various pH in the absence of oligonucleotide were also measured. Growth rate is only slightly decreased in going from pH 4.5 to 2.0 (see Fig. 11, which is published as supporting information on the PNAS web site). Also, the morphology of cells was not affected by pH (data not shown).
DMS Modification.
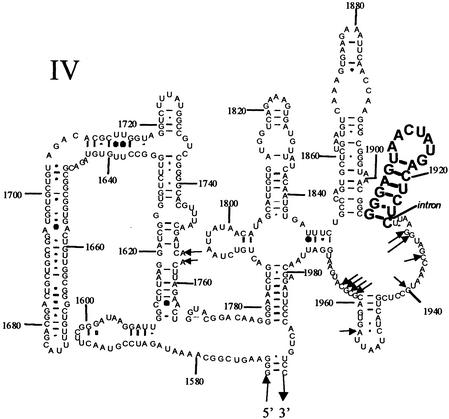
In vivo DMS modification in C. albicans strain R099 was used to determine whether 2′ OMe HP1 affects the structure of 25S rRNA, because in vitro studies have shown that nucleotides become protected when subunits are assembled (19). In our experiments, DMS modification of several nucleotides increases with increasing 2′OMe HP1 concentration, and this enhancement is absent or reduced with 2′OMe Rev HP1 (Fig. 4; and see Figs. 12 and 13, which are published as supporting information on the PNAS web site).
Figure 4.
Positions of DMS modification that are enhanced in C. albicans 25S rRNA with 2′OMe HP1 relative to 2′OMe Rev HP1. Arrows indicate positions of enhancement. The hairpin sequence is shown in bold. Nucleotides are numbered according to E. coli.
Rate of Protein Synthesis.
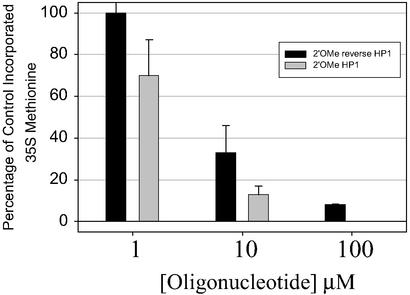
If HP1 targets the ribosome, the rate of protein synthesis will likely be reduced. Protein synthesis was measured for cells grown in the presence of 2′OMe HP1 and 2′OMe Rev HP1 (Fig. 5). At concentrations of 100 μM oligonucleotide, incorporation of [35S]methionine is reduced 39- and 14-fold with 100 μM HP1 and Rev HP1, respectively.
Figure 5.
Protein synthesis experiments with 2′OMe HP1 and 2′OMe Rev HP1. Percentage of control incorporated [35S]methionine is the percentage of [35S]methionine incorporated into protein relative to the amount incorporated in the absence of oligonucleotide.
Optical Melting Experiments.
To provide structural information on the 2′OMe HP1 oligonucleotide, absorbance versus temperature curves were measured in 1 M NaCl (1 M NaCl/5 mM EDTA/50 mM sodium cacodylate, pH 7.0) and YNB medium at pH 4.5, 3.5, and 2.5. Melting temperatures (Tm) are independent of oligonucleotide concentration for hairpins but dependent on concentration for duplexes. In 1 M NaCl, the Tm of 2′OMe HP1 (77.5°C) is independent of concentration, indicating a hairpin structure. In YNB medium at pH 4.5 and 3.5, Tm are 67.0 and 63.9°C, respectively, independent of concentration, which also indicates a hairpin structure. In YNB medium at pH 2.5, however, the Tm varies from 22.3 to 42.2°C over a concentration range of 1–25 μM, indicating a duplex structure. Thus, one potential reason for enhanced uptake at low pH is that 2′OMe HP1 forms a duplex in pH 2.5 YNB medium.
Testing Other Strains for Inhibition.
To test the generality of 2′OMe HP1 growth inhibition, several clinical isolates of C. albicans were tested for susceptibility to 25 μM HP1. R020 and R0109 have group I self-splicing introns in their precursor rRNA, whereas R099 and R0102 do not. Only growth of strain R099 is significantly slowed at these concentrations. Thus, 2′OMe HP1 is not a broad-based inhibitor of C. albicans growth (see Fig. 14, which is published as supporting information on the PNAS web site).
To test for differences between strains sensitive and insensitive to HP1, growth rate, uptake of oligonucleotide, and sensitivity to miconazole were tested. The IC50 for 2′OMe HP1 inhibition ranges from 2 to >200 μM (Table 3). In these experiments, inhibition by HP1 correlates with increased sensitivity to miconazole. In contrast, uptake of oligonucleotides is not particularly different in these four strains, suggesting that uptake does not explain why HP1 is only effective at inhibiting R099 growth.
Table 3.
Effect of miconazole and 2′OMe HP1 on growth of several C. albicans strains in pH 2.5 YNB medium
| Strain | Growth rate, min* | Miconazole IC50, μM | 2′OMe HP1 IC50, μM | Uptake of 2′OMe HP1, μM |
|---|---|---|---|---|
| R020 | 60 | 0.59 ± 0.24 | >200 | 200 ± 30 |
| R099 | 90 | 0.19 ± 0.10 | 2 | 160 ± 70 |
| R0102 | 120 | 0.33 ± 0.20 | ≈150 | 117 ± 10 |
| R0109 | 60 | 0.33 ± 0.22 | ≈100 | 64 ± 6 |
Growth rate is doubling time.
COS-7 Mammalian Cell Growth.
To assess whether HP1 affects growth of mammalian cells, 100 μM of 2′OMe, DNA, or RNA HP1 was placed in culture with COS-7 cells. Culture medium and oligonucleotide were changed every 24–36 h because oligonucleotides are degraded in the medium (t1/2 is >12 h, 5 min, and <1 min for 2′OMe, DNA, and RNA HP1, respectively). For all three oligonucleotides, growth of COS-7 cells was not affected for incubations as long as 10 days.
Discussion
This study demonstrates that C. albicans is an excellent system to test new approaches for oligonucleotide therapeutics. C. albicans is an important target for drug discovery because infections and resistant strains are increasing (1). Oligonucleotides offer several advantages, including simple design and automated synthesis. Also, studies of pharmacokinetic properties of oligonucleotides have shown that they are relatively independent of sequence (20). One oligonucleotide drug, Vitravene, has been approved by the U.S. Food and Drug Administration (21).
Antisense oligonucleotides have been shown to slow growth of both Mycobacterium smegmatis and Mycobacterium tuberculosis (22, 23). For these related organisms, there are differences in the ability of oligonucleotides to traverse cell walls. In M. smegmatis, oligonucleotide uptake is not efficient. Addition of either a small molecule to culture or to the oligonucleotide is necessary for oligonucleotide uptake and activity (22). For M. tuberculosis, oligonucleotides can simply be added to cell culture to exert an antibacterial effect (23). These studies and our work here demonstrate that oligonucleotide therapies can be developed toward pathogens that have a cell wall, in addition to a cell membrane.
Results presented here suggest that it may be easier to use oligonucleotides to target C. albicans than to target mammalian cells. Oligonucleotides enter C. albicans in an energy-dependent manner with intracellular concentrations 10- to 100-fold greater than the concentration in culture medium (Table 1). When cultured under identical conditions, C. albicans internalizes oligonucleotides with at least 10-fold higher intracellular concentration than COS-7 mammalian cells. In addition, oligonucleotides are relatively stable inside C. albicans for at least 12 h. The enhanced uptake and stability of oligonucleotides suggest that uptake and degradation provide selectivity for targeting C. albicans over human cells. Barriers other than uptake, however, can affect activity in cells. For example, oligonucleotides can be trapped in vacuoles or organelles that do not contain their target, or the oligonucleotides can be bound to protein. The demonstration that 2′OMe HP1 inhibits cell growth alleviates many of these concerns. Moreover, confocal microscopy shows that fluorescently labeled d(GCCTCT) is dispersed well in the cell.
In this study, 2′OMe HP1 is effective in slowing growth of C. albicans strain R099 (Fig. 3, Table 2). Because 2′OMe HP1 forms a stable hairpin, it is unlikely that it acts by simple base pairing. In vivo DMS modification of rRNA and reduction in protein synthesis depend on oligonucleotide sequence and suggest that the mode of action may be related to the ribosome. This is only one of many potential mechanisms. For example, HP1 could inhibit function of a helicase that is important for ribosomal assembly (24) or interfere with tertiary interactions (25). HP1 is much more potent with the R099 strain relative to other strains tested. Because the interactions for association of ribosomes are conserved, the main activity of 2′OMe HP1 may not be associated with the ribosome. Alternatively, the ribosome may be the target, but the effect of low pH on cells may vary from strain to strain. The intracellular pH is probably minimally affected by low external pH, because lowering medium pH has little effect on internal pH in S. cerevisiae (26).
Requirement for low pH does not prohibit therapeutic use of oligonucleotides in C. albicans. C. albicans infects the mouth, bladder, and skin of humans. These areas are tolerant to low pH conditions. For example, the pH of bladder washes and douches are low, and many soft drinks have a pH of 2.5.
Determining the mechanism of action may be difficult with C. albicans because of the diploid nature and plasticity of its genome (27). The correlation between miconazole and 2′OMe HP1 IC50s suggests that the sensitive strain, R099, may simply be weaker than the others (Table 3). Differential uptake is not responsible for the differences in sensitivity because the 2′OMe HP1 IC50s do not correlate with uptake of oligonucleotide into each strain (Table 3). This work, however, shows the potential of oligonucleotides to inhibit growth of C. albicans, and suggests that mimicking natural RNA structures may be a promising approach.
That C. albicans takes up oligonucleotides in high concentrations that remain stable after incubations for 12 h makes this system amenable to other therapeutic strategies that use oligonucleotides. The C. albicans genome is being sequenced, and a functional genomics approach has identified essential genes in this organism (10), so the database of targetable sequence is increasing. Biochemical information discovered using related organisms, such as S. cerevisiae, should also facilitate rational design. Taken together, these results suggest that C. albicans infections may be particularly susceptible to oligonucleotide-based therapeutics.
Supplementary Material
Acknowledgments
We thank Hiram Lyon, Liam Casey, and Prof. Joanna Olmsted for help with mammalian cell culture and confocal microscopy and Prof. Robin Gutell for providing the C. albicans 25S rRNA secondary structure. This work was supported by National Institutes of Health Grant GM22939.
Abbreviations
- 2′OMe
oligonucleotide with a 2′-O-methyl backbone
- DMS
dimethyl sulfate
- HP1
hairpin mimic of LSU rRNA sequence
- rRNA
ribosomal RNA
- YNB
yeast extract nitrogen base
References
- 1.Sternberg S. Science. 1994;266:1632–1634. doi: 10.1126/science.7702654. [DOI] [PubMed] [Google Scholar]
- 2.Disney M D, Matray T, Gryaznov S M, Turner D H. Biochemistry. 2001;40:6520–6526. doi: 10.1021/bi002009j. [DOI] [PubMed] [Google Scholar]
- 3.Testa S M, Gryaznov S M, Turner D H. Proc Natl Acad Sci USA. 1999;96:2734–2739. doi: 10.1073/pnas.96.6.2734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lebedeva I, Benimetskaya L, Stein C A, Vilenchik M. Eur J Pharm Biopharm. 2000;50:101–119. doi: 10.1016/s0939-6411(00)00088-6. [DOI] [PubMed] [Google Scholar]
- 5.Arzumanov A, Walsh A P, Rajwanshi V K, Kumar R, Wengel J, Gait M J. Biochemistry. 2001;40:14645–14654. doi: 10.1021/bi011279e. [DOI] [PubMed] [Google Scholar]
- 6.Childs J C, Disney M D, Turner D H. Proc Natl Acad Sci USA. 2002;99:11091–11096. doi: 10.1073/pnas.172391199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Caruthers M H, Beaton G, Wu J V, Wiesler W. Methods Enzymol. 1992;211:3–20. doi: 10.1016/0076-6879(92)11003-2. [DOI] [PubMed] [Google Scholar]
- 8.Freier S M, Altmann K H. Nucleic Acids Res. 1997;25:4429–4443. doi: 10.1093/nar/25.22.4429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Stein C A, Pal R, DeVico A L, Hoke G, Mumbauer S, Kinstler O, Sarngadharan M G, Letsinger R L. Biochemistry. 1991;30:2439–2444. doi: 10.1021/bi00223a020. [DOI] [PubMed] [Google Scholar]
- 10.De Backer M D, Nelissen B, Logghe M, Viaene J, Loonen I, Vandoninck S, de Hoogt R, Dewaele S, Simons F A, Verhasselt P, et al. Nat Biotechnol. 2001;19:235–241. doi: 10.1038/85677. [DOI] [PubMed] [Google Scholar]
- 11.Disney M D, Testa S M, Turner D H. Biochemistry. 2000;39:6991–7000. doi: 10.1021/bi992937m. [DOI] [PubMed] [Google Scholar]
- 12.Puglisi J D, Tinoco I., Jr Methods Enzymol. 1989;180:304–325. doi: 10.1016/0076-6879(89)80108-9. [DOI] [PubMed] [Google Scholar]
- 13.McDowell J A, Turner D H. Biochemistry. 1996;35:14077–14089. doi: 10.1021/bi9615710. [DOI] [PubMed] [Google Scholar]
- 14.Ziehler W A, Engelke D R. In: Current Protocols in Nucleic Acid Chemistry. Beaucage S L, Bergstrom D E, Glick G D, Jones R A, editors. New York: Wiley; 2001. , Unit 6. [Google Scholar]
- 15.Sherman F. Methods Enzymol. 2002;350:3–41. doi: 10.1016/s0076-6879(02)50954-x. [DOI] [PubMed] [Google Scholar]
- 16.Bonifacino J S. In: Short Protocols in Molecular Biology. Ausubel F, Brent R, Kingston R E, Moore D D, Seidman J G, Smith J A, Struhl K, editors. New York: Wiley; 1995. , Unit 10.17. [Google Scholar]
- 17.Wender P A, Mitchell D J, Pattabiraman K, Pelkey E T, Steinman L, Rothbard J B, Garlington S, Lin Q, Kirschberg T, Kreider E, et al. Proc Natl Acad Sci USA. 2000;97:13003–13008. doi: 10.1073/pnas.97.24.13003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jacobs J S, Anderson A R, Parker R P. EMBO J. 1998;17:1497–1506. doi: 10.1093/emboj/17.5.1497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Merryman C, Moazed D, Daubresse G, Noller H F, McWhirter J. J Mol Biol. 1999;285:107–113. doi: 10.1006/jmbi.1998.2243. [DOI] [PubMed] [Google Scholar]
- 20.Agrawal S, Temsamani J, Galbraith W, Tang J. Clin Pharmacokinet. 1995;28:7–16. doi: 10.2165/00003088-199528010-00002. [DOI] [PubMed] [Google Scholar]
- 21.Anderson K P, Fox M C, Brown-Driver V, Martin M J, Azad R F. Antimicrob Agents Chemother. 1996;40:2004–2011. doi: 10.1128/aac.40.9.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rapaport E, Levina A, Metelev V, Zamecnik P C. Proc Natl Acad Sci USA. 1996;93:709–713. doi: 10.1073/pnas.93.2.709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Harth G, Zamecnik P C, Tang J Y, Tabatadze D, Horwitz M A, Barker R H, Jr, Metelev V, Coakley A, Zamecnik P, Rapaport E, et al. Proc Natl Acad Sci USA. 2000;97:418–423. doi: 10.1073/pnas.97.1.418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tsu C A, Kossen K, Uhlenbeck O C. RNA. 2001;7:702–709. doi: 10.1017/s1355838201010135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yusupov M M, Yusupova G Z, Baucom A, Lieberman K, Earnest T N, Cate J H, Noller H F. Science. 2001;292:883–896. doi: 10.1126/science.1060089. [DOI] [PubMed] [Google Scholar]
- 26.Haworth R S, Fliegel L. Mol Cell Biochem. 1993;124:131–140. doi: 10.1007/BF00929205. [DOI] [PubMed] [Google Scholar]
- 27.Rustchenko E P, Howard D H, Sherman F. Microbiology. 1997;143:1765–1778. doi: 10.1099/00221287-143-5-1765. [DOI] [PubMed] [Google Scholar]
- 28.Merryman C, Moazed D, McWhirter J, Noller H F. J Mol Biol. 1999;285:97–105. doi: 10.1006/jmbi.1998.2242. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.