Abstract
Active oxygen species (AOS) are responsible for triggering defense responses in plants. Respiratory burst oxidase homologs (rboh genes) have been implicated in AOS generation. We have isolated two rboh cDNAs, NbrbohA and NbrbohB, from Nicotiana benthamiana leaves. NbrbohA was expressed constitutively at a low level and the transcripts were increased after mechanical stress of control leaf infiltration, whereas NbrbohB was induced specifically by the protein elicitor INF1 from the potato pathogen Phytophthora infestans. We examined the function of the Nbrboh genes in AOS generation and in the hypersensitive response (HR) using virus-induced gene silencing (VIGS). VIGS indicated that both genes are required for H2O2 accumulation and for resistance to Phytophthora. VIGS of Nbrboh genes also led to a reduction and delay of HR cell death caused by INF1. We further demonstrate that the induction of HR-like cell death by overexpression of a constitutively active mutant of a mitogen-activated protein kinase kinase, MEKDD, is compromised by VIGS of NbrbohB. We found that MEKDD induced NbrbohB but not NbrbohA. This work provides genetic evidence for the involvement of a mitogen-activated protein kinase cascade in the regulation of rboh genes.
INTRODUCTION
The oxidative burst is a characteristic early feature of the hypersensitive response (HR) during incompatible interactions between resistant plants and avirulent pathogens (Doke, 1983; Dietrich et al., 1996; Jabs et al., 1996; Lamb and Dixon, 1997). The rapid production of active oxygen species (AOS) could inhibit pathogen growth by direct microbicidal action (Peng and Kuc, 1992), by restricting pathogen penetration via cross-linking of cell wall glycoproteins (Bradley et al., 1992), by induction of phytoalexin accumulation or defense-related genes (Apostol et al., 1989; Desikan et al., 2001), and by activating salicylic acid biosynthesis (Léon et al., 1995), which leads to the induction of genes such as PR-1a (Chen et al., 1993).
The enzymatic complex of NADPH oxidase found in mammalian neutrophils consists of two plasma membrane proteins, gp91phox and p22phox (phox for phagocyte oxidase). The several cytosolic regulatory proteins, p47phox, p67phox, and the small G protein Rac, translocate to the plasma membrane to form the active complex after stimulation (Bokoch, 1994). Several lines of evidence demonstrated that the oxidative burst in plants is caused by activation of an NADPH oxidase closely resembling that operating in activated neutrophils (Lamb and Dixon, 1997). Incubation of potato tuber slices with an incompatible race of Phytophthora infestans, the potato late blight pathogen, or with a hyphal wall elicitor stimulated NADPH-dependent O2−-generating activity, which was located predominantly in the plasma membrane fraction (Doke, 1985; Doke and Miura, 1995). Diphenylene iodonium, an inhibitor of the phagocyte NADPH oxidase, blocks the oxidative burst in plant cells (Levine et al., 1994; Auh and Murphy, 1995; Piedras et al., 1998). Homologs of gp91phox (respiratory burst oxidase homolog [rboh]), which is the catalytic subunit of phagocyte NADPH oxidase, were isolated from several plants (Groom et al., 1996; Keller et al., 1998; Torres et al., 1998; Amicucci et al., 1999). The Arabidopsis genome contains eight rboh genes, suggesting that individual isoforms have different functions and participate in multiple distinct signaling pathways (Torres et al., 2002).
We have isolated rboh cDNAs from potato (Yoshioka et al., 2001). The cloning of these cDNAs revealed two isogenes, designated StrbohA and StrbohB. RNA gel blot analyses showed that StrbohA is expressed constitutively, whereas St-rbohB is induced by a cell wall elicitor from Phytophthora, salicylic acid, or arachidonic acid in potato tubers. The proteins have an N-terminal extension, which contains two Ca2+ binding elongation factor (EF) hands, that is absent in the phagocyte NADPH oxidase (Yoshioka et al., 2001). Earlier work provided evidence that signaling events preceding the burst include the involvement of a plant homolog of human Rac (Kawasaki et al., 1999), which is required for the assembly of an active phagocyte NADPH oxidase (Bokoch, 1994). However, no homologs of the p22phox, p47phox, or p67phox regulators of the phagocyte NADPH oxidase were found in the Arabidopsis genome (Dangl and Jones, 2001). Calcium influx into the cytoplasm (Chandra and Low, 1997; Piedras et al., 1998) and changes in protein phosphorylation (Kauss and Jeblick, 1995; Miura et al., 1995) are implicated in the activation process of the oxidative burst. To address the function of rboh genes in plant defense, we used virus-induced gene silencing (VIGS) in Nicotiana benthamiana as an efficient reverse genetics approach (Baulcombe, 1999). This allowed us to circumvent the problem of gene redundancy in plants by potentially silencing redundant members of the rboh subfamily.
Phytophthora species produce low molecular mass extracellular proteins known as elicitins, which induce HR and other biochemical changes associated with defense responses in Nicotiana species (Ricci et al., 1989; Huet et al., 1994; Kamoun et al., 1999; Sasabe et al., 2000) and some radish and turnip cultivars (Kamoun et al., 1993), presumably after binding to a plasma membrane receptor (Bourque et al., 1999). The perception of elicitin by the plant cell induces an influx of calcium ions (Tavernier et al., 1995), AOS production (Rustérucci et al., 1996), and activation of mitogen-activated protein kinase (MAPK) (Zhang et al., 1998). In contrast to strains of Phytophthora that produce elicitin INF1, strains that are engineered to be deficient in INF1 induce disease lesions on N. benthamiana, suggesting that INF1 functions as an avirulence factor that conditions resistance in this species (Kamoun et al., 1998). This finding demonstrates that the N. benthamiana–Phytophthora pathosystem is a good model for dissecting the elicitor response and resistance to Phytophthora by VIGS.
Here, we isolated the cDNAs corresponding to StrbohA and StrbohB of potato from a N. benthamiana cDNA library. The NbrbohA- and NbrbohB-silenced N. benthamiana plants were infected with Phytophthora and found to be unable to accumulate AOS during disease resistance reactions to Phytophthora. We demonstrate that the silenced plants were compromised in the INF1-mediated activation of HR. Moreover, we observed that overexpression of a constitutively active mutant of a MAPK kinase (MAPKK) (Yang et al., 2001), which activates endogenous salicylic acid–induced protein kinase (SIPK) and wound-induced protein kinase (WIPK), provoked cell death in N. benthamiana plants and also induced the NbrbohB gene. These results suggest that the MAPK pathway can regulate rboh gene expression.
RESULTS
Two cDNAs for the gp91phox Homolog in N. benthamiana
We identified two homologs of human gp91phox, designated NbrbohA and NbrbohB (Figure 1). The NbrbohA cDNA was 3732 bp in length and had an open reading frame of 962 amino acids with a predicted molecular mass of 109 kD. The longest NbrbohB cDNA sequence was 3290 bp in length and had an open reading frame of 939 amino acids with a predicted molecular mass of 106 kD. We cannot exclude the possibility that more Nbrboh genes exist. However, the analysis of cDNAs obtained under low-stringency conditions from 6 × 105 plaques of the cDNA library prepared from leaf tissues treated with hyphal wall elicitor suggests that, if there are more gp91phox homologs in N. benthamiana, they are unlikely to be expressed in elicitor-stimulated leaf tissues.
Figure 1.
Alignment of the Predicted Amino Acid Sequences of Nb-rbohA, NbrbohB, and Human gp91phox.
Amino acids that are conserved are boxed in black. Six potential transmembrane-spanning domains (TMD-1 to TMD-6) are indicated with overlines. Conserved His residues, demonstrated to bind heme in yeast FRE1 (Finegold et al., 1996), are indicated by solid triangles. Dots under the sequences indicate amino acid residues involved in N-glycosylation. EF hand motifs in the N-terminal domain of Nbrboh are overlined. Beneath these motifs is the sequence of a canonical EF hand (Kretsinger, 1996). n is usually a hydrophobic residue. Dashes indicate variable amino acid residues. X, Y, Z, and −X, contain oxygen within their side chains. Carbonyl oxygen of # serves as a ligand. −Z is usually Glu. Regions used for VIGS and DNA gel blot analysis are indicated by insert A and insert B. Dashes indicate gaps in the sequence to allow for maximal alignment. Multiple alignment of the protein sequence was made using the CLUSTAL W program (Thompson et al., 1994). FAD, flavin adenine dinucleotide.
Although the topology and functions of plant rboh proteins have not been tested experimentally, several features of the amino acid sequence and comparisons with human gp91phox allow predictions (Yu et al., 1998). NbrbohA and NbrbohB proteins are predicted to have six transmembrane-spanning domains (TMD-1 to TMD-6) that correspond to those identified in gp91phox. TMD-3 and TMD-5 contain pairs of His residues that are likely to be important for heme binding (Finegold et al., 1996). The C-terminal regions of each homolog contain conserved flavin adenine dinucleotide, NADPH-ribose, and NADPH-adenine binding sites, which are likely to be located in the cytoplasm. Moreover, human gp91phox amino acid residues Pro-415 and Asp-500, which are indispensable for the catalytic activity (Segal et al., 1992), also are conserved in NbrbohA and NbrbohB. The N-terminal regions of the proteins are hydrophilic and contain two Ca2+ binding EF hands, as seen in other plant rboh genes. Calcium binding activity of these domains was found experimentally by Keller et al. (1998) for Arabidopsis AtrbohA.
Comparison of the Nbrboh Proteins and Genes
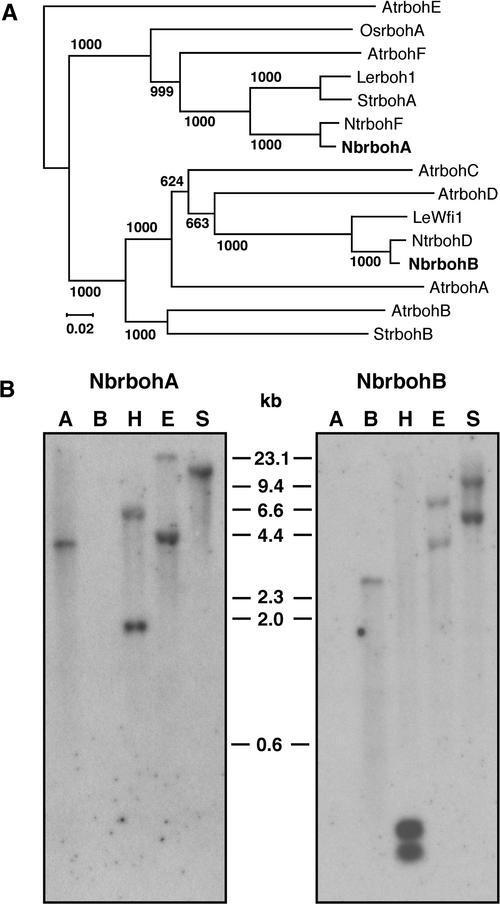
Figure 2A shows a phylogenetic tree for polypeptides of Nb-rbohA, NbrbohB, and the 13 reported plant rboh proteins. The plant rboh proteins, except for AtrbohE, were divided into two groups. The deduced amino acid sequence of NbrbohA had maximum sequence similarity to tobacco NtrbohF (96% identity). On the other hand, NbrbohB was most similar (99% identity) to tobacco NtrbohD.
Figure 2.
Nbrboh Gene Organization.
(A) Phylogenetic tree of various plant respiratory burst oxidase homologs. An unrooted tree was constructed using the neighbor-joining method (Saitou and Nei, 1987). The length of the horizontal lines connecting the sequences is proportional to the estimated genetic distance between these sequences. The number above or below each horizontal line is the frequency with which a given branch appeared in 1000 bootstrap replications. Accession numbers are listed at the end of Methods.
(B) DNA gel blot hybridization of genomic DNA. Genomic DNA (10 μg) was digested with HindIII (H), EcoRI (E), or SpeI (S) and hybridized with the 32P-labeled N-terminal region of NbrbohA or NbrbohB cDNA as indicated in Figure 1. Lane A, full-length NbrbohA cDNA (30 pg); lane B, full-length NbrbohB cDNA (30 pg). DNA molecular mass markers are indicated.
Gel blot hybridizations were performed with N. benthamiana genomic DNA samples under high-stringency conditions using regions of the cDNAs that encode the N-terminal portions of NbrbohA and NbrbohB as gene-specific probes (Figures 1 and 2B). The hybridization patterns of the blots were different, indicating that these probes detect each Nbrboh sequence. Moreover, each isogene-specific probe was shown to hybridize to another paralog in the genome, suggesting that there are two copies of NbrbohA and NbrbohB. Smith (1979) reported that the N. benthamiana genome contains 19 chromosomes (1n value), in contrast to diploid members of the Solanaceae, which have 12 chromosomes. DNA gel blot analysis is consistent with the proposed duplication of Nbrboh isogenes.
Induction of NbrbohB by INF1 and Salicylic Acid
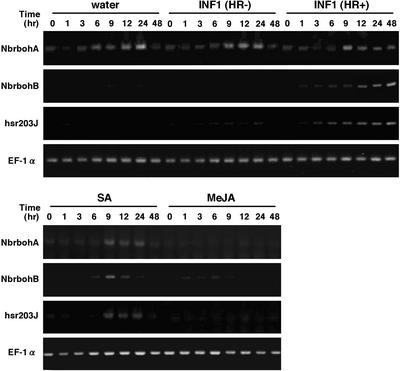
We estimated changes over time in the transcript levels of Nb-rbohA and NbrbohB in N. benthamiana leaves after elicitation with INF1 S3 (HR−) or INF1 (HR+), salicylic acid (SA), or methyl jasmonic acid (MeJA) or after mechanical stress caused by infiltrating water into leaves. INF1 S3 (HR−) contains a mutated INF1 (HR+) sequence in which the Cys at position 3 of the processed protein is mutated into a Ser and serves as a negative control (Kamoun et al., 1997). The probes from the entire sequences of NbrbohA and NbrbohB were not sensitive enough to detect a signal with RNA gel blot analysis (data not shown). Therefore, reverse transcriptase–mediated (RT) PCR with gene-specific primers was performed. We also used primers for hsr203J (Tronchet et al., 2001) and EF-1α as controls for an induced and a constitutively expressed gene, respectively (Figure 3).
Figure 3.
Expression Patterns of NbrbohA and NbrbohB after Elicitation by Wound or Pathogen Signals.
N. benthamiana leaves were treated with water, 10 μg/mL FLAG-INF1 S3 (HR−), 10 μg/mL FLAG-INF1 (HR+), 0.5 mM SA, and 0.1 mM MeJA. At the times indicated, leaf samples were harvested. Total RNA was isolated and used for RT-PCR as described in Methods, applying 35, 29, and 35 amplification cycles with specific primers for NbrbohA, NbrbohB, and hsr203J, respectively. Equal loading of cDNA was monitored by amplification of a constitutively expressed EF-1α gene (29 cycles).
NbrbohA mRNA was expressed constitutively at a low level before injection. The increase in the transcript level was detected 9 to 24 h after all injections, including water control and INF1 S3 (HR−), except injection of MeJA. This induction could be caused by the mechanical stress of the infiltration procedure. MeJA suppressed the infiltration stimulus–induced Nb-rbohA transcript accumulation. On the other hand, RT-PCR of NbrbohB and hsr203J showed that the infiltration of INF1 (HR+) led to specific elicitation in the transcript level. Moreover, exogenous SA stimulated the accumulation of NbrbohB and hsr203J transcripts at a concentration of 0.5 mM, which is sufficient to induce the oxidative burst in plant cell suspensions (Kauss and Jeblick, 1995; Shirasu et al., 1997) and plant tissues (Yoshioka et al., 2001). By contrast, the transcript level of EF-1α, an abundant protein of the cell, is unaltered under these conditions.
Isogene-Specific VIGS of the Nbrboh Subfamily
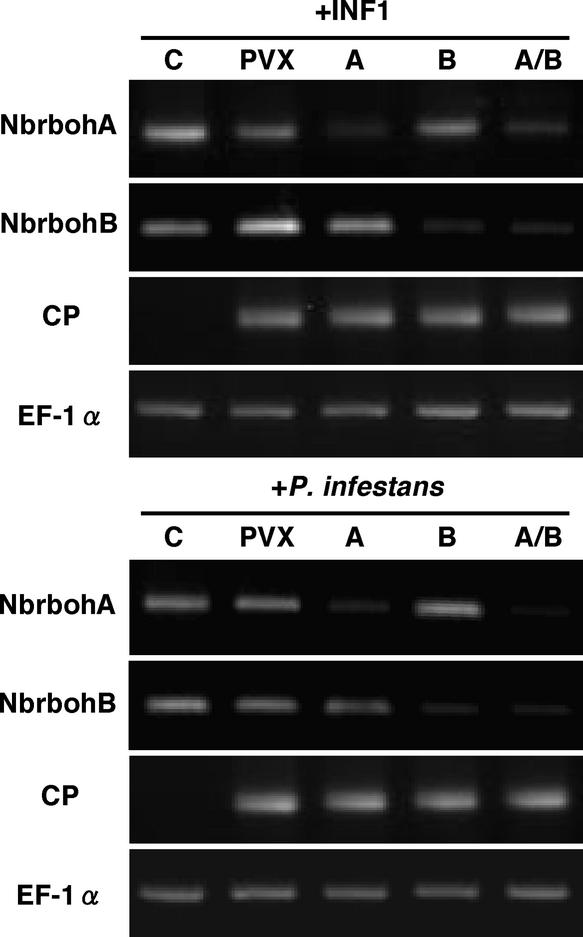
Because the N-terminal regions of NbrbohA and NbrbohB are distinct and the C-terminal regions are homologous, we conducted isogene-specific and isogene-nonspecific gene silencing using NbrbohA- and NbrbohB-specific regions or the conserved region as a dual insert for Potato virus X (PVX) vector (Figure 1). The NbrbohA and NbrbohB inserts were 278 and 197 bp and showed 41 and 40% nucleotide identity to the corresponding regions of NbrbohB and NbrbohA, respectively. For the dual insert, the silencing construct was generated by inserting a 234-bp NbrbohA cDNA fragment, which constituted most of the conserved region of rboh, into the viral vector. This fragment showed 80% nucleotide identity to NbrbohB.
Two to 3 weeks after inoculation of leaves with PVX-NbrbohA (PVX:A), PVX-NbrbohB (PVX:B), PVX-NbrbohA/B dual-silencing insert (PVX:A/B), or PVX (no insert), plants were analyzed for NbrbohA and NbrbohB transcripts 24 h after treatment with INF1 (HR+) or Phytophthora (Figure 4). The transcript levels were determined by RT-PCR using gene-specific primers that amplify regions that do not contain insert sequences for PVX vector. The transcript accumulation of NbrbohA was reduced dramatically in PVX:A- and PVX:A/B-inoculated plants but was unaffected in NbrbohB-silenced plants. On the other hand, NbrbohB was silenced in PVX:B- and PVX:A/B-inoculated plants but not in NbrbohA-silenced plants. These data indicate that the NbrbohA or NbrbohB isogene was silenced specifically when insert A or insert B was used, respectively, and that both genes were silenced when the dual insert was used. We estimated the relative amounts of NbrbohA and NbrbohB transcripts by densitometric scanning of the negative films from the DNA gels of PCR products. In all cases, >80% of the transcripts were reduced by VIGS. The coat protein (CP) transcript of PVX was amplified as a marker for viral infection. EF-1α mRNA was unaltered in all plants.
Figure 4.
Effect of VIGS on the Transcription of Nbrboh Genes.
N. benthamiana leaves were inoculated with PVX, PVX:A (A), PVX:B (B), or PVX:A/B (A/B) or treated with water (C). Total RNA was isolated from leaves at 24 h after inoculation with zoospores of Phytophthora or injection with 10 μg/mL FLAG-INF1 (HR+) and used for RT-PCR as described in Methods, applying 35, 29, and 30 amplification cycles with specific primers for NbrbohA, NbrbohB, and CP, respectively. Equal loading of cDNA was monitored by amplification of a constitutively expressed EF-1α gene (29 cycles).
Reduction of the Oxidative Burst and Disease Resistance of Nbrboh-Silenced N. benthamiana to Phytophthora
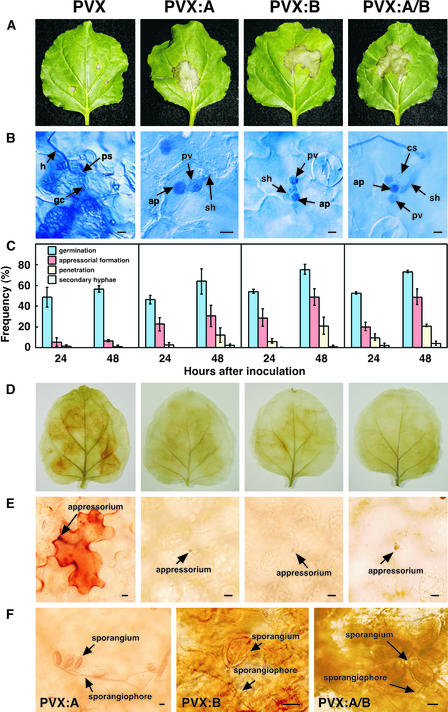
We evaluated whether silencing of Nbrboh genes had effects on the plant's resistance response against Phytophthora (Figure 5). The zoospores of Phytophthora race 0 could not infect the PVX control–inoculated leaves of N. benthamiana, and the leaf displayed macroscopic HR at 12 days after inoculation (Figure 5A, left). In some cases, no visible response was observed in the spots inoculated with Phytophthora. To determine the cytological basis of the resistance of N. benthamiana to Phytophthora, we observed the interaction microscopically using lactophenol-trypan blue–stained discs of inoculated leaves. After 3 days of inoculation, granular cytoplasm was observed in the responding cells, suggesting that a typical HR occurred in some but not all cases (Figure 5B, left). In most cases, the primary infection vesicle inside the host cell was not formed, suggesting that the pathogen is restricted to the cell during attempted penetration (Figure 5C, left). Moreover, we conducted 3,3′-diaminobenzidine (DAB) staining, which shows H2O2 accumulation visualized as a brown precipitate (Thordal-Christensen et al., 1997). Strongly polymerized DAB was detected 24 h after inoculation in the inoculated areas of whole leaf (Figure 5D, left) and in attacked cells (Figure 5E, left).
Figure 5.
A Representative Set of Nbrboh-Silenced Leaves after Inoculation with Phytophthora.
(A) After 3 weeks of inoculation with PVX (control), PVX:A, PVX:B, or PVX:A/B, N. benthamiana leaves were inoculated with four drops of Phytophthora zoospores. The Nbrboh-silenced leaves showed typical water-soaked lesions from the pathogen. Photographs were taken at 12 days after the inoculation.
(B) N. benthamiana leaf discs were stained with lactophenol-trypan blue at 3 days after inoculation with Phytophthora. ap, appressorium; cs, cystospore; gc, granular cytoplasm; h, hyphae; ps, penetration site; pv, primary vesicle; sh, secondary hyphae. Bars = 5 μm.
(C) Infection behavior of Phytophthora on Nbrboh-silenced N. benthamiana. Frequencies for cystospore germination, appressoria formation, penetration, and secondary hyphae formation of Phytophthora were assessed on leaf surfaces of N. benthamiana.
(D) H2O2 accumulation visualized by DAB in N. benthamiana leaves at 24 h after inoculation with Phytophthora. Detached leaves were stained with DAB solution as described in Methods.
(E) Microscopic observations of the DAB-stained leaves of N. benthamiana shown in (D). Bars = 10 μm.
(F) Sporulation was observed on Nbrboh-silenced N. benthamiana leaves at 12 days after inoculation with Phytophthora. Bars = 10 μm.
We performed similar analyses with PVX:A-, PVX:B-, and PVX:A/B-inoculated plants at both the macroscopic and microscopic levels, as illustrated in Figure 5. The typical water-soaked Phytophthora lesions appeared within 7 days after inoculation, and the expanding disease lesions around the inoculated spot were observed in all silenced plants at 12 days after inoculation (Figure 5A). As early as 3 days after inoculation, primary infection vesicles and secondary hyphae were observed in the infected cells at a lower rate (Figures 5B and 5C), indicating a partial loss of resistance. Importantly, brownish polymerized DAB was not detected at 24 h after inoculation in the inoculated cells, indicating that both NbrbohA and NbrbohB are required for the oxidative burst in response to Phytophthora (Figures 5D and 5E). Nbrboh silencing resulted in an increase in appressoria formation compared with that in the PVX control. These results suggest that the oxidative burst is involved in the early process of plant resistance to microbial pathogenesis. On the underside of the leaves (the leaf surface opposite to the inoculated side), sporangiophores emerging from stomata and numerous sporangia were readily observed in PVX:A-, PVX:B-, and PVX:A/B-inoculated N. benthamiana (Figure 5F).
VIGS of Nbrboh Compromises INF1-Mediated HR
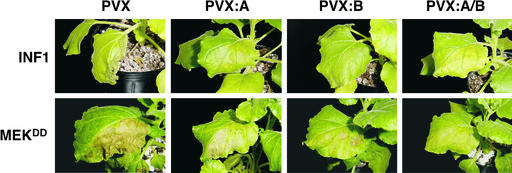
To determine whether the HR cell death observed in N. benthamiana after recognition of the protein elicitor INF1 could involve an rboh-mediated oxidative burst, inf1 was expressed under the control of the 35S promoter of Cauliflower mosaic virus (CaMV) using an agroinfiltration method in Nbrboh-silenced plants (Figure 6, top row). Because Phytophthora transformants deficient in INF1 induce disease lesions when inoculated on N. benthamiana, it has been suggested that INF1 functions as an avirulence factor in the interaction (Kamoun et al., 1998). At 3 days after infiltration, the necrotic reaction induced by INF1 was observed in PVX control–inoculated leaves of N. benthamiana, indicating that a typical HR response had occurred. By contrast, PVX:A-, PVX:B-, and PVX:A/B-inoculated plants displayed significantly reduced and delayed necrotic symptoms by INF1. This finding indicates that HR caused by INF1 is dependent on both NbrbohA and NbrbohB.
Figure 6.
HR Induction in N. benthamiana Leaves Silenced with Nbrboh.
N. benthamiana leaves were inoculated with PVX (control), PVX:A, PVX:B, or PVX:A/B. At 3 weeks after inoculation, a silenced leaf above the primary infected leaf was infiltrated with A. tumefaciens expressing INF1 or MEKDD as indicated carrying PR-1a:INF1 or the constitutive active mutant of MAPKK on one leaf half. Photographs of INF1 and MEKDD leaves were taken at 3 and 5 days after infiltration, respectively.
VIGS of NbrbohB Compromises MEKDD-Mediated HR
Recent studies have indicated that expression of a constitutively active mutant of MAPKK, MEKDD, activates endogenous SIPK and WIPK and leads to an oxidative burst (Ren et al., 2002). To address how the MAPK cascade regulates rboh-mediated HR cell death, MEKDD was expressed by agroinfiltration in Nbrboh-silenced N. benthamiana. As shown in Figure 6 (bottom row), HR-like cell death initiated by MEKDD was significantly reduced and delayed, and it developed only around the infiltration sites in PVX:B- and PVX:A/B-inoculated plants but not in PVX:A-inoculated plants. In most cases, the HR-like cell death initiated by MEKDD in PVX:B- and PVX:A/B-inoculated plants occurred 2 to 3 days later than that observed in PVX control–inoculated leaves. These results indicate that the cell death that resulted from the activation of the MAPK pathway is dependent on active NbrbohB.
Induction of NbrbohB by MAPKK Activity
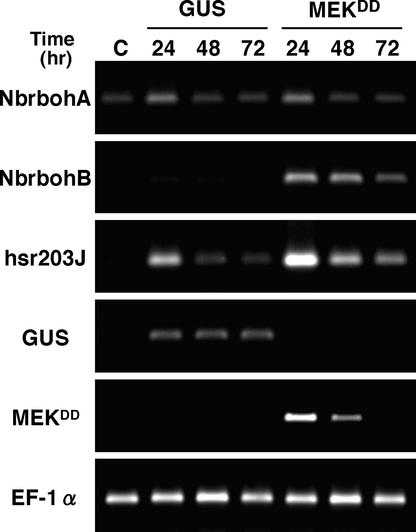
To address the question of how the MAPK cascade initiates HR-like cell death, we examined the transcript levels of Nb-rbohA and NbrbohB in leaves of N. benthamiana after infiltration of Agrobacterium tumefaciens harboring MEKDD or β-glucuronidase (GUS) as a control under the control of the 35S promoter of CaMV (Figure 7). NbrbohA was expressed constitutively at a low level before injection. The increase in the transcript levels of NbrbohA and hsr203J was detected 24 h after injection, except for in the control. This increase likely was caused by the mechanical stress of the infiltration procedure (Figure 3) or by infection stimulus from A. tumefaciens. Conversely, expression of MEKDD resulted in specific elicitation of NbrbohB transcript within 24 h after infiltration. The transcript level of EF-1α, an abundant protein of the cell, was unaltered under these conditions. When the leaves were infiltrated with A. tumefaciens harboring MEKDD, a maximum accumulation of the transcript was detected within 24 h, after which it declined. A massive H2O2 accumulation was detected by DAB staining, and HR-like necrosis appeared within 24 h (data not shown), as reported by Ren et al. (2002). Thus, we found that NbrbohB mRNA levels are influenced by a MAPK cascade.
Figure 7.
Effect of the Overexpression of MEKDD on the Transcription of Nbrboh Genes.
N. benthamiana leaves were infiltrated with A. tumefaciens harboring MEKDD or GUS (control) under the control of the 35S promoter of CaMV. Total RNA was isolated from leaves at 1 to 3 days after agroinfiltration or from nontreated leaves (C) and used for RT-PCR as described in Methods, applying 35, 29, and 29 amplification cycles with specific primers for NbrbohA, NbrbohB, and hsr203J, respectively. Transgene efficiency was monitored by amplification of GUS and MEKDD mRNAs (25 cycles). Equal loading of cDNA was monitored by amplification of a constitutively expressed EF-1α gene (29 cycles).
DISCUSSION
Function of Nbrboh Genes in the Oxidative Burst and Defense Responses
We used VIGS in N. benthamiana to define biological functions for two Nbrboh genes in the oxidative burst during the defense response. We observed that NbrbohA-, NbrbohB-, and Nb-rbohA/B-silenced plants show no DAB staining, a standard marker of H2O2 accumulation, after challenge with the nonpathogen Phytophthora (Figures 5D and 5E), suggesting that the enzyme that generates AOS in N. benthamiana is similar to phagocyte NADPH oxidase in mammalian cells. Furthermore, we found that Nbrboh-silenced N. benthamiana plants became susceptible to Phytophthora and exhibited enhanced sporangia and disease lesion formation by the pathogen (Figure 5). In PVX:A-, PVX:B-, and PVX:A/B-inoculated plants, necrotic symptoms caused by INF1 were significantly reduced and delayed (Figure 6, top row), consistent with the result that Phytophthora could infect those leaves.
The results presented here indicate that NbrbohA and NbrbohB share functions in H2O2 generation and defense responses, although the expression profiles of these genes are different in response to various stimuli. NbrbohA was expressed constitutively at a low level and was induced by the control leaf infiltration, whereas NbrbohB showed pathogen signal–specific elicitation in response to INF1 and exogenous SA (Figure 3). It is possible that in plants, elicitor induction of rboh mRNA is one of the mechanisms that control the oxidative burst machinery, perhaps leading to a long-term generation of AOS that could be responsible for cell death. Such elicitation of Arabidopsis AtrbohD and potato StrbohB has been reported (Desikan et al., 1998; Yoshioka et al., 2001). Recently, Simon-Plas et al. (2002) showed that tobacco NtrbohD, which is homologous with NbrbohB, is induced by the elicitin cryptogein and that tobacco cells transformed with antisense constructs of the gene abolished elicitor-stimulated AOS production. Their results coincide with our finding that NbrbohB-silenced leaves could not produce AOS during defense.
NbrbohA and NbrbohB are homologous with AtrbohF and AtrbohD, respectively, compared with Arabidopsis rboh proteins (Figure 2A). Using a dSpm insertion mutagenesis system, Torres et al. (2002) showed that atrbohD/F double mutations largely eliminate AOS accumulation during disease resistance reactions of Arabidopsis to avirulent Pseudomonas syringae pv tomato DC3000 and Peronospora parasitica. They also demonstrated that each mutation plays a different role in the defense of the plant. For instance, the atrbohD mutation eliminated the majority of AOS production, whereas atrbohF exhibited a strong effect on HR cell death in response to avirulent bacteria. Our analysis of the VIGS of Nbrboh genes suggested that the silencing of either NbrbohB or NbrbohA is enough to eliminate the oxidative burst in response to Phytophthora (Figures 5D and 5E). How can silencing of only one rboh gene lead to the suppression of detectable AOS production and HR in response to pathogen attack? One possible explanation is provided by Delledonne et al. (2001). Nitric oxide (NO) has been implicated in the activation of disease resistance mechanisms in plants (Noritake et al., 1996; Delledonne et al., 1998; Durner et al., 1998). Bacteria-induced HR in soybean suspension cells requires a fine balance between NO and H2O2 (Delledonne et al., 2001). Depletion of AOS production or administration of NO at high concentrations blocks HR cell death triggered by avirulent bacterial pathogens. NO and O2−, which is a precursor of H2O2, are produced independently (Durner et al., 1998) and could form peroxynitrate (ONOO−) after pathogen attack. However, HR does not appear to be mediated by ONOO−. Reduction of total AOS levels in Nbrboh-silenced plants would cause a reduction of positive signal (H2O2) for HR by a scavenging effect of NO on O2− and result in an imbalance of those molecules.
In plants, several kinds of enzyme are implicated in O2− generation in response to pathogen signals, such as NADPH oxidase in the plasma membrane, peroxidase in the cell wall, and oxalate oxidase (Kawano et al., 1998; Zhou et al., 1998). Other recent experiments using pharmacological agents suggested that at least two distinct sources, NADPH oxidase and peroxidase-type enzymes, contribute to the generation of redox cues (Allan and Fluhr, 1997; Grant et al., 2000). These reports indicate that the dominant source of AOS generation depends on the experimental system. Recently, Sagi and Fluhr (2001) confirmed that a putative plasma membrane NADPH oxidase from tobacco can produce O2− using a novel activity gel assay. Here, we provide genetic evidence for a dominant role of NADPH oxidase in the generation of AOS during the defense response in N. benthamiana leaves.
MAPK Pathway–Mediated HR-Like Cell Death Is Dependent on NbrbohB
MAPK cascades are known as major pathways by which extracellular stimuli are transduced into intracellular responses in plants (Asai et al., 2002). Recent studies have indicated that expression of a constitutively active mutant of MAPKK, MEKDD, activates endogenous SIPK and WIPK and leads to an HR that includes defense gene activation, such as Phe ammonia-lyase and 3-hydroxy-3-methylglutaryl CoA reductase (Yang et al., 2001). Ren et al. (2002) reported that the activation of endogenous Arabidopsis MAPKs by MEKDD transgenes under induced conditions leads to HR-like cell death, which is preceded by the generation of H2O2. They demonstrated that the oxidative burst might be the result of prolonged activation of a MAPK pathway. Both the activation of MAPKs and the burst precede and correlate with the HR, indicating that MAPK-induced HR-like cell death might be mediated by AOS. We demonstrated previously that two hyphal wall elicitors of Phytophthora, arachidonic acid and SA, activate a 51-kD MAPK (which may be a SIPK ortholog of tobacco) in potato tubers (Katou et al., 1999) and that these pathogen signals also stimulated both the oxidative burst and StrbohB (Yoshioka et al., 2001). Here, we showed that cell death resulting from the activation of the MAPK pathway is compromised in NbrbohB-silenced plants (Figure 7), suggesting that MAPK-induced HR-like cell death might be mediated by AOS produced by NbrbohB.
We have demonstrated that MEKDD induced the NbrbohB gene at the transcriptional level and HR-like cell death (Figures 6 and 7). However, the oxidative burst has been shown to be controlled through phosphorylation activation by its upstream kinase (Levine et al., 1994; Miura et al., 1995; Piedras et al., 1998) and dephosphorylation inactivation by its negative regulator, phosphatase. Treatment of cultured rice cells with the phosphatase inhibitor calyculin A without elicitor rapidly stimulates a diphenylene iodonium–sensitive oxidative burst (Kawasaki et al., 1999), suggesting that a small portion of its upstream kinase is active. A sudden increase in a large amount of NbrbohB by the long-lasting activation of MAPKs disrupts the balance in the cells. After the newly synthesized NbrbohB becomes activated by the basal level of upstream kinase, there is not enough phosphatase activity to inactivate NbrbohB, which leads to lethal levels of oxidative burst. Alternatively, in mammalian cells, MAPK also is implicated in the activation of phospholipase A2, protein kinase C, and Ras upstream of the MAPK to stimulate the signaling network (Bhalla et al., 2002), suggesting that a potential role of the MAPK cascade is not only the transcriptional activation of a number of defense genes but the post-translational activation of enzymes. In fact, a highly specific inhibitor of MAPKK, U0126, suppresses the rapid AOS generation induced by the Avr9 elicitor in Cf-9–expressing tobacco cell cultures (O. Rowland, and J.D.G. Jones, unpublished results). It is possible that MEKDD-mediated sustained SIPK and WIPK activities provoke the post-translational activation of NbrbohB in addition to an increase in the transcript level.
Crosstalk between the Oxidative Burst and the MAPK Cascade
It has been reported that overexpression of constitutively active deletion mutants of a MAPKK kinase activated AtMPK3 (a WIPK ortholog) and AtMPK6 (a SIPK ortholog) of the six co-overexpressed MAPKs tested in Arabidopsis protoplasts exposed to acute H2O2 stress (Kovtun et al., 2000). Moreover, exposure of Arabidopsis leaves to H2O2 by glucose/glucose oxidase infiltration activates 46- and 48-kD MAPKs orthologous with WIPK and SIPK of tobacco, respectively (Grant et al., 2000). Those authors also indicated that treatment with the MAPKK inhibitor PD98059 completely suppresses the activation of both MAPKs induced by H2O2. Here, we showed that prolonged activation of the MAPK pathway in leaf cells activated the NbrbohB gene (Figure 7), suggesting that the MAPK cascade–dependent increase of rboh mRNA levels could be an amplification circuit in the induction of defense responses (Figure 8). We confirmed that VIGS of SIPK/WIPK compromised INF1-mediated HR cell death in N. benthamiana (N. Ikeda, O. Rowland, J.D.G. Jones, and H. Yoshioka, unpublished results). However, the frequencies for the germination, appressoria formation, primary infection vesicle, and secondary hyphae in the Nbrboh-silenced leaves were low, suggesting that the silenced plants remain partially resistant to the pathogen. We speculate that the MAPK pathway could be activated after the perception of the pathogen in the absence of H2O2, which leads to partial resistance, or that Nbrboh needs to be fully silenced (no transcript) to complete the loss of resistance. Alternatively, other mechanisms may be activated for plant defense responses (Sasabe et al., 2000; Peart et al., 2002). Further investigation of the upstream and downstream regulatory components of the oxidative burst should clarify the interlinked signal network of defense pathways in plants.
Figure 8.
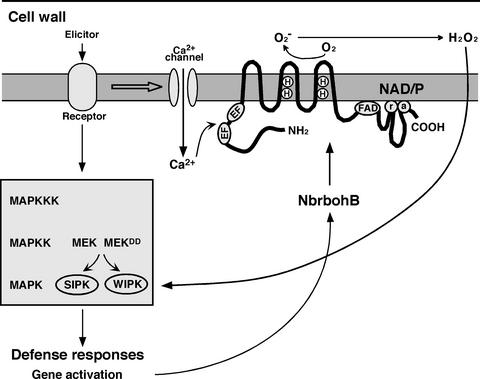
Scheme of the NbrbohB Protein and Proposed Model for the Regulatory Mechanism of NbrbohB Gene Expression.
The elicitor induces various defense-related genes, including NbrbohB, through the MAPK cascade. H2O2 produced by NbrbohB activates SIPK and WIPK. Nbrboh contains the membrane with six transmembrane domains and the putative positions of the two heme binding sites (H, histidine), as indicated in Figure 1, the flavin adenine dinucleotide (FAD), and NAD/P (a, adenine; r, ribose) binding sites, and the EF hands.
METHODS
Plant Materials, INF1 Elicitor, Chemicals, and Treatment Protocols
Nicotiana benthamiana plants were grown in environmentally controlled growth cabinets under a 16-h photoperiod and an 8-h dark period at 25°C. Elicitation with INF1 was conducted on 4-week-old plants by infiltrating INF1 solution with a needleless syringe into tiny cuts at the underside of the leaf, thereby flooding the apoplastic space. The response to osmotic stress (without the elicitation component) was investigated by infiltration of water. To prepare Phytophthora infestans elicitin FLAG-INF1 (HR+) and FLAG-INF1 S3 (HR−), in which the Cys at position 3 of the processed protein is mutated to a Ser (Kamoun et al., 1997), overnight cultures of Escherichia coli DH5α carrying either pFB52 (−) or pFB53 (+) were diluted (1:100) in Luria-Bertani medium containing ampicillin (50 μg/mL) and incubated at 37°C. When the OD600 of cultures reached 0.6, isopropylthio-β-galactoside was added to a final concentration of 0.4 mM, and the cultures were grown for 3 to 4 h to induce the elicitin into cultured medium. The E. coli cells were pelleted by centrifugation, and the supernatant was filtrated through a 0.45-μm pore filter (Kurabo, Osaka, Japan) to eliminate E. coli; this preparation then was used as elicitin. Plants were treated with salicylic acid or methyl jasmonic acid (Sigma, St. Louis, MO) infiltration with a needleless syringe as described above for INF1 infiltration.
Zoospore Inoculation and Plant Assays
Race 0 of Phytophthora was maintained on susceptible potato (Solanum tuberosum) tubers. A suspension of Phytophthora zoospores was prepared as described previously (Yoshioka et al., 1999). Ten-microliter droplets containing ∼500 zoospores were applied to the upper side of the attached leaves under high humidity at 20°C. In the case of RNA isolation, N. benthamiana leaves were inoculated with 1 × 104 Phytophthora zoospores/mL using a lens paper to disperse the zoospores. For the microscopic observation, leaf discs containing the inoculum were excised at various times after inoculation with Phytophthora and then fixed on the filter paper by immersion in a 3:1 solution of ethanol:acetic acid. The fixed samples were stained carefully with 0.1 μg/mL trypan blue solution to avoid washing spores away and then examined by microscopy for plant responses and growth of Phytophthora.
Isolation of gp91phox Homologs from N. benthamiana
Two cDNAs for potato rboh, StrbohA and StrbohB, were used as probes to screen an oligo(dT)-primed Uni-ZAP XR library (Stratagene, La Jolla, CA), which was constructed from poly(A)+ RNAs of N. benthamiana leaves treated with hyphal walls of Phytophthora for 6 h. From 6 × 105 plaques of the library, six positive clones were isolated after three rounds of plaque hybridization under low-stringency conditions by each probe (12 positive clones total). Positive phage plaques were excised directly in vivo into pBluescript SK− phagemid according to the manufacturer's instructions (Stratagene). The nucleotide sequences were determined for both strands using the Prism Dye Termination Cycle Sequencing Ready Reaction Kit and a DNA sequencing system (model 310; Applied Biosystems, Foster City, CA). The positive clones obtained by the StrbohA probe contained one NbrbohA and five NbrbohB cDNAs, whereas the StrbohB probe detected six NbrbohB cDNAs. The partial sequences of 11 NbrbohB cDNAs were identical.
Reverse Transcriptase–Mediated PCR
Total RNA from leaf tissues was prepared as described previously (Yoshioka et al., 1996). Reverse transcriptase–mediated (RT) PCR was conducted using a commercial kit (rTth DNA polymerase RT-PCR high-Plus-; Toyobo Co., Osaka, Japan). cDNA was synthesized from total RNA (1 μg). After the cDNA synthesis reaction, PCR was performed with a denaturing temperature of 94°C for 1 min and annealing plus extension temperatures of 53°C for 1.5 min, for 29 to 35 cycles. Gene-specific primers of each sequence were as follows: NbrbohA (forward, 5′-CTG-CTCAGGGCTGACGAAAACACCAAGAAA-3′; reverse, 5′-AGTACTCTT-TCTCCTTTTCGGAGAAATCTT-3′), NbrbohB (forward, 5′-TTTCTCTGA-GGTTTGCCAGCCACCACCTAA-3′; reverse, 5′-GCCTTCATGTTGTTG-ACAATGTCTTTAACA-3′), hsr203J (forward, 5′-TGTGTCAGCCATGCTGATTG-3′; reverse, 5′-CCGATAGGACCGCACGAAAC-3′), and CP (forward, 5′-TGGGACTTAGTCAGACACTG-3′; reverse, 5′-CCCCTCTTT-GGGCATGATGG-3′). As a control for equal cDNA amounts in each reaction, PCR was performed with primers for EF-1α (forward, 5′-CTTCCTACC-TCAAGAAGGTAGGATACAAC-3′; reverse, 5′-TGCCTCCTGAAGAGC-TTCGTGGTGCAT-3′).
In the case of β-glucuronidase (GUS) or MEKDD transcript amplification, cDNA was synthesized with oligo(dT) primer to distinguish plant RNA from bacterial RNA. RT-PCR was conducted using a commercial kit (High-Fidelity RNA PCR; Takara, Kusatsu, Japan). PCR was performed with denaturing, annealing, and extension temperatures of 94°C for 30 s, 53°C for 30 s, and 72°C for 1 min, respectively, for 25 cycles. Gene-specific primers of each sequence were as follows: GUS (forward, 5′-GATGTGCTGTGCCTGAACCGTTATTACGGA-3′; reverse for 3′ untranslated region sequence derived from binary vector, 5′-GCTTGACTCTAGAGG-ATCTTATGCGTAGTC-3′) and MEKDD (forward, 5′-CCTTCTCTGTGG-GGAGACAAGGAGACTGGG-3′; reverse for 3′ untranslated region sequence derived from binary vector, 5′-GCTTGACTCTAGAGGATCTTA-TGCGTAGTC-3′). The gene-specific primers of NbrbohA, NbrbohB, hsr203J, GUS, MEKDD, CP, and EF-1α generate expected product sizes of 291, 228, 354, 492, 347, 297, and 347 bp, respectively. PCR products were separated on a 1.8% agarose gel and visualized after ethidium bromide staining.
DNA Constructs and Seedling Infection for Virus-Induced Gene Silencing
A 278-bp cDNA fragment of the isogene-specific N-terminal region of NbrbohA was amplified with the following primers: NbrbohA-SalI (5′-GTCGACGCCGGTGGACATCCGATACGGTAT-3′) and NbrbohA-ClaI (5′-ATCGATTCGTCGGCGATCGAGATTCCGATA-3′) (restriction sites are underlined). A 197-bp cDNA fragment of an isogene-specific region of NbrbohB was amplified with primers NbrbohB-SalI (5′-GTCGACAATCATCATCCGCACCACCATCAC-3′) and NbrbohB-ClaI (5′-ATCGATACGCATCATCATTGGACTTGCCGC-3′). A 234-bp cDNA fragment for a conserved region of Nbrboh was amplified with primers NbrbohD-SalI (5′-GTCGACATATTTTTATTGGGTGACCCGGGA-3′) and NbrbohD-ClaI (5′-ATCGATCTAGCAAAATGTGTCCTCACCCTG-3′) using NbrbohA cDNA as a template. These cDNA fragments were subcloned into the TA cloning site of pGEM-T Easy (Promega, Madison, WI) and ligated as a ClaI-SalI fragment into the Potato virus X (PVX) vector pPC2S (Baulcombe et al., 1995). The constructs contained these inserts in the antisense orientation and were designated PVX-NbrbohA (PVX:A), PVX-NbrbohB (PVX:B), and PVX-NbrbohA/B dual-silencing insert (PVX:A/B). PVX that does not contain any inserts was used as a control. Synthesis of virus RNA from the construct and inoculation onto a leaf of N. benthamiana were as described by Chapman et al. (1992). Second leaves of 2-week-old N. benthamiana seedlings were inoculated with viral RNA using Carborundum. After an additional 2 to 3 weeks, the fourth and fifth leaves above the inoculated leaves of each plant were analyzed for transcript levels of various genes and used for Agrobacterium tumefaciens–mediated transient expression.
Transient Expression of INF1 and StMEKDD in N. benthamiana
For agroinfiltration experiments, we used the binary vector p35S-INF1 containing a fusion between the signal peptide of tobacco (Nicotiana tabacum) PR-1a and the Phytophthora inf1 gene driven by the 35S promoter of Cauliflower mosaic virus (Kamoun et al., 1999; S. Kamoun, personal communication).
To clone the ortholog of tobacco NtMEK2 (Yang et al., 2001) from potato, we performed PCR with primers StMEK-F (5′-ATGCGACCTCTT-CAACCACCCCCACCAG-3′) and StMEK-R (5′-TTCTCCAAAAATAAA-ATCCAACCACCC-3′) using a potato cDNA library as a template. The amplified 1258-bp (full-length) fragment was subcloned into the TA cloning site of pGEM-T Easy (Promega) and sequenced. The constitutively active mutant of StMEK1, StMEKDD, in which the conserved Thr-228 and Ser-234 residues are replaced by Asp (Yang et al., 2001), was generated by Mutan-Super Express Km site-directed mutagenesis (Takara) and confirmed by sequencing. StMEKDD was inserted into ClaI-BamHI sites of the vector pSLJ4K1 (Jones et al., 1992). The HindIII-EcoRI fragment then was cloned into the pGreen binary vector (Hellens et al., 2000). The StMEKDD was preceded by the 35S promoter of Cauliflower mosaic virus, the 5′ untranslated region was replaced with the ω sequence from Tobacco mosaic virus, and the nopaline synthase terminator region was on the 3′ end of the gene.
These binary plasmids were transformed into A. tumefaciens strain GV3101, which harbors the transformation helper plasmid pSoup (Hellens et al., 2000). Overnight cultures were harvested by centrifugation, and cells were resuspended in 10 mM MgCl2, 10 mM Mes, pH 5.6, and 150 μM acetosyringone to an OD600 of 0.5, incubated for 2 to 5 h at room temperature, and then infiltrated into leaves of N. benthamiana using a needleless syringe (Romeis et al., 2001). To analyze for defense-related phenotypes, the silenced fourth and fifth leaves were infiltrated with A. tumefaciens expressing PR-1a:inf1 or StMEKDD on one leaf half.
DNA Gel Blot Hybridization
Total genomic DNA of N. benthamiana was extracted from leaves according to the method described by Murray and Thompson (1980). Ten micrograms of DNA was digested with HindIII, EcoRI, or SpeI and then separated by electrophoresis on a 0.8% agarose gel. The separated DNA was transferred from gels to nylon membranes (Hybond N+; Amersham, Arlington Heights, IL) and hybridized according to the manufacturer's recommendations. The cDNA probes for NbrbohA (278 bp) and NbrbohB (197 bp) that were used for virus-induced gene silencing were labeled with 32P-dCTP using a random-primed DNA labeling kit (Takara).
H2O2 Detection by the 3,3′-Diaminobenzidine Uptake Method
To visualize H2O2 in the infection site of Phytophthora, 3,3′-diaminobenzidine staining was performed as described by Thordal-Christensen et al. (1997). N. benthamiana leaves were inoculated with 1 × 104 Phytophthora zoospores/mL using a lens paper to disperse the zoospores at 20°C. Detached leaf samples were collected at 8 h after incubation with 1 mg/mL 3,3′-diaminobenzidine solution. Leaves then were fixed with a 3:1:1 solution of ethanol:acetic acid:glycerol.
Upon request, all novel materials described in this article will be made available in a timely manner for noncommercial research purposes.
Accession Numbers
The accession numbers for the NbrbohA and NbrbohB cDNA sequences are AB079498 and AB079499, respectively. The accession numbers for the other sequences mentioned in this article are as follows: potato MAPKK (StMEK1), AB091780; Arabidopsis respiratory burst oxidase homologs (AtrbohA to AtrbohF), AF055353, AF055354, AF055355, AF055357, AF055356, and AB008111, respectively; rice rboh (OsrbohA), AP003560; tomato rboh (Lerboh1), AF088276; Wfi1 (tomato), AF147783; potato rboh (StrbohA), AB050660; StrbohB, AB050661; tobacco rboh (NtrbohD), AJ309006; and NtrbohF, AJ320505. Arabidopsis AtrbohA (AF015301), referred to by Keller et al. (1998), corresponds to AtrbohF (AB008111), referred to by Torres et al. (1998).
Acknowledgments
We deeply thank David C. Baulcombe of Sainsbury Laboratory for providing PVX vector pPC2S; Sophien Kamoun of Ohio State University for pFB52, pFB53, and p35S-INF1 and information regarding unpublished results; P. Mullineaux and R. Hellens of the John Innes Centre for pGreen binary vectors; and the Leaf Tobacco Research Center, Japan Tobacco, Inc., for N. benthamiana seeds. We also thank Takashi Tsuge for valuable suggestions and the Radioisotope Research Center, Nagoya University, for technical assistance. This work was supported in part by a Grant-in-Aid for Scientific Research (S) (14104004) from the Ministry of Education, Science, Sports, and Culture of Japan and by a Grant-in-Aid from the Research for the Future Program of the Japan Society for the Promotion of Science.
Article, publication date, and citation information can be found at www.plantcell.org/cgi/doi/10.1105/tpc.008680.
References
- Allan, A.C., and Fluhr, R. (1997). Two distinct sources of elicited reactive oxygen species in tobacco epidermal cells. Plant Cell 9, 1559–1572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amicucci, E., Gaschler, K., and Ward, J.M. (1999). NADPH oxidase genes from tomato (Lycopersicon esculentum) and curly-leaf pondweed (Potamogeton crispus). Plant Biol. 1, 524–528. [Google Scholar]
- Apostol, I., Heinstein, P.F., and Low, P.S. (1989). Rapid stimulation of an oxidative burst during elicitation of cultured plant cells: Role in defense and signal transduction. Plant Physiol. 90, 109–116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Asai, T., Tena, G., Plotnikova, J., Willmann, M.R., Chiu, W.-L., Gomez-Gomez, L., Boller, T., Ausubel, F.M., and Sheen, J. (2002). MAP kinase signalling cascade in Arabidopsis innate immunity. Nature 415, 977–983. [DOI] [PubMed] [Google Scholar]
- Auh, C.-K., and Murphy, T.M. (1995). Plasma membrane redox enzyme is involved in the synthesis of O2− and H2O2 by Phytophthora elicitor-stimulated rose cells. Plant Physiol. 107, 1241–1247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baulcombe, D.C. (1999). Fast forward genetics based on virus-induced gene silencing. Curr. Opin. Plant Biol. 2, 109–113. [DOI] [PubMed] [Google Scholar]
- Baulcombe, D.C., Chapman, S., and Cruz, S.S. (1995). Jellyfish green fluorescent protein as a reporter for virus infections. Plant J. 7, 1045–1053. [DOI] [PubMed] [Google Scholar]
- Bhalla, U.S., Ram, P.T., and Lyengar, R. (2002). MAP kinase phosphatase as a locus of flexibility in a mitogen-activated protein kinase signaling network. Science 297, 1018–1023. [DOI] [PubMed] [Google Scholar]
- Bokoch, G.M. (1994). Regulation of the human neutrophil NADPH oxidase by the RacGTP-binding proteins. Curr. Opin. Cell Biol. 6, 212–218. [DOI] [PubMed] [Google Scholar]
- Bourque, S., Binet, M.N., Ponchet, M., Pugin, A., and Lebrun-Garcia, A. (1999). Characterization of the cryptogein binding sites on plant plasma membranes. J. Biol. Chem. 274, 34699–34705. [DOI] [PubMed] [Google Scholar]
- Bradley, D.J., Kjellbom, P., and Lamb, C.J. (1992). Elicitor- and wound-induced oxidative cross-linking of a proline-rich plant cell wall protein: A novel, rapid defense response. Cell 70, 21–30. [DOI] [PubMed] [Google Scholar]
- Chandra, S., and Low, P.S. (1997). Measurement of Ca2+ fluxes during elicitation of the oxidative burst in aequorin-transformed tobacco cells. J. Biol. Chem. 272, 28274–28280. [DOI] [PubMed] [Google Scholar]
- Chapman, S., Kavanagh, T., and Baulcombe, D.C. (1992). Potato virus X as a vector for gene expression in plants. Plant J. 2, 549–557. [DOI] [PubMed] [Google Scholar]
- Chen, Z., Silva, H., and Klessig, D.F. (1993). Active oxygen species in the induction of plant systemic acquired resistance by salicylic acid. Science 262, 1883–1886. [DOI] [PubMed] [Google Scholar]
- Dangl, J.L., and Jones, J.D.G. (2001). Plant pathogens and integrated defense responses to infection. Nature 411, 826–833. [DOI] [PubMed] [Google Scholar]
- Delledonne, M., Xia, Y., Dixon, R.A., and Lamb, C. (1998). Nitric oxide functions as a signal in plant disease resistance. Nature 394, 585–588. [DOI] [PubMed] [Google Scholar]
- Delledonne, M., Zeier, J., Marocco, A., and Lamb, C. (2001). Signal interactions between nitric oxide and reactive oxygen intermediates in the plant hypersensitive disease resistance response. Proc. Natl. Acad. Sci. USA 98, 13454–13459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desikan, R., Burnett, E.C., Hancock, J.T., and Neill, S.J. (1998). Harpin and hydrogen peroxide induce the expression of a homologue of gp91-phox in Arabidopsis thaliana suspension cultures. J. Exp. Bot. 49, 1767–1771. [Google Scholar]
- Desikan, R., A.-H.-Mackerness, S., Hancock, J.T., and Neill, S.J. (2001). Regulation of the Arabidopsis transcriptome by oxidative stress. Plant Physiol. 127, 159–172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dietrich, R.A., Richberg, M.H., Schmidt, R., Dean, C., and Dangl, J.L. (1996). A novel zinc finger protein is encoded by the Arabidopsis LSD1 gene and functions as a negative regulator of plant cell death. Cell 88, 685–694. [DOI] [PubMed] [Google Scholar]
- Doke, N. (1983). Involvement of superoxide anion generation in the hypersensitive response of potato tuber tissues to infection with an incompatible race of Phytophthora infestans and to the hyphal wall components. Physiol. Plant Pathol. 23, 345–357. [Google Scholar]
- Doke, N. (1985). NADPH-dependent O2− generation in membrane fractions isolated from wounded potato tubers inoculated with Phytophthora infestans. Physiol. Plant Pathol. 27, 311–322. [Google Scholar]
- Doke, N., and Miura, Y. (1995). In vitro activation of NADPH-dependent O2− generating system in a plasma membrane-rich fraction of potato tuber tissues by treatment with an elicitor from Phytophthora infestans or with digitonin. Physiol. Mol. Plant Pathol. 46, 17–28. [Google Scholar]
- Durner, J., Wendehenne, D., and Klessig, D.F. (1998). Defense gene induction in tobacco by nitric oxide, cyclic GMP, and cyclic ADP-ribose. Proc. Natl. Acad. Sci. USA 95, 10328–10333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finegold, A.A., Shatwell, K.P., Segal, A.W., Klausner, R.D., and Dancis, A. (1996). Intramembrane bis-heme motif for transmembrane electron transport conserved in a yeast iron reductase and the human NADPH oxidase. J. Biol. Chem. 271, 31021–31024. [DOI] [PubMed] [Google Scholar]
- Grant, J.J., Yun, B.-W., and Loake, G.J. (2000). Oxidative burst and cognate redox signalling reported by luciferase imaging: Identification of a signal network that functions independently of ethylene, SA and Me-JA but is dependent on MAPKK activity. Plant J. 24, 569–582. [DOI] [PubMed] [Google Scholar]
- Groom, Q.J., Torres, M.A., Fordham-Skelton, A.P., Hammond-Kosack, K.E., Robinson, N.J., and Jones, J.D.G. (1996). rbohA, a rice homologue of the mammalian gp91 phox respiratory burst oxidase gene. Plant J. 10, 515–522. [DOI] [PubMed] [Google Scholar]
- Hellens, R.P., Edwards, A.E., Leyland, N.R., Bean, S., and Mullineaux, P.M. (2000). pGreen: A versatile and flexible binary Ti vector for Agrobacterium-mediated plant transformation. Plant Mol. Biol. 42, 819–832. [DOI] [PubMed] [Google Scholar]
- Huet, J.C., Salle-Tourne, M., and Pernollet, J.C. (1994). Amino acid sequence and toxicity of the alpha elicitin secreted with ubiquitin by Phytophthora infestans. Mol. Plant-Microbe Interact. 7, 302–304. [DOI] [PubMed] [Google Scholar]
- Jabs, T., Dietrich, R.A., and Dangl, J.L. (1996). Initiation of runaway cell death in an Arabidopsis mutant by extracellular superoxide. Science 273, 1853–1856. [DOI] [PubMed] [Google Scholar]
- Jones, J.D.G., Shlumukov, L., Carland, F., English, J., Scofield, S., Bishop, G., and Harrison, K. (1992). Effective vectors for transformation, expression of heterologous genes, and assaying transposon excision in transgenic plants. Transgenic Res. 1, 285–297. [DOI] [PubMed] [Google Scholar]
- Kamoun, S., Honée, G., Weide, R., Laugé, R., Kooman-Gersmann, M., de Groot, K., Govers, F., and de Wit, P.J.G.M. (1999). The fungal gene Avr9 and the oomycete gene inf1 confer avirulence to potato virus X on tobacco. Mol. Plant-Microbe Interact. 12, 459–462. [Google Scholar]
- Kamoun, S., van West, P., de Jong, A.J., de Groot, K.E., Vleeshouwers, V.G.A.A., and Govers, F. (1997). A gene encoding a protein elicitor of Phytophthora infestans is down-regulated during infection of potato. Mol. Plant-Microbe Interact. 10, 13–20. [DOI] [PubMed] [Google Scholar]
- Kamoun, S., van West, P., Vleeshouwers, V.G.A.A., de Groot, K.E., and Govers, F. (1998). Resistance of Nicotiana benthamiana to Phytophthora infestans is mediated by the recognition of the elicitor protein INF1. Plant Cell 10, 1413–1425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamoun, S., Young, M., Glascock, C.B., and Tyler, B.M. (1993). Extracellular protein elicitors from Phytophthora: Host-specificity and induction of resistance to bacterial and fungal phytopathogens. Mol. Plant-Microbe Interact. 6, 15–25. [Google Scholar]
- Katou, S., Senda, K., Yoshioka, H., Doke, N., and Kawakita, K. (1999). A 51 kDa protein kinase of potato activated with hyphal wall components from Phytophthora infestans. Plant Cell Physiol. 40, 825–831. [Google Scholar]
- Kauss, H., and Jeblick, W. (1995). Pretreatment of parsley suspension cultures with salicylic acid enhances spontaneous and elicited production of H2O2. Plant Physiol. 108, 1171–1178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawano, T., Sahashi, N., Takahashi, K., Uozumi, N., and Muto, S. (1998). Salicylic acid induces extracellular superoxide generation followed by an increase in cytosolic calcium ion in tobacco suspension culture: The earliest events in salicylic acid signal transduction. Plant Cell Physiol. 39, 721–730. [Google Scholar]
- Kawasaki, T., Henmi, K., Ono, E., Hatakeyama, S., Iwano, M., Satoh, H., and Shimamoto, K. (1999). The small GTP-binding protein Rac is a regulator of cell death in plants. Proc. Natl. Acad. Sci. USA 96, 10922–10926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keller, T., Damude, H.G., Werner, D., Doerner, P., Dixon, R.A., and Lamb, C. (1998). A plant homolog of the neutrophil NADPH oxidase gp91phox subunit gene encodes a plasma membrane protein with Ca2+ binding motifs. Plant Cell 10, 255–266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kovtun, Y., Chiu, W.-L., Tena, G., and Sheen, J. (2000). Functional analysis of oxidative stress-activated mitogen-activated protein kinase cascade in plants. Proc. Natl. Acad. Sci. USA 97, 2940–2945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kretsinger, R.H. (1996). EF-hands reach out. Nat. Struct. Biol. 3, 12–15. [DOI] [PubMed] [Google Scholar]
- Lamb, C., and Dixon, R.A. (1997). The oxidative burst in plant disease resistance. Annu. Rev. Plant Physiol. Plant Mol. Biol. 48, 251–275. [DOI] [PubMed] [Google Scholar]
- Léon, J., Lawton, M.A., and Raskin, I. (1995). Hydrogen peroxide stimulates salicylic acid biosynthesis in tobacco. Plant Physiol. 108, 1673–1678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levine, A., Tenhaken, R., Dixon, R., and Lamb, C. (1994). H2O2 from the oxidative burst orchestrates the plant hypersensitive disease resistance response. Cell 79, 583–593. [DOI] [PubMed] [Google Scholar]
- Miura, Y., Yoshioka, H., and Doke, N. (1995). An autophotographic determination of the active oxygen generation in potato tuber discs during hypersensitive response to fungal infection or elicitor. Plant Sci. 105, 45–52. [Google Scholar]
- Murray, M.G., and Thompson, W.F. (1980). Rapid isolation of high-molecular-weight plant DNA. Nucleic Acids Res. 8, 4321–4325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noritake, T., Kawakita, K., and Doke, N. (1996). Nitric oxide induces phytoalexin accumulation in potato tuber tissues. Plant Cell Physiol. 37, 113–116. [Google Scholar]
- Peart, J.R., et al. (2002). Ubiquitin ligase-associated protein SGT1 is required for host and nonhost disease resistance in plants. Proc. Natl. Acad. Sci. USA 99, 10865–10869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peng, M., and Kuc, J. (1992). Peroxidase-generated hydrogen peroxide as a source of antifungal activity in vitro and on tobacco leaf discs. Phytopathology 82, 696–699. [Google Scholar]
- Piedras, P., Hammond-Kosack, K.E., Harrison, K., and Jones, J.D.G. (1998). Rapid, Cf-9 and Avr9-dependent, production of active oxygen species in tobacco suspension cultures. Mol. Plant-Microbe Interact. 11, 1155–1166. [Google Scholar]
- Ren, D., Yang, H., and Zhang, S. (2002). Cell death mediated by MAPK is associated with hydrogen peroxide production in Arabidopsis. J. Biol. Chem. 277, 559–565. [DOI] [PubMed] [Google Scholar]
- Ricci, P., Bonnet, P., Huet, J.-C., Sallantin, M., Beauvais-Cante, F., Bruneteau, M., Billard, V., Michel, G., and Pernollet, J.-C. (1989). Structure and activity of proteins from pathogenic fungi Phytophthora eliciting necrosis and acquired resistance in tobacco. Eur. J. Biochem. 183, 555–563. [DOI] [PubMed] [Google Scholar]
- Romeis, T., Ludwig, A.A., Martin, R., and Jones, J.D.G. (2001). Calcium-dependent protein kinases play an essential role in a plant defence response. EMBO J. 20, 5556–5567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rustérucci, C., Stallaert, V., Milat, M.-L., Pugin, A., Ricci, P., and Blein, J.-P. (1996). Relationship between active oxygen species, lipid peroxidation, necrosis, and phytoalexin production induced by elicitins in Nicotiana. Plant Physiol. 111, 885–891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sagi, M., and Fluhr, R. (2001). Superoxide production by plant homologues of the gp91phox NADPH oxidase: Modulation of activity by calcium and by tobacco mosaic virus infection. Plant Physiol. 126, 1281–1290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saitou, N., and Nei, M. (1987). The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 4, 406–425. [DOI] [PubMed] [Google Scholar]
- Sasabe, M., Takeuchi, K., Kamoun, S., Ichinose, Y., Govers, F., Toyoda, K., Shiraishi, T., and Yamada, T. (2000). Independent pathways leading to apoptotic cell death, oxidative burst and defense gene expression in response to elicitin in tobacco cell suspension culture. Eur. J. Biochem. 267, 5005–5013. [DOI] [PubMed] [Google Scholar]
- Segal, A.W., West, I., Wientjes, F., Nugent, J.H.A., Chavan, A.J., Haley, B., Garcia, R.C., Rosen, H., and Scrace, G. (1992). Cytochrome b-245 is a flavocytochrome containing FAD and the NADPH-binding site of the microbicidal oxidase of phagocytes. Biochem. J. 284, 781–788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shirasu, K., Nakajima, H., Rajasekhar, V.K., Dixon, R.A., and Lamb, C. (1997). Salicylic acid potentiates an agonist-dependent gain control that amplifies pathogen signals in the activation of defense mechanisms. Plant Cell 9, 261–270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simon-Plas, F., Elmayan, T., and Blein, J.-P. (2002). The plasma membrane oxidase NtrbohD is responsible for AOS production in elicited tobacco cells. Plant J. 31, 137–147. [DOI] [PubMed] [Google Scholar]
- Smith, H.H. (1979). The Nicotiana genus as a genetic resource. US Dep. Agric. Tech. Bull. 1586, 1–16. [Google Scholar]
- Tavernier, E., Wendehenne, D., Blein, J.-P., and Pugin, A. (1995). Involvement of free calcium in action of cryptogein, a proteinaceous elicitor of hypersensitive reaction in tobacco cells. Plant Physiol. 109, 1025–1031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson, J.D., Higgins, D.G., and Gibson, T.J. (1994). CLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 22, 4673–4680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thordal-Christensen, H., Zhang, Z., Wei, Y., and Collinge, D.B. (1997). Subcellular localization of H2O2 in plants: H2O2 accumulation in papillae and hypersensitive response during the barley-powdery mildew interaction. Plant J. 11, 1187–1194. [Google Scholar]
- Torres, M.A., Dangl, J.L., and Jones, J.D.G. (2002). Arabidopsis gp91phox homologues AtrbohD and AtrbohF are required for accumulation of reactive oxygen intermediates in the plant defense response. Proc. Natl. Acad. Sci. USA 99, 517–522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torres, M.A., Onouchi, H., Hamada, S., Machida, C., Hammond-Kosack, K.E., and Jones, J.D.G. (1998). Six Arabidopsis thaliana homologues of the human respiratory burst oxidase (gp91phox). Plant J. 14, 365–370. [DOI] [PubMed] [Google Scholar]
- Tronchet, M., Ranty, B., Marco, Y., and Roby, D. (2001). HSR203 antisense suppression in tobacco accelerates development of hypersensitive cell death. Plant J. 27, 115–127. [DOI] [PubMed] [Google Scholar]
- Yang, K.-Y., Liu, Y., and Zhang, S. (2001). Activation of a mitogen-activated protein kinase pathway is involved in disease resistance in tobacco. Proc. Natl. Acad. Sci. USA 98, 741–746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshioka, H., Miyabe, M., Hayakawa, Y., and Doke, N. (1996). Expression of genes for phenylalanine ammonia-lyase and 3-hydroxy- 3-methylglutaryl CoA reductase in aged potato tubers infected with Phytophthora infestans. Plant Cell Physiol. 37, 81–90. [Google Scholar]
- Yoshioka, H., Sugie, K., Park, H.-J., Maeda, H., Tsuda, N., Kawakita, K., and Doke, N. (2001). Induction of plant gp91 phox homolog by fungal cell wall, arachidonic acid, and salicylic acid in potato. Mol. Plant-Microbe Interact. 14, 725–736. [DOI] [PubMed] [Google Scholar]
- Yoshioka, H., Yamada, N., and Doke, N. (1999). cDNA cloning of sesquiterpene cyclase and squalene synthase, and expression of the genes in potato tuber infected with Phytophthora infestans. Plant Cell Physiol. 40, 993–998. [DOI] [PubMed] [Google Scholar]
- Yu, L., Quinn, M.T., Cross, A.R., and Dinauer, M.C. (1998). gp91phox is the heme binding subunit of the superoxide-generating NADPH oxidase. Proc. Natl. Acad. Sci. USA 95, 7993–7998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang, S., Du, H., and Klessig, D.F. (1998). Activation of the tobacco SIP kinase by both a cell wall-derived carbohydrate elicitor and purified proteinaceous elicitins from Phytophthora spp. Plant Cell 10, 435–449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou, F., Zhang, Z., Gregersen, P.L., Mikkelsen, J.D., de Neergaard, E., Collinge, D.B., and Thordal-Christensen, H. (1998). Molecular characterization of the oxalate oxidase involved in the response of barley to the powdery mildew fungus. Plant Physiol. 117, 33–41. [DOI] [PMC free article] [PubMed] [Google Scholar]