Abstract
The mechanism regulating the pattern of leaf initiation was analyzed by using shoot organization (sho) mutants derived from three loci (SHO1, SHO2, and SHO3). In the early vegetative phase, sho mutants show an increased rate of leaf production with random phyllotaxy. The resulting leaves are malformed, threadlike, or short and narrow. Their shoot apical meristems are relatively low and wide, that is, flat shaped, although their shape and size are highly variable among plants of the same genotype. Statistical analysis reveals that the shape of the shoot meristem rather than its size is closely correlated with the variations of plastochron and phyllotaxy. Rapid and random leaf production in sho mutants is correlated with the frequent and disorganized cell divisions in the shoot meristem and with a reduction of expression domain of a rice homeobox gene, OSH1. These changes in the organization and behavior of the shoot apical meristems suggest that sho mutants have fewer indeterminate cells and more determinate cells than wild type, with many cells acting as leaf founder cells. Thus, the SHO genes have an important role in maintaining the proper organization of the shoot apical meristem, which is essential for the normal initiation pattern of leaf primordia.
INTRODUCTION
In higher plants, the shoot apical meristem (SAM) is the source of all the above-ground organs. Accordingly, key processes that elaborate shoot architecture are localized in the SAM. An aggregate of small cells located in the distal portion of the shoot, the SAM supplies cells that divide and differentiate to form the elements of the shoot (Medford, 1992). The initiation of lateral organs is related to the structure of the SAM, including its so-called zonation, which is based on anatomical features and cell division patterns. The central zone, in which cell division is less frequent, is located in the center of the SAM and acts as a pool of undifferentiated, indeterminate cells. In the peripheral zone flanking the SAM, cells divide more frequently and are incorporated into leaf primordia. The proximal region, called the rib zone, supplies the cells that form the body of the stem. At the same time, the SAM can be viewed in terms of clonally distinct cell layers (L1, L2, and L3). The outermost layer, L1, comprises epidermal cells that divide only anticlinally. Cells in the inner L2 and L3 layers divide both anticlinally and periclinally and give rise to the inner tissues of the leaves and stem (Clark, 1997; Kerstetter and Hake, 1997; Meyerowitz, 1997).
Genetic and molecular studies of several genes associated with the SAM—such as knotted1 (kn1) (Sinha et al., 1993), SHOOTMERISTEMLESS (STM) (Barton and Poethig, 1993; Long et al., 1996), NO APICAL MERISTEM (NAM) (Souer et al., 1996), CLAVATA (CLV) (Clark et al., 1993, 1997), WUSCHEL (WUS) (Laux et al., 1996; Mayer-Klaus et al., 1998), and ZWILLE/PINHEAD (Moussian et al., 1998; Lynn et al., 1999)—have helped us to understand the formation and maintenance of the SAM. Analysis of these mutants indicates that the number and initiation pattern of lateral organs are closely associated with the size of the SAM (Callos et al., 1994; Jackson and Hake, 1999). Enlarged clv1 meristems are correlated with abnormal phyllotaxy and an increased number of floral organs (Clark et al., 1993). Analogous mutations of rice, fon1 and fon2, also enlarge the floral meristem and increase the number of floral organs (Nagasawa et al., 1996). In contrast, wus and a weak allele of stm reduce the meristem size and the number of floral organs in Arabidopsis (Clark et al., 1996; Laux et al., 1996). Those studies strongly suggest an important correlation between the number of lateral organs and SAM size. However, another regulatory process involved in lateral organ production is suggested in Arabidopsis. The perianthia (pan) mutation increases the number of floral organs without affecting the size and shape of the meristem (Running and Meyerowitz, 1996). The PAN gene encodes a basic domain/leucine zipper transcription factor and regulates the number of organs by controlling the distance between the two primordia (Chuang et al., 1999). Thus, although a pathway regulates the pattern of lateral organ initiation independently of meristem size and shape, SAM size or shape (or both) appears to play an important role in the pattern of lateral organ initiation.
Throughout the vegetative phase, the SAM continues to produce leaf primordia in a regular order (phyllotaxy) on its flanks. Many studies have focused on elucidating the mechanism that regulates phyllotaxy (reviewed in Steeves and Sussex, 1989; Callos and Medford, 1994). Based on the results of surgical experiments, a chemical/physiological field model was proposed (Snow and Snow, 1931; Wardlaw, 1949), according to which a diffusible substance emanating from the center of the SAM and the preexisting leaf primordia inhibited the initiation of new leaf primordium. Alternately, a biophysical model has suggested that the phyllotactic pattern is generated spontaneously as a result of physical force in the SAM (Selker et al., 1992; Green, 1994; Green et al., 1996). This model is supported by studies in which the application of expansin, which promotes cell wall extensibility, modified phyllotaxy in tomato (Fleming et al., 1997, 1999). Recently, the maize abphyl1 mutation was reported to change from distichous to decussate phyllotaxy, accompanied by an enlarged SAM (Jackson and Hake, 1999). Despite recent progress, however, little is known about how phyllotaxy is regulated.
Another important aspect associated with leaf primordia is the rate at which they are formed, which can markedly influence shoot architecture. To date, only a few mutants have been identified that specifically affect the time span between the initiations of two successive leaf primordia (the plastochron) but not the phyllotaxy. The rice pla1 mutant is one example: The leaf production rate is doubled, but the phyllotaxy remains distichous (Itoh et al., 1998). The SAM of pla1 is enlarged but its shape is not altered, suggesting that a change in the size of the SAM affects only the rate of leaf production, if no change in shape is also present. Several known mutants affect the plastochron and phyllotaxy simultaneously. In maize, the terminal ear1 (te1) mutant shows an increased rate of leaf production, aberrant phyllotaxy, and abnormal phytomers (Veit et al., 1998). The te1 gene contains several RNA binding protein motifs and appears to inhibit phytomer/leaf initiation in the SAM. The SAM of the te1 mutant has an abnormal geometry. Another Arabidopsis mutant, amp1, also was reported to have altered plastochron and phyllotaxy, although the size and shape of the SAM were not described (Chaudhury et al., 1993). Previously, we reported on the rice shoot organization (sho) mutant, which shows random phyllotaxy, short plastochron, abnormal leaves, and a malformed SAM (Tamura et al., 1992). A better understanding of these mutants should clarify the relationships between plastochron, phyllotaxy, and SAM structure.
Another interesting problem involved in leaf initiation is how the domain of the leaf primordium is established. An early step in leaf initiation is the recruitment of leaf founder cells. In grasses such as rice and maize, leaf founder cells occupy a ringlike domain on the periphery of the SAM and eventually divide rapidly to form the leaf primordium. Recently, molecular evidence distinguishing leaf founder cells from other cells in the SAM has been reported. The maize kn1 gene is expressed in the SAM but is downregulated in P0 leaf founder cells and leaf primordia (Smith et al., 1992; Jackson et al., 1994). Analyses of kn1 and related genes indicate that the most important function of kn1 is maintaining the SAM by regulating cell fate (Kerstetter et al., 1997). Studies with kn1 as a molecular marker of determinate/indeterminate cells suggest that the narrow sheath (ns) mutant appears to lack leaf marginal domains and has fewer cells that do not express kn1 (Scanlon et al., 1996; Scanlon and Freeling, 1997). A similar situation is observed in the narrow or bladeless leaves of rough sheath2 (Schneeberger et al., 1998) and leaf bladeless1 mutants (Timmermans-Marja et al., 1998). Thus, modification of SAM organization, especially in the balance between indeterminate and determinate cells, affects leaf morphogenesis.
In this report, we describe six mutants of SHO genes in rice. They all exhibit similar phenotypes, including random phyllotaxy, short plastochron, aberrant leaves, and malformed SAMs. Using these mutants, we show how SAM structure is related to the pattern of leaf initiation.
RESULTS
We used six recessive sho mutations covering three loci (SHO1, SHO2, and SHO3): four sho1 alleles (sho1-1 to sho1-4), one sho2 allele, and one sho3 allele. SHO1 is mapped on the long arm of chromosome 4, but the map positions of the other loci are unknown. The six mutants showed very similar phenotypes in embryos, seedlings, and spikelets.
Phenotypes of sho Mutants in the Life Cycle
Embryo Development
Four days after pollination (DAP), a coleoptilar primordium appears on the ventral side of the wild-type embryo, and a SAM is just below this. At 5 DAP, the first leaf primordium is formed on the flank of the SAM opposite the coleoptilar insertion (Figure 1A). The SAM then produces the second leaf primordium at 6 to 7 DAP and the third leaf primordium at 8 to 9 DAP. The SAM in a mature embryo has a domelike appearance (Figures 1B and 1C).
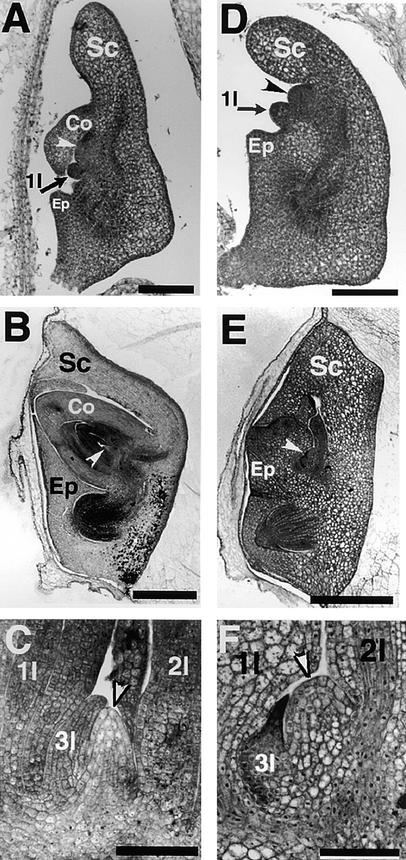
Figure 1.
Developmental Course of a sho3 Embryo.
(A) Wild-type embryo at 5 DAP differentiating scutellum, coleoptile, epiblast, and SAM (white arrowhead) with first leaf primordia (1l).
(B) Mature wild-type embryo. SAM is indicated by the white arrowhead.
(C) Shoot apex of a mature wild-type embryo with three leaf primordia (1l, 2l, and 3l) and the domelike SAM (white arrowhead).
(D) sho3 embryo at 6 DAP differentiating scutellum, epiblast, and SAM (arrowhead) with first leaf primordia (1l) but no coleoptile.
(E) Mature sho3 embryo with scutellum and abnormal shoot. SAM is indicated by the white arrowhead.
(F) Shoot apex of a sho3 embryo with three abnormal leaf primordia (1l, 2l, and 3l) and flat SAM (white arrowhead).
Co, coleoptile; Ep, epiblast; Sc, scutellum.  ;
;  .
.
The developmental course was similar in sho1, sho2, and sho3 embryos, which were indistinguishable from the wild type through 4 DAP. Early embryogenesis of sho was thus believed to be the same as that of the wild type before morphogenetic events occur. By 6 DAP, however, differences between sho and wild-type embryos were apparent, the sho embryos having a retarded initiation of the SAM and no coleoptile formation (Figure 1D). The first leaf primordium was initiated from the proximal side of the SAM, as happens in the wild type, and the mature embryo of sho also had a normal radicle, scutellum, and epiblast; however, three leaf primordia were abnormal (Figure 1E). The first leaf primordium was greatly enlarged, and its epidermal cells were palisade shaped, looking like scutellar epithelium cells. The sho SAM was broader than that of the wild type (Figure 1F). The phyllotaxy of the first three leaves in sho embryos was distichous.
Seedling Development
After germination, plants homozygous for these mutations developed unusual seedlings that exhibited various shoot organization abnormalities. A previous study had revealed that the early SAM of sho1-1 was flat, not domelike, and that the leaf primordia were formed rapidly at various positions around the SAM, resulting in a random phyllotaxy and short plastochron (Tamura et al., 1992). We analyzed the sho1, sho2, and sho3 mutants in more detail. All the sho mutants displayed similar phenotypes in 1-week-old seedlings, forming many malformed leaves (Figure 2). Most of the leaves developed into threadlike structures and did not encircle the SAM (Figure 2). The other leaves were also malformed, narrow, and short. The shape of the SAM was strongly modified. In many cases, the SAM was flat, although its shape was highly variable, even in the same genotype (Figure 2). Scanning electron microscopy revealed that the phyllotaxy was obviously irregular in all mutants (Figures 3A to 3D). Each primordium was not always positioned opposite the previous one. In some cases, two primordia of similar size were initiated simultaneously.
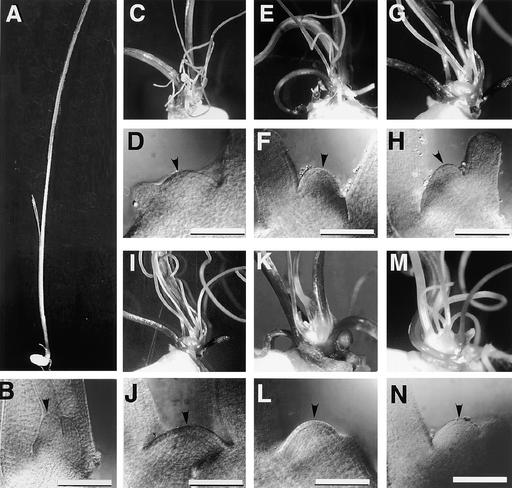
Figure 2.
Seedlings and SAMs of Wild-Type Plants and sho Mutants at 1 Week after Germination.
Each pair shows seedling and SAM, respectively. The SAM is indicated by arrowheads. In sho seedlings, many threadlike leaves are produced, and the SAMs are flat.
(A) and (B) Wild-type.
(C) and (D) sho1-1.
(E) and (F) sho1-2.
(G) and (H) sho1-3.
(I) and (J) sho1-4.
(K) and (L) sho2.
(M) and (N) sho3.
 .
.
Figure 3.
Scanning Electron Microscopy of Shoot Apices and Leaves in sho Seedlings.
(A) Side view of a wild-type shoot apex, showing foodlike P2 leaf primordium (arrow) and P1 primordium (*) in alternate phyllotaxy.
(B) to (D) Top view of sho1-1 (B), sho2 (C), and sho3 (C) seedlings showing malformed leaf primordia and irregular phyllotaxy. In several leaf primordia, a pair of small bulges (*) are observed on both sides of the threadlike structure. SAMs are indicated by arrowheads and leaf primordia by arrows.
(E) Threadlike leaf with a small ligule (arrow) on the adaxial side.
(F) Adaxial surface of a wild-type leaf blade with stomatas (arrowheads) and hairs (arrows).
(G) Adaxial surface of a threadlike leaf with stomatas (arrowheads) and hairs (arrows).
(H) Trifurcated leaf in a 3-week-old seedling of sho2. In the apical region, the leaf blade is separated into a central region and two lateral regions.
 ;
;  .
.
The threadlike leaves had a ligule on the adaxial side (Figure 3E) and a normal surface structure (Figures 3F and 3G). In addition, their vascular tissues showed normal dorsoventrality. Thus, the threadlike leaves were not caused by the loss of dorsoventrality. At the base of many threadlike leaf primordia, a pair of small bulges was observed (Figure 3C), but these did not grow further. We presume that the threadlike leaves resulted from the growth of the central region with suppression of the growth of flanking regions. In other words, most of the leaf domains may have been recruited as leaf founder cells in sho mutants, but only the central domain was allowed to grow.
Two weeks after germination, relatively long leaves began to develop. The seedlings became necrotic under field conditions, probably because the sho SAM was not covered with young leaf primordia. The seedlings were rescued when grown in aseptic culture boxes, and sho plants survived to produce nearly normal shoots after 1 month of growth. In the transition to becoming normal plants, however, some leaves had defects in central marginal patterning; that is, there was bi- or trifurcation in the apical region (Figure 3H). The mature plants appeared normal, except that they were dwarf sized and produced many tillers (Figure 4). The sho2 plants showed less severe dwarfism than the other mutants. In the late vegetative phase, the phyllotaxy of sho was also changed from random to distichous, and the SAM became dome shaped, as in the wild type. Thus, sho abnormalities were detected mainly in the early vegetative phase, and normal plants were recovered during the late phase.
Figure 4.
Mature Plants.
(A) Mature wild-type plants.
(B) Mature sho1-2 plants with apparently normal but dwarf shoot.
(C) Mature sho2 plants with apparently normal but dwarf shoot.
In the course of the allelism tests, double mutants (sho1 sho2, sho1 sho3, and sho2 sho3) must have segregated in the F2 population. However, we could not identify the double mutants on the basis of the embryo and seedling phenotypes because all of the mutant embryos and seedlings of F2 seeds were very similar,which suggests that the double mutants were phenotypically indistinguishable from the single mutants.
Inflorescence and Spikelet Development
Four mutants (sho1-1, sho1-2, sho1-3, and sho2) survived to the reproductive phase. We have not confirmed whether the other two mutants (sho1-4 and sho3) are able to reproduce because only a few plants have been used for long-term cultivation. The flowering time of sho1-1, sho1-2, sho1-3, and sho2 was normal, but the spikelets were obviously abnormal. The most remarkable feature of the sho spikelets was an awnlike elongation of the central region of the glumes (empty glumes and lemma) and degenerated palea (Figure 5B) compared with the wild type (Figure 5A). Even in a less severe case, in which lemma and palea grew relatively normally, an awn was elongated from the lemma (Figure 5C). The morphology of the floral organs, lodicule, stamens, and pistils was apparently normal but with slight variability. The phyllotaxy of spikelet and floral organs was normal.
Figure 5.
Spikelets of Wild-Type Plants and sho2 Mutants.
(A) Wild-type spikelet.
(B) sho2 spikelet with awnlike lemma and degenerated palea.
(C) Relatively normal sho2 spikelet with an elongated awn (arrowhead).
L, lemma; P, palea.
Altered SAM Structure of sho Mutants Is Correlated with Changes in Leaf Initiation
SAM Structure of sho Mutants in the Early Vegetative Phase
To understand the SAM structure of sho plants in detail in the early vegetative phase, we observed the cleared SAMs of sho seedlings and measured four variables (height, width, height-to-width ratio, and volume) that described the SAM structure (Table 1). The width of the SAM was measured just above the youngest leaf primordial (P1) insertion, and the height was the maximum distance perpendicular to this width. The volume of the SAM was approximated by a paraboloid. The mean SAM height of sho mutants was approximately two-thirds that of the wild type, and the width was larger in many seedlings. Consequently, the height-to-width ratio was smaller than in the wild type, indicating that the sho SAMs were relatively flat. Because of their increased width, the SAM volume in sho mutants was large. In addition, each evaluated characteristic of SAM structure varied widely among sho plants of the same genotype, judging from the large standard deviations shown in Table 1. These results indicate that sho SAMs are generally flat, although the shape fluctuates greatly among individuals.
Table 1.
Measurement of Traits Associated with SAM and Leaf Initiation in sho Mutants (±sd)a
| Trait | Wild Type | sho1-1 | sho1-2 | sho1-3 | sho1-4 | sho2 | sho3 |
|---|---|---|---|---|---|---|---|
| SAM width (μm) | 48.0 ± 4.8 | 68.7 ± 20.2 | 51.5 ± 5.5 | 78.2 ± 28.6 | 85.6 ± 19.4 | 66.3 ± 13.2 | 81.8 ± 18.3 |
| SAM height (μm) | 36.8 ± 4.1 | 23.3 ± 7.0 | 24.3 ± 4.7 | 26.8 ± 8.6 | 20.0 ± 5.8 | 24.5 ± 4.0 | 23.7 ± 6.6 |
| Height/width of SAM | 0.77 ± 0.02 | 0.36 ± 0.13 | 0.47 ± 0.06 | 0.37 ± 0.17 | 0.25 ± 0.12 | 0.38 ± 0.11 | 0.29 ± 0.08 |
| SAM volume (μm3) | 10,895 ± 3385 | 14,512 ± 7872 | 8397 ± 3065 | 22,290 ± 12,929 | 17,480 ± 4791 | 13,523 ± 4776 | 21,886 ± 13,919 |
| Plastochron (day) | 3.54 ± 0.04 | 1.12 ± 0.16 | 1.29 ± 0.36 | 2.33 ± 0.83 | 0.86 ± 0.25 | 0.82 ± 0.15 | 1.06 ± 0.28 |
| Divergence angle (°) | 180.0 ± 0.0 | 109.4 ± 25.0 | 127.6 ± 16.3 | 126.7 ± 20.6 | 113.2 ± 10.7 | 114.4 ± 14.6 | 102.5 ± 21.8 |
| Entropy of phyllotaxy | 0.00 ± 0.00 | 1.03 ± 0.17 | 0.645 ± 0.22 | 0.99 ± 0.21 | 0.94 ± 0.35 | 0.93 ± 0.17 | 0.95 ± 0.19 |
Five seedlings of each genotype were measured.
Leaf Initiation Pattern of sho in the Early Vegetative Phase
Next, we measured three variables (plastochron, divergence angle, and entropy of phyllotaxy) that represent the initiation pattern of leaf primordia (Table 1). In this study, the plastochron was calculated from the number of leaf primordia produced in the week after germination. The wild-type seedlings had a mean plastochron of ∼3.5 days. The plastochron for all the mutants was ∼1 day, much shorter than that of the wild type.
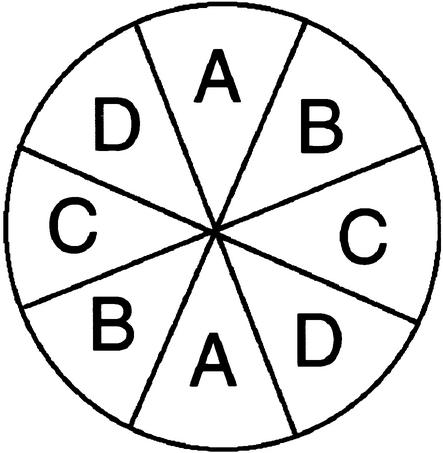
We examined the spatial distribution of leaf primordia around the shoot apex. In contrast to the distichous phyllotaxy of the wild type, which has a divergence angle of 180°, leaf primordia of sho seedlings were produced at nearly random positions around the apex (Figure 6). The mean divergence angle in sho seedlings was ∼110°. To analyze statistically the relationship between phyllotactic irregularity and SAM structure, we evaluated quantitatively the randomness of phyllotaxy. Using the scatter diagram of leaf positions shown in Figure 6, we counted the number of leaf primordia in each of the four diagonally duplicated regions of the SAM (A, B, C, and D in Figure 7) and defined the entropy of phyllotaxy as in Figure 7. In this case, completely random phyllotaxy would give a maximum value of 1.34. The entropy of phyllotaxy in each mutant was ∼1, in contrast to 0 for the wild type. These data indicate that leaf primordial production in the sho SAM was quite disordered, both temporally and spatially.
Figure 6.
Schematic Representation of Phyllotactic Pattern in sho Mutants.
Positions of the first to 10th leaf primordia are indicated by colored circles on concentric circles; the outermost circle corresponds to the first leaf, the innermost circle to the 10th leaf, and the center to the SAM. Different colors indicate different seedlings. In the wild type, leaves are formed in distichous phyllotaxis, as indicated by black circles. The positions of the first through third leaves in sho mutants are almost normal and are indicated by black circles.
Figure 7.
Definition of Entropy of Phyllotaxy.
A top view of a shoot apex is shown. It was divided into four diagonally duplicated regions (A to D), and the entropy of phyllotaxy (H) was calculated from the number of leaf primordia in each region:
H=−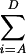 Pi 1n Pi.
Pi 1n Pi.
Pi is the probability of leaf primordium formed in region i and is expressed by the number of leaf primordia in region i divided by the total number of leaf primordia produced (n).
Correlation between SAM Structure and Leaf Initiation Pattern
These results suggest that the malformed SAM causes the abnormal pattern of leaf primordial initiation in sho. To confirm this statistically, we performed a correlation analysis between the variables (height, width, height-to-width ratio, and volume) of SAM geometry and those (plastochron, divergence angle, and the entropy of phyllotaxy) of the initiation pattern of leaf primordia (Table 2). This analysis also used three similar mutants (odm 121, odm 200, and odm 201; see Methods) in addition to the six sho mutants. To extract a relationship between SAM geometry and leaf initiation pattern, we performed a correlation analysis by using the data for 50 seedlings (five seedlings of each mutant and the wild type). Noting that the variance of each character within each mutant dominated largely over that among different mutants (genotypes) (Table 1), we treated each individual as a random sample from a population. There was no significant correlation (P > 0.05) between SAM volume and any variable of leaf primordia. This suggests that SAM volume does not affect the leaf initiation pattern. The other three variables (height, width, and height-to-width ratio) showed significant correlations (P < 0.01) with all or most characters of the leaf initiation pattern. The SAM height and height-to-width ratio were negatively correlated with the divergence angle and entropy and were positively correlated with the plastochron. SAM width was positively correlated with entropy and was negatively correlated with the divergence angle. However, the correlation coefficients between SAM width and leaf primordial variables were not very significant. The height-to-width ratio showed the greatest correlation with any leaf primordial parameter. Thus, the shape of the SAM strongly affected the spatial and temporal pattern of leaf initiation: a flatter SAM tended to produce leaf primordia more randomly and more rapidly. Similar results were obtained when only the 45 mutant seedlings were analyzed, although the correlations among several of the variables were not significant (data not shown). These results suggest that SAM shape, rather than size, primarily affects the pattern of leaf initiation.
Table 2.
Correlation Coefficientsa between SAM Parameters and Leaf Primordial Parameters in sho Mutants
| Parameter | Plastochron | Divergence Angle | Entropy of Phyllotaxy |
|---|---|---|---|
| SAM width | −0.228 | −0.425b | 0.400b |
| SAM height | 0.538b | 0.477b | −0.568b |
| Height/width | 0.617b | 0.670b | −0.733b |
| SAM volume | 0.000 | −0.197 | 0.141 |
Calculated for the data of 50 seedlings of nine mutants and the wild type. The data for 50 seedlings are treated as samples from a population.
Significant at 1% level.
In addition, the three variables of leaf primordial initiation were significantly correlated with each other (P < 0.01). Therefore, these characteristics are not regulated independently in sho mutants.
Cell Division Activity in the SAM of sho Mutants
The rapid leaf initiation in sho should be associated with high cell division activity in the SAM. Therefore, we conducted an in situ hybridization experiment using as a probe the rice histone H4 gene, which is expressed specifically in S-phase of the cell cycle. In wild-type seedlings at 1 week after germination, hybridization signals were observed in no more than two cells of the peripheral zone in many SAMs in the median longitudinal plane (Figure 8A). In the three sho mutants, however, cells with hybridization signals were more common than in the wild type (Figures 8B to 8D), indicating that sho mutations accelerated cell divisions in the SAM. In addition, cells expressing histone H4 were not restricted to the peripheral zone (in which cell division activity was relatively high in the wild-type SAM) but were also frequently detected in the central region (Figures 8B to 8D). These results indicate that the SAMs of sho mutants are organized differently from the normal SAM and that the rapid, random leaf initiation in sho mutants reflects frequent, disorganized cell divisions in the SAM.
Figure 8.
Histone H4 Expression in Wild-Type and sho Meristems at 1 Week after Germination.
Hybridization was conducted with the histone H4 antisense RNA probe.
(A) Wild type. No signal is visible in the SAM.
(B) sho1-1.
(C) sho2.
(D) sho3.
In sho mutants, signals in the SAM are observed in several cells, including those in the central zone. SAM is indicated by arrowheads.  .
.
Expression Pattern of Homeobox Gene OSH1 in sho Mutants
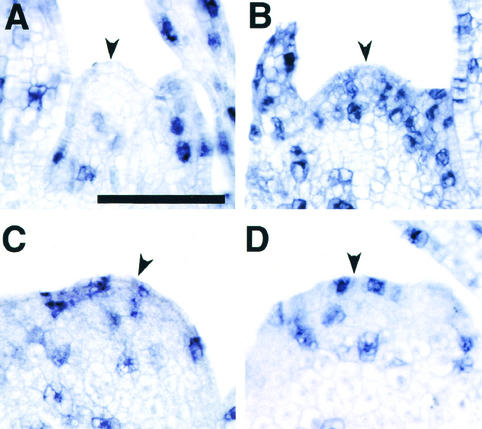
Random initiation of leaf primordia means that many cells in the sho SAM are competent to develop as leaves, or at least are not indeterminate. In the wild-type SAM, however, most cells are indeterminate; a few cells in a specific region on the flank are capable of developing into leaf primordium, as evidenced by the expression pattern of kn1 (Smith et al., 1992; Jackson et al., 1994) and its rice ortholog (Matsuoka et al., 1993). Because we expected that the distribution of indeterminate versus determinate cells in sho SAMs would differ from that in the wild-type SAM, we performed in situ hybridization of the sho SAMs, using OSH1 as a probe. OSH1, a rice ortholog of maize kn1, is expressed in the SAM but is downregulated in the P0 leaf primordium (Matsuoka et al., 1993; Sato et al., 1996) (Figures 9A to 9C). Thus, transcripts of OSH1 accumulate in indeterminate cells and play a role in maintaining the indeterminate state of the SAM.
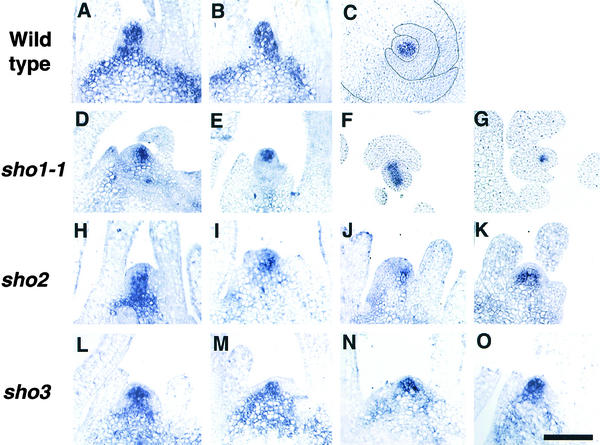
Figure 9.
OSH1 Expression in Wild-Type and sho Meristems at 1 Week after Germination.
The OSH1 antisense RNA probe was used for hybridization.
(A) to (C) Wild type.
(D) to (G) sho1-1. Four different shoot apices are shown.
(H) to (K) sho2. Four different shoot apices are shown.
(L) to (O) sho3. Four different shoot apices are shown.
(C), (F), and (G) show cross-sections; the other panels show median longitudinal sections. In (C), the outline of each leaf primordium is traced.  .
.
One week after germination, the expression pattern of OSH1 in the SAM of sho1, sho2, and sho3 seedlings was greatly modified. In many cases, the expression domain was decreased despite the large SAM size (Figure 9). In extreme cases, the expression of OSH1 was restricted to only a few cells in the median longitudinal section (Figures 9D and 9J) and in the cross-section (Figure 9G). This restricted pattern suggests that the sho SAM consists of a few indeterminate cells and a larger number of determinate cells and that many of the latter are competent to acquire leaf identity. Another interesting feature is that the expression domain of OSH1 in the SAM was not constant but fluctuated individually with each mutant (Figure 9). In many cases, the expression was biased to either side of the SAM (Figures 9D, 9I, 9J, and 9M). In some cases, OSH1 expression was downregulated on both sides of the SAM (Figures 9E, 9F, and 9L). Together with the accelerated cell division, this aberrant, unstable organization of the sho SAM causes random, rapid leaf production. These results are consistent with the variable SAM shape and random phyllotaxy in sho mutants. By 1 month after germination, this unusual pattern of OSH1 expression disappeared and a normal expression pattern was recovered, concomitant with the acquisition of normal shoot architecture (data not shown).
In conclusion, SHO genes appear to be essential for the normal organization and behavior of the SAM, especially with regard to indeterminate versus determinate cell fate, and consequently affect the leaf initiation pattern.
DISCUSSION
Because sho mutants show so many severe and unique phenotypes, their analysis should provide new insight into the regulation of shoot development. Both the pleiotropic and stage-specific functions of SHO genes suggest an important role of SHO genes in regulating shoot development and the temporal differentiation of the developmental program. The significant correlation between SAM geometry and the pattern of leaf initiation shows the important role of SAM geometry in leaf primordial development. The unusual expression pattern of histone H4 and OSH1 in sho SAMs indicates an aberrant organization of indeterminate versus determinate cells in sho SAMs, which results in modification of the leaf initiation pattern.
Requirement for SHO Function during the Life Cycle
The earliest abnormal phenotype of sho embryos is the failure of coleoptile initiation. An absence of the coleoptile is also observed in shootless (shl) mutants, in which the SAM is completely lost (Satoh et al., 1999), suggesting that coleoptile initiation depends on the presence of the SAM. Thus, the coleoptile fails to initiate in sho embryos because the SAM is defective. On the other hand, the epiblast, a small, flaplike outgrowth (the function of which is not clear) in the proximal region of embryos in some grass species (Figures 1A and 1B), forms normally in sho embryos. Both the coleoptile and epiblast are lost in shl embryos (Satoh et al., 1999). Therefore, initiation of the coleoptile may require complete SAM activity, whereas partial activity is sufficient to initiate the epiblast.
In embryogenesis and the early vegetative phase, the sho SAM was flatter than that of the wild type. Therefore, sho genes may regulate meristem structure in both embryogenesis and the early vegetative phase. Although the lack of a coleoptile in the embryo might be attributed to abnormalities of the SAM and seedlings, such is not the case, because adventitious shoots regenerated from sho calli showed the same phenotype as the seedlings (data not shown). In the very early vegetative phase, sho plants show various abnormalities of shoot architecture and have an enlarged SAM. In contrast, in the late vegetative phase, sho SAMs become normally shaped and produce leaves with distichous phyllotaxy. These phenomena are reminiscent of the maize abphl1 mutant (Jackson and Hake, 1999), in which an enlarged SAM is associated with an abnormal decussate phyllotaxy. In abphl1 plants, spontaneous reversion to distichous phyllotaxy occurs often and is accompanied by a decrease in the SAM to normal size. We also observed a close relationship between phyllotaxy and SAM structure in sho mutants. Accordingly, the SAM appears to be of primary importance in specifying phyllotaxy.
In the reproductive phase, abnormal spikelets were observed. The most conspicuous phenotype was awnlike glumes, which would be equivalent to the threadlike leaves in the seedlings. Because that abnormality was not detected in the phyllotaxy of reproductive organs, SHO genes may be more involved in the seedling phase than in the reproductive phase.
The sho mutants show pleiotrophic phenotypes during embryogenesis, the early vegetative phase, and the reproductive phase but not in the late vegetative phase. Thus, SHO genes function in a phase-specific manner and are required for maintaining SAM structure and the proper development of lateral organs in these phases.
Modification of SAM Structure Is Accompanied by Altered Leaf Initiation
In this study, we found two characteristic SAM phenotypes in sho mutants. First, the SAM of sho is flat and large compared with that of the wild type. Second, the SAM structure of sho varies widely with individual plants.
In sho mutants, the malformed SAM structure is closely associated with modification of spatial and temporal patterns of leaf initiation (phyllotaxy and plastochron). Furthermore, for regulating leaf initiation, the shape of the SAM may be more important than its size. To date, several mutations that affect SAM size or shape (or both) and phyllotaxy have been reported. For example, clv mutants of Arabidopsis have an enlarged SAM and produce leaf primordia in disrupted phyllotaxy in the vegetative phase (Leyser and Furner, 1992; Clark et al., 1993; Bowman, 1994). The maize abphl1 mutant also has an enlarged SAM with decussate phyllotaxy (Jackson and Hake, 1999). In both cases, the shape of the SAM is abnormal: flat in abphl1 and bell shaped in clv1. Therefore, these phyllotactic changes may be related to the modification of SAM shape rather than size. In pla1 mutants of rice, leaf primordia initiate rapidly without affecting phyllotaxy. The pla1 plants have an enlarged SAM, but the shape of the SAM is not altered (Itoh et al., 1998). Therefore, the change in SAM size, if not accompanied by shape modification, may affect only the production rate of leaf primordia.
In this analysis, although the SAM shape was also correlated with the plastochron, the correlation between the characteristics of SAM structure and phyllotaxy was greater than that between the characteristics of SAM structure and the plastochron (Table 2). These results suggest that the SAM structure affects the phyllotaxy more than it does the plastochron. However, we find it difficult to regard determination of the phyllotaxy and plastochron as two separable events, given the significant correlation (P < 0.01) between the phyllotactic variables and plastochron. This correlation may be a result of the specific organization of the sho SAM, because two closely positioned primordia were frequently observed to initiate simultaneously in sho mutants.
Another point is the wide variation of SAM structure among plants of each genotype. In contrast to sho, the shape and size of wild-type SAMs are uniform, as indicated by the small variation of SAM characteristics (Table 1). The variable SAM shape and the unstable expression domain of OSH1 in each mutant are evidence that the rigid regulation of SAM structure is severely disturbed in sho. These results also suggest that the SAM structure fluctuates widely during the early ontogeny of each sho plant. Although regular fluctuation of SAM structure during a plastochron has been reported (Hara, 1997), this is not the case for sho, in which the fluctuation is wide and irregular. The variability of SAM shape and the OSH1 expression domain implies the developmental instability of sho SAMs.
Possible Role of SHO Genes in the SAM of the Early Vegetative Phase
In this study, mutants of three SHO loci exhibited common phenotypes in shoot architecture: an abnormal meristem, random phyllotaxy, short plastochron, and threadlike leaves. These three loci play similar roles in shoot development and may be involved in the same cascade.
The sho genes may have functions relating to two regulatory processes: the maintenance of proper SAM organization and the regulation of leaf morphology. Two successive primordia are observed to initiate closely, suggesting a large available space for leaf initiation in the sho SAMs. This hypothesis accords well with the expression pattern of the homeobox gene OSH1. The reduction of the OSH1 expression domain indicates that indeterminate cells occupy a small portion of sho SAM, most cells being competent as leaf founder cells. Thus, the SHO genes limit the region for leaf founder cells. In the absence of SHO function, more founder cells are located in the SAM. Consequently, leaves initiate from many positions. The increased number of cells expressing histone H4 in sho SAM also indicates that the region in which cells divide rapidly is enlarged, leading to the random and rapid production of leaf primordia in the sho SAM.
Several mutations involved in leaf initiation have been reported. The te1 mutation is similar to sho in several aspects. The te1 mutants exhibit aberrant phyllotaxy and rapid leaf production (Veit et al., 1998). In contrast to sho, however, the mutant phenotype of te1 is observed throughout the life cycle. In addition, abnormalities in te1 leaves differ from those of sho leaves. General phenotypes of sho may be more similar to that of leafbladeless1 (lbl1) of maize than to te1. Mutations of lbl1 modify the dorsoventrality of lateral organs and produce radially symmetrical, threadlike leaves resembling sho leaves. The lateral propagation of leaf founder cell recruitment in the mutant SAM is affected, and the number of leaf founder cells is strongly decreased; however, its phyllotaxy remains distichous (Timmermans-Marja et al., 1998). Another mutant, rough sheath2 (rs2) in maize, also has narrower or semibladeless leaves. rs2 encodes a Myb protein that functions as a transcriptional regulator; rs2 also negatively regulates knox homeobox genes, reducing the number of leaf founder cells in the mutant SAM (Timmermans-Marja et al., 1999; Tsiantis et al., 1999). The decrease in the number of leaf founder cells in the SAM gives rise to narrower or bladeless leaves. In contrast to these two maize mutants, threadlike leaves are formed in sho despite the extended area in which OSH1 is downregulated. This implies that the downregulation of knox genes in the SAM is not necessarily reflected in the size of leaf primordium. The leaf morphology in sho may be regulated independently of the leaf initiation pattern, because threadlike leaves with normal phyllotaxy are also formed at the transition stage from early to late vegetative phase. Furthermore, the threadlike or narrow leaves of sho are caused by a defect in the growth of one or two leaf domains (J.I. Itoh and Y. Nagato, unpublished data). Thus, several functionally different genes are involved in the maintenance of proper SAM organization.
In summary, SHO genes regulate the spatial and temporal pattern of leaf primordia initiation by maintaining the proper SAM organization during the early vegetative phase.
METHODS
Plant Materials and Measurements
We used nine single recessive mutants of rice (Oryza sativa) that showed abnormal shoot architecture. They were identified among the M2 population of O. sativa cv Taichung 65 mutagenized with the chemical mutagen N-methyl-N-nitrosourea. Six mutations cover three loci: SHOOT ORGANIZATION1 (SHO1) with four alleles, sho1-1 through sho1-4; SHO2 with one allele, sho2; and SHO3 with one allele, sho3. One allele, sho1-1 (formerly named sho), has already been partially analyzed (Tamura et al., 1992). sho2 was reported elsewhere (Hong et al., 1995) but has not been characterized. SHO1 is located in the middle of the long arm on chromosome 4, but the map positions of the other genes are not known. Four mutants—sho1-2, sho1-3, sho1-4, and sho3—were newly identified. In addition to these six mutants, three mutations for which allelism to these mutations was unknown were also used for statistical analysis; they were tentatively named odm 121, odm 200, and odm 201. All nine mutants are phenotypically very similar. Because the mutants set no seeds, the mutations were maintained in a heterozygous state.
Mutant and wild-type seeds were sterilized in 2% sodium hypochlorite and germinated on filter papers in Petri dishes at 30°C. For long-term observation, mutant seeds were grown aseptically on Murashige and Skoog (1962) medium supplemented with 3% sucrose and 1% agar at pH 5.8 in a plant box at 28°C. One or 2 months after inoculation, mutant plants were transplanted to pots.
One week after germination, the number and position of leaf primordia in each seedling were measured directly under a dissecting microscope. In mutant seedlings, because the shoot apices were not covered with young leaf primordia, we could specify the position and number of leaf primordia without sectioning. To measure the structure of the shoot apical meristems, we used the same seedlings as those used for specifying the positions of leaf primordia.
Clearing the Shoot Apex
Shoot apices were fixed in formalin:glacial acetic acid:50% ethanol (1:1:18 [v/v/v]) for ∼16 hr at 4°C and were then dehydrated in a graded ethanol series. After clearing them in the benzyl-benzoate-four-and-a-half fluid devised by Herr (1982), we observed the shoot apices through a microscope equipped with Nomarski differential interference-contrast optics (model MT-2; Olympus, Tokyo, Japan).
Scanning Electron Microscopy
Early shoot apices were fixed with 2.5% glutaraldehyde in 0.1 M sodium phosphate buffer, pH 7.2, for ∼16 hr at 4°C. After rinsing with 0.1 M sodium phosphate buffer, pH 7.2, they were postfixed in 1% osmium tetroxide for 3 hr at 4°C and rinsed again with the buffer. Then samples were dehydrated in a graded ethanol series, and 100% ethanol was replaced with 3-methylbutyl acetate. Samples were dried at their critical point, sputter-coated with platinum, and observed with a scanning electron microscope (model S-4000; Hitachi, Tokyo, Japan) at an accelerating voltage of 10 kV.
In Situ Hybridization
Shoot apices were fixed in 4% paraformaldehyde and 0.25% glutaraldehyde in 0.1 M sodium phosphate buffer for ∼20 hr at 4°C. They were then dehydrated in a graded ethanol series, replaced with xylem, and embedded in Paraplast Plus (Oxford Labware, St. Louis, MO). Microtome sections 8 μm thick were applied to glass slides coated with Vectabond (Vector Laboratories, Burlingame, CA). Digoxigenin-labeled antisense and sense probes were prepared from full-length histone H4 and OSH1 cDNAs of rice without the polyA region. In situ hybridization and immunological detection of the signals were performed by the methods of Kouchi and Hata (1993). Because no hybridization signals were detected when sense probes were used, only the results with antisense probes are described.
Acknowledgments
We thank Dr. Bruce Veit (Massey University) for critical reading of the manuscript. This work was supported in part by a Grant-in-Aid for Scientific Research on Priority Areas (No. 11163205) from the Ministry of Education, Science, and Culture of Japan.
References
- Barton, M.K., and Poethig, R.S. (1993). Formation of the shoot apical meristem in Arabidopsis thaliana: An analysis of development in the wild type and in the shoot meristemless mutant. Development 119, 823–831. [Google Scholar]
- Bowman, J.L. (1994). Arabidopsis, an Atlas of Morphology and Development. (New York: Springer-Verlag).
- Callos, J.D., and Medford, J.I. (1994). Organ position and pattern formation in the shoot apex. Plant J. 6, 1–7. [Google Scholar]
- Callos, J.D., Dirado, M., Xu, B., Behringer, F.J., Link, B.M., and Medford, J.I. (1994). The forever young gene encodes an oxidoreductase required for proper development of the Arabidopsis vegetative shoot apex. Plant J. 6, 835–847. [DOI] [PubMed] [Google Scholar]
- Chaudhury, A.M., Letham, S., Craig, S., and Dennis, E.S. (1993). amp1—A mutant with high cytokinin levels and altered embryonic pattern, faster vegetative growth, constitutive photomorphogenesis and precocious flowering. Plant J. 4, 907–916. [Google Scholar]
- Chuang, C.F., Running, M.P., Williams-Robert, W., and Meyerowitz, E.M. (1999). The PERIANTHA gene encodes a bZIP protein involved in the determination of floral organ number in Arabidopsis thaliana. Genes Dev. 13, 334–344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark, S.E. (1997). Organ formation at the vegetative shoot meristem. Plant Cell 9, 1067–1076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark, S.E., Running, M.P., and Meyerowitz, E.M. (1993). CALAVATA1, a regulator of meristem and flower development in Arabidopsis. Development 119, 397–418. [DOI] [PubMed] [Google Scholar]
- Clark, S.E., Jacobsen, S.E., Levin, J.Z., and Meyerowitz, E.M. (1996). The CLAVATA and SHOOT MERISTEMLESS loci competitively regulate meristem activity in Arabidopsis. Development 122, 1567–1575. [DOI] [PubMed] [Google Scholar]
- Clark, S.E., Williams, R.W., and Meyerowitz, E.M. (1997). The CLAVATA1 gene encodes a putative receptor kinase that controls shoot and floral meristem size in Arabidopsis. Cell 89, 575–585. [DOI] [PubMed] [Google Scholar]
- Fleming, A.J., McQueen-Mason, S., Mandel, T., and Kuhlemeier, C. (1997). Induction of leaf primordia by the cell wall protein expansin. Science 276, 1415–1418. [Google Scholar]
- Fleming, A.J., Caderas, D., Wehrli, E., McQueen, M.S., and Kuhlemeier, C. (1999). Analysis of expansin-induced morphogenesis on the apical meristem of tomato. Planta 208, 166–174. [Google Scholar]
- Green, P.B. (1994). Connecting gene and hormone action to form, pattern and organogenesis, biophysical transductions. J. Exp. Bot. 45, 1775–1788. [Google Scholar]
- Green, P.B., Steele, C.S., and Rennich, S.C. (1996). Phyllotactic patterns: A biophysical mechanism for their origin. Ann. Bot. 77, 515–527. [Google Scholar]
- Hara, N. (1997). Shoot development in Aucuba japonica. IV. The shoot apex at the late stage of the first growing season. Phytomorphology 47, 437–444. [Google Scholar]
- Herr, J.M., Jr. (1982). An analysis of methods for permanently mounting ovules cleared in four-and-a-half type clearing fluid. Stain Technol. 57, 161–169. [DOI] [PubMed] [Google Scholar]
- Hong, S.-K., Aoki, T., Kitano, H., Satoh, H., and Nagato, Y. (1995). Phenotypic diversity of 188 rice embryo mutants. Dev. Genet. 16, 298–310. [Google Scholar]
- Itoh, J.-I., Hasegawa, A., Kitano, H., and Nagato, Y. (1998). A recessive heterochronic mutation, plastochron 1, shortens the plastochron and elongates the vegetative phase in rice. Plant Cell 10, 1511–1521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jackson, D., and Hake, S. (1999). Control of phyllotaxy in maize by the abphyl1 gene. Development 126, 315–329. [DOI] [PubMed] [Google Scholar]
- Jackson, D., Veit, R., and Hake, S. (1994). Expression of maize KNOTTED1 related homeobox genes in the shoot apical meristem predicts patterns of morphogenesis in the vegetative shoot. Development 120, 405–413. [Google Scholar]
- Kerstetter, R.A., and Hake, S. (1997). Shoot meristem formation in vegetative development. Plant Cell 9, 1001–1010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kerstetter, R.A., Laudencia-Chingcuanco, D., Smith, L.G., and Hake, S. (1997). Loss-of-function mutations in the maize homeobox gene, knotted1, are defective in shoot meristem maintenance. Development 124, 3045–3054. [DOI] [PubMed] [Google Scholar]
- Kouchi, H., and Hata, S. (1993). Isolation and characterization of novel nodulin cDNA representing genes expressed at early stages of soybean nodule development. Mol. Gen. Genet. 238, 106–119. [DOI] [PubMed] [Google Scholar]
- Laux, T., Mayer, K.F.X., Berger, J., and Jurgens, G. (1996). The WUSCHEL gene is required for shoot and floral meristem identity in Arabidopsis. Development 122, 87–96. [DOI] [PubMed] [Google Scholar]
- Leyser, H.M.O., and Furner, I.J. (1992). Characterization of three apical meristem mutants of Arabidopsis thaliana. Development 116, 397–413. [Google Scholar]
- Long, J.A., Moan, E.I., Medford, J.I., and Barton, M.K. (1996). A member of the KNOTTED class of homeodomain proteins encoded by the STM gene of Arabidopsis. Nature 379, 66–69. [DOI] [PubMed] [Google Scholar]
- Lynn, K., Fernandez, A., Aida, M., Sedbrook, J., Tasaka, M., Masson, P., and Barton, M.K. (1999). The PINHEAD/ZWILLE gene acts pleiotropically in Arabidopsis development and has overlapping functions with the ARGONAUTE1 gene. Development 126, 469–481. [DOI] [PubMed] [Google Scholar]
- Matsuoka, M., Ichikawa, H., Saito, A., Tada, Y., Fujimura, T., and Kano-Murakami, Y. (1993). Expression of a rice homeobox gene causes altered morphology of transgenic plants. Plant Cell 5, 1039–1048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayer-Klaus, F.X., Schoof, H., Haecker, A., Lenhard, M., Juergens, G., and Laux, T. (1998). Role of WUSCHEL in regulating stem cell fate in the Arabidopsis shoot meristem. Cell 95, 805–815. [DOI] [PubMed] [Google Scholar]
- Medford, J.I. (1992). Vegetative apical meristems. Plant Cell 4, 1029–1039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyerowitz, E.M. (1997). Genetic control of cell division patterns in developing plants. Cell 88, 299–308. [DOI] [PubMed] [Google Scholar]
- Moussian, B., Schoof, H., Haecker, A., Jurgens, G., and Laux, T. (1998). Role of the ZWILLE gene in the regulation of central shoot meristem cell fate during Arabidopsis embryogenesis. EMBO J. 17, 1799–1809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murashige, T., and Skoog, F. (1962). A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15, 473–497. [Google Scholar]
- Nagasawa, N., Miyoshi, M., Kitano, H., Satoh, H., and Nagato, Y. (1996). Mutations associated with floral organ number in rice. Planta 198, 627–633. [DOI] [PubMed] [Google Scholar]
- Running, M.P., and Meyerowitz, E.M. (1996). Mutations in the PERIANTHIA gene of Arabidopsis specifically alter floral organ number and initiation pattern. Development 122, 1261–1269. [DOI] [PubMed] [Google Scholar]
- Sato, Y., Hong, S., Tagiri, A., Kitano, H., Yamamoto, N., Nagato, Y., and Matsuoka, M. (1996). A rice homeobox gene, OSH1, is expressed before organ differentiation in a specific region during early embryogenesis. Proc. Natl. Acad. Sci. USA 93, 8117–8122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Satoh, N., Hong, S.-K., Nishimura, A., Matsuoka, M., Kitano, H., and Nagato, Y. (1999). Initiation of shoot apical meristem in rice: Characterization of four SHOOTLESS genes. Development 126, 3629–3636. [DOI] [PubMed] [Google Scholar]
- Scanlon, M.J., and Freeling, M. (1997). Clonal sectors reveal that a specific meristematic domain is not utilized in the maize mutant narrow sheath. Dev. Biol. 182, 52–66. [DOI] [PubMed] [Google Scholar]
- Scanlon, M.J., Schneeberger, R.G., and Freeling, M. (1996). The maize mutant narrow sheath fails to establish leaf margin identity in a meristematic domain. Development 122, 1683–1691. [DOI] [PubMed] [Google Scholar]
- Schneeberger, R., Tsiantis, M., Freeling, M., and Langdale, J.A. (1998). The rough sheath2 gene negatively regulates homeobox gene expression during maize leaf development. Development 12, 787–795. [DOI] [PubMed] [Google Scholar]
- Selker, J.L., Steucek, G.L., and Green, P.B. (1992). Biophysical mechanisms for morphogenetic progression at the shoot apex. Dev. Biol. 153, 29–43. [DOI] [PubMed] [Google Scholar]
- Sinha, N.R., Williams, R.E., and Hake, S. (1993). Overexpression of the maize homeobox gene, Knotted1, causes a switch from determinate to indeterminate fates. Genes Dev 7, 2857–2865. [DOI] [PubMed] [Google Scholar]
- Smith, L.G., Greene, B., Veit, B., and Hake, S. (1992). A dominant mutation in the maize homeobox gene, Knotted-1, causes its ectopic expression in leaf cells with altered fates. Development 116, 21–30. [DOI] [PubMed] [Google Scholar]
- Snow, M., and Snow, R. (1931). Experiments on phyllotaxy. I. The effect of isolating a primordium. Philos. Trans. Roy. Soc. London. 25, 301–322. [Google Scholar]
- Souer, E., Houwelingen, A.V., Kloos, D., Mol, J., and Koes, R. (1996). The No Apical Meristem gene of Petunia is required for pattern formation in embryos and flowers and is expressed at meristem and primordia boundaries. Cell 85, 159–170. [DOI] [PubMed] [Google Scholar]
- Steeves, T.A., and Sussex, I.M. (1989). Patterns in Plant Development, 2nd ed. (Cambridge, UK: Cambridge University Press).
- Tamura, Y., Kitano, H., Satoh, H., and Nagato, Y. (1992). A gene profoundly affecting shoot organization in the early phase of rice development. Plant Sci. 82, 91–99. [Google Scholar]
- Timmermans-Marja, C.P., Schultes, N.P., Jankovsky, J.P., and Nelson, T. (1998). Leafbladeless1 is required for dorsoventrality of lateral organs in maize. Development 125, 2813–2823. [DOI] [PubMed] [Google Scholar]
- Timmermans-Marja, C.P., Hudson, A., Becraft, P.W., and Nelson, T. (1999). Rough sheath2: A Myb protein that represses knox homeobox genes in maize lateral organ primordia. Science 284, 151–153. [DOI] [PubMed] [Google Scholar]
- Tsiantis, M., Schneeberger, R., Golz, J.F., Freeling, M., and Langdale, J.A. (1999). The maize rough sheath2 gene and leaf development programs in monocot and dicot plants. Science 284, 154–156. [DOI] [PubMed] [Google Scholar]
- Veit, B., Briggs, S.P., Schmidt, R.J., Yanofsky, M.F., and Hake, S. (1998). Regulation of leaf initiation by the terminal ear 1 gene of maize. Nature 393, 166–168. [DOI] [PubMed] [Google Scholar]
- Wardlaw, C.W. (1949) Experiments on organogenesis in ferns. Growth 13 (suppl.), 93.–131. [Google Scholar]