Abstract
Background
The gonads are responsible for the production of germ cells through both mitosis and meiosis. Skp2 is the receptor subunit of an SCF-type ubiquitin ligase and is a major regulator of the progression of cells into S phase of the cell cycle, which it promotes by mediating the ubiquitin-dependent degradation of p27, an inhibitor of cell proliferation. However, the role of the Skp2-p27 pathway in germ cell development remains elusive.
Results
We now show that disruption of Skp2 in mice results in a marked impairment in the fertility of males, with the phenotypes resembling Sertoli cell-only syndrome in men. Testes of Skp2-/- mice manifested pronounced germ cell hypoplasia accompanied by massive apoptosis in spermatogenic cells. Flow cytometry revealed an increased prevalence of polyploidy in spermatozoa, suggesting that the aneuploidy of these cells is responsible for the induction of apoptosis. Disruption of the p27 gene of Skp2-/- mice restored germ cell development, indicating that the testicular hypoplasia of Skp2-/- animals is attributable to the antiproliferative effect of p27 accumulation.
Conclusion
Our results thus suggest that compromised cell cycle progression caused by the accumulation of p27 results in aneuploidy and the induction of apoptosis in gonadal cells of Skp2-/- mice. The consequent reduction in the number of mature gametes accounts for the decreased fertility of these animals. These findings reinforce the importance of the Skp2-p27 pathway in cell cycle regulation and in germ cell development.
Background
Infertility affects 10 to 15% of couples with up to half of fertility problems having a genetic etiology [1]. A major type of infertility is characterized by impaired production of germ cells. Germ cell development begins with the appearance of primordial germ cells at the early stage of embryogenesis. These cells migrate to the genital ridge, where they proliferate extensively by mitosis in both male and female embryos to establish the original pool of germ cells. The germ cells then enter a state of divisional arrest and remain in this state until sexual maturity, when they complete their developmental process by undergoing spermatogenesis in males and folliculogenesis in females. Such gametogenesis is highly sensitive to deficiency of various contributing factors. Targeted disruption of several genes in mice has thus defined key roles for many extracellular and intracellular signaling proteins in germ cell development and reproductive physiology [2-5].
Germ cells undergo both mitosis and meiosis during their development. Progression through the cell cycle requires the activity of two major ubiquitin ligase complexes, the Skp1-cullin-F-box protein (SCF) complex and the anaphase-promoting complex (APC) or cyclosome [6,7]. The F-box protein component of the SCF complex is variable, binds to Skp1 through its F-box motif, and is the subunit responsible for substrate recognition [8]. The F-box protein Skp2 plays an important role in progression of S phase of the cell cycle by contributing to the ubiquitin-dependent degradation of p27, a major inhibitor of proliferation in mammalian cells [6,9-11]. Skp2 begins to accumulate in late G1 phase of the cell cycle and its abundance is maximal during S and G2 phases [12-14]. We have previously generated mice that lack Skp2 and shown that the levels of p27 and of various other regulators of the cell cycle are increased in the cells of these animals [6,15-18]. Although Skp2-/- mice are viable, their somatic cells contain markedly enlarged nuclei and manifest both polyploidy and multiple centrosomes [6]. Such defects are not apparent in Skp2-/-;p27-/- mice, suggesting that they are largely the result of the abnormal accumulation of p27 in the Skp2-/- animals [14,19].
We noticed that the fertility of male Skp2-/- mice was reduced. We now show that, unlike other organs of these animals, the testes exhibit massive apoptosis, resulting in the loss of gametes. These characteristics were not apparent in Skp2-/-;p27-/- mice, suggesting that accumulation of p27 accounts for the defect in germ cell development in the Skp2-/- animals. These results indicate that regulation of cell cycle progression by the Skp2-p27 pathway is critical for germ cell development in both males and females.
Results
Reduced male fertility of Skp2-deficient mice
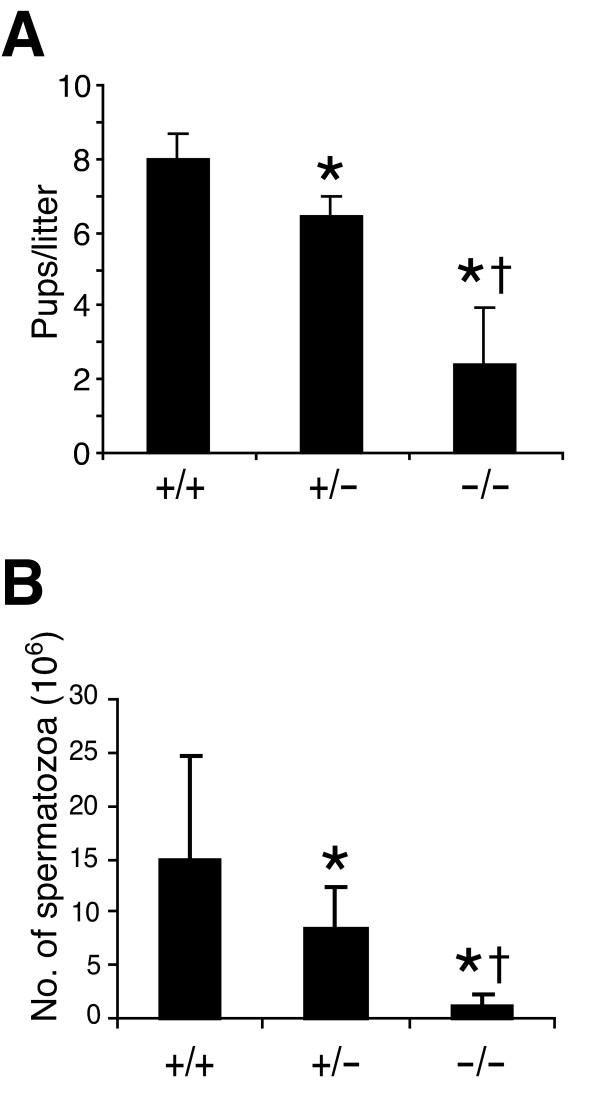
The effect of Skp2 deficiency on the male fertility was examined separately by crossing with wild-type C57BL/6 mice. Mating of Skp2-/- male mice with wild type pairs revealed a marked decrease in fertility, as evidenced by reduced litter size, compared with that of Skp2+/+ pairs or Skp2+/- pairs (Fig. 1A). The fertility of Skp2+/- pairs was also lower than that of wild-type pairs.
Figure 1.
Fertility and germ cell production in Skp2-deficient male mice. (A) Fertility rate of male wild-type and mutant mice (2 to 4 months of age) as reflected by litter size when crossed with control C57BL/6 mice. Data are means ± SD for five animals per genotype. *P < 0.05 versus Skp2+/+, †P < 0.05 versus Skp2+/-. (B) Germ cell production in male wild-type and mutant mice. Epididymal sperm in 2-month-old mice were counted. Data are means ± SD for 8–12 mice per genotype. *P < 0.05 versus Skp2+/+, †P < 0.05 versus Skp2+/-.
Epididymal spermatozoa were enumerated for evaluation of germ cell production in adult animals. Male Skp2-/- mice exhibited a markedly reduced number of spermatozoa (azoospermia in some animals) compared with wild-type or heterozygous males (Fig. 1B). Furthermore, a substantial number of abnormally large cells and degenerative bodies was apparent in the semen of Skp2-/- males (see Fig. 4D). The number of motile spermatzoa achieved >50% of the value for wild-type males in only one of 12Skp2-/- males examined.
Figure 4.
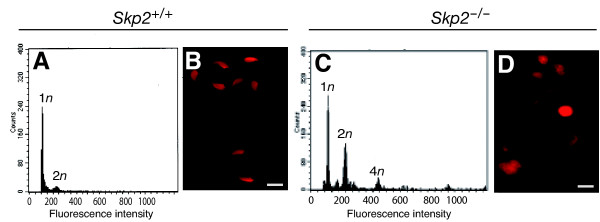
Polyploidy of epididymal sperm and ovarian granulosa cells of Skp2-/- mice. (A, C) Flow cytometric analysis of the DNA content of seminal cells isolated from wild-type (A) and Skp2-/- (C) males at 4 months of age. (B, D) Nuclear morphology of seminal cells isolated from wild-type (B) and Skp2-/- (D) mice and stained with propidium iodide. Scale bars, 50 μm.
Testicular atrophy in Skp2-deficient mice
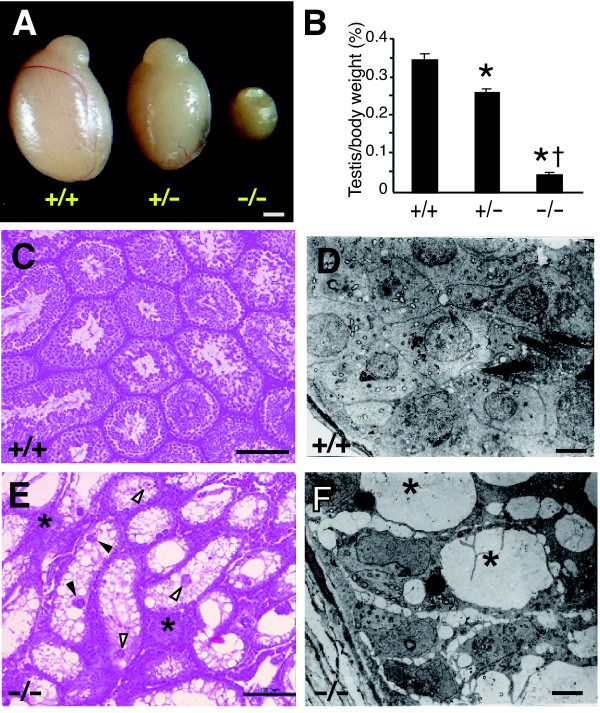
The testes and accessory reproductive organs of Skp2+/+, Skp2+/-, and Skp2-/- mice were further examined for histopathologic abnormalities that might contribute to the reduced fertility of the mutant animals. The testes of Skp2-/- males were markedly hypotrophic and hypoplastic compared with those of Skp2+/+ or Skp2+/- animals (Fig. 2A). Given that the body size of Skp2-/- mice is smaller than that of wild-type mice, we normalized testis weight by body weight for each animal; the normalized testis weight was still greatly reduced for Skp2-/- mice compared with that for Skp2+/+ or Skp2+/- animals (Fig. 2B). No gross morphological abnormalities were apparent in accessory reproductive glands of Skp2-/- males (data not shown).
Figure 2.
Testicular morphology of Skp2-deficient mice. (A) Macroscopic comparison of the testes of Skp2+/+, Skp2+/-, and Skp2-/- mice at 2 months of age. Scale bar, 1 mm. B, Ratio of the weight of testes to body weight in male mice of the three genotypes at 2 months of age. Data are means ± SD for 6–8 animals of each genotype. *P < 0.05 versus Skp2+/+, †P < 0.05 versus Skp2+/-. (C-F) Representative light (C, E) and electron (D, F) micrographs of testicular sections of wild-type (C, D) and Skp2-/- (E, F) males at 2–4 months of age. Note the pronounced loss of spermatogenic cells (asterisks in F), leaving only Sertoli cells, and the hyperplasia of the interstitial cellular population (asterisks in E) apparent adjacent to severely degenerated tubules in Skp2-/- testis. Degenerating spermatocytes and clusters of round or elongated spermatids had detached from the seminiferous epithelium and been sloughed off into the tubule lumen of Skp2-/- males (open arrowheads in E). Multinucleated giant spermatogenic cells were also present throughout the seminiferous epithelium of Skp2-/- testis (closed arrowheads in E). Scale bars, 100 μm (C, E) or 5 μm (D, F).
Light and electron microscopic examination of the testes of Skp2-/- mice revealed a progressive loss of spermatogonia with age, resulting in a marked depletion of germ cells in most seminiferous tubules (Fig. 2E, F) in comparison with wild-type testes (Fig. 2C, D). In advanced stages of germ cell loss, postmeiotic cells were rarely observed in seminiferous tubules of Skp2-/- males, with only Sertoli cells remaining. This phenotype is similar to that of Sertoli cell-only syndrome in humans. Ultrastructural analysis revealed the presence of numerous vacuolated Sertoli cells, resulting from germ cell depletion, in the tubule epithelium of such Skp2-/- males (Fig. 2F). Degenerating spermatocytes and clusters of round or elongated spermatids had detached from the seminiferous epithelium and been sloughed off into the tubule lumen (Fig. 2E). Multinucleated giant spermatogenic cells, containing from two to five or more nuclei, were also present throughout the seminiferous epithelium (Fig. 2E). The number and morphology of Sertoli cells in Skp2-/- mice were otherwise similar to those in wild-type animals, whereas interstitial cellularity was increased, especially for Leydig cells, in the mutant males (Fig. 2E). The profile of spermatogenesis in most seminiferous tubules of Skp2+/- mice was similar to that in wild-type animals, although detachment of spermatogenic cells was apparent in a few tubules of the heterozygotes (data not shown).
Increased apoptosis in the gonads of Skp2-deficient mice
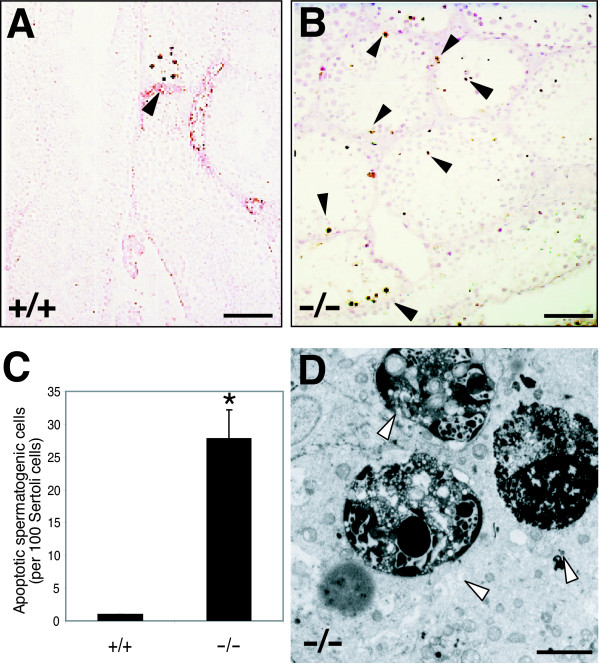
Cultured embryonic fibroblasts derived from Skp2-/- mice manifest an increased prevalence of apoptosis compared with those derived from wild-type animals [6]. We therefore performed the TUNEL assay to determine whether apoptosis contributes to the progressive loss of germ cells in Skp2-/- mice. Only a few spermatogenic cells of wild-type males, usually those at the spermatogonial stage, were found to be apoptotic (Fig. 3A). In contrast, a large proportion of spermatogenic cells, at almost all stages of spermatogenesis, was apoptotic in Skp2-/- males (Fig. 3B). Statistical analysis confirmed that the ratio of apoptotic cells was significantly increased in Skp2-/- mice (Fig. 3C). Electron microscopy also revealed the marked increase in the prevalence of apoptosis among spermatogenic cells, including those at the postmeiotic stage, in Skp2-/- males (Fig. 3D). The level of apoptosis among supporting cells, including Sertoli, Leydig, and other interstitial cells, did not differ substantially between Skp2-/- and wild-type males.
Figure 3.
Increased level of apoptosis in the gonads of Skp2-/- mice. (A, B) TUNEL staining of testicular sections of Skp2+/+ (A) or Skp2-/- (B) mice at 4 months of age. Arrowheads indicate apoptotic cells. Scale bars, 100 μm. (C) The number of apoptotic spermatogenic cells per 100 Sertoli cells. *P < 0.05 versus Skp2+/+. (D) Ultrastructural image of typical apoptotic figures (arrowheads) at late postmeiotic stages of spermatogenesis in a Skp2-/- mouse at 4 months of age. Scale bar, 5 μm.
Polyploidy and cellular dysmorphism of germ cells of Skp2-deficient mice
Isolated seminal cells were subjected to analysis of DNA content. Seminal cells from wild-type mice contained a majority of haploid (1n) cells, corresponding to normal mature spermatozoa (Fig. 4A). However, three out of seven (43%) Skp2-/- males examined manifested a substantial number of polyploid cells in their semen (Fig. 4C); the proportion of haploid cells was thus reduced and that of diploid (2n), tetraploid (4n), and >4n cells was increased. Examination of the nuclear morphology of seminal cells by propidium iodide staining revealed a majority of laterally flattened, typically curved, falciform, hock-headed spermatozoa in wild-type males (Fig. 4B). In contrast, the seminal cells of Skp2-/- males, in addition to some normally shaped haploid spermatozoa, contained a large number of cells with an abnormal nuclear size and shape (Fig. 4D).
Effects of p27 accumulation in Skp2-defcient mice
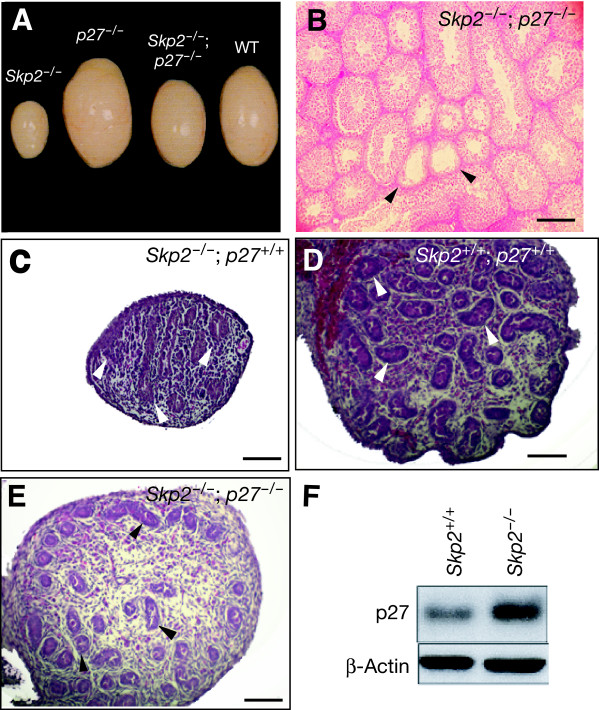
To examine the possible role of p27 in the impaired fertility of Skp2-/- mice, we analyzed Skp2-/-;p27-/- double-mutant animals. The gonads of the double-mutant mice showed a general reversal of the marked hypoplasia apparent in parental Skp2-/- mice (Fig. 5A). Disruption of the p27 gene thus largely restored the pool of germ cells in both testis (Fig. 5B). A small number of germ cell-depleted seminiferous tubules was still apparent in double-mutant males, however. The fertility rate of double-mutant males (n = 6) when crossed with C57BL/6 females was also increased to 5.66 ± 1.03 pups per litter (see Fig. 1A). The double-mutant females were completely sterile, however, which is a characteristic of p27 deficiency [20].
Figure 5.
Role of p27 accumulation in the gonadal hypoplasia of Skp2-deficient mice. (A) Macroscopic comparison of the testes of wild-type, Skp2-/-, p27-/-, and Skp2-/-;p27-/- mice. (B) Histology of the testis of Skp2-/-;p27-/- mice (4 months of age) showing pronounced recovery of germ cell production compared with that apparent in parental Skp2-/- mice. Scale bars, 100 μm. Arrowheads in B indicate seminiferous tubules with a deficiency of germ cells. (C-E) Histology of embryonic testis (15.5 days postcoitum). Severe deficiency of gonocytes (arrowheads) was evident in Skp2-/- embryos (C) compared with wild-type embryos (D). However, the gonocyte population had recovered substantially in Skp2-/-;p27-/- embryos (E). Scale bars, 100 μm. (F) Immunoblot analysis of lysates of the testes of Skp2-/- or wild-type embryos (15.5 days postcoitum) with antibodies to p27 and to β-actin (loading control).
The normalization of the reproductive systems of the double-mutant mice was apparent from early stages of germ cell development, especially in male animals. Although there was a substantial reduction in the number of gonocyte nests in the testes of Skp2-/- embryos (Fig. 5C), the gonocyte reserves in double-mutant embryos (Fig. 5E) were similar to those in wild-type embryos (Fig. 5D). In addition, the amount of p27 in the embryonic testis at the early stage of germ cell development (15.5 days postcoitum) was markedly increased in Skp2-/- mice compared with wild-type animals (Fig. 5F).
Discussion
We have shown that male Skp2-/- mice manifest a markedly reduced fertility. The gonads of male mutant mice exhibited a pronounced hypoplasia that was independent of the reduced body size of these animals. This gonadal hypoplasia was likely attributable to the lack of Skp2-dependent degradation of p27 by the proteasome [14,19]. Indeed, we have now shown that p27 accumulates in the testis of Skp2-/- embryos at the early stage of gonocyte development. An important function of p27 is to restrain progression of the cell cycle, and this protein accumulates in response to many antiproliferative signals [21]. We and others previously showed that disruption of the p27 gene removes this brake and results in uncontrolled cell proliferation and hyperplasia of most organs, especially the testis and ovary [20,22,23]. In contrast, the accumulation of p27 in Skp2-/- mice likely exerts an antiproliferative effect on embryonic germ cells, resulting in gonadal hypoplasia. This notion was supported by the observation that germ cell production was restored from an early stage of gonocyte development in Skp2-/-;p27-/- double-mutant mice.
The marked depletion of spermatogenic cells apparent in adult Skp2-/- males is reminiscent of that in infertile men with Sertoli cell-only syndrome [24]. The mutant male mice manifest hyperplasia of Leydig and other interstitial cells adjacent to the germ cell-depleted seminiferous tubules. A similar phenotype has also been described in human testicular disorders characterized by germ cell depletion and is thought to be a histological marker of testicular failure in men [25]. The accelerated depletion of germ cells in adult Skp2-/- mice appears to be mediated by apoptosis, which is responsible for the removal of cells with abnormalities, such as an incorrect DNA content [26,27]. Such physiological apoptosis is a normal feature of both testis [28] and ovary, in the latter of which atretic follicles are prevented by apoptosis from achieving the final steps of follicular growth [28-30]. Adult Skp2-/- mice manifested a greatly increased frequency of apoptosis during gametogenesis, however. In the testis, cells at various stages of spermatogenesis were found to be apoptotic, resulting in germ cell depletion.
The increased level of apoptosis apparent in the gonads of Skp2-/- mice might be attributable to polyploidy, which was previously detected in somatic organs, including the liver, kidneys, and lungs, of these animals without any evident accompanying functional defects [6,14]. In general, abnormalities of nuclear DNA content during gametogenesis disturb the development of any resulting zygote, leading to infertility or embryo loss due to aneuploidy [31,32]. We now show that both germ cells and their supporting cells of Skp2-/- mice are affected by polyploidy. In male mutant animals, polyploidy of spermatozoa likely contributed to the morphological abnormalities of these cells, given that morphologically abnormal (such as macrocephalic) spermatozoa have been shown to be polyploid both in mice [33] and in infertile men [34-38]. Polyploidy and consequent apoptosis are also likely to be responsible for detachment of spermatogonia and spermatocytes from the seminiferous epithelium and their formation of giant multinucleated structures in the tubules of Skp2-/- males. Similar structures have been shown to represent syncytia of degenerating spermatids [39-42]. Aneuploidy has also been proposed to underlie the induction of spermatogenic cell apoptosis and the development of Sertoli cell-only syndrome in men [43].
The number of ova recoverable from Skp2-/- female mice was too small to determine the presence of aneuploidy (data not shown). However, the high prevalence of apoptosis among ovarian granulosa cells was accompanied by an increased frequency of polyploidy in the mutant animals. Given the important role of granulosa cells in maintaining the female germ cells [44], follicles with apoptotic granulosa cells likely fail to progress to later stages of development and eventually undergo follicular atresia.
Impaired progression of the cell cycle caused by the absence of Skp2, an important controller of S phase, is likely responsible for the development of polyploidy in Skp2-/- mice. Various chemical agents such as colchicines and vinblastine similarly affect both mitotic and meiotic cell division [45] and induce cell cycle delays in both somatic [46] and germ [47] cells, resulting in aneuploidy. Some of these chemicals delay progression of S or G2 phases of the cell cycle and thereby prolong cell cycle time [48,49]. Given that the abundance of Skp2 is maximal during S and G2 phases [12-14], it might be expected that its absence in these phases would lead to aneuploidy through a similar mechanism. Aneuploidy may also develop in the germ cells of Skp2-/- mice by a mechanism similar to that operative in somatic cells of these animals; that is, endoreplication caused by the accumulation of p27 [14]. Indeed, the lack of p27 degradation during G2 phase in Skp2-/- cells may result in suppression of Cdc2 activity and consequent inhibition of entry into M phase [14].
p27 antagonizes the activity of not only Cdc2, but also Cdk2 and Cdk4. Interestingly, mutant mice that lack the activity of Cdk2 or Cdk4 also exhibit the abnormalities in the gonadal development. Cdk2-/- males and females are sterile with a severe atrophy of the gonads [50,51]. Cyclin E2-deficient males displayed reduced fertility, with approximately 50% of males being sterile [52]. Cyclin E2-/- males displayed reduced testicular size and greatly reduced sperm counts, as compared with wild-type littermates. Cyclin E2-deficient females develop normally and are fully fertile. Cdk4-/- mice showed reproductive dysfunction associated with hypoplastic seminiferous tubules in the testis and perturbed corpus luteum formation in the ovary [53,54]. Furthermore, cyclin D2-deficient females are sterile owing to the inability of ovarian granulosa cells to proliferate normally in response to follicle-stimulating hormone (FSH), whereas mutant males display hypoplastic testes [55]. Overall, these data suggest that Skp2-dependent control of p27 abundance plays a critical role in the regulation of the activity of Cdk2 and Cdk4, which is particularly important for the normal gonadal development.
Conclusion
Our results suggest that Skp2, as an important regulator of S phase of the cell cycle, plays a key role in establishment of the original pool of gametic cells by mitosis during early embryogenesis as well as in the proliferation and maturation of these cells at later stages of development. Our findings reinforce the importance both of cell cycle regulators in germ cell development and of impaired function of such factors in fertility problems.
Methods
Animals
Skp2-deficient mice were generated by homologous recombination in embryonic stem cells as described previously [6]. Sexually mature mice from 2 to 12 months of age were used for experimental procedures. Pairs of Skp2+/-;p27+/- mice were mated to produce Skp2-/-;p27-/- animals [14]. All animal studies conformed with the Kyushu University Animal Experimentation Act. For evaluation of fertility, pairs of Skp2+/+, Skp2+/-, or Skp2-/- mice were housed together for 8 weeks and then separated. Cages were monitored daily for the presence of seminal plugs, and the number and size of litters were recorded.
Histopathology
For light microscopic analysis, tissue samples were fixed in either Bouin's fixative or 4% paraformaldehyde, dehydrated, treated with xylene, embedded in paraffin, and sectioned at a thickness of 5 μm. After removal of paraffin, the sections were dehydrated, rehydrated, and stained with hematoxylin-eosin. They were then examined under a Nikon Eclipse E800 microscope with either Nomarski or phase-contrast optics; images were photographed with a Hamamatsu 3CCD digital camera (model 7780). For electron microscopy, samples were fixed in glutaraldehyde immediately after resection and were then embedded in resin. Sections with a thickness of 80 nm were collected on copper grids and counterstained with lead citrate and uranyl acetate. They were then observed with a JEOL (JEM 2000) electron microscope at a voltage of 80 kV.
TUNEL assay
For detection of apoptotic cells, tissue fixed in 4% paraformaldehyde or Bouin's solution was processed for the TUNEL (terminal deoxyribonucleotidyl transferase-mediated dUTP-biotin nick-end labeling)1 assay essentially as described previously [56]. Apoptotic cells were visualized with the chromogen 3,3'-diaminobenzidine tetrahydrochloride (Sigma, St. Louis, MO).
Isolation of spermatozoa
Mature male mice were killed by neck dislocation and the vas deferens and caudal epididymis were immediately exposed through a lower abdominal incision, excised, and washed briefly in phosphate-buffered saline (PBS). The tissue was then incubated in PBS for 30 min at 37°C and briefly massaged to promote the active exit of sperm from the epididymis. The released spermatozoa were isolated by centrifugation at 200 × g for 20 min, washed three times with PBS, counted with a hemocytometer attached to a light microscope, and analyzed for DNA content and morphology.
Flow cytometric analysis of DNA content
Isolated spermatozoa or granulosa cells (1 × 106) were washed in PBS, resuspended in 200 μl of PBS, and fixed by the gradual addition of 800 μl of 100% ice-cold ethanol. The fixed cells were washed twice with PBS, resuspended in 1 ml of PBS, treated with RNase (40 μg/ml) for 10 min at room temperature, and then stained with propidium iodide (25 μg/ml). The DNA content of the cells was determined by flow cytometry with a FACSCaliber instrument and CellQuest software (Becton Dickinson, San Jose, CA). The morphology of propidium iodide-stained spermatozoa was also examined with a fluorescence microscope.
Immunoblot analysis
Embryonic testes were excised under a dissecting microscope, homogenized, and lysed in the presence of protease inhibitors. The tissue lysate (30 μg of protein) was fractionated by SDS-polyacrylamide gel electrophoresis, and the separated proteins were transferred to a nitrocellulose membrane and subjected to immunoblot analysis with rabbit polyclonal antibodies to mouse p27 (Santa Cruz Biotechnology, Santa Cruz, CA) and horseradish peroxidase-conjugated secondary antibodies. Immune complexes were detected with enhanced chemiluminescence reagents (Amersham Biosciences, Little Chalfont, UK). Immunodetection of β-actin was performed as a control for protein loading.
Statistical analysis
Data are presented as means ± SD. The significance of differences between means was determined by ANOVA test. A P value of <0.05 was considered statistically significant.
Abbreviations
TUNEL: terminal deoxyribonucleotidyl transferase-mediated dUTP-biotin nick-end labeling, PBS: phosphate-buffered saline
Authors' contributions
FA performed all analyses of germ cell development. KN and KIN generated p27-/-, Skp2-/-, and Skp2-/-;p27-/- mice. KIN is the principal investigator who gave advice in designing the study and edited the manuscript. All authors read and approved the final manuscript.
Acknowledgments
Acknowledgements
We thank R. Tsunematsu and C. Oshikawa for technical advice; M. Sasaki, Y. Yamada, and S. Matsushita for technical assistance; and A. Ohta and M. Kimura for help in preparation of the manuscript. This work was supported in part by a grant from the Ministry of Education, Culture, Sports, Science, and Technology of Japan.
Contributor Information
Abbas Fotovati, Email: abbas@med.kurume-u.ac.jp.
Keiko Nakayama, Email: nakayak2@mail.tains.tohoku.ac.jp.
Keiichi I Nakayama, Email: nakayak1@bioreg.kyushu-u.ac.jp.
References
- Maduro MR, Lamb DJ. Understanding new genetics of male infertility. J Urol. 2002;168:2197–2205. doi: 10.1016/S0022-5347(05)64355-8. [DOI] [PubMed] [Google Scholar]
- Grootegoed JA BWMRHPHJH. Knockout mouse model and gametogenic failure. Mol Cell Endocrinol. 1998;145:161–166. doi: 10.1016/S0303-7207(98)00183-X. [DOI] [PubMed] [Google Scholar]
- Matzuk MM, Lamb DJ. Genetic dissection of mammalian fertility pathways. Nat Med. 2002;8 Suppl:S33–S40. doi: 10.1038/ncb-nm-fertilityS41. [DOI] [PubMed] [Google Scholar]
- Escalier D. Impact of genetic engineering on the understanding of spermatogenesis. Hum Reprod Update. 2001;7:191–210. doi: 10.1093/humupd/7.2.191. [DOI] [PubMed] [Google Scholar]
- Epifano O, Dean J. Genetic control of early folliculogenesis in mice. Trends Endocrinol Metab. 2002;13:169–173. doi: 10.1016/S1043-2760(02)00576-3. [DOI] [PubMed] [Google Scholar]
- Nakayama K, Nagahama H, Minamishima YA, Matsumoto M, Nakamichi I, Kitagawa K, Shirane M, Tsunematsu R, Tsukiyama T, Ishida N, Kitagawa M, Nakayama KI, Hatakeyama S. Targeted disruption of Skp2 results in accumulation of cyclin E and p27(Kip1), polyploidy and centrosome overduplication. EMBO J. 2000;19:2069–2081. doi: 10.1093/emboj/19.9.2069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harper JW, Burton JL, Solomon MJ. The anaphase-promoting complex: it's not just for mitosis any more. Genes Dev. 2002;16:2179–2206. doi: 10.1101/gad.1013102. [DOI] [PubMed] [Google Scholar]
- Nakayama KI, Nakayama K. Regulation of the cell cycle by SCF-type ubiquitin ligases. Semin Cell Dev Biol. 2005;16:323–333. doi: 10.1016/j.semcdb.2005.02.010. [DOI] [PubMed] [Google Scholar]
- Carrano AC, Eytan E, Hershko A, Pagano M. SKP2 is required for ubiquitin-mediated degradation of the CDK inhibitor p27. Nat Cell Biol. 1999;1:193–199. doi: 10.1038/12013. [DOI] [PubMed] [Google Scholar]
- Sutterluty H, Chatelain E, Marti A, Wirbelauer C, Senften M, Muller U, Krek W. p45SKP2 promotes p27Kip1 degradation and induces S phase in quiescent cells. Nat Cell Biol. 1999;1:207–214. doi: 10.1038/12027. [DOI] [PubMed] [Google Scholar]
- Tsvetkov LM, Yeh KH, Lee SJ, Sun H, Zhang H. p27(Kip1) ubiquitination and degradation is regulated by the SCF(Skp2) complex through phosphorylated Thr187 in p27. Curr Biol. 1999;9:661–664. doi: 10.1016/S0960-9822(99)80290-5. [DOI] [PubMed] [Google Scholar]
- Zhang H, Kobayashi R, Galaktionov K, Beach D. p19Skp1 and p45Skp2 are essential elements of the cyclin A-CDK2 S phase kinase. Cell. 1995;82:915–925. doi: 10.1016/0092-8674(95)90271-6. [DOI] [PubMed] [Google Scholar]
- Hara T, Kamura T, Nakayama K, Oshikawa K, Hatakeyama S, Nakayama KI. Degradation of p27(Kip1) at the G(0)-G(1) transition mediated by a Skp2-independent ubiquitination pathway. J Biol Chem. 2001;276:48937–48943. doi: 10.1074/jbc.M107274200. [DOI] [PubMed] [Google Scholar]
- Nakayama K, Nagahama H, Minamishima YA, Miyake S, Ishida N, Hatakeyama S, Kitagawa M, Iemura S, Natsume T, Nakayama KI. Skp2-mediated degradation of p27 regulates progression into mitosis. Dev Cell. 2004;6:661–672. doi: 10.1016/S1534-5807(04)00131-5. [DOI] [PubMed] [Google Scholar]
- Bornstein G, Bloom J, Sitry-Shevah D, Nakayama K, Pagano M, Hershko A. Role of the SCFSkp2 ubiquitin ligase in the degradation of p21Cip1 in S phase. J Biol Chem. 2003;278:25752–25757. doi: 10.1074/jbc.M301774200. [DOI] [PubMed] [Google Scholar]
- Kamura T, Hara T, Kotoshiba S, Yada M, Ishida N, Imaki H, Hatakeyama S, Nakayama K, Nakayama KI. Degradation of p57Kip2 mediated by SCFSkp2-dependent ubiquitylation. Proc Natl Acad Sci U S A. 2003;100:10231–10236. doi: 10.1073/pnas.1831009100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tedesco D, Lukas J, Reed SI. The pRb-related protein p130 is regulated by phosphorylation-dependent proteolysis via the protein-ubiquitin ligase SCF(Skp2) Genes Dev. 2002;16:2946–2957. doi: 10.1101/gad.1011202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yada M, Hatakeyama S, Kamura T, Nishiyama M, Tsunematsu R, Imaki H, Ishida N, Okumura F, Nakayama K, Nakayama KI. Phosphorylation-dependent degradation of c-Myc is mediated by the F-box protein Fbw7. EMBO Journal. 2004;23:2116–2125. doi: 10.1038/sj.emboj.7600217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kossatz U, Dietrich N, Zender L, Buer J, Manns MP, Malek NP. Skp2-dependent degradation of p27kip1 is essential for cell cycle progression. Genes Dev. 2004;18:2602–2607. doi: 10.1101/gad.321004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakayama K, Ishida N, Shirane M, Inomata A, Inoue T, Shishido N, Horii I, Loh DY, Nakayama KI. Mice lacking p27(Kip1) display increased body size, multiple organ hyperplasia, retinal dysplasia, and pituitary tumors. Cell. 1996;85:707–720. doi: 10.1016/S0092-8674(00)81237-4. [DOI] [PubMed] [Google Scholar]
- Nakayama KI, Nakayama K. Cip/Kip cyclin-dependent kinase inhibitors: brakes of the cell cycle engine during development. Bioessays. 1998;20:1020–1029. doi: 10.1002/(SICI)1521-1878(199812)20:12<1020::AID-BIES8>3.0.CO;2-D. [DOI] [PubMed] [Google Scholar]
- Fero ML, Rivkin M, Tasch M, Porter P, Carow CE, Firpo E, Polyak K, Tsai LH, Broudy V, Perlmutter RM, Kaushansky K, Roberts JM. A syndrome of multiorgan hyperplasia with features of gigantism, tumorigenesis, and female sterility in p27(Kip1)-deficient mice. Cell. 1996;85:733–744. doi: 10.1016/S0092-8674(00)81239-8. [DOI] [PubMed] [Google Scholar]
- Kiyokawa H, Kineman RD, Manova-Todorova KO, Soares VC, Hoffman ES, Ono M, Khanam D, Hayday AC, Frohman LA, Koff A. Enhanced growth of mice lacking the cyclin-dependent kinase inhibitor function of p27(Kip1) Cell. 1996;85:721–732. doi: 10.1016/S0092-8674(00)81238-6. [DOI] [PubMed] [Google Scholar]
- Skakkebaek NE. Two types of tubules containing only Sertoli cells in adults with Klinefelter's syndrome. Nature. 1969;223:643–645. doi: 10.1038/223643a0. [DOI] [PubMed] [Google Scholar]
- Holm M, Rajpert-De Meyts E, Andersson AM, Skakkebaek NE. Leydig cell micronodules are a common finding in testicular biopsies from men with impaired spermatogenesis and are associated with decreased testosterone/LH ratio. J Pathol. 2003;199:378–386. doi: 10.1002/path.1309. [DOI] [PubMed] [Google Scholar]
- Clermont Y, Leblond CP. Renewal of spermatogonia in the rat. Am J Anat. 1953;93:475–501. doi: 10.1002/aja.1000930308. [DOI] [PubMed] [Google Scholar]
- Matsui Y. Regulation of germ cell death in mammalian gonads. Apmis. 1998;106:142–7; discussion 147-8. doi: 10.1111/j.1699-0463.1998.tb01329.x. [DOI] [PubMed] [Google Scholar]
- Yan W, Suominen J, Samson M, Jegou B, Toppari J. Involvement of Bcl-2 family proteins in germ cell apoptosis during testicular development in the rat and pro-survival effect of stem cell factor on germ cells in vitro. Mol Cell Endocrinol. 2000;165:115–129. doi: 10.1016/S0303-7207(00)00257-4. [DOI] [PubMed] [Google Scholar]
- Tilly JL, Kowalski KI, Johnson AL, Hsueh AJ. Involvement of apoptosis in ovarian follicular atresia and postovulatory regression. Endocrinology. 1991;129:2799–2801. doi: 10.1210/endo-129-5-2799. [DOI] [PubMed] [Google Scholar]
- Tilly JL. Commuting the death sentence: how oocytes strive to survive. Nat Rev Mol Cell Biol. 2001;2:838–848. doi: 10.1038/35099086. [DOI] [PubMed] [Google Scholar]
- Yuan L, Liu JG, Hoja MR, Wilbertz J, Nordqvist K, Hoog C. Female germ cell aneuploidy and embryo death in mice lacking the meiosis-specific protein SCP3. Science. 2002;296:1115–1118. doi: 10.1126/science.1070594. [DOI] [PubMed] [Google Scholar]
- Hassold T, Hunt P. To err (meiotically) is human: the genesis of human aneuploidy. Nat Rev Genet. 2001;2:280–291. doi: 10.1038/35066065. [DOI] [PubMed] [Google Scholar]
- Levy ER BPS. Diploid spermatids: a manifestation of spermatogenic impairment in XOSxr and T31H/+ male mice. Cytogenet Cell Genet. 1986;42:159–163. doi: 10.1159/000132269. [DOI] [PubMed] [Google Scholar]
- Devillard F, Metzler-Guillemain C, Pelletier R, DeRobertis C, Bergues U, Hennebicq S, Guichaoua M, Sele B, Rousseaux S. Polyploidy in large-headed sperm: FISH study of three cases. Hum Reprod. 2002;17:1292–1298. doi: 10.1093/humrep/17.5.1292. [DOI] [PubMed] [Google Scholar]
- Hassold T, Abruzzo M, Adkins K, Griffin D, Merrill M, Millie E, Saker D, Shen J, Zaragoza M. Human aneuploidy: incidence, origin, and etiology. Environ Mol Mutagen. 1996;28:167–175. doi: 10.1002/(SICI)1098-2280(1996)28:3<167::AID-EM2>3.0.CO;2-B. [DOI] [PubMed] [Google Scholar]
- Lee JD, Kamiguchi Y, Yanagimachi R. Analysis of chromosome constitution of human spermatozoa with normal and aberrant head morphologies after injection into mouse oocytes. Hum Reprod. 1996;11:1942–1946. doi: 10.1093/oxfordjournals.humrep.a019521. [DOI] [PubMed] [Google Scholar]
- Martin RH, Spriggs E, Rademaker AW. Multicolor fluorescence in situ hybridization analysis of aneuploidy and diploidy frequencies in 225,846 sperm from 10 normal men. Biol Reprod. 1996;54:394–398. doi: 10.1095/biolreprod54.2.394. [DOI] [PubMed] [Google Scholar]
- In't Veld PA, Broekmans FJ, de France HF, Pearson PL, Pieters MH, van Kooij RJ. Intracytoplasmic sperm injection (ICSI) and chromosomally abnormal spermatozoa. Hum Reprod. 1997;12:752–754. doi: 10.1093/humrep/12.4.752. [DOI] [PubMed] [Google Scholar]
- Chapin RE, Morgan KT, Bus JS. The morphogenesis of testicular degeneration induced in rats by orally administered 2,5-hexanedione. Exp Mol Pathol. 1983;38:149–169. doi: 10.1016/0014-4800(83)90082-5. [DOI] [PubMed] [Google Scholar]
- Singh SK, Abe K. Light and electron microscopic observations of giant cells in the mouse testis after efferent duct ligation. Arch Histol Jpn. 1987;50:579–585. doi: 10.1679/aohc.50.579. [DOI] [PubMed] [Google Scholar]
- Print CG, Loveland KL. Germ cell suicide: new insights into apoptosis during spermatogenesis. Bioessays. 2000;22:423–430. doi: 10.1002/(SICI)1521-1878(200005)22:5<423::AID-BIES4>3.0.CO;2-0. [DOI] [PubMed] [Google Scholar]
- Totsuka Y, Kawamori T, Hisada S, Mitsumori K, Ishihara J, Sugimura T, Wakabayashi K. Testicular toxicity in F344 rats by aminophenylnorharman, formed from norharman and aniline. Toxicol Appl Pharmacol. 2001;175:169–175. doi: 10.1006/taap.2001.9236. [DOI] [PubMed] [Google Scholar]
- Kostakopoulos A, Protoyerou V, Tekerlekis P, Georgoulakis J, Louras G, Goulandris N. DNA flow-cytometric, histological and hormonal analysis of sertoli cell only syndrome (SECOS) Int Urol Nephrol. 2002;33:77–79. doi: 10.1023/A:1014417322540. [DOI] [PubMed] [Google Scholar]
- Matzuk MM, Burns KH, Viveiros MM, Eppig JJ. Intercellular communication in the mammalian ovary: oocytes carry the conversation. Science. 2002;296:2178–2180. doi: 10.1126/science.1071965. [DOI] [PubMed] [Google Scholar]
- Adler ID. Synopsis of the in vivo results obtained with the 10 known or suspected aneugens tested in the CEC collaborative study. Mutat Res. 1993;287:131–137. doi: 10.1016/0027-5107(93)90152-6. [DOI] [PubMed] [Google Scholar]
- Chen HH, Hsueh JL, Sirianni SR, Huang CC. Induction of sister-chromatid exchanges and cell cycle delay in cultured mammalian cells treated with eight organophosphorus pesticides. Mutat Res. 1981;88:307–316. doi: 10.1016/0165-1218(81)90042-2. [DOI] [PubMed] [Google Scholar]
- Miller BM, Adler ID. Aneuploidy induction in mouse spermatocytes. Mutagenesis. 1992;7:69–76. doi: 10.1093/mutage/7.1.69. [DOI] [PubMed] [Google Scholar]
- Misra NC, Roberts DW. Inhibition by 4'-demethyl-epipodophyllotoxin 9-(4,6-O-2-thenylidene-beta-D-glucopyranoside) of human lymphoblast cultures in G2 phase of the cell cycle. Cancer Res. 1975;35:99–105. [PubMed] [Google Scholar]
- Erba E, Sen S, Lorico A, D'Incalci M. Potentiation of etoposide cytotoxicity against a human ovarian cancer cell line by pretreatment with non-toxic concentrations of methotrexate or aphidicolin. Eur J Cancer. 1992;28:66–71. doi: 10.1016/0959-8049(92)90387-H. [DOI] [PubMed] [Google Scholar]
- Berthet C, Aleem E, Coppola V, Tessarollo L, Kaldis P. Cdk2 knockout mice are viable. Curr Biol. 2003;13:1775–1785. doi: 10.1016/j.cub.2003.09.024. [DOI] [PubMed] [Google Scholar]
- Ortega S, Prieto I, Odajima J, Martin A, Dubus P, Sotillo R, Barbero JL, Malumbres M, Barbacid M. Cyclin-dependent kinase 2 is essential for meiosis but not for mitotic cell division in mice. Nat Genet. 2003;35:25–31. doi: 10.1038/ng1232. [DOI] [PubMed] [Google Scholar]
- Geng Y, Yu Q, Sicinska E, Das M, Schneider JE, Bhattacharya S, Rideout WM, Bronson RT, Gardner H, Sicinski P. Cyclin E ablation in the mouse. Cell. 2003;114:431–443. doi: 10.1016/S0092-8674(03)00645-7. [DOI] [PubMed] [Google Scholar]
- Moons DS, Jirawatnotai S, Tsutsui T, Franks R, Parlow AF, Hales DB, Gibori G, Fazleabas AT, Kiyokawa H. Intact follicular maturation and defective luteal function in mice deficient for cyclin- dependent kinase-4. Endocrinology. 2002;143:647–654. doi: 10.1210/en.143.2.647. [DOI] [PubMed] [Google Scholar]
- Tsutsui T, Hesabi B, Moons DS, Pandolfi PP, Hansel KS, Koff A, Kiyokawa H. Targeted disruption of CDK4 delays cell cycle entry with enhanced p27(Kip1) activity. Mol Cell Biol. 1999;19:7011–7019. doi: 10.1128/mcb.19.10.7011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sicinski P, Donaher JL, Geng Y, Parker SB, Gardner H, Park MY, Robker RL, Richards JS, McGinnis LK, Biggers JD, Eppig JJ, Bronson RT, Elledge SJ, Weinberg RA. Cyclin D2 is an FSH-responsive gene involved in gonadal cell proliferation and oncogenesis. Nature. 1996;384:470–474. doi: 10.1038/384470a0. [DOI] [PubMed] [Google Scholar]
- Manova K, Tomihara-Newberger C, Wang S, Godelman A, Kalantry S, Witty-Blease K, De Leon V, Chen WS, Lacy E, Bachvarova RF. Apoptosis in mouse embryos: elevated levels in pregastrulae and in the distal anterior region of gastrulae of normal and mutant mice. Dev Dyn. 1998;213:293–308. doi: 10.1002/(SICI)1097-0177(199811)213:3<293::AID-AJA6>3.0.CO;2-D. [DOI] [PubMed] [Google Scholar]