Abstract
Heart failure is accompanied by important defects in metabolism. The transcriptional coactivator peroxisome proliferator-activated receptor-γ coactivator 1α (PGC-1α) is a powerful regulator of mitochondrial biology and metabolism. PGC-1α and numerous genes regulated by PGC-1α are repressed in models of cardiac stress, such as that generated by transverse aortic constriction (TAC). This finding has suggested that PGC-1α repression may contribute to the maladaptive response of the heart to chronic hemodynamic loads. We show here that TAC in mice genetically engineered to lack PGC-1α leads to accelerated cardiac dysfunction, which is accompanied by signs of significant clinical heart failure. Treating cardiac cells in tissue culture with the catecholamine epinephrine leads to repression of PGC-1α and many of its target genes, recapitulating the findings in vivo in response to TAC. Importantly, introduction of ectopic PGC-1α can reverse the repression of most of these genes by epinephrine. Together, these data indicate that endogenous PGC-1α serves a cardioprotective function and suggest that repression of PGC-1α significantly contributes to the development of heart failure. Moreover, the data suggest that elevating PGC-1α activity may have therapeutic potential in the treatment of heart failure.
Keywords: cardiac metabolism, mitochondria, transcription
Heart failure has become a leading cause of death in the industrialized world (1). It is now recognized that a major element of heart failure results from the maladaptive remodeling of the heart that occurs after hemodynamic loads, such as those after myocardial infarction or chronic hypertension (reviewed in refs. 2 and 3). The mechanisms by which this remodeling occurs remain incompletely understood. Best understood are the neurohormonal changes that accompany heart failure, most notably those of the catecholaminergic and renin/angiotensin systems. Nearly all medications currently used clinically for the treatment of heart failure target these systems (2). However, the 5-year mortality of heart failure remains in the 50% range, rivaling that of many cancers (1), which underscores the inadequacy of current treatments and calls for a deeper understanding of mechanisms leading to heart failure.
Heart failure is also accompanied by major changes in the energetics of the heart (4–9). The heart has an extremely high energy requirement for normal function, using more ATP per gram of tissue than any other organ in the body (10). Maintaining this robust production of ATP is clearly vital for the heart in both health and disease. Consistent with this notion, failing human hearts have decreased ATP production (reviewed in refs. 4 and 8). Oxidative phosphorylation in mitochondria is by far the most efficient way to produce ATP; consistent with this fact, human diseases resulting from genetic abnormalities in mitochondrial function usually lead to heart failure (11). However, primary defects in mtDNA account for only a small minority of cases of heart failure and the attendant defects in ATP production. The mechanisms by which changes in mitochondrial oxidative activity occur in the setting of more common forms of heart failure remain unclear.
Proliferator-activated receptor-γ (PPAR-γ) coactivator 1α (PGC-1α) is a dominant regulator of oxidative metabolism in many tissues (reviewed in refs. 12 and 13). This transcriptional coactivator was originally isolated as a cold-inducible PPAR-γ binding protein in brown fat (14). Subsequent work indicated that this protein bound and coactivated most members of the nuclear receptor family, and many transcription factors outside the nuclear receptor family. PGC-1α links these transcriptional factors to chromatin-modifying enzymes, the basal transcriptional machinery, and the splicing machinery, to effect powerful induction of transcriptional activity. PGC-1α has a variety of biological activities, many of which are linked to oxidative metabolism and mitochondrial function. The expression of PGC-1α in white fat cells gives them many of the properties of brown fat cells, including a marked increase in mitochondrial number and expression of UCP-1. PGC-1α is induced by exercise in skeletal muscle (e.g., refs. 15–18), and when expressed transgenically in skeletal muscle, it induces a switch in fibers toward more oxidative types I and IIa (19). PGC-1α is induced in the fasted liver, where it induces the mitochondrial beta oxidation of fatty acids and also stimulates the entire program of gluconeogenesis (20, 21). In all of these cases, PGC-1α increases respiration and many programs that go along with increased respiration in that particular tissue.
PGC-1α is abundantly expressed in the heart. A number of gain-of-function assays have shown both in vitro and in vivo that PGC-1α activates most genes of mitochondrial biology and stimulates both fatty acid oxidation and oxidative respiration in cardiac tissue (22–25). Conversely, we have recently shown that ablation of the PGC-1α gene caused important deficiencies in cardiac energy reserves and function (26). In the absence of PGC-1α, the expression of mitochondrial genes in the heart was suppressed, the activities of mitochondrial enzymes were aberrant, and ATP production was blunted. These energetic defects led to the inability to appropriately increase contractile function when the hearts were stimulated with adrenergic agents. Leone et al. (27) generated a similar mouse model, with a somewhat different ablation of the PGC-1α gene (27). Their work has shown that PGC-1α−/− animals have markedly blunted treadmill-running time and that cardiac function after treadmill running is diminished. Taken together, these studies demonstrate the vital role that PGC-1α plays in maintaining normal cardiac energetics and contractile function, especially in response to physiological stimuli.
A growing literature indicates that PGC-1α expression is altered in experimental models of heart disease (28–36). For example, PGC-1α and mitochondrial genes that are regulated by PGC-1α are repressed in rodent models of cardiac hypertrophy (30, 31, 37). This repression of PGC-1α may contribute importantly to deficiencies in ATP production and other types of dysfunction in failing hearts. To investigate the role that PGC-1α plays in the stressed heart, we subjected mice lacking PGC-1α to transverse aortic constriction (TAC), which subacutely increases the workload on the heart. We show here that PGC-1α−/− mice are profoundly predisposed to the development of heart failure in response to this stress. Moreover, induction of PGC-1α in cells in vitro reversed the inhibition of mitochondrial genes by adrenergic stimulation, suggesting that PGC-1α may be a potential therapeutic target for the treatment of heart failure.
Results
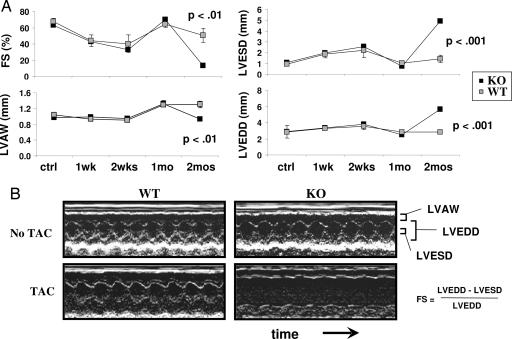
PGC-1α−/− mice and WT controls were subjected to TAC. TAC is a well established surgical procedure in which a suture is incompletely tightened around the transverse aorta, thereby increasing the resistance to blood flow out of the heart. This process leads to increased hemodynamic load on the heart, and greater force must be used by the heart to maintain normal cardiac output, eventually leading to cardiac hypertrophy. Three-month-old PGC-1α−/− and WT control mice underwent TAC and, over the ensuing 2 months, cardiac function was evaluated in vivo by using 2D echocardiography. Within the first 2 weeks after TAC, hearts in both WT and PGC-1α−/− animals developed mild dilation of the main cardiac chamber [measured as left ventricular end diastolic diameter (LVEDD); Fig. 1A], accompanied by mild reduction in contraction [measured as fractional shortening (FS); Fig. 1A]. Within 1 month, both WT and PGC-1α−/− animals compensated for the increased hemodynamic load by developing significant hypertrophy of the heart [measured as left ventricular anterior wall (LVAW) thickness], and the mild cardiac dilation observed in the first 2 weeks resolved (Fig. 1A). By 2 months, however, whereas the WT animals were able to sustain a compensated state, the PGC-1α−/− animals developed profound cardiac dysfunction. Hearts in PGC-1α−/− mice became markedly dilated, shown here with a doubling of LVEDD (Fig. 1 and Table 1). Simultaneously, the ability of these hearts to contract was severely blunted, as evidenced by a profound decrease in FS from ≈60% to ≈15% (Fig. 1 and Table 1).
Fig. 1.
Cardiac dysfunction in response to TAC in PGC-1α−/− (KO) mice. PGC-1α WT and KO mice underwent TAC as described in Methods. (A) Average values for the indicated echocardiographic parameter from WT and KO mice during the 2 months after TAC (n = 5 in each group). Error bars indicate SEM. (B) Sample 2D echocardiographs from WT and KO mice without TAC (Upper) or 2 months after TAC (Lower).
Table 1.
Cardiac dysfunction in response to TAC in PGC-1α−/− (KO) mice
| Echo parameter | WT |
KO |
WT TAC |
KO TAC |
P* | P† | P‡ | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Average | SEM | Average | SEM | Average | SEM | Average | SEM | ||||
| Heart rate, bpm | 670 | 18 | 644 | 25 | 671 | 66 | 524 | 27 | NS | NS | 0.07 |
| LVAW | 103 | 3 | 97 | 4 | 130 | 8 | 93 | 4 | NS | <0.004 | <0.007 |
| LVEDD | 279 | 15 | 288 | 8 | 285 | 21 | 565 | 21 | NS | NS | <0.0003 |
| LVESD | 94 | 16 | 107 | 6 | 143 | 34 | 493 | 22 | NS | NS | <0.0003 |
| FS | 68 | 4 | 63 | 2 | 51 | 9 | 13 | 1 | NS | NS | <0.004 |
PGC-1α WT and KO mice underwent TAC as described in Methods. Shown are average values for the indicated echocardiographic parameter from WT and KO mice without or 2 months after TAC (n = 5 in each group). LVAW, × 10−5 m. LVEDD, × 10−5 m; LVESD, × 10−5 m; FS, see Fig. 1 for derivation. NS, nonsignificant.
*KO vs. WT.
†WT TAC vs. WT.
‡KO TAC vs. KO.
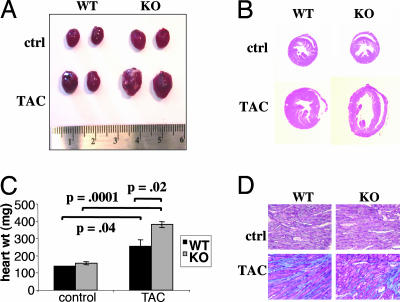
Animals were subsequently killed, and hearts were excised. Hearts from WT mice subjected to TAC were enlarged compared with controls (Fig. 2A), and their weights were increased ≈2-fold (Fig. 2C), consistent with cardiac hypertrophy. Hearts from PGC-1α−/− mice subjected to TAC, in contrast, more than tripled in weight (Fig. 2C) and were grossly enlarged (Fig. 2A), consistent with the profound dilation seen by 2D echocardiography (Fig. 1). Low-power magnifications of hematoxylin/eosin stains confirmed the marked dilation of PGC-1α−/− TAC hearts (Fig. 2B) compared with both WT TAC hearts and PGC-1α−/− hearts without TAC. Trichrome stains showed myofibrillar disarray and increased fibrosis in both the WT and −/− hearts after TAC (Fig. 2D), both characteristic of hypertrophic and failing hearts. There was no evidence of ongoing apoptosis, as determined by in situ TUNEL assay (data not shown).
Fig. 2.
PGC-1α−/− (KO) mice develop dilated cardiomyopathy in response to TAC. Hearts from the animals described in Fig. 1 were excised 2 months after TAC. (A) Photographs of sample WT and KO hearts with and without TAC. (B) Low-magnification views of hematoxylin/eosin stains of transverse sections of the same hearts as in A. (C) Average weights of hearts from all mice described in Fig. 1. (D) Sample higher-magnification views of trichrome stains from the same hearts as in B. Blue indicates the accumulation of extracellular matrix or fibrosis. (Magnifications: ×20.)
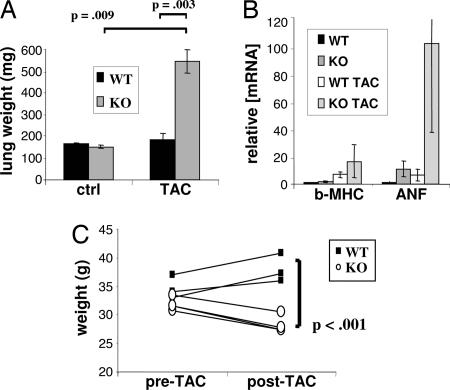
As in humans, cardiac dysfunction in mice leads to systemic manifestations. Prominent among these is congestion of the lungs and liver. As shown in Fig. 3A, the lungs in PGC-1α−/− mice nearly quadrupled in weight 2 months after TAC, likely the result of extreme congestion, as compared with only a moderate increase in WT TAC mice. mRNA expression of the β-myosin heavy chain and atrial natriuretic factor genes, markers of cardiac failure, was increased as much as 100-fold over WT control mice (Fig. 3B). Cachexia is another common end-stage consequence of heart failure. Two months after TAC, PGC-1α−/− mice were visibly emaciated (data not shown) and had significant drops in weight (Fig. 3C), unlike their WT counterparts. This weight loss occurred despite the increase in the “wet” weights of lungs (Fig. 3B) and liver (data not shown). Together, these data show that PGC-1α−/− mice develop profound cardiac dysfunction and clinical heart failure in response to TAC in vivo.
Fig. 3.
PGC-1α−/− (KO) mice develop clinical heart failure in response to TAC. (A) Average weights of lungs from WT and PGC-1α KO mice without or 2 months after TAC (n = 5 in each group). (B) mRNA expression of β-myosin heavy chain (β-MHC) and atrial natriuretic factor (ANF) in the same mice as in A. (C) Mouse body weight before and 2 months after TAC in WT and PGC-1α KO mice (n = 3). Error bars indicate SEM.
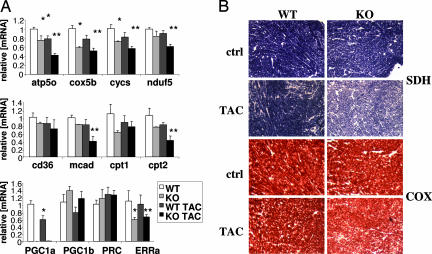
As mentioned above, the expression of PGC-1α and many of its target genes is repressed in various rodent models of cardiac failure (28–36). Consistent with these findings, PGC-1α was repressed by ≈30–40% in WT mice subjected to TAC, compared with WT controls (Fig. 4A Bottom). Target genes of PGC-1α, such as electron transport genes (atp5o, cox5b, and cycs) and fatty acid metabolism genes (mcad, cpt1, and cpt2), were similarly repressed (Fig. 4A Top and Middle). Importantly, when PGC-1α−/− hearts underwent TAC, the decrease in the expression of these genes was much more profound, decreasing by as much as 60–70% compared with WT controls and 30–40% compared with WT TAC animals, consistent with the dominant role PGC-1α plays in the regulation of these genes (Fig. 4A). Interestingly, the expression of these genes was also reduced compared with PGC-1α−/− animals that did not undergo TAC, suggesting that mechanism(s) in addition to repression of PGC-1α may be at play. To determine the consequences of the observed decreases in genetic expression, the activities of succinate dehydrogenase (SDH, electron transport complex II) and cytochrome oxidase (COX, electron transport complex IV) were evaluated in situ on transverse cardiac sections. As shown in Fig. 4B, the activities of both enzyme complexes were mildly repressed in the WT TAC and PGC-1α−/− hearts, compared with the unconstricted WT control (Fig. 4B Top Right and Bottom Left). In the PGC-1α−/− TAC hearts, however, the activities of both COX and SDH were markedly reduced (Fig. 4B Bottom Right), mirroring the effects seen on gene expression (Fig. 4A).
Fig. 4.
PGC-1α, genes regulated by PGC-1α, and activities of the electron transport chain, in WT and PGC-1α−/− hearts after TAC. (A) mRNA expression of representative genes of mitochondrial electron transport chain (cycs, nduf5, cox5b, and atp5o, Top), fatty acid metabolism (cd36, mcad, cpt1, and cpt2, Middle), and transcriptional regulation (PGC-1α, PGC-1β, PRC, and ERRα, Bottom) in a subset of the mice described in Fig. 1 (n = 3). ∗, P < 0.05 compared with WT. ∗∗, P < 0.05 compared with KO. (B) Views of sample frozen sections, cut transversely from the same hearts as in Fig. 2 and stained for SDH and COX activity. (Magnification: ×10.)
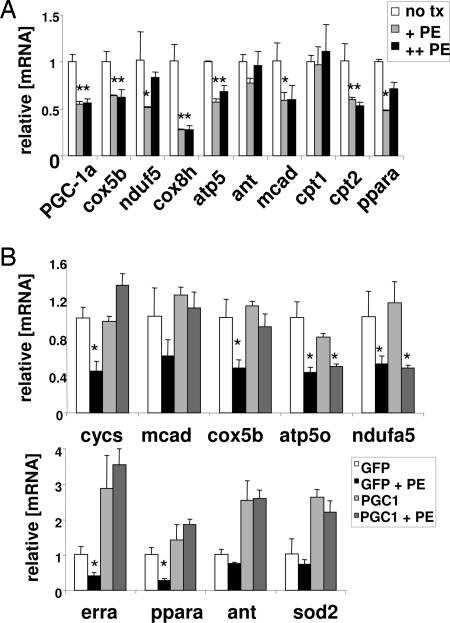
To better understand the mechanisms for this repression, we studied this process in cultured cells. Catecholamines are thought to be a key arm of the neurohormonal model of heart failure (2). In this model, cardiac stress leads to elevated neurohormonal tone, which in turn leads to further cardiac duress in a deleterious feed-forward loop. Treatment of neonatal rat ventricular myocytes (NRVMs) cells in vitro with the catecholamine phenylephrine (PE) causes a repression of expression of PPAR-α and targets of PPAR-α, recapitulating similar findings in whole animals (38). PE treatment of the cells also leads to a repression of fatty acid oxidation (38) and the development of cellular hypertrophy (39, 40), as is seen in vivo in response to TAC. To investigate the expression of PGC-1α under similar conditions, NRVMs were harvested and treated with 100 and 300 μM PE for 48 h. As shown in Fig. 5A, this treatment led to a significant 50% reduction in PGC-1α message. Moreover, genes targeted by PGC-1α, including genes of oxidative phosphorylation (e.g., electron transport genes, cox5b, atp5o, nudf5, cox8h) and fatty acid oxidation (e.g., fatty acid breakdown, mcad; transport to mitochondria, cpt2; and transcriptional regulation, ppara) were similarly repressed (Fig. 5A). Hence the treatment of primary cardiac cells with PE closely recapitulates some of the genetic changes observed in vivo in response to TAC (Fig. 4A).
Fig. 5.
PE inhibits PGC-1α and PGC-1α target genes in cardiomyocytes, and ectopic expression of PGC-1α rescues the inhibition of target genes. (A) mRNA expression of representative genes of mitochondrial electron transport chain (cox8h, nduf5, cox5b, atp5o, and ant) and fatty acid metabolism (mcad, cpt1, cpt2, and ppara) in primary NRVMs 48 h after treatment with PE (+, 100 μM; ++, 300 μM). ∗, P < 0.05 compared with untreated control. (B) NRVMs were infected (multiplicity of infection ≈5–10) with replication-incompetent adenovirus encoding for either GFP (as control) or PGC-1α and GFP. Forty-eight hours later, the cells were either left untreated or treated with 100 μM PE (+PE) for another 48 h. mRNA was then harvested. Expression of representative genes of mitochondrial electron transport chain (cycs, nduf5, cox5b, atp5o, and ant), mitochondrial protection against reactive oxygen species (sod2), and nuclear receptors involved in regulating these genes (ppara and erra) is indicated. Error bars indicate SEM.
These data suggested that the repression of genes of mitochondrial biology by PE may be in part mediated by repression of PGC-1α itself. To test this notion, the ability of PGC-1α to reverse the PE effects was studied by the introduction of exogenous PGC-1α by using adenoviral delivery. For these experiments, low titers of viruses, chosen to achieve little, if any, induction of genes targeted by PGC-1α when PE is not added, were used. Although higher amounts of virus can strongly induce the mcad gene in these cells (data not shown), the virus levels used here do not significantly induce mcad or genes of electron transport and induce certain genes like erra, ant, and sod2 only moderately (Fig. 5B). This amount of PGC-1α, however, was sufficient to nearly abolish the repression of most target genes by PE (Fig. 5B). These findings are consistent with the idea that PE represses genes of mitochondrial function, at least in part, by repression of PGC-1α. This suggests that therapeutic interventions aimed at moderately increasing PGC-1α function may reverse some of the deleterious remodeling of energetics that occurs in chronic heart failure.
Discussion
Earlier work had shown that hearts of mice lacking PGC-1α have reduced expression of many mitochondrial genes, a significant defect in ATP balance, and a blunted capacity to increase work output in response to an inotropic stimulus (26). However, the ability of hearts from mice mutant in PGC-1α to withstand a challenge in the form of pressure overload, such as seen in patients with hypertension or aortic stenosis, was unknown. We show here that mice lacking the PGC-1α transcriptional coactivator develop profound cardiac dysfunction in response to cardiac duress, as initiated by constriction of the transverse aorta. This is a commonly used model of pressure overload in rodents and other animals, in which the constriction of the aorta leads to greater resistance to cardiac outflow. The cardiac dysfunction we observe is profound and accompanied by extensive congestion of the lungs and the development of cachexia, indicating significant clinical heart failure. Importantly, the fact that PGC-1α−/− hearts do not immediately fail and are able to first develop hypertrophy in response to TAC indicates that PGC-1α is not critical for the acute response to the hemodynamic load. However, the subsequent development of profound cardiac dysfunction and failure in PGC-1α−/− mice suggests that PGC-1α plays an important role during the remodeling process. Together, the findings described here demonstrate that endogenous levels of PGC-1α are critical for the heart to defend effectively against this type of cardiac duress.
PGC-1α and a number of genes known to be regulated by PGC-1α, such as those encoding for electron transport chain subunits or proteins involved in fatty acid import and oxidation, are repressed in response to aortic banding (Fig. 4 and refs. 30, 31, and 37). Importantly, the repression is even more marked in hearts from PGC-1α−/− mice after aortic banding, compared with either unbanded −/− mice or banded WT controls (Fig. 4). This finding suggests that a threshold of mitochondrial activity might be required to cope with chronic hemodynamic load, and that the combination of PGC-1α repression and aortic banding hastens the falling below that threshold. Repression of PGC-1α and genes regulated by PGC-1α is also observed in other models of heart failure, even in cases where mitochondrial defects or pressure overload are not the primary etiology of cardiac dysfunction (28–36). Hence, the data suggest that, even in these cases, repression of PGC-1α may be causally involved in the development of heart failure.
Numerous groups have shown that hypertrophic and failing hearts change substrate preference to glucose, at the expense of fatty acids (reviewed in refs. 41–43). Evidence exists to suggest that this is an adaptive response that protects the heart from further damage (e.g., ref. 44), although the mechanism remains unclear. It will therefore be interesting to evaluate the energetic profile of hearts explanted from PGC-1α−/− mice after aortic banding. Although substrate use in PGC-1α−/− hearts has not been evaluated, it is likely that fatty acid use is decreased, because numerous genes of fatty acid import and use are known targets of PGC-1α and are repressed in −/− hearts, both with and without aortic banding. It is also important to recognize that the absence of PGC-1α in tissues other than the heart may have neurohormonal or humoral effects on cardiac function. It will therefore also be interesting to evaluate cardiac function in mice where the PGC-1α locus is deleted specifically in cardiomycytes.
If the absence of PGC-1α hastens the development of heart failure, and if PGC-1α is frequently repressed in models of heart failure, it is possible that increasing PGC-1α may be protective against certain cardiac insults. It is important to note, however, that massive induction of PGC-1α in the heart in vivo increased mitochondrial content to such an extent that the myofibrillar apparatus was displaced and cardiomyopathy ensued (23, 25). Hence, it will be necessary to increase PGC-1α activity in a more moderate fashion. Treating cardiac cells in tissue culture with the catecholamine drug PE represses the expression of PGC-1α and many of the genes it regulates, akin to the response in vivo to chronic cardiac duress. We show here that introduction of relatively low amounts of ectopic PGC-1α by low-titer adenoviral delivery rescues the inhibition of numerous genes of energy control. This suggests that the repression of PGC-1α at least in part explains the repression of these genes, and the consequent energetic deficit. Importantly, this also suggests that moderately increasing PGC-1α activity in vivo may have therapeutic benefit. It will therefore be of great interest to develop small molecules with pharmaceutical potential that can increase PGC-1α activity in vivo.
Methods
Animals.
All animal experiments were performed according to procedures approved by the Institutional Animal Care and Use Committee at Harvard Medical School. Mice were maintained on a standard rodent chow diet with 12-h light and dark cycles.
Surgeries.
Constriction of the transverse thoracic aorta was performed on 3-month-old male mice as described (45). In brief, mice were anesthetized, intubated, and placed on a respirator. Midline sternotomy was performed, the aorta was visualized, and 6.0 Prolene suture was placed around the aorta distal to the brachiocephalic artery. The suture was tightened around a blunt 27-gauge needle placed adjacent to the aorta. The needle was then removed, and the chest and overlying skin were closed. Over the ensuing 2 months, echocardiography was performed at regular intervals on awake mice to evaluate cardiac function, using a 15-Mhz linear-array probe and a Sonos 4500 ultrasonograph (Hewlett–Packard). WT and PGC-1α−/− littermates (i.e., age-matched) were used as unbanded controls. After 2 months, the animals were killed. The hearts and lungs were weighed, and the hearts were frozen in liquid nitrogen immediately upon death. Cardiac hematoxylin/eosin and trichrome stains were performed by American HistoLabs (Gaithersburg, MD). SDH and COX in situ activity stains were performed by the Boston Children’s Hospital Core Pathology Services.
Cells and Reagents.
NRVMs were harvested from 2- to 3-day-old pups as described (46) and maintained in DMEM/10% horse serum/5% FBS for 3 days before further treatment. Cells were then infected with the indicated adenovirus 48 h before treatment with PE at the indicated concentrations for an additional 48 h. Adenovirus expressing PGC-1α has been described (14).
Genetic Expression Studies.
Total RNAs were isolated by using the Trizol method (Invitrogen). Samples were reverse-transcribed (Invitrogen), and quantitative real-time PCRs were performed in the presence of fluorescent dye (Cybergreen; Bio-Rad). DNA product of the expected size was confirmed for each primer pair. All results are expressed as means ± SEM. Two-tailed independent Student’s t tests were used to determine all P values.
Acknowledgments
This work was supported by National Institutes of Health Grants HL079172-01 (to Z.A.), HL077543 (to A.R. and M.N.), DK54477 (to B.M.S.), and DK61562 (to B.M.S.).
Abbreviations
- PPAR-γ
peroxisome proliferator-activated receptor-γ
- PGC-1α
PPAR-γ coactivator 1α
- TAC
transverse aortic constriction
- FS
fractional shortening
- LVEDD
left ventricular end diastolic diameter
- LVAW
left ventricular anterior wall
- LVESD
left ventricular end systolic diameter
- SDH
succinate dehydrogenase
- COX
cytochrome oxidase
- NRVM
neonatal rat ventricular myocyte
- PE
phenylephrine.
Footnotes
Conflict of interest statement: No conflicts declared.
References
- 1.Thom T., Haase N., Rosamond W., Howard V. J., Rumsfeld J., Manolio T., Zhang Z. J., Flegal K., O’Donnell C., Kittner S., et al. Circulation. 2006;113:e85. doi: 10.1161/CIRCULATIONAHA.105.171600. [DOI] [PubMed] [Google Scholar]
- 2.Jessup M., Brozena S. N. Engl. J. Med. 2003;348:2007–2018. doi: 10.1056/NEJMra021498. [DOI] [PubMed] [Google Scholar]
- 3.Pfeffer M. A., Braunwald E. Circulation. 1990;81:1161–1172. doi: 10.1161/01.cir.81.4.1161. [DOI] [PubMed] [Google Scholar]
- 4.Ingwall J. S., Weiss R. G. Circ. Res. 2004;95:135–145. doi: 10.1161/01.RES.0000137170.41939.d9. [DOI] [PubMed] [Google Scholar]
- 5.Taegtmeyer H., Wilson C. R., Razeghi P., Sharma S. Ann. N.Y. Acad. Sci. 2005;1047:208–218. doi: 10.1196/annals.1341.019. [DOI] [PubMed] [Google Scholar]
- 6.Taegtmeyer H. Circulation. 2004;110:894–896. doi: 10.1161/01.CIR.0000139340.88769.D5. [DOI] [PubMed] [Google Scholar]
- 7.Huss J. M., Kelly D. P. J. Clin. Invest. 2005;115:547–555. doi: 10.1172/JCI200524405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.van Bilsen M. Ann. N.Y. Acad. Sci. 2004;1015:238–249. doi: 10.1196/annals.1302.020. [DOI] [PubMed] [Google Scholar]
- 9.Stanley W. C., Recchia F. A., Lopaschuk G. D. Physiol. Rev. 2005;85:1093–1129. doi: 10.1152/physrev.00006.2004. [DOI] [PubMed] [Google Scholar]
- 10.Ingwall J. S. ATP and the Heart. Boston: Kluwer; 2003. [Google Scholar]
- 11.Kelly D. P., Strauss A. W. N. Engl. J. Med. 1994;330:913–919. doi: 10.1056/NEJM199403313301308. [DOI] [PubMed] [Google Scholar]
- 12.Kelly D. P., Scarpulla R. C. Genes Dev. 2004;18:357–368. doi: 10.1101/gad.1177604. [DOI] [PubMed] [Google Scholar]
- 13.Lin J., Handschin C., Spiegelman B. M. Cell Metab. 2005;1:361–370. doi: 10.1016/j.cmet.2005.05.004. [DOI] [PubMed] [Google Scholar]
- 14.Puigserver P., Wu Z., Park C. W., Graves R., Wright M., Spiegelman B. M. Cell. 1998;92:829–839. doi: 10.1016/s0092-8674(00)81410-5. [DOI] [PubMed] [Google Scholar]
- 15.Koves T. R., Li P., An J., Akimoto T., Slentz D., Ilkayeva O., Dohm G. L., Yan Z., Newgard C. B., Muoio D. M. J. Biol. Chem. 2005;280:33588–33598. doi: 10.1074/jbc.M507621200. [DOI] [PubMed] [Google Scholar]
- 16.Cluberton L. J., McGee S. L., Murphy R. M., Hargreaves M. J. Appl. Physiol. 2005;99:1359–1363. doi: 10.1152/japplphysiol.00197.2005. [DOI] [PubMed] [Google Scholar]
- 17.Baar K., Wende A. R., Jones T. E., Marison M., Nolte L. A., Chen M., Kelly D. P., Holloszy J. O. FASEB J. 2002;16:1879–1886. doi: 10.1096/fj.02-0367com. [DOI] [PubMed] [Google Scholar]
- 18.Norrbom J., Sundberg C. J., Ameln H., Kraus W. E., Jansson E., Gustafsson T. J. Appl. Physiol. 2004;96:189–194. doi: 10.1152/japplphysiol.00765.2003. [DOI] [PubMed] [Google Scholar]
- 19.Lin J., Wu H., Tarr P. T., Zhang C. Y., Wu Z., Boss O., Michael L. F., Puigserver P., Isotani E., Olson E. N., et al. Nature. 2002;418:797–801. doi: 10.1038/nature00904. [DOI] [PubMed] [Google Scholar]
- 20.Herzig S., Long F., Jhala U. S., Hedrick S., Quinn R., Bauer A., Rudolph D., Schutz G., Yoon C., Puigserver P., et al. Nature. 2001;413:179–183. doi: 10.1038/35093131. [DOI] [PubMed] [Google Scholar]
- 21.Yoon J. C., Puigserver P., Chen G., Donovan J., Wu Z., Rhee J., Adelmant G., Stafford J., Kahn C. R., Granner D. K., et al. Nature. 2001;413:131–138. doi: 10.1038/35093050. [DOI] [PubMed] [Google Scholar]
- 22.Huss J. M., Kopp R. P., Kelly D. P. J. Biol. Chem. 2002;277:40265–40274. doi: 10.1074/jbc.M206324200. [DOI] [PubMed] [Google Scholar]
- 23.Lehman J. J., Barger P. M., Kovacs A., Saffitz J. E., Medeiros D. M., Kelly D. P. J. Clin. Invest. 2000;106:847–856. doi: 10.1172/JCI10268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Vega R. B., Huss J. M., Kelly D. P. Mol. Cell. Biol. 2000;20:1868–1876. doi: 10.1128/mcb.20.5.1868-1876.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Russell L. K., Mansfield C. M., Lehman J. J., Kovacs A., Courtois M., Saffitz J. E., Medeiros D. M., Valencik M. L., McDonald J. A., Kelly D. P. Circ. Res. 2004;94:525–533. doi: 10.1161/01.RES.0000117088.36577.EB. [DOI] [PubMed] [Google Scholar]
- 26.Arany Z., He H., Lin J., Hoyer K., Handschin C., Toka O., Ahmad F., Matsui T., Chin S., Wu P. H., et al. Cell Metab. 2005;1:259–271. doi: 10.1016/j.cmet.2005.03.002. [DOI] [PubMed] [Google Scholar]
- 27.Leone T. C., Lehman J. J., Finck B. N., Schaeffer P. J., Wende A. R., Boudina S., Courtois M., Wozniak D. F., Sambandam N., Bernal-Mizrachi C., et al. PLOS. 2005;3:e101. doi: 10.1371/journal.pbio.0030101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sano M., Wang S. C., Shirai M., Scaglia F., Xie M., Sakai S., Tanaka T., Kulkarni P. A., Barger P. M., Youker K. A., et al. EMBO J. 2004;23:3559–3569. doi: 10.1038/sj.emboj.7600351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Czubryt M. P., McAnally J., Fishman G. I., Olson E. N. Proc. Natl. Acad. Sci. USA. 2003;100:1711–1716. doi: 10.1073/pnas.0337639100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lehman J. J., Kelly D. P. Clin. Exp. Pharmacol. Physiol. 2002;29:339–345. doi: 10.1046/j.1440-1681.2002.03655.x. [DOI] [PubMed] [Google Scholar]
- 31.Garnier A., Fortin D., Delomenie C., Momken I., Veksler V., Ventura-Clapier R. J. Physiol. (London) 2003;551:491–501. doi: 10.1113/jphysiol.2003.045104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Feingold K., Kim M. S., Shigenaga J., Moser A., Grunfeld C. Am. J. Physiol. 2004;286:E201–E207. doi: 10.1152/ajpendo.00205.2003. [DOI] [PubMed] [Google Scholar]
- 33.Hsieh Y. C., Yang S., Choudhry M. A., Yu H. P., Bland K. I., Schwacha M. G., Chaudry I. H. Am. J. Physiol. 2006;290:H416–H423. doi: 10.1152/ajpheart.00865.2005. [DOI] [PubMed] [Google Scholar]
- 34.Hsieh Y. C., Yang S., Choudhry M. A., Yu H. P., Rue L. W., Bland K. I., Chaudry I. H. Am. J. Physiol. 2005;289:H2665–H2672. doi: 10.1152/ajpheart.00682.2005. [DOI] [PubMed] [Google Scholar]
- 35.van den Bosch B. J., van den Burg C. M., Schoonderwoerd K., Lindsey P. J., Scholte H. R., de Coo R. F., van Rooij E., Rockman H. A., Doevendans P. A., Smeets H. J. Cardiovasc. Res. 2005;65:411–418. doi: 10.1016/j.cardiores.2004.10.025. [DOI] [PubMed] [Google Scholar]
- 36.Suliman H. B., Welty-Wolf K. E., Carraway M., Tatro L., Piantadosi C. A. Cardiovasc. Res. 2004;64:279–288. doi: 10.1016/j.cardiores.2004.07.005. [DOI] [PubMed] [Google Scholar]
- 37.Finck B. N., Lehman J. J., Barger P. M., Kelly D. P. Cold Spring Harbor Symp. Quant. Biol. 2002;67:371–382. doi: 10.1101/sqb.2002.67.371. [DOI] [PubMed] [Google Scholar]
- 38.Barger P. M., Brandt J. M., Leone T. C., Weinheimer C. J., Kelly D. P. J. Clin. Invest. 2000;105:1723–1730. doi: 10.1172/JCI9056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Simpson P. Circ. Res. 1985;56:884–894. doi: 10.1161/01.res.56.6.884. [DOI] [PubMed] [Google Scholar]
- 40.Simpson P., McGrath A., Savion S. Circ. Res. 1982;51:787–801. doi: 10.1161/01.res.51.6.787. [DOI] [PubMed] [Google Scholar]
- 41.Lopaschuk G. D., Rebeyka I. M., Allard M. F. Circulation. 2002;105:140–142. [PubMed] [Google Scholar]
- 42.Sambandam N., Lopaschuk G. D., Brownsey R. W., Allard M. F. Heart Fail. Rev. 2002;7:161–173. doi: 10.1023/a:1015380609464. [DOI] [PubMed] [Google Scholar]
- 43.Depre C., Vanoverschelde J. L., Taegtmeyer H. Circulation. 1999;99:578–588. doi: 10.1161/01.cir.99.4.578. [DOI] [PubMed] [Google Scholar]
- 44.Liao R., Jain M., Cui L., D’Agostino J., Aiello F., Luptak I., Ngoy S., Mortensen R. M., Tian R. Circulation. 2002;106:2125–2131. doi: 10.1161/01.cir.0000034049.61181.f3. [DOI] [PubMed] [Google Scholar]
- 45.Hill J. A., Karimi M., Kutschke W., Davisson R. L., Zimmerman K., Wang Z., Kerber R. E., Weiss R. M. Circulation. 2000;101:2863–2869. doi: 10.1161/01.cir.101.24.2863. [DOI] [PubMed] [Google Scholar]
- 46.Disch D. L., Rader T. A., Cresci S., Leone T. C., Barger P. M., Vega R., Wood P. A., Kelly D. P. Mol. Cell. Biol. 1996;16:4043–4051. doi: 10.1128/mcb.16.8.4043. [DOI] [PMC free article] [PubMed] [Google Scholar]