Abstract
High levels of circulating immune complexes containing tumor-associated antigens are associated with a poor prognosis for individuals with cancer. The ability of B cells, previously exposed to tumor-associated antigens, to promote both in vitro and in vivo tumor growth formed the rationale to evaluate the mechanism by which immune complexes may promote tumor growth. In elucidating this mechanism, FcγRI expression by tumor cells was characterized by flow cytometry, polymerase chain reaction, and sequence analysis. Immune complexes containing shed tumor antigen and anti-shed tumor antigen Ab cross-linked FcγRI-expressing tumor cells, which resulted in an induction of tumor cell proliferation and of shed tumor antigen production. Use of selective tyrosine kinase inhibitors demonstrated that tumor cell proliferation induced by immune complex cross-linking of FcγRI is dependent on the tyrosine kinase signal transduction pathway. A selective inhibitor of phosphatidylinositol-3 kinase also inhibited this induction of tumor cell proliferation. These findings support a role for immune complexes and FcγRI expression by tumor cells in augmentation of tumor growth and a metastatic phenotype.
Keywords: B lymphocytes, tumor, cellular proliferation, Fc receptors, signal transduction
Introduction
Despite the activation of lymphocytes such as NK cells, T cells, and B cells by tumor antigens, the immune system is often inefficient in both eliminating the tumor and preventing its progressive growth. There have been several proposed mechanisms by which tumor cells may evade immune attack by inducing apoptosis of lymphocytes, including by expression of Fas ligand, and by some tumor-associated antigens such as RCAS1 and Muc-1 [1–4]. Additionally, it has also been proposed that B cells may be involved in lymphocyte suppression, thereby allowing tumor cells to escape mechanisms of T cell-mediated cytotoxicity [5,6]. Further, we have identified a novel mechanism by which B cells contribute to the promotion of tumor growth, invasion, and metastasis in cooperation with immune effector cells [7,8]. Briefly, B cells are activated by shed tumor-associated antigens in a humoral immune response resulting in the production of IgG antibody against a repeated carbohydrate epitope, and the shed tumor-associated antigen complexes with this IgG antibody in forming polyvalent immune complexes. The polyvalent immune complexes cross-link FcγR on FcγR-expressing immune effector cells, such as monocytes and interferon-gamma-activated polymorphonuclear cells, in a process that results in release in the tumor stroma of soluble mediators, which promote tumor cell invasion, angiogenesis, and metastasis. Thus, there is a mechanism by which B cells and antitumor antibodies, as mediated by immune effector cells, can induce tumor progression [7]. A direct antibody-tumor cell interaction has been proposed to involve IgM, which binds tumor cell-associated antigen, thereby coating and masking the tumor cells, protecting them from attack by cytotoxic lymphocytes and allowing for progressive growth of the tumor [9]. Independent of these discoveries, it has been proposed that nonlymphoid tumors, experimental and human, express FcγR identified as FcγRIIB1 [10]. Although the mechanism(s) by which FcγRIIB1 may confer augmentation of tumorigenicity remains unknown, it was suggested that complexed IgG, bound by FcγRIIB1 on tumor cells, could sterically block immune effector mechanisms from eliminating the tumor cells.
We have observed that mucin-secreting tumor cells appeared to be able to interact directly with B cells to modulate tumor cell behavior. Here, we characterized this interaction and found that the B cells require prior exposure to tumor-associated antigens, that a tumor cell receptor for mediating this interaction is FcγRI, and that it is immune complex binding and cross-linking of this receptor, which induces both tumor cell proliferation and production of mucin with polyvalent sialyl-Tn (sTn) antigen (sTn-mucin).
Materials and Methods
Cell Lines
T-47D (human ductal breast carcinoma), SW620 (human colon carcinoma), B16F1 (C57BL murine melanoma), and U937 (monocytic) cell lines were obtained from ATCC (Rockville, MD). The Met 129 (methylcholanthrene-induced mammary carcinoma in C3H mice) cell line was kindly provided by Dr J. Vaage (Roswell Park Memorial Institute, Buffalo, NY).
Mice
C3H mice and athymic nude C3H mice (nu/nu) were purchased from Harlan Sprague-Dawley. The use of experimental animals, and animal care, was in accordance with both the NIH and European Community principles of laboratory animal care.
In Vivo Tumor Growth
B cells were isolated as described previously [7] using anti-B220 mAb, selected using magnetic beads coated with sheep anti-rat IgG (Dynal), and cultured in RPMI. Flow cytometry was used to confirm that greater than 90% of these isolated cells were B220 +. C3H mice or athymic nude C3H mice were injected intrasplenically with 106 Met 129 tumor cells. Starting 5 days later, the mice were injected every 2 days for a 14-day period with either B cells (50,000 cells/100 µl) isolated from normal mouse spleen, or isolated from spleens of tumor-bearing mice, or isolated from tumors of tumor-bearing mice, or received no injection of B cells. After the 14-day period, liver metastasis and spleen tumor was scored by assigning a numerical value where “0” represents no metastasis (as proven by histology); “1” represents a single metastases in one liver lobe; “2” represents several metastases in one liver lobe; “3” represents multiple metastases involving several liver lobes; and “4” represents greater than 50% of the liver mass is tumor. The degree of spleen tumor was scored by assigning a numerical value, where: “0” represents no tumor (as proven by histology); “1” represents a small tumor; “2” represents a large tumor with visible spleen; “3” represents a large tumor in which only a portion of the spleen shape is visible; and “4” represents a tumor so large that the spleen shape is no longer visible. Each individual score was squared, and then the values for the group being analyzed were averaged to obtain a single value representative of that group.
In Vitro Tumor Growth in Presence of B Cells
Tumor infiltrating CD8+ lymphocytes were isolated from dispersed tumor tissue from C3H mice using magnetic separation techniques; and flow cytometry was used to demonstrate that greater than 90% of these isolated cells were CD8+. Ten thousand Met 129 cells were cultured in 1.5 ml of tissue culture medium supplemented with 10% fetal bovine serum per well in 24-well plates alone, or in the presence of either 10,000 CD8+ tumor-infiltrating T lymphocytes (TIL), 10,000 tumor-infiltrating B lymphocytes (B-TIL), or 10,000 B-TIL and 10,000 CD8+ TIL cells. After 72 hours of coincubation in monolayer culture, Met 129 tumor cell growth was quantitated by counting Alcian blue-stained tumor cells.
FcγR Expression
B16F1 tumor cells, T-47D tumor cells, and Met 129 tumor cells were each cultured as an adherent monolayer (37°C+5% CO2) in serum-free tissue culture medium; SW620 tumor cells were cultured in suspension with agitation (37°C) in serum-free tissue culture medium. The respective tumor cells were harvested, washed twice with PBS, centrifuged at 1200 rpm for 10 minutes, and then resuspended in PBS without calcium and magnesium. Cells were counted, and cell viability was checked using trypan blue exclusion dye. Cells were aliquoted at a concentration of 1 million cells per tube, and resuspended in 50 µl of an antibody solution selected from fluorescein isothiocyanate (FITC)-labeled anti-FcγRI (CD64), phycoerythrin (PE)-labeled anti-FcγRII (CD32), R-phycoerythrin-cyanin 5.1 (PC5)-labeled anti-FcγRIII (CD16), PE-labeled isotype control IgG1 mAb, or PE-labeled isotype control IgG2 mAb (fluorescent dye-labeled mAbs all obtained from Immunotech, Coulter). After staining for 30 minutes on ice, 800 µl of PBS was added, the cells were pelleted by centrifugation at 3000 rpm for 3 minutes, and the supernatants were removed. The cell pellets were then resuspended in 250 to 350 µl of PBS for analysis by flow cytometry. Each value was corrected for nonspecific binding by subtracting the value of the respective isotype control. For analysis of FcγRI expression at the mRNA level, RNA isolation, RT/PCR conditions, primers, PCR conditions for cDNA amplification, and analysis by agarose gel electrophoresis were performed as described by Ernst et al. [11]. U937 monocytic cell RNA was used as a control for expression of all 3 FcγRI genes. FcγRI-specific primers included 5′-ACACCACAAAGGCAGTGA-3′ and reverse primer 5′-CACCCAGAGAACAGTGTT-3′. DNA sequencing was performed commercially.
Immune Complex Induction of Tumor Proliferation
Either B72.3 mAb (IgG1; ATCC) or HB-STn mAb (IgG1; Dako) were used as the anti-sTn mAb. For initial induction experiments, sTn-mucin-secreting tumor cells were aliquoted into 96-well plates at 1000 cells per well. After culture overnight (37°C in 5% CO2), the culture medium from each well was removed, and replaced with fresh serum-free tissue culture medium alone or with anti-sTn mAb (HB-STn) to a final concentration of either 10, 1, 0.1, or 0.01 µg/ml, for a total of six wells per control or antibody dilution. In blocking FcγRI [12] experiments, 100-fold excess of Fc fragments (prepared from papain digestion of B72.3 mAb) was added to each antibody dilution and control. After incubating the 96-well plates for an additional 24 hours, proliferation was assessed by removing the culture medium, adding MTT (3-[4,5-dimethyl-thiazol-2-yl]-2,5-diphenyl-tetrazolium bromide; thiazolyl blue) dye to the cells in each well (final concentration of 0.5 mg/ml), incubating the plate for 4 hours, solubilizing the dye with 0.1 N HCl in absolute isopropanol, and measuring the absorbance at 570 and 690 nm. Cell proliferation was expressed as the absorbance measured at 570 nm minus the absorbance at 690 nm. In the case of non-sTn-mucin secreting B16F1, the same procedure was followed, except that the cells were incubated for a 72-hour period, and every 24 hours the culture medium was removed from each well and replaced with either fresh serum-free tissue culture medium (“without mucin”) or with 90 µl of fresh serum-free tissue culture medium containing 10 µl of cell-free tissue culture supernatant containing shed sTn-mucin from cultured T-47D tumor cells (“with mucin“). In assaying for inhibition of induction of tumor proliferation using tyrosine kinase inhibitors (genistein, herbimycin A; Sigma, St. Louis, MO) or PI-3 kinase inhibitor (wortmannin; Sigma), 1000 T-47D tumor cells were plated as described above, followed by treatment with either: serum-free tissue culture medium alone (1 hour); the medium containing genistein (final concentration 100 µM; for 1 hour); the medium containing herbimycin A (final concentration 10 µM; for 1 hour); or the medium containing wortmannin (final concentration of 500 nM; for 10 minutes). After the appropriate treatment period, the cells were washed with fresh serum-free tissue culture medium, and then incubated with either fresh serum-free tissue culture medium (controls) or the medium containing anti-sTn mAb (HB-STn; 0.1 µg/ml) for a 24-hour culture period, and proliferation was determined by the addition of MTT dye.
Immune Complex Induction of sTn-Mucin Production
For initial induction experiments, T-47D tumor cells were aliquoted into 96-well plates at 1000 cells per well. After culture overnight (37°C in 5% CO2), the culture medium from each well was removed, and replaced with fresh serum-free tissue culture medium alone or with anti-sTn mAb (B72.3) to a final concentration of either 10, 1, 0.1, or 0.01 ng/ml, for a total of six wells per control or antibody dilution. After incubating the 96-well plates for an additional 2 hours, the culture medium was removed and quantitated for sTn-mucin by ELISA [7] using color development with 3,3′,5,5′-tetramethylbenizidine (TMB; Gibco BRL), and absorbance readings at 450 nm with a correction at 570 nm. sTn-mucin concentration was expressed as the corrected absorbance reading. Cell proliferation was also assessed at the same time by treating the cells with MTT dye as described above. Thus, the absorbance reading for the sTn-mucin produced in a well was divided by the absorbance reading of the MTT for the same well, so as to express sTn-mucin production relative to the number of tumor cells. For assays for inhibition of induction of sTn-mucin production using tyrosine kinase inhibitors (genistein, herbimycin A) or PI-3 kinase inhibitor (wortmannin), 1000 T-47D tumor cells were plated as described above, followed by treatment with either: serum-free tissue culture medium alone (1 hour); the medium containing genistein (final concentration 100 µM; for 1 hour); the medium containing herbimycin A (final concentration 10 µM; for 1 hour); or the medium containing wortmannin (final concentration of 500 nM; for 10 minutes). After the appropriate treatment period, the cells were washed with fresh serum-free tissue culture medium, and then incubated with either fresh serum-free tissue culture medium (controls) or the medium containing anti-sTn mAb (B72.3; 0.1 µg/ml) for a 24-hour culture period, and then determination of sTn-mucin production by ELISA.
Statistical Analysis
Data from each experimental group were subjected to an analysis of normality and variance. Differences between comparative experimental groups were analyzed for statistical significance using Student's t test.
Results
B Cells with Prior Exposure to Tumor-Associated Antigens Promoted In Vivo Tumor Growth
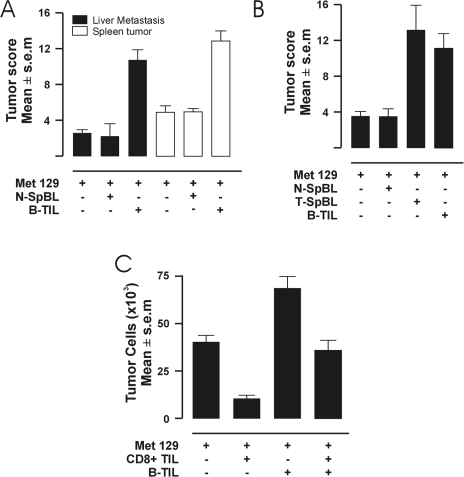
To investigate whether B cells first require exposure to tumor-associated antigens to be able to interact with tumors in promoting tumor growth in vivo, C3H mice were injected intrasplenically with high mucin-secreting, Met 129 murine mammary carcinoma cells. The mice were then divided into three groups: one group received B cells isolated from spleens of isogeneic mice in which no tumor was induced; a second group received B cells isolated from the tumor tissue of isogeneic mice that had been implanted with Met 129 cells into mammary pads; and a third group served as a control, which did not receive any exogenous B cells. Nineteen days after the tumor challenge, the mice were scored for liver metastasis and spleen tumor growth. B cells isolated from tumor tissue of tumor-bearing mice (B-TIL) promoted statistically significant tumor growth and metastasis in vivo compared with no exogenous B cells (as a control), and to B cells isolated from spleens of non-tumor-bearing mice spleen (N-SpBL) having similar scores compared with the control (Figure 1A). These results indicate that to be able to interact with tumor cells in promoting tumor growth in vivo, B cells must be exposed to tumor-associated antigens. To determine whether the promotion of tumor growth was a result of an exclusive interaction between B cells and tumor cells, as opposed to an interaction in which B cells also require a T-cell failure, the experiment was repeated in T cell-deficient mice. Athymic nude-beige mice were injected intrasplenically with mucin-secreting, Met 129 tumor cells, and then divided into three groups: a group receiving B cells isolated from spleens of isogeneic, non-tumor-bearing mice; a group receiving B cells isolated from spleens of isogeneic, Met 129 tumor-bearing mice; and a group receiving B cells isolated from the tumor tissue of isogeneic, Met 129 tumor-bearing mice. In the athymic nude-beige mice (without T cells or NK cells), B cells isolated from tumor tissue (B-TIL) or spleens (T-SpBL) of tumor-bearing mice promoted statistically significant tumor growth and metastasis in vivo compared with B cells isolated from spleens of non-tumor-bearing mice (N-SpBL) (Figure 1B). Thus, B cells in the spleens of tumor-bearing mice (i.e., B cells found outside the tumor) can also promote tumor growth and, additionally, that promotion is not due to an impairment of antitumor activity of T cells or NK cells.
Figure 1.
(A–C) Promotion of tumor growth by B cells. (A) In C3H mice, liver metastasis scores (black boxes) and spleen tumor scores (white boxes) from injection with either Met 129 tumor cells alone, Met 129 and B cells isolated from spleens of non-tumor-bearing mice spleen (N-SpBL), or Met 129 and B cells isolated from tumor tissue of Met 129 tumor-bearing mice (B-TIL). Averages of six mice per group. The means of scores from mice receiving B-TIL is significantly greater than the means of the remaining groups (P<0.01). (B) In nude mice, spleen tumor scores from injection with either Met 129 tumor cells alone, Met 129 and N-SpBL, Met 129 and B cells isolated from spleens of tumor-bearing mice spleen (T-SpBL), or Met 129 and B-TIL. Averages of six mice per group. The comparison of means of scores from mice receiving B-TIL and from mice receiving T-SpBL does not show a statistical difference. However, both are significantly greater than the means of the remaining groups (P<0.01). (C) In vitro tumor cell growth (number of tumor cells) of Met 129 tumor cells alone, Met 129 mixed with CD8+ T lymphocytes isolated from the tumor tissue of Met 129 tumor-bearing mice (CD8+ TIL), Met 129 and B-TIL, or Met 129 with CD8+ TIL and B-TIL. Averages of six wells per experiment. The mean of well values with CD8+ TIL is significantly lower than the control (P<0.01). The means of well values with B-TIL are significantly greater than the means of the remaining combinations (P<0.01). The mean of well values with both CD8+TIL and B-TIL is statistically identical to that of the control.
B Cells with Prior Exposure to Tumor-Associated Antigens Promoted In Vitro Tumor Growth
To further test this interpretation, we tested whether tumor cells and B cells (B-TIL) could interact directly to promote tumor cell growth in vitro. Met 129 tumor cells were cultured either alone, in the presence of B-TIL, in the presence of CD8+ T lymphocytes isolated from the tumor tissue of Met 129 tumor-bearing mice (CD8+ TIL), or in the presence of both B-TIL and CD8+ TIL cells; and then tumor cell growth was quantitated by counting the number of adherent Met 129 cells. CD8+ TIL cells cocultured with Met 129 cells (Figure 1C; Met129 +, CD8+ TIL+, B-TIL-) resulted in a statistically significant reduction in tumor cell growth, and thus appeared to effect growth inhibition or tumor cell death, when compared to the growth of the control of Met 129 cells alone (Figure 1C; Met 129 +, CD8+ TIL-, B-TIL-). A slight reduction in tumor cell growth, compared with that of the control, was observed when Met 129 cells were cocultured in the presence of B-TIL and CD8+ TIL cells (Figure 1C; Met 129+, CD8+ TIL+, B-TIL+). In contrast, a statistically significant increase in tumor cell growth was observed, compared with that of the control, when B-TIL were cocultured with Met 129 cells (Figure 1C; Met 129+, CD8+ TIL-, B-TIL+). These results indicate that tumor cell growth can be promoted when tumor cells interact directly, without any other cellular participation, with B cells activated by tumor-associated antigens (“specific recognition”).
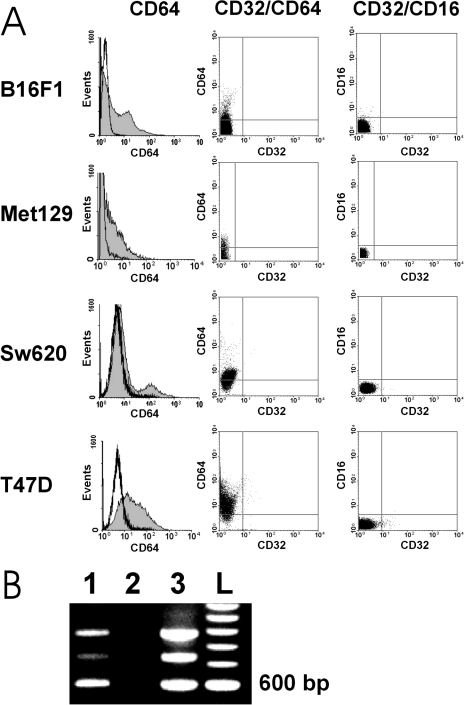
FcγRI Expression by sTn-Mucin-Secreting Tumor Cells
Because prior exposure to tumor-associated antigens was necessary for the observed promotion of tumor cell growth, suggested is a mechanism of specific recognition of tumor-associated antigens by antibody. Based on our previous experience, which identified immune complexes as triggering promotion of tumor progression [7], we investigated whether the observed promotion of tumor cell growth involved FcγR expressed by tumor cells that could be cross-linked by complexes of shed tumor-associated antigens and antibody induced against these shed antigens. To facilitate this investigation, we analyzed FcγR expression in a model involving tumor cells that either express or do not express a known tumor-associated antigen (sTn); and mAb B72.3 having binding specificity for sTn, which could be used as mediator between sTn-containing shed tumor-associated antigen and FcγR. B16F1 tumor cells (not expressing sTn), and the sTn-mucin-secreting tumor cells SW620 (human colon carcinoma), T-47D (human ductal breast carcinoma) and Met 129 were first analyzed for FcR expression by flow cytometric analysis for surface expression of FcγRI (CD64), FCγRII (CD32), and FcγRIII (CD16). This analysis showed that only FcγRI (CD64) was significantly expressed by the four different tumor cell lines, whereas expression of FcγRII (CD32) and FcγRIII (CD16) was either minimally detected or undetectable (Figure 2A). Due to this protein expression pattern, and to determine which FcγRI subtypes were expressed, we further characterized the expression of FcγRI at the mRNA level. SW620 tumor cells and T-47D tumor cells were analyzed by reverse transcription-polymerase chain reaction (RT/PCR) using FcγRI-specif icprimers [11]. By agarose gel electrophoresis, the RT/PCR of SW620 tumor cells and T-47D tumor cells showed a similar banding pattern; hence, T-47D was selected as representative of both. A primary amplified cDNA band of about 600 bp was observed (Figure 2B), and sequence analysis of the 600-bp band identified the sequence as that of the FcγRIB gene.
Figure 2.
(A–B) FcγR expression by B16F1 tumor cells and sTn-mucin-secreting tumor cells. (A) Flow cytometric analysis of human tumor cell lines SW620 and T-47D, and murine tumor cell lines B16F1 and Met 129, for cell surface expression of FcγRI (CD64), CD64 in comparison to FcγRII (CD32), and FcγRIII (CD16) in comparison to CD32 (duplicate analyses). All of these tumor cell lines show certain low percentages (10% to 40%) of cells expressing significant amounts of CD64. None showed evident expression of CD16 or CD32 (B) Ethidium bromide-stained agarose gel showing amplified FcγRI cDNA (600 bp): lane 1, from T-47D carcinoma cells; lane 2, blank (negative control); lane 3, from U937 monocytic cells (positive control); lane L, standard 1000-bp ladder.
Immune Complexes Induced Proliferation of FcγRi-Expressing Tumor Cells
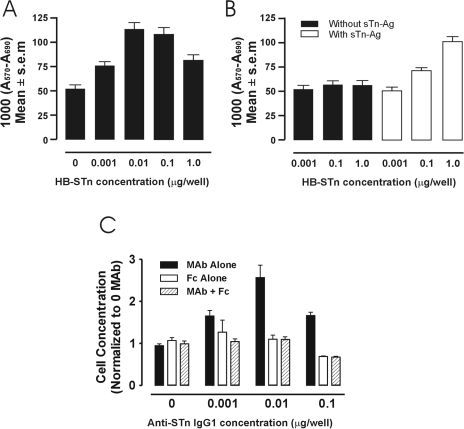
Because FcγRIB has greater affinity for binding immune complexes than monomeric IgG [13], we investigated whether immune complexes comprised of shed tumor-associated antigen and anti-shed tumor-associated antigen Ab mediated the promotion of tumor growth. Because T-47D tumor cells secrete sTn-mucin [7], murine anti-sTn IgG1 mAb was added to the cells in an in vitro culture, forming soluble immune complexes with the secreted sTn-mucin. Proliferation was then compared between T-47D tumor cells incubated in various concentrations of anti-sTn IgG1 mAb, and control T-47D tumor cells incubated with serum-free medium only. A statistically significant induction of tumor cell growth was observed for the tumor cells incubated with anti-sTn IgG1 mAb compared with the control tumor cells (Figure 3A). Similar growth induction curves were also observed for Met 129 and SW620 carcinoma cell lines. The bell-shaped, tumor growth induction curve is indicative of sTn antigen excess at the lower anti-sTn mAb concentrations, optimal immune complex formation that would therefore be more effective in cross-linking FcγRI, and antibody excess at the higher anti-sTn mAb concentrations. Additionally, the results show that the repeated sTn antigen of mucin secreted by tumor cells, and IgG antibody bound thereto, can provide the proper immune complex for cross-linking FcγRI.
Figure 3.
(A–C) Immune complex induction of tumor cell proliferation as measured by MTT assays (A570 to A690 nm). (A) Proliferation of sTn-mucin producing T-47D tumor cells after incubation with various concentrations of anti-sTn IgG1 Ab (HB-STn) (repeated in triplicate). The mean of well values with 0.01 and 0.1 µg/well of Ab are significantly greater than those observed for the remaining concentrations (P<0.01). (B) Proliferation of BWFI tumor cells after incubation with various concentrations of anti-sTn IgG1 Ab (HB-STn) in the absence of sTn-mucin (black boxes) or in the presence of sTn-mucin (white boxes). Averages of six wells per experiment. The mean of well values without sTn-Ag does not show a significant difference when compared to that of the control. The mean of well values with sTn-Ag+0.1 p.g of HB-STn Ab, and sTn-Ag + 1 µg HB-STn Ab are significantly greater than the means observed for the remaining concentrations (P<0.05 and P<0.01, respectively). (C) Proliferation of T-47D tumor cells incubated with various concentrations of anti-sTn IgG1 (solid black boxes); with various concentrations of Fc fragment alone (white boxes); and with various concentrations of anti-sTn IgG1 and Fc fragments (hatched boxes) Averages of six wells per experiment. The means of well values from each of the experiments containing anti-sTn IgG1 are significantly greater compared with that of the combination of anti-sTn IgG1 and Fc fragments or of Fc fragments alone (control) (P<0.01).
To confirm that the induction of tumor cell proliferation was a response to cross-linking of FcγRI-expressing tumor cells by immune complexes, we repeated this in vitro assay using murine metastatic melanoma cells, B16F1. We have found that these melanoma cells also express FcγRI, but do not secrete detectable amounts of sTn-mucin. The melanoma cells were treated in in vitro culture with either anti-sTn IgG1 mAb in various concentrations and supernatant containing sTn-mucin, with anti-sTn IgG1 mAb, or with fresh tissue culture medium as a control. A statistically significant induction of tumor cell growth was observed for the melanoma cells incubated with anti-sTn IgG1 mAb and sTn-mucin compared with melanoma cells incubated in either anti-sTn IgG1 mAb or no antibody (Figure 3B). These results further support the observation that it is immune complexes, which cross-link FcγRI-expressing tumor cells, which induce tumor cell proliferation. To further establish the role of FcγRI in immune complex binding and induction of tumor cell proliferation, inhibition experiments were performed with murine Fc fragments and T-47D tumor cells. T-47D tumor cells were treated with either anti-sTn IgG1 mAb alone, or anti-sTn IgG1 mAb with Fc fragments at a concentration 100-fold that of the anti-sTn IgG1 mAb. Coincubation of T-47D tumor cells and anti-sTn IgG1 mAb with Fc fragments effectively inhibited tumor cell proliferation induced by the immune complexes (Figure 3C), further demonstrating the role of FcγRI in the binding of immune complexes to the tumor cells.
Immune Complexes Induced Production of sTn-Mucin by FcγRi-Expressing Tumor Cells
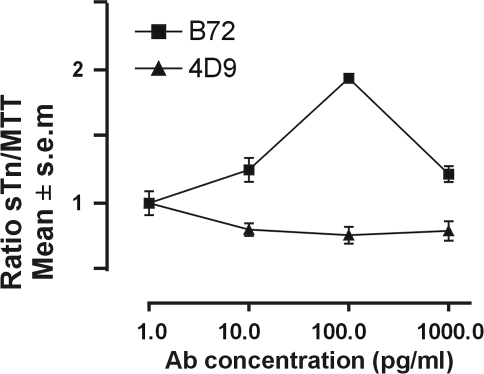
The amount of sTn-mucin produced by adenocarcinomas in vivo is associated with the metastatic potential of the tumor [14,15], may be used as a marker of progression and metastasis [15], and may be used as a prognostic indicator [16]. To investigate if cross-linking of FcγRI expressed by sTn-mucin-secreting tumor cells may also induce sTn-mucin production, T-47D tumor cells were incubated in the presence of either various concentrations of anti-sTn IgG1 mAb, tissue culture medium alone, or an isotype control mAb. The culture supernatants were then assayed for sTn-mucin production by measuring the amount of sTn epitope, and sTn-mucin production was then expressed as the ratio of the amount of sTn-mucin to the amount of cell proliferation to take induction of cell proliferation into account. sTn-mucin production by T-47D tumor cells in the presence of anti-sTn IgG1 mAb was increased approximately two-fold over comparative cells to which isotype control mAb was added (Figure 4). Further, treatment of T-47D tumor cells with an isotype control mAb did not induce a detectable increase in sTn-mucin production compared with the comparative control cells to which no IgG1 mAb was added (not shown). Based on these results, in addition to being a mechanism by which tumor cell proliferation is induced, immune complexes formed between sTn-mucin and anti-sTn Ab can induce production of sTn-mucin by sTn-mucin-secreting, FcγRI-expressing tumor cells.
Figure 4.
Immune complex induction of sTn-mucin production by tumor cells. Ratio of sTn-mucin to tumor cell proliferation for T-47D tumor cells incubated in various concentrations of anti-sTn IgG1 Ab (B72.3) or isotype control Ab (4D9). Each point represents the mean of values obtained from six wells per experiment.
Induction of Tumor Cells by FcγRi Cross-linking Is Dependent on the Tyrosine Kinase Signal Transduction Pathway
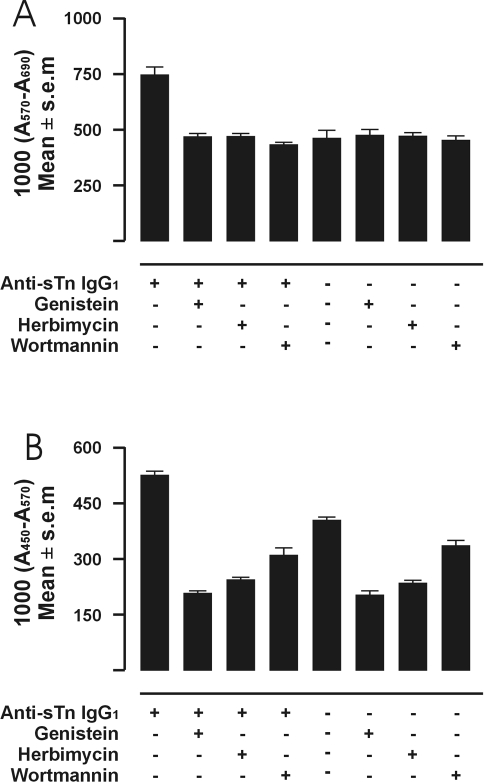
There is evidence that cross-linking of FcγRI-expressing cells can activate the tyrosine kinase signal transduction pathway [17,18]. To explore the possibility that the induction of proliferation or sTn-mucin production by tumor cells having FcγRI cross-linked by immune complexes also involved elements of the tyrosine kinase pathway, T-47D tumor cells were treated with the selective tyrosine kinase inhibitors genistein or herbimycin A. Cell proliferation or sTn-mucin production was compared between T-47D tumor cells, T-47D treated with genistein, T-47D treated with herbimycin A, T-47D incubated with anti-sTn IgG1 mAb, T-47D treated with genistein and then incubated with anti-sTn IgG1 mAb, or T-47D treated with herbimycin and then incubated with anti-sTn IgG1 mAb. A statistically significant induction of (about a 60% increase in) proliferation was observed for the tumor cells incubated with anti-sTn IgG1 mAb compared with the same basal level of proliferation exhibited by untreated (T-47D alone) or treated (T-47D + genistein or T-47D + herbimycin A) tumor cells in the absence of anti-sTn IgG1 mAb (Figure 5A). That pretreatment of the tumor cells with either genistein or herbimycin A resulted in a total inhibition of the induction of tumor cell proliferation observed in the presence of anti-sTn IgG1 mAb (Figure 5A) demonstrates that induction of tumor cell proliferation by immune complexes is dependent on the tyrosine kinase signal transduction pathway. However, the constitutive level of detectable sTn-mucin production by the tumor cells (T-47D alone) was significantly inhibited by either of the tyrosine kinase inhibitors genistein or herbimycin A (Figure 5B), thereby preventing us from assessing involvement of the tyrosine kinase pathway in sTn-mucin production induced by cross-linking FcγRI by immune complexes.
Figure 5.
(A–B) Effect of treatment with signal transduction pathway inhibitors. (A) Immune complex induction of proliferation of T-47D tumor cells with anti-sTn IgG1 with no inhibitor, after treatment with genistein, after treatment with herbimycin, and after treatment with wortmannin. Effect of treatment on constitutive level is represented by T-47D in the absence of anti-sTn IgG1 with no inhibitor, after treatment with genistein, after treatment with herbimycin, and after treatment with wortmannin. The number of cells/well is expressed as 1000x the corrected absorbance of the media after the MTT reaction. Averages of six wells per experiment. The mean of well values with sTn-Ag (first left column) reflect the induced proliferation of the tumor cells. The means of well values obtained with the inhibitors all indicate significant inhibition of the induction (P<0.01). (B) Immune complex induction of sTn-mucin production of T-47D tumor cells with anti-sTn IgG1 with no inhibitor, after treatment with genistein, after treatment with herbimycin, and after treatment with wortmannin. Effect of treatment on constitutive level is represented by T-47D in the absence of anti-sTn IgG1 with no inhibitor, after treatment with genistein, after treatment with herbimycin, and after treatment with wortmannin. The number of cells/well is expressed as 1000x the corrected absorbance of the media after the peroxidase ELISA reaction. Average of six wells per experiment. The means of well values with and without sTn-Ag are significantly different (P<0.01). The mean of well values with inhibitors show significant different degrees of inhibition (P<0.01).
Activation of tyrosine kinases is followed by events that can include phosphorylation on tyrosine of multiple cellular substrates including phosphatidylinositol-3 kinase (PI-3 kinase) [17], a tyrosine kinase-regulated enzyme. To explore the possibility that the induction of tumor cell proliferation or of sTn-mucin production by cross-linking FcγRI by immune complexes also involved PI-3 kinase, T-47D tumor cells were treated with wortmannin, a selective inhibitor of PI-3 kinase. As also shown in Figure 5A, a statistically significant induction of tumor cell growth was observed for the tumor cells incubated with anti-sTn IgG1 mAb compared with the same basal level of proliferation exhibited by untreated (T-47D alone) or treated (T-47D + wortmannin) tumor cells in the absence of anti-sTn IgG1 mAb. That pretreatment of the tumor cells with wortmannin completely inhibited the induction of tumor proliferation observed in the presence of anti-sTn IgG1 mAb (Figure 5A) demonstrates that immune complex-induced tumor cell proliferation is also dependent on the PI-3 kinase. However, the constitutive level of detectable sTn-mucin production by the tumor cells (T-47D alone) was significantly inhibited by wortmannin (Figure 5B); thereby preventing us from assessing involvement of PI-3 kinase on induction of sTn-mucin production by cross-linking FcγRI by immune complexes.
Discussion
Because tumor-associated antigens can comprise self or altered self antigens, a challenge in tumor immunotherapy is the induction of immune responses, which are capable of inducing a clinical antitumor effect, rather than a disease-promoting (e.g., immune evasive or autoimmune) effect. Our results strongly support the concept of a humoral immune response that can directly promote tumor progression. The results of the in vivo experiments show that B cells exposed in vivo to tumor-associated antigens can promote tumor growth in immune-competent animals, sustaining the suggestion of an undesired tumor promoting role of a humoral immune response induced by tumor [5–7]. Moreover, the results of the in vitro experiments, utilizing a model that isolated the interaction to only tumor cells and B cells, stress the concept that B cells exposed in vivo to tumor-associated antigens can directly interact with certain tumor cells through specific recognition, and promote tumor cell growth. As shown by flow cytometry tumor cells can express FcγRI. RT-PCR and DNA sequencing showed that those tumor cells expressed FcγRIB-RNA, which encodes a transmembrane receptor with two extracellular domains, in which the second extracellular domain splices precisely to the transmembrane/cytoplasmic domain, and does not utilize the third extra-cellular domain encoded by the FcγRIA gene [11]. FcγRIB has higher affinity for immune complexes than free Ab [13]; this suggests that an specific recognition tumor cell/B cells could be carried out, in some cases, by immune complexes produced by binding of tumor cell-secreted antigens with the correspondent antibodies produced by the lymphocytes. The experiments with adenocarcinoma cells secreting sTn-mucin incubated in the presence of anti-sTn monoclonal Ab sustain the interpretation that tumor-associated antigen secreted by tumor cells, and IgG bound thereto, can provide the required immune complex for cross-linking the tumor cell FcγRI. The interpretation that cross-linking of FcγRI is the basis of this type of induction of tumor cell proliferation is supported by the fact that Fc fragments or monomeric IgG can inhibit the induction of tumor cell proliferation, and by the requirement for both exogenous sTn-mucin and anti-sTn IgG1 mAb to induce proliferation of a nonsTn-mucin-secreting tumor cell line (B16F1 melanoma cells). Further, cross-linking can be impaired by antibody excess, i.e., higher anti-sTn monoclonal antibody concentrations. This suggests the possible existence of a feedback mechanism in which the concentration and type of both components (tumor Ag and antitumor Ab) may determine promotion or inhibition of tumor cell growth.
The concordance between FcγRI-dependent induction of tumor cell proliferation and its inhibition of induction by protein tyrosine kinase inhibitors genistein and herbimycin A is supported by the observations that cross-linking of FcγRI can result in activation of the tyrosine kinase signal transduction pathway [17,18], and that activation of tyrosine kinase signal transduction pathway has been associated with tumor cell proliferation [19,20]. For example, a significant correlation was found between tyrosine kinase signal transduction pathway activity and the proliferation capacity of human colon solid tumors and tumor cell lines [21]. Wortmannin is a potent inhibitor of PI-3 kinase, a tyrosine kinase-regulated enzyme [22]. That wortmannin also inhibited FcγRI-dependent induction of tumor cell proliferation is supported by the observation that some human tumor cell lines have elevated levels of PI-3 kinase, which correlated with the oncogenic growth of these cells [23].
We also show an induction of sTn-mucin production by tumor cells having FcγRI cross-linked by immune complexes. As related to a metastatic phenotype, production of sTn-mucin by human mucinous adenocarcinomas in vivo is associated with the metastatic potential of the tumor [14]. Metastases express a decrease in mucin core structures such as Tn and T antigens, with a reciprocal increase in sTn antigen structures, and the increased sialylation of mucin-associated carbohydrates is believed to increase metastatic potential because it enhances motility [14]. Thus, an induction in sTn-mucin production could lead to an augmentation in metastatic phenotype by several mechanisms: when surface expressed, an enhancement of motility; when shed, the formation of more polyvalent immune complexes that cross-link FcγR on FcγR-expressing immune effector cells in activating the cells to promote matrix degradation, angiogenesis, and metastasis [7]; and when shed, the formation of more polyvalent immune complexes that cross-link FcγR on FcγR-expressing tumor cells in an amplification loop, which results in more sTn-mucin production. Considering that inhibition of tyrosine kinase signal transduction pathway (e.g., by genistein) decreased constitutive mucin expression by normal epithelial cells, and that tyrosine kinase activity is critical for maintaining a high level of mucin expression in the epithelial cells [24], it is not surprising that inhibition of the tyrosine kinase signal transduction pathway appears to decrease constitutive expression of mucin by tumor cells of epithelial origin.
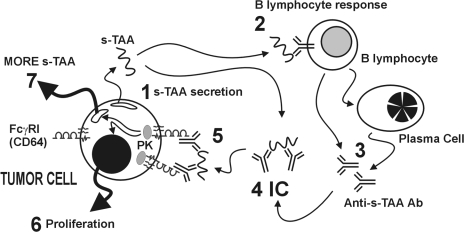
Taken together, these observations support the possibility of a direct antibody-mediated effect of B cells on tumor cells. As illustrated in Figure 6, the mechanism underlying this direct effect, which we term “specific Ab-tumor cell promotion”, involves: tumor secretion of shed tumor-associated antigens (s-TAA, step 1); activation of B cells by s-TAA (step 2) to secrete anti-s-TAA Ab (step 3), which form polyvalent immune complexes (IC, step 4) comprised of s-TAA and anti-s-TAA Ab; and IC cross-linking of FcγRI on FcγRI-expressing tumor cells, which activates one or more signal transduction pathways (e.g., protein kinase activation; step 5), resulting in the induction of cell proliferation (step 6) and sTn-mucin production (step 7). Further, we suggest a possible role of tumor-expressed FcγRI in complementing the metastatic phenotype.
Figure 6.
Model of specific Ab-tumor cell promotion. Tumor cell secrete tumor-associated antigens (s-TAA, such as sTn mucin; step 1). B cells recognize the s-TAA (step 2) and, as activated B cells and plasma cells, respond by producing and secreting anti-s-TAA IgG (step 3). Polyvalent immune complexes (step 4) comprised of s-TAA bound to anti-sTAA Ab cross-link FcγRI on FoyRI-expressing tumor cells and activate one or more signal transduction pathways (e.g., protein kinase activation PK; (step 5), resulting in the induction of cell proliferation (step 6) and sTn-mucin production (step 7).
Abbreviations
- sTn
sialyl Tn
- B-TIL
tumor-infiltrating B lymphocytes
- TIL
tumor-infiltrating T lymphocytes
- PI-3 kinase
phosphatidylinositol-3 kinase
References
- 1.Strand S, Hofmann WJ, Hug H, Muller M, Otto G, Strand D, Mariani SM, Stremmel W, Krammer PH, Galle PR. Lymphocyte apoptosis induced by CD95 (APO-1/Fas) ligand-expressing tumor cells — a mechanism of immune evasion? Nat Med. 1996;2:1361–1366. doi: 10.1038/nm1296-1361. [DOI] [PubMed] [Google Scholar]
- 2.Hahne M, Rimoldi D, Schroter M, Romero P, Schreier M, French LE, Schneider P, Bornand T, Fontana A, Lienard D, Cerottini J, Tschopp J. Melanoma cell expression of Fas (Apo-1/CD95) ligand: implications for tumor immune escape. Science. 1996;274:1363–1366. doi: 10.1126/science.274.5291.1363. [DOI] [PubMed] [Google Scholar]
- 3.Nakashima M, Sonoda K, Watanabe T. Inhibition of cell growth and induction of apoptotic cell death by human tumor-associated antigen RCAS1. Nat Med. 1999;5:938–942. doi: 10.1038/11383. [DOI] [PubMed] [Google Scholar]
- 4.Gimmi CD, Morrison BW, Mainprice BA, Gribben JG, Boussiotis VA, Freeman GJ, Park SY, Wantanabe M, Gong J, Hayes DF, Kufe DW, Nadler LM. Breast cancer-associated antigen, DF3/MUC-1, induces apoptosis of activated human T cells. Nat Med. 1996;2:1367–1370. doi: 10.1038/nm1296-1367. [DOI] [PubMed] [Google Scholar]
- 5.Monach PA, Schreiber H, Rowley DA. CD4+ and B lymphocytes in transplantation immunity: II. Augmented rejection of tumor allografts by mice lacking B cells. Transplantation. 1993;55:1356–1361. [PubMed] [Google Scholar]
- 6.Rowley DA, Stach RM. B lymphocytes secreting IgG linked to latent transforming growth factor-(beta) prevent primary cytolytic T lymphocyte responses. Int Immunol. 1998;10:355–363. doi: 10.1093/intimm/10.3.355. [DOI] [PubMed] [Google Scholar]
- 7.Barbera-Guillem E, May KF, Jr, Nyhus JK, Nelson MB. Promotion of tumor invasion by cooperation of granulocytes and macrophages activated by anti-tumor antibodies. Neoplasia. 1999;1:453–460. doi: 10.1038/sj.neo.7900054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Barbera-Guillem E, Nelson MB, Barr B, Nyhus JK, May KF, Jr, Feng L, Sampsel JW. B lymphocyte pathology in human colorectal cancer. Experimental and clinical therapeutic effects of partial B cell depletion. Cancer Immunol Immunother. 2000;48:541–549. doi: 10.1007/PL00006672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Manson LA. Does antibody-dependent epitope masking permit progressive tumor growth in the face of cell-mediated cytotoxicity? Immunol Today. 1991;12:352–355. doi: 10.1016/0167-5699(91)90065-2. [DOI] [PubMed] [Google Scholar]
- 10.Zusman T, Gohar O, Eliassi I, Avivi Y, Lisansky E, Sautes C, Even J, Bonnerot C, Fridman WH, Witz IP, Ran M. The murine Fc-gamma (Fcγ) receptor type II B1 is a tumorigenicity-enhancing factor in polyoma-virus-transformed 3T3 cells. Int J Cancer. 1996;65:221–229. doi: 10.1002/(SICI)1097-0215(19960117)65:2<221::AID-IJC16>3.0.CO;2-G. [DOI] [PubMed] [Google Scholar]
- 11.Ernst LK, van de Winkel JGJ, Chiu IM, Anderson CL. Three genes for the human high affinity Fc receptor for IgG (FcγRI) encode four distinct transcription products. J Biol Chem. 1992;267:15692–15700. [PubMed] [Google Scholar]
- 12.Gomez-Guerrero C, Duque N, Casado MT, Pastor C, Blanco J, Mampaso F, Vivanco F, Egido J. Administration of IgG Fc fragments prevents glomerular injury in experimental immune complex nephritis. J Immunol. 2000;164:2092–2101. doi: 10.4049/jimmunol.164.4.2092. [DOI] [PubMed] [Google Scholar]
- 13.Porges AJ, Redecha PB, Doebele R, Pan LC, Salmon JE, Kimberly RP. Novel Fcγ receptor I family gene products in human mononuclear cells. J Clin Invest. 1992;90:2102–2109. doi: 10.1172/JCI116094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bresalier RS, Ho SB, Schoeppner HL, Kim YS, Sleisenger MH, Brodt P, Byrd JC. Enhanced sialylation of mucin-associated carbohydrate structures in human colon cancer metastasis. Gastroenterology. 1996;110:1354–1367. doi: 10.1053/gast.1996.v110.pm8613039. [DOI] [PubMed] [Google Scholar]
- 15.Nakamori S, Ota DM, Cleary KR, Shirotani K, Irimura T. MUC1 mucin expression as a marker of progression and metastasis of human colorectal carcinoma. Gastroenterology. 1994;106:353–361. doi: 10.1016/0016-5085(94)90592-4. [DOI] [PubMed] [Google Scholar]
- 16.Kobayashi H, Terao T, Kawashima Y. Serum sialyl Tn as an independent predictor of poor prognosis in patients with epithelial ovarian cancer. J Clin Oncol. 1992;10:95–101. doi: 10.1200/JCO.1992.10.1.95. [DOI] [PubMed] [Google Scholar]
- 17.Lin C-T, Shen Z, Boros P, Unkeless JC. Fc Receptor-mediated signal transduction. J Clin Immunol. 1994;14:1–13. doi: 10.1007/BF01541170. [DOI] [PubMed] [Google Scholar]
- 18.Kiener PA, Rankin BM, Burkhardt AL, Schieven GL, Gilliland LK, Rowley RB, Bolen JB, Ledbetter JA. Cross-linking of Fcγ receptor I (FcγRI) and receptor II (FcγRII) on monocytic cells activates a signal transduction pathway common to both Fc receptors that involves the stimulation of p72 Syk protein tyrosine kinase. J Biol Chem. 1993;268:24442–24448. [PubMed] [Google Scholar]
- 19.Twaddle GM, Turbov J, Liu N, Murthy S. Tyrosine kinase inhibitors as antiproliferative agents against estrogen-dependent breast cancer cell line in vitro. J Surg Oncol. 1999;70:83–90. doi: 10.1002/(sici)1096-9098(199902)70:2<83::aid-jso4>3.0.co;2-l. [DOI] [PubMed] [Google Scholar]
- 20.Brown A, Jolly P, Wei H. Genistein modulates neuroblastoma cell proliferation and differentiation through induction of apoptosis and regulation of tyrosine kinase activity and N-myc expression. Carcinogenesis. 1998;19:991–997. doi: 10.1093/carcin/19.6.991. [DOI] [PubMed] [Google Scholar]
- 21.Keri G, Balogh A, Teplan I, Csuka O. Comparison of the tyrosine kinase activity with the proliferation rate in human colon solid tumors and tumor cell lines. Tumour Biol. 1988;9:315–322. doi: 10.1159/000217578. [DOI] [PubMed] [Google Scholar]
- 22.Barker SA, Caldwell KK, Hall A, Martinez AM, Pfeiffer JR, Oliver JM, Wilson BS. Wortmannin blocks lipid and protein kinase activities associated with PI 3-kinase and inhibits a subset of responses induced by FcεRI cross-linking. Mol Biol Cell. 1995;6:1145–1158. doi: 10.1091/mbc.6.9.1145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Xiao H, Nagai Y, Fukui Y, Tamiya-Koizumi K, Iwata H, Watanabe T, Hamaguchi M. Expression and tyrosine phosphorylation of phosphatidyl-inositol-3 kinase in human gastric cancer cells: Its correlation with cell growth. Int J Oncol. 1995;6:405–411. doi: 10.3892/ijo.6.2.405. [DOI] [PubMed] [Google Scholar]
- 24.Wegner CC, Zhou X, Ding Z-M, Kuo MT, Carson DD. Tyrosine kinase inhibition decreases Muc-1 expression in mouse epithelial cells. J Cell Physiol. 1997;170:200–208. doi: 10.1002/(SICI)1097-4652(199702)170:2<200::AID-JCP12>3.0.CO;2-L. [DOI] [PubMed] [Google Scholar]