Abstract
In the root epidermis of Arabidopsis, hair and nonhair cell types are specified in a distinct position-dependent pattern. Here, we show that transcriptional feedback loops between the WEREWOLF (WER), CAPRICE (CPC), and GLABRA2 (GL2) genes help to establish this pattern. Positional cues bias the expression of the WER MYB gene, leading to the induction of CPC and GL2 in cells located in a particular position (N) and adoption of the nonhair fate. The truncated MYB encoded by CPC mediates a lateral inhibition mechanism to negatively regulate WER, GL2, and its own gene in the alternative position (H) to induce the hair fate. These results provide a molecular genetic framework for understanding the determination of a cell-type pattern in plants.
INTRODUCTION
The appropriate specification and patterning of distinct cell types is essential for the proper development of multicellular organisms. In plants, a simple case of cell patterning occurs during development of the root epidermis in Brassicaceae species (e.g., Arabidopsis), in which the distribution of the root hair and nonhair cell types is determined by a position-dependent mechanism (Cormack, 1935; Bunning, 1951; Berger et al., 1998a). Epidermal cells in contact with two underlying cortical cells (the H position) adopt the root hair cell fate, whereas cells located outside a single cortical cell (the N position) adopt the nonhair fate. This pattern implies that positional information is responsible for guiding the specification of the epidermal cell types.
In Arabidopsis, several genes have been implicated in the establishment of the root epidermal pattern, including GLABRA2 (GL2), WEREWOLF (WER), and CAPRICE (CPC). The GL2 gene encodes a homeodomain transcription factor protein (Rerie et al., 1994) that is required to specify the nonhair cell type (DiCristina et al., 1996; Masucci et al., 1996). Accordingly, GL2 is expressed preferentially in the N cell position during epidermis development, beginning in the early embryo (Masucci et al., 1996; Lin and Schiefelbein, 2001). The WER gene encodes a MYB transcription factor required for the specification of the nonhair cell type, and WER expression occurs preferentially in the developing epidermal cells in the N position of the root (Lee and Schiefelbein, 1999). Furthermore, WER is a positive regulator of GL2 expression (Lee and Schiefelbein, 1999). The CPC gene encodes a small MYB protein without a putative transcriptional activation domain that is required for hair cell specification (Wada et al., 1997), although recent findings show that CPC is expressed preferentially in the N cell position (T. Wada and K. Okada, personal communication).
The opposing effects of the WER and CPC genes on root epidermal cell fate have been proposed to be responsible for the establishment of the cell-type pattern, perhaps as a result of competition between these MYB proteins for access to partner proteins or promoter targets (Lee and Schiefelbein, 1999). To further understand the roles of these genes in root epidermal patterning, we wished to examine their possible regulatory interactions to determine whether negative transcriptional regulation may contribute to cell patterning. Here, we report that transcriptional feedback loops exist between the WER, CPC, and GL2 genes and that they are used as part of a lateral inhibition pathway to define the cell-type pattern in the root epidermis.
RESULTS
WER Is a Positive Regulator of the CPC Gene
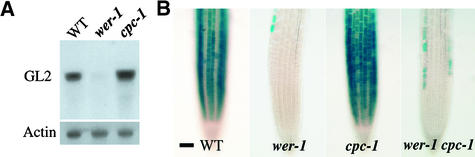
To define the possible regulatory interactions between WER, GL2, and CPC, we first examined whether WER regulates CPC gene expression. Using RNA gel blot analysis, we found that root tips of the wer-1 mutant accumulate significantly less CPC RNA than do wild-type roots (Figure 1A). In addition, we used a CPC::β-glucuronidase (GUS) reporter fusion that displays an N cell–specific expression pattern and has been found, in unpublished work, to correspond to the pattern of CPC RNA accumulation (T. Wada and K. Okada, personal communication). This CPC::GUS construct was introduced into the wer-1 mutant by crossing, and we discovered that the N cell–specific expression of CPC was diminished greatly in this background (Figure 1B). Rare GUS-expressing epidermal cells in the wer-1 CPC::GUS roots were found in both the N and H cell positions (data not shown), indicating that the wer-1 mutation abolishes the position-dependent expression of CPC. Interestingly, these effects of wer-1 on CPC expression are analogous to the effects of wer-1 on GL2 expression (Figures 2A and 2B) (Lee and Schiefelbein, 1999). These results show that despite the known role of WER in defining the nonhair cell fate (Lee and Schiefelbein, 1999), WER also is a positive regulator of the hair fate–promoting gene CPC.
Figure 1.
CPC Gene Expression Is Regulated by WER and CPC.
(A) RNA gel blot hybridization analysis using a CPC-specific gene probe. RNA was isolated from the root tips of seedlings from the genotypes indicated. The same blot was rehybridized with an 18S rRNA probe as a loading control.
(B) Expression of the CPC::GUS reporter in the developing root epidermis of 4-day-old seedlings. This reporter also is expressed in the developing stele near the root tip, but this expression is not associated with the role of CPC in epidermal development (M.M. Lee and J. Schiefelbein, unpublished observations).
WT, wild type. Bar in (B) = 50 μm.
Figure 2.
GL2 Gene Expression Is Regulated by WER and CPC.
(A) RNA gel blot hybridization analysis using a GL2-specific gene probe. RNA was isolated from root tips from the genotypes indicated and subjected to RNA gel blot analysis. The same blot was rehybridized with an actin probe as a loading control.
(B) Expression of the GL2::GUS reporter in the developing root epidermis of 4-day-old seedlings. The wer-1 and wer-1 cpc-1 plants were stained overnight, whereas the other lines were stained for 3 hr.
WT, wild type. Bar in (B) = 50 μm.
There is abundant root hair production in the epidermis of the wer-1 mutant, which lacks CPC expression (Table 1, Figure 1), and in the wer-1 cpc-1 double mutant, which lacks a functional CPC gene (Table 1) (Lee and Schiefelbein, 1999). These results suggest that CPC is not required for epidermal cells to be specified as root hair cells. Furthermore, a comparison of cell specification in wild-type, cpc-1, and wer-1 cpc-1 lines shows that the production of nonhair cells in the H position of cpc-1 is mediated by WER (Table 1). This finding suggests that in wild-type roots, CPC promotes the hair cell fate by preventing WER-directed nonhair cell specification in the H position. Given the preferential expression of WER in N cells (Lee and Schiefelbein, 1999) and the positive regulation of CPC by WER (Figure 1), the simplest interpretation is that CPC mediates a lateral inhibition pathway that originates in the N cells and prevents neighboring cells in the H position from adopting the nonhair cell fate.
Table 1.
Specification of Cell Types in the Root Epidermis
| H Cell Position
|
N Cell Position
|
||||
|---|---|---|---|---|---|
| Genotype | Hair Cells in Epidermis (%) | Hair Cells (%) | Nonhair Cells (%) | Hair Cells (%) | Nonhair Cells (%) |
| Wild type (Columbia) | 41.3 ± 6.6 | 93.8 ± 3.4 | 6.2 ± 3.4 | 2.2 ± 3.8 | 97.8 ± 3.8 |
| wer-1 | 93.2 ± 5.0 | 95.8 ± 7.2 | 4.2 ± 7.2 | 91.3 ± 9.2 | 8.7 ± 9.2 |
| cpc-1 | 8.7 ± 3.9 | 18.9 ± 5.1 | 81.1 ± 5.1 | 0 ± 0 | 100 ± 0 |
| wer-1 cpc-1 | 83.6 ± 6.1 | 94.0 ± 5.3 | 6.0 ± 5.3 | 81.3 ± 6.4 | 18.7 ± 6.4 |
| 35S::WER | 42.5 ± 5.0 | 92.9 ± 3.4 | 7.1 ± 3.4 | 1.6 ± 3.3 | 98.4 ± 3.3 |
| 35S::WER wer-1 | 59.8 ± 5.5 | 78.6 ± 10.2 | 21.4 ± 10.2 | 37.4 ± 5.1 | 62.6 ± 5.1 |
| 35S::WER wer-1 cpc-1 | 41.2 ± 6.4 | 61.0 ± 8.8 | 39.0 ± 8.8 | 37.2 ± 10.5 | 62.8 ± 10.5 |
| 35S::CPC | 86.5 ± 6.3 | 94.4 ± 3.8 | 5.6 ± 3.8 | 82.2 ± 3.8 | 17.8 ± 3.8 |
| 35S::CPC cpc-1 | 91.0 ± 5.9 | 98.3 ± 1.9 | 1.7 ± 1.9 | 83.0 ± 6.7 | 17.0 ± 6.7 |
| 35S::CPC wer-1 | 87.5 ± 5.1 | 97.3 ± 0.8 | 2.7 ± 0.8 | 91.3 ± 5.8 | 8.7 ± 5.8 |
At least 10 5-day-old seedlings were examined for each strain. Values represent means ±sd. In all strains, ∼40% of epidermal cells were in the H position.
CPC Is a Negative Regulator of the WER, GL2, and CPC Genes
If the hypothesis described above is correct, CPC might be expected to inhibit the expression of GL2 and/or WER in the H cell position. To test this possibility, we first examined the effect of the cpc-1 mutation on GL2 RNA accumulation and reporter gene expression. An increased amount of GL2 RNA was detected in root tips of the cpc-1 mutant (Figure 2A), and ectopic GUS expression in the H cell position was observed in cpc-1 GL2::GUS roots (Figure 2B). We also generated a wer-1 cpc-1 GL2::GUS line and found that the ectopic GL2::GUS expression is WER dependent (Figure 2B). Furthermore, by examining mature epidermal cells, we found that the infrequent non-GUS-expressing cells in the cpc-1 GL2::GUS line correspond to root hair cells (M.M. Lee and J. Schiefelbein, unpublished observations). These results show that CPC-dependent root hair formation is linked to the repression of GL2 transcription in the H cell position.
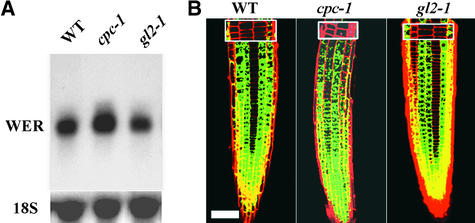
In a similar way, we examined the effect of the cpc-1 mutation on WER gene expression. We found that the cpc-1 mutant had an increased amount of WER RNA in the root (Figure 3A) and significant ectopic WER::green fluorescent protein (GFP) reporter expression in the H cell position during root epidermis development (Figure 3B). Thus, CPC acts as a negative regulator of both GL2 and WER expression in the H cell position.
Figure 3.
WER Gene Expression Is Regulated by CPC.
(A) RNA gel blot hybridization analysis using a WER-specific gene probe. RNA was isolated from root tips from the genotypes indicated and subjected to RNA gel blot analysis. The same blot was rehybridized with an 18S rRNA probe.
(B) Expression of the WER::GFP reporter in the developing root epidermis of 4-day-old seedlings analyzed by confocal fluorescence microscopy. Propidium iodide dye (red signal) was included to visualize cell boundaries. The insets are focused on the cortical cell layer and therefore indicate the positions of the epidermal cell files.
WT, wild type. Bar in (B) = 50 μm.
We next considered the possibility that the CPC-dependent lateral inhibition mechanism also might reduce the capacity of H cells to produce CPC. To test this possibility, we examined the expression of the CPC gene in the cpc-1 mutant background. We found that CPC::GUS reporter expression was increased significantly in the H cell position of cpc-1 relative to that of the wild type (Figure 1B). Furthermore, we generated a cpc-1 wer-1 CPC::GUS line and found that the ectopic CPC expression in the cpc-1 background is WER dependent (Figure 1B). Together with the results described above, this shows that CPC participates in feedback loops to negatively regulate the expression of its own gene and the WER gene in the H cells.
GL2 Does Not Regulate WER or CPC Expression
We also tested the possibility that the homeodomain protein GL2 might participate in the WER-CPC feedback loops. However, we found no significant difference in WER RNA accumulation and WER::GFP expression in the gl2-1 mutant compared with that of the wild type (Figures 3A and 3B). Likewise, a normal level of CPC RNA and a normal pattern of CPC::GUS reporter expression were observed in gl2-1 roots (Figure 1A) (M.M. Lee and J. Schiefelbein, unpublished data). Thus, GL2 is not required for the appropriate regulation of WER or CPC.
Spatial Expression of WER Determines the Cell Pattern
Together, the results described above suggest that WER influences both the nonhair cell fate (by positively regulating GL2, which represses hair cell differentiation) and the hair cell fate (by positively regulating CPC, which mediates the lateral inhibition of the nonhair fate and thereby promotes hair cell differentiation). Thus, WER may be a critical director of the epidermal cell pattern, as illustrated in Figure 4. To test this possibility, we examined the effect of expressing the WER gene throughout the epidermis using the 35S promoter of Cauliflower mosaic virus (35S). We employed two lines, 35S::WER and 35S::WER wer-1, that had been generated previously (Lee and Schiefelbein, 1999) and characterized them in greater detail.
Figure 4.
Model Showing the Regulatory Interactions between WER, CPC, and GL2 That Define Cell Pattern in the Arabidopsis Epidermis.
(A) Interactions that occur in cells with equivalent developmental potentials. In the absence of positional cues, all epidermal cells express WER, CPC, and GL2 and use CPC to inhibit their neighbors from expressing these genes.
(B) Positional cues generate a bias in WER expression as a result of induction in N cells and/or inhibition in H cells (open symbols). Feedback loops enhance this bias to generate a high level (thick lines) of WER, CPC, and GL2 expression (and the nonhair cell fate) in N cells and a low level (dotted lines) of WER, CPC, and GL2 expression (and the hair cell fate) in H cells.
In plants harboring the 35S::WER construct, a normal pattern of epidermal cell types is formed (Table 1), although there is increased GL2 expression in the H cell position (Figure 5A), as reported previously (Lee and Schiefelbein, 1999). The lack of a major effect of this construct may be attributable to the influence of the endogenous WER gene, which the 35S::WER construct cannot overcome. Therefore, we examined the effect of 35S::WER in the wer-1 mutant background. Rather than generating a single-cell fate and a similar level of GL2 expression throughout the epidermis, 35S::WER wer-1 roots produced a nonuniform distribution of epidermal cell types (Table 1) and GL2::GUS expression (Figure 5A). In particular, we found that both N and H cells were able to adopt the nonhair or hair fate (Table 1), and a similar frequency of cells at each position were able to establish GL2 expression (Table 2). Thus, in the absence of an endogenous WER gene, the expression of WER throughout the epidermis generates a pattern of cell fates that is not determined strictly by cell position. This finding indicates that WER gene regulation is critical for proper interpretation of the positional cues that define the normal cell pattern.
Figure 5.
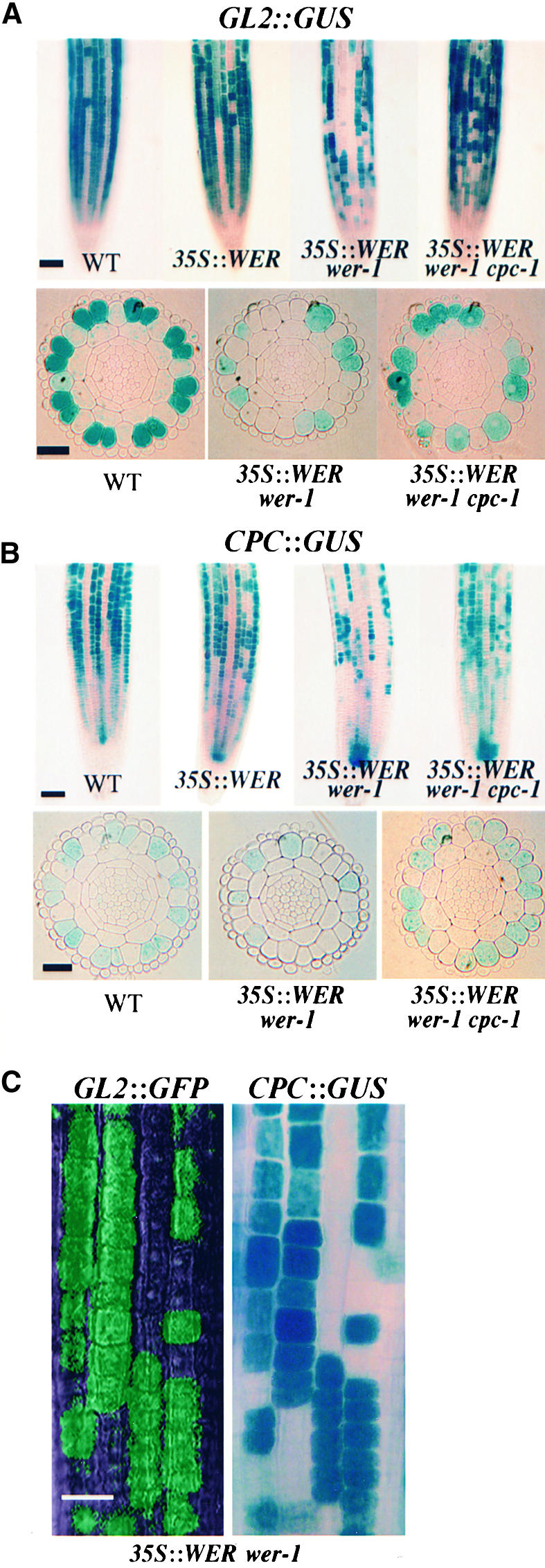
35S::WER Alters the Epidermal Cell Pattern.
(A) Expression of the GL2::GUS reporter in the developing root epidermis of 4-day-old seedlings. The top row contains surface views, and the bottom row shows transverse sections from the meristematic region. Note that a single layer of lateral root cap cells surrounds the epidermis in the transverse sections. All lines were stained for the same period of time (3 hr).
(B) Expression of the CPC::GUS reporter in the developing root epidermis of 4-day-old seedlings. The top row contains surface views, and the bottom row shows transverse sections from the meristematic region.
(C) Coordinate expression of GL2 and CPC in root epidermal cells. The accumulation of GFP and GUS is shown in a 5-day-old seedling root bearing the genotype 35S::WER wer-1 GL2::GFP CPC::GUS. An image was first recorded using fluorescence microscopy (to detect GL2::GFP expression); then, GUS activity was assessed (to detect CPC::GUS expression).
WT, wild type. Bars in (A) and (B), top rows = 50 μm; bars in (A) and (B), bottom rows = 25 μm; bar in (C) = 20 μm.
Table 2.
Location of GL2::GUS Expression in the Developing Root Epidermis
| H Cell position
|
N Cell Position
|
|||
|---|---|---|---|---|
| Genotype | Non-GUS-Expressing Cells (%) |
GUS-Expressing Cells (%) |
Non-GUS-Expressing Cells (%) |
GUS-Expressing Cells (%) |
| Wild type Columbia GL2::GUS | 85.3 ± 5.6 | 14.7 ± 5.6 | 0 ± 0 | 100 ± 0 |
| 35S::WER wer-1 GL2::GUS | 71.9 ± 6.8 | 28.1 ± 6.8 | 65.4 ± 7.7 | 34.6 ± 7.7 |
| 35S::WER wer-1 cpc-1 GL2::GUS | 56.2 ± 4.8 | 43.8 ± 4.8 | 47.6 ± 5.5 | 52.4 ± 5.5 |
A series of transverse sections from the meristematic region of at least five 4-day-old roots was examined for each strain. Values represent means ±sd.
The novel cell-type pattern in the 35S::WER wer-1 line may arise because stochastic fluctuations cause some developing epidermal cells to accumulate slightly more CPC than do their neighbors. Subsequently, as a result of the CPC-dependent feedback mechanism (Figure 4), these cells may establish a relatively high level of CPC and GL2 expression and adopt the nonhair fate while inhibiting their neighbors from expressing CPC and GL2 and forcing them to adopt the hair cell fate. If this interpretation is correct, then we would expect (1) that the pattern of CPC expression is altered (like that of GL2) in these lines and (2) that the novel cell-type pattern is dependent on CPC function.
To test the first of these expectations, we introduced the CPC::GUS reporter into the 35S::WER and 35S::WER wer-1 lines by crossing. The CPC::GUS reporter was in fact expressed in some root epidermal cells at each position in the 35S::WER wer-1 background, whereas 35S::WER CPC:: GUS plants possessed a relatively normal pattern of CPC expression (Figure 5B). To directly test the possibility that GL2 and CPC are expressed in the same set of epidermal cells, we used an available GL2::GFP reporter fusion (Lin and Schiefelbein, 2001) to generate 35S::WER wer-1 GL2::GFP CPC::GUS plants. By analyzing both GFP and GUS expression in the same plants, we determined that GL2 and CPC are expressed coordinately in a subset of root epidermal cells in this line (Figure 5C).
To test the second expectation, we introduced the cpc-1 mutation into the 35S::WER wer-1 mutant by crossing and analyzed its cell-type pattern. Indeed, the cell pattern produced by 35S::WER wer-1 cpc-1 plants was significantly different from that produced by 35S::WER wer-1 plants. A reduced number of root hair cells was formed (Table 1), and an increase in the frequency of GL2::GUS–expressing cells was observed (Figure 5A, Table 2). These results indicate that CPC is required, at least in part, for the novel 35S::WER wer-1 pattern.
Effect of CPC-Dependent Lateral Inhibition
Although the model illustrated in Figure 4 shows CPC regulating all three genes (GL2, WER, and CPC) in the H position, an alternative is that CPC inhibits only the expression of WER and that GL2 and CPC are affected indirectly through their dependence on WER. We were able to exclude this possibility by analyzing 35S::WER wer-1 and 35S::WER wer-1 cpc-1 plants. As described above, we observed CPC-dependent inhibition of nonhair cell fate and GL2 expression in these lines. Furthermore, we introduced the CPC::GUS reporter into the 35S::WER wer-1 cpc-1 line and discovered an increased frequency of GUS-expressing cells relative to the 35S::WER wer-1 CPC::GUS line (Figure 5B). Thus, CPC-dependent lateral inhibition is able to operate in the absence of a wild-type WER gene, which means that CPC does not control GL2 and CPC expression in the H cell position merely by regulating WER.
To further define the role of CPC, we analyzed plants containing a 35S::CPC construct (Wada et al., 1997). In both 35S::CPC and 35S::CPC cpc-1 roots, nearly all epidermal cells adopted the hair cell fate (Table 1) and exhibited reduced GL2 expression (Figure 6). This finding indicates that the expression of CPC alone throughout the epidermis is sufficient to inhibit cells in both the N and H positions from adopting the nonhair fate. Furthermore, the inability of the 35S::CPC construct to generate a nonuniform pattern of cell types in the cpc-1 background (as in the analogous 35S::WER wer-1 line) is consistent with Figure 4 and suggests that the CPC pathway does not regulate a secondary feedback loop. Finally, to determine whether a WER-dependent factor might contribute to the effect of 35S::CPC, we generated and examined 35S::CPC wer-1 GL2::GUS plants. These roots possessed an epidermal cell pattern (Table 1) and a GL2 expression pattern (Figure 6) similar to that of 35S::CPC, implying that WER gene function is not required for the 35S::CPC effect.
Figure 6.
35S::CPC Alters the Epidermal Cell Pattern.
Expression of the GL2::GUS reporter in the developing root epidermis of 4-day-old seedlings. 35S::CPC and 35S::CPC wer-1 plants were stained overnight, whereas the 35S::CPC cpc-1 line was stained for 3 hr. Bar = 50 μm.
DISCUSSION
Lateral inhibition is a type of cell-to-cell interaction whereby a cell adopting a particular fate prevents its neighbors from developing in the same way (Wigglesworth, 1940). Lateral inhibition is employed widely during animal development (Goriely et al., 1991; Heitzler and Simpson, 1991; Sternberg, 1993), and it has been invoked on several occasions to explain the formation of cell patterns in plants (Barlow and Carr, 1984; Korn, 1993; Hulskamp et al., 1994; Szymanski et al., 2000). However, to date, no molecular component, or its regulation, has been defined in any lateral inhibition pathway in plants.
In this study, we identified two fundamental features of the patterning mechanism responsible for the position-dependent distribution of epidermal cell types in the Arabidopsis root. First, we showed that lateral inhibition is involved in the patterning process and that two MYB-related proteins, WER and CPC, are components of this pathway. We found that this lateral inhibition pathway acts to prevent cells in a specific location (the H position) from adopting the primary cell fate (the nonhair fate). Second, we discovered regulatory feedback loops between WER and CPC that are used to establish differential expression of the downstream transcription factor gene GL2 and position-dependent cell fate. Specifically, we showed that WER is a positive regulator of CPC at the N position and that CPC is a negative regulator of the WER, GL2, and CPC genes at the H position (Figure 4). Together, these results provide a new framework for understanding the molecular determination of cell fate in plants.
This work highlights the important role of the WER gene in root epidermal patterning. WER contributes to the specification of both the nonhair cell fate, by inducing GL2, and the hair cell fate, by inducing CPC. Accordingly, proper spatial regulation of the WER gene is crucial for the normal cell-type pattern to arise. We have shown that expressing the WER gene throughout the epidermis, using the 35S::WER wer-1 line, leads to an abnormal pattern of GL2 gene expression (Figure 5, Table 2) and epidermal cell types (Table 1) that is not linked to cell position. Thus, WER is likely to be a target of positional cues, which may direct the cell-type pattern by inducing WER expression in the N position and/or inhibiting WER expression in the H position (Figure 4). Furthermore, it is likely that differential WER expression is initiated early in Arabidopsis embryogenesis, because the epidermal pattern is known to be established by the heart stage of embryogenesis (Lin and Schiefelbein, 2001).
Although we have found that CPC is an essential component of an N cell–specific lateral inhibition pathway, the precise mechanism of CPC action is unclear. It is conceivable that CPC moves from N cells into H cells via plasmodesmata and directly affects the transcription of the GL2, CPC, and WER genes. The cell-to-cell movement of a variety of plant proteins has been documented previously (Lucas et al., 1995; Perbal et al., 1996; Sessions et al., 2000; Nakajima et al., 2001), and developing epidermal cells are known to be coupled symplastically (Duckett et al., 1994). Furthermore, this possibility is consistent with our previous model for CPC and WER action, which proposes that the relative abundance of these proteins in the N and H positions is critical for establishing the cell pattern, because the truncated MYB protein encoded by CPC can interfere with WER-dependent transcriptional activation (Lee and Schiefelbein, 1999). An alternative possibility is that CPC indirectly influences transcription in the H cells by activating an as yet unidentified cell-to-cell signaling pathway in the N cells.
These results show that reciprocal regulation of the WER and CPC MYB genes, biased by positional cues, generates the position-dependent pattern of cell types in the Arabidopsis root epidermis (Figure 4). Numerous examples of cell-type patterns influenced by positional cues have been described in plants (Barlow and Carr, 1984; Berger et al., 1998b; Hung et al., 1998; Geisler et al., 2000), and these may be governed by a mechanism similar to the one described here. Furthermore, because we have shown that this same machinery can generate a position-independent cell pattern if its linkage to positional cues is severed (e.g., the pattern in the 35S::WER wer-1 line), a common mechanism involving lateral inhibition with feedback may be employed to produce both position-dependent and position-independent patterns of cell types in plants. For example, the nonrandom distribution of trichome cells in Arabidopsis may be generated by similar patterning elements, particularly given that some of the same components described here are used in trichome specification, such as the GL2 gene (Koornneef, 1981; Rerie et al., 1994) and a MYB gene (GLABROUS1; Oppenheimer et al., 1991) that is functionally equivalent to WER (Lee and Schiefelbein, 2001). Finally, a conceptually similar mechanism of lateral inhibition with feedback operates to pattern some animal cell types (e.g., the Notch signaling system; Artavanis-Tsakonas et al., 1999), which implies that despite their independent evolutionary origin, multicellular plants and animals may have adopted fundamentally similar strategies to generate defined patterns of cell fates.
METHODS
Plant Materials and Growth Conditions
The mutant alleles of Arabidopsis thaliana used in this study were isolated previously: cpc-1 (Wada et al., 1997), gl2-1 (Koornneef, 1981), and wer-1 (Masucci et al., 1996), and these are likely to represent loss-of-function alleles. The GL2::β-glucuronidase (GUS), WER::green fluorescent protein (GFP), 35S::CPC, and 35S::WER lines have been reported previously (Masucci et al., 1996; Wada et al., 1997; Lee and Schiefelbein, 1999). The CPC::GUS line was a kind gift from T. Wada and K. Okada (Kyoto University, Kyoto, Japan) and includes a 1.2-kb CPC promoter region sufficient to complement the cpc-1 mutation (Wada et al., 1997). The GL2::GFP line has been described (Lin and Schiefelbein, 2001). Lines with multiple mutations and/or various transgene combinations were generated by crossing, and their genotypes were confirmed by backcrossing to single mutants, reporter gene expression, and/or polymerase chain reaction–based tests. Seedlings were grown in vertically oriented Petri dishes on agarose-solidified medium containing mineral nutrients as described previously (Schiefelbein and Somerville, 1990).
Microscopy
The pattern of epidermal cell types was determined by staining 5-day- old roots with propidium iodide, followed by examination with a fluorescence microscope to determine the cell type and relative locations of epidermal cells. The proportion of hair and nonhair cell types in the root epidermis was determined by examining at least 10 5-day-old seedlings from each strain. An epidermal cell was scored as a root hair cell if any protrusion was visible, regardless of its length.
The GUS constructs were introduced into the indicated mutant and transgene lines by genetic crosses, and histochemical analysis of GUS activity was performed essentially as described (Masucci et al., 1996). The spatial expression of GL2::GUS was assessed in a series of transverse sections from the meristematic region of 4-day-old roots that were fixed and embedded in JB-4 resin after GUS staining, as described (Masucci et al., 1996). The proportion of cells expressing GL2::GUS was determined by examining sections from at least five 4-day-old seedlings from each strain.
The WER::GFP and GL2::GFP constructs were crossed into the various mutant and transgenic lines. GFP accumulation was visualized in 5-day-old roots after propidium iodide staining (10 μg/mL) using a Zeiss (Jena, Germany) LSM510 confocal microscope.
To assess the expression of both GFP and GUS in the same roots, 5-day-old seedlings were analyzed by fluorescence microscopy with a supporting cover slip and then subjected immediately to GUS assay by infusing 5-bromo-4-chloro-3-indolyl-β-glucuronic acid solution under the cover slip and staining for 3 hr under previously published conditions (Masucci et al., 1996).
Molecular Biology Methods
RNA was isolated from root tips from the genotypes indicated in the figures and the text and and subjected to RNA gel blot analysis as described previously (Masucci et al., 1996). The gene-specific probes used here have been described for GL2 (Masucci et al., 1996) and WER (Lee and Schiefelbein, 1999). A 300-bp fragment including most of the CPC gene was found to be CPC specific (data not shown) and was used in the CPC RNA gel blot experiments.
Acknowledgments
We are grateful to Takuji Wada and Kiyotaka Okada for providing the CPC::GUS and 35S::CPC lines and for communicating results before publication. We also thank Steve Clark, Jianming Li, and Kiyotaka Okada for helpful comments. This work was supported by National Science Foundation Grant No. IBN-0077900.
Article, publication date, and citation information can be found at www.plantcell.org/cgi/doi/10.1105/tpc.010434.
References
- Artavanis-Tsakonas, S., Rand, M.D., and Lake, R.J. (1999). Notch signaling: Cell fate control and signal integration in development. Science 284, 770–776. [DOI] [PubMed] [Google Scholar]
- Barlow, P.W., and Carr, D.J. (1984). Positional Controls in Plant Development. (Cambridge, UK: Cambridge University Press).
- Berger, F., Haseloff, J., Schiefelbein, J., and Dolan, L. (1998. a). Positional information in root epidermis is defined during embryogenesis and acts in domains with strict boundaries. Curr. Biol. 8, 421–430. [DOI] [PubMed] [Google Scholar]
- Berger, F., Linstead, P., Dolan, L., and Haseloff, J. (1998. b). Stomata patterning on the hypocotyl of Arabidopsis thaliana is controlled by genes involved in the control of root epidermis patterning. Dev. Biol. 194, 226–234. [DOI] [PubMed] [Google Scholar]
- Bunning, E. (1951). Uber die Differenzierungsvorgange in der Cruciferenwurzel. Planta 39, 126–153. [Google Scholar]
- Cormack, R.G.H. (1935). The development of root hairs by Elodea canadensis. New Phytol. 34, 19–25. [Google Scholar]
- DiCristina, M.D., Sessa, G., Dolan, L., Linstead, P., Baima, S., Ruberti, I., and Morelli, G. (1996). The Arabidopsis Athb-10 (GLABRA2) is an HD-Zip protein required for regulation of root hair development. Plant J. 10, 393–402. [DOI] [PubMed] [Google Scholar]
- Duckett, C.M., Oparka, K.J., Prior, D.A.M., Dolan, L., and Roberts, K. (1994). Dye-coupling in the root epidermis of Arabidopsis is progressively reduced during development. Development 120, 3247–3255. [Google Scholar]
- Geisler, M., Nadeau, J., and Sack, F.D. (2000). Oriented asymmetric divisions that generate the stomatal spacing pattern in Arabidopsis are disrupted by the too many mouths mutation. Plant Cell 12, 2075–2086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goriely, A., Dumont, N., Dambly-Chaudiere, C., and Ghysen, A. (1991). The determination of sense organs in Drosophila: Effect of neurogenic mutations in the embryo. Development 113, 1395–1404. [DOI] [PubMed] [Google Scholar]
- Heitzler, P., and Simpson, P. (1991). The choice of cell fate in the epidermis of Drosophila. Cell 64, 1083–1092. [DOI] [PubMed] [Google Scholar]
- Hulskamp, M., Misera, S., and Jurgens, G. (1994). Genetic dissection of trichome cell development in Arabidopsis. Cell 76, 555–566. [DOI] [PubMed] [Google Scholar]
- Hung, C.Y., Lin, Y., Zhang, M., Pollock, S., Marks, M.D., and Schiefelbein, J. (1998). A common position-dependent mechanism controls cell-type patterning and GLABRA2 regulation in the root and hypocotyl epidermis of Arabidopsis. Plant Physiol. 117, 73–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koornneef, M. (1981). The complex syndrome of ttg mutants. Arabidopsis Inf. Serv. 18, 45–51. [Google Scholar]
- Korn, R.W. (1993). Evidence in dicots for stomatal patterning by inhibition. Int. J. Plant Sci. 154, 367–377. [Google Scholar]
- Lee, M.M., and Schiefelbein, J. (1999). WEREWOLF, a MYB-related protein in Arabidopsis, is a position-dependent regulator of epidermal cell patterning. Cell 99, 473–483. [DOI] [PubMed] [Google Scholar]
- Lee, M.M., and Schiefelbein, J. (2001). Developmentally distinct MYB genes encode functionally equivalent proteins in Arabidopsis. Development 128, 1539–1546. [DOI] [PubMed] [Google Scholar]
- Lin, Y., and Schiefelbein, J. (2001). Embryonic control of epidermal cell patterning in the root and hypocotyl of Arabidopsis. Development 128, 3697–3705. [DOI] [PubMed] [Google Scholar]
- Lucas, W.J., Bouche-Pillon, S., Jackson, D.P., Nguyen, L., Baker, L., Ding, B., and Hake, S. (1995). Selective trafficking of KNOTTED1 homeodomain protein and its mRNA through plasmodesmata. Science 270, 1980–1983. [DOI] [PubMed] [Google Scholar]
- Masucci, J.D., Rerie, W.G., Foreman, D.R., Zhang, M., Galway, M.E., Marks, M.D., and Schiefelbein, J.W. (1996). The homeobox gene GLABRA2 is required for position-dependent cell differentiation in the root epidermis of Arabidopsis thaliana. Development 122, 1253–1260. [DOI] [PubMed] [Google Scholar]
- Nakajima, K., Sena, G., Nawy, T., and Benfey, P.N. (2001). Intercellular movement of the putative transcription factor SHR in root patterning. Nature 413, 307–311. [DOI] [PubMed] [Google Scholar]
- Oppenheimer, D., Herman, P.L., Sivakumaran, S., Esch, J., and Marks, M.D. (1991). A myb gene required for leaf trichome differentiation in Arabidopsis trichomes is expressed in stipules. Cell 67, 483–493. [DOI] [PubMed] [Google Scholar]
- Perbal, M., Haughn, G., Saedler, H., and Schwarz-Sommer, Z. (1996). Non-cell-autonomous function of the Antirrhinum floral homeotic proteins DEFICIENS and GLOBOSA is exerted by their polar cell-to-cell trafficking. Development 122, 3433–3441. [DOI] [PubMed] [Google Scholar]
- Rerie, W.G., Feldmann, K.A., and Marks, M.D. (1994). The GLABRA2 gene encodes a homeodomain protein required for normal trichome development in Arabidopsis. Genes Dev. 8, 1388–1399. [DOI] [PubMed] [Google Scholar]
- Schiefelbein, J.W., and Somerville, C. (1990). Genetic control of root hair development in Arabidopsis thaliana. Plant Cell 2, 235–243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sessions, A., Yanofsky, M.F., and Weigel, D. (2000). Cell–cell signaling and movement by the floral transcription factors LEAFY and APETALA1. Science 287, 779–782. [DOI] [PubMed] [Google Scholar]
- Sternberg, P.W. (1993). Falling off the knife edge. Curr. Biol. 3, 763–765. [DOI] [PubMed] [Google Scholar]
- Szymanski, D.B., Lloyd, A.M., and Marks, M.D. (2000). Progress in the molecular genetic analysis of trichome initiation and morphogenesis in Arabidopsis. Trends Plant Sci. 5, 214–219. [DOI] [PubMed] [Google Scholar]
- Wada, T., Tachibana, T., Shimura, Y., and Okada, K. (1997). Epidermal cell differentiation in Arabidopsis determined by a Myb homolog, CPC. Science 277, 1113–1116. [DOI] [PubMed] [Google Scholar]
- Wigglesworth, V.B. (1940). Local and general factors in the development of ‘pattern’ in Rhodnius prolixus (Hemiptera). J. Exp. Biol. 17, 180–200. [Google Scholar]