Abstract
Replication-competent virus vectors are attractive therapeutic agents for cancer. G207, a second-generation, multimutated herpes simplex virus type 1 (HSV-1), is one such vector that is safe in primates and efficacious against human tumors in athymic mice. Squamous cell carcinoma is the most frequently encountered malignancy of the head and neck, and the chemotherapeutic agent cisplatin is a standard treatment for recurrent head and neck cancer. In this study we examine the therapeutic potential of G207, alone and in combination with cisplatin, against squamous cell carcinoma. Human squamous cell carcinoma cell lines are sensitive to G207 replication and cytotoxicity in vitro at a multiplicity of infection of 0.01, including cisplatin sensitive (UMSCC-22A), moderately sensitive (UMSCC-38), and weakly sensitive (SQ20B) cell lines. Cisplatin did not inhibit the cytopathic effect of G207. G207 inhibited the growth of established subcutaneous head and neck tumors in athymic mice. The therapeutic effects of cisplatin and G207 in vivo were independent. However, in cisplatin-sensitive tumors (UMSCC-38), combination therapy resulted in 100% cures in contrast to 42% with G207 or 14% with cisplatin alone. We conclude that G207 should be considered for the treatment of head and neck cancer and that combination with chemotherapeutic agents may improve efficacy.
Keywords: herpes simplex virus, head and neck cancer, squamous cell carcinoma, cisplatin, gene therapy
Introduction
Cancers of the head and neck, which are mostly located in the oral cavity, pharynx and larynx, affect over 50,000 persons each year in the United States and cause about 14,000 deaths [1]. Squamous cell carcinoma accounts for 85%–90% of tumors classified as head and neck cancer [2]. Surgery and radiotherapy are effective in controlling early stages of head and neck squamous cell carcinoma (HNSCC), but overall survival is still poor due to the lack of a complete response. Recurrent head and neck cancer causes serious complications from local-regional disease. Necrosis or ulceration of the tumor creates disabling pain, infection, cranial nerve dysfunction, airway compromise, impairment of speech, and swallowing dysfunction with dehydration and malnutrition. Until recently, chemotherapy has been used almost exclusively for palliation in relapsed head and neck cancer but has had relatively little role in the curative therapy of these tumors.
Cisplatin, as a single agent, is a standard therapy for recurrent or metastatic head and neck cancer [3,4], but is more effective (70%–90%) as a combination chemotherapy in previously untreated patients [5–7]. Impressive tumor responses to chemotherapy are often initially observed, however, in patients who relapse after surgery and radiation, the tumors are usually chemotherapy resistant. Unfortunately, recurrent and metastatic disease, which generally can only be treated with chemotherapy, affects a large and important subset of patients [8]. Forty percent to 60% of patient deaths are directly attributable to uncontrolled local-regional disease [9], and 90% of patients with distant metastases die with uncontrolled tumor at the primary site or in the neck [10]. Reirradiation with chemotherapy of unresectable recurrent head and neck cancer was shown to prolong survival compared to either modality alone, and yet this dual modality of treatment caused over 10% mortality [11]. Thus safe, local, innovative therapies with or without chemotherapy are needed to palliate patients who will develop and die of local-regional recurrences.
We have developed an attenuated, multimutated, replication-competent herpes simplex virus (HSV) vector termed G207, which has proved efficacious in treating human malignant tumors [12–14]. G207 has deletions at both γ34.5 (RL1) loci and an insertion of the Escherichia coli lacZ gene inactivating the ICP6 gene (UL39) [12]. These mutations result in attenuated neurovirulence, temperature sensitivity, ganciclovir hypersensitivity, and expression of an easily detectable histochemical marker (β-galactosidase) [12]. Cytopathic effect has been demonstrated in vitro in multiple human tumor cell lines. Intraneoplastic inoculation of G207 into established subcutaneous or intracerebral tumors in mice leads to decreased tumor growth and prolonged survival [12–14]. However, G207 is nonpathogeneic when inoculated intracerebrally in mice and in HSV-sensitive, nonhuman primates [12] and is currently undergoing clinical evaluation for the treatment of recurrent malignant glioma in a phase I trial.
Combination therapies (radiation/chemotherapy, surgery/radiation, surgery/chemotherapy, multiple chemotherapeutic agents, and so on.) have proved to be generally more effective than single therapeutic agents alone in the treatment of cancer. Unfortunately, in randomized trials for head and neck cancer, combination chemotherapy failed to improve survival rate when compared with single agent chemotherapy [15–18]. Recently, such combination therapies have been extended to cancer gene therapy, in which examples include replication-competent HSV + “suicide” gene therapy [19,20], replication-competent adenovirus + “suicide” gene therapy [21], replication-competent adenovirus + chemotherapy [22], replication-competent HSV + cytokine immunotherapy [23,24], replication-competent HSV + radiation therapy [25], and p53 gene therapy + chemotherapy [26,27]. In this study we show that the combination of cisplatin with a replication-competent HSV vector, G207, provides an additive therapeutic response that can eradicate established subcutaneous human HNSSC tumors in athymic mice.
Materials and Methods
Viruses and Cells
G207 is an HSV-1 mutant containing deletions in both copies of the γ34.5 gene and an E coli lacZ insertion inactivating the ICP6 gene [12]. G207 (clinical grade) was obtained from NeuroVir, Inc (Vancouver, BC). Titration of virus was performed by plaque assay on African green monkey kidney (Vero) cells. The human squamous cell carcinoma cell lines were established in other laboratories from tumors of the aerodigestive tract. UM-SCC 22A, UM-SCC 38, PCI 51, and SCC-25/CP cell lines were grown in Dulbecco's modified Eagle's medium (DMEM) containing 10% fetal calf serum (FCS) (Hyclone, Logan, UT); 1% penicillin-streptomycin (Sigma Chemical Co., St Louis, MO); and 1% glutamine. SQ20B was grown in DMEM containing 20% FCS (Hyclone); 1% MEM nonessential amino acids (Gibco PBL, Grand Island, NY); hydrocortisone (0.4 µg/mL; Sigma Chemical Co); 1% penicillin-streptomycin; and 1% glutamine.
Cell Culture Cytotoxicity
Subconfluent monolayers of HNSSC cells in 6-well dishes were infected with G207 at multiplicities of infection (MOIs) of 0.1 and 0.01 plaque-forming units (pfu) per cell, whereas controls were treated with phosphate-buffered saline (PBS) (with glucose) and 1% heat-inactivated FCS. Viable cell numbers were determined by counting trypan blue-excluding cells.
For the cisplatin cytotoxicity assay, cells were plated in 96-well dishes (104 cells per well) in improved minimum essential medium, Eagle's (IMEM) (without phenol red) and 10% FCS. Twenty four hours after plating, cells were treated with aqueous cisplatin (cis-diamminedichloroplatinum [II] obtained from the Georgetown University Hospital Pharmacy) at concentrations ranging from 3.9 x 10−7 to 10−4 mol/L, and then grown in cisplatin for 4 days. Cell growth and viability were determined by XTT assay (Polysciences, Inc, Warrington, PA) at an optical density of 450 nm. The cells treated with cisplatin were compared with control cells not treated with cisplatin.
In Vitro Cytotoxicity with Cisplatin and G207
Subconfluent monolayers of SCC-25/CP HNSCC cells in 6-well dishes were infected in triplicate with G207 at a MOI of 0.01 pfu/cell and treated with cisplatin at 0.0075 or 0.001 mmol/L or PBS. Viable cell counts were determined by trypan blue exclusion on days 1 to 5.
Subcutaneous Tumor Model
Six-week-old female outbred athymic nu/nu mice were purchased from the National Cancer Institute (Frederick, MD). All animal procedures were approved by the Georgetown University Animal Care and Use Committee. For surgical procedures, each mouse was anesthetized with a 0.25 to 0.30 mL intraperitoneal injection of a solution consisting of 84% bacteriostatic saline; 10% sodium pentobarbital (50 mg/mL; Abbott Laboratories, Chicago, IL); and 6% ethyl alcohol.
Tumor fragments of UM-SCC 22A (3 mm x 2 mm x 2 mm) or UM-SCC 38 (3 mm x 3 mm x 2 mm) or SQ20B cells (2 x 106) were implanted subcutaneously in the left flanks of athymic mice (n = 40/tumor cell line). When the subcutaneous (s.c.) tumors were approximately 6 mm in maximal diameter (approximately 14 days postimplant), mice were randomly divided into two or four groups (n = 7 or 8). Mice harboring UM-SCC 22A or UM-SCC 38 s.c. tumors were inoculated intraneoplastically with G207 (107 pfu in 20 µL) or 20 µL mock extract. Mice harboring SQ20B s.c. tumors were inoculated intraneoplastically with 10-fold less G207 (106 pfu in 20 µL) or 20 µL mock extract. Cisplatin (4 mg/kg/100µL) or PBS was intraperitoneally injected for 5 consecutive days starting day 2 or 3 after G207 injection. Cisplatin (20 mg/kg per mouse total) will achieve maximal antitumoral activity with minimal side effects [29]. Mice were weighed before cisplatin or PBS treatment and twice weekly after treatment.
Tumor growth was determined by measuring the tumor volume (length x width x height) twice weekly. Mice were sacrificed when they appeared moribund or their tumor diameter reached 20 mm for UMSCC-38 and SQ20B tumors or tumor volume reached 1000 mm3 for UMSCC-22A tumors, and the date of sacrifice was recorded for survival studies.
Statistical differences in growth ratio were assessed using a Student t test. Statistical analysis of Kaplan-Meier survival curves was by Wilcoxon test.
Results
In Vitro Cytoxicity
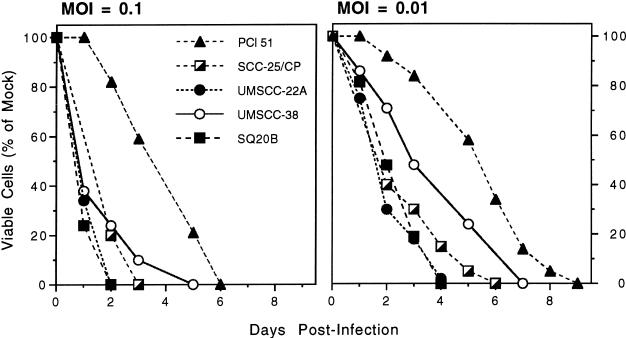
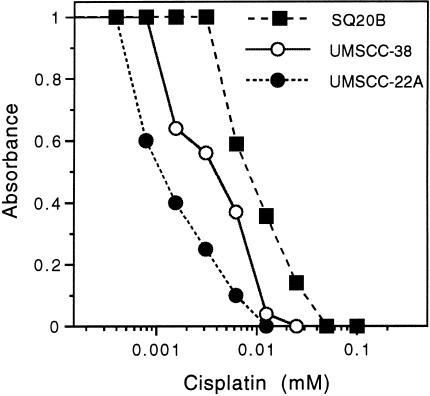
We determined the in vitro susceptibility of human HNSSC cells to G207 replication, spread, and cytotoxicity. All cell lines (PCI 51, SCC-25/CP, UMSCC-22A, UMSCC-38 and SQ20B) were destroyed (> 99% cytotoxicity) by G207 infection at a MOI = 0.01 (Figure 1). UMSCC-22A and SQ20B cells were the most sensitive and PCI-51 cells the least sensitive to G207. The sensitivity to cisplatin varied from an IC50 of approximately 0.001 mmol/L for UMSCC-22A to 0.009 mmol/L for SQ20B, with UMSCC-38 in between with an IC50 of 0.003 mmol/L (Figure 2). The IC50 was defined as the concentration of cisplatin causing a 50% decrease in control absorbance.
Figure 1.
In vitro susceptibility of human head and neck squamous cell carcinoma (HNSCC) cell lines to G207. Subconfluent monolayers of head and neck cancer cell lines (PCI 51, SCC-25/CP, UMSCC-22A, UMSCC-38, and SQ20B) were infected with G207 at a MOI of 0.1 (left) or 0.01 (right) and the viable cells counted on the days indicated. The data plotted are the mean of triplicate wells.
Figure 2.
Sensitivity of HNSCC cell lines to cisplatin cytotoxicity. Cells were plated at equal density on day 0 and treated with increasing concentrations of cisplatin on day 1. Cell viability was determined on day 4 by XTT assay. The IC50 for the most resistant cell line, SQ20B, was about 9 times greater than the least resistant, UMSCC-22A.
Effect of Cisplatin on G207
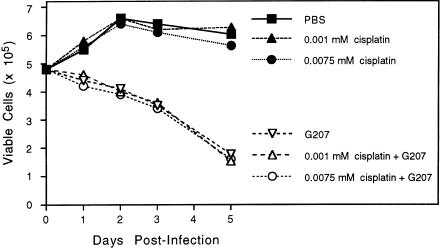
We used the most cisplatin-resistant cell line, SCC-25/CP [30], to determine the effect of cisplatin on G207 replication and cytotoxicity in vitro. Subconfluent monolayers of SCC-25/CP cells were infected with G207 at a MOI = 0.01 and treated with two dilutions of cisplatin, one being the highest clinically achievable (0.0075 mmol/L) and the other the usual clinical dosage (0.001 mmol/L). All control cells (not infected with G207) grew to confluency after 2 days, irrespective of cisplatin concentration. There was no difference in the cytotoxicity of G207 with or without cisplatin (Figure 3). SCC-25/CP cells were similar to UMSCC-38 cells in sensitivity to G207 (see Figure 1; MOI 0.01).
Figure 3.
Effect of cisplatin on G207 growth and cytotoxicity. A subconfluent monolayer of SCC-25/CP cells were infected with G207 at a MOI of 0.01 or mock infected with PBS and then treated with 0.0075 or 0.001 mmol/L cisplatin or PBS. The cells were not sensitive to cisplatin at these doses and grew to confluence in 48 hours; they remained confluent for the following 3 days. There was no significant difference in the cytopathic efficacy of G207 at either of the cisplatin concentrations.
G207 Treatment of Subcutaneous Tumors
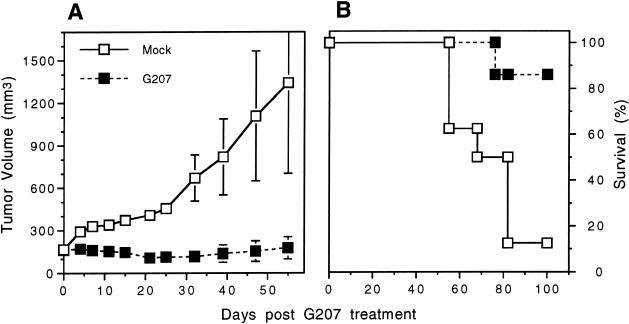
We first examined the in vivo efficacy of G207 against established UMSCC-22A s.c. tumors, as these cells were the most sensitive to G207 in vitro. When the s.c. tumors reached 6 mm in maximal diameter, they were treated with a single intraneoplastic inoculation of G207 or PBS (control). In the control group (n = 8), tumors continued to grow steadily for 4 more weeks. The mean tumor growth ratio was significantly inhibited in the G207-treated group (n = 7) compared with the control group (p < 0.05 on day 47; Student t test) (Figure 4). On day 55, one control animal was found dead with ascites and intraperitoneal metastases, and on day 78, two more control animals were sacrificed due to ascites and emaciation. Four control and one G207-treated animal were sacrificed due to tumor burden (> 1000 mm3) on day 78. All the remaining G207-treated animals (6/7) were alive at day 100, compared with only one control animal (1/8). The tumors from these surviving animals had regressed to scars or 10 to 30 mm3 necrotic masses when autopsied.
Figure 4.
G207 inhibition of UMSCC-22A subcutaneous tumor growth. Athymic mice bearing s.c. UMSCC-22A tumors, 6 mm in maximal diameter, were treated with a single intraneoplastic inoculation of G207 (107 pfu; n = 8) or PBS (mock; n = 7). (A) The tumors were measured on the days indicated and tumor volume determined. Mean tumor growth was significantly inhibited in the G207-treated tumors compared to the mock tumors (P ≤ .05 from day 11 to 47 and P = .07 on day 55; Student t test). Bars are SEM. (B) Kaplan-Meier survival curve. There was a significant difference in survival between the control and G207-treated groups (P < .01; Wilcoxon test).
Combination Therapy of Subcutaneous Tumors
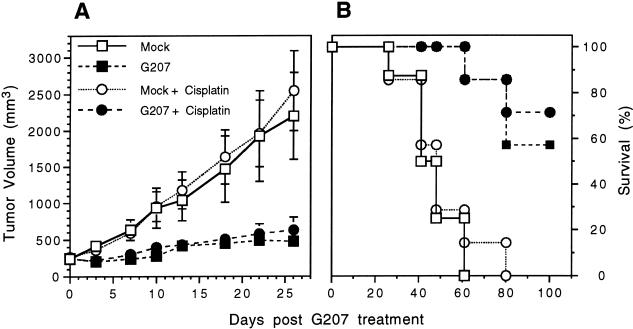
We next compared the effects of combination therapy, G207 + cisplatin, on s.c. tumor growth of a moderately cisplatin sensitive (UMSCC-38) and a weakly cisplatin sensitive (SQ20B) tumor. SQ20B cells were more sensitive to G207 in vitro than UMSCC-38 (see Figure 1) and the SQ20B s.c. tumors were inoculated with a single low dose of G207 (106 pfu). There was no inhibition or difference in the mean tumor growth of SQ20B between the cisplatin-treated group and the control group (Figure 5A; P = .35, Student t test). There was a significant inhibition of tumor growth in the G207-treated group compared with the control group (Figure 5A; P < .025, Student t test). However, there was no difference in the mean tumor growth between the combination therapy group, G207 + cisplatin, and the G207-treated group (Figure 5A; P = .2, Student t test). All control animals were sacrificed by day 61 and all cisplatin-treated animals by day 80 due to tumor burden and/or ulceration (Figure 5B). This is in contrast to the 4 of 7 surviving G207-treated animals and 5 of 7 surviving G207 + cisplatin-treated animals at day 100, which is a statistically significant difference in survival compared with the control or cisplatin-treated groups (Figure 5B; P < .005, Wilcoxon test).
Figure 5.
G207 inhibition of SQ20B subcutaneous tumor growth. Athymic mice bearing s.c. SQ20B tumors, 6 mm in maximum diameter, were treated with a single intraneoplastic inoculation of G207 (106 pfu; n = 14) or PBS (mock; n = 15). Cisplatin was injected intraperitoneally 2 days later for 5 consecutive days (4 mg/kg/day) in a subset of the G207-treated (n = 7) and mock-treated animals (n = 7). (A) The tumors were measured on the days indicated and tumor volume determined. Cisplatin treatment alone had no effect on tumor growth. Mean tumor growth was significantly inhibited in the G207-treated tumors compared with the mock tumors (P < .025; Student t test) and in the G207 + cisplatin animals compared to the mock + cisplatin animals (P < .005; Student t test). There was no significant difference between the G207 and G207 + cisplatin groups in the mean tumor growth ratio by day 26 (P = .23; Student t test). Bars are SEM. (B) Kaplan-Meier survival curve. Treatment with G207 or G207 + cisplatin significantly increased survival compared with mock or mock + cisplatin (P < .005; Wilcoxon test). There was no significant difference in survival between Mock and Mock + cisplatin groups (P = .77; Wilcoxon test) or between G207 and G207 + cisplatin groups (P = .65; Wilcoxon test). All mock-treated and mock + cisplatin-treated animals were sacrificed by days 61 and 80, respectively. On day 100, four G207- and five G207 + cisplatin-treated mice were alive. Among these survivors, one G207 + cisplatin animal had a tumor 12 mm in maximal diameter, and the remaining had tumors smaller than at the initiation of treatment (< 6 mm in maximal diameter).
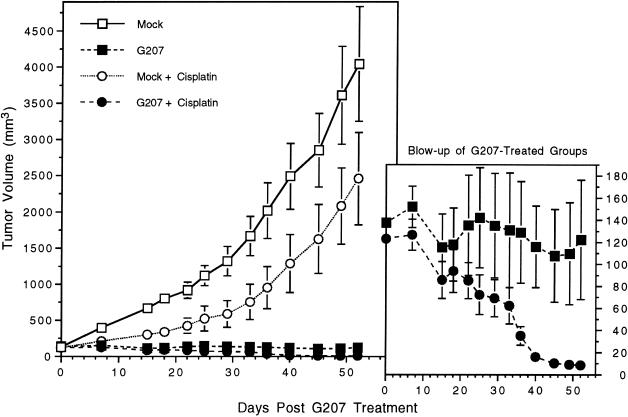
UMSCC-38 cells were moderately sensitive to cisplatin in vitro (see Figure 2), and UMSCC-38 s.c. tumors were similarly moderately sensitive to cisplatin in vivo (Figure 6). Tumor growth was inhibited by cisplatin but was not significantly different than the control group (Figure 6; P > .8, Student t test). In the cisplatin group, one tumor failed to grow and regressed to a scar after cisplatin treatment. G207 treatment (107 pfu), both with or without cisplatin, of established UMSCC-38 s.c. tumors significantly inhibited growth when compared with the control animals (Figure 6; P < .0005, Student t test) or the cisplatin-treated animals (Figure 6; P < .005, Student t test). There was also a significant difference in the mean tumor growth ratio between the G207 and G207 + cisplatin-treated groups (Figure 6; P = .04 on day 52, Student t test). In the G207 + cisplatin-treated group (n = 7) all s.c. tumors were cured in contrast to three in the G207-treated group (n = 7).
Figure 6.
G207 + cisplatin inhibition of UMSCC-38 s.c. tumor growth. Athymic mice bearing s.c. UMSCC-38 tumors, 6 mm in maximal diameter, were treated with a single intraneoplastic inoculation of G207 (107 pfu; n = 14) or PBS (mock; n = 16). Cisplatin was injected intraperitoneally 3 days later for 5 consecutive days (4 mg/kg/day) in a subset of the G207-treated (n = 7) and mock-treated animals (n = 8). Bars are SEM. Mock + cisplatin-treated animals had a decrease in tumor growth compared with mock, although the difference was not significant (P < .085; Student t test). G207 or G207 + cisplatin treatment significantly inhibited tumor growth when compared with mock (P < .0005; Student t test) or mock + cisplatin (P < .005; Student t test). There was also a significant difference between G207 and G207 + cisplatin groups on day 52 (P < .04; Student t test), with all tumors cured in the G207 + cisplatin group (7/7) compared with only 3/7 in the G207-treated group. In the Mock + cisplatin group, one tumor regressed to a scar after cisplatin treatment.
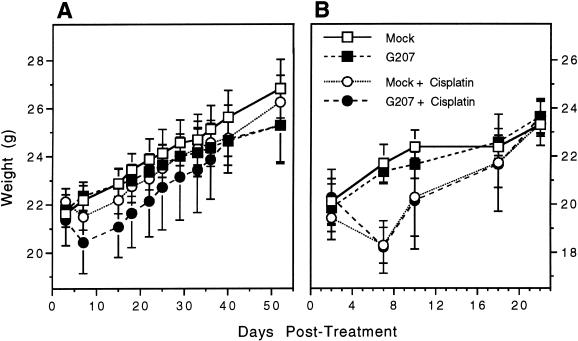
The mean starting body weight of each group before treatment was 21.0 ± 0.5 g. Cisplatin treatment resulted in a drop in body weight of 0.5 to 2 g (2.5%–10% body weight change) within 3 to 4 days (Figure 7). Within 3 weeks after treatment, body weights from all groups were about the same, 22.5 ± 0.5 g.
Figure 7.
Toxicity of tumor therapy. Weight of mice bearing UMSCC-38 (A) and SQ20B (B) tumors. Mice were weighed before initiation of cisplatin treatment and at every tumor measurement after treatment. The starting mean weights were 22.0 ± 0.5 g for UMSCC-38 and 20.0 ± 0.5 g for SQ20B. Bars are SEM. Cisplatin treatment resulted in a decrease in body weight of about 1.5 g (0.5–2.0 g or 2.5%–10% of body weight) within 3 to 4 days of treatment. By 10 to 12 days post-treatment, body weights recovered to their original weight, and by 3 weeks post-treatment body weights were equal to those of the animals not treated with cisplatin. There was no difference in body weights between mock and G207-treated animals.
Discussion
We initially demonstrated that G207, with deletions at both and γ34.5 loci and lacZ insertion in the ICP6 gene, has therapeutic potential for central nervous system tumors such as meningioma [13] and glioma [12]. We have now expanded the use of G207 to the treatment of HNSCC, which constitute about 10% of cancers worldwide and cause approximately 5% of all cancer deaths [31]. The rational for using this new treatment modality in head and neck cancers include 1) standard therapeutic intervention such as surgery, radiation and chemotherapy continue to have a low 5-year survival rate (8%–62%) [32]; 2) to date, no trial has demonstrated improved survival as a consequence of adjuvant chemotherapy or combination chemotherapy [31]; and 3) the location of HNSCC is easily accessible to local G207 inoculation because local invasion is the primary cause of patient morbidity and mortality.
All human HNSCC cell lines tested were very sensitive to G207 cytotoxicity in vitro. G207 was able to significantly inhibit the growth of established UMSCC-22A, UMSCC-38, and SQ20B s.c. tumors in vivo. In addition, G207 treatment prevented metastasis in UMSCC-22A tumor-bearing animals, whereas 37% of the control-treated animals had signs of tumor metastases. It has recently been shown that retroviral vector HSV-tk/GCV therapy can have a distant bystander effect on noninoculated human head and neck tumors in outbred athymic mice, possibly because of the actions of natural killer cells, monocytes, macrophages, and/or B cells [33,34]. In patients, surgery or radiotherapy improve local and regional control, but this has not had a significant impact on survival because of deaths due to metastatic disease [35,36].
Cisplatin, a cell cycle nonspecific drug, produces predominantly intrastrand DNA cross-links [37]. Cisplatin cross-links act as blocks to DNA replication [38,39], with cisplatin-resistant human cancer cells having higher levels of replicative bypass than sensitive cells [40]. G207 causes cell death during viral replication and generation of new infectious virus and spreads to neighboring tumor cells to repeat the process. In order for G207 to spread and destroy the tumor, it must undergo extensive DNA replication and transcription, processes that could be blocked by cisplatin. It was, therefore, essential to determine whether G207 replication, spread, and cytotoxicity were inhibited by cisplatin. In order to test the effect of high cisplatin doses on G207, we used a HNSCC cell line that was relatively resistant to cisplatin so that cytopathic effects of the drug would not complicate the analysis. The SCC-25/CP cell line was derived from SCC-25 by passage in escalating concentrations of cisplatin [28] and is a little more resistant to cisplatin than SQ20B [30]. Cisplatin had no effect on the cytopathic ability of G207. It has been shown that intraperitoneal injection of cisplatin was unable to significantly inhibit HSV-2 infection in a mouse genital model [41]. Biochemical studies indicate that HSV DNA helicase-primase complex in the presence of HSV DNA binding protein ICP8 is able to bypass cisplatin intrastrand cross-links in DNA [42]. The demonstration that DNA damaging agents, cisplatin and cyclophosphamide [20], are not inhibitory to HSV replication at doses that are toxic to growing cells provides an additional means to preferentially target tumor cells for death without affecting the oncolytic virus.
When the combination therapy G207 + cisplatin was used in vivo, we found that it had an additive tumoricidal effect on established UMSCC-38 s.c. tumors, which are moderately sensitive to cisplatin, but no increased efficacy over G207 alone against SQ20-B s.c. tumors, which are only weakly sensitive to cisplatin. This supports the notion that cisplatin and G207 are acting independently in this system. G207 is effective against most human tumor cells, irrespective of their chemotherapy or radiation sensitivity. In addition, G207 did not increase the overall toxicity associated with cisplatin treatment, as determined by weight loss. There was no ulceration of the skin at the site of the tumor nor any neurological changes associated with the combination treatment (data not shown). Combination therapies with other oncolytic viral vectors and chemotherapy or radiation have shown increased antitumor efficacy; the E1B-mutated adenovirus in combination with cisplatin or 5-fluorouracil was more effective at inhibiting human laryngeal carcinoma xenografts than either therapy alone [22], and radiation therapy enhanced the antitumor activity of an attenuated, replication-competent HSV against human glioma xenografts [25].
These experiments demonstrated that G207 is an effective treatment for head and neck cancer, even for tumors that are relatively cisplatin resistant, and should be considered for clinical evaluation. The combination of G207 + cisplatin significantly improved efficacy in tumors that were moderately sensitive to cisplatin without increasing toxicity. It may also be worthwhile to explore such combination therapies for other tumor types.
Acknowledgements
The human squamous cell carcinoma cell lines UM-SCC 22A and UM-SCC 38 were kindly provided by Dr. Thomas Carey (University of Michigan Cancer Center, Ann Arbor, Michigan); PCI 51 by Dr. Theresa Whiteside (University of Pittsburgh, Pittsburgh, PA); SCC-25/CP (28) by Dr. J. Lazo (University of Pittsburgh); and SQ20B by Dr. R. Weichselbaum (University of Chicago, Chicago, IL). We thank Dr. Sundaresan Periasamy for his suggestions and help with the animals, Anu Iyer for technical assistance, Dr. Joseph T. Newsome and the Georgetown University Research Resource Facility staff for assistance with the animals, and Paul Johnson for providing purified G207. We also thank Dr. Kevin Cullen for his suggestions and members of his laboratory for technical assistance. This work was supported in part by a grant from the National Institutes of Health (NS32677 to R.L.M.). Samuel D. Rabkin and Robert L. Martuza are consultants to NeuroVir, Inc, which has a license from Georgetown University for G207.
Abbreviations
- HNSCC
head and neck squamous cell carcinoma
- HSV
herpes simplex virus
- MOI
multiplicity of infection
- pfu
plaque-forming unit
- s.c.
subcutaneous
References
- 1.Parker SL, Tong T, Bolden S, Wingo PA. Cancer statistics, 1997. CA Cancer J Clin. 1997;47:5–27. doi: 10.3322/canjclin.47.1.5. [DOI] [PubMed] [Google Scholar]
- 2.Carey TE. Head And Neck Tumor Cell Lines. In: Hay R J, Park J-G, Gazdar A, editors. Atlas of Human Tumor Cell Lines. San Diego: Academic Press; 1994. pp. 79–120. [Google Scholar]
- 3.Hong WK, Bromer R. Chemotherapy in head and neck cancer. N Engl J Med. 1983;308:75–79. doi: 10.1056/NEJM198301133080204. [DOI] [PubMed] [Google Scholar]
- 4.Clark JR, Dreyfuss AI. The role of cisplatin in treatment regimens for squamous cell carcinoma of the head and neck. Semin Oncol. 1991;18:34–48. [PubMed] [Google Scholar]
- 5.The Department of Veterans Affairs Laryngeal Cancer Study Group, author. Induction chemotherapy plus radiation compared with surgery plus radiation in patients with advanced laryngeal cancer. N Engl J Med. 1991;325:1656–1657. doi: 10.1056/NEJM199106133242402. [DOI] [PubMed] [Google Scholar]
- 6.Hong WK, Shapshay SM, Bhutani R, Craft ML, Ucmakli A, Yamaguchi KT, Vaughan CW, Strong MS. Induction chemotherapy in advanced squamous head and neck carcinoma with high-dose cisplatinum and bleomycin infusion. Cancer. 1979;44:19–25. doi: 10.1002/1097-0142(197907)44:1<19::aid-cncr2820440104>3.0.co;2-r. [DOI] [PubMed] [Google Scholar]
- 7.Kish JA, Weaver A, Jacobs J, Cummings G, Al-Sarraf M. Cisplatin and 5-fluorouracil infusion in patients with recurrent and disseminated epidermoid cancer of the head and neck. Cancer. 1984;53:1819–1824. doi: 10.1002/1097-0142(19840501)53:9<1819::aid-cncr2820530903>3.0.co;2-r. [DOI] [PubMed] [Google Scholar]
- 8.Jacobs C, Lyman G, Velez-Garcia E, Sridhar KS, Knight W, Hochster H, Goodnough LT, Mortimer JE, Einhorn LH, Schacter L, Cherng N, Dalton T, Burroughs J, Rozeneweig M. A phase III randomized study comparing cisplatin and fluorouracil as single agents and in combination for advanced squamous cell carcinoma of the head and neck. J Clin Oncol. 1992;10:257–263. doi: 10.1200/JCO.1992.10.2.257. [DOI] [PubMed] [Google Scholar]
- 9.Tupchong L, Scott CB, Blitzer PH, Marcial VA, Lowry LD, Jacobs JR, Stetz J, Davis LW, Snow JB, Chandler R, Kramer S, Pajak TF. Randomized study of preoperative versus postoperative radiation therapy in advanced head and neck carcinoma: Long-term follow-up of RTOG Study 73-03. Int J Radiat Oncol Biol Phys. 1991;20:21–28. doi: 10.1016/0360-3016(91)90133-o. [DOI] [PubMed] [Google Scholar]
- 10.Kotwall C, Sako K, Razack MS, Rao U, Bakamjian V, Shedd DP. Metastatic patterns in squamous cell cancer of the head and neck. Am J Surg. 1987;154:439–442. doi: 10.1016/0002-9610(89)90020-2. [DOI] [PubMed] [Google Scholar]
- 11.Haraf DJ, Weichselbaum RR, Vokes EE. Re-irradiation with concomitant chemotherapy of unresectable recurrent head and neck cancer: a potentially curable disease. Ann Oncol. 1996;7:913–918. doi: 10.1093/oxfordjournals.annonc.a010793. [DOI] [PubMed] [Google Scholar]
- 12.Mineta T, Rabkin SD, Yazaki T, Hunter WD, Martuza RL. Attenuated multi-mutated herpes simplex virus-1 for the treatment of malignant gliomas. Nat Med. 1995;1:938–943. doi: 10.1038/nm0995-938. [DOI] [PubMed] [Google Scholar]
- 13.Yazaki T, Manz HJ, Rabkin SD, Martuza RL. Treatment of human malignant meningiomas by G207, a replication-competent multimutated herpes simplex virus 1. Cancer Res. 1995;55:4752–4756. [PubMed] [Google Scholar]
- 14.Toda M, Rabkin SD, Martuza RL. Treatment of human breast cancer in a brain metastatic model by G207, a replication-competent multimutated herpes simplex virus 1. Hum Gene Ther. 1998;9:2177–2185. doi: 10.1089/hum.1998.9.15-2177. [DOI] [PubMed] [Google Scholar]
- 15.Liverpool Head and Neck Oncology Group, author. A phase III randomised trial of cisplatinum, methotrextate, cisplatinum + methotrexate and cisplatinum +5-FU in end stage squamous carcinoma of the head and neck. Br J Cancer. 1990;61:311–315. doi: 10.1038/bjc.1990.59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Forastiere AA, Urba SG. Experimental therapeutic approaches for recurrent head and neck cancer. Cancer Treat Res. 1995;74:263–281. doi: 10.1007/978-1-4615-2023-8_14. [DOI] [PubMed] [Google Scholar]
- 17.Williams SD, Velez-Garcia E, Essessee I, Ratkin G, Birch R, Einhorn LH. Chemotherapy for head and neck cancer. Comparison of cisplatin + vinblastine + bleomycin versus methotrexate. Cancer. 1986;57:18–23. doi: 10.1002/1097-0142(19860101)57:1<18::aid-cncr2820570106>3.0.co;2-v. [DOI] [PubMed] [Google Scholar]
- 18.Boviatsis EJ, Park JS, Sena-Esteves M, Kramm CM, Chase M, Efird JT, Wei MX, Breakfield XO, Chiocca EA. Long-term survival of rats harboring brain neoplasms treated with ganciclovir and a herpes simplex virus vector that retains an intact thymidine kinase gene. Cancer Res. 1994;54:5745–5751. [PubMed] [Google Scholar]
- 19.Miyatake S, Martuza RL, Rabkin SD. Defective herpes simplex virus vectors expressing thymidine kinase for the treatment of malignant glioma. Cancer Gene Ther. 1997;4:222–228. [PubMed] [Google Scholar]
- 20.Chase M, Chung RY, Chiocca EA. An oncolytic viral mutant that delivers the CYP2B1 transgene and augments cyclophosphamide chemotherapy. Nat Biotechnol. 1998;16:444–448. doi: 10.1038/nbt0598-444. [DOI] [PubMed] [Google Scholar]
- 21.Freytag SO, Rogulski KR, Paielli DL, Gilbert JD, Kim JH. A novel three-pronged approach to kill cancer cells selectively: concomitant viral, double suicide gene, and radiotherapy. Hum Gene Ther. 1998;9:1323–1333. doi: 10.1089/hum.1998.9.9-1323. [DOI] [PubMed] [Google Scholar]
- 22.Heise C, Sampson-Johannes A, Williams A, McCormick F, Von Hoff DD, Kirn DH. ONYX-015, an E1B gene-attenuated adenovirus, causes tumor-specific cytolysis and antitumoral efficacy that can be augmented by standard chemotherapeutic agents. Nat Med. 1997;3:639–645. doi: 10.1038/nm0697-639. [DOI] [PubMed] [Google Scholar]
- 23.Toda M, Martuza RL, Kojima H, Rabkin SD. in situ cancer vaccination: an IL-12 defective vector/replication-competent herpes simplex virus vector combination induces local and systemic antitumor activity. J Immunol. 1998;160:4457–4464. [PubMed] [Google Scholar]
- 24.Andreansky S, He B, van Cott J, McGhee J, Markert JM, Gillespie GY, Roizman B, Whitley RJ. Treatment of intracranial gliomas in immunocompetent mice using herpes simplex viruses that express murine interleukins. Gene Ther. 1998;5:121–130. doi: 10.1038/sj.gt.3300550. [DOI] [PubMed] [Google Scholar]
- 25.Advani SJ, Sibley GS, Song PY, Hallahan DE, Kataoka Y, Roizman B, Weichselbaum RR. Enhancement of replication of genetically engineered herpes simplex viruses by ionizing radiation: a new paradigm for destruction of therapeutically intractable tumors. Gene Ther. 1998;5:160–165. doi: 10.1038/sj.gt.3300546. [DOI] [PubMed] [Google Scholar]
- 26.Nielsen LL, Lipari P, Dell J, Gurnani M, Hajian G. Adenovirus-mediated p53 gene therapy and paclitaxel have synergistic efficacy in models of human head and neck, ovarian, prostate, and breast cancer. Clin Cancer Res. 1998;4:835–846. [PubMed] [Google Scholar]
- 27.Kanamori Y, Kigawa J, Minagawa Y, Irie T, Oishi T, Shimada M, Takahashi M, Nakamura T, Sato K, Terakawa N. A newly developed adenovirus-mediated transfer of a wild-type p53 gene increases sensitivity to cis-diamminedichloroplatinum (II) in p53-deleted ovarian cancer cells. Eur J Cancer [A] 1998;34:1802–1806. doi: 10.1016/s0959-8049(98)00199-3. [DOI] [PubMed] [Google Scholar]
- 28.Teicher BA, Holden SA, Kelly MJ, Shea TC, Cucchi CA, Rosowsky A, Henner WD, Frei E. Characterization of a human squamous carcinoma cell line resistant to cis-diamminedichloroplatinum (II) Cancer Res. 1987;47:388–393. [PubMed] [Google Scholar]
- 29.Kurihara N, Kubota T, Hoshiya Y, Otani Y, Watanabe M, Kumai K, Kitajima M. Antitumor activity of cis-diamminedichloroplatinum (II) against human tumor xenografts depends on its area under the curve in nude mice. J Surg Oncol. 1996;61:138–142. doi: 10.1002/(SICI)1096-9098(199602)61:2<138::AID-JSO9>3.0.CO;2-D. [DOI] [PubMed] [Google Scholar]
- 30.Newkirk K, Heffern J, Sloman-Moll E, Sessions RB, Rasmussen AA, Andrews PA, Cullen KJ. Glutathione content but not gamma glutamyl cysteine synthase mRNA expression predicts cisplatin resistance in head and neck cancer cell lines. Cancer Chemother Pharmacol. 1997;40:75–80. doi: 10.1007/s002800050629. [DOI] [PubMed] [Google Scholar]
- 31.Vokes EE, Weichselbaum RR, Lippman SM, Hong WK. Head and Neck Cancer. N Engl J Med. 1993;328:184–194. doi: 10.1056/NEJM199301213280306. [DOI] [PubMed] [Google Scholar]
- 32.Rao DN, Shroff PD, Chattopadhyay G, Dinshaw KA. Survival analysis of 5595 head and neck cancers-results of conventional treatment in a high-risk population. Br J Cancer. 1998;77:1514–1518. doi: 10.1038/bjc.1998.249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bi W, Kim YG, Feliciano ES, Pavelic L, Wilson KM, Pavelic ZP, Stambrook PJ. An HSVtk-mediated local and distant antitumor bystander effect in tumors of head and neck origin in athymic mice. Cancer Gene Ther. 1997;4:246–252. [PubMed] [Google Scholar]
- 34.Wilson KM, Stambrook PJ, Bi WL, Pavelic ZP, Pavelic L, Gluckman JL. HSV-tk gene therapy in head and neck squamous cell carcinoma. Enhancement by the local and distant bystander effect. Arch Otolaryngol Head Neck Surg. 1996;122:746–749. doi: 10.1001/archotol.1996.01890190042011. [DOI] [PubMed] [Google Scholar]
- 35.Schuller DE, McGuirt WF, Krause CJ, McCabe BF, Pflug BK. Increased survival with surgery alone vs. combined therapy. Laryngoscope. 1979;89:582–594. doi: 10.1288/00005537-197904000-00007. [DOI] [PubMed] [Google Scholar]
- 36.Jesse RH, Lindberg RD. The efficacy of combining radiation therapy with a surgical procedure in patients with cervical metastasis from squamous cancer of the oropharynx and hypopharynx. Cancer. 1975;35:1163–1166. doi: 10.1002/1097-0142(197504)35:4<1163::aid-cncr2820350422>3.0.co;2-c. [DOI] [PubMed] [Google Scholar]
- 37.Pinto AL, Lippard SJ. Binding of the antitumor drug cis-diamminedichloroplatinum(II) (cisplatin) to DNA. Biochim Biophys Acta. 1985;780:167–180. doi: 10.1016/0304-419x(85)90001-0. [DOI] [PubMed] [Google Scholar]
- 38.Pinto AL, Lippard SJ. Sequence-dependent termination of in vitro DNA synthesis by cis- and trans-diamminedichloroplatinum (II) Proc Natl Acad Sci USA. 1985;82:4616–4619. doi: 10.1073/pnas.82.14.4616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Heiger-Bernays WJ, Essigmann JM, Lippard SJ. Effect of the antitumor drug cis-diamminedichloroplatinum(II) and related platinum complexes on eukaryotic DNA replication. Biochemistry. 1990;29:8461–8466. doi: 10.1021/bi00488a037. [DOI] [PubMed] [Google Scholar]
- 40.Mamenta EL, Poma EE, Kaufmann WK, Delmastro DA, Grady HL, Chaney SG. Enhanced replicative bypass of platinum-DNA adducts in cisplatin-resistant human ovarian carcinoma cell lines. Cancer Res. 1994;54:3500–3505. [PubMed] [Google Scholar]
- 41.Snyder MB, Saravolatz LD, Markowitz N, Pohlod D, Taylor RC, Ward SG. The in-vitro and in-vivo efficacy of cisplatin and analogues in the treatment of herpes simplex virus-II infections. J Antimicrob Chemother. 1987;19:815–822. doi: 10.1093/jac/19.6.815. [DOI] [PubMed] [Google Scholar]
- 42.Le Gac NT, Villani G, Boehmer PE. Herpes simplex virus type-1 single-strand DNA-binding protein (ICP8) enhances the ability of the viral DNA helicase-primase to unwind cisplatin-modified DNA. J Biol Chem. 1998;273:13801–13807. doi: 10.1074/jbc.273.22.13801. [DOI] [PubMed] [Google Scholar]