Abstract
Lutropin (LH) directs ovulation and implantation by regulating the production of estrogen and progesterone. We have shown that the circulatory half-life of LH is controlled by the Man/GalNAc-4-SO4 receptor, which binds GalNAc-4-SO4 on LH oligosaccharides. The short half-life in conjunction with episodic release of LH from the pituitary accounts for the pulsatile rise and fall in circulating LH. Complete genetic ablation of the Man/GalNAc-4-SO4 receptor results in death in utero. Heterozygous female mice clear LH from the circulation more slowly and have smaller litters due to a reduction in the rate of implantation. This reduction is fully correctable by exogenous progesterone and estrogen, indicating that the rate of LH clearance is critical for the production of sufficient progesterone and estrogen for implantation. Thus, the Man/GalNAc-4-SO4 receptor regulates the endocrinological status of the female and is essential for an early event in embryonic development.
Introduction
Ovulation, fertilization, and implantation are complex processes that depend on the exquisitely coordinated interaction of neurons in the hypothalamus, gonadotroph cells in the anterior pituitary, and target cells in the ovary. These three participants form the gonad/pituitary/hypothalamic axis. Abnormalities in the regulation of this axis can result in infertility and miscarriage (1–3). Peptide-releasing factors produced in the hypothalamus stimulate gonadotroph cells to release the glycoprotein hormones follitropin (FSH) and lutropin (LH) into the circulation. FSH directs follicular development and oocyte maturation, whereas LH is responsible for the rupture of antral follicles to release the ovum and for the conversion of postovulatory follicles into corpora lutea (4). The precisely controlled production of estrogen and progesterone, initially by developing follicles and subsequently by corpora lutea, is essential for differentiation of the uterus into a receptive state for the fertilized egg and for implantation (5–9).
LH characteristically demonstrates an episodic rise and fall in the circulation that is critical for the biologic activity of LH in vivo. Gonadotropin-releasing hormone–stimulated release of LH from dense core granules in pituitary gonadotrophs generates these pulses at specific intervals. The amplitude and frequency of LH pulses produced in the circulation are in turn regulated by progesterone and estrogen levels (10–14). The pulsatile rise and fall in circulating LH levels results in the episodic stimulation of the LH receptor, a G protein–coupled receptor (15). Episodic stimulation may be critical because the activated form of the LH receptor is recognized by β-arrestin, resulting in uncoupling from adenylyl cyclase and internalization of the occupied receptor (16). Replenishment at the cell surface with unoccupied LH receptor requires periods between pulses when the cells are not exposed to the hormone.
The episodic rise and fall in LH levels also requires that LH have a short half-life in the circulation. The circulatory half-life of LH, but not FSH, is precisely controlled by the Man/GalNAc-4-SO4 receptor that we have identified in hepatic endothelial cells (17, 18). We initially demonstrated that LH bears unique oligosaccharide structures that terminate with β1,4-linked GalNAc-4-SO4 whereas FSH bears structures that terminate with sialic acid linked to galactose (19–21). We have shown that it is the cysteine-rich domain located at the amino-terminus of the Man/GalNAc-4-SO4 receptor, expressed at high levels in hepatic endothelial cells, that specifically binds terminal β1,4-linked GalNAc-4-SO4 located on the N-linked oligosaccharides of LH (22–24). This binding mediates the rapid clearance of LH from the circulation. The Man/GalNAc-4-SO4 receptor expressed by hepatic endothelial cells is present in the form of a dimer that must simultaneously bind to two terminal GalNAc-4-SO4 moieties on LH to achieve the apparent Kd of 1.6 × 10–7 M we have observed with isolated cells (25). The bound hormone is internalized and rapidly transported to lysosomes where it is degraded. Each endothelial cell in rat liver expresses 600,000 receptors at the cell surface (18), and it has been estimated that the half-time for internalization of the surface pool of receptor-ligand complexes is 10.1 seconds (26). We have calculated that there is sufficient receptor to mediate the clearance of LH from the blood with the t1/2 of 7.5 minutes observed for clearance in the rat (17). Furthermore, only changes in receptor level and/or the rate of internalization from the cell surface are predicted to have an effect on the rate of LH clearance from the blood.
We have generated mice expressing reduced levels of the Man/GalNAc-4-SO4 receptor in order to examine the role of LH clearance rate on follicle development, ovulation, and implantation. While animals homozygous (–/–) for genetic ablation of the Man/GalNAc-4-SO4 receptor do not survive in utero, mice heterozygous (+/–) for ablation of the receptor are normal. Female mice, but not male mice, that are heterozygous for deletion of the receptor display sufficient alterations in the regulation of their endocrinologic state to markedly reduce the efficiency of implantation, but not ovulation.
Methods
Generation of Man/GalNAc-4-SO4 receptor–deficient mice by gene targeting.
A 129/SvJ BAC library (Genome Systems Inc., St. Louis, Missouri, USA) was screened with a probe derived from the cysteine-rich domain of the murine Man/GalNAc-4-SO4 receptor cDNA. An 8-kb PstI fragment containing exon 2 encoding the cysteine-rich domain of the receptor was isolated. The targeting vector was constructed by subcloning the fragment into pZero (Invitrogen Corp., Carlsbad, California, USA). Exon 2 of the Man/GalNAc-4-SO4 receptor was deleted by inverse PCR with primers containing NotI sites at their 5′ ends: 5′TTCCATCTGCGGCCGCTTGTGATCTTC-ATTATTAGATTAAAAATTG3′ and 5′GATCACAAGCGGCCGCAGATGGAAGGTCTATGGAACCACGGATGAC3′. A cassette containing the neomycin phosphotransferase cDNA driven by the phosphoglycerate kinase promoter (PGK-neo) flanked by loxP sites was inserted into the NotI site created during deletion of exon 2 (Figure 1a). Embryonic stem (ES) cells were electroporated with the targeting plasmid that had been linearized with SnaBI. Homologous recombination occurred in 22 of 140 ES cell clones surviving selection with G418 that were analyzed by Southern blotting. Two colonies were individually injected into C57BL/6J blastocysts. Chimeric males were mated with C57BL/6J females, and the first generation of agouti offspring were screened by Southern blot analysis of tail genomic DNA for germline transmission of the targeted allele. Genotypes of the mice from later generations were determined by multiplex PCR using the primers P1: 5′GGGACCACTCAAGGATATGTT3′; P2: 5′GCTCTAAGTGCCATCTCAGTT3′; P3: 5′TGCTCGACGTTGTCACTGAAG3′; and P4: 5′CTCGTCAAGAAGGCGATAGAA3′. The expected sizes for the wild-type and mutated alleles were 412 bp and 548 bp, respectively (Figure 1).
Figure 1.
Generation of Man/GalNAc-4-SO4 receptor+/– mice. (a) The Man/GalNAc-4-SO4 receptor targeting construct. The 5′ and 3′ homologies were derived from exon 2, which encodes the entire cysteine-rich domain of the receptor, and its flanking introns. Exon 2 was replaced with a PGK-neo cassette. (b) DNA from the live progeny of heterozygous matings was genotyped by PCR or Southern blotting. The wild-type locus was detected using primers P1 and P2, which yielded a 421-bp product, and the mutant locus using primers P3 and P4, which yielded a 548-bp product. No Man/GalNAc-4-SO4 receptor–/– animals lived to birth. Examples of PCR reactions from M/G4S-R+/+ and M/G4S-R+/– mice are shown. Std, oligonucleotide standard.
Analysis of ovulation and implantation.
The number of ovulated oocytes released during the estrous cycle was determined by sacrificing female Man/GalNAc-4-SO4+/+ (M/G4S-R+/+) and M/G4S-R+/– mice the morning following estrus. The enlarged, translucent ampulla of each oviduct was excised and trimmed, and the ovulated oocyte-cumulus complexes were collected into a Petri dish by washing with Dulbecco’s PBS. The oocytes were then manually counted. Normal implantation was examined by housing M/G4S-R+/+ and M/G4S-R+/– female mice in estrus with M/G4S-R+/+ male mice overnight. The morning of finding a vaginal plug was designated day 1 of pregnancy. Implantation sites were visualized on day 5 by intravenous injection of 0.1 ml of Chicago sky blue 6B dye (Sigma Chemical Co., St. Louis, Missouri, USA) (8). Exogenous hormone was administered to M/G4S-R+/– females as follows: day 3 of pregnancy at 0900 hours, progesterone (P4) (2 mg per mouse); day 4 of pregnancy at 0900 hours, P4 (2 mg per mouse) + 17β-estradiol (E2) (10 ng per mouse). Implantation sites were counted on day 5. P4 and E2 were dissolved in peanut oil and injected subcutaneously (0.1 ml/ per mouse). Mice without implantation sites were not included in the analyses.
Quantitation of Man/GalNAc-4-SO4 receptor steady state mRNA levels.
Total RNA was isolated using TRIzol reagent (Life Technologies Inc., Rockville, Maryland, USA). Three micrograms of total RNA was used to synthesize the first-strand cDNA with Superscript II RNaseH-Reverse Transcriptase (Life Technologies Inc.). Quantitative PCR was performed on a LightCycler (Roche Molecular Biochemicals, Indianapolis, Indiana, USA) in a total volume of 20 μl of 10× reaction buffer (Taq polymerase, deoxynucleoside triphosphates, (MgCl2, SYBR Green I; Roche Molecular Biochemicals) and 1 μl of first-strand cDNA. Ten pmol of each primer (5′TGGGGACCTGGCAAGTAT3′ and 5′TGTCACTGGGGTT-CCATC3′) was added. Real-time PCR was performed in glass capillaries with an initial denaturation step of 2 minutes at 95°C, followed by 50 cycles of 0 seconds at 95°C, 10 seconds at 60°C, and 20 seconds at 72°C. At the end of each cycle the fluorescence emitted by SYBR Green was measured at 85°C. The PCR products from tissue with the most abundant message were detected after 35 cycles. A dilution series of Man/GalNAc-4-SO4 receptor cDNA of known concentration was included in each run to generate a standard curve. Duplicate samples for each analysis were quantified according to the LightCycler analysis software.
Detection of Man/GalNAc-4-SO4 receptor protein expression.
Livers from M/G4S-R+/+ and M/G4S-R+/– mice were homogenized with a Polytron homogenizer (Brinkmann Instruments, Westbury, New York, USA) in 5 volumes of buffer containing 25 mM HEPES, 50 mM KCl, 2 mM Mg acetate, 1 mM DTT, and 10% sucrose. Homogenates were layered over a 65% sucrose cushion and sedimented at 100,000 g for 75 minutes. The membrane fraction was collected and resuspended in 20 mM Tris, pH 7.8, 150 mM NaCl using a Dounce homogenizer. Protein concentrations were determined using the Bradford method (Bio-Rad Laboratories, Hercules, California, USA). Binding assays were performed with SO4-4-GalNAcβ1,4GlcNAcβ1,4Man-[125I]BSA (S4GGnM-BSA) and [125I]asialo-orosomucoid (ASOR) in the presence of 1% Triton X-100 as described previously (22, 27).
Clearance studies were performed as described previously (17) in mice anesthetized with ketamine (87 mg/kg) and xylazine (13 mg/kg) by cannulating both jugular veins. Mice were heparinized and 2 × 106 disintegrations per minute of [125I]bLH (1 μCi/10 ng LH) in 100 μl of saline was injected through the right jugular vein. Samples were withdrawn from the left jugular vein and weighed, and the amount of [125I]bLH present per 50 μl was determined by γ-counting.
Results
Man/GalNAc-4-SO4 receptor–deficient mice.
The role of LH clearance by the Man/GalNAc-4-SO4 receptor was examined by preparing mice deficient in receptor. The targeting construct was prepared by replacing exon 2 with a PGK-neo cassette (Figure 1). Exon 2 was chosen because it encodes the entire cysteine-rich domain of the Man/GalNAc-4-SO4 receptor and contains the longest contiguous coding sequence, 413 bp (28). The targeting vector was introduced into 129/SvJ ES cells by electroporation and G418-resistant ES clones selected. Southern blot analysis identified 22 recombinants out of 140 screened. Chimeric male mice were bred to C57BL/6J females to generate heterozygous male and female mice on a mixed background.
Wild-type (M/G4S-R+/+) and heterozygous (M/G4S-R+/–) male mice were bred with M/G4S-R+/+ and M/G4S-R+/– female mice. The M/G4S-R+/– × M/G4S-R+/+ crosses both yielded the expected 1:1 ratio of M/G4S-R+/+ and M/G4S-R+/– progeny (Table 1). In contrast, M/G4S-R+/– × M/G4S-R+/– breedings yielded M/G4S-R+/+ : M/G4S-R+/– progeny in a ratio of 1:2.27 but did not yield any live animals homozygous (–/–) for the deletion (Table 1). Analyses of the embryos in utero indicated that loss of M/G4S-R–/– embryos occurred prior to day 6.5 (not shown). The results were not affected by the sex of the M/G4S-R+/– mice used in the breedings.
Table 1.
No live M/G4S-R–/– mice are obtained from M/G4S-R+/– × M/G4S-R–/– matings
Female mice heterozygous for deletion of the Man/GalNAc-4-SO4 receptor have reduced litter sizes due to reduced implantation.
When females heterozygous for deletion of the Man/GalNAc-4-SO4 receptor were bred with either M/G4S-R+/+ or M/G4S-R+/– males, there was a marked reduction of the median number of pups per litter to five from the eight to nine obtained when M/G4S-R+/+ or M/G4S-R+/– males were bred to M/G4S-R+/+ females (Figure 2). Furthermore, only 15–20% of the litters born to M/G4S-R+/– females fell within the normal range of litter size for M/G4S-R+/+ females (Figure 2). The majority of litters delivered by M/G4S-R+/– females contained only 1–5 pups. We also noted that the number of pups per litter delivered by individual M/G4S-R+/– females tended to remain constant through multiple breedings. Since the reduction in litter size is only seen when female mice are heterozygous for Man/GalNAc-4-SO4 receptor deletion, this reduction reflects an alteration in the reproductive endocrinologic status of the female rather than a defect in the development of the embryo. While a greater reduction in litter size would be predicted for the M/G4S-R+/– × M/G4S-R+/– cross than for the M/G4S-R+/+ × M/G4S-R+/– cross due to the lethality of the homozygous deletion, the difference between the respective mean values of 4.7 and 5.2 for litter sizes was not significant.
Figure 2.
Female mice heterozygous for deletion of the Man/GalNAc-4-SO4 receptor have smaller numbers of pups per litter. M/G4S-R+/+ and M/G4S-R+/– female mice were bred with both M/G4S-R+/+ and M/G4S-R+/– male mice. The number of pups born was determined the morning of delivery. There was no apparent difference in appearance or survival rate following delivery. Bars indicate the median value for each group. Mean values of 4.7 (n = 63) for female M/G4S-R+/– × male M/G4S-R+/–, 5.2 (n = 25) for female M/G4S-R+/– × male M/G4S-R+/+, 9.0 (n = 11) for female M/G4S-R+/+ × male M/G4S-R+/–, and 9.8 (n = 20) for female M/G4S-R+/+ × male M/G4S-R+/+ pups per litter were obtained. ANOVA analysis using Tukey’s multiple comparison test indicated that differences between the means of female M/G4S-R+/– × male M/G4S-R+/– and female M/G4S-R+/– × male M/G4S-R+/+, or between female M/G4S-R+/+ × male M/G4S-R+/– and female M/G4S-R+/+ × male M/G4S-R+/+, were not significant (P > 0.05). Differences between the means for either female M/G4S-R+/– × male M/G4S-R+/– or female M/G4S-R+/– × male M/G4S-R+/+, and either female M/G4S-R+/+ × male M/G4S-R+/– or female M/G4S-R+/+ × male M/G4S-R+/+, were highly significant (P < 0.001).
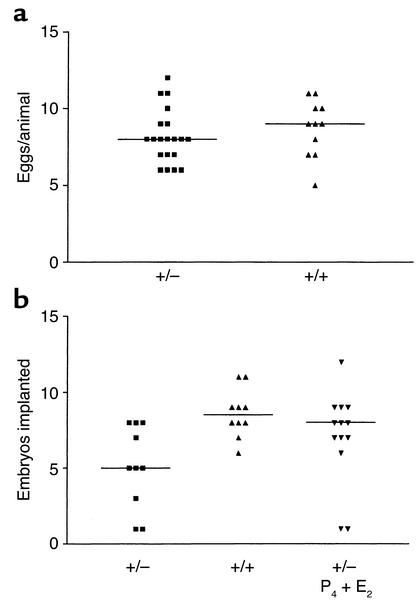
M/G4S-R+/– mice are predicted to express a reduced number of Man/GalNAc-4-SO4 receptors in their hepatic endothelial cells and clear LH from their circulation more slowly than M/G4S-R+/+ mice. Since LH regulates the production of estrogen and progesterone in the ovary, altered rates of clearance could have an impact on ovulation, implantation, or maintenance of the fetus and result in the changes shown in Figure 2. The ovulation rate for M/G4S-R+/+ and M/G4S-R+/– females was examined by counting the number of eggs released (Figure 3a). The difference in the mean values for the number of eggs released by M/G4S-R+/+ and M/G4S-R+/– animals, 8.7 and 8.2, respectively, is not significant. This suggests that the levels of estrogen and progesterone produced in response to the preovulatory surge of LH are sufficient for the normal number of eggs to be released from the ovary. In contrast, there is a significant difference in the number of embryos that are able to implant (Figure 3b) in M/G4S-R+/+ as compared with M/G4S-R+/– female mice, with means of 8.7 and 5.1 embryos implanted, respectively.
Figure 3.
Female mice heterozygous for deletion of the Man/GalNAc-4-SO4 receptor ovulate normally but have a reduced rate of implantation. (a) Eggs were collected from genotyped mice the morning following estrus and counted. An unpaired t test indicated that the mean values of 8.2 for M/G4S-R+/– female mice and 8.7 for M/G4S-R+/+ female mice were not significant (P = 0.40). (b) Genotyped mice were given subcutaneous injections of P4 (2 mg per mouse) on day 3, and P4 (2 mg per mouse) + E2 (10 ng per mouse) on day 4. Mice were injected with Chicago sky blue 6B in saline on day 5 shortly before sacrifice, and the number of implantation sites demarcated as localized blue bands (8) was counted. Bars indicate the median. ANOVA analysis using Tukey’s multiple comparison test indicated that the difference between the mean value of 5.1 for M/G4S-R+/– female mice and 8.6 for M/G4S-R+/+ female mice is significant (P < 0.05) whereas the difference between the means of 7.1 for M/G4S-R+/– female mice treated with P4 and E2 and 8.6 for M/G4S-R+/+ female mice was not significant (P > 0.05).
Implantation occurs around midnight of day 4 following copulation. In addition to increasing levels of progesterone, a transient increase in estrogen levels is seen during the morning prior to implantation. Implantation does not occur in the absence of this transient rise in estrogen levels. Exogenous progesterone will maintain the blastula in a condition termed delayed implantation until exogenous estrogen is provided (5–9). Heterozygous females were, therefore, given progesterone (P4) on day 3, and P4 + 17β-estradiol (E2) on day 4, by injection. The number of embryos implanted was determined on day 5 (Figure 3b). Providing exogenous progesterone and estrogen increased the mean for the number of embryos implanted in the M/G4S-R+/– female mice to 7.1, a value not significantly different from that of the M/G4S-R+/+ female mice. A small number of animals treated with E2 alone on day 4 also showed an increased rate of implantation consistent with the known requirement for estrogen (data not shown). Since LH regulates the production of progesterone and estrogen at this critical time for implantation, it appears that, in females heterozygous for ablation of the Man/GalNAc-4-SO4receptor, there is a sufficient alteration in the clearance rate for LH to have an impact on postovulatory hormone production and implantation but not on ovulation itself.
mRNA and protein levels are reduced in mice heterozygous for Man/GalNAc-4-SO4 receptor gene deletion.
The Man/GalNAc-4-SO4receptor is expressed in macrophages as well as in hepatic endothelial cells (29). In addition we have detected the Man/GalNAc-4-SO4receptor at the tubulovesicular junction of sperm and sertoli cells (Y. Mi and J.U. Baenziger, unpublished observation). The phenotype of the M/G4S-R+/– mice indicates that levels of the Man/GalNAc-4-SO4receptor are sufficiently reduced in these animals to affect the physiologic regulation of their hormones. Message levels were therefore determined for lung (macrophages), testis (sertoli cells, sperm, and macrophages), spleen (macrophages), and liver (endothelial cells and Kupffer cells) as shown in Figure 4. The primer pair utilized is specific for exon 7 of the receptor. Levels of Man/GalNAc-4-SO4receptor message containing exon 7 were reduced by at least half in the tissues examined with the exception of the spleen. Thus, message for the mutant form of the Man/GalNAc-4-SO4receptor does not appear to be expressed, even though only exon 2 was deleted, and there is no evidence of significant compensation in Man/GalNAc-4-SO4receptor message levels for the wild-type allele in M/G4S-R+/– mice.
Figure 4.
Man/GalNAc-4-SO4 receptor message is reduced in M/G4S-R+/– mice as compared with M/G4S-R+/+ mice. Total RNA was prepared from the tissues indicated. Three micrograms of total RNA were used to synthesize first-strand cDNA for each tissue. The amount of Man/GalNAc-4-SO4 receptor message for identical inputs of total RNA was quantitated by PCR on a LightCycler by comparison to Man/GalNAc-4-SO4 receptor cDNA of known concentration using the primers specific for exon 7. The values shown represent the mean of two determinations in each case. The duplicates for each determination differed by less than 5%.
The amounts of GalNAc-4-SO4–specific binding activity in the membranes prepared from livers of M/G4S-R+/+ and M/G4S-R+/– mice were also compared (Figure 5). The amount of S4GGnM-[125I]BSA bound per microgram of membrane protein was significantly reduced in preparations from M/G4S-R+/– mice. The GalNAc-4-SO4–specific binding activity in membranes from M/G4S-R+/– mice was 67% of that in membranes from M/G4S-R+/+ mice. This reduction in binding was not observed for another carbohydrate-specific receptor, the asialoglycoprotein receptor, examined with ASOR as the ligand (Figure 5). Thus, based on both message levels and binding activity in the liver, the amount of Man/GalNAc-4-SO4receptor expressed by hepatic endothelial cells of M/G4S-R+/– mice is significantly reduced when compared with that of M/G4S-R+/+ mice.
Figure 5.
GalNAc-4-SO4–specific binding is reduced in membranes from M/G4S-R+/– mice as compared with M/G4S-R+/+ mice. Membranes were prepared from livers of three M/G4S-R+/+ and three M/G4S-R+/– mice and tested for binding of S4GGnM-BSA by the Man/GalNAc-4-SO4 receptor and ASOR by the asialoglycoprotein receptor in triplicate at three different concentrations of membranes. The results are presented as cpm bound per microgram of membrane protein. The difference between the mean of 31.3 cpm bound per microgram for M/G4S-R+/+ mice and 20.8 cpm bound per microgram for M/G4S-R+/– mice for S4GGnM-BSA was highly significant (P = 0.0009), whereas the difference between the mean of 279 cpm bound per microgram for M/G4S-R+/+ mice and 298 cpm bound per microgram for M/G4S-R+/– mice for ASOR was not significant (P = 0.58).
The clearance of LH from the blood is slower in mice heterozygous for deletion of the Man/GalNAc-4-SO4 receptor gene.
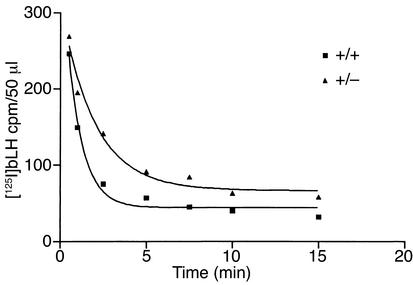
Alterations in the number of Man/GalNAc-4-SO4receptors expressed at the surface of hepatic endothelial cells are predicted to have an effect on the clearance rate for LH in vivo. The reduction in GalNAc-4-SO4–specific binding by M/G4S-R+/– liver membranes indicated that the rate of LH clearance from the circulation would likely be slower in M/G4S-R+/– than in M/G4S-R+/+ mice. Differences in the rates of clearance were observed upon injection of [125I]bLH into M/G4S-R+/+ and M/G4S-R+/– mice, as shown in Figure 6 and summarized from multiple studies in Table 2. Nonlinear regression analyses performed using Prism 2.0 indicated that the best fit was obtained with parameters for single-component exponential decay. The half-life of LH in the circulation was 0.47 minutes for M/G4S-R+/+ and 2.04 minutes for M/G4S-R+/– mice (Table 2). Thus, LH clearance is fourfold slower in M/G4S-R+/– mice than in M/G4S-R+/+ mice. The increased time spent in the circulation results in the levels of LH remaining elevated for longer periods of time, with an apparent impact on activation and deactivation of the LH receptor in the ovary that is sufficient to alter the levels of E2 produced at the time of implantation.
Figure 6.
LH is cleared from the circulation more slowly by M/G4S-R+/– mice than by M/G4S-R+/+ mice. [125I]bLH was injected into M/G4S-R+/– and M/G4S-R+/+ mice, and 50 μl of blood was withdrawn from a cannula in the jugular vein at the times indicated. The amount of radioactivity in each sample was determined on a γ-counter. The curves shown were generated with Prism 2.0 (GraphPad Software Inc., San Diego, California, USA) using the equation for exponential decay with a single component.
Table 2.
M/G4S-R+/+ mice remove LH from the circulation more rapidly than M/G4S-R+/– mice
Discussion
The N-linked oligosaccharides on the four members of the glycoprotein hormone family contribute to the biologic function of these hormones at multiple levels. The carbohydrates are important for folding and assembly, for secretion, and for providing sufficient mass to prevent rapid filtration by the kidney (30–32). However, most striking is the critical role played by the unique sulfated carbohydrate structures found on LH and thyrotropin (TSH). LH and TSH bear unique N-linked oligosaccharides that terminate with β1,4-linked GalNAc-4-SO4(20, 21, 31, 33–35). In contrast, the N-linked oligosaccharides FSH and chorionic gonadotropin terminate with the sequence sialic acid α2,3/6-galactose (20, 21, 36). The presence of this unique carbohydrate modification on LH and TSH reflects the action of a protein-specific β1,4-N-acetylgalactosaminyltransferase (37–40) and a recently cloned GalNAc-4-sulfotransferase (41, 42). The sulfated carbohydrate structures synthesized by these transferases are present on these glycoprotein hormones produced in the pituitaries of all vertebrate species (43). Thus, the unique features of the N-linked carbohydrate structures like those of the protein have been conserved throughout vertebrate evolution, indicating that they are of vital importance to the action of LH in vivo. Our observation that LH bearing oligosaccharides terminating with β1,4-linked GalNAc-4-SO4 is rapidly cleared from the circulation by a receptor we identified as the Man/GalNAc-4-SO4receptor led us to propose that the rate of clearance is critical for attaining optimal activation of ovarian LH receptors in vivo (17, 30). The pulsatile rise and fall in LH levels reflects stimulated release of LH from gonadotrophs by gonadotropin-releasing hormone and the short half-life due to Man/GalNAc-4-SO4receptor binding and internalization. Notably, both the amplitude and the frequency of LH pulses can be modulated by P4 and E2 in vivo (11). The pulse frequency is considered particularly critical for the initiation of ovulation (44, 45). Furthermore, in humans the episodic rise and fall in circulating LH levels first appears during puberty and continues throughout the reproductive years (11, 46).
The affinity of the Man/GalNAc-4-SO4receptor for LH, 1.7 × 10–7 M (18), and the number of receptors in hepatic endothelial cells, 600,000 LH-binding sites per cell (18), have been determined for the rat. The combination of this affinity and abundance of the Man/GalNAc-4-SO4receptor make it ideal for maintaining a specific rate of clearance at any LH concentration that falls within the physiologic range. LH levels in the circulation of the rat, 0.1 × 10–9 to 1.0 × 10–9 M, fall far below the Kd for binding to the Man/GalNAc-4-SO4receptor. However, the abundance of the receptor at the endothelial cell surface and the large number of endothelial cells present in the liver together provide sufficient capacity to bind on the order of 10% of the total pool of circulating LH if 0.5% of the receptor sites are occupied. The Man/GalNAc-4-SO4receptor has one of the fastest rates of internalization measured to date for an endocytic receptor, with a reported half-time of 10.1 seconds for internalization of the entire pool of occupied receptor (26). As a result, effectively all of the bound LH, representing 10% of the total circulating pool of LH, will be internalized and degraded every 30 seconds, yielding a predicted half-life of 2.5 minutes for the rat. This prediction is in close agreement with the half-lives we have measured in the mouse, 0.48 minutes, and the rat, 7.5 minutes (17). An important feature of this mechanism for LH clearance is that the rate of clearance will remain constant at LH concentrations below the Kd for binding to the receptor and will only be altered by a change in either the number of Man/GalNAc-4-SO4receptors expressed at the cell surface or their rate of internalization. Thus, the reduced number of receptors in M/G4S-R+/– mice should, as observed, result in an increase in the circulatory half-life of LH. Both the height and the width of the LH pulse but not the level of LH between pulses should be altered by a change in half-life. We have not, however, been able to document changes in peak width and height for LH pulses, because technical considerations including the amount of serum and the number of samples required are prohibitive in mice.
Our present studies demonstrate that reducing the levels of Man/GalNAc-4-SO4receptor on hepatic endothelial cells by roughly half increases the circulatory half-life of injected LH by fourfold. These studies underscore the relationship of the Man/GalNAc-4-SO4receptor and the terminal β1,4-linked GalNAc-4-SO4 moieties on LH to the regulation of P4 and E2 levels. While the decrease in the clearance rate for LH in the M/G4S-R+/– mice does not have an impact on the ovulation rate, the slower rate of clearance does affect implantation. Implantation in the mouse occurs on day 4 and requires a brief increase in E2 levels on day 3 (5, 7, 9). In the absence of this brief increase in E2 levels, the blastocysts remain viable but dormant in a condition termed delayed implantation. Correction of the implantation delay in the M/G4S-R+/– mice by providing exogenous P4 and E2 indicates that the decreased rate of LH clearance in these heterozygotes is sufficient to perturb the endocrinologic state of the female so that inadequate levels of E2 are produced.
The regulation of LH levels and their relationship to levels of P4 and E2 production in the context of the gonad/pituitary/hypothalamic axis are complex. Elevated levels of circulating LH are associated with infertility and miscarriage in humans. For example, women with polycystic ovary syndrome exhibit continuous hypersecretion of LH. The reduced rate of implantation seen in M/G4S-R+/– mice suggests that alterations in the rate of internalization of the Man/GalNAc-4-SO4receptor, the affinity of the cysteine-rich domain for terminal GalNAc-4-SO4, the ability of the receptor to form dimeric species, or the pattern of LH glycosylation could all result in changes in the regulation of P4 and/or E2 production in response to LH. The levels of Man/GalNAc-4-SO4receptors in liver increase in response to E2 in mice (ref. 47; and Y. Mi and J.U. Baenziger, unpublished observation). This is consistent with the observation that in postmenopausal women the half-life for LH increases from 57 minutes to 139 minutes (48). The change in half-life may reflect changes in the level of Man/GalNAc-4-SO4receptor expression and in the levels of GalNAc-4-SO4 addition to LH, which are also sensitive to E2 levels (49). Such changes could contribute to the decline in fertility with increasing age.
The current studies provide a strong link between the carbohydrate structures on LH, the Man/GalNAc-4-SO4receptor, and the regulation of E2 and P4 production in vivo. Furthermore, the phenotype of the M/G4S-R+/– female demonstrates that the in vivo regulation of circulatory half-life for proteins may be of critical importance for biologic activity. The role of the Man/GalNAc-4-SO4receptor during development of the embryo represents a different function that remains to be elucidated. Our studies emphasize that the Man/GalNAc-4-SO4receptor is multifunctional, having at least two critical functions during the ovulatory cycle and development that are distinct from its potential role in innate immunity as a mannose-specific receptor in macrophages (50, 51).
Acknowledgments
We thank Carla Weinheimer for performing the clearance studies in mice. This work was supported by NIH grant R37-CA21923.
Footnotes
See the related Commentary beginning on page 169.
References
- 1.Balen AH, Tan S-L, Jacobs HS. Hypersecretion of luteinizing hormone: a significant cause of infertility and miscarriage. Br J Obstet Gynaecol. 1993;100:1082–1089. doi: 10.1111/j.1471-0528.1993.tb15170.x. [DOI] [PubMed] [Google Scholar]
- 2.Franks S. Polycystic ovary syndrome. N Engl J Med. 1995;333:853–861. doi: 10.1056/NEJM199509283331307. [DOI] [PubMed] [Google Scholar]
- 3.Shoham Z, Jacobs HS, Insler V. Luteinizing hormone: its role, mechanism of action, and detrimental effects when hypersecreted during the follicular phase. Fertil Steril. 1993;59:1153–1161. doi: 10.1016/s0015-0282(16)55968-8. [DOI] [PubMed] [Google Scholar]
- 4.Tsafriri, A., and Chun, S.-Y. 1996. Ovulation. In Reproductive endocrinology, surgery, and technology. E.Y. Adashi, J.A. Rock, and Z. Rosenwaks, editors. Lippincott-Raven Publishers. Philadelphia, Pennsylvania, USA. 235–249.
- 5.Finn CA, Martin L. The control of implantation. J Reprod Fertil. 1974;39:195–206. doi: 10.1530/jrf.0.0390195. [DOI] [PubMed] [Google Scholar]
- 6.Huet-Hudson YM, Andrews GK, Dey SK. Cell type-specific localization of c-Myc protein in the mouse uterus: modulation by steroid hormones and analysis of the periimplantation period. Endocrinology. 1989;125:1683–1690. doi: 10.1210/endo-125-3-1683. [DOI] [PubMed] [Google Scholar]
- 7.McCormack JT, Greenwald GS. Evidence for a preimplantation rise in oestradiol-17β levels on day 4 of pregnancy in the mouse. J Reprod Fertil. 1974;41:297–301. doi: 10.1530/jrf.0.0410297. [DOI] [PubMed] [Google Scholar]
- 8.Paria BC, Huet-Hudson YM, Dey SK. Blastocyst’s state of activity determines the “window” of implantation in the mouse receptive uterus. Proc Natl Acad Sci USA. 1993;90:10159–10162. doi: 10.1073/pnas.90.21.10159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yoshinaga K, Adams CE. Delayed implantation in the spayed, progesterone treated adult mouse. J Reprod Fertil. 1966;12:593–595. doi: 10.1530/jrf.0.0120593. [DOI] [PubMed] [Google Scholar]
- 10.1987. The episodic secretion of hormones. W.F. Crowley, Jr., and J.G. Hofler, editors. John Wiley & Sons. New York, New York, USA.
- 11.Marshall JC, et al. Gonadotropin-releasing hormone pulses: regulators of gonadotropin synthesis and ovulatory cycles. Recent Prog Horm Res. 1991;47:155–187. doi: 10.1016/b978-0-12-571147-0.50009-3. [DOI] [PubMed] [Google Scholar]
- 12.Marshall JC, Griffin ML. The role of changing pulse frequency in the regulation of ovulation. Hum Reprod. 1993;8(Suppl. 2):57–61. doi: 10.1093/humrep/8.suppl_2.57. [DOI] [PubMed] [Google Scholar]
- 13.Veldhuis JD, Beitins IZ, Johnson ML, Serabian MO, Dufau ML. Biologically active luteinizing hormone is secreted in episodic pulsations that vary in relation to stage of the menstrual cycle. J Clin Endocrinol Metab. 1984;58:1050–1058. doi: 10.1210/jcem-58-6-1050. [DOI] [PubMed] [Google Scholar]
- 14.Veldhuis JD, Carlson ML, Johnson ML. The pituitary gland secretes in bursts: appraising the nature of glandular secretory impulses by simultaneous multiple-parameter deconvolution of plasma hormone concentrations. Proc Natl Acad Sci USA. 1987;84:7686–7690. doi: 10.1073/pnas.84.21.7686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Segaloff DL, Ascoli M. The lutropin/choriogonadotropin receptor ... 4 years later. Endocr Rev. 1993;14:324–347. doi: 10.1210/edrv-14-3-324. [DOI] [PubMed] [Google Scholar]
- 16.Mukherjee S, et al. A direct role for arrestins in desensitization of the luteinizing hormone/choriogonadotropin receptor in porcine ovarian follicular membranes. Proc Natl Acad Sci USA. 1999;96:493–498. doi: 10.1073/pnas.96.2.493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Baenziger JU, Kumar S, Brodbeck RM, Smith PL, Beranek MC. Circulatory half-life but not interaction with the lutropin/chorionic gonadotropin receptor is modulated by sulfation of bovine lutropin oligosaccharides. Proc Natl Acad Sci USA. 1992;89:334–338. doi: 10.1073/pnas.89.1.334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Fiete D, Srivastava V, Hindsgaul O, Baenziger JU. A hepatic reticuloendothelial cell receptor specific for SO4-4GalNAcb1,4GlcNAcb1,2Mana that mediates rapid clearance of lutropin. Cell. 1991;67:1103–1110. doi: 10.1016/0092-8674(91)90287-9. [DOI] [PubMed] [Google Scholar]
- 19.Baenziger JU, Green ED. Pituitary glycoprotein hormone oligosaccharides: structure, synthesis and function of the asparagine-linked oligosaccharides on lutropin, follitropin and thyrotropin. Biochim Biophys Acta. 1988;947:287–306. doi: 10.1016/0304-4157(88)90012-3. [DOI] [PubMed] [Google Scholar]
- 20.Green ED, Baenziger JU. Asparagine-linked oligosaccharides on lutropin, follitropin, and thyrotropin. I. Structural elucidation of the sulfated and sialylated oligosaccharides on bovine, ovine, and human pituitary glycoprotein hormones. J Biol Chem. 1988;263:25–35. [PubMed] [Google Scholar]
- 21.Green ED, Baenziger JU. Asparagine-linked oligosaccharides on lutropin, follitropin, and thyrotropin. II. Distributions of sulfated and sialylated oligosaccharides on bovine, ovine, and human glycoprotein hormones. J Biol Chem. 1988;263:36–44. [PubMed] [Google Scholar]
- 22.Fiete D, Baenziger JU. Isolation of the SO4-4-GalNAcb1,4GlcNAcb1,2Mana-specific receptor from rat liver. J Biol Chem. 1997;272:14629–14637. doi: 10.1074/jbc.272.23.14629. [DOI] [PubMed] [Google Scholar]
- 23.Fiete D, Beranek MC, Baenziger JU. The macrophage/endothelial cell mannose receptor cDNA encodes a protein that binds oligosaccharides terminating with SO4-4-GalNAcb1,4GlcNAcb or Man at independent sites. Proc Natl Acad Sci USA. 1997;94:11256–11261. doi: 10.1073/pnas.94.21.11256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fiete DJ, Beranek MC, Baenziger JU. A cysteine-rich domain of the “mannose” receptor mediates GalNAc-4-SO4binding. Proc Natl Acad Sci USA. 1998;95:2089–2093. doi: 10.1073/pnas.95.5.2089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Roseman DS, Baenziger JU. Molecular basis of lutropin recognition by the mannose/GalNAc-4-SO4 receptor. Proc Natl Acad Sci USA. 2000;97:9949–9954. doi: 10.1073/pnas.170184597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Magnusson S, Berg T. Extremely rapid endocytosis mediated by the mannose receptor of sinusoidal endothelial rat liver cells. Biochem J. 1989;257:651–656. doi: 10.1042/bj2570651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Baenziger JU, Maynard Y. Human hepatic lectin: physiochemical properties and specificity. J Biol Chem. 1980;255:4607–4613. [PubMed] [Google Scholar]
- 28.Harris N, et al. The exon-intron structure and chromosomal localization of the mouse macrophage mannose receptor gene Mrcl: identification of a Ricin-like domain at the N-terminus of the receptor. Biochem Biophys Res Commun. 1994;198:682–692. doi: 10.1006/bbrc.1994.1099. [DOI] [PubMed] [Google Scholar]
- 29.Takahashi K, Donovan MJ, Rogers RA, Ezekowitz RA. Distribution of murine mannose receptor expression from early embryogenesis through to adulthood. Cell Tissue Res. 1998;292:311–323. doi: 10.1007/s004410051062. [DOI] [PubMed] [Google Scholar]
- 30.Baenziger JU. Glycosylation: to what end for the glycoprotein hormones? Endocrinology. 1996;137:1520–1522. doi: 10.1210/endo.137.5.8612480. [DOI] [PubMed] [Google Scholar]
- 31.Baenziger, J.U., and Green, E.D. 1991. Structure, synthesis, and function of the asparagine-linked oligosaccharides on pituitary glycoprotein hormones. In Biology of carbohydrates. Volume 3. V. Ginsberg and P.W. Robbins, editors. JAI Press Ltd. London, United Kingdom. 1–46.
- 32.Feng W, et al. The asparagine-linked oligosaccharides of the human chorionic gonadotropin beta subunit facilitate correct disulfide bond pairing. J Biol Chem. 1995;270:11851–11859. doi: 10.1074/jbc.270.20.11851. [DOI] [PubMed] [Google Scholar]
- 33.Green ED, Van Halbeek H, Boime I, Baenziger JU. Structural elucidation of the disulfated oligosaccharide from bovine lutropin. J Biol Chem. 1985;260:15623–15630. [PubMed] [Google Scholar]
- 34.Stockell Hartree A, Renwick AGC. Molecular structures of glycoprotein hormones and functions of their carbohydrate components. Biochem J. 1992;287:665–679. doi: 10.1042/bj2870665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Thotakura NR, Blithe DL. Glycoprotein hormones: glycobiology of gonadotropins, thyrotropin, and free α subunit. Glycobiology. 1995;5:3–10. doi: 10.1093/glycob/5.1.3. [DOI] [PubMed] [Google Scholar]
- 36.Endo Y, Yamashita K, Tachibana Y, Tojo S, Kobata A. Structures of the asparagine-linked sugar chains of human chorionic gonadotropin. J Biochem (Tokyo) 1979;85:669–679. [PubMed] [Google Scholar]
- 37.Mengeling BJ, Manzella SM, Baenziger JU. A cluster of basic amino acids within an α-helix is essential for α-subunit recognition by the glycoprotein hormone N-acetylgalactosaminyltransferase. Proc Natl Acad Sci USA. 1995;92:502–506. doi: 10.1073/pnas.92.2.502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Smith PL, Baenziger JU. A pituitary N-acetylgalactosamine transferase that specifically recognizes glycoprotein hormones. Science. 1988;242:930–933. doi: 10.1126/science.2460923. [DOI] [PubMed] [Google Scholar]
- 39.Smith PL, Baenziger JU. Recognition by the glycoprotein hormone-specific N-acetylgalactosaminetransferase is independent of hormone native conformation. Proc Natl Acad Sci USA. 1990;87:7275–7279. doi: 10.1073/pnas.87.18.7275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Smith PL, Baenziger JU. Molecular basis of recognition by the glycoprotein hormone-specific N-acetylgalactosamine-transferase. Proc Natl Acad Sci USA. 1992;89:329–333. doi: 10.1073/pnas.89.1.329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Okuda T, et al. Molecular cloning and characterization of GalNAc-4-sulfotransferase expressed in human pituitary gland. J Biol Chem. 2000;275:40605–40613. doi: 10.1074/jbc.M007983200. [DOI] [PubMed] [Google Scholar]
- 42.Xia G, Evers MR, Kang H-G, Schachner M, Baenziger JU. Molecular cloning and expression of the pituitary glycoprotein hormone N-acetylgalactosamine-4-O-sulfotransferase. J Biol Chem. 2000;275:38402–38409. doi: 10.1074/jbc.M007821200. [DOI] [PubMed] [Google Scholar]
- 43.Manzella SM, Dharmesh SM, Beranek MC, Swanson P, Baenziger JU. Evolutionary conservation of the sulfated oligosaccharides on vertebrate glycoprotein hormones that control circulatory half-life. J Biol Chem. 1995;270:21665–21671. doi: 10.1074/jbc.270.37.21665. [DOI] [PubMed] [Google Scholar]
- 44.Marshall JC, Kelch RP. Gonadotropin-releasing hormone: role of pulsatile secretion in the regulation of reproduction. N Engl J Med. 1986;315:1459–1468. doi: 10.1056/NEJM198612043152306. [DOI] [PubMed] [Google Scholar]
- 45.Ream N, Saunder SE, Kelch RP, Marshall JC. Pulsatile gonadotropin secretion during the menstrual cycle: evidence for altered frequency of gonadotropin-releasing hormone secretion. J Clin Endocrinol Metab. 1984;59:328–337. doi: 10.1210/jcem-59-2-328. [DOI] [PubMed] [Google Scholar]
- 46.Knobil E. The neuroendocrine control of the menstrual cycle. Recent Prog Horm Res. 1980;36:53–78. doi: 10.1016/b978-0-12-571136-4.50008-5. [DOI] [PubMed] [Google Scholar]
- 47.Lansink M, et al. Increased clearance explains lower plasma levels of tissue-type plasminogen activator by estradiol: evidence for potently enhanced mannose receptor expression in mice. Blood. 1999;94:1330–1336. [PubMed] [Google Scholar]
- 48.Sharpless JL, Supko JG, Martin KA, Hall JE. Disappearance of endogenous luteinizing hormone is prolonged in postmenopausal women. J Clin Endocrinol Metab. 1999;84:688–694. doi: 10.1210/jcem.84.2.5433. [DOI] [PubMed] [Google Scholar]
- 49.Dharmesh SM, Baenziger JU. Estrogen modulates expression of the glycosyltransferases that synthesize sulfated oligosaccharides on lutropin. Proc Natl Acad Sci USA. 1993;90:11127–11131. doi: 10.1073/pnas.90.23.11127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Hoffmann JA, Kafatos FC, Janeway CA, Ezekowitz RA. Phylogenetic perspectives in innate immunity. Science. 1999;284:1313–1318. doi: 10.1126/science.284.5418.1313. [DOI] [PubMed] [Google Scholar]
- 51.Stahl PD, Ezekowitz RA. The mannose receptor is a pattern recognition receptor involved in host defense. Curr Opin Immunol. 1998;10:50–55. doi: 10.1016/s0952-7915(98)80031-9. [DOI] [PubMed] [Google Scholar]