Abstract
Hypoxic necrosis of dentate gyrus neurons in primary culture required the activation of an orderly cell death program independent of protein synthesis. Early mitochondrial swelling and loss of the mitochondrial membrane potential were accompanied by release of cytochrome c and followed by caspase-9-dependent activation of caspase-3. Caspase-3 and -9 inhibitors reduced neuronal necrosis. Calcium directly induced cytochrome c release from isolated mitochondria. Hypoxic neuronal necrosis may be an active process in which the direct effect of hypoxia on mitochondria may lead to the final common pathway of caspase-3-mediated neuronal death.
Apoptosis and necrosis are two distinct modes of cell death initially defined by morphological criteria (1). In its most classical form, such as death by deprivation of nerve growth factor in sympathetic neurons, apoptosis activates a genetic program in which cell surface receptors trigger the expression of immediate early genes and death-signaling proteins, which activate a cascade of proteolytic enzymes named caspases (2, 3). Necrosis, by contrast, is usually seen as the passive explosion of a cell overwhelmed by ionic fluxes. Although molecular mechanisms of apoptosis in the nervous system have been extensively studied (4), mechanisms of neuronal necrosis have received little attention, although necrosis is the predominant form of neuronal death induced by hypoxia-ischemia, and seizures in the adult brain (5–9). We studied the mechanism of necrosis induced by hypoxia or hypoxia-ischemia in primary cultures of dentate gyrus (DG) neurons and found that it involves the rapid but orderly activation of caspases from their constitutionally expressed precursors. This activation may result from the release of cytochrome c by calcium-loaded mitochondria and does not require gene expression or protein synthesis.
Materials and Methods
DG Cultures.
Cultures were prepared as described by Lowenstein and Arsenault (10) with minor modifications. DG were dissected from postnatal day 3 (P3) Wistar rat pups, incubated for 15 min in Neurobasal/B-27 containing trypsin 0.25% (wt/vol) at 37°C, and triturated with glass pipettes in Neurobasal/FBS (10% vol/vol) to disperse the tissue. The supernatant (containing dissociated cells) was centrifuged (130 × g) , and dissociated cells were redispersed in Neurobasal/FBS. Cells were plated at 400 cells per mm2 onto 96-, 12-, or 8-well plates and incubated at 37°C in a 5% CO2/95% O2 incubator. Culture medium was removed and replaced by Neurobasal/B-27 at day in vitro (DIV) 1 and DIV 5. Hypoxia was induced at DIV 7 by 45 min exposure to 10 mM NaCN. Some cultures were treated with 300 nM staurosporine. The research protocol was approved by the Institutional Animal Care and Use Committee at the performance site.
Ultrastructural Studies.
Cultures were fixed with 2.5% glutaraldehyde and 1.6% paraformaldehyde in 0.1 M cacodylate buffer for 1 h. After washing, the cultures were postfixed with 1% osmium tetroxide in cacodylate buffer for 45 min, rinsed with water, and en bloc stained with 1% uranyl acetate for 1 h. The cultures were then dehydrated with graded ethanols and embedded in Spurr Epon. The thin sections were stained with uranyl acetate and lead citrate and viewed with a Philips (Eindhoven, The Netherlands) 201C transmission electron microscope.
Light Microscopic Immunocytochemistry.
Cultures, plated on eight-well chamber slides (Lab-Tek), were fixed with paraformaldehyde 4% (wt/vol) for 30 min, washed three times with phosphate buffer (PB) 0.1 M, and incubated with PB 0.1 M containing gelatin 0.2% (wt/vol; PB-G) for 1 h, then incubated for 90 min with a rabbit polyclonal Ab to the p20 active fragment of caspase-3 (caspase-3a) (R & D Systems; AF835; see ref. 11 for Ab specificity) diluted in PB-G (0.3 μg/ml), or with rabbit polyclonal Ab to fractin (product of actin cleavage by active caspase-3; PharMingen; 551527; see ref. 12 for Ab specificity) diluted in PB-G (1:1000), or with a mouse mAb to cytochrome c (PharMingen, 556432) diluted in PB-G (1:100). Cultures were then washed three times with PB-G and incubated for 90 min with anti-rabbit polyclonal or anti-mouse mAbs conjugated to peroxidase (Amersham Pharmacia, NA934 and NA931) diluted in PB-G (1:100). After washing, staining was revealed with diaminobenzidine for 10 min. Control experiments, including incubation without primary Ab or with a nonimmune rabbit or mouse serum, did not show any staining. Cultures were counterstained with the nuclear dye Hoechst 33342. We counted the percentage of caspase-3a or cytochrome c immunoreactive cells in eight fields in each group. Some cultures were preincubated with 200 nM Mitotracker orange (Molecular Probes; M-7510) 30 min before paraformaldehyde fixation.
Caspase-3a and Cytochrome c Double Staining.
After fixation and a 1-h saturation with PB-G, cultures were incubated for 10 min with PB-G containing Triton X-100 0.1%, then with anti-caspase-3a and cytochrome c Abs as described previously. Double staining was revealed with anti-mouse mAbs conjugated to FITC (1:100, Jackson ImmunoResearch) and anti-rabbit polyclonal Abs conjugated to Cy3 (1:100, Jackson ImmunoResearch). After counterstaining with Hoechst 33342, the triple label was viewed on a scanning wide-field microscope with image deconvolution (Scanalytics, Billerica, MA).
Electron Microscopic Immunocytochemistry.
Preembedding immunogold labeling was used to localize caspase-3a and cytochrome c immunoreactivity. Cultures were fixed and incubated with primary Abs as described previously for light microscopic immunocytochemistry. After washing, cultures were incubated with PBS containing 0.1% cold fish gelatin (CFG) and 1% normal goat serum (NGS) for 15 min and incubated for 90 min with anti-rabbit polyclonal or anti-mouse mAbs conjugated to 1 nm of gold particles and diluted in PBS containing 0.1% CFG. The cultures were washed with PBS containing 0.1% CFG and 2.5 M NaCl for 20 min, and washed for 10 min with CFG and NGS, then several times with distilled water. Gold staining was intensified by silver enhancement. Cultures were postfixed with 0.1% osmium, stained with 1% uranyl acetate, dehydrated with ethanol, embedded with Spurr Epon, and thin-sectioned. Sections were stained with uranyl acetate and lead citrate and viewed on a Philips 201C TEM.
Caspase Inhibitors.
Cultures were preincubated with caspase inhibitors (Enzyme Systems Products, Livermore, CA) 45 min before NaCN exposure and this procedure was continued during and after hypoxic treatment. Control and untreated hypoxic wells were incubated with vehicle (0.2% DMSO) 24 h after hypoxia, and neuronal survival was determined by trypan blue assay in 4 wells in each group. Neurons were easily recognized by their birefringent cytoplasmic membranes.
Western Blots.
Western blots were performed according to Niquet et al. (13) and incubated with Abs recognizing both the precursor and the active form of caspase-3 (Santa Cruz Biotechnology, H-277) or exclusively the active form (Chemicon, AB3623).
Isolated Mitochondria.
Isolated mitochondria were prepared according to Brustovetsky and Dubinsky (14) with minor modifications. During brain homogenization, EGTA was substituted by EDTA. The purity of the preparation was confirmed by electron microscopy (EM). Western blots were performed as described by Gogvadze et al. (15).
Results
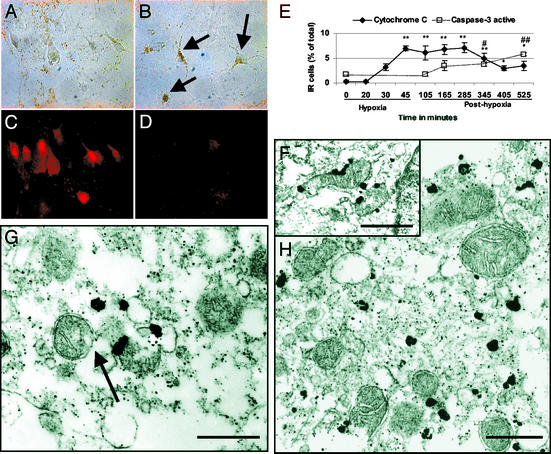
In DG cultures exposed to 10 mM NaCN for 45 min, ≈90% of neurons (identified by immunoreactivity against NeuN, a neuronal marker) died 24 h after hypoxia, because they stained with propidium iodide. Survival of GFAP-immunoreactive glia was not affected (data not shown). Combining the same cyanide treatment with glucose deprivation (“ischemia”) had very similar effects on the neurons. By EM, many hypoxic neurons showed early swelling of mitochondria and endoplasmic reticulum, in the presence of an intact nucleus (Fig. 1B), and evolved later toward necrosis with severe cytoplasmic swelling and membrane rupture (Fig. 1C). This finding was in sharp contrast to staurosporine-treated apoptotic neurons, which displayed nuclear breakdown into large, round masses of chromatin (Fig. 1D). Hypoxic necrosis evolved rapidly: we observed a progressive loss of the mitochondrial membrane potential Δψ within a few minutes of cyanide application (Fig. 2 A–D). This Δψ decay was always synchronous with release of cytochrome c. Even in the absence of the Δψ-sensitive dye Mitotracker orange, which may accelerate mitochondrial depolarization (16), cytochrome c release was already visible 30 min after NaCN application, and was maximal after 45 min of hypoxia (Fig. 2E). At that time, exclusion of trypan blue or propidium iodide was completely preserved, excluding the possibility that cytochrome c release is a consequence of cell death. Cytochrome c release was not cyanide-specific but was shared with other inhibitors of the mitochondrial electron transport chain, because it was observed after 45 min exposure to 2 μM rotenone (23.4 ± 6% of cells were cytochrome c-immunoreactive), 20 mM azide (39.5 ± 5.1%), and 20 μM antimycin (13 ± 2.7%). In controls, the localization of cytochrome c between the inner and outer mitochondrial membranes could be seen clearly (Fig. 2F), and by the time that cytochrome c release was visualized immunocytochemically, some breaks of the outer mitochondrial membrane could be identified (Fig. 2 G and H).
Figure 1.
Hypoxia induces early swelling of mitochondria and necrotic neuronal death. Ultrastructure of control (A), hypoxic (1 and 24 h after hypoxia; B and C), and staurosporine-treated (24 h after staurosporine treatment; D) neurons was studied by EM. One hour after hypoxia, most cytosolic and nuclear morphology is intact, and mitochondrial swelling (arrows in B) is the first sign of cell damage. It evolves toward necrosis with severe cytoplasmic swelling, including mitochondrial swelling, membrane rupture, and tigroid-type chromatin fragmentation (24 h after hypoxia; C). (D) By contrast, the staurosporine-treated neuron displays a characteristic apoptotic morphology with large masses of chromatin. (Bars = 1 μM.)
Figure 2.
(A–D) Control neurons (no Triton X-100, peroxidase technique) are not cytochrome c-immunoreactive (A) and have a high mitochondrial membrane potential indicated by Mitotracker orange dye (C). Hypoxic neurons 3 min after NaCN application are cytochrome c-immunoreactive (arrows in B) and show a severe loss of mitochondrial membrane potential (D). (E) Cytochrome c release preceded caspase-3 activation. Sister cultures were stained with anti-cytochrome c and anti-caspase-3a Abs. Data are mean ± SEM of four independent determinations in each group, analyzed by one-factor ANOVA with post hoc t tests using the pooled SD and α = 0.05. *, <0.05 or **, < 0.001 vs. 0 min for cytochrome c. #, <0.05 or ##, < 0.001 vs. 0 min for caspase-3a. (F–H) Electron photomicrographs of mitochondria stained with anti-cytochrome c Abs in control (F) and after 45 min of NaCN exposure (G and H). In control cultures, mitochondria are undamaged and cigar-shaped. Cytochrome c staining, revealed by gold particles, is located in the intermembrane space. In hypoxic cultures, mitochondria are swollen and outer membranes are sometimes ruptured (arrow). Cytochrome c immunoreactivity is located in the cytosol. (Bars = 0.5 μM.)
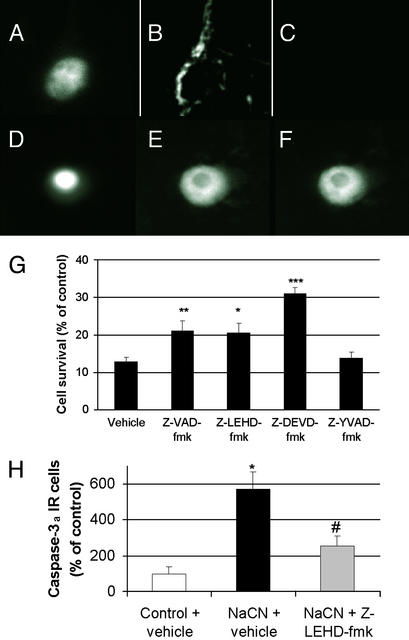
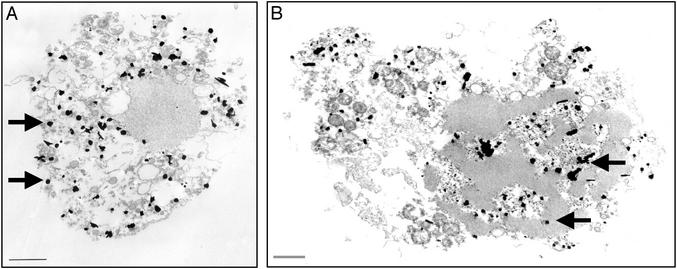
Caspase-3 activation was present 5 h after hypoxic exposure, when 75% of the activated form of caspase-3 (caspase-3a) immunoreactivity colocalized with cytochrome c (Fig. 3 A–F), and was maximal at 24 h, when cytochrome c was almost entirely degraded. Western blots incubated with the H-277 Abs confirmed the presence of the precursor of caspase-3 (32 kD; p32) in control cultures and the appearance of caspase-3a (20 kD; p20) in hypoxia-treated cultures. Western blots incubated with the AB3623 Abs showed a faint caspase-3a immunoreactivity in control cultures and a dramatic increase in hypoxia-treated cultures (data not shown). Fractin immunoreactivity, revealing actin cleavage by caspase-3a, increased in hypoxia-treated cultures (16.8 ± 1.7% IR cells in hypoxic cultures vs. 7.7 ± 1.3% IR cells in control cultures; P < 0.05 by Student's t test), confirming that caspase-3a was functional. The general caspase inhibitor ZVAD-fmk, the caspase-9 inhibitor LEHD-fmk, and the caspase-3 inhibitor DEVD-fmk all improved neuronal survival (Fig. 3G). All caspase-3a-immunoreactive cells were double-stained with propidium iodide after hypoxia, suggesting that caspase-3 activation kills the neurons (data not shown). This result is consistent with the protective effect of the relatively specific caspase-3 inhibitor DEVD-fmk (Fig. 3G). Activation of caspase-9 was located upstream of caspase-3, because the caspase-9 inhibitor LEHD-fmk inhibited hypoxia-induced caspase-3 activation (Fig. 3H). By EM, caspase-3a immunoreactive cells had a necrotic morphology. Most caspase-3a staining was limited to the cytoplasm 5 h after hypoxia, explaining the predominant location of proteolytic damage near the mitochondrial site of release, and spread to the nucleus by 24 h (Fig. 4).
Figure 3.
(A–F) In the control neuron (A–C, Triton X-100-treated), the nucleus (stained with Hoechst 33342, A) is large, cytochrome c staining is punctate, reflecting its mitochondrial localization (B), and no caspase-3a immunoreactivity is visible (C). Five hours after hypoxia, a hypoxic neuron has a shrunken nucleus (D) and diffuse cytochrome c staining throughout the cytoplasm (E), and is immunoreactive for caspase-3a (F). (G) Inhibitors of caspase-3 (DEVD-fmk), caspase-9 (LEHD-fmk), and the general caspase inhibitor ZVAD-fmk reduce hypoxic neuronal death, whereas caspase-1 inhibitor (YVAD-fmk) has no effect. Cell survival was determined 24 h after NaCN by using trypan blue. All these inhibitors had no effect on control cultures. Data are mean ± SEM of seven independent determinations in each group, analyzed by one-factor ANOVA with post hoc t tests using the pooled SD and α = 0.05 (*, <0.05; **, < 0.01; ***, <0.001). (H) A caspase-9 inhibitor (LEHD-fmk) substantially decreases the number of caspase-3a-positive cells in hypoxic cultures, suggesting the existence of a cascade leading to caspase-3 activation. LEHD-fmk (100 μM) was added 45 min before NaCN application and again after hypoxic treatment. Control and hypoxic wells were treated with vehicle (0.2% DMSO). Cultures were fixed with 4% paraformaldehyde 24 h after hypoxic treatment. Data are mean ± SEM of four independent determinations in each group, analyzed by one-factor ANOVA with post hoc t tests by using the pooled SD and α = 0.05. *, <0.001 vs. control; #, <0.001 vs. NaCN.
Figure 4.
Five hours after NaCN treatment, a neuron with relatively intact nucleus, ruptured plasma membrane, and grossly swollen organelles is clearly necrotic but activated caspase-3 (caspase-3a) immunoreactivity (gold particles, arrows) is present throughout the cytoplasm (A). In another necrotic neuron at 24 h (B), caspase-3a has diffused into the nucleus as well. (Bars = 1 μM.)
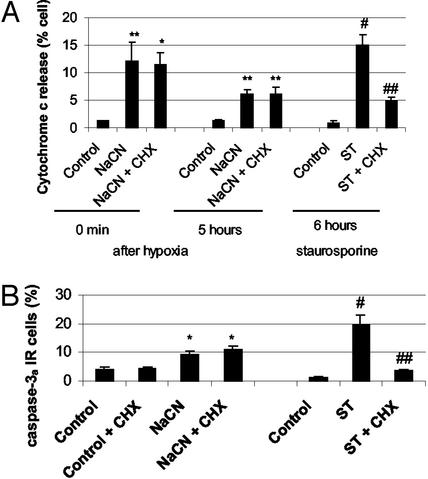
These results suggest that hypoxia-induced necrotic death depends on cytochrome c release and caspase activation. Because this hypoxia-induced “programmed necrosis” (17) and classical apoptosis share a common final pathway, we asked whether they were triggered by an identical mechanism upstream of cytochrome c release. Protein synthesis is usually required for the translation of immediate early genes and/or signaling proteins that trigger the apoptotic cascade, for example when apoptosis is induced by lower concentration of cyanide (18). Cycloheximide (CHX; 1 μg/ml) was effective in blocking cytochrome c release and caspase activation in stauroporine-induced apoptosis of DG neurons but was totally ineffective in hypoxic necrosis of the same cells (Fig. 5). This finding is not surprising, because during energy-poor states such as hypoxia or ischemia, the large amounts of energy needed for synthesis of new proteins are not available. It is notable that the caspase-3 proenzyme is already expressed in control cultures and does not need to be synthesized. The proteolytic cleavage of caspases from a precursor, which activates them, would be expected to require little energy, and it is not surprising that it can take place in an energy-poor state.
Figure 5.
(A) The protein synthesis inhibitor CHX does not alter hypoxia-induced cytochrome c release but inhibits staurosporine-induced cytochrome c release. The graph represents the percentage of cells immunoreactive for cytochrome c. Cultures were preincubated 1 h before NaCN or staurosporine treatment with CHX (1 μg/ml). CHX was added again after the hypoxic episode. Hypoxic cultures were fixed after 45 min of NaCN exposure (0 min after hypoxia) or 5 h after hypoxia to study cytochrome c release. Staurosporine cultures were fixed 6 h after treatment. Data are mean ± SEM of three to six independent determinations in each group, analyzed by one-factor ANOVA with post hoc t tests using the pooled SD and α = 0.05. *, <0.05 and **, <0.001 vs. control. #, <0.0001 vs. control and ##, <0.0001 vs. staurosporine. (B) The protein synthesis inhibitor CHX reduces caspase-3 activation in staurosporine-induced apoptosis but has no effect on hypoxic neuronal necrosis. The graph represents the percentage of immunoreactive cells for caspase-3a. Cultures were preincubated with CHX as in A and fixed 6 or 24 h after staurosporine or NaCN treatment, respectively. Data are mean ± SEM of 9–10 independent determinations in each group, analyzed by one-factor ANOVA with post hoc t tests using the pooled SD and α = 0.05. *, <0.001 vs. control; #, <0.0001 vs. control and ##, <0.0001 vs. ST.
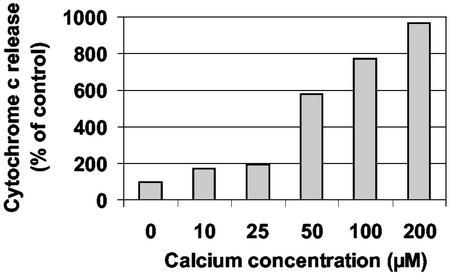
If genes that trigger cytochrome c release from mitochondria are not expressed, would mitochondrial dysfunction induced by hypoxia accomplish the same result? Calcium overloading of mitochondria is known to cause mitochondrial depolarization (19–21). A possible role for mitochondrial calcium accumulation from energy failure and/or excitotoxic injury was suggested by the observation that Δψ decay was synchronous with cytochrome c release (Fig. 2). Indeed, Western blots showed that very high Ca2+ levels, such as might be achieved by anoxic depolarization, released cytochrome c in a cell-free mitochondrial preparation (Fig. 6), as shown (22). Further work is needed to determine whether the opening of the mitochondrial transition pore (which would be expected from the Δψ decay), breaks of the mitochondrial membrane, or yet other mechanisms are responsible for cytochrome c release.
Figure 6.
Purified mitochondria were incubated for 30 min in a solution containing various calcium concentrations. Cytochrome c release was measured in Western blots of supernatants (15 μl per lane).
Discussion
These results suggest that, in this model, hypoxic neuronal necrosis, far from being a passive event with cell swelling and lysis from overwhelming ionic fluxes, is an orderly process that requires the activation of a cell death program. Our results provide EM evidence of early cytochrome c release into the cytoplasm with concomitant loss of mitochondrial Δψ, and biochemical and pharmacological evidence of caspase-9-dependent caspase-3 activation, with protection by caspase inhibitors. This “final common pathway” is the same cell death program involved in many forms of apoptotic death. However, programmed necrosis and classical apoptosis differ in their requirement for gene expression and protein synthesis. Initial events of apoptosis include immediate early gene up-regulation (23), causing increased expression of death-signaling proteins such as Bax (24), which is translocated from the cytoplasm to the mitochondria (25, 26), where it induces release of cytochrome c (27–29), which activates caspases (30). The energy-intensive process of expressing new genes is not required for programmed necrosis nor would it be expected in the presence of energy failure. The initial event of programmed necrosis seems to be hypoxia-induced mitochondrial dysfunction, which releases cytochrome c, activating constitutionally expressed procaspases. Because all mitochondrial toxins used in this study cause energy failure, which in turn induces calcium influx into mitochondria, this is one possible mechanism of mitochondrial dysfunction, which would explain why activation of DNA repair enzymes can paradoxically enhance cell death (31), presumably through energy depletion, and would imply that other types of mitochondrial malfunction may well trigger similar cascades. This hypothetical mechanism would also explain the relationship between energy failure and excitotoxicity, both of which might trigger calcium accumulation and cytochrome c release, and the reports of a necrotic morphology (7, 8) and an apoptotic-like biochemistry (32–35) in pathological situations characterized by depletion of energy reserves, such as cerebral ischemia.
Acknowledgments
Dr. Jeffrey A. Gornbein (University of California) provided expert assistance in the statistical analysis of data. This study was funded by National Institute of Neurological Disorders and Stroke Grant NS13515, the National Institutes of Health, and the Research Service of the Veterans Health Administration.
Abbreviations
- DG
dentate gyrus
- PB-G
phosphate buffer containing gelatin
- EM
electron microscopy
- caspase-3a
active fragment of caspase-3
- CHX
cycloheximide
References
- 1.Kerr J F, Wyllie A H, Currie A R. Br J Cancer. 1972;26:239–257. doi: 10.1038/bjc.1972.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Troy C M, Rabacchi S A, Hohl J B, Angelastro J M, Greene L A, Shelanski M L. J Neurosci. 2001;21:5007–5016. doi: 10.1523/JNEUROSCI.21-14-05007.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Whitfield J, Neame S J, Paquet L, Bernard O, Ham J. Neuron. 2001;29:629–643. doi: 10.1016/s0896-6273(01)00239-2. [DOI] [PubMed] [Google Scholar]
- 4.Lipton P. Physiol Rev. 1999;79:1431–1568. doi: 10.1152/physrev.1999.79.4.1431. [DOI] [PubMed] [Google Scholar]
- 5.Fujikawa D G, Shinmei S S, Cai B. Eur J Neurosci. 1999;11:1605–1614. doi: 10.1046/j.1460-9568.1999.00573.x. [DOI] [PubMed] [Google Scholar]
- 6.Fujikawa D G, Shinmei S S, Cai B. Neuroscience. 2000;98:41–53. doi: 10.1016/s0306-4522(00)00085-3. [DOI] [PubMed] [Google Scholar]
- 7.Colbourne F, Sutherland G R, Auer R N. J Neurosci. 1999;19:4200–4210. doi: 10.1523/JNEUROSCI.19-11-04200.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Colbourne F, Li H, Buchan A M, Clemens J A. Stroke (Dallas) 1999;30:662–668. doi: 10.1161/01.str.30.3.662. [DOI] [PubMed] [Google Scholar]
- 9.Tsukada T, Watanabe M, Yamashima T. J Neurochem. 2001;79:1196–1206. doi: 10.1046/j.1471-4159.2001.00679.x. [DOI] [PubMed] [Google Scholar]
- 10.Lowenstein D H, Arsenault L. J Neurosci. 1996;16:1759–1769. doi: 10.1523/JNEUROSCI.16-05-01759.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.See V, Loeffler J P. J Biol Chem. 2001;276:35049–35059. doi: 10.1074/jbc.M104988200. [DOI] [PubMed] [Google Scholar]
- 12.Yang F, Sun X, Beech W, Teter B, Wu S, Sigel J, Vinters H V, Frautschy S A, Cole G M. Am J Pathol. 1998;152:379–389. [PMC free article] [PubMed] [Google Scholar]
- 13.Niquet J, Perez-Martinez L, Guerra M, Grouselle D, Joseph-Bravo P, Charli J. Brain Res Dev Brain Res. 2000;120:49–56. doi: 10.1016/s0165-3806(99)00190-x. [DOI] [PubMed] [Google Scholar]
- 14.Brustovetsky N, Dubinsky J M. J Neurosci. 2000;20:8229–8237. doi: 10.1523/JNEUROSCI.20-22-08229.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gogvadze V, Robertson J D, Zhivotovsky B, Orrenius S. J Biol Chem. 2001;276:19066–19071. doi: 10.1074/jbc.M100614200. [DOI] [PubMed] [Google Scholar]
- 16.Scorrano L, Petronilli V, Colonna R, Di Lisa F, Bernardi P. J Biol Chem. 1999;274:24657–24663. doi: 10.1074/jbc.274.35.24657. [DOI] [PubMed] [Google Scholar]
- 17.Kitana C, Kuchino Y. Cell Death Differ. 1999;6:508–515. doi: 10.1038/sj.cdd.4400526. [DOI] [PubMed] [Google Scholar]
- 18.Jensen M S, Ahlemeyer B, Ravati A, Thakur P, Mennel H D, Krieglstein J. Neurochem Int. 2002;40:285–293. doi: 10.1016/s0197-0186(01)00096-1. [DOI] [PubMed] [Google Scholar]
- 19.Carafoli E, Lehninger A L. Biochem J. 1971;122:681–690. doi: 10.1042/bj1220681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Nicholls D G, Budd S L. Physiol Rev. 2000;80:315–360. doi: 10.1152/physrev.2000.80.1.315. [DOI] [PubMed] [Google Scholar]
- 21.Brustovetsky N, Dubinsky J M. J Neurosci. 2000;20:103–113. doi: 10.1523/JNEUROSCI.20-01-00103.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Andreyev A Y, Fahy B, Fiskum G. FEBS Lett. 1998;439:373–376. doi: 10.1016/s0014-5793(98)01394-5. [DOI] [PubMed] [Google Scholar]
- 23.Didenko V V, Wang X, Yang L, Hornsby P J. J Clin Invest. 1996;97:1723–1731. doi: 10.1172/JCI118599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Miyashita T, Reed J C. Cell. 1995;80:293–299. doi: 10.1016/0092-8674(95)90412-3. [DOI] [PubMed] [Google Scholar]
- 25.Martinou J C, Green D R. Nat Rev Mol Cell Biol. 2001;2:63–67. doi: 10.1038/35048069. [DOI] [PubMed] [Google Scholar]
- 26.Zamzami N, Kroemer G. Nat Rev Mol Cell Biol. 2001;2:67–71. doi: 10.1038/35048073. [DOI] [PubMed] [Google Scholar]
- 27.Vayssiere J L, Petit P X, Risler Y, Mignotte B. Proc Natl Acad Sci USA. 1994;91:11752–11756. doi: 10.1073/pnas.91.24.11752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Liu X, Kim C N, Yang J, Jemmerson R, Wang X. Cell. 1996;86:147–157. doi: 10.1016/s0092-8674(00)80085-9. [DOI] [PubMed] [Google Scholar]
- 29.Jurgensmeier J M, Xie Z, Deveraux Q, Ellerby L, Bredesen D, Reed J C. Proc Natl Acad Sci USA. 1998;95:4997–5002. doi: 10.1073/pnas.95.9.4997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kluck R M, Martin S J, Hoffmann B M, Zhou J S, Green D R, Newmeyer D D. EMBO J. 1997;16:4639–4649. doi: 10.1093/emboj/16.15.4639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ying W, Sevigny M B, Chen Y, Swanson R A. Proc Natl Acad Sci USA. 2001;98:12227–12232. doi: 10.1073/pnas.211202598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chen J, Nagayama T, Jin K, Stetler R A, Zhu R L, Graham S H, Simon R P. J Neurosci. 1998;18:4914–4928. doi: 10.1523/JNEUROSCI.18-13-04914.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ouyang Y B, Tan Y, Comb M, Liu C L, Martone M E, Siesjo B K, Hu B R. J Cereb Blood Flow Metab. 1999;19:1126–1135. doi: 10.1097/00004647-199910000-00009. [DOI] [PubMed] [Google Scholar]
- 34.Niwa M, Hara A, Iwai T, Wang S, Hotta K, Mori H, Uematsu T. Neurosci Lett. 2001;300:103–106. doi: 10.1016/s0304-3940(01)01559-2. [DOI] [PubMed] [Google Scholar]
- 35.Honkaniemi J, Massa S M, Breckinridge M, Sharp F R. Brain Res Mol Brain Res. 1996;42:79–88. doi: 10.1016/s0169-328x(96)00121-0. [DOI] [PubMed] [Google Scholar]