Abstract
Background
To compare the corneal healing response between conventional and phototherapeutic keratectomy through a masking agent, in rabbit corneas.
Methods
24 adult rabbits underwent phototherapeutic keratectomy. Animals were divided in two groups: 12 received photoablation through a masking agent (PALM gel) and the remaining 12 received conventional phototherapeutic keratectomy of equal depth and served as control. Light and transmission electron microscopy was performed in specimens of both groups obtained: immediately after, four hours, one week, one, three and six months after treatment.
Results
Reepitheliazation was complete within five days in all eyes. Light and transmission electron microscopy did not reveal any differences of the healing process in the experimental eyes compared to the controls.
Conclusion
Photoablation through the PALM technique did not result any evident alterations of the reepithelisation and stromal healing process.
Keywords: corneal healing, gel, masking, PALM, phototherapeutic
Background
The excimer laser ablation is currently widely used primarily to correct refractive errors by altering the anterior curvature of the cornea. Due to its potential for accurate ablation of anterior stromal corneal lesions its initial clinical utilization was removal of diseased corneal tissue in a procedure called photo therapeutic keratectomy (PTK). In 1985 Serdareviz et al proposed the first therapeutic use of the excimer laser to treat an experimental Candida infectious keratitis [1]. Despite the lack of controlled clinical trials comparing PTK with other treatment modalities such as superficial keratectomy or lamellar keratoplasty, the US Food and Drug Administration (FDA) identified PTK as an alternative therapeutic approach for the treatment of superficial corneal and epithelial membrane dystrophies, irregular corneal surfaces and corneal scars and opacities.
The major inherent obstacle of PTK for the management of corneal surface irregularities is that through photoablation these irregularities are reproduced deeper within the stroma.
Masking techniques refer to photorefractive procedures utilizing various masking means or so called modulators to protect flatter corneal areas while steeper areas are excised with the excimer laser.
Kornmehl et al [2] have shown that an ideal masking agent should have moderate viscosity (between that of saline and 1% carboxymethylcellulose) and concluded that very viscous fluids would not cover irregular surfaces uniformly whereas fluids of inadequate viscosity would run off quickly exposing both peaks and valleys thus resulting in irregular surfaces after ablation.
Numerous previous investigators have used different masking agents as methylcellulose [3-6] and sodium hyaluronate [5,7,8] at various concentrations or performed transepithelial (so that the epithelium acts as a natural masking agent) treatments [5,6,9,10] and reported the beneficial effect of masked PTK. Fasano et al [3] reported that the ideal masking agent should have the same ablation rate to that of the cornea, be biocompatible and adhere well to the cornea.
These properties have been met with the use of collagen solutions as modulators. Their applicability as masking agents for corneal photoablation was examined in experimental models of animal and cadaver eyes (Scott et al: Collagen modulator fluids for use during photoablation keratectomy: ARVO 1992, Margaritis et al. Properties of a new two component gel material as an aid in PRK corneal remodeling: ARVO 1994) [11-13] as well as in limited clinical trials of partially sighted eyes (Pallikaris et al: Photoablated lenticular modulator PALM technique: A report of ten cases: ARVO 1998) [14,15].
Photo-Ablatable Lenticular Modulator (PALM) technique refers to the use of a modified collagen gel solution for the photorefractive correction of corneal surface irregularities [14]. The PALM gel similarly to other collagen modulators [12,13,15] is thermo reversible. The gel is in liquid state when heated to solidify to a firm gel as its temperature lowers. Its use for masking purposes requires its application onto the corneal stoma at a temperature of 49°C where it can be molded to form a stable lenticule that serves as the final masking agent.
At temperatures of 40° to 53°C, the collagen individual fibril bonds are broken and the collagen denatures to its parent gelatinous form. When the tissue temperature drops to below 40°C, these bonds can reunite with possible rearrangement of the matrix but without any cellular damage [16]. Under this consideration the thermal contact of the 49°C preheated PALM gel onto the bare corneal stroma for the in situ molding of the lenticule, would not be expected to induce any irreversible changes of the corneal collagen chains. However its sequential use as masking agent for PTK could affect the laser-tissue interaction and probably the corneal healing response to irradiation.
In the current study we examined the possible implication of the PALM gel when used as described above, to the corneal wound healing on rabbit corneas.
We examined the reepithelization process of 12 rabbit eyes after PTK through PALM gel lenticules formed in situ. We used a moving slit delivery system in a 5 mm ablation zone confined by means of a metal iris diaphragm. An equal group of rabbit eyes received conventional PTK of the same depth and treatment zone and served as control.
Methods
The PALM gel as a masking agent
The gel used, as modulator is a soluble mixture of purified type A gelatin from porcine skin and a polysaccharide, type III carrageenan K in nanopure water [17]. Both components are commercially available for laboratory use (Sigma Chemical Co, St Louis, Mo).
Being thermoreversible, the gel can be stored at room temperature in solid form. When heated to 49°C it liquefies to a high viscosity fluid that solidifies within minutes when exposed to room temperature. When still in liquid form the gel can be molded by means of a rigid contact lens to form a stable lenticule that its upper surface will reproduce the inner surface of the molding lens.
If the lenticule is molded in situ onto the cornea its bottom surface will fill any corneal surface irregularity. As shown by testing on freshly excised porcine corneas (fig. 1) the mixture for the preparation of the gel is conditioned so that its solid form to have the same ablation to that of the cornea [13]. The full removal of the lenticule with photoablation in PTK mode would be expected to reproduce the lenticule's upper surface onto the surface of the cornea. Selecting the appropriate inner radius of curvature of the molding lens, the upper surface of the formed lenticule can be of variable curvature. Instillation of 100 μm of the preheated liquid solution [18] on the corneal surface requires 4 minutes for the lenticule to acquire the mechanical stiffness required to withstand the stress induced by the lens removal, at room temperature of 25°C. After this time the lens mold is removed and the lenticule is ablated through with irradiation in PTK mode. An effective amount of fluorescein in the lenticule mixture provides the surgeon visual feedback for the ablation progress. Fluorescence fades as soon as the lenticule is fully removed.
Figure 1.

Ablation rate of the gel was examined on irregular porcine cornea developed by means of 60 microns deep phototherapeutic keratectomy through a metal grid. The irregular cornea created was fully covered with PALM gel. Half of the cornea was ablated until the gel was fully removed. Ablation resulted in smooth cornea. Microphotograph. Original magnification ×
Surgical procedure
A total of 24 adult New Zealand white rabbits (average weight 4–5 Kg) were anaesthetized by intramuscular injection of ketamine hydrochloride (40 mg/kg) and xylazine hydrochloride (7 mg/kg) mixture. All experimental procedures were carried out in accordance with the Guiding Principles in the Care and Use of Animals (DHEW Publication, NIH 80–23) and were approved from the University's of Crete ethics committee.
One drop of 0.5% proparacaine hydrochloride (Alcon, Ft Worth, TX) was instilled into each eye and the corneas were deepithelized with the use of a rotating brush. We treated one eye of each animal. 12 were treated with PTK through PALM gel (experimental eyes) and the remaining 12 received conventional treatment and served as controls. The laser system used in all eyes was the Aesculap Meditec Mel 60 (Karl-Zeiss, Jena, Germany), with laser fluence of 180 mJ/cm2 pulse rate at 20 Hz, in PTK mode.
Experimental eyes received PTK through PALM gel lenticules formed in situ onto deepithelized corneas as described above. In order to shorten the time of the surgical procedures we obtained the thinner possible lenticules using as molds the "best fitting" contact lenses for each operative eye. Molds used in the current study had inner radii of curvature ranging from 8.25 to 9 mm. After the removal of the mold, an iris diaphragm having an inner aperture of 5 mm was centrated over the pupil onto the upper surface of the lenticule to define the ablation zone. The gel lenticules were ablated through with irradiation in PTK mode. As soon as fluorescence faded each experimental eye received an additional 60-micron deep PTK.
The control eyes were submitted to a phototherapeutic keratectomy of 5 mm diameter ablation zone (confined by the same iris diaphragm), depth of 60 μm, on deepithelised cornea without the use of any masking agent.
Postoperative care
Postoperative treatment of all treated eyes included daily application of combined tobramycin (0.3%), dexamethasone (0.1%) ointment (Alcon Ft Worth, TX) until reepithelisation was complete. No topical steroids were used subsequently in the study.
The reepithelization process was examined daily with staining of the epithelial defect area using 2% fluorescein dye and a hand held slit-lamp. Corneal clarity was recorded using a predetermined 5-scale grading of haze [19].
Two animals of each group (experimental and control) were sacrificed with an overdose of penthobarbital sodium through the marginal ear vein at each of the following postoperative intervals: immediately after treatment, 4 hours after treatment, 1 week and at one, three and six postoperative months.
Histology processing
Immediately after enucleation the eyes were fixed using 2.5% gludaraldehyde in 0.1 mol/l cacodylate buffer (pH 7.4). After short prefixation the corneas were excised and placed in the same fresh fixative overnight. The tissue samples were post fixated in 2% osmium tetroxide in 0.1 M cacodylate buffer (Ph 7.4) for 2 hours at 4°C, dehydrated in a series of alcohol and propylene oxide and embedded in epoxy resin.
1-μm sections were stained with trichrome stain and were processed for light microscopic examination whereas ultra thin sections were stained with uranyl acetate and lead citrate and were examined with transmit ion electron microscopy.
Results
Clinical course
Reepithelisation was complete (no epithelial defect was observed with fluorescein staining) within five days in all treated eyes (within 3 days in two control and one experimental eye, 4 days in three experimental and one control eye and on the fifth postoperative day to the rest of the treated eyes). No persistent defects or recurrent erosions were recorded.
Corneal haze was evident from the first week in all treated eyes ranging from grade 1 to grade 2.
Corneal haze reached its maximum on the first postoperative month ranging from grade 2 to 3 (grade 3 in one experimental eye).
All treated eyes were clear or had minimal haze by the third month (grade 1 in one experimental and two control eyes), which was resolved in all eyes by the sixth postoperative month.
Histology
In the specimens taken immediately after treatment, the surface of corneal stroma was covered by a pseudomembrane typical after excimer laser. The control group didn't demonstrate any difference in pseudomembrane thickness or regularity in comparison to the experiment. The most typical corneal morphology is demonstrated in fig 2. The appearance of collagen fibrils and stromal keratocytes was close to normal in both groups of specimens.
Figure 2.
Experimental cornea. Immediately after the procedure, the pseudomembrane (arrowheads) that was formed during ablation, has local thickness irregularities. cf – collagen fibers Electron microphotograph. Original magnification × 6600.
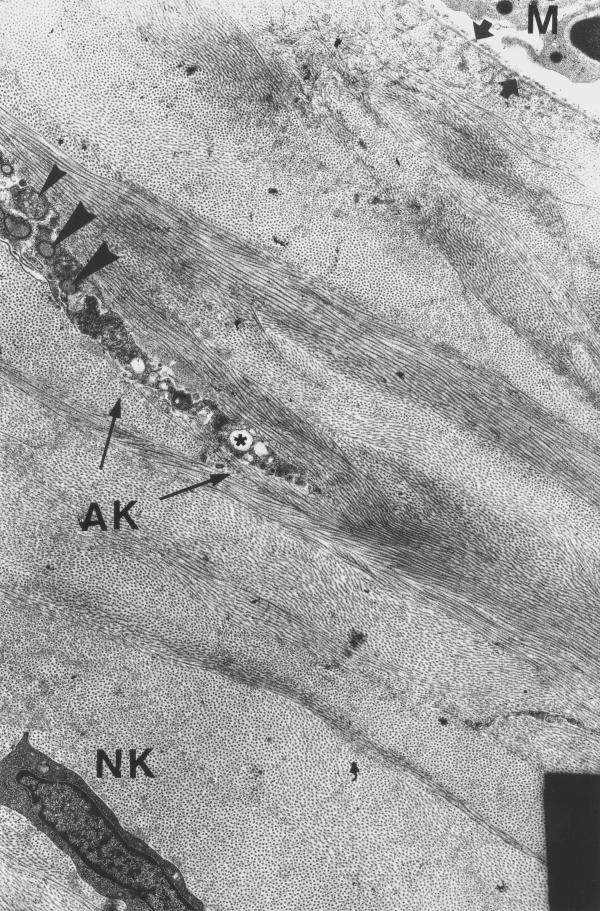
4 hours after irradiation, the distribution of collagen fibers under the ablated area was irregular due to the occurrence of corneal edema. Isolated macrophages were observed on the stromal surface. Keratocytes situated within the 20–40 μm of the upper stroma, demonstrated various pathological changes including cytoplasm vacuolization and blebbing, with formation of cellular fragments (fig 3). Keratocytes located deeper than 40 μm under ablated stromal surface did not demonstrate any noticeable morphological alterations. We observed no differences in the type, degree and extent of the pathological changes of keratocytes under the ablated surface between the control and experimental specimens. In the endothelial corneal layer, both inside and outside the ablation area, we didn't observe any morphological abnormalities, neither in the experimental nor in the control groups. The nuclear chromatin, abundant mitochondria, reticulum and coated vesicles, had normal appearance. The cells preserved their apical junctional complexes and well developed intercellular gap junctions.
Figure 3.
Experimental cornea 4 hours after the procedure. Macrophages (M) appear on the corneal surface covered by pseudomembrane (arrows). Abnormal keratocyte (AK) with vacuolized cytoplasm (asterisk), blebbing (big arrowheads) and cytoplasmic fragments surrounded by cell's membrane (small arrowheads). Deeper in the cornea, a normal keratocyte (NK) may be seen, whose cytoplasmic and nuclear structures are typical for corneal stroma. Electron microphotograph. Original magnification × 3300.
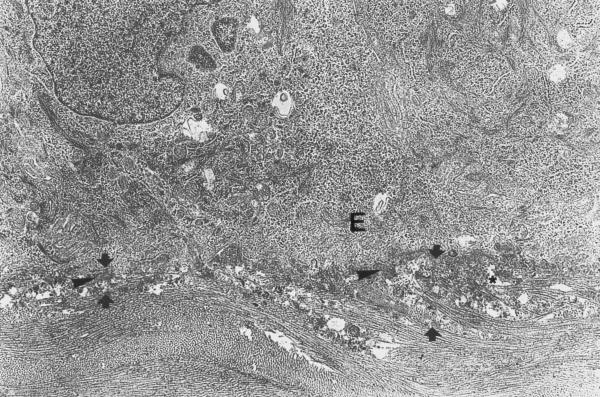
In the specimens obtained 1 week after the procedure, the epithelium covering the stroma in the ablated zone demonstrated mild hyperplasia. The typical desmosomal contacts between the basal cells, which are very characteristic for the intact stromal epithelium, were rather rare (fig 4). However, adhesive contacts with significant resemblance with zonula adherens were observed. The morphological appearance of the regenerating basement membrane that was removed during the procedure, corresponded to the initial phases of its reconstruction: formation of characteristic amorphous layer of extracellular matrix was evident in some contact zones of the epithelial cells with stroma. In some places this layer had focal discontinuities or duplication. Typical hemidesmosomes were not present. However, few small sized hemidesmosomes with low electron density of cytoplasmic plaque were observed. The presence of these immature hemidesmosomes was considered as an evidence of adhesive contacts' reconstruction between the epithelium and the basement membrane. At this stage, inflammatory cells infiltration was rarely observed in all obtained specimens. The anterior stroma beneath the treatment zone consisted of newly synthesized extracellular matrix with collagen fibers of irregular size and distribution, as well as few amorphous inclusions and "empty" spaces giving this layer a "foamy" appearance. The thickness of this locally discontinued "foam" layer did not exceed 3–4 μm. The corneal endothelium of the 1 week specimens of both groups had normal appearance. A discontinuous layer of an electron dense substance was observed beneath the boarder between the endothelial layer and Descemet's membrane (fig 5).
Figure 4.
Experimental cornea 1 week after the procedure. In the anterior stroma under the epithelium, appearance of chaotically distributed collagen fibers is observed (between the arrows), as well as of amorphous inclusions strictly under the basal parts of epithelial cells (arrowheads), and of small "empty" spaces in the extracellular matrix (asterisk). Original magnification × 3300.
Figure 5.
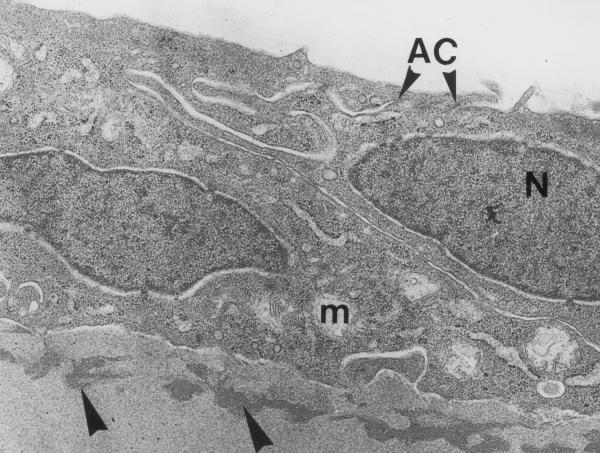
Experimental cornea 1 week after the procedure. The cells of corneal endothelium demonstrate normal appearance of cell structures and intercellular contacts. Under the endothelial layer, the Descemet's membrane has a discontinuous layer of an electrondense substance (big arrowheads). m – mitochondria AC – apical junctional complex N – nucleus Original magnification × 3300.
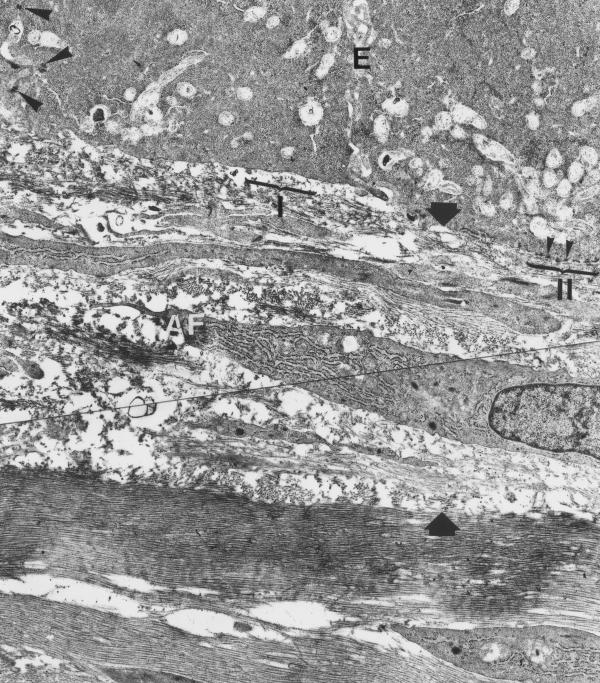
One month postoperatively, no epithelial irregularities were observed (fig 6). The desmosomes between epithelial cells, as well as the hemidesmosomes between the basal epithelial cells and the basal lamina, were more frequently observed and although irregularly distributed, their morphology was close to the normal structure of contacts of the intact epithelium. The irregular distribution of hemidesmosomes can be justified by their concurrent reconstruction with the basal lamina formation. The thickness of the "foam" layer mostly consisted of activated fibroblasts, achieved its maximum (10–15 μm) by this interval. The deeper corneal layers demonstrated exquisitely regular structure (fig 7).
Figure 6.
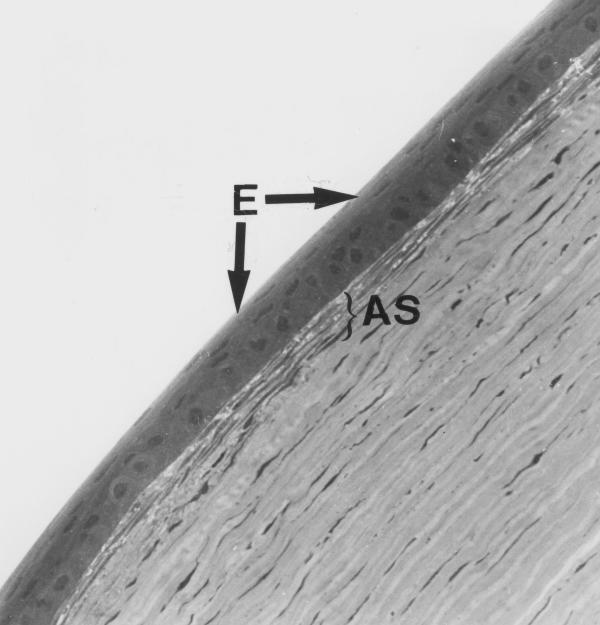
Experimental cornea. Light microphotograph of the ablation zone 1 month after the procedure. The epithelial layer (E) covering the stroma has normal appearance, with the possible exception of minor thickness variations. The anterior stroma (AS) under the epithelium may be distinguished by a big number of cell elements (fibroblasts); the structure of extracellular matrix in the subepithelial stroma differs significantly from that of extracellular matrix of deeper layers. Note the absence of inflammatory cells in the subepithelial stroma. Original magnification × 130.
Figure 7.
Experimental cornea 1 month after the procedure. Adhesive contacts between epithelial cells are represented by normal desmosomes typical for corneal epithelial layer (big arrowheads). Hemidesmosomes (small arrrows) between basal epithelial cells and basement membrane are distributed extremely irregularly (compare zones I and II). The foamy layer (between big arrows) is significantly thicker in comparison to 1-week post-op specimens. Numerous activated fibroblasts (AF) are observed within this layer. Electron microphotograph. Original magnification × 3300.
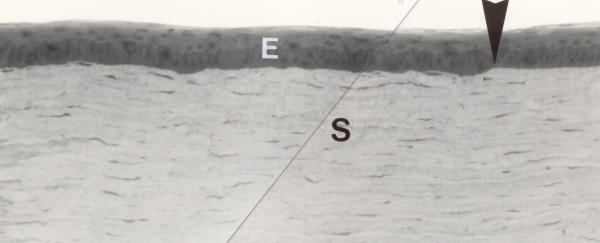
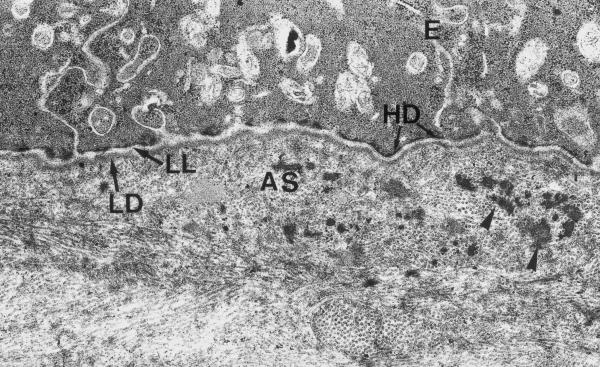
Three months postoperatively, the epithelium covering the ablation zone exceeded normal thickness only in very few locations (fig 8). The morphology of epithelial cells, the number of layers and the intercellular contacts were close to normal. The structure of the epithelial basal lamina was also appeared close to normal: lamina densa and lamina lucida were well distinguishable with rare branching and duplication. The hemidesmosomes were regularly distributed, and had normal morphology. The thin (2–3 μm) layer of anterior stroma beneath the treatment zone was close to normal, with rare exceptions of aggregations of electron dense substance, few irregularly located collagen fibres with small variations of their diameter (fig 9). The deeper stromal layers had normal architecture, with no signs of scarring, inflammatory infiltration or deformation, neither in the experimental nor in the control specimens.
Figure 8.
Experimental cornea 3 months after the procedure. Light microscopy of the ablation zone. The border of the ablation zone is indicated by a big arrowhead. The epithelium (E) in the ablated zone (to the left of the arrowhead) has local thickness irregularities. However, the cells have mostly normal appearance. No signs of inflammatory reaction. The "foamy" layer in the stroma (S) is practically indistinguishable in light microphotographs. Original magnification × 130.
Figure 9.
Experimental cornea 3 months after the procedure. The basal part of epithelial cells demonstrates well-developed adhesive contacts. Hemidesmosomes (HD) have normal structure and regular distribution along basal lamina. The thickness and electron density of lamina densa (LD) and lamina lucida (LL) have returned back to normal values. The layer of anterior stroma (AS) contains aggregations of electrondense substance (arrowheads). The fibers of this layer have attained more regular distribution in comparison to 1-month post-op specimens. Electron microphotograph. Original magnification × 10 000.
Six months postoperatively, we didn't find any significant morphological difference compared to the three-month interval obtained specimens, neither in the experimental nor in the control group.
Discussion
Application of a heated agent on denuded cornea or even ablation through a chemical agent could affect laser-tissue interaction, corneal reepithelization or stromal healing response after photo ablation.
We used rabbits to examine the probable effect of the PALM gel if used as a masking agent for PTK since stromal healing is very similar in rabbits and humans [20].
The healing course proved similar in the experimental and control eyes. No specimens obtained at any postoperative interval had any sign of necrosis or macrophages infiltration.
The simultaneous reepithelization as well as the similar histological findings between experimental specimens and controls is indicative of negligible gel's impact on corneal healing response. The newly synthesized extracellular matrix presented as foam layer at the early specimens is a common finding reported after photo refractive keratectomy or even simple epithelial scrape injury in a number of previous studies [21-24] and is supposed to manifest the healing response of rabbit corneas [21,22,24].
The epithelial hyperplasia observed on the seventh postoperative day was more intense than that reported in the literature [21,24,25]. The possibility of the gel's implication to this response is minimized since it was also observed in the control eyes. We assume that this finding is related to the ablation profile of our treatments (a rather deep keratectomy with sharp edges), which would justify an intense epithelial healing response [26].
The extrusion of electron dense fibro-granular material into the descemet's membrane is also a common finding observed in a number of previous studies [20,21,27-29] and has been attributed to acoustic shock waves by irradiation [24] or the distraction of the epithelial integrity after trauma [29]. The presence of this finding in only one of our specimens may be related to the homogenous and smooth removal of the corneal epithelium with the rotating brush.
In conclusion the use of the PALM gel did not seem to seriously affect the healing process after PTK on rabbit corneas as compared to controls. A larger number of animals would allow for statistical analysis of pathologic findings such as activated fibroblasts by depth and thickness of newly formed extracellular matrix between experimental and control eyes. Furthermore, tenascin and fibronectin staining of the specimens would allow for better understanding of the corneal healing process.
The use of new generation laser systems that offer phototherapeutic ablation mode with transition zones are expected to minimize epithelial hyperplasia and corneal stroma healing response. The major remaining drawback of the PALM as well as of similar PTK techniques [15] in order to obtain an optimal refractive result is the accurate centration and placement of the molding lens for the proper formation of the lenticule before irradiation.
Competing Interests
Authors Ioannis G Pallikaris and Harilaos S Ginis are patent holders and have proprietary interest in PALM technique. The rest of the authors have no proprietary interest.
Acknowledgements
The study was supported by a research grant (August 1999) from the LASIK Institute (LASIK Institute, 750 Washington Street, box 450, Boston Massachusetts 02111, USA).
Pre-publication history
The pre-publication history for this paper can be accessed here:
Contributor Information
Vikentia J Katsanevaki, Email: vikatsan@med.uoc.gr.
Harilaos S Ginis, Email: ginis@med.uoc.gr.
Irini I Naoumidi, Email: maoumidis@hotmail.com.
Ioannis G Pallikaris, Email: pallikar@her.forthnet.gr.
References
- Serdareviz O, Darrell RW, Krueger RR, et al. Excimer laser therapy for experimental Candida Keratitis. Am J Ophthalmol. 1985;99:534–538. doi: 10.1016/s0002-9394(14)77952-8. [DOI] [PubMed] [Google Scholar]
- Kornmehl EW, Steinert RF, Puliafito CA. A comparative study of masking fluids for excimer laser phototherapeutic keratectomy. Arch Ophthalmol. 1991;109:860–863. doi: 10.1001/archopht.1991.01080060124039. [DOI] [PubMed] [Google Scholar]
- Fasano PA, Moreira H, Mc Donnel PJ, Sinbawy A. Excimer laser smoothing with a re producible model of anterior corneal surface irregularity. Ophthalmology. 1991;98:1782–1785. doi: 10.1016/s0161-6420(91)32050-5. [DOI] [PubMed] [Google Scholar]
- Gartry D, Kerr Muir M, Maarshall J. Excimer laser treatment of corneal surface pathology a laboratory and clinical study. Br J Ophthalmol. 1991;75:258–269. doi: 10.1136/bjo.75.5.258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fagerholm P, Fitzimmmons TD, Orndhal M, et al. Phototherapeutic keratectomy: long term results in 166 eyes. Refract Corneal Surg. 1993;9:S76–S81. [PubMed] [Google Scholar]
- Hersh PS, Spinak A, Garrana R, Mayer M. Phototherapeutic keratectomy: Strategies and results in 12 eyes. Refract Corneal Surg. 1993;9:S90–S95. [PubMed] [Google Scholar]
- Alio JL, Belda JI, Shalaby MM. Correction of irregular astigmatism with excimer laser assisted by sodium hyaluronate. Ophthalmology. 2001;108:1246–1260. doi: 10.1016/S0161-6420(01)00602-9. [DOI] [PubMed] [Google Scholar]
- Dogru M, Katakami C, Yamanka A. Refractive changes after excimer laser phototherapeutic keratectomy. J Cataract Refract Surg. 2001;27:686–692. doi: 10.1016/S0886-3350(01)00802-1. [DOI] [PubMed] [Google Scholar]
- Mc Donnel PJ, Seiler T. Phototherapeutic keratectomy for Reis-Buckler's corneal dystrophy. Refract Corneal Surg. 1992;8:306–310. [PubMed] [Google Scholar]
- Talamo JH, Wagoner MD, Lee SY. Management of ablation decentration following excimer photorefractive keratectomy. Arch Ophthalmol. 1995;113:706–707. doi: 10.1001/archopht.1995.01100060030018. [DOI] [PubMed] [Google Scholar]
- Englanoff JS, Kolahdouz-Isfahani AH, Moreira H, Cheung DT, Nimni ME, Trockel SL, Mc Donnel PJ. In situ collagen mold as an aid in excimer laser superficial keratectomy. Ophthalmology. 1992;99:1201–1208. doi: 10.1016/s0161-6420(92)31822-6. [DOI] [PubMed] [Google Scholar]
- De Vore DP, Scott JB, Nordquist RE, Hoffman RS, Nguyen H, Eifferman RA. Rapidly polymerized collagen gel as a smoothing agent in excimer laser photoablation. J Refract Surg. 1995;11:50–55. doi: 10.3928/1081-597X-19950101-13. [DOI] [PubMed] [Google Scholar]
- Stevens SX, Bowyer BL, Sanchez-Thorin JC, Rocha G, Young DA, Rowsy JJ. The BioMask for treatment of corneal surface irregularities with excimer Laser phototherapeutic keratectomy. Cornea. 1999;18:155–63. doi: 10.1097/00003226-199903000-00003. [DOI] [PubMed] [Google Scholar]
- Pallikaris IG, Katsanevaki VJ, Ginis HS. The PALM technique as an alternative to Customized Ablation. Seminars in Ophthalmology. 2000;15:160–169. [Google Scholar]
- Kremer F, Aronsky M, Bowyer BL, Stevens SX. Treatment of corneal surface irregularities using BioMask as an adjunct to excimer laser phototherapeutic keratectomy. Cornea. 2002;21:28–32. doi: 10.1097/00003226-200201000-00007. [DOI] [PubMed] [Google Scholar]
- Ohshiro T, Galderhead RG. Reversible and irreversible photobioreactions. In: Ohshiro T, Galderhead RG, editor. Low level laser therapy: A practical introduction. John Willey & sons Ltd; 1988. pp. 25–26. [Google Scholar]
- Pallikaris IG. inventor: Photoablatable Lenticular Modulator:US Patent 09/139,368. 1998 August 25.
- Pallikaris IG, Ginis HS. inventors: Device for the shaping of a substance on the surface of the cornea. PCT/1B01/01088. April 20 2001.
- Fantes FE, Hanna KD, Warring GO, III, Pouliquen Y, Thompson KP, Salvodelli M. Wound healing after excimer laser keratomileusis (photorefractive keratetomy) in monkeys. Arch Ophthalmol. 1990;108:665–675. doi: 10.1001/archopht.1990.01070070051034. [DOI] [PubMed] [Google Scholar]
- Talamo JH, Gollamudi S, Green WR, De la Cruiz Z, Filatov V, Stark WJ. Modulation of corneal wound healing after excimer laser keratomileusis using topical mitomycin C and steroids. Arch Ophthalmol. 1991;109:1141–1146. doi: 10.1001/archopht.1991.01080080101040. [DOI] [PubMed] [Google Scholar]
- Hanna KD, Pouliquen Y, Waring GO, III, Salvodelli M, Cotter J, Morton K, Menache M. Corneal stromal wound healing in rabbits after excimer laser surface ablation. Arch Ophthalmol. 1989;107:895–901. doi: 10.1001/archopht.1989.01070010917041. [DOI] [PubMed] [Google Scholar]
- Amm M, Wetzel W, Winter M, Uthof D, Duncker GIW. Histopathological comparison of photorefractive keratectomy and laser in situ keratomileusis in rabbits. J Refract Surg. 1996;12:758–766. doi: 10.3928/1081-597X-19961101-07. [DOI] [PubMed] [Google Scholar]
- Wilson SE. Molecular cell biology for the refractive corneal surgeon: Programmed cell death and wound healing. J Refract Surg. 1997;13:171–175. doi: 10.3928/1081-597X-19970301-15. [DOI] [PubMed] [Google Scholar]
- Wang Y, Zhao K, Wang H. Histopathology of corneal wound healing after photorefractive keratectomy in rabbit eyes. J Refract Surg. 1998;14:S209–11. doi: 10.3928/1081-597X-19980401-15. [DOI] [PubMed] [Google Scholar]
- Del Pero RA, Gigstad JE, Roberts JR. A refractive and histopathologic study of excimer laser keratectomy in primates. Am J Ophthalmol. 1990;109:419. doi: 10.1016/s0002-9394(14)74608-2. [DOI] [PubMed] [Google Scholar]
- Heitzman J, Binder PS, Kassar BS, Nordan ST. The correction of high myopia with the excimer laser. Arch Ophthalmol. 1993;111:1627–1634. doi: 10.1001/archopht.1993.01090120049021. [DOI] [PubMed] [Google Scholar]
- Seiler T, Fantes FE, Waring GO, III, Hanna K. Laser corneal surgery. In: Warring GO III, editor. Refractive keratectomy for myopia and astigmatism. St Louis Mo: Mosby year book Inc; 1992. pp. 669–745. [Google Scholar]
- Krueger RR, Binder PS, McDonnel PJ. The effects of excimer laser photoablation of the cornea. In: Salz JJ, editor. Corneal laser surgery. St Louis Mo: Mosby year book Inc; 1995. pp. 11–44. [Google Scholar]
- Sano Y, Itoh Y, Tsuneoka H, Ohki K, Sakabe I, Kitahara K, Okamoto S. Changes in descemet membrane and endothelium after corneal epithelial abrasion alone and with photorefractive keratectomy in rabbits. Arch Ophthalmol. 1996;114:1105–1108. doi: 10.1001/archopht.1996.01100140307009. [DOI] [PubMed] [Google Scholar]