Abstract
Bilirubin clearance is one of the numerous important functions of the liver. Defects in this process result in jaundice, which is particularly common in neonates. Elevated bilirubin levels can be decreased by treatment with phenobarbital. Because the nuclear hormone receptor constitutive androstane receptor (CAR) mediates hepatic effects of this xenobiotic inducer, we hypothesized that CAR could be a regulator of bilirubin clearance. Activation of the nuclear hormone receptor CAR increases hepatic expression of each of five components of the bilirubin-clearance pathway. This induction is absent in homozygous CAR null mice but is observed in mice expressing human CAR instead of mouse CAR. Pretreatment with xenobiotic inducers markedly increases the rate of clearance of an exogenous bilirubin load in wild-type but not CAR knockout animals. Bilirubin itself can also activate CAR, and mice lacking CAR are defective in clearing chronically elevated bilirubin levels. Unexpectedly, CAR expression is very low in livers of neonatal mice and humans. We conclude that CAR directs a protective response to elevated bilirubin levels and suggest that a functional deficit of CAR activity may contribute to neonatal jaundice.
Bilirubin is an oxidative end product of heme catabolism. A significant amount of bilirubin is produced every day (250–400 mg in adult humans), primarily from breakdown of hemoglobin. This is ordinarily removed from the body by the liver, which excretes it into the bile. Failure of this process results in the accumulation of this yellow pigment and jaundice. Bilirubin is very hydrophobic and in chronic jaundice it accumulates in the CNS, resulting in neurotoxicity that can progress to potentially fatal encephalopathy.
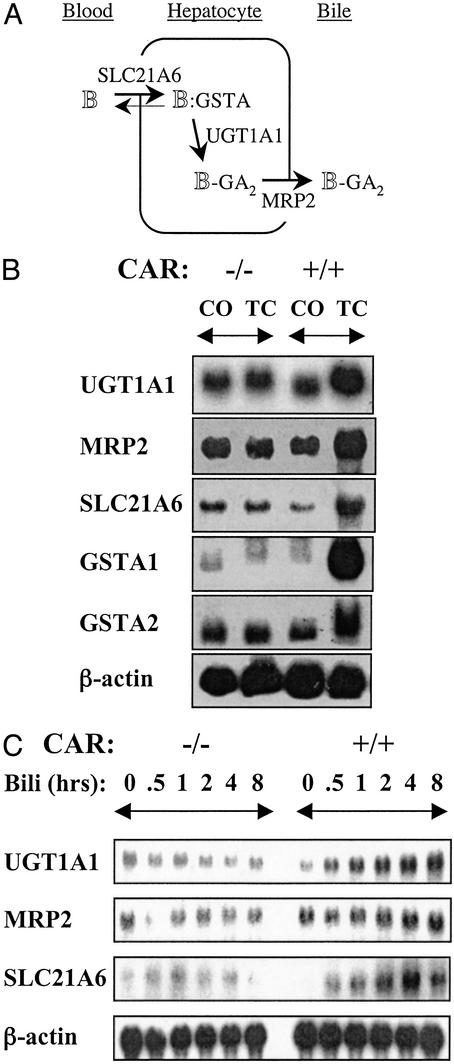
The pathway for clearance of this toxic product is relatively well defined (Fig. 1A; refs. 1 and 2). In the circulation, bilirubin is bound noncovalently to serum albumin. Free bilirubin enters the liver via the sinusoidal surface of the hepatocytes by facilitated diffusion, which is mediated by the organic anion transporter SLC21A6 (3). Although this transport is bidirectional, binding to cytosolic ligandin, a homodimer or heterodimer of glutathione S-transferase (GST)A1 and GSTA2, reduces efflux and increases net uptake. Bilirubin is glucuronidated in the endoplasmic reticulum by a specific bilirubin UDP-glucuronosyltransferase (UGT1A1). Complete loss of UGT1A1 function in the type 1 form of Crigler–Najjar syndrome results in severe hyperbilirubinemia that is fatal unless treated by liver transplantation (4). Bilirubin diglucuronide is secreted across the bile–canalicular membrane of the hepatocyte by an active transporter, MRP2 (multidrug resistance-associated protein 2, also known as cMOAT or ABC-C2). Abnormalities of excretion of conjugated bilirubin due to the loss of function of MRP2 result in Dubin–Johnson syndrome, which is characterized by accumulation of the glucuronidated product (5).
Figure 1.
(A) CAR mediates xenobiotic and bilirubin induction of genes involved in bilirubin clearance. (B) Wild-type (+/+) and CAR knockout (−/−) animals (n = 3) were pretreated with TCPOBOP (TC, 3 mg/kg) or corn oil (CO). Total liver RNA was prepared, and equivalent amounts of RNA pooled from three individuals were analyzed by Northern blotting with the indicated probes. (C) Mouse primary hepatocytes from wild-type (+/+) or CAR knockout (−/−) animals were cultured in the presence of 100 μM bilirubin (Bili) for different times as indicated. Total RNA was prepared and analyzed by Northern blotting with the indicated probes.
It has long been known that phenobarbital (PB), a well studied inducer of drug metabolism, can increase bilirubin-conjugating activity and decrease elevated bilirubin levels in humans (6, 7). Results with knockout mice demonstrate that the nuclear hormone receptor constitutive androstane receptor (CAR, NR1I3) mediates the response of the liver to specific xenobiotics, particularly PB and other “PB-like” inducers (8). Thus, we hypothesized that CAR plays an important role in bilirubin metabolism. Results with wild-type and CAR knockout mice support this hypothesis by confirming that CAR can activate expression of five known components of the bilirubin pathway. In wild-type but not knockout mice, activation of CAR results in increased clearance of an acute dose of bilirubin. Bilirubin can induce its own conjugation (9, 10), and this response is also lost in the CAR knockout animals, which show defective clearance of chronically elevated bilirubin levels. Unexpectedly, CAR expression is low in neonatal mice and humans. We suggest that this functional deficit may contribute to neonatal jaundice.
Materials and Methods
Animals and Treatments.
Mice were housed in a pathogen-free animal facility under a standard 12-h light/dark cycle. Mice were fed standard rodent chow and water ad libitum or given water containing 0.1 mg/ml phenylhydrazine in aluminum foil-wrapped bottles. The drug-containing water was changed every 2 days. After 30 days, blood was collected and transferred to T-MGA tubes (Terumo Medical Corporation, Elkton, MD) and serum was prepared by centrifugation at 1,200 × g for 10 min. Mice were injected via the tail vein with bilirubin (10 mg/kg) dissolved in an isotonic solution containing 0.5 g of Na2CO3 and 0.52 g of NaCl per 100 ml. After 1 h, blood samples were collected, and bilirubin levels were determined by using total and directed bilirubin kits (Sigma).
The albumin promoter was used to generate a transgene designed to direct expression of human CAR (hCAR) in the liver. This construct includes an intron from β-globin and a polyadenylation site from bovine growth hormone to promote expression and is analogous to that used previously to generate mice expressing only human pregnane X receptor (PXR) (11). The hCAR transgenic line was crossed with CAR nulls, resulting in mice expressing hCAR but not murine CAR (mCAR) in the liver as described (12).
RNA Analysis.
Total RNA was extracted from mouse liver by using the midi RNeasy kit (Qiagen, Valencia, CA). Equivalent amounts of RNA from three mice were pooled, and 10 μg was used for Northern blot analysis. Except for β-actin, cDNA probes were generated by PCR from reverse-transcribed liver poly(A)+ RNA. The primers used were UGT1A1 (5′-AGATGCTGATCCAGCAACTG-3′ and 5′-GGCTTCTTGGTAACACAG-3′), MRP2 (5′-GTTCTGTCCAACGCCCTCAAC-3′ and 5′-CTCTGCCCTATGCTCAGGTTG-3′), SLC21A6 (5′-GCTGTGTTTCAAACAACCC-3′ and 5′-CAATGTTTGGTACAACATGTGC-3′), GSTA1 (5′-CCGGGAAGCCCGTGCTTCAC-3′ and 5′-CTGAATCTTGAAAGCCTTCC-3′), GSTA2 (5′-AGAAGGAGTGGCGGATCTGG-3′ and 5′-TATCCGAGGAAGTGATCATG-3′), CAR (5′-CCGCCTCTAGATGGAGGACGCAGTCCAT-3′ and 5′-CGCGCGGATCCTGTTCAGCATTTCATTGCC-3′), and PXR (5′-CCGCCTCTAGAAGCTACAGCTGCATAAGG-3′ and 5′-CCGCCGGATCCTTAACTCATAGGCCAGCAC-3′). A single blot was hybridized sequentially with the different probes.
Mouse primary hepatocytes were prepared and maintained in William's E defined medium (Invitrogen) supplemented with 10 μg/ml insulin (Sigma)/10 μg/ml transferrin (Sigma)/25 ng/ml epidermal growth factor (Collaborative Research)/25 μg/ml gentamycin/100 nM somatotropin (Sigma)/1 μM dexamethasone (Sigma). Twenty-four hours after isolation, cells were treated with 100 μM bilirubin for various times. Bilirubin was initially dissolved in a small volume of 0.1 M NaOH and adjusted to the desired final concentration with sterile PBS (pH 7.4). Bilirubin solutions were freshly prepared under low-light conditions for each experiment.
For studies of CAR expression in humans, neonatal tissues were normal livers from autopsies of 1-, 3-, and 8-week-old individuals. One adult liver sample was obtained surgically from a normal liver; the two others were from autopsies. Samples were obtained by R.G. under appropriate guidelines as described (13). Equivalent amounts of total RNA extracted from the liver tissue were pooled, and 10 μg of each sample was used for Northern blotting by using an hCAR-specific cDNA probe from the ligand-binding domain. The same blot was reprobed with β-actin as loading control.
Protein Analysis.
Primary mouse hepatocytes were prepared and maintained in control medium. After 24 h, 1 mM PB or 100 μM bilirubin was added, and cells were incubated for an additional 3 h. Cells were collected, and nuclear extract was prepared as described (14). Protein (20 μg) was fractionated on a 10% polyacrylamide gel and immunoblotted with an mCAR antibody generously provided by Masahiko Negishi (14). Western blotting was done by using the ECL kit from Amersham Pharmacia.
For expression of hCAR, extracts were prepared from the three human neonatal and adult liver samples, and equivalent amounts were pooled. Fifty micrograms of each pooled sample was fractionated on 10% polyacrylamide gel and immunoblotted with an hCAR antibody generated by Quality Controlled Biochemical (Camarillo, CA) using the hCAR peptide QRDEIDQLQEEMALTLQSYIK. The same blot was stripped and then immunoblotted with actin monoclonal antibody (1:1,000, Santa Cruz Biotechnology).
Transfections.
HepG2 cells were plated into 24-well dishes with DMEM supplemented with 10% charcoal-stripped serum. Transfections used Fugene 6 reagent according to manufacturer instructions with 100 ng of pCMX-Gal4-mCAR-LBD or pCMX-Gal4 as control, 300 ng of pG5E1b luciferase reporter, and 100 ng of pSV2-β-galactosidase as internal control for transfection efficiency. Twelve hours after transfection, different concentrations of bilirubin were added and incubated for an additional 24 h.
Results
CAR Activation of the Bilirubin-Clearance Pathway.
To test the potential role of CAR in bilirubin clearance, we examined the response of the genes encoding the known components of the clearance pathway diagrammed in Fig. 1A to the potent CAR agonist 1,4-bis-[2-(3,5-dichloropyridyloxy)]benzene (TCPOBOP) in wild-type and CAR null animals. CAR is sequestered in the cytoplasm of hepatocytes under normal conditions (15), and loss of CAR function did not affect basal expression. However, TCPOBOP induced expression of each of five known components of the bilirubin-clearance pathway in the livers of wild-type but not CAR-deficient mice (Fig. 1B). The induction of UGT1A1, GSTA1, and MRP2 by TCPOBOP is generally consistent with both recent reports identifying them as primary CAR targets (16–18) and the accompanying paper by Xie et al. (19). The 2- to 3-fold response of SLC21A6 to TCPOBOP observed in several experiments contrasts with a report that SLC21A6 expression is not altered in PB-treated rats (20). In additional studies, PB treatment resulted in a limited response of this gene in some but not all mice examined (data not shown). We conclude that CAR can activate expression of the genes known to be involved in the bilirubin-clearance pathway.
To examine the role of CAR in the reported ability of bilirubin to induce its own glucuronidation (9, 10), we exposed mouse primary hepatocytes from either wild-type or CAR knockout animals to bilirubin for different times. SLC21A6, UGT1A1, and MRP2 mRNAs were clearly increased by bilirubin treatment in the wild-type but not CAR knockout cells, indicating that CAR is required for this response (Fig. 1C). Markedly elevated basal level of GSTA1 and GSTA2 expression in the primary hepatocytes relative to livers may have masked their responses to bilirubin.
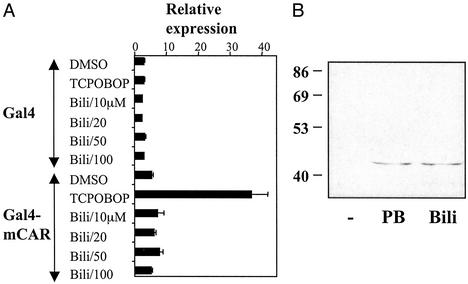
In contrast to these effects, transient transfection experiments using either full-length CAR or a fusion protein containing the CAR ligand-binding domain and the Gal4 DNA-binding domain showed no responses to bilirubin, biliverdin, or related compounds (Fig. 2A and data not shown). Similar results were obtained with Gal4 fusions to the ligand-binding domains of PXR and a number of other nuclear hormone receptors (data not shown), suggesting that bilirubin is not an agonist ligand for CAR or any of these other receptors. This lack of response is not due to failure of these compounds to get into the cells, because similar approaches have shown that bilirubin can directly activate the aryl hydrocarbon receptor (21, 22).
Figure 2.
Bilirubin activates CAR indirectly. (A) HepG2 cells were cotransfected with expression vectors for a fusion of the Gal4 DNA-binding domain to the CAR ligand-binding domain or Gal4 alone, the pG5E1b-Luc reporter, and pSV2-β-galactosidase. After 12 h, increasing concentrations of bilirubin (Bili) solution were added as indicated. Cells were cultured for an additional 24 h, and luciferase activity was measured and normalized by using the level of β-galactosidase activity. (B) Mouse primary hepatocytes were cultured in a William's E defined medium for 24 h. PB (1 mM), bilirubin (100 μM, Bili), or vehicle (−) was added, and the cells were cultured for an additional 3 h. Equal amounts of nuclear extracts were fractionated by SDS/PAGE, and CAR levels in the nucleus were assayed by Western blotting with a previously described (15) CAR antibody.
Similar to bilirubin, PB also shows no evidence of direct binding to CAR (14, 23). Instead, it is thought that PB causes translocation of CAR from the hepatocyte cytoplasm to the nucleus (15, 24), where its constitutive activity is responsible for the response of appropriate target genes. Thus, we tested the ability of bilirubin to induce nuclear translocation of CAR in primary hepatocytes. As shown in Fig. 2B, treatment with PB or bilirubin resulted in comparable levels of CAR accumulation in the nucleus. We conclude that bilirubin is an endogenous activator of CAR, although apparently not a direct agonist ligand, and can induce the expression of essential genes involved in its own clearance.
Defects in Acute and Chronic Bilirubin Clearance in CAR Null Mice.
To test the functional role of CAR in bilirubin metabolism, we injected wild-type or CAR null mice pretreated with PB, TCPOBOP, or vehicle for 3 days with a single bolus of bilirubin. Mice have a high capacity to eliminate circulating bilirubin, and ≈90% of the bilirubin dose was cleared in the control-treated wild-type animals in 1 h. However, even this high rate of clearance was increased markedly by pretreatment with PB or TCPOBOP, resulting in an ≈3- to 5-fold lower level of residual total serum bilirubin (Fig. 3A). As expected from the gene-expression results, the inducers had no effect on clearance of this acute bilirubin dose in the knockout animals.
Figure 3.
CAR activation increases bilirubin clearance. (A) A single dose of bilirubin (10 mg/kg) was injected into the tail veins of wild-type (+/+) or CAR knockout (−/−) animals (n = 3) pretreated with PB, TCPOBOP (TC), or corn oil (CO) control for 3 days. Total serum bilirubin was measured at 60 min after the injection. At this time, ≈90% of the bilirubin measured 5 min after injection had been cleared in the untreated wild-type animals, but the remaining levels were significantly above those in animals not injected with bilirubin. Measurement of direct bilirubin showed that the majority (60–70%) of this serum bilirubin is conjugated. (B) Total serum bilirubin was measured after incorporation of phenylhydrazine (0.1 mg/ml, PHZ) (+) into the drinking water of wild-type (+/+) and CAR knockout (−/−) animals (n = 5) for 30 days. Controls (−) received the normal drinking water. Measurement of direct bilirubin showed very similar results, indicating that essentially all of this circulating bilirubin is conjugated. (C) Total liver RNA from the same mice was analyzed by Northern blotting using the indicated probes.
To study the effect of loss of CAR on chronically elevated bilirubin levels, we induced hemolytic anemia, one of the major causes of hyperbilirubinemia in humans, by adding phenylhydrazine (0.1 mg/ml) to the drinking water of wild-type or CAR null mice. Phenylhydrazine increases the rate of breakdown of erythrocytes and increases the bilirubin level in plasma (25). As predicted, this treatment increased levels of serum bilirubin in the wild-type mice, and these levels were strikingly elevated in the CAR knockout mice (Fig. 3B). Serial tail-vein injections of bilirubin into individual mice also resulted in higher plasma bilirubin accumulation in the CAR-deficient mice (data not shown), indicating that the observed differences between the wild-type and knockout animals are due to changes in bilirubin levels, and not secondary effects of phenylhydrazine or anemia.
To explore the basis for this defective response to a chronic bilirubin challenge, the expression of the components of the bilirubin-clearance pathway was examined. As expected, UGT1A1 expression was increased in wild-type but not CAR knockout mice treated with phenylhydrazine (Fig. 3C). GSTA1 was also induced in these animals, and expression of SLC21A6 and MRP2 was slightly increased (data not shown). Together, these results demonstrate that activation of CAR increases the rate of bilirubin clearance and that loss of CAR function results in a defect in clearance of chronically elevated bilirubin.
hCAR Can Activate Bilirubin Clearance.
The differences in response of the xenobiotic receptors in different species (8, 14, 23) raise the important question of whether the effects observed with mCAR can also be observed with hCAR. To test this, a line of transgenic mice expressing only hCAR in the liver was generated by using a construct based on the liver-specific albumin promoter (Fig. 4A; ref. 12). hCAR-expressing animals were bred with the CAR null mice to produce a humanized line expressing only hCAR in the liver. These mice are analogous to a previously described line expressing human instead of mouse PXR (11). Treatment of the CAR-humanized animals with the general CAR activator PB increased clearance of bilirubin as expected (Fig. 4B). As in the wild-type mice, PB treatment of these humanized mice also induced expression of UGT1A1, MRP2, SLC21A6, and GSTA1 (data not shown). In addition, treatment of primary hepatocytes from the humanized livers with bilirubin induced expression of UGT1A1 (Fig. 4C). These results demonstrate that the responses observed with mouse CAR can also be observed with hCAR.
Figure 4.
Function of hCAR in bilirubin clearance. (A) The indicated construct was used to generate transgenic mice by standard approaches. CAR-humanized mice were generated by crossing the transgenic line with the CAR null background, resulting in animals expressing hCAR but not mCAR in the liver. (B) A single dose of bilirubin (10 mg/kg) was injected into the tail veins of humanized mice (n = 3) pretreated with corn oil (CO) or PB (100 mg/kg) for 3 days. Total serum bilirubin was measured 60 min after the injection. PB treatment significantly lowered bilirubin levels (P < 0.01). (C) Mouse primary hepatocytes from CAR knockout (KO) or CAR-humanized (hCAR) mice were cultured in the presence of bilirubin (10 μM) for 24 h. Total RNA was prepared and analyzed by Northern blotting with the indicated probes.
Decreased CAR Expression in Neonates.
Because bilirubin metabolism is particularly important in the neonatal period, the expression of CAR mRNA was examined in neonatal mouse livers (Fig. 5A). In contrast to results with PXR (26) but in agreement with results with CAR in the fetal liver (27), the amount of CAR mRNA was much lower in neonatal livers than adult livers.
Figure 5.
Decreased CAR expression in neonatal mice and humans. (A) Livers were collected from neonatal mice of the indicated ages. Total liver RNA was prepared, and equal amounts of RNA from three animals were pooled and analyzed by Northern blotting with CAR or PXR probes as indicated. (B and C) RNA and protein extracts were prepared from liver samples from three human neonates and three adults. (B) Equal amounts of RNA from each were pooled, and hCAR mRNA expression was analyzed by Northern blotting. Equivalent amounts of protein were pooled, and 50 μg of the neonatal and adult samples were separated by SDS/PAGE. (C) Relative expression of CAR was assayed by using an antibody directed against hCAR antibody. The blot was stripped and reanalyzed with an antiactin monoclonal antibody (1:1,000) (Santa Cruz Biotechnology).
To determine whether this decreased CAR expression is also observed in humans, we examined CAR mRNA and protein levels in three neonatal livers and three adult livers. CAR expression is markedly lower but not absent in the neonatal samples (Fig. 5 B and C). These initial studies are clearly consistent with the expression of mouse CAR, although it remains possible that CAR expression could have been increased in some adult individuals by activation of PXR (18) or the glucocorticoid receptor (28).
Discussion
The results described here demonstrate that CAR responds to elevated bilirubin levels by increasing the rate of bilirubin clearance. This role of CAR is consistent with the well documented ability of PB to lower bilirubin levels in humans (4, 29, 30), recent results indicating that UGT1A1, MRP2, and GSTA1 are primary CAR targets (16–18), and the accompanying article by Xie et al. (19) demonstrating broader regulatory effects of CAR and PXR on expression of UGT isoforms.
The primary function of CAR in this pathway is to direct a coordinate response to elevated levels of bilirubin. Under normal conditions, CAR is sequestered in the cytoplasm and does not contribute to basal levels of bilirubin clearance. Thus, defects in bilirubin clearance are not observed in the CAR null mice until the appropriate stress is applied. This specific response of CAR to the stimulus of bilirubin excess is very similar to those observed with other nuclear receptors, for example the protective responses of PXR to elevated levels of toxic bile acids (31, 32), or the liver X receptors to elevated levels of cholesterol (33). However, it differs in that bilirubin is apparently not a direct agonist ligand for CAR. It is likely that the translocation induced by bilirubin is similar to that induced by PB (15, 24), but the molecular basis for this process remains unclear.
Approximately 60% of infants are affected by icterus neonatorum or neonatal jaundice (34, 35). This complex condition has many potential risk factors including hemolytic conditions, familial hyperbilirubinemia, decreased gestational age, and breastfeeding. In physiologic jaundice, serum bilirubin levels are elevated for up to 1 week after birth but decline to normal adult levels over the next few weeks. Hyperbilirubinemia is considered pathologic when serum levels exceed 17 mg/dl (35). In such cases, bilirubin can be deposited in the CNS, resulting in neurotoxicity and encephalopathy. Pathologic hyperbilirubinemia can be treated effectively by phototherapy or by exchange transfusion in severe cases. Until recently, these treatments had essentially eliminated kernicterus as a health problem in developed countries. However, the combination of the relaxation of the guidelines for treatment of hyperbilirubinemia (34), an increase in the rate of breastfeeding, and decreases in the hospital stay and therefore monitoring of neonates has resulted in an increase in the incidence of this serious condition (36–38). With recent results suggesting that the neurologic impact of relatively moderate hyperbilirubinemia may be more significant than thought previously (39), the management of neonatal jaundice has become a controversial area with significant public-health concerns (40).
Physiologic neonatal jaundice is associated with both an increased rate of erythrocyte turnover (41) and low levels of bilirubin UDP glucuronosyltransferase activity (42–44). The results described here suggest that decreased expression of CAR may account for the apparent failure of expression of UGT1A1 and other components of the pathway to increase in response to the increased demand. Thus, we propose that a functional deficit of CAR activity in neonates may contribute to the etiology of neonatal jaundice. Although it could clearly have pathologic consequences in some cases, it is an interesting possibility that the decreased CAR expression allows potentially beneficial effects of transient elevation of bilirubin, a potent antioxidant, in the neonate.
A significant question raised by the decreased neonatal CAR expression is the basis for the reported clinical effectiveness of PB treatment for neonatal jaundice (4, 29, 30). There are several possible explanations, the simplest of which is that the residual CAR expression is sufficient to confer sufficient response to this potent activator. In humans, PB can also function as an agonist ligand for PXR. Such PXR activation could contribute to the effects of PB both directly via induction of UGT1A1 and other components of the clearance pathway and indirectly by increasing expression of CAR (18, 19).
Because of its sedating effects, PB is not a desirable therapeutic approach for neonatal or other forms of jaundice. Although TCPOBOP is a potent and specific agonist of mCAR (23), analogous high-affinity ligands remain to be identified for hCAR. It is an exciting possibility that such potent activators of hCAR could have direct therapeutic applications in neonatal, acquired, and genetic forms of jaundice.
Acknowledgments
We thank Drs. Masahiko Negishi for kindly providing the mouse CAR antibody and Dennis H. Dowhan, Ping Wei, and Saul Karpen for advice. This work was supported by National Institutes of Health Grant R01 DK46546.
Abbreviations
- UGT1A1
UDP-glucuronosyltransferase
- PB
phenobarbital
- CAR
constitutive androstane receptor
- hCAR
human CAR
- mCAR
murine CAR
- PXR
pregnane X receptor
- TCPOBOP
1,4-bis-[2-(3,5-dichloropyridyloxy)]benzene
Note Added in Proof.
J. M. Maglich et al. (45) have recently identified a specific human CAR agonist.
References
- 1.Jansen P L, Bosma P J, Chowdhury J R. Prog Liver Dis. 1995;13:125–150. [PubMed] [Google Scholar]
- 2.Kamisako T, Kobayashi Y, Takeuchi K, Ishihara T, Higuchi K, Tanaka Y, Gabazza E C, Adachi Y. J Gastroenterol. 2000;35:659–664. doi: 10.1007/s005350070044. [DOI] [PubMed] [Google Scholar]
- 3.Cui Y, Konig J, Leier I, Buchholz U, Keppler D. J Biol Chem. 2001;276:9626–9630. doi: 10.1074/jbc.M004968200. [DOI] [PubMed] [Google Scholar]
- 4.Jansen P L. Eur J Pediatr. 1999;158, Suppl. 2:S89–S94. doi: 10.1007/pl00014330. [DOI] [PubMed] [Google Scholar]
- 5.Kadakol A, Ghosh S S, Sappal B S, Sharma G, Chowdhury J R, Chowdhury N R. Hum Mutat. 2000;16:297–306. doi: 10.1002/1098-1004(200010)16:4<297::AID-HUMU2>3.0.CO;2-Z. [DOI] [PubMed] [Google Scholar]
- 6.Catz C, Yaffe S J. Am J Dis Child. 1962;104:516–517. [Google Scholar]
- 7.Yaffe S J, Levy G, Matsuzawa T, Baliah T. N Engl J Med. 1966;275:1461–1466. doi: 10.1056/NEJM196612292752602. [DOI] [PubMed] [Google Scholar]
- 8.Wei P, Zhang J, Egan-Hafley M, Liang S, Moore D D. Nature. 2000;407:920–923. doi: 10.1038/35038112. [DOI] [PubMed] [Google Scholar]
- 9.Munoz M E, Esteller A, Gonzalez J. Clin Sci (London) 1987;73:371–375. doi: 10.1042/cs0730371. [DOI] [PubMed] [Google Scholar]
- 10.Li Y Q, Prentice D A, Howard M L, Mashford M L, Desmond P V. J Gastroenterol Hepatol. 2000;15:865–870. doi: 10.1046/j.1440-1746.2000.02223.x. [DOI] [PubMed] [Google Scholar]
- 11.Xie W, Barwick J L, Downes M, Blumberg B, Simon C M, Nelson M C, Neuschwander-Tetri B A, Brunt E M, Guzelian P S, Evans R M. Nature. 2000;406:435–439. doi: 10.1038/35019116. [DOI] [PubMed] [Google Scholar]
- 12.Zhang J, Huang W, Chua S S, Wei P, Moore D D. Science. 2002;298:422–424. doi: 10.1126/science.1073502. [DOI] [PubMed] [Google Scholar]
- 13.Granata R, Broglio F, Migliorino D, Cutrupi S, Baldanzi G, Sireno M, Fubini A, Grazian A, Ghigo E, Pucci A. J Endocrinol Invest. 2000;23:724–726. doi: 10.1007/BF03345060. [DOI] [PubMed] [Google Scholar]
- 14.Moore L B, Parks D J, Jones S A, Bledsoe R K, Consler T G, Stimmel J B, Goodwin B, Liddle C, Blanchard S G, Willson T M, et al. J Biol Chem. 2000;275:15122–15127. doi: 10.1074/jbc.M001215200. [DOI] [PubMed] [Google Scholar]
- 15.Kawamoto T, Sueyoshi T, Zelko I, Moore R, Washburn K, Negishi M. Mol Cell Biol. 1999;19:6318–6322. doi: 10.1128/mcb.19.9.6318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sugatani J, Kojima H, Ueda A, Kakizaki S, Yoshinari K, Gong Q H, Owens I S, Negishi M, Sueyoshi T. Hepatology. 2001;33:1232–1238. doi: 10.1053/jhep.2001.24172. [DOI] [PubMed] [Google Scholar]
- 17.Kast H R, Goodwin B, Tarr P T, Jones S A, Anisfeld A M, Stoltz C M, Tontonoz P, Kliewer S, Willson T M, Edwards P A. J Biol Chem. 2002;277:2908–2915. doi: 10.1074/jbc.M109326200. [DOI] [PubMed] [Google Scholar]
- 18.Maglich J M, Stoltz C M, Goodwin B, Hawkins-Brown D, Moore J T, Kliewer S A. Mol Pharmacol. 2002;62:638–646. doi: 10.1124/mol.62.3.638. [DOI] [PubMed] [Google Scholar]
- 19.Xie W, Yeuh M-F, Radominska-Pandya A, Saini S P S, Negishi Y, Bottroff B S, Cabrera G Y, Tukey R H, Evans R M. Proc Natl Acad Sci USA. 2003;100:4150–4155. doi: 10.1073/pnas.0438010100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hagenbuch N, Reichel C, Stieger B, Cattori V, Fattinger K E, Landmann L, Meier P J, Kullak-Ublick G A. J Hepatol. 2001;34:881–887. doi: 10.1016/s0168-8278(01)00097-6. [DOI] [PubMed] [Google Scholar]
- 21.Sinal C J, Bend J R. Mol Pharmacol. 1997;52:590–599. doi: 10.1124/mol.52.4.590. [DOI] [PubMed] [Google Scholar]
- 22.Phelan D, Winter G M, Rogers W J, Lam J C, Denison M S. Arch Biochem Biophys. 1998;357:155–163. doi: 10.1006/abbi.1998.0814. [DOI] [PubMed] [Google Scholar]
- 23.Tzameli I, Pissios P, Schuetz E G, Moore D D. Mol Cell Biol. 2000;20:2951–2958. doi: 10.1128/mcb.20.9.2951-2958.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zelko I, Sueyoshi T, Kawamoto T, Moore R, Negishi M. Mol Cell Biol. 2001;21:2838–2846. doi: 10.1128/MCB.21.8.2838-2846.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bauer A, Tronche F, Wessely O, Kellendonk C, Reichardt H M, Steinlein P, Schutz G, Beug H. Genes Dev. 1999;13:2996–3002. doi: 10.1101/gad.13.22.2996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Masuyama H, Hiramatsu Y, Mizutani Y, Inoshita H, Kudo T. Mol Cell Endocrinol. 2001;172:47–56. doi: 10.1016/s0303-7207(00)00395-6. [DOI] [PubMed] [Google Scholar]
- 27.Wei P, Zhang J, Dohan D, Han Y, Moore D D. Pharmacogenomics J. 2002;2:117–126. doi: 10.1038/sj.tpj.6500087. [DOI] [PubMed] [Google Scholar]
- 28.Pascussi J M, Busson-Le Coniat M, Maurel P, Vilarem M J. Mol Endocrinol. 2003;17:42–55. doi: 10.1210/me.2002-0244. [DOI] [PubMed] [Google Scholar]
- 29.Valaes T, Kipouros K, Petmezaki S, Solman M, Doxiadis S A. Pediatr Res. 1980;14:947–952. doi: 10.1203/00006450-198008000-00011. [DOI] [PubMed] [Google Scholar]
- 30.Wallin A, Boreus L O. Acta Paediatr Scand. 1984;73:488–497. doi: 10.1111/j.1651-2227.1984.tb09960.x. [DOI] [PubMed] [Google Scholar]
- 31.Xie W, Radominska-Pandya A, Shi Y, Simon C M, Nelson M C, Ong E S, Waxman D J, Evans R M. Proc Natl Acad Sci USA. 2001;98:3375–3380. doi: 10.1073/pnas.051014398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Staudinger J L, Goodwin B, Jones S A, Hawkins-Brown D, MacKenzie K I, LaTour A, Liu Y, Klaassen C D, Brown K K, Reinhard J, et al. Proc Natl Acad Sci USA. 2001;98:3369–3374. doi: 10.1073/pnas.051551698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Peet D J, Turley S D, Ma W, Janowski B A, Lobaccaro J M, Hammer R E, Mangelsdorf D J. Cell. 1998;93:693–704. doi: 10.1016/s0092-8674(00)81432-4. [DOI] [PubMed] [Google Scholar]
- 34.Provisional Committee for Quality Improvement and Subcommittee on Hyperbilirubinemia. Pediatrics. 1994;94:558–565. [PubMed] [Google Scholar]
- 35.Dennery P A, Seidman D S, Stevenson D K. N Engl J Med. 2001;344:581–590. doi: 10.1056/NEJM200102223440807. [DOI] [PubMed] [Google Scholar]
- 36.Ebbesen F. Acta Paediatr. 2000;89:1213–1217. doi: 10.1080/080352500750027592. [DOI] [PubMed] [Google Scholar]
- 37.Harris M C, Bernbaum J C, Polin J R, Zimmerman R, Polin R A. Pediatrics. 2001;107:1075–1080. doi: 10.1542/peds.107.5.1075. [DOI] [PubMed] [Google Scholar]
- 38.Carter K, Dixon K. Morbid Mortal Wkly Rep. 2001;50:491–494. [Google Scholar]
- 39.Soorani-Lunsing I, Woltil H A, Hadders-Algra M. Pediatr Res. 2001;50:701–705. doi: 10.1203/00006450-200112000-00012. [DOI] [PubMed] [Google Scholar]
- 40.AAP Subcommittee on Neonatal Hyperbilirubinemia. Pediatrics. 2001;108:763–765. doi: 10.1542/peds.108.3.763. [DOI] [PubMed] [Google Scholar]
- 41.Brouillard R P. J Am Med Assoc. 1974;230:1304–1305. [PubMed] [Google Scholar]
- 42.Leakey J E, Hume R, Burchell B. Biochem J. 1987;243:859–861. doi: 10.1042/bj2430859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Burchell B, Coughtrie M, Jackson M, Harding D, Fournel-Gigleux S, Leakey J, Hume R. Dev Pharmacol Ther. 1989;13:70–77. doi: 10.1159/000457587. [DOI] [PubMed] [Google Scholar]
- 44.Strassburg C P, Strassburg A, Kneip S, Barut A, Tukey R H, Rodeck B, Manns M P. Gut. 2002;50:259–265. doi: 10.1136/gut.50.2.259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Maglich, J. M., Parks, D. J., Moore, L. B., Collins, J. L., Goodwin, B., Billin, A. N., Stoltz, C. A., Kliewer, S. A., Lambert, M. H., Willson, T. M. & Moore, J. T. J. Biol. Chem., 10.1074/jbc.M300138200.