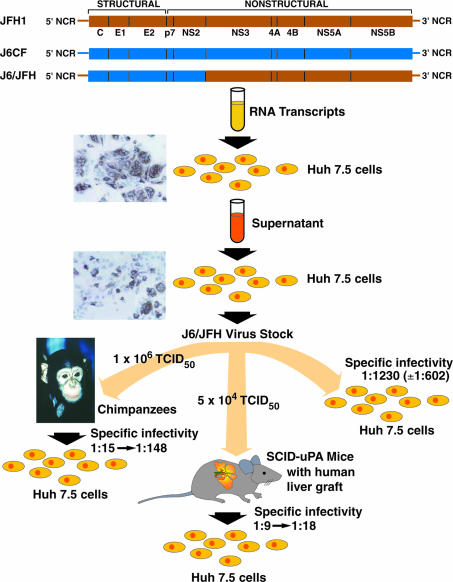
Chronic hepatitis C virus (HCV) infection is a leading cause of end-stage liver disease in the United States, and the Centers for Disease Control has predicted that ≈10,000 Americans die annually from this disease. Since the discovery of HCV in 1989, basic research on this important pathogen has been difficult because of the lack of a robust cell-culture system. Thus, the recent development of a cell culture resulting in production of significant levels of infectious virus particles (1–3) will without doubt lead to rapid progress in our understanding of the HCV viral life cycle and, it is hoped, development of better therapies. The development of molecular clones of strain H77 (genotype 1a) infectious for chimpanzees in 1997 was an important initial advance, but these clones and subsequent infectious clones of other genotype strains did not produce viruses in cell culture (4–7). Another critical advance was the development of subgenomic replicons in 1999 that permitted studies of RNA replication (8). Efficient replication, however, depended on adaptive mutations (9, 10). In 2003, Kato et al. (11) observed that the WT JFH1 strain of HCV, belonging to genotype 2a, replicated to high levels in the replicon system. Subsequently, in 2005, Wakita and coworkers (2) demonstrated that RNA transcripts from the full-length JFH1 genome could produce viruses in a human liver hepatoma cell line (Huh-7 cells), and it was demonstrated by Chisari and coworkers (3) that this system could be developed to generate relatively high titers of HCV. In a different approach, Rice and coworkers (1) developed a chimeric genome in which the structural genes (C, E1, and E2), p7, and NS2 from an infectious clone of another genotype 2a strain (strain HC-J6) (6) were inserted into the subgenomic replicon of the JFH1 strain and demonstrated that RNA transcripts from this full-length chimeric genome could produce relatively high titers of HCV. In the current issue of PNAS, a study by Lindenbach et al. (12) demonstrated that chimeric J6/JFH1 viruses generated in vitro were fully viable also in vivo, as tested in chimpanzees and in SCID-uPA mice with human liver grafts, and that virus recovered from infected animals efficiently infected naïve Huh-7 cells. An overview of this important study is shown in Fig. 1. Therefore, it appears that not only has a cell-culture system been developed for HCV, but this system produces viruses that are biologically relevant.
Fig. 1.
Generation of J6/JFH1 viruses in cell culture and demonstration of their infectivity in vivo. Diagram showing study design and results in Lindenbach et al. (12). RNA transcripts from the chimeric genome were transfected into Huh-7.5 cells, and filtered supernatant was used to infect naïve Huh-7.5 cells. The immunostaining shown was performed with a mouse monoclonal NS5A antibody (1). Titrated virus from a virus stock was used to inoculate two chimpanzees i.v. and two SCID-uPA mice with a human liver graft i.p. Transfer of plasma from one of these viremic mice resulted in HCV infection in a third mouse. The specific infectivity (ratio of infectivity titer to RNA titer) was determined in naïve Huh-7 cells for chimpanzee and mouse serum containing the J6/JFH1 chimeric virus. TCID50, tissue culture 50% infective dose. Figure adapted, in part, from Lindenbach et al. (1).
The chimpanzee model is the only animal model available for HCV that permits studies of infectivity and pathogenesis (reviewed in ref. 13). The course of infection observed with the J6/JFH1 viruses derived from cell culture was similar to experimental acute infections with other HCV strains, with early appearance of viremia, peak viral titers of 104 to 105 units/ml, seroconversion, and induction of intrahepatic innate and adaptive immune responses. Neither of the two chimpanzees developed clear evidence of acute viral hepatitis, but that has also been observed in animals infected with WT virus. In 2001, Mercer et al. (14) introduced a mouse model that could be used to test the infectivity of HCV. In this SCID-uPA mouse, degenerated mouse hepatocytes are replaced by primary human hepatocytes (14, 15). It is a difficult model, in part because of high mortality and the need for fresh primary hepatocytes for engraftment (reviewed in ref. 16). However, if the mice have a successful graft as measured by a human albumin level of >1 mg/ml, they appear to be readily susceptible to HCV. Again in this animal model, the J6/JFH1 viruses behaved as WT virus and reached titers of 106 to 107 units/ml. Overall, therefore, the conclusion that these cell-culture-derived viruses are fully viable in vivo is justified. It will be important to confirm by sequence analysis that the virus recovered from the animals did not acquire adaptive mutations but the kinetics observed in the animals, and the fact that these viruses could readily infect naïve Huh-7 cells would suggest that adaptation did not play a major role.
As mentioned, efficient replication in Huh-7 cells of replicons of most HCV strains depends on adaptive mutations located at various sites in the nonstructural proteins (reviewed in ref. 17). It was previously demonstrated that such mutations severely attenuate viability of full-length genomes in chimpanzees (18) and also interfere with virus production in vitro (19). The exception, however, was the JFH1 strain, where the WT sequence apparently replicated to high levels in the replicon system. Two observations, however, suggest that the JFH1 full-length genome might be attenuated in vitro and in vivo. First, the JFH1 genome apparently has a delayed kinetics of virus release (2, 3, 12, 19), and it will be important to determine whether the JFH1 virus that eventually spreads efficiently in cell culture has acquired adaptive mutations. Second, the JFH1 virus collected from transfected Huh-7 cells resulted in an attenuated infection in a chimpanzee with low viremia titers, early viral clearance, and no seroconversion (2). The JFH1 and J6/JFH1 genomes have identical untranslated regions and NS3 through NS5B genes. The C, E1, E2, p7, and NS2 genes in the J6/JFH1 genome originate from a clone that was previously shown to be infectious in vivo (6). Thus the fact that the J6/JFH1 genome appears to be fully viable would suggest that the JFH1 full-length sequence might have sequences in the C through NS2 genes that are attenuating in vitro and in vivo. The fact that a chimera consisting of two different strains of the same genotype is highly replication-competent in vivo is somewhat surprising, in particular because the chimeric NS2–NS3 junction contains the NS2–NS3 autoprotease. Further studies are needed to determine whether the chimeric J6/JFH1 virus eventually acquires mutations that might further optimize replication of this cell-culture-derived virus.
A very intriguing observation was made by Lindenbach et al. (12) on the relative specific infectivity, as determined by comparing the infectivity titer in Huh-7 cells with the HCV genome titers of cell-cultured viruses from Huh-7 cells and those recovered from chimpanzees and SCID-uPA mice grafted with human hepatocytes (Fig. 1). The J6/JFH1 viruses recovered from animals had a specific infectivity that was 10- to 100-fold greater than viruses recovered from infected Huh-7 cells. This higher infectivity of animal-derived viruses was lost after reculture in Huh-7 cells. The difference in specific infectivity of these viruses was reflected by a difference in their buoyant density, with the animal-derived viruses having a lower density. This finding raises the possibility that specific proteins released by human or chimpanzee hepatocytes enhance infectivity of HCV. This phenomenon has recently been observed in the HCV pseudovirus system, in which the entry of retroviral pseudovirus particles bearing the envelope proteins of HCV was found to be enhanced by serum from chimpanzees and humans, human lipoproteins, and human apolipoproteins (20–23). Thus, it is a possibility that such lipoproteins and apolipoproteins might also enhance the infectivity of HCV in cell culture.
The development of the J6/JFH1 culture system by Lindenbach et al. (1) that produces viruses that are fully viable in vivo (12) is a major accomplishment. This culture system is still limited to this particular genotype 2a strain, and even the full-length HC-J6 genome does not replicate in Huh-7 cells. Thus, it will be important to develop this system for HCV strains representing all six major genotypes of HCV. Such diversity will probably be essential in the testing of new virus-specific antivirals, such as protease and polymerase inhibitors, which most likely will have different efficacies against different viral sequences. Alternatively, other JFH1 chimeric viruses that contain the C through NS2 sequence from other strains could be developed and would be sufficient for studying virus neutralization and specific mechanisms of how HCV enters the host cell. Another limitation of the current cell-culture system is its dependence on a single cell line, which is a hepatoma cell line known to have numerous genetic defects compared with hepatocytes. Such defects could lead, in particular, to data on virus–host interactions that are not biologically relevant. Finally, to consider this cell-culture system for the development of an inactivated whole virus vaccine, it would be critical to identify cell lines, approved for vaccine production, in which HCV can grow to even higher titers than is currently observed in Huh-7 cells. Thus, although the HCV research field has reached an important milestone with the development of a true cell-culture system that permits classical virological studies, new developments are required to expand research beyond one virus and one cell.
Conflict of interest statement: No conflicts declared.
See companion article on page 3805 .
References
- 1.Lindenbach B. D., Evans M. J., Syder A. J., Wolk B., Tellinghuisen T. L., Liu C. C., Maruyama T., Hynes R. O., Burton D. R., McKeating J. A., Rice C. M. Science. 2005;309:623–626. doi: 10.1126/science.1114016. [DOI] [PubMed] [Google Scholar]
- 2.Wakita T., Pietschmann T., Kato T., Date T., Miyamoto M., Zhao Z., Murthy K., Habermann A., Krausslich H. G., Mizokami M., et al. Nat. Med. 2005;11:791–796. doi: 10.1038/nm1268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhong J., Gastaminza P., Cheng G., Kapadia S., Kato T., Burton D. R., Wieland S. F., Uprichard S. L., Wakita T., Chisari F. V. Proc. Natl. Acad. Sci. USA. 2005;102:9294–9299. doi: 10.1073/pnas.0503596102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kolykhalov A. A., Agapov E. V., Blight K. J., Mihalik K., Feinstone S. M., Rice C. M. Science. 1997;277:570–574. doi: 10.1126/science.277.5325.570. [DOI] [PubMed] [Google Scholar]
- 5.Yanagi M., Purcell R. H., Emerson S. U., Bukh J. Proc. Natl. Acad. Sci. USA. 1997;94:8738–8743. doi: 10.1073/pnas.94.16.8738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yanagi M., Purcell R. H., Emerson S. U., Bukh J. Virology. 1999;262:250–263. doi: 10.1006/viro.1999.9889. [DOI] [PubMed] [Google Scholar]
- 7.Yanagi M., St. Claire M., Shapiro M., Emerson S. U., Purcell R. H., Bukh J. Virology. 1998;244:161–172. doi: 10.1006/viro.1998.9092. [DOI] [PubMed] [Google Scholar]
- 8.Lohmann V., Korner F., Koch J., Herian U., Theilmann L., Bartenschlager R. Science. 1999;285:110–113. doi: 10.1126/science.285.5424.110. [DOI] [PubMed] [Google Scholar]
- 9.Blight K. J., Kolykhalov A. A., Rice C. M. Science. 2000;290:1972–1974. doi: 10.1126/science.290.5498.1972. [DOI] [PubMed] [Google Scholar]
- 10.Lohmann V., Korner F., Dobierzewska A., Bartenschlager R. J. Virol. 2001;75:1437–1449. doi: 10.1128/JVI.75.3.1437-1449.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kato T., Date T., Miyamoto M., Furusaka A., Tokushige K., Mizokami M., Wakita T. Gastroenterology. 2003;125:1808–1817. doi: 10.1053/j.gastro.2003.09.023. [DOI] [PubMed] [Google Scholar]
- 12.Lindenbach B. D., Meuleman P., Ploss A., Vanwolleghem T., Syder A. J., McKeating J. A., Lanford R. E., Feinstone S. M., Major M. E., Leroux-Roels G., et al. Proc. Natl. Acad. Sci. USA. 2006;103:3805–3809. doi: 10.1073/pnas.0511218103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bukh J. Hepatology. 2004;39:1469–1475. doi: 10.1002/hep.20268. [DOI] [PubMed] [Google Scholar]
- 14.Mercer D. F., Schiller D. E., Elliott J. F., Douglas D. N., Hao C., Rinfret A., Addison W. R., Fischer K. P., Churchill T. A., Lakey J. R., et al. Nat. Med. 2001;7:927–933. doi: 10.1038/90968. [DOI] [PubMed] [Google Scholar]
- 15.Meuleman P., Libbrecht L., De Vos R., de Hemptinne B., Gevaert K., Vandekerckhove J., Roskams T., Leroux-Roels G. Hepatology. 2005;41:847–856. doi: 10.1002/hep.20657. [DOI] [PubMed] [Google Scholar]
- 16.Kneteman N. M., Mercer D. F. Hepatology. 2005;41:703–706. doi: 10.1002/hep.20681. [DOI] [PubMed] [Google Scholar]
- 17.Bartenschlager R., Lohmann V. Antiviral Res. 2001;52:1–17. doi: 10.1016/s0166-3542(01)00164-4. [DOI] [PubMed] [Google Scholar]
- 18.Bukh J., Pietschmann T., Lohmann V., Krieger N., Faulk K., Engle R. E., Govindarajan S., Shapiro M., St. Claire M., Bartenschlager R. Proc. Natl. Acad. Sci. USA. 2002;99:14416–14421. doi: 10.1073/pnas.212532699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bartenschlager R., Pietschmann T. Proc. Natl. Acad. Sci. USA. 2005;102:9739–9740. doi: 10.1073/pnas.0504296102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bartosch B., Verney G., Dreux M., Donot P., Morice Y., Penin F., Pawlotsky J. M., Lavillette D., Cosset F. L. J. Virol. 2005;79:8217–8229. doi: 10.1128/JVI.79.13.8217-8229.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Meunier J.-C., Engle R. E., Faulk K., Zhao M., Bartosch B., Alter H., Emerson S. U., Cosset F.-L., Purcell R. H., Bukh J. Proc. Natl. Acad. Sci. USA. 2005;102:4560–4565. doi: 10.1073/pnas.0501275102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Voisset C., Callens N., Blanchard E., Op De Beeck A., Dubuisson J., Vu-Dac N. J. Biol. Chem. 2005;280:7793–7799. doi: 10.1074/jbc.M411600200. [DOI] [PubMed] [Google Scholar]
- 23.Lavillette D., Morice Y., Germanidis G., Donot P., Soulier A., Pagkalos E., Sakellariou G., Intrator L., Bartosch B., Pawlotsky J. M., Cosset F. L. J. Virol. 2005;79:6023–6034. doi: 10.1128/JVI.79.10.6023-6034.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]