Abstract
Statins inhibit 3-hydroxy-3-methylglutaryl (HMG-CoA) reductase, the rate limiting step in cholesterol synthesis. They are, therefore, used clinically to lower cholesterol and prevent atherosclerosis. Statins have beneficial effects on multiple organ systems. Some of these effects are found in the absence of significant changes in cholesterol levels. Polyunsaturated fatty acids also inhibit HMG-CoA reductase and have many of the same beneficial effects of statins. Four statins (mevastatin, lovastatin, simvastatin and atorvastatin) have been tested in rat liver cells for their effect on arachidonic acid (AA) release and prostaglandin I2 production induced in the presence of lactacystin and 12-O-tetradecanoylphorbol-13-acetate. Each statin stimulated release of AA and induced prostaglandin I2 production. Mevalonate, the product of HMG-CoA reductase, did not reduce the stimulation observed in the presence of simvastatin indicating that HMG-CoA reductase activity is not involved. In view of the multiple biologic properties of AA, the AA released as a result of the action of the statins may play a role in some of the pharmacological effects attributed to these drugs.
Background
The statins, by inhibiting HMG-CoA reductase activity, reduce cholesterol and isoprenoid synthesis [1]. They are being used primarily in the prevention of atherosclerosis [2,3]. They have proven to be beneficial in preventing stroke [4] and may increase bone formation [5] and lower the risk of dementia [6]. Some of the benefits in patients with myocardial infarction are independent of cholesterol levels. Statins also inhibit cellular proliferation and induce apoptosis of tumor cells [7]. Essential fatty acids have several properties similar to the statins [8,9] and Das [10] has suggested that "essential fatty acids and their metabolites may serve as second messengers of the actions of statins."
Statins affect prostaglandin (PG) production. Mevastatin or lovastatin, at 25 μM, induce PGI2 production and cyclooxygenase (COX)-2 in human aorta smooth muscle cells [11]. Mevalonate and geranylgeranyl-pyrophosphate block these stimulations implicating the cholesterol biosynthetic pathway in this up-regulation. However, fluvastatin down-regulates rather than up-regulates COX-2 expression in human umbilical vein endothelial cells [12] and lovastatin reduces PGI2 production in bovine endothelial cells, human skin fibroblasts and arterial smooth muscle cells [13].
I observed that in rat liver cells, the statins stimulate the release of an essential fatty acid, arachidonic acid (AA) and stimulate production of its metabolite, PGI2. These stimulations are independent of HMG-CoA reductase activity as measured by the lack of any suppression by mevalonate.
Results
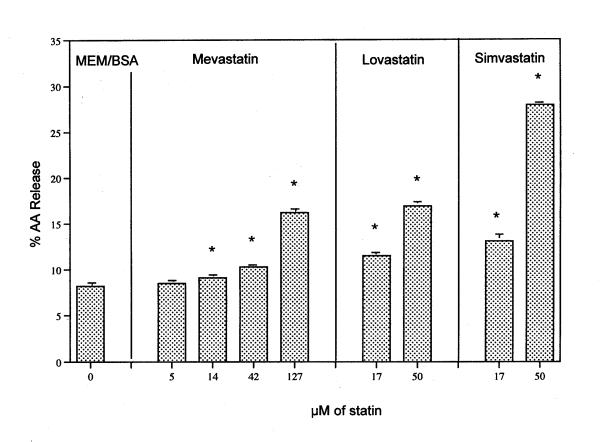
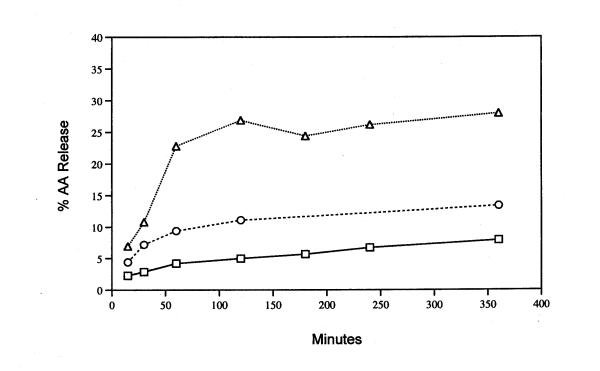
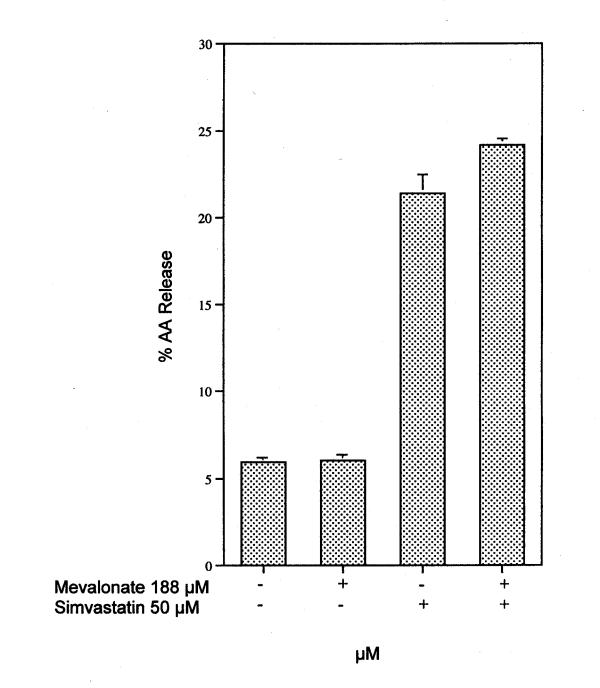
The release of AA from rat liver cells after 6 h incubation with mevastatin, lovastatin and simvastatin is shown in Fig. 1. An extract of the pharmaceutical product Lipitor® also significantly stimulates AA release (data not shown). The release of AA as a function of time in the presence of 50 μM lovastatin or simvastatin is shown in Fig. 2. Stimulation by lovastatin and simvastatin is observed after as little as 15 min incubation with cells (the time of release with mevastatin was not studied). Preincubation of the cells for 2 h with 1 μM actinomycin did not alter the quantity of AA release stimulated by 6 h incubation with simvastatin [17.0 ± 0.30 % (5) was released in the absence of actinomycin compared to 17.6 ± 0.60 % (5) released in its presence]. These results indicate that transcription is not required. I have shown previously that under these conditions, induced PG production is inhibited [14,15]. Nor does mevalonate, the direct product of HMG-CoA reductase, affect the stimulated release by 50 μM simvastatin (Fig. 3).
Figure 1.
AA release by mevastatin, lovastatin and simvastatin. Cells were incubated with these statins at the concentrations shown for 6 h. The analyses were performed on triplicate culture dishes. * Statistically significant vs MEM/BSA control (P < 0.05). Data are representative of several independent experiments.
Figure 2.
Time course of release of AA after incubation with 50 μM lovastatin (○), 50 μM simvastatin (△) and the MEM/BSA controls (□). Analyses were performed on triplicate or duplicate dishes. The average value is recorded.
Figure 3.
Effect of 188 μM mevalonate on AA release from rat liver cells incubated 6 h in the presence of 50 μM simvastatin. The analyses were performed on triplicate dishes. The bars show the mean values and brackets the SEM. This experiment was repeated two times with similar results.
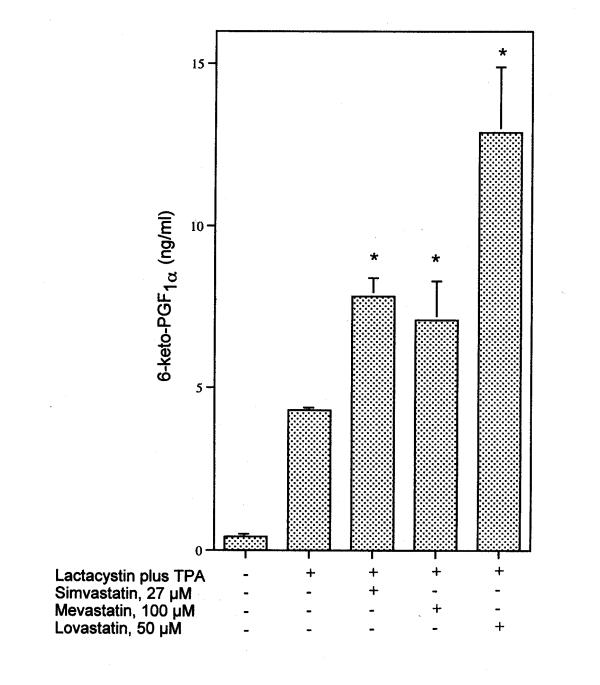
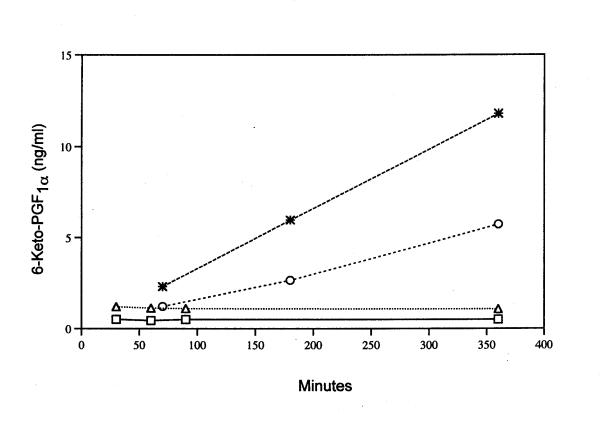
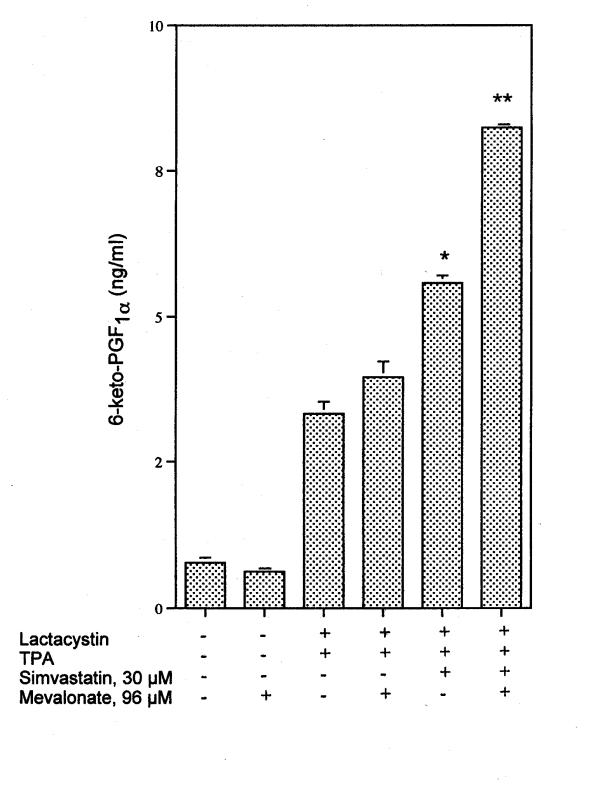
PGI2 production is enhanced in cells incubated with lactacystin in the presence of TPA (Fig. 4). Simvastatin, mevastatin and lovastatin enhance the induced PGI2 levels (Fig. 4). Even at 70 min the quantity of PGI2 produced by lactacystin plus TPA increased in the presence of 30 μM simvastatin (Fig. 5). The effect of 96 μM mevalonate on simvastatin's amplification of induced PGI2 in the presence of lactacystin plus TPA is shown in Fig. 6. Mevalonate does not suppress the stimulated PGI2 production. Mevalonate amplifies even further the effect of simvastatin on lactacystin plus TPA induced PGI2 production, suggesting a role for a downstream isoprenoid in the induction pathway (Fig. 6).
Figure 4.
Effect of 27 μM simvastatin, 100 μM mevastatin and 50 μM lovastatin on lactacystin plus TPA induced 6-keto-PGF1α production after 6 h incubation. At the levels tested, simvastatin, mevastatin and lovastatin alone do significantly stimulate basal PGI2 production, but by < 1 ng/ml. The analyses were performed on triplicate dishes. * Statistically significant vs lactacystin plus TPA (P < 0.05). This experiment was repeated with similar results.
Figure 5.
Time course of 6-keto-PGF1α production during incubation with MEM/BSA (□), 30 μM simvastatin (△), lactacystin plus TPA (○) and lactacystin plus TPA plus 30 μM simvastatin (*). Analyses were performed on triplicate or duplicate dishes. The average value is recorded.
Figure 6.
Effect of 96 μM mevalonate on 6-keto-PGF1α produced by stimulation with 30 μM simvastatin in the presence of lactacystin plus TPA after a 6 h incubation. Analyses were performed on triplicate dishes. * Statistically significant vs lactacystin plus TPA (P < 0.05) ** Statistically significant vs lactacystin plus TPA and simvastatin (P < 0.05).
Discussion
Mevastatin, lovastatin, simvastatin and atorvastatin stimulate the release of AA from rat liver cells. They do so even after preincubation with actinomycin. Mevalonate, the product of HMG-CoA reductase, does not suppress simvastatin's stimulated AA release. Therefore, it appears that neither transcription nor cholesterol biosynthesis is required for this stimulated AA release. HMG-CoA reductase activity also is not required for up-regulation of induced PGI2 production by simvastatin.
AA has a number of important bioactivities. It serves as substrate for cyclooxygenases, lipoxygenases and cytochrome P450 epoxygenases. After additional enzyme reactions the products yield a variety of biologically active prostanoids [16]. AA also activates leukocyte NADPH oxidase [17], activates PPAR receptors [18], affects H+ channel [19], K+ channel [20] and Ca 2+ channel activities [21]. It elevates heat shock gene expression [22], induces apoptosis [23] and inhibits Ca 2+ induced Ca 2+ release [24]. In human embryonic kidney cells it down-regulates sterol regulatory element-binding proteins (SREBPs), especially the SREBP-1a isoform [25]. The release of AA after incubation with sulindac and the stimulation of apoptosis via conversion of sphingomyelin to ceramide [26] is a mechanism proposed to mediate cancer chemoprevention by nonsteroidal anti-inflammatory drugs [26]. I have postulated that, in addition to nonsteroidal anti-inflammatory drugs, the continuous use of other commonly prescribed drugs that release AA is potentially cancer preventative [27].
The major product of PGH2 metabolism by these rat liver cells is PGI2, although 10% or less of the metabolites are PGE2 and PGF2α [28]. In this study, only PGI2 was measured. Although PGI2 synthase is an induced enzyme [29], the increased PGI2 production probably reflects COX activity [27]. Up-regulation of PGI2 production is in agreement with the findings of Degreave et al [11] who showed that mevastatin and lovastatin up-regulate COX activity in human aortic smooth muscle cells. However, while up-regulation in the muscle cell system involves cholesterol biosynthesis, the up-regulation observed in rat liver cells does not. Statins also have been shown to down-regulate COX activity in human umbilical vein endothelial cells [12]. In addition to differences inherent in the type of cells being studied by the various investigators, the apparently contradictory findings may also be due to examination of the cells at different stages of growth. The published studies [11-13] use cells at confluent densities and also may have been in the G0 stage of growth. The rat liver cells studied in this report were at low density (dividing cells).
Since statins activate at μM concentrations, it is likely that the release of AA and effects on PGI2 production are reflections of interaction with cell membranes.
Conclusions
Statins stimulate AA release from and PGI2 production in rat liver cells. It is not yet possible to assign a biological consequence to either stimulation. However, in view of the many biological activities of AA, its release could generate the essential fatty acid that is postulated to be a mediator of statin action [10]. Such mediation does not involve cholesterol biosynthesis.
Materials
The C-9 rat liver cell line was purchased from the American Type Culture Collection (Manassa, VA, USA). The cells were maintained in Eagle's minimum essential medium (MEM) supplemented with 10% fetal bovine serum. The [3H] AA (91.8 Ci/mmol) was from NEN Life Science Products, Inc., (Boston, MA, USA). Mevastatin, lovastatin, DL-mevalonic acid lactone were from Sigma Chemical Co. (St. Louis, MO, USA). I thank Dr. Gregory R. Mundy, Institute for Drug Development, San Antonio, TX, for the generous supply of simvastatin. For the experiments with atorvastatin, CH3CH2OH and CH3OH extracts of Lipitor® tablets were used.
Methods
On the day prior to experiments, the rat liver cells were treated with 0.25% trypsin-EDTA and, after addition of minimal essential media (MEM) containing 10% fetal calf serum, the floating cells were seeded onto 35 mm culture dishes. The plating densities varied from 0.1 to 0.5 × 105 cells/35 mm dish. The freshly seeded cultures were incubated 24 h to allow for cell attachment. After decantation of MEM containing the fetal bovine serum, 1.0 ml fresh MEM containing 10% fetal bovine serum and [3H] AA (0.2 μCi/ml) was added and the cells incubated for another 24 h. The cells were washed 4 times with MEM and incubated for various periods of time with 1.0 ml of different concentrations of each compound in MEM containing 1.0 mg BSA/ml. The culture fluids were then decanted, centrifuged at 2000 × g for 10 min, and 200 μl of the supernate counted for radioactivity. Radioactivity recovered in the washes before the 6 h incubation was compared to input radioactivity to calculate the % radioactivity incorporated into the cells [30].
For PGI2 production, 1.0 ml of MEM supplemented with 10% fetal bovine serum, void of [3H] AA, was added after the first 24 h incubation. The cells were incubated for another 24 h, washed three times with MEM, then incubated with lactacystin plus TPA and the compounds in MEM containing BSA for various periods of time. The culture fluids were decanted and analyzed for 6-keto-PGF1α, the stable hydrolytic product of PGI2, by radioimmunoassay [31].
Presentation of data
The [3H] AA release is presented as a percentage of the radioactivity incorporated by the cells. Three to six culture dishes were used for each experimental point. The data are expressed as mean values ± SEM (number of dishes). The data were statistically evaluated by unpaired Student's t-test. A P value < 0.05 was considered significant.
Acknowledgments
Acknowledgements
I thank Hilda B. Gjika for preparation of the manuscript and Dr. Armen Tashjian Jr., Department of Cancer Cell Biology, Harvard School of Public Health, for his continuing interest in these studies.
References
- Goldstein JL, Brown MS. Regulation of the mevalonate pathway. Nature. 1990;343:425–430. doi: 10.1038/343425a0. [DOI] [PubMed] [Google Scholar]
- Bellosta S, Bernini F, Ferri N, Quarato P, Canavesi M, Arnaboldi L, Fumagalli R, Paoletti R, Corsini A. Direct vascular effects of HMG-CoA reductase inhibitors. Atherosclerosis. 1998;137:S101–109. doi: 10.1016/s0021-9150(97)00319-5. [DOI] [PubMed] [Google Scholar]
- Koh KK. Effects of statins on vascular wall: vasomotor function, inflammation, and plaque stability. Cardiovasc Res. 2000;47:648–657. doi: 10.1016/S0008-6363(00)00146-2. [DOI] [PubMed] [Google Scholar]
- Vaughan CJ, Delanty N. Neuroprotective properties of statins in cerebral ischemia and stroke. Stroke. 1999;30:1969–1973. doi: 10.1161/01.str.30.9.1969. [DOI] [PubMed] [Google Scholar]
- Mundy G, Garrett R, Harris S, Chan J, Chen D, Rossini G, Boyce B, Zhao M, Gutierrez G. Stimulation of bone formation in vitro and in rodents by statins. Science. 1999;286:1946–1949. doi: 10.1126/science.286.5446.1946. [DOI] [PubMed] [Google Scholar]
- Jick H, Zornberg GL, Jick SS, Seshadri S, Drachman DA. Statins and the risk of dementia. Lancet. 2000;356:1627–1631. doi: 10.1016/S0140-6736(00)03155-X. [DOI] [PubMed] [Google Scholar]
- Knapp AC, Huang J, Starling G, Kiener PA. Inhibitors of HMG-CoA reductase sensitize human smooth muscle cells to Fas-ligand and cytokine-induced cell death. Atherosclerosis. 2000;152:217–227. doi: 10.1016/S0021-9150(99)00462-1. [DOI] [PubMed] [Google Scholar]
- Ihara-Watanabe M, Umekawa H, Takahashi T, Furuichi Y. Effects of dietary alpha- or gamma-linolenic acid on levels and fatty acid compositions of serum and hepatic lipids, and activity and mRNA abundance of 3-hydroxy-3-methylglutaryl CoA reductase in rats. Comp Biochem Physiol A Mol Integr Physiol. 1999;122:213–220. doi: 10.1016/S1095-6433(98)10176-9. [DOI] [PubMed] [Google Scholar]
- Proksch E, Feingold KR, Elias PM. Epidermal HMG CoA reductase activity in essential fatty acid deficiency: barrier requirements rather than eicosanoid generation regulate cholesterol synthesis. J Invest Dermatol. 1992;99:216–220. doi: 10.1111/1523-1747.ep12650440. [DOI] [PubMed] [Google Scholar]
- Das UN. Essential fatty acids as possible mediators of the actions of statins. Prostaglandins Leukot Essent Fatty Acids. 2001;65:37–40. doi: 10.1054/plef.2001.0285. [DOI] [PubMed] [Google Scholar]
- Degraeve F, Bolla M, Blaie S, Creminon C, Quere I, Boquet P, Levy-Toledano S, Bertoglio J, Habib A. Modulation of COX-2 expression by statins in human aortic smooth muscle cells. Involvement of geranylgeranylated proteins. J Biol Chem. 2001;276:46849–46855. doi: 10.1074/jbc.M104197200. [DOI] [PubMed] [Google Scholar]
- Inoue I, Goto S, Mizotani K, Awata T, Mastunaga T, Kawai S, Nakajima T, Hokari S, Komoda T, Katayama S. Lipophilic HMG-CoA reductase inhibitor has an anti-inflammatory effect: reduction of mRNA levels for interleukin-1beta, interleukin-6, cyclooxygenase-2, and p22phox by regulation of peroxisome proliferator-activated receptor alpha (PPARalpha) in primary endothelial cells. Life Sci. 2000;67:863–876. doi: 10.1016/S0024-3205(00)00680-9. [DOI] [PubMed] [Google Scholar]
- Falke P, Mattiasson I, Stavenow L, Hood B. Effects of a competitive inhibitor (mevinolin) of 3-hydroxy-3-methylglutaryl coenzyme A reductase on human and bovine endothelial cells, fibroblasts and smooth muscle cells in vitro. Pharmacol Toxicol. 1989;64:173–176. doi: 10.1111/j.1600-0773.1989.tb00624.x. [DOI] [PubMed] [Google Scholar]
- Pong SS, Hong SL, Levine L. Prostaglandin production by methylcholanthrene-transformed mouse BALB/3T3. Requirement for protein synthesis. J Biol Chem. 1977;252:1408–1413. [PubMed] [Google Scholar]
- Levine L. Nuclear receptor agonists stimulate release of arachidonic acid from rat liver cells. Prostaglandins Leukot Essent Fatty Acids. 2002;67:453–459. doi: 10.1054/plef.2002.0456. [DOI] [PubMed] [Google Scholar]
- Smith WL. The eicosanoids and their biochemical mechanisms of action. Biochem J. 1989;259:315–324. doi: 10.1042/bj2590315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dana R, Leto TL, Malech HL, Levy R. Essential requirement of cytosolic phospholipase A2 for activation of the phagocyte NADPH oxidase. J Biol Chem. 1998;273:441–445. doi: 10.1074/jbc.273.1.441. [DOI] [PubMed] [Google Scholar]
- Kliewer SA, Sundseth SS, Jones SA, Brown PJ, Wisely GB, Koble CS, Devchand P, Wahli W, Willson TM, Lenhard JM, Lehmann JM. Fatty acids and eicosanoids regulate gene expression through direct interactions with peroxisome proliferator-activated receptors alpha and gamma. Proc Natl Acad Sci U S A. 1997;94:4318–4323. doi: 10.1073/pnas.94.9.4318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henderson LM, Thomas S, Banting G, Chappell JB. The arachidonate-activatable, NADPH oxidase-associated H+ channel is contained within the multi-membrane-spanning N-terminal region of gp91-phox. Biochem J. 1997;325:701–705. doi: 10.1042/bj3250701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lesage F, Maingret F, Lazdunski M. Cloning and expression of human TRAAK, a polyunsaturated fatty acids-activated and mechano-sensitive K(+) channel. FEBS Lett. 2000;471:137–140. doi: 10.1016/S0014-5793(00)01388-0. [DOI] [PubMed] [Google Scholar]
- Mignen O, Shuttleworth TJ. I(ARC), a novel arachidonate-regulated, noncapacitative Ca(2+) entry channel. J Biol Chem. 2000;275:9114–9119. doi: 10.1074/jbc.275.13.9114. [DOI] [PubMed] [Google Scholar]
- Jurivich DA, Sistonen L, Sarge KD, Morimoto RI. Arachidonate is a potent modulator of human heat shock gene transcription. Proc Natl Acad Sci U S A. 1994;91:2280–2284. doi: 10.1073/pnas.91.6.2280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Surette ME, Fonteh AN, Bernatchez C, Chilton FH. Perturbations in the control of cellular arachidonic acid levels block cell growth and induce apoptosis in HL-60 cells. Carcinogenesis. 1999;20:757–763. doi: 10.1093/carcin/20.5.757. [DOI] [PubMed] [Google Scholar]
- Siegel G, Sternfeld L, Gonzalez A, Schulz I, Schmid A. Arachidonic acid modulates the spatiotemporal characteristics of agonist-evoked -Ca2+ waves in mouse pancreatic acinar cells. J Biol Chem. 2001;276:16986–16991. doi: 10.1074/jbc.M101136200. [DOI] [PubMed] [Google Scholar]
- Hannah VC, Ou J, Luong A, Goldstein JL, Brown MS. Unsaturated fatty acids down-regulate SREBP isoforms 1a and 1c by two mechanisms in HEK-293 cells. J Biol Chem. 2001;276:4365–4372. doi: 10.1074/jbc.M007273200. [DOI] [PubMed] [Google Scholar]
- Chan TA, Morin PJ, Vogelstein B, Kinzler KW. Mechanisms underlying nonsteroidal antiinflammatory drug-mediated apoptosis. Proc Natl Acad Sci U S A. 1998;95:681–686. doi: 10.1073/pnas.95.2.681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levine l. Does the release of arachidonic acid from cells play a role in cancer chemoprevention? FASEB Journal. 2003. [DOI] [PubMed]
- Rigas A, Levine L. Arachidonic acid metabolism by rat liver cells (the C-9 cell line). J Pharmacol Exp Ther. 1984;231:230–235. [PubMed] [Google Scholar]
- Ulrich V, Brugger R, Lottspeich F, Siegle I. Properties of prostacyclin synthase in Honn er all. Eicosanoids and other bioactive lipids in cancer, inflammation and radiation injury. 1997. pp. 113–119. [DOI] [PubMed]
- Levine L. Stimulated release of arachidonic acid from rat liver cells by celecoxib and indomethacin. Prostaglandins Leukot Essent Fatty Acids. 2001;65:31–35. doi: 10.1054/plef.2001.0284. [DOI] [PubMed] [Google Scholar]
- Levine L. Measurement of arachidonic acid metabolites by radioimmunoassay. In: Rose NR, Friedman H, fahey JL, editor. Manual of Clinical Laboratory Immunology. 3. Washington DC: American Society for Microbiology; 1986. pp. 685–691. [Google Scholar]