Abstract
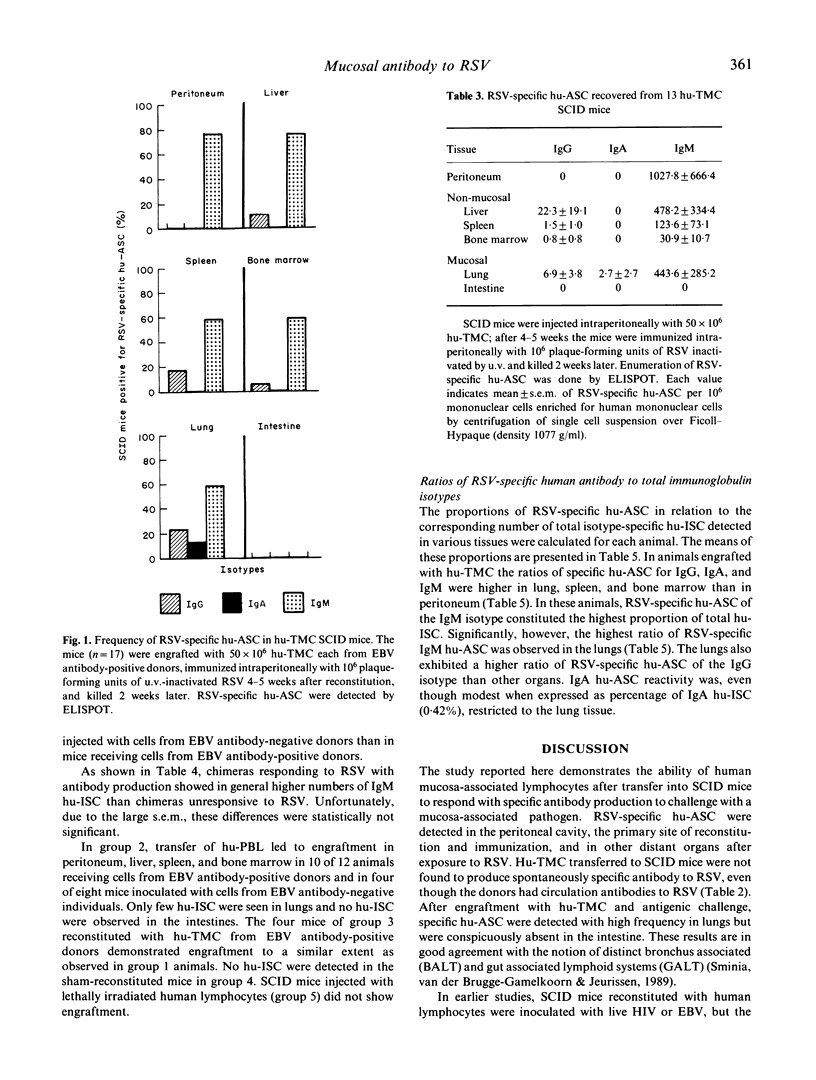
Groups of C.B-17 SCID mice were reconstituted intraperitoneally with human tonsillar mononuclear cells (hu-TMC) from children seropositive for antibody to respiratory syncytial virus (RSV) and subsequently challenged intraperitoneally with inactivated RSV or sham-immunized. The synthesis and the distribution characteristics of human antibody to RSV in various murine tissues were studied using an enzyme-linked immunospot assay (ELISPOT). No specific antibody was observed in sham-immunized animals. In contrast, mice engrafted with hu-TMC exhibited the appearance of specific human antibody secreting cells (hu-ASC) after i.p. immunization with inactivated RSV. RSV-specific hu-ASC were detected only in animals engrafted with cells from donors seropositive for antibodies to Epstein-Barr virus. Hu-TMC engrafted mice showed RSV-specific IgM and, in lower numbers, IgG hu-ASC in several tissues including the lungs. Numbers of RSV-specific IgA hu-ASC were low, however, and detected only in the lung. No RSV-specific hu-ASC were detected in the intestine. These data demonstrate for the first time that hu-TMC-SCID chimeras respond to immunization with viral antigen. Furthermore, the results suggest that hu-TMC engraft in lungs but not in the intestinal tissue.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Beagley K. W., Eldridge J. H., Lee F., Kiyono H., Everson M. P., Koopman W. J., Hirano T., Kishimoto T., McGhee J. R. Interleukins and IgA synthesis. Human and murine interleukin 6 induce high rate IgA secretion in IgA-committed B cells. J Exp Med. 1989 Jun 1;169(6):2133–2148. doi: 10.1084/jem.169.6.2133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bosma G. C., Custer R. P., Bosma M. J. A severe combined immunodeficiency mutation in the mouse. Nature. 1983 Feb 10;301(5900):527–530. doi: 10.1038/301527a0. [DOI] [PubMed] [Google Scholar]
- Cahill R. N., Poskitt D. C., Hay J. B., Heron I., Trnka Z. The migration of lymphocytes in the fetal lamb. Eur J Immunol. 1979 Mar;9(3):251–253. doi: 10.1002/eji.1830090315. [DOI] [PubMed] [Google Scholar]
- Cannon M. J., Pisa P., Fox R. I., Cooper N. R. Epstein-Barr virus induces aggressive lymphoproliferative disorders of human B cell origin in SCID/hu chimeric mice. J Clin Invest. 1990 Apr;85(4):1333–1337. doi: 10.1172/JCI114573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Czerkinsky C., Moldoveanu Z., Mestecky J., Nilsson L. A., Ouchterlony O. A novel two colour ELISPOT assay. I. Simultaneous detection of distinct types of antibody-secreting cells. J Immunol Methods. 1988 Nov 25;115(1):31–37. doi: 10.1016/0022-1759(88)90306-7. [DOI] [PubMed] [Google Scholar]
- Dorshkind K., Keller G. M., Phillips R. A., Miller R. G., Bosma G. C., O'Toole M., Bosma M. J. Functional status of cells from lymphoid and myeloid tissues in mice with severe combined immunodeficiency disease. J Immunol. 1984 Apr;132(4):1804–1808. [PubMed] [Google Scholar]
- Duchosal M. A., McConahey P. J., Robinson C. A., Dixon F. J. Transfer of human systemic lupus erythematosus in severe combined immunodeficient (SCID) mice. J Exp Med. 1990 Sep 1;172(3):985–988. doi: 10.1084/jem.172.3.985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernandez-Botran R., Vitetta E. S. A soluble, high-affinity, interleukin-4-binding protein is present in the biological fluids of mice. Proc Natl Acad Sci U S A. 1990 Jun;87(11):4202–4206. doi: 10.1073/pnas.87.11.4202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gray D., Leanderson T. Expansion, selection and maintenance of memory B-cell clones. Curr Top Microbiol Immunol. 1990;159:1–17. doi: 10.1007/978-3-642-75244-5_1. [DOI] [PubMed] [Google Scholar]
- Hall J. G., Hopkins J., Orlans E. Studies on the lymphocytes of sheep. III. Destination of lymph-borne immunoblasts in relation to their tissue of origin. Eur J Immunol. 1977 Jan;7(1):30–37. doi: 10.1002/eji.1830070108. [DOI] [PubMed] [Google Scholar]
- Henle W., Henle G. E., Horwitz C. A. Epstein-Barr virus specific diagnostic tests in infectious mononucleosis. Hum Pathol. 1974 Sep;5(5):551–565. doi: 10.1016/s0046-8177(74)80006-7. [DOI] [PubMed] [Google Scholar]
- Husband A. J., Gowans J. L. The origin and antigen-dependent distribution of IgA-containing cells in the intestine. J Exp Med. 1978 Nov 1;148(5):1146–1160. doi: 10.1084/jem.148.5.1146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamel-Reid S., Letarte M., Sirard C., Doedens M., Grunberger T., Fulop G., Freedman M. H., Phillips R. A., Dick J. E. A model of human acute lymphoblastic leukemia in immune-deficient SCID mice. Science. 1989 Dec 22;246(4937):1597–1600. doi: 10.1126/science.2595371. [DOI] [PubMed] [Google Scholar]
- Kawanishi H., Saltzman L. E., Strober W. Characteristics and regulatory function of murine con A-induced, cloned T cells obtained from Peyer's patches and spleen: mechanisms regulating isotype-specific immunoglobulin production by Peyer's patch B cells. J Immunol. 1982 Aug;129(2):475–483. [PubMed] [Google Scholar]
- Kawanishi H., Saltzman L. E., Strober W. Mechanisms regulating IgA class-specific immunoglobulin production in murine gut-associated lymphoid tissues. I. T cells derived from Peyer's patches that switch sIgM B cells to sIgA B cells in vitro. J Exp Med. 1983 Feb 1;157(2):433–450. doi: 10.1084/jem.157.2.433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawanishi H., Saltzman L., Strober W. Mechanisms regulating IgA class-specific immunoglobulin production in murine gut-associated lymphoid tissues. II. Terminal differentiation of postswitch sIgA-bearing Peyer's patch B cells. J Exp Med. 1983 Sep 1;158(3):649–669. doi: 10.1084/jem.158.3.649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kiyono H., Cooper M. D., Kearney J. F., Mosteller L. M., Michalek S. M., Koopman W. J., McGhee J. R. Isotype specificity of helper T cell clones. Peyer's patch Th cells preferentially collaborate with mature IgA B cells for IgA responses. J Exp Med. 1984 Mar 1;159(3):798–811. doi: 10.1084/jem.159.3.798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kiyono H., McGhee J. R., Mosteller L. M., Eldridge J. H., Koopman W. J., Kearney J. F., Michalek S. M. Murine Peyer's patch T cell clones. Characterization of antigen-specific helper T cells for immunoglobulin A responses. J Exp Med. 1982 Oct 1;156(4):1115–1130. doi: 10.1084/jem.156.4.1115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kiyono H., Mosteller-Barnum L. M., Pitts A. M., Williamson S. I., Michalek S. M., McGhee J. R. Isotype-specific immunoregulation. IgA-binding factors produced by Fc alpha receptor-positive T cell hybridomas regulate IgA responses. J Exp Med. 1985 Apr 1;161(4):731–747. doi: 10.1084/jem.161.4.731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krams S. M., Dorshkind K., Gershwin M. E. Generation of biliary lesions after transfer of human lymphocytes into severe combined immunodeficient (SCID) mice. J Exp Med. 1989 Dec 1;170(6):1919–1930. doi: 10.1084/jem.170.6.1919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y. J., Joshua D. E., Williams G. T., Smith C. A., Gordon J., MacLennan I. C. Mechanism of antigen-driven selection in germinal centres. Nature. 1989 Dec 21;342(6252):929–931. doi: 10.1038/342929a0. [DOI] [PubMed] [Google Scholar]
- Mayer L., Fu S. M., Kunkel H. G. Human T cell hybridomas secreting factors for IgA-specific help, polyclonal B cell activation, and B cell proliferation. J Exp Med. 1982 Dec 1;156(6):1860–1865. doi: 10.1084/jem.156.6.1860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayer L., Posnett D. N., Kunkel H. G. Human malignant T cells capable of inducing an immunoglobulin class switch. J Exp Med. 1985 Jan 1;161(1):134–144. doi: 10.1084/jem.161.1.134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCune J. M., Namikawa R., Kaneshima H., Shultz L. D., Lieberman M., Weissman I. L. The SCID-hu mouse: murine model for the analysis of human hematolymphoid differentiation and function. Science. 1988 Sep 23;241(4873):1632–1639. doi: 10.1126/science.241.4873.1632. [DOI] [PubMed] [Google Scholar]
- McCune J. M., Namikawa R., Shih C. C., Rabin L., Kaneshima H. Suppression of HIV infection in AZT-treated SCID-hu mice. Science. 1990 Feb 2;247(4942):564–566. doi: 10.1126/science.2300816. [DOI] [PubMed] [Google Scholar]
- McIntosh K., Ellis E. F., Hoffman L. S., Lybass T. G., Eller J. J., Fulginiti V. A. The association of viral and bacterial respiratory infections with exacerbations of wheezing in young asthmatic children. J Pediatr. 1973 Apr;82(4):578–590. doi: 10.1016/S0022-3476(73)80582-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore K. W., Vieira P., Fiorentino D. F., Trounstine M. L., Khan T. A., Mosmann T. R. Homology of cytokine synthesis inhibitory factor (IL-10) to the Epstein-Barr virus gene BCRFI. Science. 1990 Jun 8;248(4960):1230–1234. doi: 10.1126/science.2161559. [DOI] [PubMed] [Google Scholar]
- Mosier D. E., Gulizia R. J., Baird S. M., Wilson D. B. Transfer of a functional human immune system to mice with severe combined immunodeficiency. Nature. 1988 Sep 15;335(6187):256–259. doi: 10.1038/335256a0. [DOI] [PubMed] [Google Scholar]
- Namikawa R., Kaneshima H., Lieberman M., Weissman I. L., McCune J. M. Infection of the SCID-hu mouse by HIV-1. Science. 1988 Dec 23;242(4886):1684–1686. doi: 10.1126/science.3201256. [DOI] [PubMed] [Google Scholar]
- Okamoto Y., Brodsky L., Bernstein J. M., Ogra P. L. Characteristics of in vitro production of mucosal antibody to respiratory syncytial virus in tonsillar tissue lymphocytes. Clin Immunol Immunopathol. 1988 Nov;49(2):299–307. doi: 10.1016/0090-1229(88)90120-1. [DOI] [PubMed] [Google Scholar]
- Parrott D. M., Ferguson A. Selective migration of lymphocytes within the mouse small intestine. Immunology. 1974 Mar;26(3):571–588. [PMC free article] [PubMed] [Google Scholar]
- Pierce N. F., Cray W. C., Jr Determinants of the localization, magnitude, and duration of a specific mucosal IgA plasma cell response in enterically immunized rats. J Immunol. 1982 Mar;128(3):1311–1315. [PubMed] [Google Scholar]
- Sminia T., van der Brugge-Gamelkoorn G. J., Jeurissen S. H. Structure and function of bronchus-associated lymphoid tissue (BALT). Crit Rev Immunol. 1989;9(2):119–150. [PubMed] [Google Scholar]
- Ueba O. Purification and polypeptides of respiratory syncytial virus. Microbiol Immunol. 1980;24(4):361–364. doi: 10.1111/j.1348-0421.1980.tb02839.x. [DOI] [PubMed] [Google Scholar]
- Welliver R. C., Kaul A., Ogra P. L. Cell-mediated immune response to respiratory syncytial virus infection: relationship to the development of reactive airway disease. J Pediatr. 1979 Mar;94(3):370–375. doi: 10.1016/s0022-3476(79)80573-9. [DOI] [PubMed] [Google Scholar]
- Welliver R. C., Kaul T. N., Ogra P. L. The appearance of cell-bound IgE in respiratory-tract epithelium after respiratory-syncytial-virus infection. N Engl J Med. 1980 Nov 20;303(21):1198–1202. doi: 10.1056/NEJM198011203032103. [DOI] [PubMed] [Google Scholar]
- Welliver R. C., Wong D. T., Sun M., Middleton E., Jr, Vaughan R. S., Ogra P. L. The development of respiratory syncytial virus-specific IgE and the release of histamine in nasopharyngeal secretions after infection. N Engl J Med. 1981 Oct 8;305(15):841–846. doi: 10.1056/NEJM198110083051501. [DOI] [PubMed] [Google Scholar]
- Wong D. T., Rosenband M., Hovey K., Ogra P. L. Respiratory syncytial virus infection in immunosuppressed animals: implications in human infection. J Med Virol. 1985 Dec;17(4):359–370. doi: 10.1002/jmv.1890170408. [DOI] [PubMed] [Google Scholar]
- Yarchoan R., Barrow L. A., Kurman C., Strober W., Nelson D. L. Human peripheral blood mononuclear cells produce IgA anti-influenza virus antibody in a secondary in vitro antibody response. J Immunol. 1985 Aug;135(2):1033–1039. [PubMed] [Google Scholar]


