Abstract
β-Catenin is a transcriptional activator that regulates embryonic development as part of the Wnt pathway and also plays a role in tumorigenesis. The mechanisms leading to Wnt-induced stabilization of β-catenin, which results in its translocation to the nucleus and activation of transcription, have been an area of intense interest. However, it is not clear whether stimuli other than Wnts can lead to important stabilization of β-catenin and, if so, what factors mediate that stabilization and what biologic processes might be regulated. Herein we report that β-catenin is stabilized in cardiomyocytes after these cells have been exposed to hypertrophic stimuli in culture or in vivo. The mechanism by which β-catenin is stabilized is distinctly different from that used by Wnt signaling. Although, as with Wnt signaling, inhibition of glycogen synthase kinase-3 remains central to hypertrophic stimulus-induced stabilization of β-catenin, the mechanism by which this occurs involves the recruitment of activated PKB to the β-catenin-degradation complex. PKB stabilizes the complex and phosphorylates glycogen synthase kinase-3 within the complex, inhibiting its activity directed at β-catenin. Finally, we demonstrate via adenoviral gene transfer that β-catenin is both sufficient to induce growth in cardiomyocytes in culture and in vivo and necessary for hypertrophic stimulus-induced growth. Thus, in these terminally differentiated cells, β-catenin is stabilized by hypertrophic stimuli acting via heterotrimeric G protein-coupled receptors. The stabilization occurs via a unique Wnt-independent mechanism and results in cellular growth.
Glycogen synthase kinase-3 (GSK-3)-α and -β function as inhibitors of Wnt signaling during the development of the embryonic axis (1). GSK-3 is also a negative regulator of growth in cardiomyocytes, cells that are terminally differentiated and can only undergo hypertrophic growth (2–4). Inhibition of GSK-3 is necessary for the hypertrophic response both in vitro and in vivo (2–5), and at least some of the antihypertrophic effects of active GSK-3 are mediated by regulating activity of the nuclear factor of activated T cells (NF-AT) family of transcription factors (2, 5, 6). However, we found that gene transfer of an activated NF-AT3 failed to recapitulate the full hypertrophic response and asked whether additional GSK-3 targets could play a role. β-Catenin, which plays critical roles in development and tumorigenesis (7, 8), is one potential target. The protein exists in the cell in two pools, membrane associated and cytosolic. In the membrane, β-catenin links cadherins to the cytoskeleton (9). β-Catenin also functions as a transcriptional coactivator (8), the source of this being the cytosolic pool, which is negatively regulated by GSK-3. GSK-3 phosphorylates the amino-terminal region of β-catenin, targeting it for ubiquitination and degradation by the proteasome (10, 11). β-Catenin is phosphorylated by GSK-3 when part of a complex that includes the scaffolding protein Axin and the adenomatous polyposis coli gene product, APC (12). Inhibition of GSK-3 is therefore essential for the stabilization and accumulation of β-catenin.
GSK-3 activity is inhibited via two primary mechanisms. One, phosphorylation of an amino-terminal serine residue (Ser-21 for α, Ser-9 for β; ref. 13), is catalyzed by PKB (14). This phosphorylation inhibits GSK-3 activity directed toward primed substrates that have been previously phosphorylated at a site four residues carboxy terminal to the GSK-3 phosphorylation site but does not inhibit kinase activity directed toward unprimed substrates (15, 16). This mechanism is used in growth factor signaling but is not believed to be important in Wnt signaling and has been reported to be insufficient to induce β-catenin accumulation (17, 18). Although these data are compatible with β-catenin being unprimed in situ (19), recent studies indicate that β-catenin can exist as a primed target for GSK-3, when phosphorylated on Ser-45 by casein kinase 1α (20), and raise the possibility that, in certain circumstances, Ser-9 phosphorylation of GSK-3β could stabilize β-catenin. A second mechanism of inhibition of GSK-3, used by Wnts, involves, in part, complex formation of GSK-3 with GSK-3-binding protein/Frat1 (21, 22). Complex formation is believed to sequester GSK-3 and primarily inhibit phosphorylation of unprimed substrates, at least in kinase assays in vitro (12, 15).
Increases in β-catenin levels in the cytosol, together with less well defined signals, lead to its translocation to the nucleus, where it acts in tandem with T cell factor (Tcf)/lymphocyte enhancer factor (Lef) family members to induce expression of several genes involved in cell cycle reentry, as well as in transformation of postnatal cells (23, 24). In this manuscript, we ask what role, if any, this pathway might be playing in terminally differentiated cells that cannot enter the cell cycle and whether the mechanisms regulating β-catenin stability differed in these cells (11).
Methods
Adenoviruses.
AdGFP, Adβ-catenin, and Adβ-cateninΔ contain cytomegalovirus-driven expression cassettes for enhanced GFP and either β-galactosidase or vesicular stomatitis virus-tagged β-catenin or β-cateninΔ (β-catenin deleted for the N-terminal 134 amino acids, a region that contains the GSK-3 phosphorylation sites), respectively, substituted for E1 through homologous recombination (24). AdGSK-3β(S9A), encoding GSK-3β with a Ser-9-to-Ala mutation has been described (2). AdNF-ATΔ, provided by Jeffery Molkentin (Children's Hospital Medical Center, Cincinnati), encodes NF-AT3 deleted for the first 317 amino acids and is constitutively active (6).
Cell Culture.
Neonatal rat ventricular myocytes (NRVM).
Cardiomyocytes were prepared from 1- to 2-d-old rats by using standard methods (2).
S2-Wingless (Wg)-secreting cells.
S2 cells expressing Drosophila Wg under the control of the metallothionein promoter were as described (25). Production of Wg was induced by addition of CdCl2 to the culture medium to a final concentration of 0.1 mM. Media was collected 6 h later. Although Drosophila in origin, when added to mammalian cells, Wg activates the Wnt pathway (see Fig. 1D).
Figure 1.
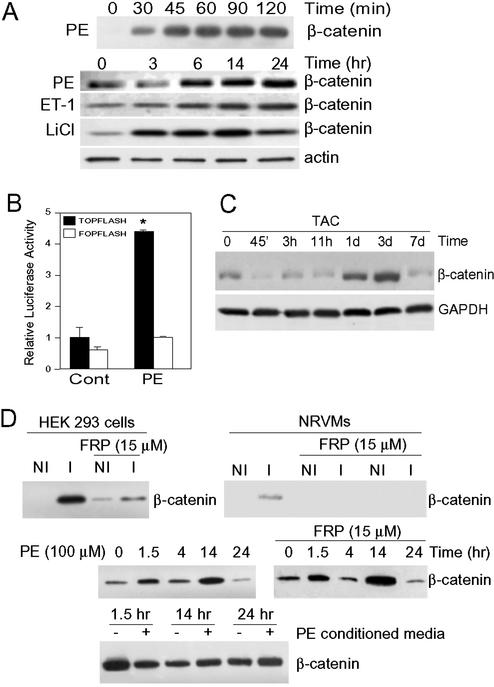
Hypertrophic stimuli induce β-catenin accumulation in vitro and in vivo. (A) NRVMs were treated with PE (100 μM), endothelin-1 (100 nM), or, as a positive control, with the GSK-3 inhibitor LiCl (10 mM), for the times indicated. Cytosolic fractions were blotted with anti-β-catenin antibody. Antiactin blot confirms equivalent protein loading. (B) NRVMs were transfected with pTOPFlash or pFOPFlash and 24 h later were stimulated with PE or vehicle (Cont) for an additional 24 h before luciferase assay. Luciferase activity of vehicle-treated, pTOPFlash-transfected cells was set at 1, and other results are expressed as fold activation relative to that value (n = 5 independent experiments, done in triplicate; *, P < 0.01 vs. all other values). (C) Rats were subjected to TAC or sham surgery (control) for the times indicated. Lysates of cytosolic fractions were blotted with anti-β-catenin antibody. Anti-GAPDH blot confirms equivalent loading. Shams had no increase in β-catenin throughout the 7-d protocol (data not shown). (D Top), NRVMs and HEK293 cells were treated for 2 h with media from S2 cells that had not been induced to secrete Wg (NI) or were induced (I) by the addition of CdCl2 (see Methods). A separate group of cells was pretreated with Frizzled-related protein-1 (FRP, 15 μM) for 1 h before the addition of NI or I S2 media for 2 h. Cytosolic fractions were blotted with anti-β-catenin antibody. (Middle) NRVMs were incubated with PE (100 μM), with or without Frizzled-related protein-1 pretreatment for 1 h, for the times indicated. Cytosolic fractions were blotted with anti-β-catenin antibody. (Bottom) NRVMs were treated with or without PE for 14 h, after which the conditioned media were collected and incubated for the times indicated with fresh cultures of NRVMs. Cytosolic fractions were blotted with anti-β-catenin antibody.
Luciferase Assays.
Plasmids: pTOPFlash contains three copies of the Tcf/Lef binding site upstream of the thymidine kinase minimal promoter and luciferase cDNA. pFOPFlash has three copies of a mutated Tcf/Lef binding site (Upstate Biotechnology). NRVMs were transfected by using FuGENE 6 (Roche) with 0.2 μg of plasmid per 1 × 105 cells. Luciferase activity was determined by using a commercially available assay system (Promega) and was normalized for transfection efficiency with pcDNA3-β-galactosidase.
[3H]Leucine Incorporation.
Cardiomyocytes in 12-well dishes were infected with Adβ-catenin, Adβ-cateninΔ, AdNF-ATΔ, or AdGFP for 48 h. For the final 14 h, [3H]leucine (1 μCi per well) was added. [3H]Leucine incorporation was determined as described (2).
Immunoprecipitation and Immunoblotting.
Immunoblotting of whole-cell lysates from cardiomyocytes in culture or the intact heart was performed as described (2, 26). Lysates were routinely blotted with antibodies against actin or GAPDH to confirm equivalent loading. All immunoblots shown are representative of at least three.
Cytosolic Fractionation.
NRVMs were fractionated by hypotonic lysis as described (2), except that the centrifugation was performed at 100,000 × g. Immunoblotting with an anticaveolin antibody has confirmed that, with this protocol, there is no contamination of the cytosolic fractions with membrane components.
Immunocytochemistry.
NRVMs on coverslips were transduced with viruses or transfected with plasmids, and cells were stained with antibodies noted in figure legends. Cardiomyocyte area was determined as described (2) by using openlab software from Improvision, Inc. (Quincy, MA).
Adenovirus Injections in Vivo.
Rats were anesthetized, and the myocardium of the left ventricle was directly injected at five sites on the lateral free wall with 1 μl (1 × 1011 particles per milliliter) of AdGFP, or Adβ-cateninΔ diluted in 10 μl of PBS. Eight days after injection, the hearts were arrested in cardioplegia solution and fixed. GFP-positive cardiomyocytes were identified in transverse sections by using an anti-GFP antibody (Santa Cruz Biotechnology), followed by a horseradish peroxidase-conjugated secondary Ab, and counterstained with hematoxylin. Cardiomyocyte cross-sectional area (CSA) was determined on GFP-positive myocytes (infected with AdGFP or Adβ-cateninΔ) and adjacent nontransduced myocytes.
Thoracic Aortic Constriction (TAC).
Rats were subjected to supravalvular aortic banding as described (26). Control rats underwent a sham operation.
Results
Stabilization of β-Catenin by Hypertrophic Stimuli.
We examined the response of NRVMs to hypertrophic agonists. Phenylephrine (PE) and endothelin-1 induced a biphasic pattern of stabilization of β-catenin with an initial peak at 90–120 min, a return to baseline by 3 h, and then a much more sustained peak lasting from ≈6 to 24 h (Fig. 1A). The increase in cytosolic β-catenin was equivalent to that induced by direct inhibition of GSK-3β activity with LiCl (Fig. 1A). Importantly, this accumulation of β-catenin was sufficient to activate transcription from a Tcf/Lef reporter, pTOPFlash, but not from a mutated Tcf/Lef reporter, pFOPFlash (Fig. 1B). β-Catenin was also stabilized in vivo in the rat heart exposed to the hypertrophic stress of pressure overload, although, unlike the cardiomyocytes in culture, there was no early peak after TAC (Fig. 1C). β-Catenin levels increased at 1 d and peaked by 3 d after TAC (Fig. 1C).
One mechanism by which hypertrophic agonists could stabilize β-catenin is by inducing release of a Wnt from cardiomyocytes and recruiting the classical Wnt pathway. We found that stimulation of cardiomyocytes with the Drosophila Wnt homolog, Wg, led to stabilization of β-catenin, although the response was relatively weak compared with that of HEK293 cells (Fig. 1D Top). Stabilization of β-catenin in response to Wg was blocked in both HEK293 cells and cardiomyocytes by addition to the media of Frizzled-related protein-1 (provided by Jeffrey Rubin, National Cancer Institute, Bethesda), which binds to Wg and Wnts, blocking activation of the Wnt pathway (Fig. 1D Top and ref. 27). In contrast, stabilization of β-catenin by PE was not blocked by Frizzled-related protein-1, suggesting it was not mediated by recruitment of the Wnt pathway (Fig. 1D Middle). In addition, PE-conditioned media did not lead to stabilization of β-catenin (Fig. 1D Bottom). These data suggest that PE-induced stabilization of β-catenin is not mediated by recruitment of the classical Wnt pathway and is also not secondary to release of a stable factor acting in a paracrine fashion.
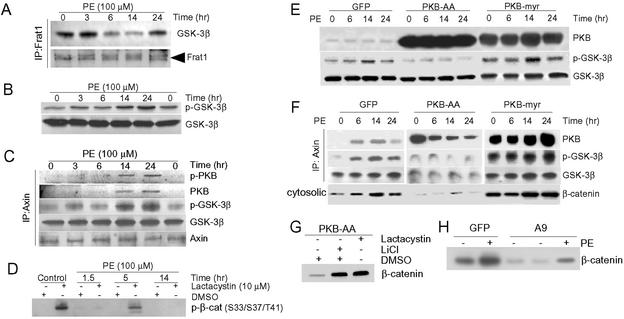
We examined the mechanisms by which hypertrophic agonists stabilized β-catenin in cardiomyocytes. We focused on the delayed/sustained peak of β-catenin because, based on the absence of an early peak in vivo (Fig. 1C), the later peak appeared to be more relevant to the hypertrophic response. We found that sequestration of GSK-3β by Frat1 played no significant role in inhibition of GSK-3β, because the amount of GSK-3β complexed with Frat1 decreased 6–14 h after PE (Fig. 2A), times at which β-catenin levels were increasing (Fig. 1A). We therefore turned to the alternative mechanism of GSK-3β inhibition, Ser-9 phosphorylation, and found an increase in phosphorylation at 3 h, which peaked at 14–24 h after PE (Fig. 2B), when β-catenin levels were maximal (Fig. 1A). Thus, the time course was consistent with Ser-9 phosphorylation playing a role in the accumulation of β-catenin. However, the critical pool of GSK-3 is believed to be that which is complexed with Axin in the β-catenin-degradation complex and, therefore, has access to β-catenin. The amount of GSK-3β in the complex that was phosphorylated on Ser-9 increased markedly, coincident with both the early peak (data not shown) and the late peak of PE-induced β-catenin accumulation (Fig. 2C). Thus, although GSK-3β was present in the Axin complex, and therefore had access to β-catenin, the GSK-3β was phosphorylated on Ser-9 and inhibited.
Figure 2.
Mechanisms of β-catenin stabilization in cardiomyocytes. NRVMs were stimulated with PE (100 μM) for the times indicated. (A) Lysates were subjected to immunoprecipitation with anti-Frat1 antibody followed by blotting with antibodies to GSK-3β or Frat1. (B) Lysates were blotted with antibodies to Ser-9-phosphorylated GSK-3β (p-GSK-3β) or to GSK-3β. (C) Lysates were subjected to immunoprecipitation with anti-Axin antibody followed by blotting with antibodies to p-GSK-3β, GSK-3β, Axin, or with an antibody recognizing Ser-473-phosphorylated (active) PKB (p-PKB) or total PKB. (D) NRVMs were pretreated with either DMSO or lactacystin (10 μM) for 1 h before stimulation with PE for the times indicated. Cytosolic fractions were prepared and then blotted with anti-phospho-specific Ser-33/37/Thr-41 β-catenin antibody. (E and F) NRVMs were transduced with AdPKB-AA, AdPKB-myr, or AdGFP (control) for 48 h by using a multiplicity of infection of 25 plaque-forming units per cell and then stimulated with PE for the times indicated. Cell lysates were blotted with antibodies to PKB, p-GSK-3β, and GSK-3β (E), or lysates were immunoprecipitated with anti-Axin antibody followed by blotting with antibodies to PKB, p-GSK-3β, GSK-3β (F). Cytosolic fractions were blotted with anti-β-catenin antibody (Bottom). (G) NRVMs were transduced with AdPKB-AA and 48 h later were incubated for 2 h with LiCl, lactacystin, or DMSO, as indicated. Cytosolic fractions were blotted with anti-β-catenin antibody. (H) NRVMs were transduced with either AdGSK-3β(S9A) or AdGFP for 24 h and then stimulated with PE or vehicle (-) for 14 h. Cytosolic fractions were blotted with anti-β-catenin antibody.
We also examined a time course of phosphorylation of β-catenin on three GSK-3β sites, Ser-33, Ser-37, and Thr-41, reasoning that this should correlate with the activation state of the kinase and inversely correlate with Ser-9 phosphorylation, if phosphorylation of GSK-3β on Ser-9 is the critical determinant of stabilization of β-catenin. We were unable to detect any phosphorylation of β-catenin on these sites at any point in the time course (Fig. 2D). However, this finding could be consistent with rapid clearance of β-catenin by the proteasome once these sites were phosphorylated. Therefore, we performed a PE time course in the presence of the proteasome inhibitor lactacystin. We found significant phosphorylation of β-catenin only in the presence of lactacystin and only in the control and 5-h-after-PE time points, when GSK-3β was most active (i.e., not phosphorylated on Ser-9; Fig. 2D). Thus, at the control and 5-h time points, when GSK-3β is not phosphorylated on Ser-9 and is active, inhibition of the proteasome exposes the phosphorylation of β-catenin on the GSK-3β sites. Conversely, when GSK-3β is phosphorylated on Ser-9 and inhibited (1.5 and 14 h after PE), there is no detectable phosphorylation of β-catenin, even in the presence of lactacystin. These studies suggest a central role for Ser-9 phosphorylation of GSK-3β in the stabilization of β-catenin by hypertrophic stimuli.
Coincident with the increase in phosphorylation of GSK-3β in the complex, active PKB, as evidenced by Ser-473 phosphorylation, was identified in the complex (Fig. 2C). To confirm that the association of activated PKB with the complex resulted in the observed phosphorylation of GSK-3β, we expressed a dominant inhibitory mutant of PKB, PKB-AA, in which the activating phosphorylation sites, Ser-473 and Thr-308, were replaced with Ala residues (28). PKB-AA inhibited the PE-induced phosphorylation of total cellular GSK-3β (Fig. 2E Middle) and, as shown in Fig. 2F Middle, inhibited phosphorylation of Axin-associated GSK-3β on Ser-9, promoted destabilization of the Axin complex as evidenced by significant dissociation of GSK-3β from Axin, and blocked the cytosolic accumulation of β-catenin. Two alternative dominant negatives, PKB-K179M and PKB-AAA, produced comparable results (data not shown). Conversely, when we transduced cells with myristoylated and active PKB (28), we observed increased Ser-9 phosphorylation of total cellular GSK-3β (Fig. 2E) and Axin-associated GSK-3β (Fig. 2F Right), enhanced stabilization of the β-catenin-degradation complex, and accumulation of β-catenin (Fig. 2F Right).
β-Catenin is most efficiently phosphorylated by GSK-3β and targeted for ubiquitination when it is in the Axin complex. Thus, the findings shown in Fig. 2F, demonstrating that expression of PKB-AA destabilizes β-catenin despite the fact that it disrupts the Axin complex, appear at odds with this principle. Therefore, we asked whether PKB-AA-induced destabilization of β-catenin might be via an Axin/GSK-3β-independent mechanism. We exposed NRVMs that had been transduced with AdPKB-AA to either LiCl or lactacystin. Both agents blocked PKB-AA-mediated degradation of β-catenin (Fig. 2G). These data confirm that PKB-AA is acting predominantly via GSK-3β and the proteasome to destabilize β-catenin, although a minor GSK-3β-independent mechanism cannot be ruled out. The data in Fig. 2 E–G suggest that, in the presence of PKB-AA, GSK-3β is maximally activated and efficiently phosphorylates β-catenin. Thus, even the minimal amount of Axin complex evident in Fig. 2F Middle, appears to be sufficient to maintain β-catenin at low levels.
Finally, we transduced cells with adenovirus encoding GSK-3β(S9A), a mutant unable to be inhibited by Ser-9 phosphorylation. This construct, which abrogates the hypertrophic response in cardiomyocytes in culture and in vivo (2, 5), dramatically reduced the accumulation of β-catenin after PE (Fig. 2H).
In summary, the studies in NRVMs suggest that levels of β-catenin increase following hypertrophic stress, due, in large part, to GSK-3β phosphorylation on Ser-9, likely catalyzed by the recruitment of PKB to the Axin complex. Activated PKB also enhances the stability of the complex. Furthermore, the accumulation of β-catenin is sufficient to induce transcription from promoters containing Tcf/Lef elements.
Mechanisms of Stabilization of β-Catenin in Vivo.
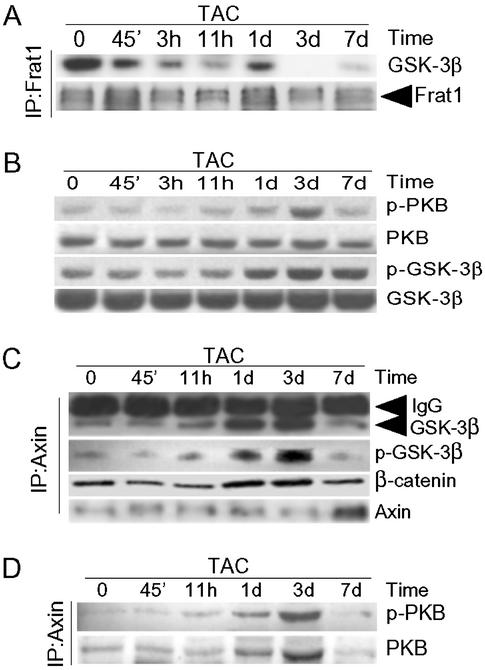
We found that the mechanisms regulating stabilization of β-catenin in the heart of the intact rat exposed to pressure overload induced by TAC were similar to those in the PE-stimulated NRVMs. TAC induced the rapid dissociation of GSK-3β from Frat1 (Fig. 3A), suggesting that sequestration of GSK-3 by Frat1 did not account for the stabilization of β-catenin at 1 and 3 d after TAC.
Figure 3.
Mechanisms of hypertrophic stress-induced stabilization of β-catenin in vivo. Rats were subjected to TAC for the times indicated. (A) Myocardial lysates were immunoprecipitated with anti-Frat1 antibody, followed by blotting with anti-GSK-3β and anti-Frat1 antibodies. (B) Myocardial lysates were blotted with antibodies to p-PKB, PKB, p-GSK-3β, or GSK-3β. (C and D) Myocardial lysates were immunoprecipitated with an anti-Axin antibody followed by blotting with antibodies to GSK-3β, p-GSK-3β, and β-catenin (C) or with anti-p-PKB or anti-PKB (D).
Phosphorylation of total cellular GSK-3β on Ser-9 increased after TAC, and the time course of the phosphorylation, which peaked 3 d after TAC, was compatible with this phosphorylation playing a role in stabilizing β-catenin (Fig. 3B). Phosphorylation of total cellular PKB followed a similar profile of activation (Fig. 3B).
We again examined signaling within the Axin complex. As with the cells in culture, only more strikingly, complex formation of GSK-3β with Axin increased markedly after TAC, peaking at 1–3 d (Fig. 3C). Furthermore, more β-catenin was present in the Axin complex after TAC (Fig. 3C), which would be expected to increase access of GSK-3β to β-catenin, leading to destabilization of β-catenin. However, at 1 and 3 d after TAC, coincident with the increase in β-catenin levels, there was a marked increase in the association of activated PKB with the complex (Fig. 3D) and a corresponding increase in phosphorylation on Ser-9 of GSK-3β (Fig. 3C). Thus, the mechanisms of stabilization of β-catenin in vivo by hypertrophic stress appear to be very similar to those in cultured cardiomyocytes.
β-Catenin Regulates Growth of Cardiomyocytes.
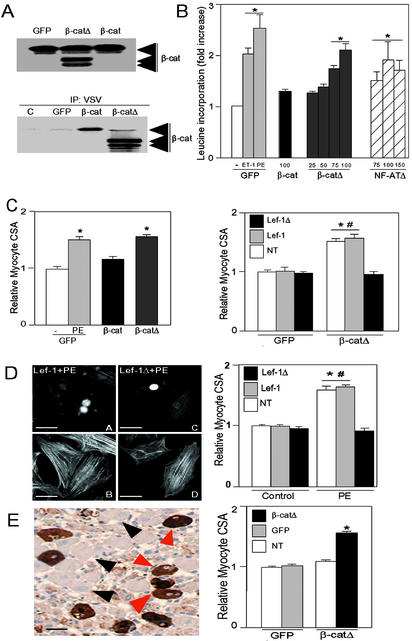
We next examined whether stabilization of β-catenin produced a phenotype in NRVMs. We transduced NRVMs with adenoviruses encoding either vesicular stomatitis virus-tagged wild-type β-catenin or β-cateninΔ, a stabilized mutant lacking the GSK-3 phosphorylation sites. Expression of the transgenes was confirmed (Fig. 4A Upper). Gene transfer with wild-type β-catenin led to only modest increases in levels of β-catenin, indicating efficient compensation by the cells (Fig. 4A Upper). Gene transfer of the stabilized mutant led to somewhat greater increases, although, compared with endogenous levels, expression was increased only ≈2- to 3-fold (Fig. 4A Upper).
Figure 4.
β-Catenin is sufficient and necessary for cardiomyocyte hypertrophy. (A) NRVMs were transduced with AdGFP (GFP), Adβ-catenin (β-cat), or Adβ-cateninΔ (β-catΔ), each with a multiplicity of infection of 100 plaque-forming units per cell. Lysates were blotted with anti-β-catenin antibody (Upper) or were subjected to immunoprecipitation with anti-vesicular stomatitis virus antibody followed by blotting with anti-β-catenin antibody (Lower). (B) NRVMs were transduced with AdGFP, Adβ-catenin, Adβ-cateninΔ, or AdNF-ATΔ at the multiplicities of infection noted. Incorporation of [3H]leucine was determined 48 h later. As a reference, [3H]leucine incorporation was determined in AdGFP-transduced cells stimulated with endothelin-1 or PE for 48 h. Values for AdGFP-transduced cells treated with vehicle were normalized to 1 (n = 5–6 experiments, each done in triplicate). *, P < 0.01 vs. AdGFP-transduced cells treated with vehicle. (C Left), β-CateninΔ induces an increase in cardiomyocyte size in vitro. NRVMs on coverslips were transduced with AdGFP, Adβ-catenin, or Adβ-cateninΔ at a multiplicity of infection of 100 plaque-forming units per cell. AdGFP-transduced cells were stimulated with PE or vehicle (-) for 48 h. Transduced cells were identified by GFP positivity, and cardiomyocyte CSA was determined on images of anti-α-actinin-stained myocytes (n = 4 experiments; ≥75 myocytes measured per experiment). *, P < 0.01 vs. AdGFP-transduced cells treated with vehicle. (Right) Inhibition of β-cateninΔ-induced hypertrophy by Lef-1Δβ-catenin. NRVMs were transduced with AdGFP or Adβ-cateninΔ and then were transfected with plasmids encoding Myc epitope-tagged wild-type Lef-1 or Lef-1Δβ-catenin. Cardiomyocyte CSA was determined 48 h later in cells successfully transfected with either Lef-1 or Lef-1Δβ-catenin (Lef-1Δ) and in cells on the same coverslips that had undergone the transfection protocol but were not successfully transfected (NT; n = 4 experiments; ≥75 myocytes measured per experiment). #, P < 0.01 vs. CSA of Adβ-catΔ-transduced cells that were transfected with Lef-1Δβ-cat. *, P < 0.01 vs. CSA of all AdGFP-transduced cells. (D) Inhibition of PE-induced hypertrophy by Lef-1Δβ-catenin. (Left) NRVMs were transfected with plasmids encoding Myc-tagged wild-type Lef-1 (A and B) or Myc-tagged Lef-1Δβ-cat (C and D). Twenty-four hours later, cells were stimulated with PE. Cardiomyocyte CSA was determined 48 h later. (A and C) Immunostained with anti-Myc tag to identify transfected cells. (B and D) The same fields stained with FITC-conjugated phalloidin. (Right) Graph compares CSA of cells successfully transfected with either Lef-1 or Lef-1Δβ-cat with cells that were not transfected (NT), after stimulation with PE or vehicle (control). Data are from four experiments; ≥75 myocytes measured per experiment. *, P < 0.01 vs. CSA of all controls. #, P < 0.01 vs. CSA of Lef-1Δβ-cat-transfected cells. (Bar, 20 μm.) (E) β-CateninΔ increased cardiomyocyte size in vivo. (Left) AdGFP or Adβ-cateninΔ were injected directly into the left ventricular myocardium of rats. Eight days, later cardiomyocyte CSA was determined on GFP-positive myocytes infected with Adβ-cateninΔ (red arrows) and adjacent nontransduced myocytes (black arrows). (Bar, 20 μm.) (Right) Graph compares CSA of cells successfully transduced with Adβ-cateninΔ, AdGFP, or cells that were not transduced (NT; n = 4 hearts per treatment group; ≥50 myocytes measured per heart). *, P < 0.01 vs. AdGFP transduced.
The essential feature of hypertrophic growth, protein synthesis (as determined by leucine incorporation), was significantly increased by gene transfer of β-cateninΔ (Fig. 4B). The increase was equivalent to that induced by gene transfer of NF-ATΔ, a positive regulator of hypertrophy (6), and to endothelin-1 and only marginally less than that with PE (Fig. 4B). These changes were not attributable to proliferation of cardiomyocytes because β-cateninΔ did not induce cell cycle entry, as determined by BrdUrd incorporation (data not shown). Another marker of hypertrophy, cardiomyocyte size, was also significantly increased by β-cateninΔ (Fig. 4C Left). Gene transfer of wild-type β-catenin increased protein synthesis and cardiomyocyte size slightly but not significantly, compatible with the minimal increase in β-catenin levels achieved (Fig. 4 B and C Left).
The prohypertrophic effects of β-cateninΔ could have been mediated via effects at the cell membrane. However, coexpression of a dominant inhibitory mutant of its transcriptional coactivator Lef-1, in which the DNA binding domain is preserved but the N-terminal 56 amino acids, containing the β-catenin interaction domain, has been deleted (Lef-1Δβ-cat; ref. 29), abrogated the β-catenin-induced increase in cell size (Fig. 4C Right). Of note, two other markers of hypertrophy in cardiomyocytes, sarcomere organization and expression of the fetal gene atrial natriuretic factor, were not induced by β-catenin (data not shown). These data confirm that β-catenin is sufficient to induce two of the critical components of the hypertrophic response, protein synthesis and cellular growth, and confirm that the effect is mediated via its transcriptional activating activity.
We next asked whether β-catenin is necessary for the hypertrophic response of cardiomyocytes to physiological stimuli. β-Catenin constructs lacking the transcriptional activation domain do not consistently function as dominant inhibitory mutants because they can increase cytosolic and nuclear levels of β-catenin, possibly by displacing endogenous wild-type β-catenin from the membrane (30). Therefore, we again expressed Lef-1Δβ-cat or wild-type Lef-1 and determined the effect on PE-induced hypertrophy. Expression of Lef-1 had no significant effect on PE-induced hypertrophy, compatible with β-catenin being the limiting factor (Fig. 4D). In contrast, expression of Lef-1Δβ-cat abrogated the PE-induced increase in cell size (Fig. 4D).
To determine whether β-catenin might have a role in cardiomyocyte growth in vivo, we performed adenoviral gene transfer in the intact rat heart. At 8 d after injection, cardiomyocytes expressing β-cateninΔ were significantly larger than those transduced with a control virus (AdGFP) or nontransduced cells (Fig. 4E). Mild inflammation was observed in myocardial sections when counterstained with hematoxylin. However, the fact that there was a significant increase in cell size in only the β-cateninΔ-transduced cardiomyocytes indicates that paracrine effects from cytokines released by inflammatory cells could not have accounted for the changes noted. These data confirm that β-catenin is both sufficient and necessary for the growth response of NRVMs to hypertrophic stimuli in vitro and sufficient to induce cardiomyocyte growth in vivo.
Discussion
β-Catenin is best known for its role in development and cancer (7). To our knowledge, its role in terminally differentiated cells or in normal tissues of adult mammals has not been explored (11). Herein, we have identified a biological role for the transactivating activity of β-catenin in terminally differentiated cells, and that role is as a regulator of hypertrophic stress-induced cardiomyocyte growth. Furthermore, the studies identify mechanisms of regulation of β-catenin in cultured cardiomyocytes and in vivo in response to stimuli other than Wnts. We found striking differences between the mechanisms regulating cytosolic β-catenin levels in cardiomyocytes exposed to hypertrophic stress and those reported previously from studies in Drosophila, Xenopus, and in mammalian cells stimulated with Wnts (11). We found that what is believed to be an important mechanism of inhibition of GSK-3 used by Wnt signaling, complex formation of GSK-3β with Frat1, was not a major factor after hypertrophic stress (21). In fact, we found that hypertrophic stress disrupted the GSK-3β/Frat1 complex, releasing GSK-3 from this inhibitor. Hypertrophic stress induced the formation of a multiprotein complex that included Axin, GSK-3β, and β-catenin. Surprisingly, the stabilization of the β-catenin-degradation complex, which is normally associated with destabilization of β-catenin (31), was associated with increased cytosolic levels (or stabilization) of β-catenin (Fig. 1 A and C). Our data demonstrating the continued presence of β-catenin in the Axin complex suggest that release of β-catenin from the complex, which occurs with Wnt signaling possibly mediated by Dishevelled (Dvl), casein kinase Iɛ/δ (31), or by GSK-3 inhibition, is not the primary mechanism contributing to its stabilization. Rather, our data suggest that the stabilization of β-catenin is achieved via Ser-9 phosphorylation of GSK-3β, and that this, in turn, occurs via recruitment of PKB to the Axin complex.
Ding et al. (17), studying regulation of β-catenin by stimuli other than Wnts, reported that PKB-induced Ser-9 phosphorylation (and inhibition) of GSK-3β in response to insulin was insufficient to stabilize β-catenin. In contrast, we have found that insulin-like growth factor-1 and insulin, which also induce hypertrophic growth in cardiomyocytes, lead to a biphasic pattern of stabilization of β-catenin that is similar to that induced by PE (data not shown). Although we have not clarified the mechanism of stabilization of β-catenin by insulin like growth factor-1, the findings suggest that the different conclusions reached by Ding et al. (17) may be because of the fact that stabilization of β-catenin by hypertrophic agonists is restricted to certain cell types, cardiomyocytes being one.
Wnts also stabilize β-catenin via inhibition of GSK-3β, but, in contrast to hypertrophic signaling, this is not believed to involve PKB-induced Ser-9 phosphorylation (16, 32). Fukumoto et al. (18) have reported Wnt-induced recruitment of PKB to the Axin complex, and, although this recruitment was not necessary for stabilization of β-catenin, it did augment Wnt-induced stabilization. Thus, hypertrophic stimuli and Wnt signaling lead to an identical outcome stabilization of β-catenin; and, although different mechanisms are used, recruitment of PKB to the Axin complex may be a common thread of both signaling systems.
The cyclin D1 gene is a known target of β-catenin (33). Although gene transfer of β-catenin led to increased cyclin D1 expression in cycling cells, cyclin D1 was not induced in cardiomyocytes (data not shown). The inability of terminally differentiated cardiomyocytes to up-regulate cyclin D1 might, in part, explain the differences in the growth responses observed between these cells and cells with the ability to proliferate, highlighting a possible point of divergence in the regulation of hyperplastic vs. hypertrophic growth potential.
It has been unclear how hypertrophic stimuli, which primarily produce transient activation (or inhibition) of cellular signaling pathways, can produce the profound and long-term alterations in gene expression that culminate in the hypertrophic response. The delayed (and prolonged) recruitment of the GSK-3β/β-catenin module demonstrated here suggests one mechanism whereby signaling networks may be sequentially recruited to allow the full expression of the hypertrophic phenotype.
We have shown that β-catenin is stabilized by hypertrophic stimuli and that inhibition of GSK-3β, via phosphorylation of Ser-9 by PKB, appears to be the mechanism by which β-catenin is stabilized. β-Catenin is both sufficient to induce hypertrophic growth when expressed in cardiomyocytes in vitro and in vivo, and its transcriptional activating activity is necessary for the hypertrophic response to physiologically relevant stimuli. These studies implicate β-catenin as a substrate of GSK-3 that regulates growth of terminally differentiated cells and identify a new Wnt-independent mechanism of regulation of the β-catenin-degradation complex that is triggered by hypertrophic stimuli acting via members of the heterotrimeric G protein-coupled receptor superfamily.
Acknowledgments
We thank Paul Hamel, Jeff Molkentin, and Jeffrey Rubin for reagents and invaluable advice. This work was supported by the Wellcome Trust (064925/Z/01/Z to S.H.), a Scientist Development Grant and Established Investigator Award from the American Heart Association, and the National Institutes of Health (Grants HL61688 and HL67371).
Abbreviations
- GSK-3
glycogen synthase kinase-3
- NF-AT
nuclear factor of activated T cells
- Tcf
T cell factor
- Lef
lymphocyte enhancer factor
- NRVM
neonatal rat ventricular myocytes
- Wg
Wingless
- CSA
cross-sectional area
- TAC
thoracic aortic constriction
- PE
phenylephrine
References
- 1.He X, Saint-Jeannet J P, Woodgett J R, Varmus H E, Dawid I B. Nature. 1995;374:617–622. doi: 10.1038/374617a0. [DOI] [PubMed] [Google Scholar]
- 2.Haq S, Choukroun G, Kang Z B, Lee K-H, Ranu H, Matsui T, Rosenzweig A, Alessandrini A, Molkentin J D, Woodgett J, et al. J Cell Biol. 2000;151:117–129. doi: 10.1083/jcb.151.1.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Morisco C, Zebrowski D, Condorelli G, Tsichlis P, Vatner S F, Sadoshima J. J Biol Chem. 2000;275:14466–14475. doi: 10.1074/jbc.275.19.14466. [DOI] [PubMed] [Google Scholar]
- 4.Badorff C, Ruetten H, Mueller S, Stahmer M, Gehring D, Jung F, Ihling C, Zeiher A M, Dimmeler S. J Clin Invest. 2002;3:373–381. doi: 10.1172/JCI13779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Antos C L, McKinsey T A, Frey N, Kutschke W, McAnally J, Shelton J M, Richardson J A, Hill J A, Olson E N. Proc Natl Acad Sci USA. 2002;99:907–912. doi: 10.1073/pnas.231619298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Molkentin J D, Lu J R, Antos C L, Markham B, Richardson J, Robbins J, Grant S R, Olson E N. Cell. 1998;93:215–228. doi: 10.1016/s0092-8674(00)81573-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bienz M, Clevers H. Cell. 2000;103:311–320. doi: 10.1016/s0092-8674(00)00122-7. [DOI] [PubMed] [Google Scholar]
- 8.Miller J R, Moon R T. Genes Dev. 1996;10:2527–2539. doi: 10.1101/gad.10.20.2527. [DOI] [PubMed] [Google Scholar]
- 9.Sadot E, Simcha I, Shututman M, Ben-Ze'ev A, Geiger B. Proc Natl Acad Sci USA. 1998;95:15339–15344. doi: 10.1073/pnas.95.26.15339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Aberle H, Bauer A, Stappert J, Kispert A, Kemler R. EMBO J. 1997;16:3797–3804. doi: 10.1093/emboj/16.13.3797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Polakis P. Cell. 2001;105:563–566. doi: 10.1016/s0092-8674(01)00379-8. [DOI] [PubMed] [Google Scholar]
- 12.Frame S, Cohen P. Biochem J. 2001;359:1–16. doi: 10.1042/0264-6021:3590001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Stambolic V, Woodgett J R. Biochem J. 1994;303:701–704. doi: 10.1042/bj3030701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cross D A, Alessi D R, Cohen P, Andjelkovich M, Hemmings B A. Nature. 1995;378:785–789. doi: 10.1038/378785a0. [DOI] [PubMed] [Google Scholar]
- 15.Dajani R, Fraser E, Roe S M, Young N, Good V, Dale T C, Pearl L H. Cell. 2001;105:721–732. doi: 10.1016/s0092-8674(01)00374-9. [DOI] [PubMed] [Google Scholar]
- 16.Frame S, Cohen P, Biondi R M. Mol Cell. 2001;7:1321–1327. doi: 10.1016/s1097-2765(01)00253-2. [DOI] [PubMed] [Google Scholar]
- 17.Ding V W, Chen R H, McCormick F. J Biol Chem. 2000;275:32475–32481. doi: 10.1074/jbc.M005342200. [DOI] [PubMed] [Google Scholar]
- 18.Fukumoto S, Hsieh C-M, Maemura K, Layne M D, Ye S-F, Lee K-H, Matsui T, Rosenzweig A, Taylor W G, Rubin J S, et al. J Biol Chem. 2001;276:17479–17483. doi: 10.1074/jbc.C000880200. [DOI] [PubMed] [Google Scholar]
- 19.Harwood A J. Cell. 2001;105:821–824. doi: 10.1016/s0092-8674(01)00412-3. [DOI] [PubMed] [Google Scholar]
- 20.Liu C, Li Y, Semenov M, Han C, Baeg G-H, Tan Y, Zhang Z, Lin X, He X. Cell. 2002;108:837–847. doi: 10.1016/s0092-8674(02)00685-2. [DOI] [PubMed] [Google Scholar]
- 21.Yost C, Farr G H, Pierce S B, Ferkey D M, Chen M M, Kimelman D. Cell. 1998;93:1031–1041. doi: 10.1016/s0092-8674(00)81208-8. [DOI] [PubMed] [Google Scholar]
- 22.Li L, Yuan H, Weaver C D, Mao J, Farr G H, Sussman D J, Jonkers J, Kimelman D, Wu D. EMBO J. 1999;18:4233–4240. doi: 10.1093/emboj/18.15.4233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Morin P J, Sparks A B, Korinek V, Barker N, Clevers H, Vogelstein B, Kinzler K W. Science. 1997;275:1787–1790. doi: 10.1126/science.275.5307.1787. [DOI] [PubMed] [Google Scholar]
- 24.He T C, Zhou S, Da Costa L T, Yu J, Kinzler K W, Vogelstein B. Proc Natl Acad Sci USA. 1998;95:2509–2514. doi: 10.1073/pnas.95.5.2509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ruel L, Stambolic V, Ali A, Manoukian A S, Woodgett J R. J Biol Chem. 1999;274:21790–21796. doi: 10.1074/jbc.274.31.21790. [DOI] [PubMed] [Google Scholar]
- 26.Choukroun G, Hajjar R, Fry S, del Monte F, Haq S, Guerrero J L, Picard M, Rosenzweig A, Force T. J Clin Invest. 1999;104:391–398. doi: 10.1172/JCI6350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Uren A, Reichsman F, Anest V, Taylor W G, Muraiso K, Bottaro D P, Cumberledge S, Rubin J S. J Biol Chem. 2000;275:4374–4382. doi: 10.1074/jbc.275.6.4374. [DOI] [PubMed] [Google Scholar]
- 28.Songyang Z, Baltimore D, Cantley L C, Kaplan D R, Franke T. Proc Natl Acad Sci USA. 1997;94:11345–11350. doi: 10.1073/pnas.94.21.11345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hsu S-C, Galceran J, Grosschedl R. Mol Cell Biol. 1998;18:4807–4818. doi: 10.1128/mcb.18.8.4807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zhurinsky J, Shtutman M, Ben-Ze'ev A. Mol Cell Biol. 2000;20:4238–4252. doi: 10.1128/mcb.20.12.4238-4252.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gao Z H, Seeling J M, Hill V, Yochum A, Virshup D M. Proc Natl Acad Sci USA. 2002;99:1182–1187. doi: 10.1073/pnas.032468199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Yuan H, Mao J, Wu D. J Biol Chem. 1999;274:30419–30423. doi: 10.1074/jbc.274.43.30419. [DOI] [PubMed] [Google Scholar]
- 33.Tetsu O, McCormick F. Nature. 1999;398:422–426. doi: 10.1038/18884. [DOI] [PubMed] [Google Scholar]