Abstract
Production of extended-spectrum β-lactamases (ESBLs) is an important mechanism of β-lactam resistance in Enterobacteriaceae. Identification of ESBLs based on phenotypic tests is the strategy most commonly used in clinical microbiology laboratories. The Phoenix ESBL test (BD Diagnostic Systems, Sparks, Md.) is a recently developed automated system for detection of ESBL-producing gram-negative bacteria. An algorithm based on phenotypic responses to a panel of cephalosporins (ceftazidime plus clavulanic acid, ceftazidime, cefotaxime plus clavulanic acid, cefpodoxime, and ceftriaxone plus clavulanic acid) was used to test 510 clinical isolates of Escherichia coli, Klebsiella pneumoniae, Klebsiella oxytoca, Proteus mirabilis, Providencia stuartii, Morganella morganii, Enterobacter aerogenes, Enterobacter cloacae, Serratia marcescens, Citrobacter freundii, and Citrobacter koseri. Of these isolates, 319 were identified as ESBL producers, and the remaining 191 were identified as non-ESBL producers based on the results of current phenotypic tests. Combined use of isoelectric focusing, PCR, and/or DNA sequencing demonstrated that 288 isolates possessed blaTEM-1- and/or blaSHV-1-derived genes, and 28 had a blaCTX-M gene. Among the 191 non-ESBL-producing isolates, 77 isolates produced an AmpC-type enzyme, 110 isolates possessed TEM-1, TEM-2, or SHV-1 β-lactamases, and the remaining four isolates (all K. oxytoca strains) hyperproduced K1 chromosomal β-lactamase. The Phoenix ESBL test system gave positive results for all the 319 ESBL-producing isolates and also for two of the four K1-hyperproducing isolates of K. oxytoca. Compared with the phenotypic tests and molecular analyses, the Phoenix system displayed 100% sensitivity and 98.9% specificity. These findings suggest that the Phoenix ESBL test can be a rapid and reliable method for laboratory detection of ESBL resistance in gram-negative bacteria.
Among members of the family Enterobacteriaceae, extended-spectrum β-lactamase (ESBL) production is the most important mechanism of resistance to β-lactam antimicrobial agents, and there are an increasing number of reports of the clinical failure of these drugs, especially in high-risk wards, such as intensive care units (1, 17). ESBLs hydrolyze broad-spectrum cephalosporins and monobactams, including oxyimino-cephalosporins (expanded-spectrum cephalosporins, e.g., cefpodoxime, ceftazidime, cefotaxime, or ceftriaxone), as well as older β-lactam drugs; in addition, they are inhibited by clavulanic acid (CA) (2). The vast majority of ESBLs are derivatives of the classic TEM and SHV β-lactamases with one or more amino acid substitutions (5). This explains the extended substrate profile of ESBLs compared with that of parental enzymes (5, 9, 27) and the variety of in vitro antimicrobial susceptibility patterns observed in several gram-negative species (11).
Identification of ESBL-producing isolates based on phenotypic traits may be difficult because resistance to the extended-spectrum cephalosporins and aztreonam of many strains producing ESBLs is not readily detectable with routine susceptibility tests based on the current National Committee for Laboratory Standards (NCCLS) breakpoints (13, 15). The NCCLS thus recommends the use of ESBL breakpoints for the indicator drugs (expanded-spectrum cephalosporins, e.g., ceftazidime or cefpodoxime) to screen for potential ESBL production in Escherichia coli and Klebsiella sp. Isolates for which the MICs of ceftazidime or those of cefpodoxime are ≥2 or ≥8 μg/ml, respectively, should then be confirmed as ESBL-producing strains by phenotypic testing based on synergy between CA and extended-spectrum cephalosporins, ceftazidime, and cefotaxime, and then reported as resistant for all penicillins, cephalosporins, and aztreonam if this is found (22). However, in some microorganisms, additional mechanisms of resistance, such as AmpC-type enzymes, porin changes, and TEM and SHV β-lactamases with reduced affinities for β-lactamases inhibitors, can mask CA inhibition (30). In addition, other ESBLs, such as CTX-M and some OXA β-lactamases are also inhibited by CA (2, 26).
The sensitivity of ESBL screening can be improved by testing isolates against more than one antimicrobial agent, although positive results must still be verified with the confirmatory tests mentioned above. Recently, these procedures have been revised and validated by Steward et al. (30) although CA inhibition was ultimately observed for only 83.5% of the Klebsiella pneumoniae isolates initially identified as presumptive ESBL producers. In this study, an algorithm based on phenotypic methods was suggested for evaluation of such isolates. In 1999, Thomson et al. (32) demonstrated the utility of using a panel of various β-lactam drugs, alone and in combination with CA, for detection of important β-lactamases in members of the family Enterobacteriaceae, emphasizing the fact that no single drug alone can discriminate between ESBLs and high-AmpC β-lactamases.
In this study, we evaluated the reliability of the Phoenix ESBL test, a recently developed automated system for detecting ESBLs in gram-negative bacteria, on a large number of clinical isolates of Enterobacteriaceae. Isolates were characterized as ESBL and non-ESBL producers based on phenotypic responses to five extended-spectrum cephalosporins, alone or in combination with CA inhibitor. The results were then compared with those obtained with ESBL phenotypic tests and molecular analyses.
MATERIALS AND METHODS
Study isolates.
The study was conducted on 510 nonrepetitive clinical isolates of Enterobacteriaceae, many of which had been collected as part of a previously published nationwide survey (29) (Table 1). The organisms were selected as suspected ESBL producers based on findings of resistance or reduced susceptibility to one or more extended-spectrum cephalosporins and/or ampicillin in routine susceptibility studies using the standard broth microdilution method (20). The study isolates were identified with the VITEK 2 system (bioMérieux, Marcy l'Etoile, France) and/or the API 20E system (bioMérieux).
TABLE 1.
Distribution of bacterial strains studied according to β-lactamase production
| Species (no. of strains) | No. of strains producing:
|
|||
|---|---|---|---|---|
| ESBL | AmpC | High K1 β-lactamase | Other β-lactamases | |
| E. coli (88) | 48 | 0 | 0 | 40 |
| K. pneumoniae (158) | 118 | 0 | 0 | 40 |
| K. oxytoca (18) | 14 | 0 | 4 | 0 |
| P. mirabilis (106) | 76 | 0 | 0 | 30 |
| P. stuartii (26) | 17 | 9 | 0 | 0 |
| M. morganii (12) | 3 | 9 | 0 | 0 |
| E. aerogenes (44) | 18 | 26 | 0 | 0 |
| E. cloacae (23) | 8 | 15 | 0 | 0 |
| S. marcescens (6) | 1 | 5 | 0 | 0 |
| C. freundii (21) | 12 | 9 | 0 | 0 |
| C. koseri (8) | 4 | 4 | 0 | 0 |
| Total (510) | 319 | 77 | 4 | 110 |
Quality control strains (E. coli ATCC 25922 and ATCC 35218, Pseudomonas aeruginosa ATCC 27853, and K. pneumoniae ATCC 700603) were included.
Double-disk synergy test and E-test.
The double-disk synergy test was performed by a standard disk diffusion assay on Mueller-Hinton agar (Oxoid, Milan, Italy) (10). Disks containing aztreonam, ceftazidime, cefepime, ceftriaxone, and cefotaxime (30 μg each) were placed at variable distances(20 to 30 mm from center to center, depending on the species) around a disk containing amoxicillin (20 μg) plus CA (10 μg). Enhancement of the inhibition zone toward the amoxicillin-plus-CA disk was considered suggestive of ESBL production. The synergistic activity of CA with both ceftazidime and cefotaxime was confirmed by means of E-test special strips (AB Biodisk, Solna, Sweden) containing ceftazidime or ceftazidime plus CA and cefotaxime or cefotaxime plus CA. Results were interpreted according to NCCLS criteria only for the isolates of E. coli and Klebsiella sp. (21).
Phoenix ESBL test.
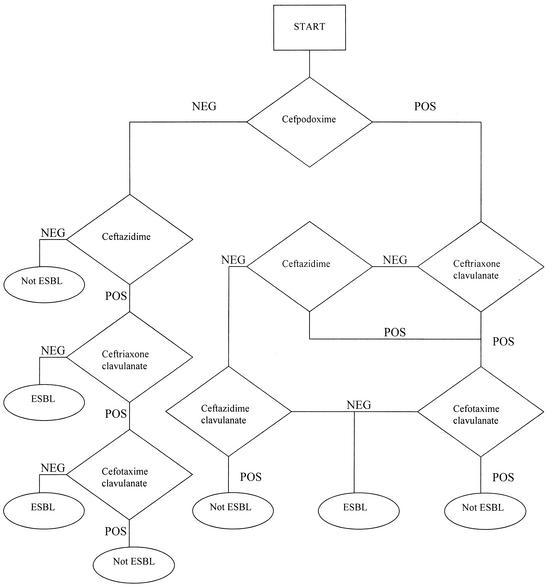
The Phoenix ESBL test used five wells containing the fixed concentrations of the following drugs or drug combinations: cefpodoxime, ceftazidime, ceftazidime plus CA, cefotaxime plus CA, and ceftriaxone plus CA. After inoculation with each of the isolates, the panel was placed in the instrument and continually monitored for growth. Results were interpreted according to the algorithm shown in Fig. 1. At each decision point, the growth curve derived from each well was evaluated. Growth curves were evaluated based on a series of functions describing their intensity and shape. A series of mathematical functions was used to determine a positive or negative growth response to a predetermined threshold; when the decision point was at terminal node, the results were reported.
FIG. 1.
Phoenix ESBL test algorithm. Abbreviations: POS, positive growth response to a predetermined threshold at 6 h; NEG, negative growth response to a predetermined threshold at 6 h.
Molecular characterization of β-lactamases.
Preliminary characterization of β-lactamases was performed by isoelectric focusing (IEF) (18, 33). Crude preparations of β-lactamases from isolates were extracted from 100 ml of Trypticase soy broth culture subjected to five cycles of freezing and thawing followed by centrifugation. The β-lactamase pIs were determined by IEF with a Bio-Rad (Hercules, Calif.) apparatus on prepared Ampholine PAG plate gels (pH 3.5 to 9.5; Bio-Rad). Gels were developed with 0.5 mM nitrocefin (Oxoid). Extracts from strains producing TEM-1, TEM-2, and SHV-1 β-lactamases were used as standards for pIs of 5.4, 5.6, and 7.6, respectively. TEM and SHV β-lactamase genes were amplified by PCR with primers 5′-ATGAGTATTCAACATTTCCGTG (nucleotides [nt] 1 to 23, numbered from the start of the enzyme-coding region) and 5′-TTACCAATGCTTAATCAGTGAG (nt 861 to 840) for blaTEM, and with 5′-ATGCGTTATATTCGCCTGTG (nt 1 to 20) and 5′-TTAGCGTTGCCAGTGCTC (nt 864 to 847) for blaSHV. PCR conditions included a 3-min denaturation at 94°C; 35 cycles of 94°C for 45 s, 59°C for 1 min, and 72°C for 1 min 30 s; and a final extension period of 72°C for 10 min. Isolates that were PCR-negative for TEM and SHV genes were subjected to PCR to amplify blaOXA, blaCTX-M, blaOXY, and ampC-type genes using separate sets of primers and conditions, as described elsewhere (8, 26, 30).
Sequencing of TEM, SHV, and CTX-M genes.
The PCR blaSHV, blaTEM and blaCTX-M products were purified with the PCR Clean Up kit (Roche Diagnostic Molecular Biochemicals, Monza, Italy), cloned in a pCR II vector (Advantage PCR cloning kit; Clontech, Palo Alto, Calif.), and sequenced with an ABI Prism 377 Sequencer Analyzer (Applied Biosystems, Foster City, Calif.). Sequences on both strands were analyzed with DNASIS software for Windows, version 2.1 (Hitachi Software Genetic Systems, San Francisco, Calif.) and confirmed by two independent PCR experiments.
RESULTS
Initial classification of the isolates as ESBL positive or negative.
Double-disk synergy test was used for preliminary classification of the isolates as ESBL positive or negative. Synergy between CA and at least one of the tested β-lactams was observed for 319 of the 510 isolates studied. The E-test method confirmed ESBL production in 180 (E. coli, K. pneumoniae, and Klebsiella oxytoca) of the 319 isolates as evidenced by ratios of the MIC of ceftazidime and/or cefotaxime to the MIC of ceftazidime and/or cefotaxime plus CA of ≥8, according to NCCLS criteria (21). For other 80 isolates (Proteus mirabilis and Citrobacter koseri), E-test results suggested the presence of an ESBL. All 191 double-disk-negative isolates had MIC ratios less than 8 and were thus considered non-ESBL producers.
IEF and PCR testing.
The β-lactamases produced by the 319 isolates identified as ESBL positive were characterized by IEF and PCR. Most of these isolates (288 of 319) showed electrofocusing bands in the typical regions of TEM and/or SHV enzymes (pI 5.2 to 6.5 and pI 7.0 to 8.2, respectively), and these findings were consistent with the results of PCR, which revealed the presence of blaTEM, blaSHV, or both in all 288 isolates (Table 2). Twenty-eight of the 31 remaining non-TEM/non-SHV isolates presenting an IEF band with a pI of >8 contained a blaCTX-M gene, as shown by PCR. All these isolates possessed a CTX-M-1 enzyme. Three isolates were repeatedly negative for blaCTX-M and blaOXA genes, and the identity of these β-lactamases is still under investigation (Table 2).
TABLE 2.
TEM, SHV, and CTX-M genes in ESBL-producing isolates
| Species | ESBL type (no. of genes)
|
Total no. of genes | |||
|---|---|---|---|---|---|
| TEM | SHV | TEM and SHV | Non-TEM or non-SHV | ||
| E. coli | TEM-20 (5) | SHV-5 (2) | TEM-1/SHV-5 (7) | CTX-M-1 (11) | 48 |
| TEM-60 (3) | SHV-12 (18) | TEM-1/SHV-12 (2) | |||
| K. pneumoniae | TEM-20 (2) | SHV-5 (25) | TEM-52/SHV-1 (4) | 118 | |
| TEM-26 (5) | SHV-12 (70) | TEM-1/SHV-5 (10) | |||
| TEM-52 (2) | |||||
| K. oxytoca | TEM-26 (6) | SHV-12 (3) | TEM-1/SHV-12 (2) | 3a | 14 |
| P. mirabilis | TEM-20 (3) | 76 | |||
| TEM-26 (4) | |||||
| TEM-47 (2) | |||||
| TEM-52 (66) | |||||
| TEM-87 (1) | |||||
| P. stuartii | TEM-52 (14) | TEM-52/SHV-1 (3) | 17 | ||
| M. morganii | TEM-43 (1) | SHV-5 (2) | 3 | ||
| E. aerogenes | TEM-26 (1) | SHV-12 (4) | CTX-M-1 (10) | 18 | |
| TEM-52 (3) | |||||
| E. cloacae | TEM-20 (5) | SHV-12 (3) | 8 | ||
| S. marcescens | TEM-12 (1) | 1 | |||
| C. freundii | TEM-26 (1) | SHV-12 (4) | CTX-M-1 (7) | 12 | |
| C. koseri | TEM-20 (3) | SHV-12 (1) | 4 | ||
| Total | 128 | 132 | 28 | 31 | 319 |
Unidentified genes.
Among the 191 isolates classified as non-ESBL producers on the basis of negative synergy test results (Table 1), there were 4 isolates of K. oxytoca that were resistant or less susceptible to cefuroxime, ceftriaxone, cefotaxime, and aztreonam but susceptible to ceftazidime and carbapenems, suggesting hyperproduction of K1 β-lactamase enzyme. In IEF, each of the isolates presented multiple β-lactamase bands with a single major band at pI 5.2, 5.6, 5.9, or 6.1. When these isolates were tested by PCR, a 155-bp amplicon was observed, consistent with the presence of the OXY-2 subtype enzyme (8). Within the group of non-ESBL producers (Table 1), 77 isolates (the majority of which were Enterobacter spp. and Citrobacter freundii strains) for which the MICs of cefoxitin were ≥32 μg/ml showed IEF bands with pIs of ≥8.3 and were PCR positive for the ampC-type gene, indicating the presence of an AmpC-type β-lactamase. As for the remaining 110 isolates (E. coli, K. pneumoniae, and P. mirabilis strains) (Table 1), whose MIC patterns suggested the presence of Bush group 2b enzymes, IEF findings were compatible with the production of TEM-1 (pI 5.4), TEM-2 (pI 5.6), and SHV-1 (pI 7.6) (25). Sequencing of PCR products obtained from these 110 isolates using oligonucleotide primers for blaTEM and blaSHV demonstrated that all produced a TEM or SHV ancestral β-lactamase.
Phoenix ESBL testing.
The performance of the Phoenix ESBL test was evaluated on all 510 isolates included in the study. This system identified 321 isolates as ESBL producers, including 319 that were ESBL positive and two (K. oxytoca isolates) that were ESBL negative in synergy testing. The remaining 189 isolates were correctly identified as ESBL negative by the Phoenix test. When the test was repeated for those isolates with discordant results, the discrepancies between the two methods remained. Comparison of Phoenix results with those of the double-disk synergy test revealed 100% sensitivity and 98.9% specificity for the Phoenix test and concordant results for 99.6% of the strains tested. For the two false-positive K. oxytoca isolates, results of synergy testing had been confirmed by molecular analyses that revealed the presence of blaOXY-2 gene. As expected (based on their susceptibility profiles), these two strains were K1 enzyme hyperproducers.
DISCUSSION
β-Lactam resistance among clinical isolates is a growing problem, and the identification of Enterobacteriaceae producing ESBLs and other emerging β-lactamases will soon be imperative for clinical microbiology laboratories (2, 14). Any method used for this purpose must be capable of discriminating between the various enzymes conferring resistance to these agents, and the ideal method will provide this information rapidly. However, laboratory detection of ESBLs can be problematic, because in many cases conventional breakpoints for resistance are not reached (7, 12), and in Europe up to 35% of putative ESBL-producing Klebsiella spp. are incorrectly reported to be susceptible to expanded-spectrum cephalosporins or monobactams (1, 16). Because of their different enzymatic properties, ESBL producers vary in their capacities to hydrolyze specific β-lactam drugs. For example, organisms that produce TEM-10 and TEM-26, the most common ESBLs in the United States, have very high rates of resistance to ceftazidime and aztreonam, while the growing family of CA-sensitive CTX-M β-lactamase producers can hydrolyze cefotaxime at least 150 times more efficiently than ceftazidime (3, 4, 23). Confounding factors, such as the production of different β-lactamases by the same organism, can also lead to erroneous phenotypic conclusions. As reported by Bush (4), clinical isolates producing as many as five distinct β-lactamases have been identified, and this makes it difficult to assess the contribution of each enzyme to a given resistance phenotype. Moreover, the phenotypic response is the result not only of the hydrolytic affinity of a given enzyme for its β-lactam substrate but also of the amount of enzyme produced. For this reason, initial efforts to improve detection of ESBLs in clinical isolates have included the use of higher than standard inocula in MIC tests (7, 31). However, interpretative criteria for tests with high inocula have proved to be controversial, since they may lead to false reports of resistance (31).
ESBL production must also be differentiated from chromosomal β-lactamase (called K1 or KOXY) hyperproduction, which is encountered in some isolates of K. oxytoca (7). K1 hyperproducer organisms have a typical antibiogram characterized by resistance to aztreonam and cefuroxime, moderate resistance to ceftriaxone, and susceptibility to ceftazidime. For this reason, ceftazidime must be included in the panel of antibiotics used in routine susceptibility testing (14, 15). In addition, CTX-M type ESBLs, which are more closely related to K1 than to TEM and SHV, produce resistance to cefotaxime, ceftriaxone, and (usually) aztreonam, but, again, not to ceftazidime (15).
Considerable attention has therefore been focused on the selection of drugs to use in ESBL detection tests, and a number of approaches have been proposed (4). Currently, two NCCLS-approved methods are the procedures most widely used to detect ESBL in E. coli, K. pneumoniae, and K. oxytoca (21). In the first (screening test), ceftazidime or cefpodoxime is used as an indicator drug, and isolates with reduced susceptibilities to these drugs are considered to be resistant to all oxyimino-cephalosporins. The second (confirmatory test) detects synergy between cephalosporins and CA. The double-disk test (10), three-dimensional test (31), E-test ESBL, and, alternatively, automated susceptibility testing systems like the Vitek ESBL can be used to screen for ESBL production. The NCCLS (21) has also recommended combined-disk methods; such combined disks are marketed by Oxoid (Combination Disk), BD Diagnostics, and Mast (MAST double disk [MDD]): this approach involves the comparison of inhibition zones produced by cefotaxime (30 μg) or ceftazidime (30 μg) disks with those produced by the same drug combined with CA (10 μg). M'Zali et al. (19) compared the MDD test, standard double-disk method, and E-test ESBL on a wide range of ESBL, non-ESBL, and class 1 β-lactamase-producing isolates. MDD correctly identified 93% of ESBL producers using both agents, whereas use of the ceftazidime and cefotaxime pairs alone achieved 86 and 65.5% sensitivity, respectively. However, another combination disk method (Oxoid), which uses cefpodoxime (10 μg) as a partner agent for CA (1 μg), has been reported to detect ESBL-positive Klebsiella strains with 100% sensitivity and specificity (6). This method makes it possible to distinguish isolates with AmpC or K1 enzymes, whose cefpodoxime inhibition zone is not enhanced by CA. This is not surprising, because cefpodoxime itself has proved to be useful in ESBL screening tests. This compound has proven to be an accurate indicator of ESBL or AmpC production in isolates of E. coli or K. pneumoniae, although no single drug was able to completely discriminate between the two types of β-lactamases (28).
The automated short-incubation Phoenix system uses five cephalosporins, alone or in combination with CA (see Materials and Methods). The system is easy to use and does not require subjective interpretation of results. After the five-well panel has been manually inoculated, it is continually monitored, and final readings of growth (compared to a preset threshold) are provided after a very short period of incubation (approximately 6 h). Although both the Phoenix and Vitek ESBL tests have automated growth monitoring systems, the latter method utilizes only two antibiotics, cefotaxime and ceftazidime, alone (0.5 μg/ml) and in combination with CA (4 μg/ml) (27). The use of a more complete drug panel has been recommended by the NCCLS (20). Thomson et al. (32) demonstrated that maximum differentiation of ESBL, high AmpC, high K1, and other β-lactamase production in Enterobacteriaceae could be achieved with a panel of five indicator drugs: cefpodoxime, cefpodoxime plus CA, ceftazidime, ceftriaxone, and ceftriaxone plus sulbactam.
In the present study, which is the first evaluation of the Phoenix ESBL test, the system provided accurate results when tested against the double-disk synergy test, with 100% sensitivity and 98.9% specificity. Good discrimination between ESBL and non-ESBL producers was observed for all species tested, with a false-positive rate of 0.6% and no false negatives. The Phoenix ESBL test performed well with K. pneumoniae, E. coli, P. mirabilis, Providencia stuartii, and also with Enterobacter aerogenes, Enterobacter cloacae, Morganella morganii, Serratia marcescens, C. freundii, and C. koseri. For K. oxytoca isolates, the Phoenix panel should in theory be able to distinguish ESBL producers from high-level K1 producers, but two K1 β-lactamase-hyperproducing isolates were incorrectly identified as ESBL producers, resulting in the only two observed false-positive results. The occurrence of false-positive and false-negative results has also been reported with other ESBL detection methods (6, 27). Sanders et al. (27), for example, observed that, of the 60 strains of K. oxytoca studied, all but 1 of the 19 strains producing ESBL or high levels of non-ESBL Bush group 2be enzymes were classified as ESBL positive by the Vitek ESBL system. Unfortunately, the system did not distinguish ESBL producers from K1 hyperproducers among these isolates. As previously observed (6), this information is important for surveillance purposes as well as for patient care since it influences the choice of further antibiotics to test.
The Phoenix ESBL system proved to be capable of identifying ESBL producers within various genera of the family Enterobacteriaceae, including Enterobacter, Citrobacter, Morganella, and Serratia. These four genera were included in minor part by Sanders et al. (27) in their evaluation of the Vitek ESBL test, although the method was found to be highly sensitive and specific, when compared to the double-disk test, for detection of ESBLs in E. coli, K. pneumoniae, and K. oxytoca isolates. Thomson et al. (32) maintain that separate criteria should be used for interpreting tests with K. pneumoniae and K. oxytoca due to the presence of the K1 β-lactamase in the latter species. In the flow chart proposed by the authors for the identification of β-lactamase groups in Enterobacteriaceae isolates, Enterobacter spp./C. freundii, Serratia spp., and E. coli and K. pneumoniae were separated from each other.
In the present study, molecular analyses confirmed the results of double-disk synergy test for all of the isolates, including the two K. oxytoca isolates (K1 hyperproducers) that produced false-positive results in the Phoenix ESBL test. Considerations on the prevalence and types of ESBLs in our isolates are beyond the aim of this report since the isolates we examined were a selected population. We would like, however, to underline the relative abundance of TEM-52, especially in isolates of P. mirabilis, and SHV-12 in isolates of K. pneumoniae, which is consistent with the results of Perilli et al. (24).
Despite the introduction of very promising molecular methods (e.g., DNA probes, PCR, nucleotide sequencing, and chip technology) and the known limitations of phenotypic methods, the latter are currently considered by many the simplest and most cost-effective strategies for detection of ESBLs among gram-negative bacteria. The Phoenix ESBL test may prove to be a valid alternative to the methodologies currently used for routine evaluation of β-lactam resistance in clinical microbiology laboratories.
Acknowledgments
This work was in part supported by a Strategic Research Program 1999 grant from the Istituto di Ricovero e Cura a Carattere Scientifico (IRCCS) “Lazzaro Spallanzani,” Rome, Italy.
REFERENCES
- 1.Babini, G. S., and D. M. Livermore. 2000. Antimicrobial resistance amongst Klebsiella spp. collected from intensive care units in Southern and Western Europe in 1997-1998. J. Antimicrob. Chemother. 45:183-189. [DOI] [PubMed] [Google Scholar]
- 2.Bradford, P. A. 2001. Extended-spectrum β-lactamases in the 21st century: characterization, epidemiology, and detection of this important resistance threat. Clin. Microbiol. Rev. 14:933-951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bradford, P. A., Y. Yang, D. Sahm, I. Grope, D. Gardovska, and G. Storch. 1998. CTX-M-5, a novel cefotaxime-hydrolyzing 32 β-lactamase from an outbreak of Salmonella typhimurium in Latvia. Antimicrob. Agents Chemother. 42:1980-1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bush, K. 2001. New β-lactamases in gram-negative bacteria: diversity and impact on the selection of antimicrobial therapy. Clin. Infect. Dis. 32:1085-1089. [DOI] [PubMed] [Google Scholar]
- 5.Bush, K., G. A. Jacoby, and A. A. Medeiros. 1995. A functional classification scheme for β-lactamases and its correlation with molecular structure. Antimicrob. Agents Chemother. 39:1211-1233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Carter, M. W., K. J. Oakton, M. Warner, and D. M. Livermore. 2000. Detection of extended-spectrum β-lactamases in klebsiellae with the Oxoid combination disk method. J. Clin. Microbiol. 38:4228-4232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Essack, S. Y. 2000. Laboratory detection of extended-spectrum β-lactamases (ESBLs)-the need for a reliable, reproducible method. Diagn. Microbiol. Infect. Dis. 37:293-295. [DOI] [PubMed] [Google Scholar]
- 8.Gheorghiu, R., M. Yuan, L. M. Hall, and D. M. Livermore. 1997. Bases of variation in resistance to β-lactams in Klebsiella oxytoca isolates hyperproducing K1 β-lactamase. J. Antimicrob. Chemother. 40:533-541. [DOI] [PubMed] [Google Scholar]
- 9.Jacoby, G. A., and A. A. Medeiros. 1991. More extended-spectrum β-lactamases. Antimicrob. Agents Chemother. 35:1697-1704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jarlier, V., M. H. Nicolas, G. Fournier, and A. Philippon. 1988. Extended broad-spectrum β-lactamases conferring transferable resistance to newer β-lactam agents in Enterobacteriaceae: hospital prevalence and susceptibility patterns. Rev. Infect. Dis. 10:867-878. [DOI] [PubMed] [Google Scholar]
- 11.Jorgensen, J. H., J. D. Turnidge, and J. A. Washington. 1999. Antibacterial susceptibility tests: dilution and disk diffusion methods, p. 1526-1577. In P. R. Murray, E. J. Baron, M. A. Pfaller, F. C. Tenover, and R. H Yolken (ed.), Manual of clinical microbiology, 7th ed. American Society for Microbiology, Washington, D.C.
- 12.Katsanis, G. P., J. Spargo, M. J. Ferraro, L. Sutton, and G. A. Jacoby. 1994. Detection of Klebsiella pneumoniae and Escherichia coli strains producing extended-spectrum β-lactamases. J. Clin. Microbiol. 32:691-696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Livermore, D. M. 1995. β-Lactamases in laboratory and clinical resistance. Clin. Microbiol. Rev. 8:557-584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Livermore, D. M. 1998. β-Lactamase-mediated resistance and opportunities for its control. J. Antimicrob. Chemother. 41:25-41. [DOI] [PubMed] [Google Scholar]
- 15.Livermore, D. M., and D. F. Brown. 2001. Detection of β-lactamase-mediated resistance. J. Antimicrob. Chemother. 48:59-64. [DOI] [PubMed] [Google Scholar]
- 16.Livermore, D. M., and M. Yuan. 1996. Antibiotic resistance and production of extended-spectrum β-lactamases amongst Klebsiella spp. from intensive care units in Europe. J. Antimicrob. Chemother. 38:409-424. [DOI] [PubMed] [Google Scholar]
- 17.Luzzaro, F., M. Perilli, R. Migliavacca, G. Lombardi, P. Micheletti, A. Agodi, S. Stefani, G. Amicosante, and L. Pagani. 1998. Repeated epidemics caused by extended-spectrum beta-lactamase-producing Serratia marcescens strains. Eur. J. Clin. Microbiol. Infect. Dis. 17:629-636. [DOI] [PubMed] [Google Scholar]
- 18.Matthew, M., A. M. Harris, M. J. Marshall, and G. W. Ross. 1975. The use of analytical isoelectric focusing for detection and identification of β-lactamases. J. Gen. Microbiol. 88:169-178. [DOI] [PubMed] [Google Scholar]
- 19.M'Zali, F. H., A. Chanawong, K. G. Kerr, D. Birkenhead, and P. M. Hawkey. 2000. Detection of extended-spectrum β-lactamases in members of the family Enterobacteriaceae: comparison of the MAST DD test, the double disc and the Etest ESBL. J. Antimicrob. Chemother. 45:881-885. [DOI] [PubMed] [Google Scholar]
- 20.National Committee for Clinical Laboratory Standards. 2000. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. Approved standard M7-A5 and informational supplement M100-S10. National Committee for Clinical Laboratory Standards, Wayne, Pa.
- 21.National Committee for Clinical Laboratory Standards. 2000. Performance standards for antimicrobial disk susceptibility tests, 7th ed. Approved standard M2-A7 (M100-S10). National Committee for Clinical Laboratory Standards, Wayne, Pa.
- 22.National Committee for Clinical Laboratory Standards. 2002. Performance standards for antimicrobial susceptibility testing; twelfth informational supplement. M100-S12. National Committee for Clinical Laboratory Standards, Wayne, Pa.
- 23.Oliver, A., J. C. Perez-Diaz, T. M. Coque, F. Baquero, and R. Canton. 2001. Nucleotide sequence and characterization of a novel cefotaxime-hydrolyzing β-lactamase (CTX-M-10) isolated in Spain. Antimicrob. Agents Chemother. 45:616-620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Perilli, M., E. Dell'Amico, B. Segatore, M. R. de Massis, C. Bianchi, F. Luzzaro, G. M. Rossolini, A. Toniolo, G. Nicoletti, and G. Amicosante. 2002. Molecular characterization of extended-spectrum β-lactamases produced by nosocomial isolates of Enterobacteriaceae from an Italian nationwide survey. J. Clin. Microbiol. 40:611-614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Philippon, A., R. Labia, and G. Jacoby. 1989. Extended-spectrum β-lactamases. Antimicrob. Agents Chemother. 33:1131-1136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Saladin, M., V. T. Cao, T. Lambert, J. L. Donay, J. L. Herrmann, Z. Ould-Hocine, C. Verdet, F. Delisle, A. Philippon, and G. Arlet. 2002. Diversity of CTX-M β-lactamases and their promoter regions from Enterobacteriaceae isolated in three Parisian hospitals. FEMS Microbiol. Lett. 209:161-168. [DOI] [PubMed] [Google Scholar]
- 27.Sanders, C. C., A. L. Barry, J. A. Washington, C. Shubert, E. S. Moland, M. M. Traczewski, C. Knapp, and R. Mulder. 1996. Detection of extended-spectrum-β-lactamase-producing members of the family Enterobacteriaceae with Vitek ESBL test. J. Clin. Microbiol. 34:2997-3001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Smith Moland, E., C. C. Sanders, and K. S. Thomson. 1998. Can results obtained with commercially available MicroScan microdilution panels serve as an indicator of β-lactamase production among Escherichia coli and Klebsiella isolates with hidden resistance to expanded-spectrum cephalosporins and aztreonam? J. Clin. Microbiol. 36:2575-2579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Spanu, T., F. Luzzaro, M. Perilli, G. Amicosante, A. Toniolo, G. Fadda, and The Italian ESBL Study Group. 2002. Occurrence of extended-spectrum β-lactamases in members of the family Enterobacteriaceae in Italy: implications for resistance to β-lactams and other antimicrobial drugs. Antimicrob. Agents Chemother. 46:196-202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Steward, C. D., J. K. Rasheed, S. K. Hubert, J. W. Biddle, P. M. Raney, G. J. Anderson, P. P. Williams, K. L. Brittain, A. Oliver, J. E. McGowan, Jr, and F. C. Tenover. 2001. Characterization of clinical isolates of Klebsiella pneumoniae from 19 laboratories using the National Committee for Clinical Laboratory Standards extended-spectrum β-lactamase detection methods. J. Clin. Microbiol. 39:2864-2872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Thomson, K. S., and C. C. Sanders. 1992. Detection of extended-spectrum β-lactamases in members of the family Enterobacteriaceae: comparison of the double-disk and three-dimensional tests. Antimicrob. Agents Chemother. 36:1877-1882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Thomson, K. S., C. C. Sanders, and E. Smith Moland. 1999. Use of microdilution panels with and without 32 β-lactamase inhibitors as a phenotypic test for β-lactamase production among Escherichia coli, Klebsiella spp., Enterobacter spp., Citrobacter freundii, and Serratia marcescens. Antimicrob. Agents Chemother. 43:1393-1400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yang, Y., N. Bhachech, P. A. Bradford, B. D., Jett, D. F. Sahm, and K. Bush. 1998. Ceftazidime-resistant Klebsiella pneumoniae and Escherichia coli isolates producing TEM-10 and TEM-43 32 β-lactamases from St. Louis, Missouri. Antimicrob. Agents Chemother. 42:1671-1676. [DOI] [PMC free article] [PubMed] [Google Scholar]