Abstract
Nasopharyngeal tonsils (adenoids) are part of human nasopharynx-associated lymphoid tissue, which may play an important role in local defense against pneumococci. Recent studies with animals have suggested that several pneumococcal proteins, including CbpA and pneumolysin (Ply), may be vaccine candidates. Our recent data obtained with children suggest that antibodies to these proteins may protect against carriage. This study was performed to investigate the regulation of the T-cell-dependent antibody responses to CbpA and pneumolysin by cytokines in adenoidal immune cells from children. Adenoidal mononuclear cells (MNC) were cultured with pneumococcal concentrated culture supernatants (CCS) or recombinant proteins. Cytokine expression profiles in adenoidal MNC after antigen stimulation were analyzed by reverse transcription-PCR, protein array analysis, and an immunoassay, along with an antibody production analysis. The roles, interactions, and cellular sources of the main cytokines identified were evaluated further. Pneumococcal CCS induced production of CbpA- and Ply-specific antibodies in association with several chemokines and cytokines, including gamma interferon (IFN-γ) and interleukin-10 (IL-10) in MNC. The antibody production correlated well with the concentrations of these two cytokines. Addition of recombinant IFN-γ or IL-10 enhanced antibody production, and monoclonal antibodies to these two cytokines and T-cell depletion significantly reduced antibody production. Intracellular cytokine staining showed that T cells are a major source of IFN-γ and IL-10. Recombinant Ply and, to a lesser extent, recombinant CbpA induced significant production of IFN-γ and IL-10 in MNC. T-cell-derived IFN-γ and IL-10 may be key regulators of production of mucosal antibody to pneumococcal protein antigens in the nasopharynx and may play an important role in local protection against pneumococcal infection in children.
Streptococcus pneumoniae is an encapsulated bacterium that is associated with significant global morbidity and mortality (37). Due to the high cost and limited coverage of capsular polysaccharide-based vaccines, several candidate protein antigens are currently being studied, including choline-binding protein A (CbpA), and pneumolysin (Ply). CbpA, also called pneumococcal surface protein C (PspC) or S. pneumoniae secretory immunoglobulin A (IgA) binding protein (SpsA) (8, 14, 33), is exposed on the pneumococcal surface and can act as an adhesin (33, 39); recently, the solution structure of the adhesion domains (R1 and R2) of CbpA has been studied, which has provided insight into the mechanism by which this protein binds polymeric immunoglobulin receptor, through which pneumococci adhere and invade human cells (25, 39). Ply is produced by virtually all clinical isolates of pneumococci. Immunization with a genetically detoxified Ply derivative has been shown to protect mice against multiple serotypes of pneumococci (1).
Recent studies have demonstrated the advantage of intranasal mucosal immunization for elicitation of pneumococcal polysaccharide-specific memory responses early in the life of mice (4). Nasopharyngeal tonsils (adenoids) are mucosa-associated lymphoid tissue and are thought to be functionally related to the nasopharynx-associated lymphoid tissues (NALT) of rodents (22). As adenoids are in direct contact with local mucosal pathogens, such as the pneumococcus, adenoidal immune cells may play an important role in local immunity. We have shown previously that cells secreting antibodies to pneumococcal protein antigens are present in adenoidal mononuclear cells (MNC) isolated from children undergoing adenoidectomies (42) and that children colonized with pneumococcus tend to have lower levels of serum and salivary antibodies to CbpA and Ply than culture-negative children have, suggesting that the existing levels of systemic and local mucosal antibodies to these antigens in vivo may protect against carriage (41). In in vitro cell cultures, adenoidal B cells stimulated with pneumococcal antigens produce significant antibodies to protein antigens, including CbpA and Ply, especially in children who are colonized with pneumococcus (41). Our results have also shown that the levels of both immunoglobulin J chain-expressing and nonexpressing IgG immunocytes are increased after antigen stimulation, which suggests that adenoids can be induction sites for both mucosal and systemic antibody production (6, 41).
It is generally thought that B-cell responses to protein antigens are T cell dependent and modulated by cytokines. This study was designed to determine which cytokines are induced by pneumococcal antigens and which cytokine(s) is crucial in the regulation of production of local antibodies to pneumococcal protein antigens in nasopharngeal tonsils in children. We show here that production of antibodies to CbpA and Ply by adenoidal cells is closely correlated with the production of cytokines, especially gamma interferon (IFN-γ) and interleukin-10 (IL-10). The results suggest that these cytokines are key independent regulators of mucosal anti-pneumococcal protein antibody production in the nasopharynx and are likely to be important in local protection against pneumococci in children.
MATERIALS AND METHODS
Subjects and samples.
Adenoids were obtained from children who were 2 to 12 years old (median age, 5 years), were undergoing adenoidectomies for adenoidal hypertrophy, and were otherwise healthy at Bristol Royal Hospital for Children, Bristol, United Kingdom. Patients who were immunized against pneumococcus previously, who had received antibiotics within 2 weeks of the operation or steroids, or who had an immunodeficiency or serious infection were excluded. The study was approved by the South Bristol local research ethics committee (approval number E5142), and written informed consent was obtained in all cases.
Pneumococcal culture supernatants.
The pneumococcal strains used in this study included a standard encapsulated type 2 strain (strain D39), isogenic CbpA− and Ply− mutant strains, and two carriage strains (types 9v and 14) that were isolated from healthy children previously involved in vaccine studies (40). Pneumococcal concentrated culture supernatant (CCS) was prepared from each strain and concentrated by using previously described methods (41). The protein concentrations in the CCS were determined using the Bio-Rad protein assay according to the manufacturer's instructions (Bio-Rad, Hemel Hempstead, United Kingdom). Western blotting performed as described previously (41, 42) confirmed that there was similar expression of CbpA and Ply in the CCS derived from D39 and the carriage strains and that there was no expression of CbpA or Ply in the CCS derived from the CbpA− and Ply− mutant strains (data not shown). The CCS were subsequently used at a protein concentration of 1 μg/ml (previously determined to be an optimal dose [data not shown]) in cell stimulation experiments. In some experiments, the CCS were first treated with proteinase K, followed by boiling (41).
Isolation and culture of adenoidal mononuclear cells.
Adenoids were transported and processed, and MNC were isolated by using methods described previously (41, 42). Adenoidal MNC were washed in sterile phosphate-buffered saline (PBS) and resuspended at a concentration of 4 × 106 cells/ml in RPMI medium containing HEPES, 2 mM glutamine, 100 U/ml penicillin, 100 μg/ml streptomycin, and 10% fetal bovine serum (Sigma, Dorset, United Kingdom). Cells were cultured in 96-well culture plates (Corning Inc, Corning, NY), and cell culture supernatants were collected at predetermined times and stored at −70°C until assays for antigen-specific antibodies and cytokines were performed. In some experiments recombinant cytokines, including IL-2, IL-4, and IL-5 (Sigma, Dorset, United Kingdom) and IL-10 and IFN-γ (R&D Systems, Abingdon, United Kingdom), were added with pneumococcal CCS, and in other experiments neutralizing monoclonal antibodies against IL-10 and IFN-γ (R&D Systems) or isotype-matched control antibodies were added 1 h before stimulation with pneumococcal CCS.
T-cell depletion from adenoidal MNC.
In some experiments, adenoidal MNC were depleted of T cells by using magnetic beads according to the manufacturer's instructions before cells were cultured with stimulants. In brief, MNC were washed in PBS with 1% bovine serum albumin and incubated with human CD3-positive selection cocktail for 10 min, and this was followed by addition of magnetic beads (StemCell Technologies, Vancouver, Canada) and incubation at 4°C for 10 min. The mixture was then washed and passed through a column on a magnetic cell separator (Miltenyi Biotec, Bisley, United Kingdom). The T-cell-depleted MNC were collected, washed, and then passed through a second identical column, and phenotypes were determined by CD3/CD19 two-color monoclonal antibody immunofluorescence staining and fluorescence-activated cell sorting (FACS) analysis performed by standard procedures to confirm the purity.
Recombinant antigens and immunoassays.
Recombinant SpsA (rSpsA), recombinant CbpA (rCbpA), and recombinant Ply toxoid (rPly) proteins were used as antigen stimulants and capture antigens for assaying antigen-specific antibodies with an immunoassay. The rCbpA (consisting of amino acids 1 to 445) was expressed and purified from Escherichia coli expressing the cbpA gene (31). The original source of the gene was the encapsulated type 2 pneumococcal strain D39 (= NCTC7466). The truncated rSpsA protein SH2 (amino acids 38 to 283) (15) was expressed and purified in E. coli and was derived from pneumococcal type 1 strain (ATCC 33400) PspC2.1 (8). The truncated SpsA protein does not contain the proline-rich region of the CbpA protein which may be important for the cross-reaction between anti-CbpA and PspA antigens (8). The rPly antigen was a toxoid form of Ply with a Trp433-Phe mutation, which reduced cytolytic activity but retained antigenicity (32). The endotoxin levels of these recombinant proteins were <0.01 ng/μg of protein, as determined by the Limulus assay (BioWhittaker, Walkersville, MD). A range of concentrations of each protein were tested to determine the optimal dose for stimulation.
Immunoassays for anti-CbpA (or anti-SpsA) and anti-Ply antibodies in cell culture supernatants (day 8 after stimulation) and inhibition assays to confirm their specificities were performed as described previously (41, 42). The latter assays showed 94 to 100% specificity when 10 μg/ml of each recombinant antigen (rCbpA, rSpsA, or rPly) was added. The antigen concentrations required for 50% inhibition were in the range from 0.1 to 1 μg/ml.
RT-PCR for mRNA expression of cytokines.
RNA was isolated using Trizol (Sigma), and reverse transcription (RT)-PCR for examining mRNA expression of IL-2, IL-4, IL-5, IL-10, IL-12, and IFN-γ was performed using a one-step RT-PCR kit (QIAGEN, Crawley, United Kingdom); the manufacturers' instructions were used for both procedures. Primer sequences were obtained from previous studies (Table 1) and were synthesized by Invitrogen (Gibco Life Sciences, Paisely, United Kingdom). For each reaction, 0.4 μg RNA was used. The PCRs consisted of 30 to 39 cycles of denaturation (94°C, 1 min), annealing (55°C to 57°C, 1 min), and extension (72°C, 1 min), followed by a final extension step at 72°C for 10 min. Human glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used to ensure equal sample loading. A mock PCR (without RNA) was performed for each cytokine reaction as a negative control, and a PCR performed with RNA derived from phytohemagglutinin-stimulated adenoidal MNC was used as a positive control. Amplified PCR products were separated on 1.5% agarose gels and visualized by ethidium bromide staining, and the results were digitized using a gel documentation system (Bio-Rad, Hemel Hempstead, United Kingdom). The mean density and the area were determined for each band on the gel using Scion Image for Windows (Scion Corporation, Frederick, MD). The level of mRNA expression for each band was expressed as the mean density × area, and the ratio of expression of each cytokine to expression of GAPDH was calculated.
TABLE 1.
Primer sequences used in RT-PCR for cytokine mRNA expression
| Cytokine | Primer sequence (5′-3′) | Product size (bp) | Reference |
|---|---|---|---|
| IL-2 | ATGTACAGGATGCAACTCCTGTCTT | 457 | 8a |
| GTTAGTGTTGAGATGATGCTTTGAC | |||
| IL-4 | AGCCTCACAGAGCAGAAGACTCTG | 99 | 37a |
| CAGCCCTGCAGAAGGTTTCCTTCTC | |||
| IL-5 | GCTTCTGCATTTGAGTTTGCTAGCT | 293 | 8a |
| TGGCCGTCAATGTATTTCTTTATTAAG | |||
| IL-10 | CTTCGAGATCTCCGAGATGCCTTC | 121 | 37a |
| GGATCATCTCAGACAAGGCTTGGC | |||
| IL-12 | CCTGCTGGTGGCTGACGACAAT | 311 | 15a |
| CTTCAGCTGCAAGTTCTTGGGT | |||
| IFN-γ | GGACCCATATGTAAAAGAAGCAGA | 121 | 37a |
| TGTCACTCTCCTCTTTCCAATTCT | |||
| TNF-α | AGAGGGAAGAGTCCCCCAGGA | 81 | 37a |
| TCAGCTTGAGGGTTTGCTACAA | |||
| GAPDH | CGGATTTGGTCGTATTGG | 203 | 36a |
| AGATGGTGATGGGATTTC |
Cytokine protein array.
A human cytokine protein array (RayBio, Norcross, Ga.) which detects 79 cytokines and chemokines was used to analyze the expression profile of adenoidal MNC by following the manufacturer's instructions, with slight modifications. The MNC culture supernatants were collected after 3 days of culture with or without stimulation with pneumococcal CCS derived from type 2 pneumococcus (strain D39). Each array membrane was incubated with blocking buffer at room temperature for 30 min to block nonspecific binding. One milliliter of cell culture supernatant was incubated with each membrane at room temperature for 2 h. The membrane was incubated with a solution containing mixed biotinylated detection antibodies (anti-cytokine and anti-chemokine) for 1 h. Horseradish peroxidase-conjugated streptavidin was then added and incubated for 45 min. The membrane was washed, incubated with the ECL chemiluminescence detection agent (Amersham Bioscience, Little Chalfont, United Kingdom) for 1 min, and exposed to Hyperfilm (Amersham Bioscience), and the image was developed using an HC-110 developer (Sigma, United Kingdom). The spot images were scanned, and each spot signal was measured by using the ScanAlyze software (Eisen Lab, Berkeley, CA). The signals from negative control spots were used as the background level. The density of expression of each protein was the intensity of the spot signal after subtraction of the background level. The relative level of expression of each protein was calculated by determining the percentage of the protein spot density based on the mean density of the positive control spots in each array. An increase of ≥2-fold in the level of expression of each cytokine after CCS stimulation was considered significant.
Immunoassays for cytokines.
Cell culture supernatants (day 3 after stimulation) were assayed to determine the levels of IL-2, IL-4, IL-5, IL-10, IL-12(p70), tumor necrosis factor alpha (TNF-α), and IFN-γ using Eli-pair kits (Diaclone Research, IDS, Boldon Tyne & Wear, United Kingdom) by following the manufacturer's instructions; the lower detection limits were 15.5 pg, 1.1 pg, 7.8 pg, 4.8 pg, 6.25 pg, 12.5 pg, and 15 pg, respectively.
Intracellular staining for cytokines.
Stimulated adenoidal MNC were cultured for 5 h; then brefeldin A (10 μg/ml; Sigma, Dorset, United Kingdom) was added, and the culture was incubated for another 2 h. Cells were then harvested, washed in PBS, stained with fluorescein isothiocyanate-labeled anti-human CD3 or CD4 monoclonal antibodies, and fixed in 2% paraformaldehyde (Sigma, Dorset, United Kingdom) for 20 min. After washing, cells were permeabilized using 0.5% saponin (Sigma, Dorset, United Kingdom) in PBS, incubated with phycoerythrin-labeled monoclonal antibodies to IL-10 or IFN-γ (Caltag Medsystems, Towcester, United Kingdom) for 45 min at 4°C, and analyzed by flow cytometry (FACScan; Becton Dickinson, United Kingdom).
Statistical analysis.
The geometric mean (with the 95% confidence interval) or the arithmetic mean (with the standard deviation) were used to express antibody titers. Log transformation of data was used when data were analyzed by parametric statistical methods as follows: the significance of differences between two groups was analyzed using Student's t test, and the significance of differences between stimulated and unstimulated cell samples from a group of subjects was analyzed by paired Student's t test. A P value of <0.05 was considered to indicate statistical significance. The analyses were performed using the SPSS software, version 11.
RESULTS
Induction of T-cell-dependent production of antigen-specific antibody by pneumococcal proteins.
Coculture of adenoidal MNC with pneumococcal CCS derived from wild-type strain D39 CCS induced significant production of anti-CbpA (or anti-SpsA) (see Materials and Methods) and anti-Ply IgG antibodies (at day 8) by adenoidal MNC, but CCS from the CbpA− and Ply− mutants did not induce significant production of antibody to the deleted antigen (Table 2). The levels of antibody to SpsA detected correlated well with the levels of antibody to the CbpA antigen (r = 0.952; n = 30; P < 0.01). There were no significant differences between the levels of IgG antibody induced by the CCS from the wild type (D39) and the levels of IgG antibody induced by the CCS from carriage strains 9F and 14 (data not shown). No significant production of IgA antibodies was detected in the cell culture supernatants after antigen stimulation (data not shown).
TABLE 2.
Anti-SpsA, anti-CbpA, and anti-Ply IgG antibody levels in adenoidal MNC from 20 children after stimulation by CCS derived from wild-type (D39), Ply−, and CbpA− pneumococci
| Prepn | Level of IgG antibodies (U/ml) toa:
|
||
|---|---|---|---|
| SpsA | CbpA | Ply | |
| Wild-type CCS | 5.78 (1.19) | 5.95 (1.29) | 1.95 (0.36) |
| Ply− CCS | 5.98 (1.25) | 6.02 (1.32) | 0.58 (0.19)b |
| CbpA− CCS | 1.52 (0.31)b | 1.61 (0.33)b | 1.89 (0.34) |
| Mediumc | 1.28 (0.27) | 1.32 (0.29) | 0.49 (0.16) |
The values are means (standard deviations).
P < 0.01 compared to the data for wild-type CCS.
Bacterial culture medium only.
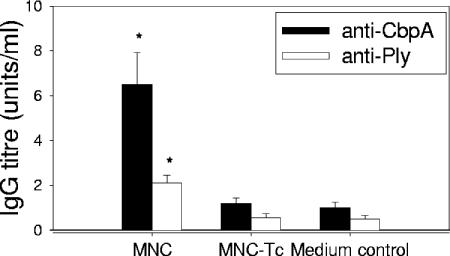
To confirm that T cells are required for the production of antigen-specific antibodies in this model, the production of anti-CbpA and anti-Ply antibodies in adenoidal MNC was measured after stimulation with pneumococcal CCS with or without prior CD3+ T-cell depletion, which significantly reduced antibody production to the baseline levels (Fig. 1).
FIG. 1.
Production of anti-CbpA and anti-Ply IgG by adenoidal MNC and T-cell-depleted MNC (MNC-Tc) stimulated with pneumococcal (D39) CCS, showing significantly lower antibody production in the latter MNC. The medium control contained only pneumococcal culture medium. The data are means and standard deviations of six experiments. An asterisk indicates that the P value is <0.01 compared to the data for T-cell-depleted MNC.
Induction of cytokines by a pneumococcal protein(s).
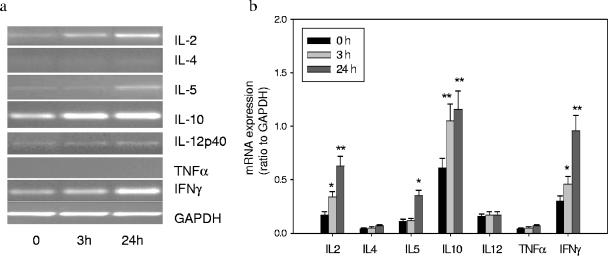
To elucidate the regulation of anti-CbpA- and anti-Ply-specific antibody responses, we investigated induction of Th1 and Th2 cytokines in adenoidal cells following stimulation by pneumococcal CCS at both the mRNA and protein levels. IL-10 mRNA was constitutively expressed, and the expression was upregulated by CCS as early as 3 h. Expression of both IFN-γ and IL-2 mRNA was upregulated significantly at 3 h and was upregulated further at 24 h. Expression of IL-5 mRNA was also increased at 24 h. No induction of IL-4 or TNF-α mRNA was observed (Fig. 2). Low levels of constitutively expressed IL-12 mRNA were detected, but no change was observed after stimulation.
FIG. 2.
Th1/Th2 cytokine mRNA expression at time zero, 3 h, and 24 h after pneumococcal CCS stimulation (1 μg/ml). (a) PCR product for each cytokine on a 1.5% agarose gel. (b) Expression of mRNA of each cytokine relative to GAPDH expression. The data are means and standard deviations of six experiments. One asterisk indicates that the P value is <0.05 compared to the data for time zero (before stimulation), and two asterisks indicate that the P value is <0.01 compared to the data for time zero (before stimulation).
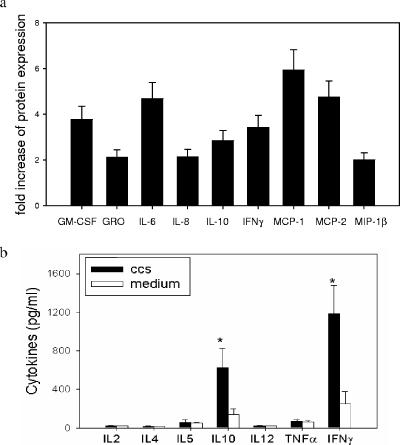
Samples from five patients, including three patients whose MNC produced significant anti-CbpA and anti-Ply antibodies and two patients whose MNC produced low levels of antibodies, were selected for analysis by the cytokine protein array. No significant baseline expression or induction of cytokine or chemokine expression (apart from IL-8 expression) was observed in MNC culture supernatants from the latter two patients (data not shown). However, significant induction of granulocyte-macrophage colony-stimulating factor, growth-related oncogene, IL-6, IL-8, IL-10, IFN-γ, monocyte chemoattractant protein 1, monocyte chemoattractant protein 2, and macrophage inflammatory protein 1β was observed in MNC cultures from the former three patients after stimulation with pneumococcal strain D39 CCS. The fold increases in the levels of expression of these cytokines after stimulation are shown in Fig. 3a. There were no significant changes in the levels of expression of other cytokines and proteins included in the array (Table 3). Further investigation of induction of chemokines by pneumococcal proteins is in progress in our laboratory.
FIG. 3.
(a) Protein array analysis of production of cytokines by adenoidal cells after stimulation by CCS derived from a wild-type pneumococcus (strain D39). The increases in the levels of protein expression of the cytokines that were upregulated significantly (≥2-fold) after stimulation are shown. The data are means and standard deviations for the level of expression based on three individual experiments (performed in duplicate) using adenoidal MNC from three subjects. GM-CSF, granulocyte-macrophage colony-stimulating factor; GRO, growth-related oncogene; MCP-1, monocyte chemoattractant protein 1; MCP-2, monocyte chemoattractant protein 2; MIP-1β, macrophage inflammatory protein 1β. (b) Concentrations of cytokines as determined by immunoassay in adenoidal MNC culture supernatants after stimulation with pneumococcal CCS. The data are means and standard deviations for cytokine concentrations (n = 30). An asterisk indicates that the P value is <0.01 compared to the data for the unstimulated medium control.
TABLE 3.
Fold increases in the levels of expression of cytokines after pneumococcal CCS stimulation (compared to medium control) in adenoidal MNC
| Cytokinea | Increase in expression (fold) | Cytokinea | Increase in expression (fold) | |
|---|---|---|---|---|
| ENA-78 | <1.5 | VEGF | <1.5 | |
| G-CSF | <1.5 | PDGF-B | <1.5 | |
| GM-CSF | 3.8 | Leptin | <1.5 | |
| GRO | 2.1 | BDNF | <1.5 | |
| GRO-α | <1.5 | BLC | <1.5 | |
| I-309 | <1.5 | Ckb 8-1 | <1.5 | |
| IL-1α | <1.5 | Eotaxin | <1.5 | |
| IL-1β | <1.5 | Eotaxin-2 | <1.5 | |
| IL-2 | <1.5 | Eotaxin-3 | <1.5 | |
| IL-3 | <1.5 | FGF-4 | <1.5 | |
| IL-4 | <1.5 | FGF-6 | <1.5 | |
| IL-5 | <1.5 | FGF-7 | <1.5 | |
| IL-6 | 4.7 | FGF-9 | <1.5 | |
| IL-7 | <1.5 | Flt-3 ligand | <1.5 | |
| IL-8 | 2.1 | Franctl kine | <1.5 | |
| IL-10 | 2.9 | GCP-2 | <1.5 | |
| IL-12 | <1.5 | GDNF | <1.5 | |
| IL-13 | <1.5 | HGF | <1.5 | |
| IL-15 | <1.5 | IGFBP-1 | <1.5 | |
| IFN-γ | 3.4 | IGFBP-2 | <1.5 | |
| MCP-1 | 5.9 | IGFBP-3 | <1.5 | |
| MCP-2 | 4.7 | IGFBP-4 | <1.5 | |
| MCP-3 | <1.5 | IL-16 | <1.5 | |
| M-CSF | <1.5 | IP-10 | <1.5 | |
| MDC | <1.5 | LIF | <1.5 | |
| MIG | <1.5 | LIGHT | <1.5 | |
| MIP-1β | 2.0 | MCP-4 | <1.5 | |
| MIP-1δ | <1.5 | MIF | <1.5 | |
| RANTES | <1.5 | MIP-3α | <1.5 | |
| SCF | <1.5 | NAP-2 | <1.5 | |
| SDF-1 | <1.5 | NT-3 | <1.5 | |
| TARC | <1.5 | NT-4 | <1.5 | |
| TGF-β | <1.5 | Osteoprotegerin | <1.5 | |
| TNF-α | <1.5 | PARC | <1.5 | |
| TNF-β | <1.5 | PIGF | <1.5 | |
| EGF | <1.5 | TGF-β2 | <1.5 | |
| IGF-1 | <1.5 | TGF-β3 | <1.5 | |
| Ang | <1.5 | TIMP-1 | 1.8 | |
| OSM | <1.5 | TIMP-2 | <1.5 | |
| Tpo | <1.5 |
G-CSF, granulocyte colony-stimulating factor; GM-CSF, granulocyte-macrophage colony-stimulating factor; GRO, growth-related oncogene; GRO-α, growth-related oncogene alpha; MCP-1, monocyte chemoattractant protein 1; MCP-2, monocyte chemoattractant protein 2; MCP-3, monocyte chemoattractant protein 3; M-CSF, macrophage colony-stimulating factor; MIP-1β, macrophage inflammatory protein 1β; MIP-1δ, macrophage inflammatory protein 1δ; TGF-β, transforming growth factor beta; EGF, epidermal growth factor; PDGF-B, platelet-derived growth factor B; FGF-4, fibroblast growth factor 4; FGF-6, fibroblast growth factor 6; FGF-7, fibroblast growth factor 7; FGF-9, fibroblast growth factor 9; HGF, hepatocyte growth factor; MCP-4, monocyte chemoattractant protein 4; MIF, migration inhibition factor; MIP-3α, macrophage inflammatory protein 3α; TGF-β2, transforming growth factor beta 2; TGF-β3, transforming growth factor beta 3. ANG, Angiogenin; BLC, B-lymphocyte chemoattractant; ENA-78, epithelial neutrophil-activating protein 78; Flt-3 ligand, fms-like tyrosine kinase-3 ligand; GCP-2, granulocyte chemotactic protein 2; GDNF, glial-derived neurotrophic factor; IGFBP, insulin-like growth factor binding proteins; IP-10, IFN-γ inducible protein 10; LIF, leukemia inhibitory factor; NDC, macrophage-derived chemokine; MIG, monokine induced by IFN-γ; NAP-2, neutrophil-activating peptide 2; OSM, oncostatin M; PARC, pulmonary and activation-regulated chemokine; P1GF, placenta growth factor; SCF, stem cell factor; SDF-1, stromal cell-derived factor; TARC, thymus and activation-regulated chemokine; TIMP, tissue inhibitor of metalloproteinases; TPO, thrombopoietin; VEGF, vascular endothelial growth factor.
To confirm these findings and to investigate further the induction of B-cell regulatory cytokines, we determined the concentrations of IL-2, IL-4, IL-5, IL-10, IL-12p70, TNF-α, and IFN-γ in supernatants from cultured adenoidal MNC with or without prior CCS stimulation. Significantly increased IFN-γ and IL-10 concentrations were observed after stimulation; in contrast, the concentrations of IL-2, IL-4, IL-5, IL-12, and TNF-α did not increase significantly (Fig. 3b). Cytokine induction by CCS was reduced by >80% by pretreatment of CCS with proteinase K and boiling (data not shown).
Production of antigen-specific antibodies is regulated by IL-10 and IFN-γ.
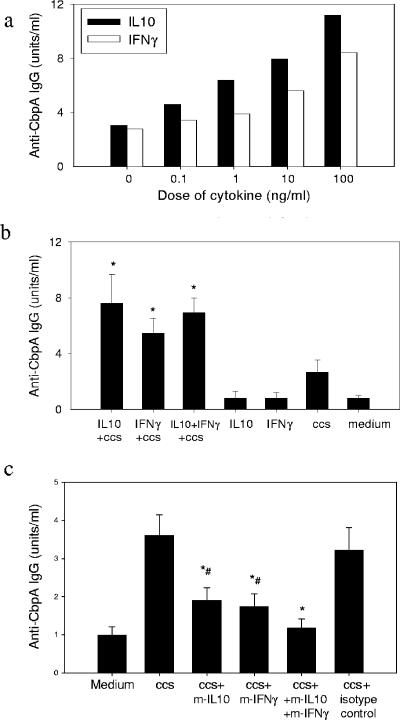
The relationship between IL-10 and IFN-γ and production of antigen-specific antibodies was studied further. For 30 subjects, there was a significant correlation between the production of anti-CbpA antibody and the concentrations of both IL-10 and IFN-γ in MNC culture supernatants after CCS stimulation (r = 0.80 and r = 0.83, respectively; P < 0.01). A similar correlation was found between anti-Ply IgG and IFN-γ and IL-10 concentrations (r = 0.68 and r = 0.73, respectively; P < 0.01). When both recombinant IL-10 and IFN-γ were added with CCS to adenoidal MNC cultures, they induced dose-dependent increases in production of anti-CbpA IgG (Fig. 4a) and anti-Ply IgG (data not shown) antibodies, but there were no significant changes in total IgG production (data not shown). Addition of these cytokines alone without CCS did not induce production of antigen-specific antibodies, suggesting that the enhancing effect is antigen dependent (Fig. 4b). There was no further significant additive effect of IFN-γ and IL-10 on antibody production when the two cytokines were mixed at submaximal doses (Fig. 4b). Coincubation of CCS with recombinant IL-2, IL-4, IL-5, or lipopolysaccharide had no effect on the antibody response (data not shown). Monoclonal neutralizing antibodies to IFN-γ and IL-10 inhibited production of anti-CbpA (Fig. 4c) and anti-Ply (data not shown) antibodies induced by CCS. When the two monoclonal antibodies were used together, the antibody production was inhibited even further (Fig. 4c).
FIG. 4.
(a) Dose-dependent increase in production of anti-CbpA IgG antibody induced by recombinant IFN-γ and IL-10 in adenoidal MNC stimulated with pneumococcal CCS. The results of one of six representative experiments are shown. (b) Effect of IFN-γ (50 ng/ml) and IL-10 (10 ng/ml) on antibody production. An asterisk indicates that the P value is <0.05 compared to the data for CCS stimulation alone. (c) Effects of monoclonal antibodies to IFN-γ (m-IFN-γ) (10 μg/ml) and IL-10 (m-IL-10) (10 μg/ml) on production of anti-CbpA IgG by adenoidal MNC. There was no inhibition of IgG production by isotype control antibodies. An asterisk indicates that the P value is <0.05 compared to the data for CCS-stimulated cells, and a number sign indicates that the P value is <0.05 compared to the data for CCS plus monoclonal antibodies to both IFN-γ and IL-10, which are not significantly different from the data for the medium control. In panels b and c the data are the means and standard deviations of six experiments.
IL-10 inhibits IFN-γ production in adenoidal cell cultures.
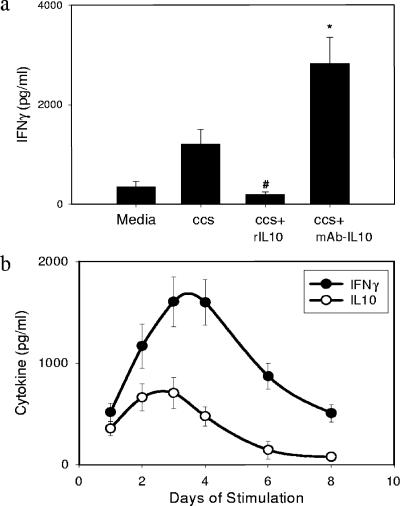
Addition of recombinant IL-10 to adenoidal MNC cultures significantly inhibited IFN-γ production induced by pneumococcal CCS, and conversely, addition of neutralizing antibody to IL-10 increased the IFN-γ concentration (Fig. 5a). However, neither addition of recombinant IFN-γ nor addition of neutralizing antibody to IFN-γ altered the concentration of IL-10 detected (data not shown).
FIG. 5.
(a) Concentrations of IFN-γ in adenoidal MNC stimulated with CCS. Recombinant IL-10 (rIL10) (10 ng/ml) inhibited IFN-γ production, and monoclonal antibody to IL-10 (mAb-IL10) (10 μg/ml) enhanced IFN-γ production. The data are means and standard deviations of eight experiments. An asterisk indicates that the P value is <0.01 compared to the data for CCS-stimulated cells. (b) Kinetics of IFN-γ and IL-10 secretion in adenoidal MNC after stimulation by CCS. The data are means and standard deviations of six experiments.
Figure 5b shows time courses for IL-10 and IFN-γ production in adenoidal MNC cultures after stimulation with pneumococcal CCS at a protein concentration of 1 μg/ml. Serial samples of supernatant from each cell culture were collected, and the culture medium was replenished daily. The IL-10 concentrations peaked at around day 2 or 3, which was slightly earlier than the IFN-γ concentrations peaked. This pattern of cytokine production is similar to the observed kinetics of mRNA expression for these two cytokines, with upregulation of IL-10 expression by CCS occurring earlier than upregulation of IFN-γ expression (Fig. 2a).
Adenoidal T cells are a major source of IFN-γ and IL-10.
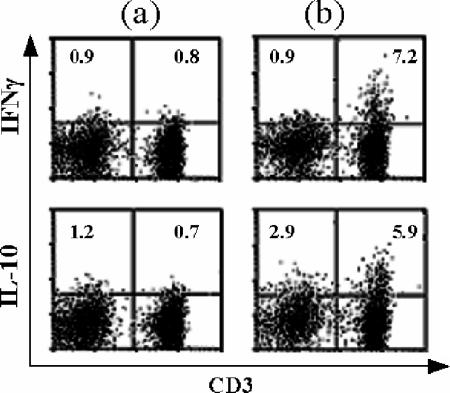
Dual staining for intracellular IFN-γ or IL-10 and cell surface markers followed by FACS analysis of lymphocytes among adenoidal MNC after CCS stimulation revealed that CD3+ T cells are a major cellular source of IFN-γ (Fig. 6), and further analysis showed that the IFN-γ-secreting cells are mainly CD4+ T cells (data not shown). CD3+ T cells are also a major source of IL-10, although the number of CD3− cells (presumably B cells) expressing IL-10 also increased after stimulation (Fig. 6).
FIG. 6.
Dual staining for intracellular IFN-γ or IL-10-phycoerythrin and CD3-flourescein isothiocyanate, followed by FACS analysis of adenoidal lymphocytes (defined by gating of typical events using forward and side scatter) after CCS stimulation. The data are the data from one of six representative experiments. (a) Medium control. (b) CCS-stimulated MNC. The values indicate the percentages of total acquired events in quadrants.
Ply and CbpA contribute to IFN-γ and IL-10 production.
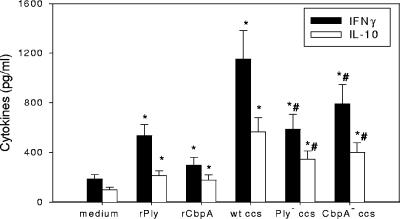
To investigate whether CbpA and Ply contribute to the induction of IFN-γ and IL-10, CCS derived from the wild-type strain (D39) and isogenic Ply− and CbpA− mutants of pneumococci and purified recombinant CbpA and Ply toxoid were cocultured with adenoidal MNC. Figure 7 shows that CCS derived from the Ply− or CbpA− mutant strains induced less production of IFN-γ and IL-10 than the wild-type CCS induced. Recombinant Ply toxoid and, to a lesser extent, recombinant CbpA induced significant production of IFN-γ and IL-10 in adenoidal MNC. No significant difference was found between recombinant CbpA and SpsA in the induction of cytokines (data not shown).
FIG. 7.
Cytokine induction in adenoidal MNC (n = 10) by purified rPly toxoid (1 μgml), rCbpA (1 μgml), and CCS (1 μgml) derived from wild-type (D39) and Ply− and CbpA− mutant pneumococci. An asterisk indicates that the P value is <0.05 compared to the data for the medium control, and a number sign indicates that the P value is <0.05 compared to the data for wild-type (wt) CCS-stimulated cells.
DISCUSSION
Effective pneumococcal protein vaccines would be easier and cheaper to manufacture than the conjugated polysaccharide vaccines currently available. Much research has been directed at exploring such antigens in animal models (7, 30). Nasal colonization rates peak in early childhood and then fall, suggesting that local immunity develops either with increasing maturity or following exposure to colonization or both. It is not clear whether this local immunity is antigen specific and, if it is, whether it is antibody mediated, cell mediated, or both. Anti-CbpA and anti-Ply IgG antibodies may protect against pneumococcal carriage in children, and adenoids may be important induction and effector sites for the production of such antibodies (41).
This is the first study of the regulation of B-cell responses to pneumococcal proteins in humans. We modeled in vitro the local production of anti-pneumococcal protein antibodies in NALT using cultures of mixed adenoidal MNC from children. The subclasses of IgG antibodies produced in this model closely resemble those seen in vivo in serum; there is a predominance of IgG1, along with lower levels of IgG3 and IgG4 and little or no IgG2 (41).
Several cytokines and also chemokines were induced in these cells following stimulation with secreted pneumococcal antigens, as expected (9, 20, 29). Among the cytokines known to have prominent B-cell regulatory functions, IL-10 and IFN-γ stood out as molecules that were consistently present and significantly induced by pneumococcal antigen stimulation, whereas IL-2, IL-4, IL-5, IL-12, and TNF-α were not generally detectable in culture supernatants (Fig. 4). In contrast, stimulation of human peripheral blood mononuclear cells with killed pneumococci has been shown to induce Th1-type cytokines, including both IFN-γ and IL-12 (2), while mice inoculated with whole pneumococci exhibit increases in expression of TNF-α, IL-1, IL-6, IL-12, IFN-γ, IL-4, and IL-10 along with evidence of effects on antibody production for several of these cytokines (21).
The relatively strong correlation between the concentrations of IL-10 and IFN-γ and the quantities of anti-CbpA and anti-Ply antibodies produced following antigenic stimulation of cells from different individuals, along with the observed dose-dependent enhancement of antibody generation by both cytokines (Fig. 4a) and inhibition by exposure of cell cultures to blocking antibodies to each cytokine (Fig. 4c), is evidence that supports a regulatory role for both of these cytokines in B-cell antibody responses in this model. Similar correlations were also observed between the concentrations of the two cytokines and the levels of anti-PspA and anti-PsaA antibodies (data not shown), suggesting that IL-10 and IFN-γ have a more generalized regulatory role in production of pneumococcal protein antigen-specific antibodies. The absence of any increase in antibody responses following stimulation with each cytokine alone in the absence of antigen (Fig. 4b) supports the hypothesis that there are specific effects on antigen-specific responses rather than nonspecific stimulation of B-cell function. The enhancing effects of the two cytokines on antibody production when they were mixed together at submaximal doses were not additive (Fig. 4b). This suggests that there are overlapping effects or interactions between the two cytokines, and an inhibitory effect of IL-10 on IFN-γ production was observed (Fig. 5a). Conversely, the combined effects of blocking antibodies to both cytokines exceeded the effects of the same antibodies used alone (Fig. 4c), suggesting that the two cytokines may modulate antigen-specific antibody production through different mechanisms or pathways. It is possible that distinct T-cell subsets are involved in the production of IFN-γ and IL-10, which may regulate antibody production independently.
The most important role that IFN-γ plays in host defense against microbial infections is thought to be its capacity to activate macrophages (28). Stimulation in vitro of adenotonsillar cells with other respiratory pathogens, such as Candida albicans and influenza virus, can enhance IFN-γ production (5). Mice lacking IFN-γ are more sensitive to infection by S. pneumoniae (36), and administration of IFN-γ has been shown to enhance survival following challenge with S. pneumoniae (38). However, a role for this cytokine in regulation of mucosal immunity to pneumococcus has not been suggested previously. Although IFN-γ is known to influence the growth and differentiation of immunoglobulin-secreting B cells (17), the only previous suggestion of a regulatory role in the production of antibodies against pneumococci was the demonstration that mice deficient in IFN-γ had reduced IgG anti-PspA responses when they were immunized with intact pneumococci (21).
Originally classified as a Th2 cytokine because it was first reported to be a product of Th2 clones, IL-10 is chiefly known as a downregulator of inflammation (13). The importance of IL-10 in the elimination of infections has become increasingly evident recently (3). IL-10 has direct effects on B cells, including prevention of apoptosis (23), enhancement of proliferation and differentiation of plasma cells (34, 35), and effects in combination with other cytokines on immunoglobulin class switching (11) (16). Its main regulatory effects appear to be effects on the differentiation and function of myeloid lineage antigen-presenting cells, including antigen presentation (10). In this study, recombinant IL-10 inhibited the generation of IFN-γ by adenoidal cell cultures, and blocking antibody to IL-10 increased IFN-γ production (Fig. 5a). The capacity of IL-10 to inhibit IFN-γ generation in Th1 cell clones is well established and was one of the first properties of IL-10 described (12). Conversely, in our model, IFN-γ and anti-IFN-γ did not alter IL-10 generation. This is also consistent with previous findings, as no such inhibitory effect has been reported previously. The earlier appearance of IL-10 RNA expression (Fig. 2a and b) and the earlier peak in IL-10 protein production (Fig. 5) than in IFN-γ production are consistent with this regulatory role for IL-10.
The factors involved in the induction of IFN-γ and IL-10 by pneumococcal CCS in this model are not clear at present. Treatment of CCS by proteinase K followed by boiling significantly reduced cytokine production, which suggests that a protein(s) may be important in the induction of these cytokines. The results of stimulation of adenoidal MNC using wild-type CCS compared to the results of stimulation of adenoidal MNC using CCS derived from Ply− or CbpA− pneumococci and purified recombinant Ply and CbpA antigens suggest that Ply and, to a lesser extent, CbpA contribute to the induction of IFN-γ and IL-10. However, our data do not rule out the possibility that other protein antigens, such as PspA and PsaA, may also contribute to cytokine induction to some extent.
Our data also suggest that T cells are a significant source of both IL-10 and IFN-γ (Fig. 6) in this system and that antibody responses are T cell dependent (Fig. 1). Thus, T cells may exert their effects on anti-pneumococcal B-cell function in NALT in part through release of these cytokines. Following stimulation, the presence of CD3− IL-10+ cells with light-scattering properties of lymphocytes, probably B cells, is consistent with recent reports of cytokine-expressing regulatory B cells (24). However, adherent macrophages may also contribute to IL-10 production (3) in such cell cultures.
The predominant production of IFN-γ in adenoidal MNC after pneumococcal antigen stimulation suggests that there is a Th1-type response. The transient disappearance of T cells with type 1 cytokine profiles in patients with invasive pneumococcal infections (19) has been interpreted as being indicative of the importance of these cells in the immune response to this infection.
This study provides the first information on the nature of cytokine regulation of acquired mucosal immune responses to pneumococcus in humans. Very recent studies with mice identified a critical role for T cells in protection against pneumococcal infection (18, 26). Our data show that there is an absolute requirement for T cells in the elaboration of anti-CbpA and anti-Ply antibodies, perhaps in part through the release of IFN-γ and IL-10 induced by pneumococcal protein antigens. In addition to regulation of B cells in the production of antigen-specific antibodies, the release of cytokines such as IFN-γ by CD4+ T cells in human NALT shown in this study could also contribute directly to antibody-independent T-cell-mediated immunity against carriage or infection, as suggested by Malley et al. (26) and McCool and Weiser (27), who used mouse models. The balance between the Th1 cytokine IFN-γ and the potent regulatory cytokine IL-10 may be crucial in specific mucosal immunity against pneumococcal infection in children.
Acknowledgments
This work was supported by the David Telling Charitable Trust, the Special Trustees of the United Bristol Healthcare Trust, United Kingdom, and the North Bristol NHS Trust Research Foundation, United Kingdom.
We are grateful to members of the ENT departments of Bristol Royal Hospital for Children and Southmead Hospital for assisting with the collection of adenoid tissues and to the patients and their families who took part in the study. We also thank Jeffrey Weiser of the University of Pennsylvania, Philadelphia, Pa., for kindly providing pneumococcal strain D39 and the CbpA-deficient mutant and Howard Jenkinson of the University of Bristol, Bristol, United Kingdom, for critical reading of the manuscript.
Editor: J. N. Weiser
REFERENCES
- 1.Alexander, J. E., R. A. Lock, C. C. Peeters, J. T. Poolman, P. W. Andrew, T. J. Mitchell, D. Hansman, and J. C. Paton. 1994. Immunization of mice with pneumolysin toxoid confers a significant degree of protection against at least nine serotypes of Streptococcus pneumoniae. Infect. Immun. 62:5683-5688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Arva, E., and B. Andersson. 1999. Induction of phagocyte-stimulating and Th1-promoting cytokines by in vitro stimulation of human peripheral blood mononuclear cells with Streptococcus pneumoniae. Scand. J. Immunol. 49:417-423. [DOI] [PubMed] [Google Scholar]
- 3.Asadullah, K., W. Sterry, and H. D. Volk. 2003. Interleukin-10 therapy—review of a new approach. Pharmacol. Rev. 55:241-269. [DOI] [PubMed] [Google Scholar]
- 4.Bjarnarson, S. P., H. Jakobsen, G. Del Giudice, E. Trannoy, C. A. Siegrist, and I. Jonsdottir. 2005. The advantage of mucosal immunization for polysaccharide-specific memory responses in early life. Eur. J. Immunol. 35:1037-1045. [DOI] [PubMed] [Google Scholar]
- 5.Boyaka, P. N., P. F. Wright, M. Marinaro, H. Kiyono, J. E. Johnson, R. A. Gonzales, M. R. Ikizler, J. A. Werkhaven, R. J. Jackson, K. Fujihashi, S. Di Fabio, H. F. Staats, and J. R. McGhee. 2000. Human nasopharyngeal-associated lymphoreticular tissues. Functional analysis of subepithelial and intraepithelial B and T cells from adenoids and tonsils. Am. J. Pathol. 157:2023-2035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Brandtzaeg, P. 2003. Immunology of tonsils and adenoids: everything the ENT surgeon needs to know. Int. J. Pediatr. Otorhinolaryngol 67(Suppl. 1):S69-S76. (Erratum, Int. J. Pediatr. Otorhinolaryngol. 68:387, 2004.) [DOI] [PubMed] [Google Scholar]
- 7.Briles, D. E., S. K. Hollingshead, J. C. Paton, E. W. Ades, L. Novak, F. W. Van Ginkel, and W. H. Benjamin, Jr. 2003. Immunizations with pneumococcal surface protein A and pneumolysin are protective against pneumonia in a murine model of pulmonary infection with Streptococcus pneumoniae. J. Infect. Dis. 188:339-348. [DOI] [PubMed] [Google Scholar]
- 8.Brooks-Walter, A., D. E. Briles, and S. K. Hollingshead. 1999. The pspC gene of Streptococcus pneumoniae encodes a polymorphic protein, PspC, which elicits cross-reactive antibodies to PspA and provides immunity to pneumococcal bacteremia. Infect. Immun. 67:6533-6542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8a.Butch, A. W., G. H. Chung, J. W. Hoffmann, and M. H. Nahm. 1993. Cytokine expression by germinal center cells. J. Immunol. 150:39-47. [PubMed] [Google Scholar]
- 9.Cockeran, R., C. Durandt, C. Feldman, T. J. Mitchell, and R. Anderson. 2002. Pneumolysin activates the synthesis and release of interleukin-8 by human neutrophils in vitro. J. Infect. Dis. 186:562-565. [DOI] [PubMed] [Google Scholar]
- 10.Creery, W. D., F. Diaz-Mitoma, L. Filion, and A. Kumar. 1996. Differential modulation of B7-1 and B7-2 isoform expression on human monocytes by cytokines which influence the development of T helper cell phenotype. Eur. J. Immunol. 26:1273-1277. [DOI] [PubMed] [Google Scholar]
- 11.Defrance, T., B. Vanbervliet, F. Briere, I. Durand, F. Rousset, and J. Banchereau. 1992. Interleukin 10 and transforming growth factor beta cooperate to induce anti-CD40-activated naive human B cells to secrete immunoglobulin A. J. Exp. Med. 175:671-682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fiorentino, D. F., M. W. Bond, and T. R. Mosmann. 1989. Two types of mouse T helper cell. IV. Th2 clones secrete a factor that inhibits cytokine production by Th1 clones. J. Exp. Med. 170:2081-2095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Grütz, G. 2005. New insights into the molecular mechanism of interleukin-10-mediated immunosuppression. J. Leukoc. Biol. 77:3-15. [DOI] [PubMed] [Google Scholar]
- 14.Hammerschmidt, S., S. R. Talay, P. Brandtzaeg, and G. S. Chhatwal. 1997. SpsA, a novel pneumococcal surface protein with specific binding to secretory immunoglobulin A and secretory component. Mol. Microbiol. 25:1113-1124. [DOI] [PubMed] [Google Scholar]
- 15.Hammerschmidt, S., M. P. Tillig, S. Wolff, J. P. Vaerman, and G. S. Chhatwal. 2000. Species-specific binding of human secretory component to SpsA protein of Streptococcus pneumoniae via a hexapeptide motif. Mol. Microbiol. 36:726-736. [DOI] [PubMed] [Google Scholar]
- 15a.Hida, N., T. Shimoyama, Jr., P. Neville, M. F. Dixon, A. T. Axon, T. Shimoyama, Sr., and J. E. Crabtree. 1999. Increased expression of IL-10 and IL-12(p40) mRNA in Helicobacter pylori infected gastric mucosa: relation to bacterial cag status and peptic ulceration. J. Clin. Pathol. 52:658-664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jeannin, P., S. Lecoanet, Y. Delneste, J. F. Gauchat, and J. Y. Bonnefoy. 1998. IgE versus IgG4 production can be differentially regulated by IL-10. J. Immunol. 160:3555-3561. [PubMed] [Google Scholar]
- 17.Jelinek, D. F., J. B. Splawski, and P. E. Lipsky. 1986. The roles of interleukin 2 and interferon-gamma in human B cell activation, growth and differentiation. Eur. J. Immunol. 16:925-932. [DOI] [PubMed] [Google Scholar]
- 18.Kadioglu, A., W. Coward, M. J. Colston, C. R. Hewitt, and P. W. Andrew. 2004. CD4-T-lymphocyte interactions with pneumolysin and pneumococci suggest a crucial protective role in the host response to pneumococcal infection. Infect. Immun. 72:2689-2697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kemp, K., H. Bruunsgaard, P. Skinhoj, and P. B. Klarlund. 2002. Pneumococcal infections in humans are associated with increased apoptosis and trafficking of type 1 cytokine-producing T cells. Infect. Immun. 70:5019-5025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kerr, A. R., J. J. Irvine, J. J. Search, N. A. Gingles, A. Kadioglu, P. W. Andrew, W. L. McPheat, C. G. Booth, and T. J. Mitchell. 2002. Role of inflammatory mediators in resistance and susceptibility to pneumococcal infection. Infect. Immun. 70:1547-1557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Khan, A. Q., Y. Shen, Z. Q. Wu, T. A. Wynn, and C. M. Snapper. 2002. Endogenous pro- and anti-inflammatory cytokines differentially regulate an in vivo humoral response to Streptococcus pneumoniae. Infect. Immun. 70:749-761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kuper, C. F., P. J. Koornstra, D. M. Hameleers, J. Biewenga, B. J. Spit, A. M. Duijvestijn, P. J. Breda Vriesman, and T. Sminia. 1992. The role of nasopharyngeal lymphoid tissue. Immunol. Today 13:219-224. [DOI] [PubMed] [Google Scholar]
- 23.Levy, Y., and J. C. Brouet. 1994. Interleukin-10 prevents spontaneous death of germinal center B cells by induction of the bcl-2 protein. J. Clin. Investig. 93:424-428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lund, F. E., B. A. Garvy, T. D. Randall, and D. P. Harris. 2005. Regulatory roles for cytokine-producing B cells in infection and autoimmune disease. Curr. Dir. Autoimmun. 8:25-54. [DOI] [PubMed] [Google Scholar]
- 25.Luo, R., B. Mann, W. S. Lewis, A. Rowe, R. Heath, M. L. Stewart, E. Agnes, A. E. Hamburger, S. Sivakolundu, E. R. Lacy, P. J. Bjorkman, E. Tuomanen, and R. W. Kriwacki. 2005. Solution structure of choline binding protein A, the major adhesin of Streptococcus pneumoniae. EMBO J. 24:34-43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Malley, R., K. Trzcinski, A. Srivastava, C. M. Thompson, P. W. Anderson, and M. Lipsitch. 2005. CD4 T cells mediate antibody-independent acquired immunity to pneumococcal colonization. Proc. Natl. Acad. Sci. USA 102:4848-4853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.McCool, T. L., and J. N. Weiser. 2004. Limited role of antibody in clearance of Streptococcus pneumoniae in a murine model of colonization. Infect. Immun. 72:5807-5813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Morris, A., and I. Zvetkova. 1997. Cytokine research: the interferon paradigm. J. Clin. Pathol. 50:635-639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Murdoch, C., R. C. Read, Q. Zhang, and A. Finn. 2002. Choline-binding protein A of Streptococcus pneumoniae elicits chemokine production and expression of intercellular adhesion molecule 1 (CD54) by human alveolar epithelial cells. J. Infect. Dis. 186:1253-1260. [DOI] [PubMed] [Google Scholar]
- 30.Ogunniyi, A. D., R. L. Folland, D. E. Briles, S. K. Hollingshead, and J. C. Paton. 2000. Immunization of mice with combinations of pneumococcal virulence proteins elicits enhanced protection against challenge with Streptococcus pneumoniae. Infect. Immun. 68:3028-3033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ogunniyi, A. D., M. C. Woodrow, J. T. Poolman, and J. C. Paton. 2001. Protection against Streptococcus pneumoniae elicited by immunization with pneumolysin and CbpA. Infect. Immun. 69:5997-6003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Paton, J. C., R. A. Lock, C. J. Lee, J. P. Li, A. M. Berry, T. J. Mitchell, P. W. Andrew, D. Hansman, and G. J. Boulnois. 1991. Purification and immunogenicity of genetically obtained pneumolysin toxoids and their conjugation to Streptococcus pneumoniae type 19F polysaccharide. Infect. Immun. 59:2297-2304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Rosenow, C., P. Ryan, J. N. Weiser, S. Johnson, P. Fontan, A. Ortqvist, and H. R. Masure. 1997. Contribution of novel choline-binding proteins to adherence, colonization and immunogenicity of Streptococcus pneumoniae. Mol. Microbiol. 25:819-829. [DOI] [PubMed] [Google Scholar]
- 34.Rousset, F., E. Garcia, T. Defrance, C. Peronne, N. Vezzio, D. H. Hsu, R. Kastelein, K. W. Moore, and J. Banchereau. 1992. Interleukin 10 is a potent growth and differentiation factor for activated human B lymphocytes. Proc. Natl. Acad. Sci. USA 89:1890-1893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rousset, F., S. Peyrol, E. Garcia, N. Vezzio, M. Andujar, J. A. Grimaud, and J. Banchereau. 1995. Long-term cultured CD40-activated B lymphocytes differentiate into plasma cells in response to IL-10 but not IL-4. Int. Immunol. 7:1243-1253. [DOI] [PubMed] [Google Scholar]
- 36.Rubins, J. B., and C. Pomeroy. 1997. Role of gamma interferon in the pathogenesis of bacteremic pneumococcal pneumonia. Infect. Immun. 65:2975-2977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36a.Shibakura, M., K. Niiya, T. Kiguchi, I. Kitajima, M. Niiya, N. Asaumi, N. Huh, Y. Nakata, M. Harada, and M. Tanimoto. 2003. Induction of IL-8 and monocyte chemoattractant protein-1 by doxorubicin in human small cell lung carcinoma cells. Int. J. Cancer 103:380-386. [DOI] [PubMed] [Google Scholar]
- 37.Siber, G. R. 1994. Pneumococcal disease: prospects for a new generation of vaccines. Science 265:1385-1387. [DOI] [PubMed] [Google Scholar]
- 37a.Teruya-Feldstein, J., D. W. Kingma, A. Weiss, L. Sorbara, P. R. Burd, M. Raffeld, B. U. Mueller, G. Tosato, and E. S. Jaffe. 2001. Chemokine gene expression and clonal analysis of B cells in tissues involved by lymphoid interstitial pneumonitis from HIV-infected pediatric patients. Mod. Pathol. 14:929-936. [DOI] [PubMed] [Google Scholar]
- 38.Weigent, D. A., T. L. Huff, J. W. Peterson, G. J. Stanton, and S. Baron. 1986. Role of interferon in streptococcal infection in the mouse. Microb. Pathog. 1:399-407. [DOI] [PubMed] [Google Scholar]
- 39.Zhang, J. R., K. E. Mostov, M. E. Lamm, M. Nanno, S. Shimida, M. Ohwaki, and E. Tuomanen. 2000. The polymeric immunoglobulin receptor translocates pneumococci across human nasopharyngeal epithelial cells. Cell 102:827-837. [DOI] [PubMed] [Google Scholar]
- 40.Zhang, Q., K. Arnaoutakis, C. Murdoch, R. Lakshman, G. Race, R. Burkinshaw, and A. Finn. 2004. Mucosal immune responses to capsular polysaccharides in immunized pre-school children and controls with similar nasal pneumococcal colonization rates. Pediatr. Infect. Dis. J. 23:307-313. [DOI] [PubMed] [Google Scholar]
- 41.Zhang, Q., J. Bernatoniene, L. Bagrade, A. J. Pollard, T. J. Mitchell, J. C. Paton, and A. Finn. 2006. Serum and mucosal antibody responses to pneumococcal protein antigens in children: relationships with carriage status. Eur. J. Immunol. 36:46-57. [DOI] [PubMed] [Google Scholar]
- 42.Zhang, Q., S. Choo, and A. Finn. 2002. Immune responses to novel pneumococcal proteins (pneumolysin, PspA, PsaA and CbpA) in adenoidal B cells from children. Infect. Immun. 70:5363-5369. [DOI] [PMC free article] [PubMed] [Google Scholar]