Abstract
Background
Estrogen/progestin replacement therapy (EPRT), alcohol consumption, physical activity, and breast-feeding duration differ from other factors associated with breast cancer in being immediately modifiable by the individual, thereby representing attractive targets for future breast cancer prevention efforts. To justify such efforts, it is vital to quantify the potential population-level impacts on breast cancer considering population variations in behavior prevalence, risk estimate, and baseline incidence.
Methods
For each of these four factors, we calculated population attributable risk percents (PARs) using population-based survey (2001) and cancer registry data (1998–2002) for 41 subpopulations of white, non-Hispanic California women aged 40–79 years, and ranges of relative risk (RR) estimates from the literature.
Results
Using a single RR estimate, subpopulation PARs ranged from 2.5% to 5.6% for hormone use, from 0.0% to 6.1% for recent consumption of >= 2 alcoholic drinks daily, and 4.6% to 11.0% for physical inactivity. Using a range of RR estimates, PARs were 2–11% for EPRT use, 1–20% for alcohol consumption and 2–15% for physical inactivity. Subpopulation data were unavailable for breastfeeding, but PARs using published RR estimates ranged from 2% to 11% for lifetime breastfeeding >= 31 months. Thus, of 13,019 breast cancers diagnosed annually in California, as many as 1,432 attributable to EPRT use, 2,604 attributable to alcohol consumption, 1,953 attributable to physical inactivity, and 1,432 attributable to never breastfeeding might be avoidable.
Conclusion
The relatively feasible lifestyle changes of discontinuing EPRT use, reducing alcohol consumption, increasing physical activity, and lengthening breastfeeding duration could lower population breast cancer incidence substantially.
Background
Reducing the morbidity and mortality associated with breast cancer, the most commonly diagnosed malignancy in women, is a pressing public health concern. One part of the response to the growing breast cancer burden is to develop effective prevention strategies. Although many breast cancer risk factors have been identified that might form the basis of such strategies, prevention remains challenging, due in large part to practical difficulties in modifying risk-increasing factors like nulliparity, late age at first full-term pregnancy, early age at menarche, and late age at menopause[1,2]. However, some established risk factors for breast cancer could be considered for practicable modification because they involve immediately modifiable personal behaviors of adulthood. Examples include recent use of estrogen/progestin -containing replacement therapy (EPRT)[3], recent alcohol consumption[4], shorter breastfeeding duration[5], and physical inactivity[6,7].
Implementing modifications in any of these behaviors would require considerable efforts in health education and policy. To justify such preventive strategies, it is important first to quantify the theoretical impacts of changes on breast cancer incidence at the population level, but to date few analyses of this sort have occurred[8-10]. A reasonable epidemiologic metric, the population attributable risk (PAR), exists for estimating the effect on disease rates of removing a risk factor from the population. However, since the PAR takes into consideration both disease incidence and risk factor prevalence, and since these are well-established to vary across population subgroups, a PAR determined for a single population has limited utility for understanding the broader range of impact that risk factor changes might have on breast cancer incidence.
Data from California, USA provide an as-yet unexplored opportunity to circumvent this problem, since this state has a large, demographically heterogeneous population and routinely collects detailed breast cancer incidence and risk factor data at the relatively small subpopulation level of the county. In addition, California counties demonstrate variation in both breast cancer incidence rates and socioeconomic status generally representative of national variation[11]. Therefore, we took advantage of California county-level cancer registry and risk factor surveillance data to estimate potential reductions in breast cancer incidence for changes in four modifiable behaviors–usage of EPRT, consumption of alcohol, physical inactivity and sustained breastfeeding.
Methods
Analytic rationale
The PAR is calculated using the population prevalence of the exposure (p) and the relative risk associated with the exposure (r), as follows: PAR = p(r-1)/(1+p(r-1))[12]. We obtained estimates of the prevalence and effect of the exposures under consideration from population risk factor surveys and published epidemiological studies, as described below. We then applied the PARs to breast cancer incidence rates for each of 41 California county-based subpopulations defined by the California Health Interview Survey (CHIS) to estimate the potential change in case counts from reductions in exposure prevalence. We limited our assessments to women of white, non-Hispanic race/ethnicity (hereafter referred to as white) in order to minimize the influence of racial/ethnic distributions on differences among subpopulations, and because the most reliable data on the risks of breast cancer associated with these exposures are from studies that included mostly white women[3,4]. Furthermore, 2000 US Census data show that Californian white women are generally similar to white women nationally on most sociodemographic characteristics but with slightly higher levels of education (21% vs.17% with bachelor's degree) and annual household income ($53K vs. $45K)[13]. Lastly, we restricted our analyses to women aged 40–79 years, who are at highest risk of breast cancer.
We estimated PAR point estimates for two circumstances: 1) for each of the 41 California subpopulations while utilizing a single value for the relative risk (RR), in order to determine the range of likely PAR values across defined subpopulations; and 2) for hypothetical populations with varying exposure prevalences and varying degrees of risk associated with those exposures, utilizing a range of likely values for the RR and a realistic range of exposure prevalences based on the observed subpopulation distributions.
Breast cancer incidence and population data
Using data from the population-based California Cancer Registry and population estimates from the California Department of Finance, we calculated average annual incidence rates of invasive breast cancer (ICD-O-3 site codes 50.0–50.9) for the period 1998–2002 separately for each of the 41 county groups. The software program SEER*Stat. ([14] was used for all calculations.
Risk factor prevalence data
Risk factor prevalence ranges were estimated using data collected by the CHIS program in 2001. The overall response rate to the adult survey was 38%, reflecting both the screener completion rate (i.e., success in surveying a household to select a respondent) of 59%, and the extended interview completion rate (i.e., success in interviewing the selected respondent) of 64%[15]. We used the internet-based AskCHIS[16], to obtain prevalences of EPRT use, alcohol consumption, and physical inactivity for each of the 41 county groups. To assess hormone therapy (HT) use, CHIS asked women aged 40 and older who were not pregnant: "Are you currently taking any hormone replacement supplements prescribed by a medical doctor to control the symptoms of menopause?" As this question referred to any kind of HT, to estimate more closely the population prevalence of women using EPRT we excluded from the numerator of our prevalence calculations any women who reported both HT use and hysterectomy (approximately half of all women reporting HT use), because women with intact uteri are not prescribed estrogen-only HT because of demonstrated risks of endometrial cancer[17].
To assess current alcohol consumption, CHIS asked all adults: "During the past month, have you had at least one drink of any alcoholic beverage such as beer, wine, wine coolers, or liquor?"; those answering yes were also asked: "During the past month, how many days per week or per month did you drink any alcoholic beverages, on the average?" and "On the days when you drank, about how many drinks did you drink on the average?" Answers to these questions were used to calculate the prevalence of women who reported drinking two or more alcoholic drinks per day on 10 or more days in the last month.
To assess physical inactivity, CHIS asked all adults a series of questions about their physical activity in the past 30 days, including whether respondents walked or bicycled to work/errands. The CHIS variable "level of physical activity" consolidated all of these questions into an indicator with three levels: "no vigorous/moderate activity", "some vigorous/moderate activity but doesn't walk/bicycle", and "some vigorous/moderate activity and does walk/bicycle". We used this variable to calculate the percentage of white women aged 40–79 years who reported no vigorous/moderate activity in the last month.
Information regarding breastfeeding is not available from CHIS. However, a recent review of population-based surveys of breastfeeding prevalence[18], estimated that 60–75% of parous white women initiated breastfeeding during the period 1994–2001, suggesting that the prevalence of not initiating breastfeeding ranged from 25% to 40%.
Estimates of relative risk
We obtained estimates of RRs for each risk factor as follows:
EPRT
The Women's Health Initiative (WHI), the only randomized controlled trial of EPRT vs. placebo[3] found an elevated risk of invasive breast cancer in participants after a mean 5.2 years of follow-up, with a hazard ratio of 1.26 (95% confidence interval (CI) 1.00–1.59). A subsequent meta-analysis (including this trial) concluded that RRs of breast cancer among current users of estrogen/progestin ranged from 1.2–1.4 and increased with duration of use[19], compared to never users, although other studies estimated the RR for current, long-term use to be as high as 1.5[20,21]. Therefore, we used an RR of 1.26 for the county-specific PAR calculations and a range of RRs from 1.2–1.4 to estimate PARs for hypothetical populations.
Alcohol consumption
A pooled analysis of 53 epidemiologic studies[4] identified RRs of breast cancer associated with consumption of two or more alcoholic drinks per day (≥ 30 grams per day) as compared to no drinks in the range of 1.3–1.4. A pooled analysis of cohort studies[22] found that, compared with non-drinkers, women consuming 30–60 grams of alcohol daily had a multivariate-adjusted RR of 1.41 (95% CI 1.18–1.69). For one of the California subpopulations (Marin county), RRs associated with drinking alcohol daily were 2.3 (95% CI 1.2–4.4) for 2 glasses and 3.6 (95% CI 1.2–11.5) for 3+ glasses[23]. Therefore, we used an RR of 1.4 for the county-specific calculations and a range of 1.4–2.2 for estimating PARs for hypothetical populations.
Physical inactivity
A review of 36 epidemiologic studies identified RRs for breast cancer associated with inactivity compared to moderate physical activity in the range of 1.3–1.4 irrespective of menopausal status[6]. This range was further supported in a subsequent review by a National Cancer Institute working group[7]. Therefore, we used an RR of 1.3 for the county-specific calculations and a range of RRs from 1.2–1.4 for estimating PARs for hypothetical populations.
Breastfeeding
A pooled analysis of 47 international breast cancer studies (cohort, and hospital-based and population-based case-control)[5] found that the RR for breastfeeding among parous women decreased by 4.3% (95% CI 2.9%–5.8%) for every 12 months beyond the influence of additional births. With adjustment for parity and compared to women who never breastfed, the RR of breast cancer was 0.88 (95% CI 0.82–0.94) for women who breastfed 31–54 months and 0.73 (95% CI 0.63–0.83) for women who breastfed for 55+ months over their lifetimes. For analyses estimating PARs in hypothetical populations, we used RR estimates in the range of 1.1–1.3 for not breastfeeding compared with breastfeeding for at least 31 months.
Results
Subpopulation variation in breast cancer incidence
Overall, the average annual age-adjusted incidence rate of invasive breast cancer among California white women aged 40–79 years (331.6 per 100,000) was slightly higher than the comparable national rate reported by SEER (315.6 per 100,000). Table 1 presents breast cancer incidence rates for each county-group subpopulation. Counties with the highest incidence rates (the urban areas of Marin and San Francisco) had rates 45% higher than counties with the lowest rates (the rural areas of Imperial and Siskiyou/Lassen/Modoc/Trinity).
Table 1.
Average annual population counts, number of invasive breast cancer cases, and incidence rates in California Health Interview Survey-defined California county groups for non-Hispanic white women aged 40–79, 1998–2002.
| California county group | Population | Cases | Ratea |
| Siskiyou/Lassen/Modoc/Trinity | 20,055 | 55 | 258.3 |
| Imperial | 6,795 | 21 | 273.5 |
| Tulare | 37,287 | 112 | 281.0 |
| Tehama/Colusa/Glenn | 17,096 | 53 | 284.8 |
| Kern | 72,647 | 215 | 285.1 |
| Stanislaus | 58,641 | 179 | 294.6 |
| Mendocino/Lake | 29,368 | 93 | 295.9 |
| Madera | 15,356 | 48 | 299.2 |
| San Joaquin | 62,562 | 195 | 300.0 |
| Humboldt/Del Norte | 28,529 | 88 | 304.6 |
| San Luis Obispo | 46,319 | 151 | 306.0 |
| San Bernardino | 163,317 | 512 | 307.1 |
| Butte | 38,390 | 131 | 309.7 |
| Monterey/San Benito | 46,548 | 151 | 309.8 |
| Riverside | 194,069 | 666 | 309.9 |
| Merced | 20,340 | 67 | 310.4 |
| Tuolumne/Alpine/Amador/Calaveras/Inyo/Mariposa/Mono | 40,654 | 138 | 312.3 |
| Shasta | 34,222 | 115 | 318.1 |
| Yolo | 20,875 | 66 | 318.6 |
| Santa Cruz | 40,217 | 123 | 318.7 |
| Sonoma | 87,289 | 283 | 322.7 |
| El Dorado | 33,311 | 106 | 322.8 |
| Fresno | 76,816 | 262 | 323.2 |
| Santa Barbara | 54,635 | 190 | 324.2 |
| Placer | 49,738 | 165 | 328.2 |
| Sutter/Yuba | 19,714 | 68 | 329.6 |
| Nevada/Plumas/Sierra | 28,648 | 100 | 329.8 |
| San Diego | 349,009 | 1,193 | 330.0 |
| Kings | 10,115 | 35 | 333.5 |
| Los Angeles | 744,519 | 2,656 | 338.5 |
| Solano | 42,593 | 142 | 338.6 |
| Orange | 348,248 | 1,217 | 340.2 |
| Ventura | 98,544 | 344 | 348.3 |
| Sacramento | 161,327 | 581 | 349.8 |
| Alameda | 139,371 | 490 | 353.1 |
| Santa Clara | 177,331 | 643 | 354.9 |
| Contra Costa | 136,317 | 488 | 355.7 |
| Napa | 22,552 | 87 | 361.1 |
| San Mateo | 90,534 | 341 | 362.6 |
| San Francisco | 63,036 | 242 | 374.4 |
| Marin | 54,688 | 206 | 374.8 |
| All California | 3,781,621 | 13,019 | 331.6 |
| SEER | 80,072 | 315.6 | |
a per 100,000/year and age-adjusted to the 2000 U.S. standard.
EPRT
In 2001, prior to the release of WHI results, EPRT use varied by over 100% among counties, ranging from 10.0% to 22.9% in a manner generally correlating with breast cancer incidence rate (Table 2). Using an RR estimate of 1.26[3], we found that the county-specific PAR estimates for current EPRT use ranged from 2.5–5.6%, (Table 2). Statewide, we estimated that approximately 4.5% of breast cancer cases (567 cases) annually could be attributed to current use of EPRT. With a range of RR estimates and population prevalences, PAR values for EPRT use ranged from 2% (assuming EPRT utilization of 10% and a relative risk of 1.2) to 11% (assuming EPRT utilization of 30% and a relative risk of 1.4) (Figure 1).
Table 2.
Exposure prevalence and population attributable risk (PAR) for breast cancer among white non-Hispanic women ages 40–79 associated with current use of estrogen/progestin-containing hormone therapy, consumption of 2+ alcoholic beverages/day, and physical inactivity in California Health Interview Survey-defined California county groups.
| Estrogen/progestin hormone therapy | Consumption of 2+ alcoholic beverages | Physical inactivity | ||||||||||
| California county group | Exposure prevalence | PAR for RR = 1.26 | Ratea if exposure removed | N cases due to exposure | Exposure prevalence | PAR for RR = 1.4 | Ratea if exposure removed | N cases due to exposure | Exposure prevalence | PAR for RR = 1.3 | Ratea if exposure removed | N cases due to exposure |
| Siskiyou/Lassen/Trinity/Modoc | 10.00% | 2.50% | 251.8 | 1 | 5.00% | 2.00% | 253.2 | 1 | 30.8 | 8.50% | 236.5 | 8 |
| Imperial | 16.70% | 4.20% | 262.1 | 1 | 0.00% | 0.00% | 273.5 | 0 | 34.1 | 9.30% | 248.1 | 4 |
| Tulare | 19.40% | 4.80% | 267.5 | 5 | 5.60% | 2.20% | 274.9 | 2 | 33 | 9.00% | 255.7 | 17 |
| Tehama/Glenn/Colusa | 11.80% | 3.00% | 276.4 | 2 | 0.00% | 0.00% | 284.8 | 0 | 32.1 | 8.80% | 259.8 | 9 |
| Kern | 11.40% | 2.90% | 276.9 | 6 | 8.60% | 3.30% | 275.7 | 7 | 30.7 | 8.40% | 261.1 | 25 |
| Stanislaus | 13.00% | 3.30% | 285 | 6 | 7.30% | 2.80% | 286.3 | 5 | 34.6 | 9.40% | 266.9 | 22 |
| Mendocino, Lake | 13.30% | 3.40% | 286 | 3 | 6.50% | 2.50% | 288.5 | 2 | 30.5 | 8.40% | 271.1 | 13 |
| Madera | 13.30% | 3.40% | 289.2 | 2 | 0.00% | 0.00% | 299.2 | 0 | 24.8 | 6.90% | 278.5 | 5 |
| San Joaquin | 12.10% | 3.10% | 290.8 | 6 | 4.60% | 1.80% | 294.6 | 3 | 29.4 | 8.10% | 275.7 | 23 |
| Humboldt/Del Norte | 17.90% | 4.40% | 291.1 | 4 | 7.10% | 2.80% | 296.1 | 2 | 27.5 | 7.60% | 281.4 | 8 |
| San Luis Obispo | 15.70% | 3.90% | 294 | 6 | 5.90% | 2.30% | 299 | 3 | 23.5 | 6.60% | 285.8 | 19 |
| San Bernadino | 16.80% | 4.20% | 294.3 | 21 | 5.80% | 2.30% | 300.2 | 12 | 29.8 | 8.20% | 281.9 | 52 |
| Butte | 12.50% | 3.20% | 300 | 4 | 7.50% | 2.90% | 300.7 | 4 | 26.9 | 7.50% | 286.6 | 21 |
| Monterey/San Benito | 18.20% | 4.50% | 295.8 | 7 | 9.10% | 3.50% | 298.9 | 5 | 26.5 | 7.40% | 287 | 17 |
| Riverside | 13.10% | 3.30% | 299.7 | 22 | 7.50% | 2.90% | 300.8 | 19 | 30.2 | 8.30% | 284.2 | 115 |
| Merced | 16.70% | 4.20% | 297.5 | 3 | 0.00% | 0.00% | 310.4 | 0 | 36.7 | 9.90% | 279.6 | 10 |
| Tuolumne/Calaveras/Amador/Inyo/Mariposa/Mono/Alpine | 14.00% | 3.50% | 301.4 | 5 | 11.60% | 4.40% | 298.4 | 6 | 23.7 | 6.60% | 291.6 | 19 |
| Shasta | 14.30% | 3.60% | 306.7 | 4 | 5.70% | 2.20% | 311 | 3 | 26.8 | 7.40% | 294.4 | 14 |
| Yolo | 19.10% | 4.70% | 303.6 | 3 | 9.50% | 3.70% | 306.9 | 2 | 30.8 | 8.50% | 291.7 | 5 |
| Santa Cruz | 21.10% | 5.20% | 302.2 | 6 | 12.80% | 4.90% | 303.2 | 6 | 19.4 | 5.50% | 301.2 | 2 |
| Sonoma | 18.60% | 4.60% | 307.8 | 13 | 12.80% | 4.90% | 307 | 14 | 22.8 | 6.40% | 302 | 19 |
| El Dorado | 13.20% | 3.30% | 312.1 | 3 | 12.80% | 4.90% | 307.1 | 5 | 20.7 | 5.80% | 303.9 | 5 |
| Fresno | 16.90% | 4.20% | 309.6 | 11 | 6.50% | 2.50% | 315 | 7 | 28.7 | 7.90% | 297.6 | 33 |
| Santa Barbara | 16.70% | 4.20% | 310.7 | 8 | 7.40% | 2.90% | 314.9 | 5 | 24.8 | 6.90% | 301.7 | 25 |
| Placer | 13.20% | 3.30% | 317.3 | 5 | 9.40% | 3.60% | 316.3 | 6 | 20.5 | 5.80% | 309.2 | 11 |
| Sutter/Yuba | 10.00% | 2.50% | 321.3 | 2 | 5.00% | 2.00% | 323.1 | 1 | 30.9 | 8.50% | 301.6 | 9 |
| Nevada/Plumas/Sierra | 17.20% | 4.30% | 315.7 | 4 | 13.80% | 5.20% | 312.6 | 5 | 21.5 | 6.10% | 309.8 | 11 |
| San Diego | 19.30% | 4.80% | 314.2 | 57 | 9.90% | 3.80% | 317.4 | 46 | 23.4 | 6.60% | 308.4 | 117 |
| Kings | 11.10% | 2.80% | 324.1 | 1 | 0.00% | 0.00% | 333.5 | 0 | 41.2 | 11.00% | 296.8 | 5 |
| Los Angeles | 18.10% | 4.50% | 323.3 | 119 | 7.90% | 3.10% | 328.2 | 81 | 30.5 | 8.40% | 310.1 | 347 |
| Solano | 15.60% | 3.90% | 325.4 | 6 | 8.90% | 3.40% | 327 | 5 | 23.6 | 6.60% | 316.2 | 7 |
| Orange | 19.30% | 4.80% | 324 | 58 | 9.20% | 3.60% | 328.1 | 43 | 27.7 | 7.70% | 314.1 | 123 |
| Ventura | 17.00% | 4.20% | 333.6 | 15 | 5.00% | 1.90% | 341.5 | 7 | 26.2 | 7.30% | 322.9 | 26 |
| Sacramento | 18.20% | 4.50% | 334 | 26 | 10.70% | 4.10% | 335.5 | 24 | 25.1 | 7.00% | 325.3 | 56 |
| Alameda | 22.90% | 5.60% | 333.3 | 27 | 12.10% | 4.60% | 336.7 | 23 | 24.1 | 6.70% | 329.3 | 31 |
| Santa Clara | 18.80% | 4.70% | 338.4 | 30 | 9.30% | 3.60% | 342.1 | 23 | 26.7 | 7.40% | 328.6 | 60 |
| Contra Costa | 20.10% | 5.00% | 338 | 24 | 11.00% | 4.20% | 340.8 | 20 | 24.2 | 6.80% | 331.6 | 36 |
| Napa | 22.70% | 5.60% | 341 | 5 | 9.10% | 3.50% | 348.4 | 3 | 24.1 | 6.70% | 336.8 | 11 |
| San Mateo | 13.30% | 3.40% | 350.5 | 11 | 14.10% | 5.40% | 343.2 | 18 | 25 | 7.00% | 337.3 | 36 |
| San Francisco | 20.30% | 5.00% | 355.7 | 12 | 13.50% | 5.10% | 355.2 | 12 | 22.2 | 6.20% | 351 | 21 |
| Marin | 20.40% | 5.00% | 356 | 10 | 16.40% | 6.10% | 351.8 | 13 | 16.2 | 4.60% | 357.4 | 11 |
| All California | 17.50% | 4.40% | 317.2 | 567 | 9.00% | 3.50% | 320.1 | 450 | 27.1 | 7.50% | 306.7 | 1422 |
a per 100,000/year and age-adjusted to the 2000 U.S. standard
Figure 1.
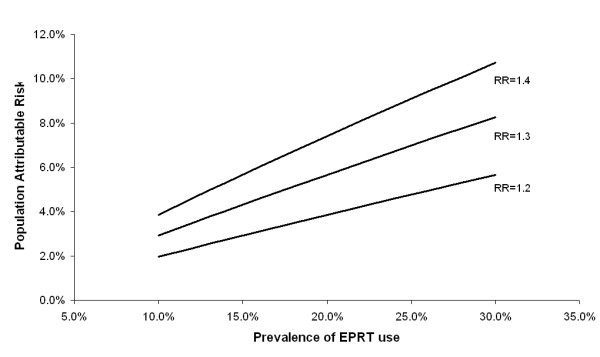
Population attributable risk of breast cancer associated with use of combined estrogen/progestin replacement therapy (EPRT) for various levels of exposure and relative risk.
Alcohol consumption
Consumption of at least two alcoholic beverages per day among white women aged 40–79 varied by over 100% among counties, ranging from 0% to 16.4% also in a manner generally correlating with breast cancer incidence rate (Table 2). Prevalence was notably higher in northern (Marin, Napa) than southern (Orange, Los Angeles) urban counties. Using an RR estimate of 1.4[22], PAR estimates for consumption of 2+ alcoholic drinks per day ranged between 0.0% and 6.1% for county groups (Table 2). Overall in California, we estimated that about 3.5% of breast cancer cases, or 450 per year were attributable to this level of alcohol consumption. Considering ranges of RR estimates from 1.4 to 2.2 and population prevalences from 2% to 22%, we produced a corresponding range of PAR values for alcohol consumption from 1% to 20% (Figure 2).
Figure 2.
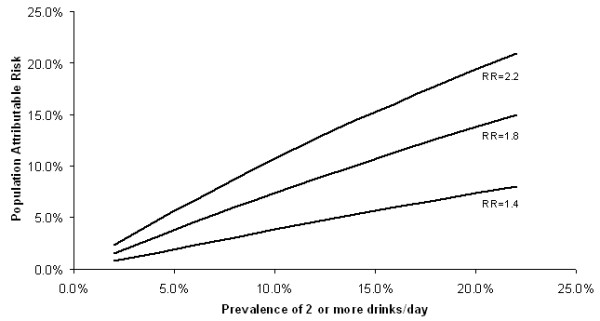
Population attributable risk of breast cancer associated with consumption of 2 or more alcoholic drinks per day for various levels of population prevalence and relative risk.
Physical inactivity
White women aged 40–79 years reporting no vigorous/moderate physical activity in the last month ranged from 16.1 to 41.0% by county (Table 2). Using an RR estimate of 1.3[6], PAR estimates for physical inactivity ranged between 0.0% and 6.1% for county groups (Table 2). We estimated that statewide, 7.5% of breast cancer cases, or 1,422 cases per year, were attributable to a sedentary lifestyle. Considering ranges of RR estimates from 1.2 to 1.4 and population prevalences from 15% to 45%, we estimated a corresponding range of PARs of 2%–15% (Figure 3).
Figure 3.
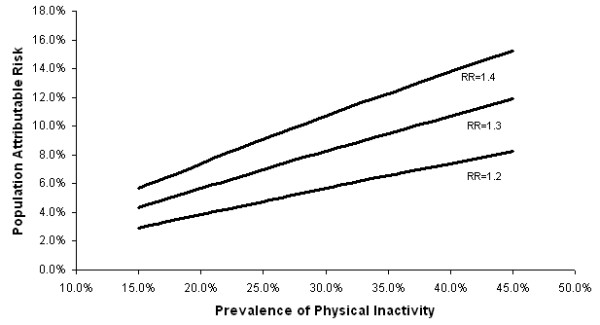
Population attributable risk of breast cancer associated with physical inactivity (reporting no moderate/vigorous activity in past 30 days) for various levels of population prevalence and relative risk.
Breastfeeding
Using prevalences of never breastfeeding ranging from 25% to 40% and RRs ranging from 1.1 to 1.3, PAR estimates for never breastfeeding (compared with breastfeeding for at least 31 months over a lifetime) ranged from 2% to 11% (Figure 4). According to CHIS, 16.4% of white California women aged 40–79 years had never given birth. Therefore, applying the PARs to the California population of parous white women aged 40–79 years, we estimated that never breastfeeding (compared to breastfeeding for 31+ months) could explain between 218 and 1,306 breast cancer cases yearly.
Figure 4.
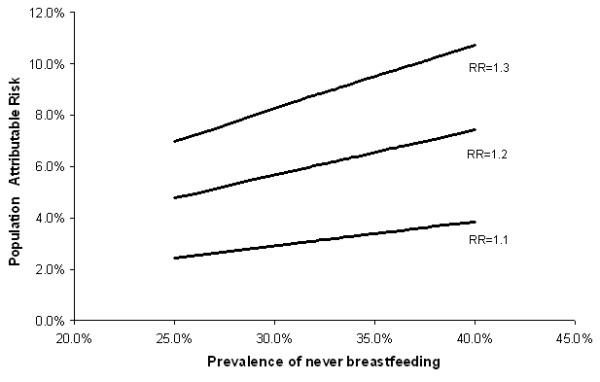
Population attributable risk of breast cancer associated with not breast feeding (compared to breastfeeding for 31 months or more over a lifetime) for various levels of population prevalence and relative risk.
Discussion
This study quantified the theoretical reductions in breast cancer to be expected from changes in EPRT use, alcohol consumption, physical activity and breastfeeding duration for a range of socioeconomically heterogeneous populations. Because the variation in breast cancer incidence rates, risk factor prevalences, and socioeconomic status across California is generally representative of national variation[11], we believe that the range of PAR estimates we obtained have relevance not only for 8 million white California women, but also could reflect the range across the US white population. Of 13,019 breast cancers diagnosed annually in Californian white women aged 40–79 years, our data suggest that as many as 1,432 (11%) may be attributable to EPRT use, 2,604 (20%) may be attributable to consuming 2 or more alcoholic drinks daily, 1,953 (15%) may be attributable to physical inactivity and 1,432 (11%) may be attributable to never breastfeeding.
Our data suggest that the proportions of women consuming two or more alcoholic drinks daily may have a greater influence on breast cancer incidence than previously estimated, particularly in high-incidence subpopulations. A pooled analysis of 53 studies estimated that 4% of breast cancer cases in developed countries were attributable to alcohol, based on average consumption of 6.0 g/day in controls[4]. Data from the Third National Health and Nutrition Examination Survey, reporting prevalences of light (0.1–6.4 g/day), moderate (6.5–25.9 g/day) and heavy (26+ g/day) drinking of 27%, 13% and 3% respectively, were used to produce a PAR of 2.1% across all levels[10]. Our estimates of possible PARs for consuming two or more drinks daily included a range with much higher values (1% to 20%). At the low end of our RR range, PAR estimates exceeded 5% for Marin and San Francisco counties, which have among the highest breast cancer rates among all counties in California and in the non-California regions of the SEER program[24]. If prevalence of alcohol consumption now is higher than in previously examined populations; a public health effort aimed at reducing alcohol consumption below two drinks daily may be an increasingly important strategy for breast cancer prevention in particular subpopulations of white women. A more specific recommendation needs to consider the balance of health risks and benefits for alcohol consumption, which may protect against ischemic heart disease and myocardial infarction[25], diseases with greater mortality risks than breast cancer. An alternative strategy to reducing alcohol intake[26] may be for women to increase folate intake through supplementation or food supply fortification, as recent evidence from the Nurses Health Study showed no adverse influence of alcohol on breast cancer occurrence among women with high plasma folate[27].
Although the presumed effect of physical activity on breast cancer risk is not large (RR = 1.3–1.4), the highly prevalent nature of physical inactivity corresponded to a California-wide PAR of 7.5%. Especially at the high end of our estimated PAR range (15%), the potential for physical activity interventions to reduce breast cancer incidence may be underestimated. The CHIS statewide average of 27% of women reporting inactivity is higher than that reported in previous PAR estimation efforts[28] and underscores the importance of ongoing public health efforts to increase physical activity for its well-known, wide range of health benefits.
We found that use of EPRT at the levels occurring in 2001 may have explained 2.5% to 5.6% of breast cancer in white California women, but could have been responsible for as much as 11%, depending on the associated RR. Coombs et al. recently used CHIS data to estimate that HT (irrespective of formulation) was attributable for 4.3–17.4% of breast cancer in California in 2001, depending on the RR[9]. Findings from the Women's Health Initiative (WHI) on risks of long-term EPRT use, released in 2002 after the collection of the data used in this analysis, appear to have had a dramatic effect on EPRT prescribing and usage patterns, with declines of up to 50% reported in some cohorts. Thus, long-term use is now much less likely to be recommended by physicians to reduce risks of osteoporosis and cardiovascular disease[29], although some women continue to use EPRT for treatment of menopausal symptoms. Our results would predict that reductions in EPRT use related to the WHI results could result in breast cancer incidence rate declines between 5% and 15%, depending on population characteristics.
Our findings also indicate that the number of breast cancer cases occurring in California might be reduced by as much as 11% by encouraging breastfeeding for 31 months or more (a time period derived from a meta-analysis that could be refined in further analyses). Lengthening the duration of breastfeeding among childbearing women depends in part on individual circumstances, including parity and medical conditions interfering with breastfeeding, as well as on societal influences beyond individual control (e.g., cultural support of breastfeeding, workplace policies for maternal leave and breast pumping). Nevertheless, our data support continued public policy efforts to encourage longer duration of breastfeeding in childbearing women, which for a woman bearing two children equates to breastfeeding each child slightly more than one year, particularly given the overwhelming evidence of breastfeeding benefit to the infant.
The PAR has had many previous applications to breast cancer, predominantly to estimate the percentage of cases "explained" by established risk factors using results from a single study[10,28,30,31]. Madigan et al. estimated that well-established risk factors for breast cancer (later age at first birth, nulliparity, higher income, and family history of breast cancer) together accounted for 41% of U.S. breast cancer cases[30]. Mezzetti et al. estimated that the attributable risk in an Italian population was 10.7% for high alcohol consumption, 15.0% for low beta-carotene intake, and 11.6% for low physical activity, with the three factors together accounting for 33% of breast cancer cases[28]. Rockhill et al. estimated that 25% (95% CI 6% – 48%) of breast cancer cases could be attributed to menarche before age 14 years, nulliparity/first birth after age 20 years, family history, and history of benign breast biopsy[31]. However, these studies did not aim to quantify potential case count reductions from changes in risk factor prevalence, as we have done.
Our study has several limitations. Our statistic of choice, the PAR, is a useful measure for quantifying the extent of disease reduction possible through behavior modification, but its interpretation can be complex, particularly as it relates to the interrelationships of factors of interest with other established risk factors and changes in population prevalence. In addition, the PAR does not incorporate processes of risk latency and reversibility for exposures; for example, breast cancer risk increases with duration of EPRT use and declines after EPRT cessation[21], effects we could not capture. The PAR also is sensitive to the reference (i.e., minimum risk) category chosen. To minimize the effect of these limitations, we restricted our analysis to a relatively homogeneous group, white women aged 40–79 years, and used systematically collected estimates of exposure prevalence in categories for which multiple RR estimates were available. Exposure prevalence estimates, although based on the best available population data, are subject to sampling variation, response and misclassification bias. The AskCHIS program uses a weighting procedure to compensate for differential selection probabilities for households and persons, which attempts to minimize selection bias and adjusts, to the extent possible, for undercoverage in the sampling frames[15]. Although RR estimates were taken from pooled analyses or other multi-study efforts, they remain subject to bias or uncontrolled confounding. To help account for this problem, we examined a range of possible RR values. Empirically derived ranges of values for RR and exposure prevalence may be more difficult to interpret but are more useful for future public health consideration. Lastly, our PAR estimates cannot be summed together to produce a combined PAR estimate for removing all three risk factors from the population, because these factors are likely to be correlated; moreover, we did not calculate multivariate PARs because RRs for the combinations of all three risk factors were not available.
We restricted our analysis to white non-Hispanic women because few studies have included sufficient numbers of women from non-white racial/ethnic groups to obtain reliable RR estimates for the exposures we considered here[32]. Based on the limited data available[32,33], it is likely that incidence reduction in nonwhite groups also may be anticipated from changes in these behaviors. Statewide CHIS data suggested that the prevalences of combined EPRT use and consumption of at least two alcoholic drinks per day were considerably lower in African American, Asian and Hispanic women in 2001 than in white women, at 7%, 13% and 9%; and 2.7%, 0.8% and 0.9%, respectively. Average annual age-adjusted incidence rates of breast cancer were also much lower in these ethnic groups, at 263, 194 and 197 per 100,000 women per year for the period 1998–2002 in California. Thus, for these factors, PARs for non-white women may be considerably lower than those we estimated in white women. On the other hand, prevalence of physical inactivity was notably higher in African American, Asian and Hispanic women (45%, 45%, 57%, respectively), such that PARs and presumable breast cancer preventive benefits could be higher for remediating physical inactivity in non-white women[33] than our estimates for white women.
Conclusion
Our results suggest that public health interventions to further reduce the proportions of women taking EPRT, drinking two or more alcoholic beverages daily, being physically inactive or not breastfeeding could substantially decrease the numbers of new breast cancer cases and, furthermore, may have a greater impact on breast cancer risks than previously estimated by others[4,10], particularly in high incidence populations where these risk factors tend to be more prevalent. Given the relative feasibility of implementing these changes and the other health benefits already associated with them, public health efforts toward this end are warranted.
Abbreviations
EPRT estrogen/progestin replacement therapy
PAR population attributable risk
CHIS California Health Interview Survey
HT hormone therapy
WHI Womens' Health Initiative
CI confidence intervals
RR relative risk
Competing interests
Drs. Clarke and Purdie have consulted for attorneys preparing litigation regarding hormone therapy.
Authors' contributions
CC conceived of the study, performed statistical analyses and drafted the manuscript. DP performed statistical analyses, and drafted the manuscript. SG participated in the study design and helped to draft the manuscript. All authors read and approved the final manuscript.
Pre-publication history
The pre-publication history for this paper can be accessed here:
Acknowledgments
Acknowledgements
The authors thank Dr. Susan Stewart for her contributions. This study was supported by the SEER Rapid Response Surveillance Study program by a supplement to contract N01-CN-35136 with the National Cancer Institute. The collection of cancer incidence data used in this study was supported by the California Department of Health Services as part of the statewide cancer reporting program mandated by California Health and Safety Code Section 103885; the National Cancer Institute's Surveillance, Epidemiology and End Results Program under contract N01-PC-35136 awarded to the Northern California Cancer Center, contract N01-PC-35139 awarded to the University of Southern California, and contract N02-PC-15105 awarded to the Public Health Institute; and the Centers for Disease Control and Prevention's National Program of Cancer Registries, under agreement #U55/CCR921930-02 awarded to the Public Health Institute. The ideas and opinions expressed herein are those of the author(s) and endorsement by the State of California, Department of Health Services, the National Cancer Institute, and the Centers for Disease Control and Prevention or their Contractors and Subcontractors is not intended nor should be inferred.
Contributor Information
Christina A Clarke, Email: tina@nccc.org.
David M Purdie, Email: davidpurdie@fastmail.fm.
Sally L Glaser, Email: sglaser@nccc.org.
References
- Kelsey JL, Gammon MD, John EM. Reproductive factors and breast cancer. Epidemiol Rev. 1993;15:36–47. doi: 10.1093/oxfordjournals.epirev.a036115. [DOI] [PubMed] [Google Scholar]
- Hulka BS, Brinton LA. Hormones and breast and endometrial cancers: preventive strategies and future research. Environ Health Perspect. 1995;103 Suppl 8:185–189. doi: 10.1289/ehp.95103s8185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rossouw JE, Anderson GL, Prentice RL, LaCroix AZ, Kooperberg C, Stefanick ML, Jackson RD, Beresford SA, Howard BV, Johnson KC, Kotchen JM, Ockene J. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results From the Women's Health Initiative randomized controlled trial. JAMA. 2002;288:321–333. doi: 10.1001/jama.288.3.321. [DOI] [PubMed] [Google Scholar]
- Hamajima N, Hirose K, Tajima K, Rohan T, Calle EE, Heath CWJ, Coates RJ, Liff JM, Talamini R, Chantarakul N, Koetsawang S, Rachawat D, Morabia A, Schuman L, Stewart W, Szklo M, Bain C, Schofield F, Siskind V, Band P, Coldman AJ, Gallagher RP, Hislop TG, Yang P, Kolonel LM, Nomura AM, Hu J, Johnson KC, Mao Y, De Sanjose S, Lee N, Marchbanks P, Ory HW, Peterson HB, Wilson HG, Wingo PA, Ebeling K, Kunde D, Nishan P, Hopper JL, Colditz G, Gajalanski V, Martin N, Pardthaisong T, Silpisornkosol S, Theetranont C, Boosiri B, Chutivongse S, Jimakorn P, Virutamasen P, Wongsrichanalai C, Ewertz M, Adami HO, Bergkvist L, Magnusson C, Persson I, Chang-Claude J, Paul C, Skegg DC, Spears GF, Boyle P, Evstifeeva T, Daling JR, Hutchinson WB, Malone K, Noonan EA, Stanford JL, Thomas DB, Weiss NS, White E, Andrieu N, Bremond A, Clavel F, Gairard B, Lansac J, Piana L, Renaud R, Izquierdo A, Viladiu P, Cuevas HR, Ontiveros P, Palet A, Salazar SB, Aristizabel N, Cuadros A, Tryggvadottir L, Tulinius H, Bachelot A, Le MG, Peto J, Franceschi S, Lubin F, Modan B, Ron E, Wax Y, Friedman GD, Hiatt RA, Levi F, Bishop T, Kosmelj K, Primic-Zakelj M, Ravnihar B, Stare J, Beeson WL, Fraser G, Bullbrook RD, Cuzick J, Duffy SW, Fentiman IS, Hayward JL, Wang DY, McMichael AJ, McPherson K, Hanson RL, Leske MC, Mahoney MC, Nasca PC, Varma AO, Weinstein AL, Moller TR, Olsson H, Ranstam J, Goldbohm RA, van den Brandt PA, Apelo RA, Baens J, de la Cruz JR, Javier B, Lacaya LB, Ngelangel CA, La Vecchia C, Negri E, Marubini E, Ferraroni M, Gerber M, Richardson S, Segala C, Gatei D, Kenya P, Kungu A, Mati JG, Brinton LA, Hoover R, Schairer C, Spirtas R, Lee HP, Rookus MA, van Leeuwen FE, Schoenberg JA, McCredie M, Gammon MD, Clarke EA, Jones L, Neil A, Vessey M, Yeates D, Appleby P, Banks E, Beral V, Bull D, Crossley B, Goodill A, Green J, Hermon C, Key T, Langston N, Lewis C, Reeves G, Collins R, Doll R, Peto R, Mabuchi K, Preston D, Hannaford P, Kay C, Rosero-Bixby L, Gao YT, Jin F, Yuan JM, Wei HY, Yun T, Zhiheng C, Berry G, Cooper Booth J, Jelihovsky T, MacLennan R, Shearman R, Wang QS, Baines CJ, Miller AB, Wall C, Lund E, Stalsberg H, Shu XO, Zheng W, Katsouyanni K, Trichopoulou A, Trichopoulos D, Dabancens A, Martinez L, Molina R, Salas O, Alexander FE, Anderson K, Folsom AR, Hulka BS, Bernstein L, Enger S, Haile RW, Paganini-Hill A, Pike MC, Ross RK, Ursin G, Yu MC, Longnecker MP, Newcomb P, Kalache A, Farley TM, Holck S, Meirik O. Alcohol, tobacco and breast cancer - collaborative reanalysis of individual data from 53 epidemiological studies, including 58,515 women with breast cancer and 95,067 women without the disease. Br J Cancer. 2002;87:1234–1245. doi: 10.1038/sj.bjc.6600596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collaborative Group on Hormonal Factors in Breast Cancer Breast cancer and breastfeeding: collaborative reanalysis of individual data from 47 epidemiological studies in 30 countries, including 50302 women with breast cancer and 96973 women without the disease. Lancet. 2002;360:187–195. doi: 10.1016/S0140-6736(02)09454-0. [DOI] [PubMed] [Google Scholar]
- Friedenreich CM. Physical activity and cancer prevention: from observational to intervention research. Cancer Epidemiol Biomarkers Prev. 2001;10:287–301. [PubMed] [Google Scholar]
- Physical activity across the cancer continuum: report of a workshop: review of existing knowledge and innovative designs for future research. Cancer. 2002;95:1134–1143. doi: 10.1002/cncr.10771. [DOI] [PubMed] [Google Scholar]
- Coombs NJ, Taylor R, Wilcken N, Boyages J. HRT and breast cancer: impact on population risk and incidence. Eur J Cancer. 2005;41:1775–1781. doi: 10.1016/j.ejca.2005.03.030. [DOI] [PubMed] [Google Scholar]
- Coombs NJ, Taylor R, Wilcken N, Fiorica J, Boyages J. Hormone replacement therapy and breast cancer risk in California. Breast J. 2005;11:410–415. doi: 10.1111/j.1075-122X.2005.00132.x. [DOI] [PubMed] [Google Scholar]
- Tseng M, Weinberg CR, Umbach DM, Longnecker MP. Calculation of population attributable risk for alcohol and breast cancer (United States) Cancer Causes Control. 1999;10:119–123. doi: 10.1023/A:1008853115901. [DOI] [PubMed] [Google Scholar]
- Reynolds P, Hurley S, Goldberg DE, Anton-Culver H, Bernstein L, Deapen D, Horn-Ross PL, Peel D, Pinder R, Ross RK, West D, Wright WE, Ziogas A. Regional variations in breast cancer among California teachers. Epidemiology. 2004;15:746–754. doi: 10.1097/01.ede.0000134863.45834.50. [DOI] [PubMed] [Google Scholar]
- Breslow NE, Day NE. Methods in Cancer Research Vol. 1: The Analysis of Case-Control Studies. Lyon , IARC Sci. Publ.; 1980. pp. 73–76. [PubMed] [Google Scholar]
- US Census Bureau Census 2000 Summary File 3, Matrices P19, P36, P37, P38, PCT24, and PCT25
- National Cancer Institute DCCPSSRPCSB. Surveillance, Epidemiology, and End Results (SEER) Program (www.seer.cancer.gov) SEER*Stat Database: Incidence - SEER 9 Regs Public-Use, Nov 2004 Sub (1973-2002. National Cancer Institute, DCCPS, Surveillance Research Program, Cancer Statistics Branch; 2005. [Google Scholar]
- California Health Interview Survey . CHIS 2001 Methodology Series: Report 4 – Response Rates. Los Angeles, CA , UCLA Center for Health Policy Research; 2002. [Google Scholar]
- California Health Interview Survey . CHIS 2001 Adult Public Use File, Release 3 [computer file] Los Angeles, CA , UCLA Center for Health Policy Research; 2004. [Google Scholar]
- Grady D, Gebretsadik T, Kerlikowske K, Ernster V, Petitti D. Hormone replacement therapy and endometrial cancer risk: a meta-analysis. Obstet Gynecol. 1995;85:304–313. doi: 10.1016/0029-7844(94)00383-O. [DOI] [PubMed] [Google Scholar]
- Newton ER. The epidemiology of breastfeeding. Clin Obstet Gynecol. 2004;47:613–623. doi: 10.1097/01.grf.0000137068.00855.a0. [DOI] [PubMed] [Google Scholar]
- Nelson HD, Humphrey LL, Nygren P, Teutsch SM, Allan JD. Postmenopausal hormone replacement therapy: scientific review. JAMA. 2002;288:872–881. doi: 10.1001/jama.288.7.872. [DOI] [PubMed] [Google Scholar]
- Chen WY, Colditz GA, Rosner B, Hankinson SE, Hunter DJ, Manson JE, Stampfer MJ, Willett WC, Speizer FE. Use of postmenopausal hormones, alcohol, and risk for invasive breast cancer. Ann Intern Med. 2002;137:798–804. doi: 10.7326/0003-4819-137-10-200211190-00008. [DOI] [PubMed] [Google Scholar]
- Collins JA, Blake JM, Crosignani PG. Breast cancer risk with postmenopausal hormonal treatment. Hum Reprod Update. 2005;11:545–560. doi: 10.1093/humupd/dmi028. [DOI] [PubMed] [Google Scholar]
- Smith-Warner SA, Spiegelman D, Yaun SS, van den Brandt PA, Folsom AR, Goldbohm RA, Graham S, Holmberg L, Howe GR, Marshall JR, Miller AB, Potter JD, Speizer FE, Willett WC, Wolk A, Hunter DJ. Alcohol and breast cancer in women: a pooled analysis of cohort studies. JAMA. 1998;279:535–540. doi: 10.1001/jama.279.7.535. [DOI] [PubMed] [Google Scholar]
- Wrensch M, Chew T, Farren G, Barlow J, Belli F, Clarke C, Erdmann CA, Lee M, Moghadassi M, Peskin-Mentzer R, Quesenberry CPJ, Souders-Mason V, Spence L, Suzuki M, Gould M. Risk factors for breast cancer in a population with high incidence rates. Breast Cancer Res. 2003;5:R88–102. doi: 10.1186/bcr605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clarke CA, Glaser SL, West DW, Ereman RR, Erdmann CA, Barlow JM, Wrensch MR. Breast cancer incidence and mortality trends in an affluent population: Marin County, California, USA, 1990-1999. Breast Cancer Res. 2002;4:R13. doi: 10.1186/bcr458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rimm EB, Klatsky A, Grobbee D, Stampfer MJ. Review of moderate alcohol consumption and reduced risk of coronary heart disease: is the effect due to beer, wine, or spirits. Bmj. 1996;312:731–736. doi: 10.1136/bmj.312.7033.731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colditz GA. Epidemiology and prevention of breast cancer. Cancer Epidemiol Biomarkers Prev. 2005;14:768–772. doi: 10.1158/1055-9965.EPI-04-0157. [DOI] [PubMed] [Google Scholar]
- Zhang SM, Willett WC, Selhub J, Hunter DJ, Giovannucci EL, Holmes MD, Colditz GA, Hankinson SE. Plasma folate, vitamin B6, vitamin B12, homocysteine, and risk of breast cancer. J Natl Cancer Inst. 2003;95:373–380. doi: 10.1093/jnci/95.5.373. [DOI] [PubMed] [Google Scholar]
- Mezzetti M, La Vecchia C, Decarli A, Boyle P, Talamini R, Franceschi S. Population attributable risk for breast cancer: diet, nutrition, and physical exercise. J Natl Cancer Inst. 1998;90:389–394. doi: 10.1093/jnci/90.5.389. [DOI] [PubMed] [Google Scholar]
- Vogel RA. The changing view of hormone replacement therapy. Rev Cardiovasc Med. 2003;4:68–71. [PubMed] [Google Scholar]
- Madigan MP, Ziegler RG, Benichou J, Byrne C, Hoover RN. Proportion of breast cancer cases in the United States explained by well-established risk factors. J Natl Cancer Inst. 1995;87:1681–1685. doi: 10.1093/jnci/87.22.1681. [DOI] [PubMed] [Google Scholar]
- Rockhill B, Weinberg CR, Newman B. Population attributable fraction estimation for established breast cancer risk factors: considering the issues of high prevalence and unmodifiability. Am J Epidemiol. 1998;147:826–833. doi: 10.1093/oxfordjournals.aje.a009535. [DOI] [PubMed] [Google Scholar]
- Bernstein L, Teal CR, Joslyn S, Wilson J. Ethnicity-related variation in breast cancer risk factors. Cancer. 2003;97:222–229. doi: 10.1002/cncr.11014. [DOI] [PubMed] [Google Scholar]
- John EM, Horn-Ross PL, Koo J. Lifetime physical activity and breast cancer risk in a multiethnic population: the San Francisco Bay area breast cancer study. Cancer Epidemiol Biomarkers Prev. 2003;12:1143–1152. [PubMed] [Google Scholar]


