Abstract
The effect of intracerebroventricular (i.c.v.) administration of different potassium channel blockers (tetraethylammonium, apamin, charybdotoxin, gliquidone), potassium channel openers (pinacidil, minoxidil, cromakalim) and aODN to mKv1.1 on immobility time was evaluated in the mouse forced swimming test, an animal model of depression.
Tetraethylammonium (TEA; 5 μg per mouse i.c.v.), apamin (3 ng per mouse i.c.v.), charybdotoxin (1 μg per mouse i.c.v.) and gliquidone (6 μg per mouse i.c.v.) administered 20 min before the test produced anti-immobility comparable to that induced by the tricyclic antidepressants amitriptyline (15 mg kg−1 s.c.) and imipramine (30 mg kg−1 s.c.).
By contrast pinacidil (10–20 μg per mouse i.c.v.), minoxidil (10–20 μg per mouse i.c.v.) and cromakalim (20–30 μg per mouse i.c.v.) increased immobility time when administered in the same experimental conditions.
Repeated administration of an antisense oligonucleotide (aODN) to the mKv1.1 gene (1 and 3 nmol per single i.c.v. injection) produced a dose-dependent increase in immobility time of mice 72 h after the last injection. At day 7, the increasing effect produced by aODN disappeared. A degenerate mKv1.1 oligonucleotide (dODN), used as control, did not produce any effect in comparison with saline- and vector-treated mice.
At the highest effective dose, potassium channels modulators and the mKv1.1 aODN did not impair motor coordination, as revealed by the rota rod test, nor did they modify spontaneous motility as revealed by the Animex apparatus.
These results suggest that modulation of potassium channels plays an important role in the regulation of immobility time in the mouse forced swimming test.
Keywords: Potassium channel, forced swimming test, minoxidil, pinacidil, cromakalim, gliquidone, apamin, charybdotoxin, tethraethylammonium, antisense oligonucleotide
Introduction
Several kinds of potassium channels with different electrophysiological characteristics and pharmacological sensitivities have been described in neurones (Halliwell, 1990; Aronson, 1992). In general, open potassium channels inhibit excitable cells by drawing the membrane potential toward the potassium equilibrium potential and away from the action potential threshold. Potassium channels that are tonically open set the resting membrane potential. Stimuli that activate potassium channels hyperpolarize cells and lower the effectiveness of excitatory imputs (Christie, 1995). Evidence in the literature suggests that antidepressant drugs may modulate neuronal excitability via potassium channel inhibition and this may even be the final common pathway of pharmacological action of these drugs (Tytgat et al., 1997).
Recently it was suggested that, in the forced swimming test, the effect of lithium pretreatment was additive with effects on antidepressant drugs which was proposed by Nixon et al. (1994) to be a result of an inhibition of potassium channels. Quinine, a calcium-activated potassium channel blocker (Cherubini et al., 1984), decreased the immobility time of mice administered 5-hydroxytriptamine (5-HT) and noradrenaline uptake inhibitors (Guo et al., 1995). Furthermore, the KATP-dependent potassium channel blocker glyburide, when combined with antidepressants acting through a serotoninergic or noradrenergic mechanism, was more effective in decreasing the amount of time mice were immobile than when these drugs were administered alone (Guo et al., 1996). By contrast, the KATP-dependent potassium channel activator cromakalim induced an effect opposite to that seen following pretreatment with potassium channel blockers in the mouse forced swimming test. Cromakalim antagonized the anti-immobility effect of imipramine, amitriptyline, desipramine and paroxetine through a central mechanism (Redrobe et al., 1996).
In the present work we thought it worthwhile to employ potassium channel modulators to better elucidate the role of potassium channels in the forced swimming test. Charybdotoxin, as well as apamin, was originally reported to specifically block currents through the calcium-activated potassium channel (Miller et al., 1985; Cook, 1988). In recent years, it has become clear that charybdotoxin can also block voltage-gated potassium channels (Stühmer et al., 1989; Strong, 1990). Sulphonylureas such as gliquidone block KATP potassium channels in neurones whereas minoxidil, pinacidil and cromakalim open the same type (KATP) of potassium channel (Edwards & Weston, 1993; Longman & Hamilton, 1992). TEA blocks different types of potassium channels in neurones including calcium-activated and voltage-dependent potassium channels (Cook & Quast, 1990; Halliwell, 1990). Since TEA is not selective for any of them in particular (Cook & Quast, 1990; Halliwell, 1990), an antisense oligonucleotide (aODN) was used as a selective blocker of mKv1.1, a voltage-gated potassium channel. aODNs are short synthetic DNA segments complementary to sequences of an mRNA target. By forming DNA/mRNA heteroduplexes, aODNs can transiently inactivate single genes.
To this purpose we have evaluated the effects produced by the potassium channel blockers TEA, apamin, charybdotoxin and gliquidone, the potassium channel activators minoxidil, pinacidil and cromakalim, as well as an mKv1.1 aODN in the mouse forced swimming test.
Methods
Animals
Male Swiss albino mice (23–30 g) from the Morini (San Polo d'Enza, Italy) breeding farm were used. Fifteen mice were housed per cage. The cages were placed in the experimental room 24 h before the test for acclimatization. The animals were fed a standard laboratory diet and tap water ad libitum and kept at 23±1°C with a 12 h light/dark cycle, light on at 19.00 h. All experiments, conducted between 10.00 h to 14.00 h, were carried out according to the guidelines of the European Community Council for experimental animal care.
Intracerebroventricular injection technique
Intracerebroventricular administration was performed under ether anaesthesia, according to the method described by Haley & McCormick (1957). Briefly, during anaesthesia, mice were grasped firmly by the loose skin behind the head. A 0.4 mm external diameter, hypodermic needle attached to a 10 μl syringe was inserted perpendicularly through the skull and no more than 2 mm into the brain of the mouse, where 5 μl were then administered. The injection site was 1 mm to the right or left from the midpoint on a line drawn through to the anterior base of the ears. Injections were performed into the right or left ventricle randomly. To ascertain that the drugs were administered exactly into the cerebral ventricle, some mice (20%) were injected with 5 μl of diluted 1 : 10 India ink and their brains examined macroscopically after sectioning. The accuracy of the injection technique was evaluated and the percentage of correct injections was 95%.
Forced swimming test
The forced swimming test used was the same as described by Porsolt et al. (1977). Briefly, mice were dropped individually into glass cylinders (height: 25 cm, diameter: 10 cm) containing 6 cm of water maintained at 22–23°C and left there for 6 min. A mouse was judged to be immobile when it floated in the water, in an upright position, and made only small movements to keep its head above water. The duration of immobility was recorded during the last 4 min of the 6-min test. A decrease in the duration of immobility is indicative of an antidepressant-like effect. The test was performed 20 min after administration of all potassium channel modulators used and 30 min after tricyclic antidepressants injection. Concerning ODNs experiments, the test was performed 72 h and 7 days after the last i.c.v. injection of either vector or ODNs.
Spontaneous activity meter (Animex)
Locomoter activity in mice was quantified using an Animex activity meter Type S (LKB, Farad, Sweden) set to maximum sensitivity. Every movement of mice, which were placed on the top of the Animex activity meter, produced a signal due to variation in inductance and capacity of the apparatus resonance circuit. Then signals were automatically converted to numbers. On the day of the experiment the mice were treated and then the cage, containing five mice, was put on the measuring platform. Activity counts were made for 10 min starting 15 min after injection of the drug. Because of the arbitrary scale adopted to quantify movements, drug-treated mice were always compared with saline-treated ones. The test was performed 15 min after administration of all potassium channel modulators used and tricyclic antidepressants. Concerning ODNs experiments, the test was performed 72 h and 7 days after the last i.c.v. injection of either vector or ODNs.
Rota-rod test
The apparatus consisted of a base platform and a rotating rod 3 cm in diameter and with a non-slippery surface. This rod was placed at a height of 15 cm from the base. The rod, 30 cm in length, was divided into five equal sections by six disks. Thus up to five mice were tested simultaneously on the apparatus, with a rod-rotating speed of 16 r.p.m. The integrity of motor coordination was assessed on the basis of endurance time of the animals on the rotating rod.
One day before the test, the animals were trained twice. On the day of the test only the mice that were able to stay balanced on the rotating rod between 80 and 120 s (cut-off-time) were selected for testing. The performance time was measured before and at various times after treatment. The test was performed 15 min after administration of all potassium channel modulators used and tricyclic antidepressants. Concerning ODNs experiments, the test was performed 72 h and 7 days after the last i.c.v. injection of either vector or ODNs.
Antisense oligonucleotides
The 24mer phosphodiester oligonucleotides were capped by a terminal phosphorothioate double substitution and purified by chromatography (Genosys, The Woodlands, U.S.A.). the antisense ODN (5′-CGA CAT CAC CGT CAT GAT GAA AGC-3′) was designed to target the 5′ portion of the murine Kv1.1 (mKv1.1) mRNA, residues 575–598 of the published cDNA sequence (Chandy et al., 1990). A fully degenerate 24mer ODN was used as control.
Mice were randomly assigned to antisense oligodeoxyribonucleotide (mKv1.1 aODN), degenerate oligodeoxyribonucleotide (mKv1.1 dODN) or vector groups. 100, 200 or 600 μM ODNs were preincubated at 37°C for 30 min with 13 μM DOTAP (N-[1-(2,3-dioleoyloxy)propyl]-N,N,N-trimethylammonium methyl sulphate), used as vector. Each group received a single i.c.v. injection on days 1, 4 and 7. The behavioural tests were performed 72 h and 7 days after the last i.c.v. injection of either vector or ODNs.
Drugs
The following drugs were used: minoxidil, pinacidil, tetraethylammonium chloride, apamin, charybdotoxin, imipramine hydrochloride (RBI); gliquidone (Boehringer Ingelheim); cromakalim, amitriptyline hydrochloride (Sigma).
Drugs were dissolved in isotonic (NaCl 0.9%) saline solution, with the exception of the highest doses used of pinacidil, minoxidil and cromakalim that were dissolved in a water and dimethyl sulphoxide (DMSO) (3 : 1) vehicle, immediately before use. Drug concentrations were prepared in such a way that the necessary dose could be administered in a volume of 5 μl per mouse by i.c.v. injection and 10 ml kg−1 by s.c. injection.
The potassium channel modulators were administered i.c.v. to avoid peripheral effects. Moreover, since the compounds are poorly able to cross the blood-brain barrier and apamin and charybdotoxin are peptides and therefore easily degradable by systemic administration, the i.c.v. administration route appears to be the most appropriate.
In every behavioural experiment, each treated animal was i.c.v. injected with a potassium channel modulator and s.c. injected with a tricyclic antidepressant. Therefore, control animals received two injections (saline i.c.v.+saline s.c. or vehicle i.c.v.+saline s.c.) in order to reproduce the same experimental conditions in both treated and control groups. Since each control (saline or vehicle) animal received two injections, there are only two control groups; saline or vehicle group, and there are no subgroups (i.e. saline s.c., saline i.c.v. etc).
Statistical analysis
All experimental results are given as the means±s.e.mean. Analysis of variance (ANOVA), followed by Fisher's Protected Least Significant Difference (PLSD) procedure for post-hoc comparison, was used to verify significance between two means. The statistical significances for each compound have been calculated versus the corresponding control treatment (saline or vehicle). The data have not been pooled, but in each Figure is represented just one value for controls since there was not significant difference among groups. Data were analysed with the StatView software for the Macintosh (1992). P values of less than 0.05 were considered significant.
Results
Reduction of immobility time by potassium channel blockers on forced swimming test
The dose response curve for tetraethylammonium (TEA, 0.1–5 μg per mouse i.c.v.), apamin (0.3–3 ng per mouse i.c.v.), charybdotoxin (0.1–1 μg per mouse i.c.v.) and gliquidone (1–6 μg per mouse i.c.v.) in the mouse forced swimming test were reported in Figure 1. All compounds, injected 20 min before the test, produced a dose-dependent reduction of the immobility time of mice. The maximum effect of TEA, apamin, charybdotoxin and gliquidone was reached, respectively, at the doses of 5, 0.003, 1 and 6 μg per mouse i.c.v. The reductions in immobility time were comparable to that produced by the well known antidepressant drugs amitriptyline (15 mg kg−1 s.c.) and imipramine (30 mg kg−1 s.c.), administered 30 min before the test (Figure 1). Higher doses of TEA, charybdotoxin and gliquidone were not investigated since the maximum effect was already reached. Doses of apamin higher than 3 ng per mouse i.c.v. induced convulsions and were therefore not investigated.
Figure 1.
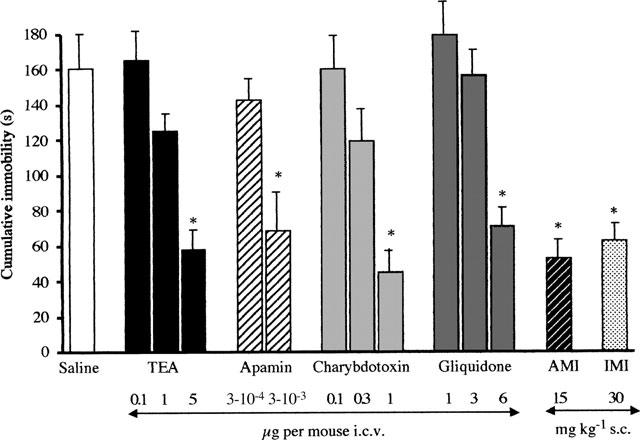
Effect of tetraethylammonium (TEA), apamin, charybdotoxin, gliquidone, amitriptyline (AMI) and imipramine (IMI) in the mouse forced swimming test. Potassium channel blockers were administered 20 min before test whereas amitriptyline and imipramine were injected 30 min before test. *P<0.01 versus saline-treated mice (see statistical analysis session). Vertical lines give s.e.mean. Each column represents the mean of at least 12 mice.
Enhancement of immobility time by potassium channel openers on forced swimming test
The dose response curves for pinacidil (3–20 μg per mouse i.c.v.), minoxidil (1–20 μg per mouse i.c.v.) and cromakalim (1–30 μg per mouse i.c.v.) in the mouse forced swimming test were reported in Figure 2. All compounds, injected 20 min before the test, produced dose-dependent increases in immobility time reaching maximum effect at the dose of 10 μg per mouse i.c.v. for minoxidil and pinacidil, and 20 μg per mouse i.c.v. for cromakalim (Figure 2). Higher doses of the three potassium channel openers, because of their poor solubility in water, needed to be dissolved in a vehicle comprising of water and DMSO 3 : 1. However, a stronger effect on forced swimming test was not obtained. A dose of pinacidil (20 μg per mouse i.c.v.), minoxidil (20 μg per mouse i.c.v.) and cromakalim (30 μg per mouse i.c.v.), higher than the maximal dose soluble in water, produced an effect on mouse immobility time comparable to that obtained with the highest dose soluble in water (Figure 2). The vehicle employed did not modify mouse immobility time in comparison with saline control when given alone (Figure 2).
Figure 2.
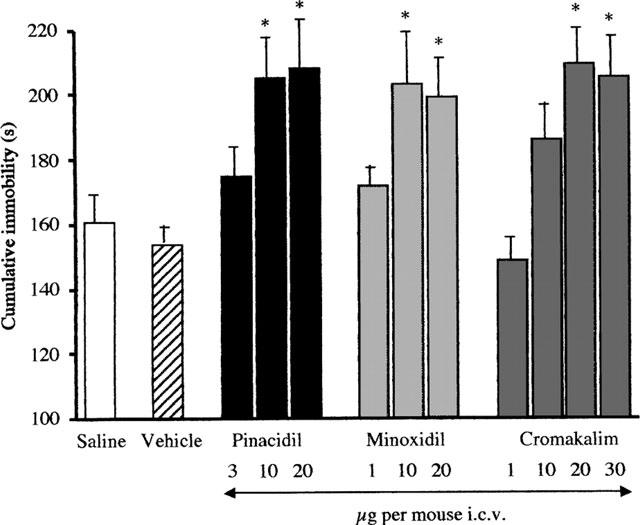
Effect of pinacidil, minoxidil and cromakalim in the mouse forced swimming test. The test was performed 20 min after treatment. *P<0.01 versus saline or vehicle-treated mice. Vertical lines give s.e.mean. Each column represents the mean of at least 14 mice. The statistical significance of all drugs has been performed versus vehicle group. The saline value reported has been inserted to indicate that there is no difference between saline and vehicle treatments.
Effect of aODN to mKv1.1 in the mouse forced swimming test
Repeated administration of mKv1.1 aODN produced a dose-dependent increase in immobility time as illustrated in Figure 3.
Figure 3.
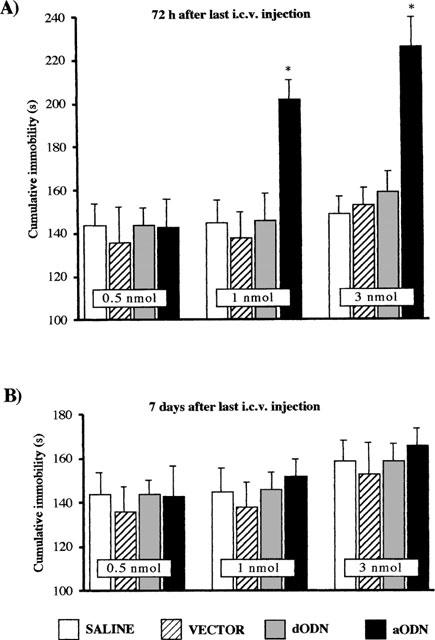
Effect of mKv1.1 aODN potassium channel in the mouse forced swimming test 72 h (A) and 7 days (B) after the last i.c.v. injection. *P<0.01 versus saline or vector-treated mice (see statistical analysis section). Vertical lines give s.e.mean. The number of mice was between 17–21 mice. See Methods for administration schedule.
Seventy-two hours after the last i.c.v. injection, 0.5 nmol mKv1.1 aODN was devoid of any effect while doses of 1 and 3 nmol of mKv1.1 aODN increased the mouse immobility time in the forced swimming test in comparison with vector-treated mice (Figure 3A). The mKv1.1 dODN, in the same experimental conditions, produced no effect as compared with either saline or vector alone (Figure 3A). In contrast, at day 7, the increasing effect on immobility time produced by mKv1.1 aODN at the doses of 1 and 3 nmol per i.c.v. injection disappeared (Figure 3B).
Effect of potassium channel modulators on mouse rota rod test and Animex apparatus
It should be noted that the potassium channel blockers (TEA, apamin, charybdotoxin, gliquidone), potassium channel openers (minoxidil, pinacidil, cromakalim), the tricyclic antidepressants (amitriptyline, imipramine) and the mKv1.1 aODN under investigation elicited their modulatory effects on immobility time in the forced swimming test without changing either gross behaviour or motor coordination as revealed by the rota rod test (Table 1).
Table 1.
Effect of potassium channel modulators in the mouse rota rod test
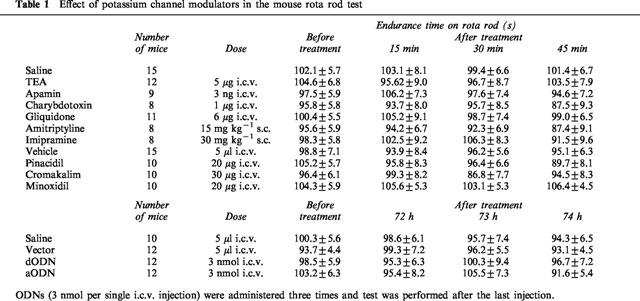
None of the drugs, administered at the highest active doses, decreased endurance time on the rotating rod in comparison with saline and vehicle-treated mice (Table 1). Furthermore, repeated administration of mKv1.1 aODN did not modify the time spent by mice on the rotating rod in comparison with mKv1.1 dODN- and vector-treated animals (Table 1).
The spontaneous motility of mice was unmodified by administration of the above-mentioned potassium channel modulators and aODN as revealed by the Animex apparatus in comparison with saline-, vehicle- and vector-treated mice (Figure 4).
Figure 4.
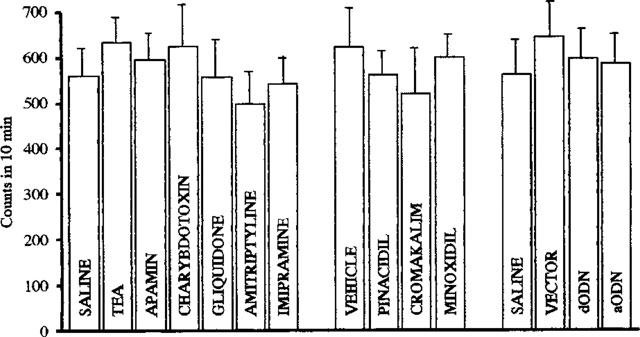
Effect of TEA (5 μg, per mouse i.c.v.), apamin (3 ng per mouse i.c.v.), charybdotoxin (1 μg per mouse i.c.v.), gliquidone (6 μg per mouse i.c.v.), amitriptyline (15 mg kg−1 s.c.), imipramine (30 mg kg−1 s.c.), pinacidil (20 μg per mouse i.c.v.), cromakalim (30 μg per mouse i.c.v.), minoxidil (20 μg per mouse i.c.v.), aODN (3 nmol per single i.c.v. injection) and dODN (3 nmol per single i.c.v. injection) in the mouse Animex apparatus test. The test was performed respectively 20 min after treatment of potassium channels modulators, 30 min after antidepressants and 72 h after the last i.c.v. injection of ODNs. Vertical lines give s.e.mean. Each column represents the mean of 15 mice. See Methods for aODN administration schedule. Since each control (saline or vehicle) animals received two injections, there are only two control groups: saline or vehicle group, and there are no subgroups (i.e. saline s.c., saline i.c.v. etc).
Discussion
Potassium channel modulators appear to be involved in the modulation of depressive states as evidenced by the forced swimming test in mice. Our results demonstrate that the administration of potassium channel blockers (TEA, apamin, charybdotoxin, gliquidone) provoke a reduction of mouse immobility time of intensity comparable to that produced by the tricyclic antidepressants amitriptyline and imipramine. By contrast, the potassium channel openers (minoxidil, pinacidil, cromakalim) increased the time of immobility in the same experimental conditions. Therefore, from the present results, the potassium channel functionally appears fundamental in the modulation of the mouse immobility time.
The forced swimming test is widely used to predict the antidepressant action of drugs in humans. The immobility time of mice in this test is reduced by the majority of antidepressants including tricyclic and atypical antidepressants, MAO inhibitors and 5-HT uptake inhibitors (Porsolt et al., 1977; Bourin et al., 1991) and their effectiveness correlates significantly with clinical potency (Willner, 1984). However, this animal model has also some drawbacks represented by the possibility to obtain some false positives or negatives (Borsini & Meli, 1988; Detke & Lucki, 1996). Drugs enhancing motor activity, such as anticholinergics and antihistaminics, may give a ‘false' positive effect in the forced swimming test and antidepressants such as bupropion, nomifensine and amineptine would then be rejected since they increase motor activity (Borsini & Meli, 1988).
An antidepressant-like effect was produced in mice by blocking the KATP potassium channels whereas a depressant-like effect was obtained by opening KATP potassium channels. This is the first evidence that KATP potassium channel modulators influence immobility time when given alone. So far, only a potentiation by glyburide, a KATP potassium channel blocker, or an antagonism by cromakalim, a KATP potassium channel opener, of the anti-immobility activity of some antidepressant drugs has been reported (Guo et al., 1996; Redrobe et al., 1996).
However, an anti-immobility effect is also obtained by antagonizing other types of potassium channels such as calcium-activated channels. Apamin and charybdotoxin, two calcium-activated potassium channel blockers (Miller et al., 1985; Cook, 1988), dose-dependently reduced the time of immobility in the forced swimming test producing an antidepressant-like effect comparable to that observed with the tricyclic antidepressants amitriptyline and imipramine.
The present study also provides evidence for the involvement of voltage-gated potassium channels in the induction of an antidepressant-like activity. The administration of TEA, a blocker of voltage-gated potassium channels, reduced immobility time to an extent similar to tricyclic antidepressants. The role of voltage-gated potassium channels in the relief of a depressive state is also confirmed by the inhibition of delayed-rectifier current (KV) and transient current (KA) by tricyclic antidepressants (Wooltorton & Mathie, 1993; 1995). Furthermore, Tytgat et al. (1997) reported that the antidepressant drug fluoxetine blocks Kv1.1 when expressed in oocytes of Xenopus laevis. Molecular cloning has confirmed the presence of at least four subfamilies of voltage-gated potassium channel genes, homologous to the Drosophila genes Shaker, Shab, Shaw and Shal, in various mammalian species (Tempel et al., 1988; Christie et al., 1989; Stühmer et al., 1989; Chandy et al., 1990; Swanson et al., 1990; Pak et al., 1991). The mouse Kv1.1 is, a Shaker-like potassium channel widely distributed in the central nervous system (Wang et al., 1994) endowed with delayed rectifier properties (Hopkins & Tempel, 1992) and primarily involved in the repolarization of many types of neurones (Wang et al., 1994). In order to better elucidate the role of Kv1.1 potassium channel in promoting a depressant-like condition, an aODN that specifically inhibited mKv1.1 gene expression was used in the mouse forced swimming test. Repeated administration of anti-mKv1.1 ODN produced a dose-dependent increase in immobility time confirming the importance of a complete integrity and functionality of Kv1.1 in the control of mood.
The depressant-like effect produced by mKv1.1 aODN treatment disappeared 7 days after the last i.c.v. injection of the aODN. This return of sensitivity implies both the total reversal of aODN-induced specific inhibition of mKv1.1 gene expression and a lack of damage or toxicity associated with aODN treatment. In comparison with saline i.c.v. treatment, dODN and vector treatments did not affect immobility time, ruling out the possibility that the effect exerted by aODN could be caused by a sequence-independent action on cerebral structures. This claim is supported by previous results obtained from the quantitative RT–PCR analysis of ODN effects on mKv1.1 gene expression as compared with those on the housekeeping β-actin gene. dODN modified neither mKv1.1 nor β-actin mRNA brain levels, whereas the anti-mKv1.1 aODN specifically lowered mKv1.1 mRNA brain levels in a dose-dependent manner and did not affect β-actin mRNA brain levels (Galeotti et al., 1997). The choice of the range of doses of aODN used in the present study (0.5–3 nmol per single i.c.v. injection) appeared appropriate since they are able not only to reduce mKv1.1 mRNA brain levels, but also to influence different behavioural paradigms such as learning and memory (Meiri et al., 1997) and food intake (Ghelardini et al., 1997).
Since drugs which modify motor activity may give false positive or negative effect, it is suitable to carry out a test to check this aspect, in parallel with forced swimming test. The highest doses of the drugs used in the present work were the maximum that did not produce behavioural side effects. The potassium channel modulators, as well as the tricyclic antidepressant amitriptyline and imipramine, at the highest doses used, did not modify animals gross behaviour. All the substances used were tested on the rota-rod test before the forced swimming test was performed, to make sure that they did not influence the normal motor coordination of the mice. Since ataxic mice are not able to coordinate movements and fall from the rotating rod, while excited animals tend to jump off the rod, the good performance on the rod by mice in the present study indicates the results obtained with the forced swimming test are not due to altered motor activity induced by substances as the doses used. Furthermore, not only altered motor coordination but also a modified spontaneous motility could lead to a misinterpretation of the results obtained in the forced swimming test. An influence of the substances used on spontaneous motility has, therefore, been excluded by using the Animex apparatus. Moreover, drugs which have a known psychostimulant effect, like (+)-amphetamine and caffeine, at the same doses at which they are able to decrease the time of immobility in the rat forced swimming test, also show a significant increased motor activity in an open field (Porsolt et al., 1978).
In our experimental conditions, the mKv1.1 aODN did not cause any detectable modification in gross behaviour of mice. Moreover, the motor coordination and spontaneous motility of mice treated with the highest dose of mKv1.1 aODN were unimpaired as compared with mKv1.1 dODN- and vector-treated groups of naïve mice.
In conclusion, these results suggest potassium channels play an important role in the modulation of immobility time in mice swimming test and that more than one type of potassium channel is involved. It is hypothesized that potassium channel openers induce a depressant effect through an hyperpolarization of neurones whereas potassium channel blockers produce an antidepressant-like effect by inhibiting the hyperpolarization thus leading to an increased excitatory response.
Acknowledgments
This work was supported by grants from MURST.
Abbreviations
- TEA
tethraethylammonium
- aODN
antisense oligonucleotide
References
- ARONSON J.K. Potassium channels in nervous tissue. Biochem. Pharmacol. 1992;43:11–14. doi: 10.1016/0006-2952(92)90653-z. [DOI] [PubMed] [Google Scholar]
- BORSINI F., MELI A. Is the forced swimming test a suitable model for revealing antidepressant activity. Psychopharmacology. 1988;94:147–160. doi: 10.1007/BF00176837. [DOI] [PubMed] [Google Scholar]
- BOURIN M., COLOMBEL M.C., MALIGNE M, BRADWEJN J. Clonidine as a sensitizing agent in the forced swimming test for revealing antidepressant activity. J. Psychiatr. Neurosci. 1991;16:199–203. [PMC free article] [PubMed] [Google Scholar]
- CHANDY K.G., WILLIAMS C.B., SPENCER R.H., AGUILAR B.A., GHANSHANI S., TEMPEL B.L., GUTMAN G.A. A family of three mouse potassium channel genes with intronless coding regiones. Science. 1990;247:973–975. doi: 10.1126/science.2305265. [DOI] [PubMed] [Google Scholar]
- CHERUBINI E., NORTH R.A., SUPRENANT A. Quinine blocks a calcium-activated potassium conductance in mammalian enteric neurons. Br. J. Pharmacol. 1984;83:3–5. doi: 10.1111/j.1476-5381.1984.tb10112.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CHRISTIE M.J. Molecular and functional diversity of K+ channels. Clin. Exp. Pharmacol. Physiol. 1995;22:944–951. doi: 10.1111/j.1440-1681.1995.tb02331.x. [DOI] [PubMed] [Google Scholar]
- CHRISTIE M.J., ADELMAN J.P., DOUGLASS J., NORTH R.A. Expression of a cloned rat brain potassium channel in Xenopus oocytes. Science. 1989;244:221–224. doi: 10.1126/science.2539643. [DOI] [PubMed] [Google Scholar]
- COOK N.S. The pharmacology of potassium channels and their therapeutic potential. Trend Pharmacol. Sci. 1988;9:21–28. doi: 10.1016/0165-6147(88)90238-6. [DOI] [PubMed] [Google Scholar]
- COOK N.S., QUAST U.Potassium channel pharmacology Potassium channels: Structure, classification, function and therapeutic potential 1990Chichester: Ellis Horwood Limited; 181–255.ed. Cook, N.S. [Google Scholar]
- DETKE M.J., LUCKI I. Detection of serotoninergic and noradrenergic antidepressants in the rat forced swimming test: the effects of water depth. Behav. Brain Res. 1996;73:43–46. doi: 10.1016/0166-4328(96)00067-8. [DOI] [PubMed] [Google Scholar]
- EDWARDS G., WESTON A.H. The pharmacology of ATP-sensitive potassium channels. Annu. Rev. Pharmacol. Toxicol. 1993;33:597–637. doi: 10.1146/annurev.pa.33.040193.003121. [DOI] [PubMed] [Google Scholar]
- GALEOTTI N., GHELARDINI C., PAPUCCI L., QUATTRONE A., CAPACCIOLI S., BARTOLINI A. An antisense oligonucleotide to the mouse Shaker-like potassium channel Kv1.1 gene prevents the antinociception induced by morphine and baclofen. J. Pharmacol. Exp. Ther. 1997;281:941–949. [PubMed] [Google Scholar]
- GHELARDINI C., GALEOTTI N., PECORI VETTORI A., CAPACCIOLI S., QUATTRONE A., BARTOLINI A. Effect of potassium channels modulation on mouse feeding behaviour. Eur. J. Pharmacol. 1997;329:1–8. doi: 10.1016/s0014-2999(97)10102-9. [DOI] [PubMed] [Google Scholar]
- GUO W., TODD K.G., BOURIN M., HASCOET M. The additive effect of quinine on antidepressant drugs in the forced swimming test in mice. Psychopharmacology. 1995;121:173–179. doi: 10.1007/BF02245627. [DOI] [PubMed] [Google Scholar]
- GUO W., TODD K.G., BOURIN M., HASCOET M., KOUADIO F. Additive effects of glyburide and antidepressants in the forced swimming test: evidence for the involvement of potassium channel blockade. Pharmacol. Biochem. Behav. 1996;54:725–730. doi: 10.1016/0091-3057(95)02226-0. [DOI] [PubMed] [Google Scholar]
- HALEY T.J., MCCORMICK W.G. Pharmacological effects produced by intracerebral injection of drugs in the conscious mouse. Br. J. Pharmacol. Chemother. 1957;12:12–15. doi: 10.1111/j.1476-5381.1957.tb01354.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HALLIWELL J.V.K+ channels in the central nervous system Potassium channels: Structure, classification, function and therapeutic potential 1990Chichester: Ellis Horwood Limited; 348–381.ed. Cook, N.S. [Google Scholar]
- HOPKINS W.F., TEMPEL B.L. Members of a mouse subfamily of genes encoding voltage-gated potassium channel subunits form heteromultimers when coexpressed in Xenopus oocytes. Soc. Neurosci. Abstr. 1992;18:1093. [Google Scholar]
- LONGMAN S.D., HAMILTON T.C. Potassium channel activator drugs: mechanism of action, pharmacological properties, and therapeutic potential. Med. Res. Rev. 1992;12:73–148. doi: 10.1002/med.2610120202. [DOI] [PubMed] [Google Scholar]
- MEIRI N., GHELARDINI C., TESCO G., GALEOTTI N., DAHAL D., TOMSIC D., CAVALLARO S., QUATTRONE A., CAPACCIOLI S., BARTOLINI A., ALKON D.L. Antisense inhibition of the rodent Shaker-like potassium channel Kv1.1: disruption of memory without effects on LTP. Proc. Natl. Acad. Sci. USA. 1997;94:4430–4434. doi: 10.1073/pnas.94.9.4430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MILLER C., MOCZYDLOWSKI E., LATORRE R., PHILLIPS M. Charybdotoxin, a protein inhibitor of single Ca++ activated K+ channels from mammalian skeletal muscle. Nature. 1985;313:316–318. doi: 10.1038/313316a0. [DOI] [PubMed] [Google Scholar]
- NIXON M.K., HASCOET M., BOURIN M., COLOMBEL M.C. Additive effects of lithium and antidepressants in the forced swimming test: Further evidence for the involvement of the serotoninergic system. Psychopharmacology. 1994;115:59–64. doi: 10.1007/BF02244752. [DOI] [PubMed] [Google Scholar]
- PAK M.D., COVARRUBIAS M., RATCLIFFE A., SALKOFF L. A mouse brain homolog of the Drosophila Shab K+ channel with conserved delayed-rectifier properties. J. Neurosci. 1991;11:869–880. doi: 10.1523/JNEUROSCI.11-03-00869.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- PORSOLT R.D., ANTON G., BLAVET M., JALFRE M. Behavioural despair in rats: a new model sensitive to antidepressant treatments. Eur. J. Pharmacol. 1978;47:379–391. doi: 10.1016/0014-2999(78)90118-8. [DOI] [PubMed] [Google Scholar]
- PORSOLT R.D., BERTIN A., JALFRE M. Behavioural despair in mice: A primary screening test for antidepressants. Arch. Int. Pharmacodyn. 1977;229:327–336. [PubMed] [Google Scholar]
- REDROBE J.P., PINOT P., BOURIN M. The effect of the potassium channel activator, cromakalim, on antidepressant drugs in the forced swimming test in mice. Fund. Clin. Pharmacol. 1996;10:524–528. doi: 10.1111/j.1472-8206.1996.tb00610.x. [DOI] [PubMed] [Google Scholar]
- STRONG P.N. Potassium channel toxins. Pharmacol. Ther. 1990;46:137–162. doi: 10.1016/0163-7258(90)90040-9. [DOI] [PubMed] [Google Scholar]
- STÜHMER W., RUPPERSBERG J.P., SCHRÖTER K.H., SAKMANN B., STOCKER M., GIESE K.P., PERSCHKE A., BAUMANN A., PONGS O. Molecular basis of functional diversity of voltage-gated potassium channels in mammalian brain. EMBO J. 1989;8:3235–3244. doi: 10.1002/j.1460-2075.1989.tb08483.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SWANSON R., MARSHALL J., SMITH J.S., WILLIAMS J.B., BOYLE M.B., FOLANDER K., LUNEAU C.J., ANTANAVAGE J., OLIVA C., BUHROW S.A., BENNET C., STEIN R.B., KACZMAREK L.K. Cloning and expression of cDNA and genomic clones encoding three delayed rectifier potassium channels in rat brain. Neuron. 1990;4:929–939. doi: 10.1016/0896-6273(90)90146-7. [DOI] [PubMed] [Google Scholar]
- TEMPEL B.L., JAN Y.N., YAN L.Y. Cloning of a probable potassium channel gene from the mouse brain. Nature. 1988;332:837–839. doi: 10.1038/332837a0. [DOI] [PubMed] [Google Scholar]
- TYTGAT J., MAERTENS C., DAENENS P. Effect of fluoxetine on a neuronal, voltage-dependent potassium channel (Kv1.1) Br. J. Pharmacol. 1997;122:1417–1424. doi: 10.1038/sj.bjp.0701545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WANG H., KUNKEL D.D., SCHWARTZKROIN P.A., TEMPEL B.L. Localization of Kv1.1 and Kv1.2, two K channel proteins, to synaptic terminals, somata, and dendrites in the mouse brain. J. Neurosci. 1994;14:4588–4599. doi: 10.1523/JNEUROSCI.14-08-04588.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WILLNER P. The validity of animal models for depression. Psychopharmacology. 1984;83:1–16. doi: 10.1007/BF00427414. [DOI] [PubMed] [Google Scholar]
- WOOLTORTON J.R.A., MATHIE A. Block of potassium currents in rat sympathetic neurones by tricyclic antidepressants and structurally related compounds. Br. J. Pharmacol. 1993;110:1126–1132. doi: 10.1111/j.1476-5381.1993.tb13931.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WOOLTORTON J.R.A., MATHIE A. Potent block of potassium currents in rat isolated sympathetic neurones by the uncharged form of amitriptyline and related tricyclic compounds. Br. J. Pharmacol. 1995;116:2191–2200. doi: 10.1111/j.1476-5381.1995.tb15053.x. [DOI] [PMC free article] [PubMed] [Google Scholar]


