Abstract
Airway smooth muscle thickening is a characteristic feature of airway wall remodelling in chronic asthma. We have investigated the role of the leukotrienes in airway smooth muscle (ASM) and epithelial cell DNA synthesis and ASM thickening following repeated allergen exposure in Brown Norway rats sensitized to ovalbumin.
There was a 3 fold increase in ASM cell DNA synthesis, as measured by percentage bromodeoxyuridine (BrdU) incorporation, in repeatedly ovalbumin-exposed (4.1%, 3.6–4.6; mean, 95% c.i.) compared to chronically saline-exposed rats (1.3%, 0.6–2.1; P<0.001).
Treatment with a 5-lipoxygenase enzyme inhibitor (SB 210661, 10 mg kg−1, p.o.) and a specific cysteinyl leukotriene (CysLT1) receptor antagonist, pranlukast (SB 205312, 30 mg kg−1, p.o.), both attenuated ASM cell DNA synthesis. Treatment with a specific leukotriene B4 (BLT) receptor antagonist (SB 201146, 15 mg kg−1, p.o.) had no effect.
There was also a significant, 2 fold increase in the number of epithelial cells incorporating BrdU per unit length of basement membrane after repeated allergen exposure. This response was not inhibited by treatment with SB 210661, pranlukast or SB 201146.
A significant increase in ASM thickness was identified following repeated allergen exposure and this response was attenuated significantly by SB 210661, pranlukast and SB 201146.
Rats exposed to chronic allergen exhibited bronchial hyperresponsiveness to acetylcholine and had significant eosinophil recruitment into the lungs. Treatment with SB 210661, pranlukast or SB 201146 significantly attenuated eosinophil recruitment into the lungs, whilst having no significant effect on airway hyperresponsiveness.
These data indicate that the cysteinyl leukotrienes are important mediators in allergen-induced ASM cell DNA synthesis in rats, while both LTB4 and cysteinyl leukotrienes contribute to ASM thickening and eosinophil recruitment following repeated allergen exposure.
Keywords: Allergic reactions, cysteinyl leukotrienes, airway smooth muscle, DNA synthesis, eosinophils, airway remodelling
Introduction
Asthma is a disease characterized by chronic inflammation, recruitment of pro-inflammatory cells into the airways and airway wall remodelling. Airway remodelling is distinguished by a number of processes including increased airway smooth muscle mass (Dunnill et al., 1969), epithelial cell disruption (Laitinen et al., 1985) and sub-epithelial fibrosis (Roche et al., 1989). Increased airway smooth muscle mass consists of both smooth muscle cell hyperplasia and hypertrophy (Heard & Hossain 1973; Ebina et al., 1993), and may contribute to bronchial narrowing and airway hyperresponsiveness.
Little is known about the mediators that induce changes in airway wall structure in vivo, but there is evidence to suggest that cysteinyl leukotrienes may be important in this process. The cysteinyl leukotrienes (CysLT: LTC4, LTD4 and LTE4) are lipid mediators that are present at elevated concentrations in the bronchoalveolar lavage fluid of asthmatics (Lam et al., 1988; Wardlaw et al., 1989). They also cause potent contraction of airway smooth muscle (Dahlén et al., 1980; Barnes et al., 1984), increase microvascular leakage (Woodward et al., 1983; Hua et al., 1985), stimulate mucus hypersecretion (Marom et al., 1982; Johnson & McNee, 1983) but decrease mucocilliary clearance (Bisgaard & Pedersen 1987), and also promote eosinophil accumulation (Laitinen et al., 1993; Spada et al., 1994). LTD4 also potentiates human airway smooth muscle cell proliferation in vitro (Panettieri et al., 1998b).
In Brown Norway rats, as few as three allergen exposures have been reported to increase airway smooth muscle mass (Sapienza et al., 1991). LTD4 has been implicated in allergen-induced airway smooth muscle thickening following repeated challenge (Wang et al., 1993) and cysteinyl leukotriene breakdown products have been demonstrated to be increased after allergen exposure in Brown Norway rats (Martin et al., 1993). Increased rates of DNA synthesis in both airway smooth muscle cells and epithelial cells have been observed in models of chronic allergen exposure using sensitized Brown Norway rats (Salmon et al., 1997; Panettieri et al., 1998a).
In the current study, we have investigated the influence of the leukotrienes on bronchial responsiveness to acetylcholine, DNA synthesis in airway smooth muscle and epithelial cells, smooth muscle area and eosinophil recruitment in a repeated allergen exposure model. We have used a 5-lipoxygenase (5-LO; E.C. 1.13.11.34) inhibitor, SB 210661 (McLoughlin et al., 1998), and a cysteinyl leukotriene (CysLT1) receptor antagonist, pranlukast (SB 205312, ONO-1078) (Obata et al., 1992; Fujiwara et al., 1993; Taki et al., 1994), in order to evaluate the role of CysLT in bronchial responsiveness and airway wall remodelling. In addition, the potential contribution of another 5-LO product, leukotriene B4, in these processes has been studied using a specific LTB4 (BLT) receptor antagonist, SB 201146 (Daines et al., 1993, 1996).
Methods
Sensitization and challenges
Pathogen-free, male Brown Norway rats weighing 220–250 g (Harlan, Bicester, U.K.) were sensitized on days 1, 2 and 3 using 1 mg kg−1 intraperitoneal injections of ovalbumin in 0.9% sterile saline containing 100 mg Al(OH)3 as adjuvant. On days 6, 9, 12, 15, 18 and 21, animals were exposed to 1% ovalbumin aerosol for 20 min. Challenges took place in a 0.8 m3 chamber, with free-breathing animals exposed to either saline or a 1% ovalbumin aerosol mist produced by a DeVilbiss PulmoSonic nebulizer (DeVilbiss Health Care Ltd., Feltham, U.K.). The aerosol mist was pumped at a rate of 0.6 l min−1 by a small animal ventilator. At all other times, rats were housed in a caging system receiving clean filtered air (Maximiser, Thorens Caging System Inc., Hazleton, PA, U.S.A.). Five groups of animals were studied in total (n=6 in each group):
group A: ovalbumin-sensitized, saline-challenged, vehicle-treated;
group B: ovalbumin-sensitized, ovalbumin-challenged, vehicle-treated;
group C: ovalbumin-sensitized, ovalbumin-challenged, SB 210661-treated;
group D: ovalbumin-sensitized, ovalbumin-challenged, SB 201146-treated;
group E: ovalbumin-sensitized, ovalbumin-challenged, pranlukast-treated.
Drug pre-treatments
All oral drug pre-treatments were given at a time prior to allergen challenges such that blood plasma levels were maximal or near-maximal at the time of the challenge. A second dose was then administered so that drug plasma levels were near-maximal approximately 7 h after the allergen challenge. The 5-LO inhibitor, SB 210661, was administered 1 h before allergen challenge at a dose of 10 mg kg−1 (in 1 ml of a 1 : 1 solution of 10% polyethyleneglycol 200 and 1% methylcellulose). The BLT receptor antagonist, SB 201146, was administered 30 min before allergen challenge at a dose of 15 mg kg−1 (in 1 ml of 0.9% saline). The CysLT1 receptor antagonist, pranlukast, was administered 2 h before allergen challenge at a dose of 30 mg kg−1 (in 1 ml of a 1 : 1 solution of 0.5 M NaOH and polyethyleneglycol 400). Vehicle-treated rats received 1 ml of the same vehicle at the same time points as those rats receiving drugs.
Bromodeoxyuridine dosing
5-Bromo-2′-deoxyuridine (BrdU; Sigma Chemical Co., Poole, U.K.) was dissolved in dimethylsulphoxide (DMSO) and diluted with sterile water, giving a final concentration of DMSO <7%. Rats were injected intraperitoneally with 50 mg kg−1 BrdU in 1 ml of solution immediately following each allergen challenge and received a second dose 8 h later (total of 12 injections).
Measurement of bronchial responsiveness to acetylcholine
Bronchial responsiveness was measured 18–24 h after the final allergen challenge as previously described (Elwood et al., 1991). Briefly, rats were anaesthetized, tracheostomy performed and lung resistance calculated using a computer program (LabVIEW 2, National Instruments, Austin, TX, U.S.A.) by the method of von Neergard & Wirz (1927). Increasing half-log10 concentrations of acetylcholine were administered by inhalation for 45 breaths and lung resistance was measured. Challenges were halted when lung resistance exceeded 200% of initial baseline values. The concentration of acetylcholine required to increase baseline resistance by 200% (PC200) was determined by linear interpolation of log concentration-lung resistance curves.
Tissue collection
Lung resistance was allowed to return to baseline levels and then rats were killed using an overdose of sodium pentobarbitone (500 mg kg−1; i.p.). The lungs were rapidly removed and insufflated with O.C.T. Tissue Tek™ mounting medium (Raymond A. Lamb, London, U.K.) diluted 1 : 1 with phosphate-buffered saline. Regions of the left and right lung lobes were mounted on cork blocks with the main bronchi uppermost, snap-frozen in melting isopentane and stored at −25°C.
Bromodeoxyuridine and α-smooth muscle actin immunohistochemistry
Cryostat sections 5 μm thick were cut and thaw-mounted onto glass slides treated with Vectabond (Vector Laboratories, Peterborough, U.K.). Left lung blocks were cut between the first and second division of the main bronchi, at an angle perpendicular and lateral to the conducting airways. Tissue sections were fixed in a 1 : 1 mixture of acetone and methanol for 10 min at 12°C and endogenous peroxidase was blocked by immersing sections in methanol containing 0.3% (v v−1) hydrogen peroxide for 20 min. A primary anti-BrdU monoclonal antibody (mAb) solution (clone BU-1; Amersham International, Buckinghamshire, U.K.) was applied at 37°C for 75 min. A secondary biotinylated rat adsorbed antiserum to mouse IgG (Vector Laboratories) was then applied followed by a 45-min incubation with a peroxidase-linked avidin-biotin complex solution (ABC-Elite kit, Vector Laboratories). BrdU-positive cells were visualized using 3,3-diaminobenzidinetetrachloride solution (Sigma) with glucose oxidase-nickel enhancement to give a black end-product (Shu et al., 1988). Sections were then rinsed and a primary anti-α-smooth muscle actin mAb (clone 1A4; Sigma) was applied for 1 h at room temperature. A secondary biotinylated rat adsorbed antiserum to mouse IgG (Vector Laboratories) was applied to the sections, followed by an avidin-biotin complex reagent conjugated to alkaline phosphatase (Vector Laboratories).
The α-smooth muscle actin staining was visualized using Sigma FAST (4chloro-2-methylbenzenediazonium/3-hydroxy-2-naphthoic acid 2,4-dimethylanilide phosphate (α-naphthol AS-MX) and Fast Red TR) in Tris buffer (Sigma) to give a red end-product. Nuclei that were not immunoreactive for BrdU were counterstained by application of the fluorescent DNA ligand 4,6-diamidino-2-phenylindole hydrochloride (DAPI; Sigma) at a concentration of 0.00001% and mounted under glass coverslips.
Tissue sections of spleen, colon and thymus were used to confirm positive BrdU immunoreactivity. Negative controls were performed on sections of lung for BrdU and α-smooth muscle actin with antibodies of the same immunoglobulin class or in the absence of primary antibody.
Eosinophil major basic protein immunohistochemistry and counting
Sections were fixed in acetone and then incubated with a mAb against human major basic protein (MBP; clone BMK-13; Monosan, Uden, The Netherlands) at a concentration of 1 : 80 for 1 h at room temperature for detection of rat eosinophils. After labelling with a biotinylated horse anti-mouse mAb, positive cells were visualized by using an avidin-biotin complex reagent conjugated to alkaline phosphatase (Vector Laboratories). Eosinophils were visualized using Sigma FAST in Tris buffer, with positive cells appearing red. Sections were counterstained with haematoxylin (BDH, Lutterworth, U.K.) and mounted under glass coverslips. Eosinophil recruitment around the airways and in parenchyma was evaluated. Eosinophil influx around the five largest airways in each lung section was assessed as the number of positive cells in the airway wall (consisting of epithelium, submucosa, smooth muscle and lamina propria), and expressed per millimetre of basement membrane length. Counts in parenchyma were assessed as the number of positive cells in five randomly selected low power fields.
Quantification of DNA synthesis and airway smooth muscle area using computer-assisted image analysis
Quantification of images was performed using a Zeiss microscope fitted for both transmitted light and fluorescence imaging, with images captured using a monochrome camera at maximum sensitivity, and analysed using an image analysis system (Sonata model, Seescan, Cambridge, U.K.).
The field of interest containing the whole airway was visualized with a ×10 objective using DAPI fluorescence and converted to a monochrome image. The airway of interest was identified and internal perimeter, cross-sectional area and breadth (feret diameter) measured. The airway was then visualized using a ×20 objective, the transmitted light image containing BrdU-positive cells captured and converted to a monochrome image. Without moving the section, a red fluorescence image of the alkaline phosphatase-Fast Red labelled α-smooth muscle actin immunoreactivity was captured. A blue fluorescence image for DAPI-positive nuclei was then captured and all three images were converted to stored monochrome images. The area of α-smooth muscle actin immunoreactivity was measured and then a mask created of this region which was overlaid onto the transmitted light image of the same area and the number of BrdU-positively stained nuclei was counted. The mask was then overlaid onto the DAPI fluorescent image and the number of nuclei counted. DNA synthesis in epithelium was measured in a similar way by creation of an epithelial mask by interactive delineation using the DAPI-fluorescence image, and then counting the number of BrdU-positive nuclei within the mask and measuring basement membrane length.
DNA synthesis in airway smooth muscle cells was measured as the number of BrdU-immunoreactive nuclei divided by the total number of nuclei (BrdU plus DAPI nuclei) within the α-smooth muscle actin-defined immunoreactive area. Epithelial DNA synthesis was measured as the number of BrdU-positive cells per unit length of basement membrane within the DAPI-defined epithelial mask. Airway smooth muscle thickness was measured as the total α-smooth muscle actin immunoreactive area around each airway per unit length of basement membrane.
Airway number and section variability
BrdU indices were measured from the five largest conducting airways in a section of lung tissue cut perpendicular and lateral to the plane of the airways between the first and second division of the main bronchi. In preliminary experiments, the airway smooth muscle BrdU index was measured in the five largest airways from five consecutive lung tissue sections from a sensitized and ovalbumin-challenged rat. The coefficient of variation for BrdU index decreased with increasing section number, but the differences were small, with a standard error of less than ±15% of the mean index when comparing one against five consecutive sections. BrdU indices in this study were therefore calculated from the five largest airways from a single lung section for each rat in all treatment groups. All counts in this study were performed with the investigator blinded to treatment group.
Analysis of data
All data are expressed as the arithmetic mean index derived from the five largest airways in one section of lung from each rat. Mean indices were statistically analysed after logarithmic transformation by one-way analysis of variance (ANOVA), followed by t-tests with Bonferroni correction used to evaluate significant differences between groups. Values are expressed as means with 95% confidence intervals, with P values of <0.05 considered significant.
Results
Bronchial responsiveness to acetylcholine
There was no significant difference in the baseline lung resistance (RL) values following saline challenge among the five groups of rats. There was a significant increase in bronchial responsiveness to acetylcholine in the sensitized and repeatedly allergen-exposed rats (PC200: 5.62 mM, 3.89–8.13) (mean, 95% c.i.) compared to sensitized and repeated-saline exposed rats (PC200: 17.3 mM, 10.2–29.5; P<0.01). There was no significant attenuation of this increased bronchial responsiveness in rats treated with either SB 210661 or SB 201146. There was a trend towards inhibition of bronchial responsiveness in the pranlukast-treated rats compared to the repeatedly allergen- and vehicle-treated group, but this was not significant (Figure 1).
Figure 1.
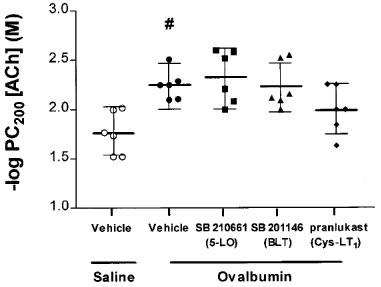
Bronchial responsiveness to acetylcholine in sensitized Brown Norway rats exposed repeatedly to saline or ovalbumin. There was a half-log order decrease in the mean −log PC200 value in repeatedly allergen-exposed rats compared to the saline-exposed group (#P<0.05). Following repeated allergen exposure, treatment with SB 210661, SB 201146 or pranlukast caused no significant attenuation of mean −log PC200 values compared to vehicle-treated rats. Data are expressed as mean −log PC200 values with error bars representing 95% confidence intervals.
Airway smooth muscle and epithelial cell DNA synthesis
The BrdU index of airway smooth muscle cells in the sensitized and repeatedly saline-challenged rats was 1.3% (0.6–2.1) and this was increased to 4.1% (3.6–4.6) in sensitized and repeatedly allergen-exposed rats (P<0.001). Treatment with the 5-LO inhibitor SB 210661 (2.0%, 1.3–2.7; P<0.01) or the CysLT1 receptor antagonist pranlukast (2.1%, 1.5–2.7; P⩾0.05) attenuated airway smooth muscle cell DNA synthesis. The BLT receptor antagonist SB 201146 did not, however, significantly inhibit airway smooth muscle cell DNA synthesis (Figure 2A).
Figure 2.
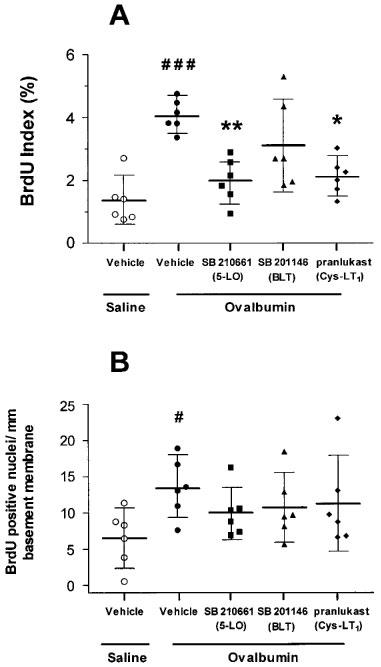
Effects of SB 210661, SB 201146 and pranlukast on airway smooth muscle cell (A) and epithelial cell (B) bromodeoxyuridine (BrdU) incorporation following repeated allergen challenge. (A) There was a greater than 3 fold increase in mean BrdU index of the airway smooth muscle cells of repeated allergen compared to repeated saline-exposed rats (###P<0.001). Treatment with SB 210661 and pranlukast but not SB 201146 caused significant attenuation of airway smooth muscle BrdU index compared to vehicle-treated rats (*P<0.05, **P<0.01). (B) There was a greater than 2 fold increase in epithelial cell BrdU incorporation of repeated allergen compared to repeated saline-exposed rats (#P<0.05). Treatment with any of the three drugs did not cause an attenuation in epithelial cell BrdU incorporation. Horizontal bars are the mean values for each group of rats with error bars representing the 95% confidence intervals.
There was a greater than 2 fold increase in epithelial cell DNA synthesis in sensitized and repeatedly allergen-exposed rats, with 13.5 BrdU-positive cells per mm basement membrane (9.3–17.7) compared to 6.6 (2.5–10.6) in sensitized and repeatedly saline-exposed rats (P<0.05). Treatment with any of the three drugs did not cause significant inhibition of epithelial cell DNA synthesis (Figure 2B).
Airway smooth muscle area and cell number
In sensitized rats, repeated allergen challenge significantly increased airway smooth muscle area compared to repeated saline challenge. The saline-challenged group had a mean airway smooth muscle thickness (defined as ASM area/basement membrane length) of 19.3 μm per mm basement membrane (17.5–21.2), compared to 25.9 μm (19.7–32.1) in the allergen-challenged group (P<0.05). There was a significant attenuation of the increased ASM thickness with SB 210661 (17.9, 15.0–20.8; P<0.05), SB 201146 (16.6, 13.9–19.3; P<0.01) and pranlukast (19.0, 13.1–24.9; P<0.05) compared to vehicle-treated and repeatedly allergen-exposed rats (Figure 3A). The total number of airway smooth muscle cells was counted to determine if increases in ASM area were due to an increase in the overall number of cells (i.e. ASM cell hyperplasia). Repeatedly allergen-exposed rats had more smooth muscle nuclei per mm basement membrane, with 82.6 (57.9–107.3) compared to 64.9 (47.9–82.0) in the saline-exposed group, but this increase was not significant (Figure 3B).
Figure 3.
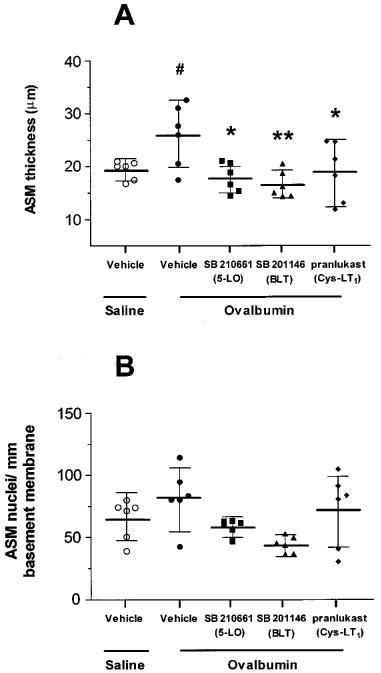
Effect of SB 210661, SB 201146 and pranlukast on airway smooth muscle thickness (A) and cell number (B). (A) Following repeated allergen exposure there was a significant increase in airway smooth muscle thickness per unit length of basement membrane compared to the repeated saline-exposed group (#P<0.05). Treatment with SB 210661, SB 201146 or pranlukast caused a significant reduction in ASM thickness compared to the vehicle-treated group (*P<0.05, **P<0.01). (B) There was no significant increase in the total number of ASM nuclei per mm of basement membrane in the repeated-allergen compared to the repeated saline-exposed group of rats.
Eosinophils in the airway wall and parenchyma
Sensitized and repeatedly allergen-exposed rats had significantly more eosinophils in the airways, with 26.9 (19.7–34.2) MBP-positive cells per mm basement membrane compared to 4.5 (1.6–7.4) in the sensitized and repeatedly saline-exposed (P<0.001). There was a significant reduction in eosinophil numbers around the airways following drug treatment with SB 210661 (14.9, 12.2–17.6; P<0.001), SB 201146 (17.4, 14.2–20.7; P<0.01) or pranlukast (12.4, 7.0–17.8; P<0.001) compared to vehicle-treated rats (Figure 4A). The trend in eosinophil migration in parenchyma was similar to that observed around the airways. The sensitized and chronically saline-exposed rats had a mean of 23.2 MBP-positive cells (10.1–36.3) in five low-power microscope fields, compared to 74.0 (44.1–103.9) in the chronically allergen-exposed group (P<0.01). Again, significant attenuation of eosinophil recruitment was observed following treatment with all three drugs (Figure 4B).
Figure 4.
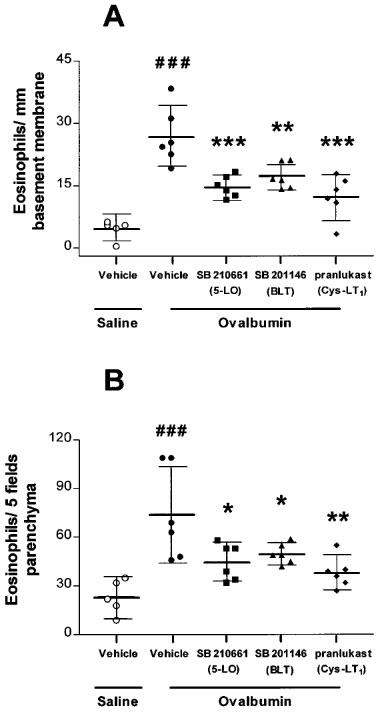
Effect of SB 210661, SB 201146 and pranlukast on eosinophil recruitment to the airways (A) and parenchyma (B). (A) There was a significant number of MBP-positive eosinophils around the airways following repeated allergen exposure of rats compared to repeated saline exposure (###P<0.001). Following treatment with SB 210661, SB 201146 or pranlukast there was a significant attenuation of eosinophil recruitment to the airways compared to the vehicle-treated group (**P<0.01, ***P<0.001). (B) There was also a significant increase in MBP-positive eosinophil recruitment into the parenchyma of repeatedly allergen-exposed compared to repeatedly saline-exposed rats (###P<0.001). Treatment with SB 210661, SB 201146 or SB 205312 caused a significant attenuation of eosinophil recruitment compared to vehicle-treated rats (*P<0.05, **P<0.01).
Discussion
Following repeated allergen exposure of sensitized Brown Norway rats we detected significant increases in both airway smooth muscle and epithelial cell DNA synthesis, as measured by BrdU incorporation, compared to saline-exposed controls. Treatment of sensitized and repeatedly allergen-exposed rats with the 5-LO inhibitor SB 210661 and the CysLT1 receptor antagonist SB 205312 (pranlukast) significantly attenuated airway smooth muscle cell DNA synthesis whereas the BLT receptor antagonist SB 201146 was without effect. These data indicate that airway smooth muscle cell DNA synthesis was attenuated by blocking the production or action of cysteinyl leukotrienes. In contrast, increased epithelial cell DNA synthesis following chronic allergen exposure was not inhibited by the 5-LO inhibitor nor by either of the receptor antagonists, indicating that factors other than leukotrienes are involved in this response.
The increase in airway smooth muscle cell DNA synthesis following repeated allergen exposure was accompanied by a significant increase in airway smooth muscle thickness. Increased ASM thickness may be due to ASM cell hyperplasia or hypertrophy, or a combination of both these events (Heard & Hossain 1973; Ebina et al., 1993). Leukotriene-enhanced ASM cell proliferation may contribute to this increased ASM thickening. Treatment with the 5-LO inhibitor or either the BLT or CysLT1 receptor antagonist significantly attenuated allergen-induced increases in ASM thickness to control levels. This indicates that leukotrienes may be important mediators in allergen-induced ASM thickening.
Eosinophil recruitment into the lungs has been reported following both single and repeated allergen exposure in sensitized Brown Norway rats (Elwood et al., 1991). In the present study, eosinophil recruitment was increased both in the airways and in parenchyma of repeatedly allergen-exposed rats. There was an attenuation of eosinophil recruitment to the airways and parenchyma after treatment with the 5-LO inhibitor (SB 210661), and BLT (SB 201146) or CysLT1 (pranlukast) receptor antagonists. The cysteinyl leukotrienes LTD4 and LTE4 stimulate human eosinophil chemotaxis in vitro (Spada et al., 1994), and stimulate the recruitment of pulmonary eosinophils in humans (Laitinen et al., 1993; Diamant et al., 1997). Further evidence in guinea-pigs indicates that this chemotactic role of cysteinyl leukotrienes for eosinophils is mediated via an interleukin-5 dependent mechanism (Underwood et al., 1996). Our data suggest that LTB4 may also be involved in eosinophil recruitment. This concept is supported by previous in vivo studies in guinea-pigs in which LTB4 administered directly to the lungs induces pulmonary eosinophilia (Silbaugh et al., 1987). Treatment with a BLT receptor antagonist, U-75302, has been demonstrated to attenuate allergen-induced eosinophil recruitment (Richards et al., 1989). Therefore, both cysteinyl leukotrienes and leukotriene B4 appear to contribute to eosinophil recruitment in the airways following chronic allergen challenge in guinea-pigs and rats.
Bronchial responsiveness to acetylcholine was significantly increased in the repeatedly allergen-exposed rats compared to the saline-exposed controls, 24 h after the final challenge. Treatment with SB 210661, SB 201146 or pranlukast caused a significant attenuation of ASM thickening and eosinophil recruitment without having a significant influence on bronchial hyperresponsiveness. Two previous studies have reported a significant attenuation of airway hyperresponsiveness following a single allergen challenge in 5-LO-deficient mice (Irvin et al., 1997), or after treatment with a CysLT1 receptor antagonist following repeated allergen challenge of sensitized rats (Wang et al., 1993). The discrepancy in these findings from our own may be due to the species and experimental differences between the studies. In both our study and that of Wang et al. (1993) attenuation of ASM thickness is reported following treatment with a CysLT1 receptor antagonist. The model we used has, however, a much more extreme protocol, with more challenges in a shorter time-period, and is one where the IgE levels of rats are still high at the time of the final allergen challenge (Pauwels et al., 1979). This may therefore produce a much greater inflammatory response, with the potential for many mediators to be involved in the genesis of bronchial hyperresponsiveness. Our data indicate that additional factors must contribute to bronchial hyperresponsiveness in this model, as attenuation of ASM thickening and eosinophil infiltration did not prevent bronchial hyperresponsiveness. This phenomenon, however, warrants further investigation.
Our evidence implicates cysteinyl leukotrienes in allergen-induced airway smooth muscle cell DNA synthesis and increases in airway smooth muscle thickening. The attenuation of increased airway smooth muscle cell DNA synthesis and thickening by the CysLT1 receptor antagonist pranlukast could be mediated through two distinct mechanisms. Firstly, this could have occurred by inhibiting direct receptor-mediated effects of cysteinyl leukotrienes to induce airway smooth muscle cell DNA synthesis. Secondly, if airway smooth muscle cell DNA synthesis was secondary to cysteinyl leukotriene release from infiltrating eosinophils, inhibition of eosinophil recruitment into the lung by cysteinyl leukotriene antagonists may indirectly reduce the capacity for further release of cysteinyl leukotrienes from migrating eosinophils. Differential effects have recently been described for three structurally different CysLT1 receptor antagonists: pranlukast, pobilukast and zafirlukast. All three compounds inhibited LTD4-induced contraction of human bronchus but only pranlukast and pobilukast inhibited the effect of LTD4 on epidermal growth factor (EGF)-induced proliferation of human ASM cells (Panettieri et al., 1998b). This indicates that subtypes of the CysLT1 receptor may exist which augment human ASM cell mitogenesis but are distinct from those which mediate LTD4-induced contraction of ASM. It is not known if this is also the case in rat ASM cells but, to date, evidence suggests that the CysLT1 receptor in rat and human have similar characteristics especially with respect to agonist and antagonist affinities. CysLT1 receptor antagonists from different structural classes have been shown to have similar potencies against cysteinyl leukotriene-induced contractions in rat lung compared to those reported for human bronchus (Tudhope et al., 1994).
Little is known about the mechanisms and the mediators influencing airway smooth muscle thickening, or the cellular processes underlying these events. Growth factors such as EGF (Tomlinson et al., 1994), platelet-derived growth factor (Hirst et al., 1992) and thrombin (Panettieri et al., 1995), and contractile agonists such as histamine (Panettieri et al., 1990) and endothelin-1 (Glassberg et al., 1994) have been demonstrated to induce airway smooth muscle cell proliferation. Leukotriene D4 has also been shown to augment the influence of the growth factors insulin-like growth factor (Cohen et al., 1995) and EGF (Panettieri et al., 1998b) to potentiate airway smooth muscle cell proliferation. A combination of the cytokines IL-1β and IL-6 induces both guinea-pig tracheal smooth muscle cell hyperplasia and hypertrophy in vitro (De et al., 1995). A study with human airway smooth muscle cells has implicated a role for angiotensin II in cellular hypertrophy but not hyperplasia (McKay et al., 1998). In our model, treatment with the 5-LO inhibitor and either BLT or CysLT1 receptor antagonists significantly inhibited increases in airway smooth muscle thickening, suggesting that both LTB4 and cysteinyl leukotrienes may influence this process. In a previous in vivo study in rats, the CysLT1 receptor antagonist MK-571 also significantly inhibited allergen-induced increases in airway smooth muscle mass (Wang et al., 1993). The BLT receptor antagonist had a significant inhibitory effect on allergen-induced increases in airway smooth muscle thickness but not on increased DNA synthesis, suggesting that factors other than cell proliferation may contribute to airway smooth muscle thickening. The possibility remains therefore that ASM cell proliferation and ASM thickening may be differentially regulated through the release of different types of mediators.
One confounding factor in this study is that, following repeated allergen exposure, rats exhibited a significant increase in ASM cell DNA synthesis but not a significant increase in the total number of ASM cells, although there was an overall increase compared to the saline-exposed control group. The reason for this may be related to the temporal delay between detection of DNA synthesis by BrdU incorporation and the actual onset of cell proliferation. Alternatively, it is possible that ASM cell proliferation may be accompanied by a similar rate of ASM cell apoptosis, such that no overall increase in cell number is observed.
In summary, we demonstrate that cysteinyl leukotrienes mediate increased airway smooth muscle cell DNA synthesis induced by repeated allergen exposure in Brown Norway rats. In addition, the appearance of airway smooth muscle thickening is also dependent on products of the 5-LO pathway, including both LTB4 and cysteinyl leukotrienes. Both LTB4 and cysteinyl leukotrienes are also involved in allergen-induced airway eosinophilia, but are not directly responsible for bronchial hyperresponsiveness. These results support the concept that leukotrienes participate in increased ASM cell proliferation and ASM thickening following repeated allergen exposure, processes underlying airway wall remodelling in chronic asthma.
Acknowledgments
The authors would like to thank SmithKline Beecham for supporting this study and for providing the compounds SB 210661, SB 201146 and pranlukast.
Abbreviations
- ASM
airway smooth muscle
- BLT
leukotriene B4 receptor
- BrdU
5-bromo-2′-deoxyuridine
- CysLT
cysteinyl leukotrienes
- CysLT1
cysteinyl leukotriene receptor 1
- CysLT2
cysteinyl leukotriene receptor 2
- DAPI
4,6-diamidino-2-phenylindole
- PC200
concentration of acetylcholine required to increase baseline airways resistance 200%
References
- BARNES N.C., PIPER P.J., COSTELLO J.F. Comparative effects of inhaled leukotriene C4, leukotriene D4, and histamine in normal human subjects. Thorax. 1984;39:500–504. doi: 10.1136/thx.39.7.500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BISGAARD H., PEDERSEN M. SRS-A leukotrienes decrease the activity of human respiratory cilia. Clin. Allergy. 1987;17:95–103. doi: 10.1111/j.1365-2222.1987.tb02326.x. [DOI] [PubMed] [Google Scholar]
- COHEN P., NOVERAL J.P., BHALA A., NUNN S.E., HERRICK D.J., GRUNSTEIN M.M. Leukotriene D4 facilitates airway smooth muscle cell proliferation via modulation of the IGF axis. Am. J. Physiol. 1995;269:L151–L157. doi: 10.1152/ajplung.1995.269.2.L151. [DOI] [PubMed] [Google Scholar]
- DAHLÉN S.-E., HEDQVIST P., HAMMARSTRÖM S., SAMUELSSON B. Leukotrienes are potent constrictors of human bronchi. Nature. 1980;288:484–486. doi: 10.1038/288484a0. [DOI] [PubMed] [Google Scholar]
- DAINES R.A., CHAMBERS P.A., FOLEY J.J., GRISWOLD D.E., KINGSBURY W.D., MARTIN L.D., SCHMIDT D.B., SHAM K.K., SARAU H.M. (E)-3-[6-[[2,6-dichlorophenyl)thio]methyl]-3-(2-phenylethoxy)pyridinyl]-2-propenoic acid: a high-affinity leukotriene B4 receptor antagonist with oral antiinflammatory activity. J. Med. Chem. 1996;39:3837–3841. doi: 10.1021/jm960248s. [DOI] [PubMed] [Google Scholar]
- DAINES R.A., CHAMBERS P.A., PENDRAK I., JAKAS D.R., SARAU H.M., FOLEY J.J., SCHMIDT D.B., KINGSBURY W.D. Trisubstituted pyridine leukotriene B4 receptor antagonists: synthesis and structure-activity relationships. J. Med. Chem. 1993;36:3321–3332. doi: 10.1021/jm00074a013. [DOI] [PubMed] [Google Scholar]
- DE S., ZELAZNY E.T., SOUHRADA J.F., SOUHRADA M. IL1β and IL-6 induce hyperplasia and hypertrophy of cultured guinea-pig airway smooth muscle cells. J. Appl. Physiol. 1995;78:1555–1563. doi: 10.1152/jappl.1995.78.4.1555. [DOI] [PubMed] [Google Scholar]
- DIAMANT Z., HILTERMANN J.T., VAN RENSEN E.L., CALLENBACH P.M., VESELIC-CHARVAT M., VAN DER VEEN H., SONT J.K., STERK P.J. The effect of inhaled leukotriene D4 and methacholine on sputum cell differentials in asthma. Am. J. Respir. Crit. Care Med. 1997;155:1247–1253. doi: 10.1164/ajrccm.155.4.9105062. [DOI] [PubMed] [Google Scholar]
- DUNNILL M.S., MASSARELLA G.R., ANDERSON J.A. A comparison of the quantitative anatomy of the bronchi in normal subjects, in status asthmaticus, in chronic bronchitis, and in emphysema. Thorax. 1969;24:176–179. doi: 10.1136/thx.24.2.176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- EBINA M., TAKAHASHI T., CHIBA T., MOTOMIYA M. Cellular hypertrophy and hyperplasia of airway smooth muscles underlying bronchial asthma: a 3-D morphometric study. Am. Rev. Respir. Dis. 1993;148:720–726. doi: 10.1164/ajrccm/148.3.720. [DOI] [PubMed] [Google Scholar]
- ELWOOD W, , LTVALL J.O., BARNES P.J., CHUNG K.F. Characterization of allergen-induced bronchial hyperresponsiveness and airway inflammation in actively sensitized Brown-Norway rats. J. Allergy Clin. Immunol. 1991;88:951–960. doi: 10.1016/0091-6749(91)90253-k. [DOI] [PubMed] [Google Scholar]
- FUJIWARA H., KURIHARA N., OHTA, HIRATA K., MATSUSHITA H., KANAZAWA H., TAKEDA T. Effect of a new leukotriene receptor antagonist, ONO-1078, on human bronchial smooth muscle in vitro. Prostaglandins Leukot. Essent. Fatty Acids. 1993;48:241–246. doi: 10.1016/0952-3278(93)90092-b. [DOI] [PubMed] [Google Scholar]
- GLASSBERG M.K., ERGUL A., WANNER A., PUETT D. Endothelin-1 promotes mitogenesis in airway smooth muscle cells. Am. J. Respir. Cell Mol. Biol. 1994;10:316–321. doi: 10.1165/ajrcmb.10.3.7509612. [DOI] [PubMed] [Google Scholar]
- HEARD B.E., HOSSAIN S. Hyperplasia of bronchial muscle in asthma. J. Pathol. 1973;110:319–331. doi: 10.1002/path.1711010212. [DOI] [PubMed] [Google Scholar]
- HIRST S.J., BARNES P.J., TWORT C.H.C. Quantifying proliferation of cultured human and rabbit airway smooth muscle in response to serum and platelet-derived growth factor. Am. J. Respir. Cell Mol. Biol. 1992;7:574–581. doi: 10.1165/ajrcmb/7.6.574. [DOI] [PubMed] [Google Scholar]
- HUA X.Y., DAHLÉN S.-E., LUNDBERG J.M., HAMMERSTRÖM S., HEDQVIST P. Leukotrienes C4, D4 and E4 cause widespread and extensive plasma extravasation in the guinea-pig. Naunyn Schmiederbergs Arch. Pharmacol. 1985;330:136–141. doi: 10.1007/BF00499906. [DOI] [PubMed] [Google Scholar]
- IRVINC C.G., TU Y-P., SHELLER J.R., FUNK C.D. 5-Lipoxygenase products are necessary for ovalbumin-induced airway responsiveness in mice. Am. J. Physiol. 1997;272:L1053–L1058. doi: 10.1152/ajplung.1997.272.6.L1053. [DOI] [PubMed] [Google Scholar]
- JOHNSON H.G., McNEE M.L. Secretagogue responses of leukotriene C4, D4: comparison of potency in canine trachea in vivo. Prostaglandins. 1983;25:237–243. doi: 10.1016/0090-6980(83)90107-7. [DOI] [PubMed] [Google Scholar]
- LAITINEN L.A., HEINO M., LAITINEN A., KAVA T., HAAHTELA T. Damage of the airway epithelium and bronchial reactivity in patients with asthma. Am. Rev. Respir. Dis. 1985;131:599–606. doi: 10.1164/arrd.1985.131.4.599. [DOI] [PubMed] [Google Scholar]
- LAITINEN L.A., LAITINEN A., HAAHTELA T., VILKKA V., SPUR B.W., LEE T.H. Leukotriene E4 and granulocytic infiltration in the asthmatic airways. Lancet. 1993;341:989–990. doi: 10.1016/0140-6736(93)91073-u. [DOI] [PubMed] [Google Scholar]
- LAM S., CHAN H., LERICHE J.C., CHAN YEUNG M., SALARI H. Release of leukotrienes in patients with bronchial asthma. J. Allergy Clin. Immunol. 1988;81:711–717. doi: 10.1016/0091-6749(88)91043-3. [DOI] [PubMed] [Google Scholar]
- MAROM Z., SHELHAMER J.H., BACH M.K., MORTON D.R., KALINER M. Slow-reacting substances, leukotrienes C4 and D4, increase the release of mucus from human airways in vitro. Am. Rev. Respir. Dis. 1982;126:449–451. doi: 10.1164/arrd.1982.126.3.449. [DOI] [PubMed] [Google Scholar]
- MARTIN J.G., XU L.-J., TOH M-Y., OLIVENSTEIN R., POWELL W.S. Leukotrienes in bile during the early and the late airway responses after allergen challenge of sensitized rats. Am. Rev. Respir. Dis. 1993;147:104–110. doi: 10.1164/ajrccm/147.1.104. [DOI] [PubMed] [Google Scholar]
- MCKAY S., DE JONGSTE J.C., SAXENA P.R., SHARMA H.S. Angiotensin II induces hypertrophy of human airway smooth muscle cells: expression of transcription factors and transforming growth factor-β1. Am. J. Respir. Cell Mol. Biol. 1998;18:823–833. doi: 10.1165/ajrcmb.18.6.2924. [DOI] [PubMed] [Google Scholar]
- MCLOUGHLIN M., KEARNEY A.S., PALEPU N.R. Aqueous stability of SB210661: kinetics and primary degradation mechanisms of an N-hydroxyurea-containing 5-lipoxygenase inhibitor. J. Pharm. Pharmacol. 1998;50:127–132. doi: 10.1111/j.2042-7158.1998.tb06166.x. [DOI] [PubMed] [Google Scholar]
- OBATA T., OKADA Y., MOTOISHI M., NAKAGAWA N., TERAWAKI T., AISHITA H. In vitro antagonism of ONO-1078, a newly, developed anti-asthma agent, against peptide leukotrienes in isolated guinea-pig tissues. Jpn. J. Pharmacol. 1992;60:227–237. doi: 10.1254/jjp.60.227. [DOI] [PubMed] [Google Scholar]
- PANETTIERI R.A., JR, HALL I.P., MAKI C.S., MURRAY R.K. α-Thrombin increases cytosolic calcium and induces human airway smooth muscle cell proliferation. Am. J. Respir. Cell Mol. Biol. 1995;13:205–216. doi: 10.1165/ajrcmb.13.2.7626288. [DOI] [PubMed] [Google Scholar]
- PANETTIERI R.A., JR, MURRAY R.K., ESZTERHAS A.J., BILGEN G., MARTIN J.G. Repeated allergen inhalations induce DNA synthesis in airway smooth muscle and epithelial cells in vivo. Am. J. Physiol. 1998a;274:L417–L424. doi: 10.1152/ajplung.1998.274.3.L417. [DOI] [PubMed] [Google Scholar]
- PANETTIERI R.A., JR, TAN E.M.L., CIOCCA V., LUTTMANN M.A., LEONARD T.B., HAY D.W.P. Effects of LTD4 on human airway smooth muscle cell proliferation, matrix expression, and contraction in vitro: differential sensitivity to cysteinyl leukotriene receptor antagonists. Am. J. Respir. Cell Mol. Biol. 1998b;19:453–461. doi: 10.1165/ajrcmb.19.3.2999. [DOI] [PubMed] [Google Scholar]
- PANETTIERI R.A., JR, YADVISH P.A., KELLY A.M., RUBINSTEIN N.A., KOTLIKOFF M.I. Histamine stimulates proliferation of airway smooth muscle and induces c-fos expression. Am. J. Physiol. 1990;259:L365–L371. doi: 10.1152/ajplung.1990.259.6.L365. [DOI] [PubMed] [Google Scholar]
- PAUWELS R., BAZIN H., PLATTEAU B., VAN DER STRAETEN M. The influence of antigen dose on IgE production in different rat strains. Immunology. 1979;36:151–157. [PMC free article] [PubMed] [Google Scholar]
- RICHARDS I.M., GRIFFIN R.L., OOSTVEEN J.A., MORRIS J., WISHKA D.G., DUNN C.J. Effect of the selective leukotriene B4 antagonist U75302 on antigen-induced bronchopulmonary eosinophilia in sensitized guinea-pigs. Am. Rev. Respir. Dis. 1989;140:1712–1716. doi: 10.1164/ajrccm/140.6.1712. [DOI] [PubMed] [Google Scholar]
- ROCHE W.R., BEASLEY R., WILLIAMS J.H., HOLGATE S.T. Subepithelial fibrosis in the bronchi of asthmatics. Lancet. 1989;i:520–524. doi: 10.1016/s0140-6736(89)90067-6. [DOI] [PubMed] [Google Scholar]
- SALMON M., WALSH D.A., KOTO H., BARNES P.J., CHUNG K.F.Chronic inhalational ovalbumin challenge induces proliferation in airway smooth muscle and epithelium of sensitised Brown-Norway rats Inflamm. Res. 199746S199[Abstract] [Google Scholar]
- SAPIENZA S., DU T., EIDELMAN D.H., WANG N.S., MARTIN J.G. Structural changes in the airways of sensitized Brown Norway rats after antigen challenge. Am. Rev. Respir. Dis. 1991;144:423–427. doi: 10.1164/ajrccm/144.2.423. [DOI] [PubMed] [Google Scholar]
- SHU S.Y., JU G., FAN L.Z. The glucose oxidase-DAB-nickel method in peroxidase histochemistry of the central nervous system. Neurosci. Lett. 1988;85:169–171. doi: 10.1016/0304-3940(88)90346-1. [DOI] [PubMed] [Google Scholar]
- SILBAUGH S.A., STENGEL P.W., WILLIAMS G.D., HERRON D.K., GALLAGHER P., BAKER S.R. Effects of leukotriene B4 inhalation: airway sensitization and lung granulocyte infiltration in the guinea-pig. Am. Rev. Respir. Dis. 1987;136:930–934. doi: 10.1164/ajrccm/136.4.930. [DOI] [PubMed] [Google Scholar]
- SPADA C.S., NIEVES A.L., KRAUSS A.H.-P., WOODWARD D.F. Comparison of leukotriene B4 and D4 effects on human eosinophil and neutrophil motility in vitro. J. Leukoc. Biol. 1994;55:183–191. doi: 10.1002/jlb.55.2.183. [DOI] [PubMed] [Google Scholar]
- TAKI F., SUZUKI R., TORII K., MATSUMOTO S., TANIGUCHI H., TAKAGI K. Reduction of the severity of bronchial hyperresponsiveness by the novel leukotriene antagonist 4-oxo-8-[4-(4-phenyl-butoxy)benzoylamino]-2-(tetrazol-5-yl)-4H-1-benzopyran hemihydrate. Arzneimittelforschung. 1994;44:330–333. [PubMed] [Google Scholar]
- TOMLINSON P.R., WILSON J.W., STEWART A.G. Inhibition by salbutamol of the proliferation of human airway smooth muscle cells grown in culture. Br. J. Pharmacol. 1994;111:641–647. doi: 10.1111/j.1476-5381.1994.tb14784.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- TUDHOPE S.R., CUTHBERT N.J., ABRAM T.S., JENNINGS M.A., MAXEY R.J., THOMPSON A.M., NORMAN P., GARDINER P.J. BAY u9773, a novel antagonist of cysteinyl leukotrienes with activity against two receptor subtypes. Eur. J. Pharmacol. 1994;264:317–323. doi: 10.1016/0014-2999(94)00485-4. [DOI] [PubMed] [Google Scholar]
- UNDERWOOD D.C., OSBORN R.R., NEWSHOLME S.J., TORPHY T.J., HAY D.W.P. Persistent airway eosinophilia after leukotriene (LT) D4 administration in the guinea-pig: modulation by the LTD4 receptor antagonist, pranlukast, or an interleukin-5 monoclonal antibody. Am. J. Respir. Crit. Care Med. 1996;154:850–857. doi: 10.1164/ajrccm.154.4.8887574. [DOI] [PubMed] [Google Scholar]
- VON NEERGARD K., WIRZ K. Die messung der Strömungswiderstande in den Atemwegen des Menschen, insbesondere bei Asthma und Emphysem. Z. Klin. Med. 1927;105:51–58. [Google Scholar]
- WANG C.G., DU T., XU L.-J., MARTIN J.G. Role of leukotriene D4 in allergen-induced increases in airway smooth muscle in the rat. Am. Rev. Respir. Dis. 1993;148:413–417. doi: 10.1164/ajrccm/148.2.413. [DOI] [PubMed] [Google Scholar]
- WARDLAW A.J., HAY H., CROMWELL O., COLLINS J.V., KAY A.B. Leukotrienes, LTC4 and LTB4, in bronchoalveolar lavage in bronchial asthma and other respiratory diseases. J. Allergy Clin. Immunol. 1989;84:19–26. doi: 10.1016/0091-6749(89)90173-5. [DOI] [PubMed] [Google Scholar]
- WOODWARD D.F., WASSERMAN M.A., WEICHMANN B.M. Investigation of leukotriene involvement in the vasopermeability response associated with guinea-pig tracheal anaphylaxis: comparison with cutaneous anaphylaxis. Eur. J. Pharmacol. 1983;93:9–19. doi: 10.1016/0014-2999(83)90025-0. [DOI] [PubMed] [Google Scholar]


