Abstract
Smooth muscle is a major component of human tissues and is essential for the normal function of a multitude of organs including the intestine, urinary tract and the vascular system. The use of stem cells for cell-based tissue engineering and regeneration strategies represents a promising alternative for smooth muscle repair. For such strategies to succeed, a reliable source of smooth muscle precursor cells must be identified. Adipose tissue provides an abundant source of multipotent cells. In this study, the capacity of processed lipoaspirate (PLA) and adipose-derived stem cells to differentiate into phenotypic and functional smooth muscle cells was evaluated. To induce differentiation, PLA cells were cultured in smooth muscle differentiation medium. Smooth muscle differentiation of PLA cells induced genetic expression of all smooth muscle markers and further confirmed by increased protein expression of smooth muscle cell-specific α actin (ASMA), calponin, caldesmon, SM22, myosin heavy chain (MHC), and smoothelin. Clonal studies of adipose derived multipotent cells demonstrated differentiation of these cells into smooth muscle cells in addition to trilineage differentiation capacity. Importantly, smooth muscle-differentiated cells, but not their precursors, exhibit the functional ability to contract and relax in direct response to pharmacologic agents. In conclusion, adipose-derived cells have the potential to differentiate into functional smooth muscle cells and, thus, adipose tissue can be a useful source of cells for treatment of injured tissues where smooth muscle plays an important role.
Smooth muscle, an active component of cardiovascular, gastrointestinal, reproductive, and urinary systems, has been the subject of intense research in the field of cellular therapeutics involving these tissues. However, one major limitation to this approach has been finding a reliable source of smooth muscle cells (SMC) because smooth muscle biopsies of these systems can be impractical and morbid. In addition, biopsies usually lead to limited amounts of cells that need to be extensively expanded before they can be used for therapeutic applications. Furthermore, it has been shown that SMC derived from diseased organs can lead to abnormal cells that are different from healthy SMC, thus limiting their use (1, 2). Therefore, there is a great need for alternative sources of healthy SMC.
Several groups have suggested the use of bone marrow-derived cells (3–5) and embryonic stem cells to repair smooth muscle tissues (6, 7) because of their stem cell-like properties. Adipose tissue represents a potential alternative reservoir of cells with stem cell properties such as self-renewal and pluripotency. A large population of cells isolated from the stromal vascular fraction of human adipose tissue termed processed lipoaspirate (PLA) cells possesses multilineage potential (8, 9). Recent reports on PLA cells have demonstrated improvement of postnatal neovascularization (10), muscle regeneration (11), high potential to heal critical-size calvarial defects (osteogenic capacity) (12), and rescue of lethally irradiated mice (stromal capacity) (13). Adipose-derived cells also appear to possess cardiomyogenic capacity in vitro; however, the significance of this capacity remains to be proven (14). Adipose stem cells (ASC), a pluripotent clonal population derived from PLA cells, have shown higher differentiation capacity when compared to PLA cells (8). The multilineage capacity of PLA cells and ASC makes them promising candidates for reconstruction of human smooth muscle related tissues and organs. Moreover, adipose tissue can be harvested in large quantity with minimal morbidity.
This study demonstrates the ability of PLA cells to be differentiated both phenotypically and functionally into SMC. Upon induction, PLA cells assumed typical smooth muscle morphology and expressed characteristic leiomyogenic markers both at the genetic and protein levels. In addition, differentiated PLA cells exhibited a contractile function that is similar to that of smooth muscle in vivo. ASCs, a clonal enriched stem cell population, maintained their multilineage potential and differentiated into functional SMC. Furthermore, the clonally enriched adipose stem cell maintain their multilineage potential and can be differentiated into functional SMC. Differentiation rather than cell selection appears to be responsible for the observed phenomena. This study describes a source of cells in adipose tissue for smooth muscle applications, extends the differentiation repertoire of ASC, provides a model for the study of smooth muscle differentiation, and describes an in vitro system to test the functional capacity of PLA-differentiated SMC to contract and relax in response to common pharmacologic agents.
Results
Smooth Muscle Differentiation of PLA Cells.
Optimal smooth muscle differentiation was observed when cells were cultured in smooth muscle inductive medium (SMIM; Medium MCDB 131 supplemented with 1% FBS plus 100 units/ml heparin). After 6 weeks, PLA cells (P0, P1, and P2) cultured in uncoated dishes in control media (CM) grew to confluence and formed a monolayer of fibroblast-like PLA cells (Fig. 1A) presenting a flat morphology and stress-fiber pattern. In the presence of SMIM in uncoated substratum, the cells acquired typical SMC morphology with spindle-like shape with a “hill and valley morphology” and proliferated at a lower rate compared to those cultured in CM (Fig. 1B). Neither spontaneous adipogenic differentiation nor apparent mineralization was observed in the culture conditions studied. Laminin alone, in the absence of SMIM, did not change the cells to SMC morphology but instead cells retracted and formed nodule like structures, leaving areas sparsely populated (data not shown).
Fig. 1.
PLA cells acquire SMC morphology. Contrast micrographs (×100) are shown. (A) Typical morphology of PLA cells cultured in CM for 4 weeks, which grow in a monolayer. (B) In the presence of SMIM, PLA cells show the typical “hills and valleys” growth pattern of SMC.
Gene Expression at Transcriptional Level.
For gene expression studies, six well-known markers of leiomyogenic differentiation [SMC-specific α actin (ASMA), smoothelin, calponin, caldesmon, MHC, and SM22] were studied. The expression of these markers was confirmed by RT-PCR (Fig. 2 A and B). Like other pluripotent stem cells, some early markers of smooth muscle differentiation such as ASMA, caldesmon, and SM22 were found to be spontaneously transcribed in undifferentiated PLA cells. Upon induction, there were increases in the expression of these markers confirmed by real-time PCR, as well as new expression of MHC and caldesmon. At a population level, the most dramatic effect was observed in the expression of calponin. Induction resulted in 9-fold increase in calponin expression when compared to control. There also were increases in the expression of ASMA (2.5-fold) and SM22 (4-fold) (Fig. 2C). No additional effects on smooth muscle differentiation were seen in cells grown on laminin or collagen-coated dishes (data not shown).
Fig. 2.
Induction of SMC markers when PLA cells are grown in smooth muscle differentiation conditions. (A) Induction of gene expression of smooth muscle markers when PLA are exposed to smooth muscle differentiation conditions for 3 and 6 weeks as determined by RT-PCR. (B) Induction of gene expression of all smooth muscle markers as determined by semiquantitative RT-PCR using GAPDH as a control gene. (C) Increases in the expression of ASMA, calponin, and SM22 were confirmed with real-time PCR.
Gene Expression at Translational Level.
Immunohistochemistry and immunofluorescence revealed an increase in the expression of ASMA, calponin, and caldesmon when cells were cultured in SMIM (Fig. 3 A and B). Paralleling the transcriptional studies, late markers of differentiation such as MHC were present only when cells were exposed to SMIM. Western blot confirmed the immunohistochemistry data with increases in the expression of ASMA and expression of MHC only in cells grown in SMIM (Fig. 3C). Although a few scattered cells in CM expressed ASMA, cells exposed to SMIM formed colonies of smooth muscle marker-positive cells. PLA cells cultured in any of the conditions mentioned above were homogenously positive for prolyl-hydrohylase, vWfactor, and CD105, but negative for CD31.
Fig. 3.
Induction of smooth muscle marker protein expression when PLA cells are cultured in SMIM. (A) There is an increase in the expression of ASMA, calponin, caldesmon, and MHC in PLA cells cultured in SMIM compared to cells grown in CM, as determined by immunohistochemistry (×100). (B) Increase in expression of ASMA and induction of MHC confirmed by immunofluorescence (×200). (C) Increase in ASMA and induction of MHC expression confirmed by Western blot analysis.
Smooth Muscle Differentiation of ASC.
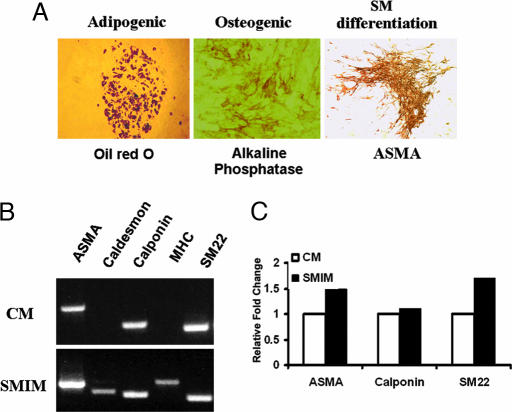
To ascertain that the induction of smooth muscle markers was due to differentiation and not SMC selection in the aspirates, we evaluated the differentiation capacity of ASCs. PLA cells from passage P0 were cloned. Ten clones were evaluated for smooth muscle differentiation capacity as well as pluripotency (osteogenic and adipogenic differentiation, Fig. 4A). Like PLA cells, clones were found to express early markers of smooth muscle differentiation at the genetic level such as ASMA and caldesmon. Consistent with the PLA cell data, clonal analysis showed that expression of MHC and caldesmon was induced in previously nonexpressing cells upon differentiation in SMIM (Fig. 4B). Real-time PCR showed increased ASMA, calponin, and SM22 gene expression with exposure to SMIM (1.5-, 1.1-, and 1.7-fold respectively, Fig. 4C). All clones maintained their potential for osteogenic and adipogenic differentiation, reinforcing the idea that multilineage precursors are present within adipose tissue.
Fig. 4.
Clonal adipose-derived cells maintain multipotent capacity. (A) When exposed to adipogenic, osteogenic, or leiomyogenic differentiation conditions, ASC clones maintain adipose (Oil red O), osteogenic (alkaline phosphatase), and leiomyogenic (ASMA) phenotype (×100). (B) Representative clone showing new expression of caldesmon and MHC when grown in SMIM as determined by RT-PCR. (C) Representative clone showing increased ASMA, calponin, and SM22 expression when grown in SMIM compared to clone grown in CM as determined by real-time PCR.
Cell Contraction.
Regardless of the tissue of origin, the primary function of SMC is to contract and relax. To examine whether the smooth muscle-differentiated PLA were functional, these cells were subjected to the effects of carbachol and atropine. Qualitative and quantitative assessments were used to measure contractile effects.
Qualitative Carbachol Contraction of Cell Cultures.
Differentiated cells were photographed before and after exposure to 10−5 M carbachol. One min after exposure to carbachol, the cells began to contract, remaining in this state for up to 10 min (Movie 1, which is published as supporting information on the PNAS web site). Furthermore, the muscarinic antagonist atropine was shown to block the carbachol-mediated effects. Similar results were observed in primary rat bladder SMC (rBSMC).
Contraction of Collagen Gels.
After mechanically releasing the collagen gels, only the gels seeded with smooth muscle-differentiated PLA cells and rBSMC showed baseline contractions, whereas unseeded gels and gels with human T lymphocytes remained static (Fig. 5A). Human suppressor T lymphocytes showed no contraction to carbachol. On the other hand, smooth muscle-differentiated PLA cells showed a strong contraction to carbachol similar to the response of rBSMC (Fig. 5A). A dose–response relationship to carbachol was demonstrated by evaluating gel contractility at different agonist concentrations (Fig. 5B). When the cholinergic response was first inhibited by atropine, the subsequent response to carbachol was greatly reduced (Fig. 5C). Undifferentiated PLA cell lacked responses to carbachol.
Fig. 5.

Smooth muscle-differentiated PLA cells have a response to carbachol that is similar to that seen for SMC. (A) Collagen gels embedded with smooth muscle-differentiated PLA cells contract to carbachol similar to rBSMC. Collagen gels are released at time 0. Only gels with rBSMC and smooth muscle-differentiated PLA cells have contraction after release. Gels are allowed to stabilize for 45 min, at which point 10−5 M carbachol is added. There is further contraction in rBSMC and smooth muscle-differentiated PLA cells but no response in blank gels or gels embedded with T cells. (B) Smooth muscle-differentiated PLA cells respond to carbachol in a dose-dependent manner. Gels are released at time 0 and allowed to stabilize, and varying doses of carbachol (none, 10−11 M, 10−8 M, and 10−5 M) are added at 45 min. (C) Atropine blocks carbachol response. Collagen gels embedded with smooth muscle-differentiated PLA cells were released at time 0 and allowed to stabilize. 10−4 M atropine was added at 45 min, and 10−5 M carbachol was added at 90 min (open diamonds). The response to carbachol was attenuated compared to a similar experiment where the gel was exposed only to carbachol 10−5 M at 45 min (filled diamonds).
Discussion
Cell therapies involving SMC differentiation may provide an alternative treatment for diseases involving SMC pathology such as cardiovascular disease, gastrointestinal diseases, urinary incontinence, and bladder dysfunction, among others. A major obstacle for such an approach has been finding a reliable source of healthy SMC that can be safely harvested and that requires minimal manipulation.
There is significant heterogeneity in the origin of SMC in both development and adult life. During development, most smooth muscle progenitors arise mainly from the splanchnic mesoderm and from the neural crest (15). At postnatal life, progenitors for SMC have been found in circulating blood (16) and bone marrow (17). Bone marrow has been the hallmark site in postnatal life known to harbor at least two populations of stem cells: the hematopoietic and the nonhematopoietic mesenchymal stem cell (MSC). In vitro, marrow stromal cells have been shown to differentiate into bone, fat, cartilage, neuron, or muscle when treated with specialized induction media. Recent studies have indicated that smooth muscle-like cells also can be derived from bone marrow cells both in vitro (17, 18) or in vivo (3, 5). However, bone marrow is not the only site of adult stem cells capable of multiple differentiation. ADCs have the capacity to differentiate along adipogenic, osteogenic, chondrogenic, myogenic, neurogenic, and cardiomyogenic pathways (9, 14, 19, 20). Unlike bone marrow, adipose tissue can be obtained in relatively large quantities with minimal morbidity and might represent an ideal source of autologous progenitor cells for engineering tissues. These cells have been shown to be phenotypically similar to MSCs but lack expression of CD106, among other markers, suggesting that they are a truly distinct population of cells from mesenchymal origin (9). They have been shown to be stable in long-term culture, maintaining a consistent population doubling rate, and exhibiting low levels of senescence (8, 9). This stability, combined with their ability to be differentiated into multiple pathways, has led investigators to believe that ASCs are a stem cell population derived from adipose tissue.
The present study demonstrates that a population of adherent cells obtained from the stromal vascular fraction of human adipose tissue can differentiate into SMC. Although, morphologically and physiologically, SMC form a very heterogenous population, the majority have some common features such as a spindle shape, low proliferative rate, reduced synthesis of extracellular matrix, and ligand-induced contractility (21, 22). It is the sequential expression of cytoskeletal and contractile proteins that generates the diversity of phenotypes ranging from immature cells to mature SMC expressing the complete repertoire of proteins involved in smooth muscle contraction.
To establish that a particular cell is a mature SMC, certain unique markers need to be present. These smooth muscle markers include ASMA, SM22, calponin, caldesmon, smoothelin, and smooth muscle MHC. Although ASMA is an early marker of developing smooth muscle, its expression alone does not provide definitive evidence for a smooth muscle lineage (15). Thus, to characterize leiomyogenic differentiation, we chose to evaluate not only ASMA expression, but also the expression of all other smooth muscle markers. These other markers are highly restricted to differentiated smooth muscle and, in particular, MHC and smoothelin are not detected in any other cell type and are only expressed in contractile SMC (15, 23). At the genetic level, we found that PLA cells, like other pluripotent stem cell populations, express some smooth muscle markers at baseline. For this reason, real-time PCR analysis was performed to assess the capacity of our media conditions to induce increases in gene expression. In the presence of SMIM, PLA cells acquired typical SMC morphology and increases in baseline expression of early and mid markers of smooth muscle differentiation such as ASMA, caldesmon, calponin, and SM22 at the genetic level. In addition, late markers of SMC differentiation which are only present in contractile mature SMC (such as smoothelin and smooth muscle MHC) were present only in cells cultured with SMIM. These findings were confirmed at the protein level. PLA cells cultured in any of these conditions, showed to be homogenously positive for vWfactor, CD 105, and polyl-hydroxylase but negative for CD31, a phenotype common to other cultured mesenchymal stem cells.
Mature SMCs can undergo reversible changes of phenotype (phenotype modulation or switching) in response to changes in the local environment (15, 24). Previous studies of smooth muscle differentiation and phenotype modulation indicate that SMC express the contractile differentiated phenotype when the cells are not in a proliferated state (15). In an attempt to induce PLA smooth muscle differentiation, we controlled cell proliferation. Heparin has been shown to prevent proliferation, as well as to induce ASMA expression (25, 26). Phenotypic modulation of SMC by extracellular matrix has also been documented (1, 2, 27–29). Based on these observations, the effects of media and substrate on the induction of a differentiated smooth muscle phenotype were evaluated. PLA cells were cultured with low levels of FBS and varying concentration of heparin to control their differentiation and proliferation. Optimal differentiation occurred when PLA cells cultured in medium MCDB131 supplemented with low concentration of FBS plus 100 units/ml heparin. In addition, the effects of collagen and laminin on differentiation were studied. Although exposure to laminin had an effect on cell morphology, overall the media rather than the substrate seemed to be the predominant differentiating factor (data not shown).
SMC are highly specialized cells whose principal function is contraction and relaxation. The ability of SMC to contract in response to a physiological stimulus has been considered to be the main proof that myocytes are in a differentiated state (30, 31). In this study, we developed a system to evaluate the ability of cells to respond to a variety pharmacologic agents by tracking the movement of glass beads dispersed in a collagen gel embedded with cells. The differentiated cells showed dose-dependent contraction in response to a potent muscarinic contractile agonist, carbachol. In addition, the contractile response was inhibited by atropine. Thus, the data suggest that a population of multipotent cells derived from adipose tissue can be differentiated into phenotypical and functional SMC.
To confirm that differentiation occurred and to rule out the possibility that media conditions had selected for a subpopulation of smooth muscle progenitor cells, similar experiments were conducted on clonal population of cells obtained from PLA cells. Smooth muscle marker expression was induced on all clones studied. In addition, the multipotential capacity of these clones was evaluated by demonstrating adipogenic and osteogenic differentiation. Interestingly, there was decreased smooth muscle marker expression at the genetic level on the ASC when compared to their PLA counterparts in culture under smooth muscle differentiation conditions. The extensive proliferation to which clonal cells are submitted during the cloning process might render them senescent and account for this discrepancy. An alternative possibility is that isolating one individual cell upon cloning may eliminate necessary paracrine signals that may occur between cells in the heterogeneous PLA population. These signals may act to facilitate differentiation. Some of the soluble factors known to modulate SMC differentiation are TGF-β1, platelet-derived growth factor, retinoic acid, interleukin 1, and ascorbic acid (32–34). Some of these factors are known to be produced by endothelial cells, one of the cell types identified in PLA cultures. It has also been reported that the interaction of cell surface receptors and ligands, the formation of gap junction structures, as well as the involvement of short-range-acting molecules between differentiated and stem cells contribute to stem cell differentiation (35). It is thus possible that cell–cell interaction among different cell types in the PLA may be important in smooth muscle differentiation of the progenitor cells.
In the current study, the expression of leiomyogenic genes was not repressed during adipogenic differentiation (results not shown). The persistent expression of genes restricted to SMCs in PLA and ASC lead us to speculate that PLA cells might represent a hierarchical organization similar to that proposed to be operating in the bone marrow (35, 36). Thus, the PLA population could be seen as a peripheral stromal cell compartment with cells displaying distinct differentiation capacities elicited by culture conditions or in vivo needs.
Regardless of the mechanism of differentiation, adipose tissue appears to provide a population of cells with multilineage potential capable of inducing the expression of a smooth muscle phenotype. In addition, these differentiated cells can contract and respond to common pharmacologic agents as native SMC. Therefore, adipose tissue can be an abundant source of functional SMC for tissue engineering or therapeutic modalities for diseases that affect smooth muscle.
Materials and Methods
Isolation and Culture of Multipotent Cells.
All experiments were approved by the Office of Protection of Research Subjects Internal Review Board at University of California, Los Angeles. Human PLA cells were isolated as described (8, 9). Cells were plated in Dulbecco's Modified Eagle's Media (DMEM; Mediatech, Herndon, VA) supplemented with 10% FBS (FBS; HyClone, Logan, UT) and 1% from a 100× concentrated antibiotic–antimycotic solution (penicillin G/amphotericin B/streptomycin; Mediatech) at a density of 5 × 106 nucleated cells per 100-mm tissue culture dish. This medium is referred as CM. Cells cultured as described are referred to as P0 cells. To expand the cells, adherent cells were allowed to grow to near confluency, washed with PBS, and harvested by using 0.25% trypsin/1 mM EDTA. The cells were replated 1:4 and allowed to grow.
Isolation of rBSMC.
Differentiated PLA cells were compared against primary rBSMC. Harvested bladder specimens from 3-day-old Sprague–Dawley rat pups (Charles River Laboratories, Wilmington, MA) were rinsed with sterile PBS containing penicillin and streptomycin. The perivesical fat and bladder mucosa were removed with fine scissors. The bladder tissue was then placed into DMEM supplemented with 10% FBS and 1% antimicrobial solution (penicillin G/amphoteracin B/streptomycin; Mediatech), minced, immersed in 4 ml of 0.075% type IV collagenase (Sigma, St. Louis, MO) at 37°C, and agitated for 30 min. The digest was centrifuged at 1,000 rpm (200 × g) for 5 min. rBSMC were suspended and plated on T25-ml cell culturing flasks in 5 ml of growth media with 10% FBS and penicillin (100 units/ml) and streptomycin (1 μg/ml). The culture was then incubated in humidified 5% CO2 air atmosphere at 37°C. The cells were cultured with fresh medium 2 days thereafter. Passaging and subculturing of cells were performed as per routine (37).
Isolation of ASC.
Passage P0 PLA cells were diluted to a density of 10 cells per ml. Cells were cloned and subcloned by using limited dilution. The single PLA cell-derived colonies were expanded in cloning medium (15% FBS, 1% antibiotic–antimycotic in F-12/DMEM; a 1:1 mixture). To assess lineage plasticity, isolated clones were subjected to leiomyogenic, osteogenic, and adipogenic differentiation.
Smooth Muscle Differentiation.
The effect of the medium and the substrate on the induction of a differentiated smooth muscle phenotype was evaluated. To study the effect of the medium, cells were cultured either in CM or in SMIM consisting of MCDB131 (Sigma) supplemented with 1% FBS at varying concentrations of heparin (VWR International, West Chester, PA). To study the effect of the substrate, cells were plated either in regular uncoated culture dishes or in regular dishes coated with laminin or collagen. P2 cells were harvested, plated in control medium at 5,000 cells per cm2 in regular and coated dishes for 72 h, and then incubated in either SMIM or control medium for an additional 6 weeks. The medium was replaced every 3–4 days. Smooth muscle differentiation was assessed by cell morphology, immunohistochemistry, and Western blot expression of smooth muscle markers and genetic characterization using semiquantitative and real-time PCR.
RNA Isolation and RT-PCR.
All reagents are from Promega (Madison, WI) unless otherwise stated. DNase-treated total cellular RNA was isolated from cell cultures. All amplifications were performed in a Mastercycler thermal cycler (Eppendorf, Westbury, NY) for 35 cycles after an initial 2-min denaturation at 94°C. The same reaction profile was used for all primers. The PCR primer sequences are listed in Table 1, which is published as supporting information on the PNAS web site. Reaction products were analyzed by electrophoresis of 10-μl aliquots in 1.5% agarose gels, and the DNA fragments were visualized by ethidium bromide staining. Semiquantitative analysis of mRNA expression was performed by using the ImageJ software (http://rsb.info.nih.gov//ij) using human glyceraldehyde-3-phosphate dehydrogenase (GAPDH) as a housekeeping gene.
Quantitative Real-Time PCR.
The expression of ASMA, calponin, and SM22 was quantified for all differentiation conditions described above. Total cellular RNA was reverse transcribed by using TaqMan Gold RT-PCR kit for real-time PCR (Applied Biosystems, Foster City, CA). Quantitative real-time PCR was performed in an ABI 7000 Prism Sequence detection system. Primer and probe sequences were designed by the UCLA Sequencing Core Facility, and probes were synthesized by BioSource (Camarillo, CA) having a 5′ fluorogenic probe 6FAM and a 3′ quencher TAMRA. The relative abundance of transcripts was normalized to the constitutive expression of GAPDH.
Immunohistochemistry.
Immunohistochemistry was performed by using R.T.U. VECTASTAIN Elite ABC kit (Vector Laboratories, Burlingame, CA). Briefly, adherent cells were washed and then fixed with acetone/methanol 1:1 for 5 min. Endogenous biotin was blocked by using the Avidin/Biotin Blocking kit (Vector Laboratories). Cells were incubated with primary antibody for 30 min, washed, and then incubated with biotinylated secondary antibody for an additional 30 min, followed by a final incubation with the streptavidin–peroxidase complex. The peroxidase reaction was developed by using the DAB substrate chromogen (DAB Substrate kit; Vector Laboratories). Counterstaining was performed with Mayer's hematoxylin (Dako, Carpinteria, CA). Cells were examined by using optical microscopy (Zeiss) equipped with a SPOT digital imaging acquisition system (Diagnostic Instruments). Control experiments were performed by using an irrelevant primary antibody.
Western Blot.
Cells were washed with PBS, and proteins were extracted through treatment with triton lysis buffer, subjected to SDS polyacrylamide gels, and blotted onto nitrocellulose membranes. Visualization of secondary antibody binding was performed by chemiluminescence (Tropix, Bedford, MA). Primary antibodies used were anti-smoothelin (Chemicon, Temecula, CA), anti-calponin (Dako), anti-smooth muscle myosin (Biomedical Technologies, Stoughton, MA), anti-ASMA (Dako), anti-heavy caldesmon (Dako), anti-CD105 (BD PharMingen, San Diego, CA), anti-vWfactor (Dako), and anti-prolyl-4-hydroxylase (Dako).
Multilineage Differentiation.
Expanded clones were analyzed for leiomyogenic (as described above), osteogenic, and adipogenic differentiation. Osteogenic differentiation was evaluated at 3 weeks by histochemical presence of alkaline phosphatase and mineralized extracellular matrix. Adipogenic differentiation was evaluated at 2 weeks by detection of lipid accumulation by oil red O staining.
Functional Studies.
Qualitative carbachol contraction.
PLA and ADC were cultured under SMIM for 6 weeks, washed with PBS, and incubated with a bath solution consisting of 155 mM NaCl, 4.5 mM KCl, 2 mM CaCl2, 1 mM MgCl2, 5 mM glucose, and 10 mM Hepes. Contraction was elicited by incubation with 10−5 M carbachol (Calbiochem), and relaxation was subsequently elicited by addition of 10−4 M atropine (Sigma). Bright-field images were obtained by using a Leica DM IRB microscope attached to a real-time camera.
Pharmacologic studies using collagen gels.
To determine whether smooth muscle-differentiated PLA cells gained the ability to contract when exposed to pharmacologic agents, we mixed the cells into a 3D collagen matrix in the form of a soft, compressible gel disk to visualize contractions that might not be easily discernible if cells were anchored on a hard surface such as a tissue culture dish. The experimental groups were: (i) smooth muscle-differentiated PLA cell, (ii) rBSMCs at passage no more than 5 as positive control, (iii) human T lymphocyte line (SupT1, T lymphoblastoid cell line, NIH) as negative control, and (iv) unseeded gels. To prepare the collagen gels, mixtures of 50% volume collagen solution (3.0 mg/ml; Vitrogen), 25% volume 4× DMEM prepared from powder (Gibco, Grand Island, NY) with supplemented sodium bicarbonate, ≈4-μl volume of 120- 150-μm-diameter blue-tinted soda lime glass microspheres (Mo-Scix, Rolla, MO), 1-μl volume of 210- to 250-μm-diameter clear glass beads (Polysciences, Warrington, PA), and 5 × 105 cells in a total volume of 400 μl were prepared on ice. The blue and clear glass beads were randomly mixed into the gel disk to produce positional markers in easily distinguishable patterns to facilitate tracking and measurement. The cold slurries were transferred to the wells on a 24-well tissue culture plate and polymerized overnight in a 37°C incubator. The thickness of the collagen gels was ≈2 mm and reproducibly controlled by delivering identical volume of gel into each well. Appropriate culture medium was added the next day to supplement cell growth, and gels were incubated for an additional 24 h.
To quantify the degree of contraction, pictures of the gel at marked fields were taken at ×25 and/or ×50 with a real-time camera connected to a microscope. The gel was released 280° around the circumference so that it was allowed to contract freely, while a portion of the gel remained attached to a small part of the well to prevent gels from floating in the medium and facilitate bead tracking for contraction measurements relative to a fixed point. Pictures were taken every 5–15 min. Once gel contraction from the mechanical release alone had stabilized, drugs premixed in DMEM were added at specific concentrations to test their effects on contractility. The ability of differentiated PLA cells to contract in response to 10−11 M, 10−8 M, and 10−5 M carbachol was evaluated. Similarly, the ability of differentiated PLA cells to respond to atropine was investigated by incubation in 10−4 M atropine for 45 min followed by addition of carbachol. Results were interpreted by measuring the change of the distance between glass beads over time.
Supplementary Material
Acknowledgments
We thank Drs. Daniel Blander and Shlomo Raz for comments on the manuscript. This work was funded in part by National Institute of Child Health and Human Development Building Interdisciplinary Research Careers in Women's Health (BIRCWH) Grant 5-K12-HD01400, the American Geriatrics Society Jahnigen Career Development Scholars Award, and National Institute of Diabetes and Digestive and Kidney Diseases Grant R01 DK067198-01.
Abbreviations
- PLA
processed lipoaspirate
- ASC
adipose stem cells
- CM
control media
- SMC
smooth muscle cell
- ASMA
SMC-specific α actin
- rBSMC
rat bladder SMC
- SMIM
smooth muscle inductive medium.
Footnotes
Conflict of interest statement: No conflicts declared.
References
- 1.Lai J. Y., Yoon C. Y., Yoo J. J., Wulf T., Atala A. J. Urol. 2002;168:1853–1857. doi: 10.1097/01.ju.0000030040.76258.5a. discussion, 1858. [DOI] [PubMed] [Google Scholar]
- 2.Lin H. K., Cowan R., Moore P., Zhang Y., Yang Q., Peterson J. A., Jr., Tomasek J. J., Kropp B. P., Cheng E. Y. J. Urol. 2004;171:1348–1352. doi: 10.1097/01.ju.0000108800.47594.8b. [DOI] [PubMed] [Google Scholar]
- 3.Orlic D., Kajstura J., Chimenti S., Jakoniuk I., Anderson S. M., Li B., Pickel J., McKay R., Nadal-Ginard B., Bodine D. M., et al. Nature. 2001;410:701–705. doi: 10.1038/35070587. [DOI] [PubMed] [Google Scholar]
- 4.Silva G. V., Litovsky S., Assad J. A., Sousa A. L., Martin B. J., Vela D., Coulter S. C., Lin J., Ober J., Vaughn W. K., et al. Circulation. 2005;111:150–156. doi: 10.1161/01.CIR.0000151812.86142.45. [DOI] [PubMed] [Google Scholar]
- 5.Religa P., Bojakowski K., Maksymowicz M., Bojakowska M., Sirsjo A., Gaciong Z., Olszewski W., Hedin U., Thyberg J. Transplantation. 2002;74:1310–1315. doi: 10.1097/00007890-200211150-00019. [DOI] [PubMed] [Google Scholar]
- 6.Lakshmanan Y., Frimberger D., Gearhart J. D., Gearhart J. P. Urology. 2005;65:821–826. doi: 10.1016/j.urology.2004.11.022. [DOI] [PubMed] [Google Scholar]
- 7.Sinha S., Hoofnagle M. H., Kingston P. A., McCanna M. E., Owens G. K. Am. J. Physiol. 2004;287:C1560–C1568. doi: 10.1152/ajpcell.00221.2004. [DOI] [PubMed] [Google Scholar]
- 8.Zuk P. A., Zhu M., Ashjian P., De Ugarte D. A., Huang J. I., Mizuno H., Alfonso Z. C., Fraser J. K., Benhaim P., Hedrick M. H. Mol. Biol. Cell. 2002;13:4279–4295. doi: 10.1091/mbc.E02-02-0105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zuk P. A., Zhu M., Mizuno H., Huang J., Futrell J. W., Katz A. J., Benhaim P., Lorenz H. P., Hedrick M. H. Tissue Eng. 2001;7:211–228. doi: 10.1089/107632701300062859. [DOI] [PubMed] [Google Scholar]
- 10.Miranville A., Heeschen C., Sengenes C., Curat C. A., Busse R., Bouloumie A. Circulation. 2004;110:349–355. doi: 10.1161/01.CIR.0000135466.16823.D0. [DOI] [PubMed] [Google Scholar]
- 11.Bacou F., el Andalousi R. B., Daussin P. A., Micallef J. P., Levin J. M., Chammas M., Casteilla L., Reyne Y., Nougues J. Cell Transplant. 2004;13:103–111. doi: 10.3727/000000004773301771. [DOI] [PubMed] [Google Scholar]
- 12.Cowan C. M., Shi Y. Y., Aalami O. O., Chou Y. F., Mari C., Thomas R., Quarto N., Contag C. H., Wu B., Longaker M. T. Nat. Biotechnol. 2004;22:560–567. doi: 10.1038/nbt958. [DOI] [PubMed] [Google Scholar]
- 13.Cousin B., Andre M., Arnaud E., Penicaud L., Casteilla L. Biochem. Biophys. Res. Commun. 2003;301:1016–1022. doi: 10.1016/s0006-291x(03)00061-5. [DOI] [PubMed] [Google Scholar]
- 14.Planat-Benard V., Menard C., Andre M., Puceat M., Perez A., Garcia-Verdugo J. M., Penicaud L., Casteilla L. Circ. Res. 2004;94:223–229. doi: 10.1161/01.RES.0000109792.43271.47. [DOI] [PubMed] [Google Scholar]
- 15.Miano J. M. Results Probl. Cell Differ. 2002;38:39–59. doi: 10.1007/978-3-540-45686-5_2. [DOI] [PubMed] [Google Scholar]
- 16.Simper D., Stalboerger P. G., Panetta C. J., Wang S., Caplice N. M. Circulation. 2002;106:1199–1204. doi: 10.1161/01.cir.0000031525.61826.a8. [DOI] [PubMed] [Google Scholar]
- 17.Kashiwakura Y., Katoh Y., Tamayose K., Konishi H., Takaya N., Yuhara S., Yamada M., Sugimoto K., Daida H. Circulation. 2003;107:2078–2081. doi: 10.1161/01.CIR.0000070082.64414.B5. [DOI] [PubMed] [Google Scholar]
- 18.Wolf N. S., Penn P. E., Rao D., McKee M. D. Exp. Cell Res. 2003;290:346–357. doi: 10.1016/s0014-4827(03)00321-5. [DOI] [PubMed] [Google Scholar]
- 19.Erickson G. R., Gimble J. M., Franklin D. M., Rice H. E., Awad H., Guilak F. Biochem. Biophys. Res. Commun. 2002;290:763–769. doi: 10.1006/bbrc.2001.6270. [DOI] [PubMed] [Google Scholar]
- 20.Safford K. M., Hicok K. C., Safford S. D., Halvorsen Y. D., Wilkison W. O., Gimble J. M., Rice H. E. Biochem. Biophys. Res. Commun. 2002;294:371–379. doi: 10.1016/S0006-291X(02)00469-2. [DOI] [PubMed] [Google Scholar]
- 21.Small J. V., Gimona M. Acta Physiol. Scand. 1998;164:341–348. doi: 10.1046/j.1365-201X.1998.00441.x. [DOI] [PubMed] [Google Scholar]
- 22.Yang Y., Relan N. K., Przywara D. A., Schuger L. Development (Cambridge, U.K.) 1999;126:3027–3033. doi: 10.1242/dev.126.13.3027. [DOI] [PubMed] [Google Scholar]
- 23.van der Loop F. T., Schaart G., Timmer E. D., Ramaekers F. C., van Eys G. J. J. Cell Biol. 1996;134:401–411. doi: 10.1083/jcb.134.2.401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Worth N. F., Rolfe B. E., Song J., Campbell G. R. Cell Motil. Cytoskeleton. 2001;49:130–145. doi: 10.1002/cm.1027. [DOI] [PubMed] [Google Scholar]
- 25.Desmouliere A., Rubbia-Brandt L., Grau G., Gabbiani G. Lab. Invest. 1992;67:716–726. [PubMed] [Google Scholar]
- 26.Horiuchi A., Nikaido T., Ya-Li Z., Ito K., Orii A., Fujii S. Mol. Hum. Reprod. 1999;5:139–145. doi: 10.1093/molehr/5.2.139. [DOI] [PubMed] [Google Scholar]
- 27.Qin H., Ishiwata T., Wang R., Kudo M., Yokoyama M., Naito Z., Asano G. Exp. Mol. Pathol. 2000;69:79–90. doi: 10.1006/exmp.2000.2321. [DOI] [PubMed] [Google Scholar]
- 28.Relan N. K., Yang Y., Beqaj S., Miner J. H., Schuger L. J. Cell Biol. 1999;147:1341–1350. doi: 10.1083/jcb.147.6.1341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yang Y., Palmer K. C., Relan N., Diglio C., Schuger L. Development (Cambridge, U.K.) 1998;125:2621–2629. doi: 10.1242/dev.125.14.2621. [DOI] [PubMed] [Google Scholar]
- 30.Kinner B., Zaleskas J. M., Spector M. Exp. Cell Res. 2002;278:72–83. doi: 10.1006/excr.2002.5561. [DOI] [PubMed] [Google Scholar]
- 31.Oishi K., Ogawa Y., Gamoh S., Uchida M. K. J. Physiol. (London) 2002;540:139–152. doi: 10.1113/jphysiol.2001.013278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Arakawa E., Hasegawa K., Irie J., Ide S., Ushiki J., Yamaguchi K., Oda S., Matsuda Y. J. Cardiovasc. Pharmacol. 2003;42:745–751. doi: 10.1097/00005344-200312000-00008. [DOI] [PubMed] [Google Scholar]
- 33.Hirschi K. K., Rohovsky S. A., D'Amore P. A. J. Cell Biol. 1998;141:805–814. doi: 10.1083/jcb.141.3.805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gualandris A., Annes J. P., Arese M., Noguera I., Jurukovski V., Rifkin D. B. Mol. Biol. Cell. 2000;11:4295–4308. doi: 10.1091/mbc.11.12.4295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Remy-Martin J. P., Marandin A., Challier B., Bernard G., Deschaseaux M., Herve P., Wei Y., Tsuji T., Auerbach R., Dennis J. E., et al. Exp. Hematol. 1999;27:1782–1795. doi: 10.1016/s0301-472x(99)00122-8. [DOI] [PubMed] [Google Scholar]
- 36.Charbord P., Oostendorp R., Pang W., Herault O., Noel F., Tsuji T., Dzierzak E., Peault B. Exp. Hematol. 2002;30:1202–1210. doi: 10.1016/s0301-472x(02)00895-0. [DOI] [PubMed] [Google Scholar]
- 37.Toma J. G., Akhavan M., Fernandes K. J., Barnabe-Heider F., Sadikot A., Kaplan D. R., Miller F. D. Nat. Cell Biol. 2001;3:778–784. doi: 10.1038/ncb0901-778. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.