Abstract
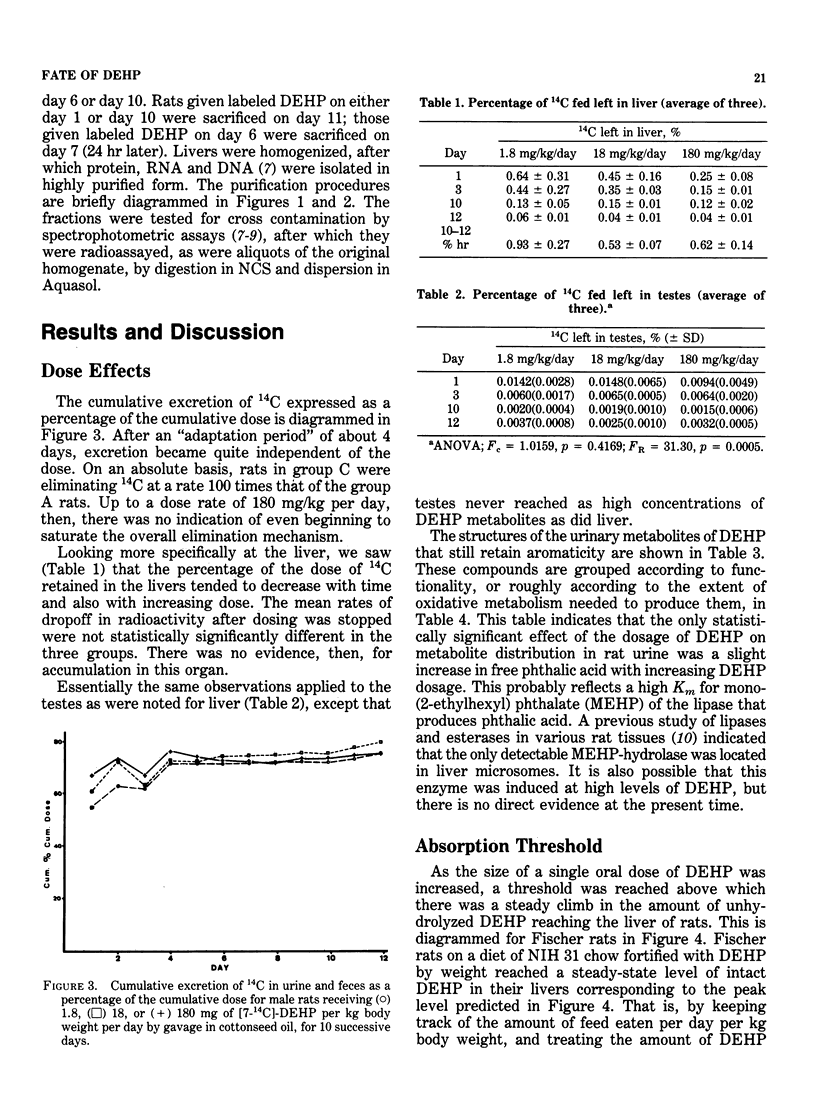
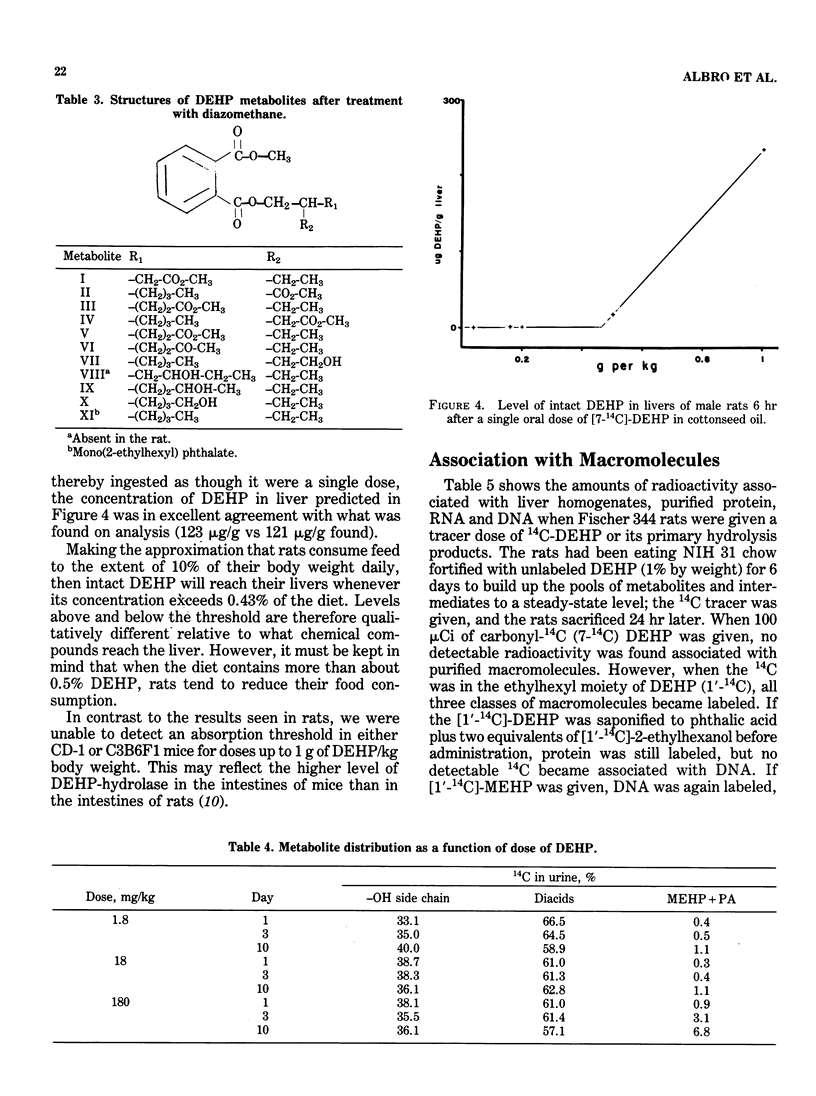
The recent long-term carcinogenesis bioassay of di(2-ethylhexyl) phthalate (DEHP) in rats and mice reported by the National Toxicology Program was the first such bioassay to implicate DEHP as a hepatocarcinogen. At the levels of DEHP fed (up to 1.2% of the diet for two years), the livers of the rats would have been exposed to unhydrolyzed diester; this would not have been the case at lower dosages. Extrapolation to lower dosages is therefore questionable. We do not have sufficient pharmacokinetic data in mice to evaluate the dose relationships as yet. Rodents differ conspicuously from primates in their manner of metabolizing DEHP, both in terms of the demand made on the oxidation potential of the liver and in the chemical properties of the major metabolites. The relevance of these differences must be determined before rodent species can be considered models for the effects of DEHP in humans. Radioactivity from carbonyl-labeled DEHP did not associate with purified protein, RNA or DNA from rat liver in vivo. Label from 2-ethyl-(1-14C)-hexyl-labeled DEHP or mono(2-ethylhexyl) phthalate (MEHP) did appear to associate strongly with purified DNA, but label from free 14C-labeled 2-ethylhexanol did not. The apparent binding from DEHP and MEHP was not exchangeable, but was not proven to be covalent. This phenomenon needs additional study.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Albro P. W., Hass J. R., Peck C. C., Odam D. G., Corbett J. T., Bailey F. J., Blatt H. E., Barrett B. B. Identification of the metabolites of di-(2-ethylhexyl) phthalate in urine from the African green monkey. Drug Metab Dispos. 1981 May-Jun;9(3):223–225. [PubMed] [Google Scholar]
- Albro P. W., Moore B. Identification of the metabolites of simple phthalate diesters in rat urine. J Chromatogr. 1974 Jul 17;94(0):209–218. doi: 10.1016/s0021-9673(01)92368-4. [DOI] [PubMed] [Google Scholar]
- Albro P. W., Thomas R. O. Enzymatic hydrolysis of di-(2-ethylhexyl) phthalate by lipases. Biochim Biophys Acta. 1973 Jun 21;306(3):380–390. doi: 10.1016/0005-2760(73)90176-8. [DOI] [PubMed] [Google Scholar]
- Albro P. W., Thomas R., Fishbein L. Metabolism of diethylhexyl phthalate by rats. Isolation and characterization of the urinary metabolites. J Chromatogr. 1973 Feb 28;76(2):321–330. doi: 10.1016/s0021-9673(01)96915-8. [DOI] [PubMed] [Google Scholar]
- Daniel J. W., Bratt H. The absorption, metabolism and tissue distribution of di(2-ethylhexyl)phthalate in rats. Toxicology. 1974 Mar;2(1):51–65. doi: 10.1016/0300-483x(74)90042-0. [DOI] [PubMed] [Google Scholar]
- FOLCH J., LEES M., SLOANE STANLEY G. H. A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem. 1957 May;226(1):497–509. [PubMed] [Google Scholar]
- Harvan D. J., Hass J. R., Albro P. W., Friesen M. D. Mass spectrometry of di-(2-ethylhexyl)phthalate metabolites. Biomed Mass Spectrom. 1980 Jun;7(6):242–246. doi: 10.1002/bms.1200070604. [DOI] [PubMed] [Google Scholar]
- JEVONS F. R. DETECTION OF NON-REDUCING CARBOHYDRATE WITH ORCINOL IN PHOSPHORIC ACID. J Chromatogr. 1964 Oct;16:252–253. doi: 10.1016/s0021-9673(01)82475-4. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Markov G. G., Ivanov I. G. Hydroxyapatite column chromatography in procedures for isolation of purified DNA. Anal Biochem. 1974 Jun;59(2):555–563. doi: 10.1016/0003-2697(74)90309-1. [DOI] [PubMed] [Google Scholar]
- Stephen J. M., Waterlow J. C. Use of carbon-14-labelled arginine to measure the catabolic rate of serum and liver proteins and the extent of amino-acid recycling. Nature. 1966 Aug 27;211(5052):978–980. doi: 10.1038/211978a0. [DOI] [PubMed] [Google Scholar]


