Abstract
The biosynthesis of endothelin-1 is increased in the diabetic state. So this peptide may cause diabetic vascular complications. We tested this possibility by chronically administering J-104132, a potent orally active mixed antagonist of endothelin A and B (ETA/ETB) receptors to streptozotocin (STZ)-induced diabetic rats and focusing on changes in endothelial function.
The acetylcholine (ACh)-induced endothelium-dependent relaxation was impaired in diabetic rats and this impairment was significantly attenuated following chronic administration of J-104132 (10 mg kg−1, p.o., daily for 4 weeks).
In an in vitro experiment using aortae from diabetic rats, the ACh-induced relaxation was not changed by the presence of J-104132 (3×10−9 M).
The expression levels of the mRNA for endothelial nitric oxide synthase was comparable among aortae from the three groups (control, diabetic and chronically J-104132-treated diabetic).
The amount of superoxide anion was significantly greater in aortae from diabetic rats than in controls. Chronic J-104132 treatment significantly decreased the level of superoxide anion in diabetic rats.
The expression of the p22phox mRNA for the NADH/NADPH oxidase subunit was significantly increased in STZ-induced diabetic rats and this increase was completely prevented by chronic administration of J-104132.
These results suggest that in STZ-induced diabetic rats, ET-1 may be directly involved in impairing endothelium-dependent relaxation via increased superoxide-anion production.
Keywords: Endothelin-1, endothelin antagonist, aorta, diabetes, superoxide-anion
Introduction
Endothelin-1 (ET-1), a vasoconstrictor peptide secreted from endothelial cells, is thought to play a pathological role in a number of vascular diseases (Goto et al., 1996). Plasma ET-1 levels are increased in the diabetic state (Takahashi et al., 1990; Makino & Kamata, 1998; 2000; Makino et al., 2001) and the plasma concentration of big endothelin-1, the precursor of ET-1, is elevated in patients with diabetes mellitus (Tsunoda et al., 1991). Thus, although ET-1 may also contribute to the normal maintenance of vascular tone, a pathogenetic role is implied by its overproduction in patients with diabetes (Takahashi et al., 1990; Haak et al., 1992).
An accumulating body of evidence indicates that the relaxation responses induced in aortic strips by endothelium-dependent agents are weak in streptozotocin (STZ)-induced diabetic rats (Oyama et al., 1986; Kamata et al., 1989; Poston & Taylor, 1995; Pieper, 1998; Kamata & Kobayashi, 1996; Kobayashi & Kamata, 1999a, 1999b; 2001; Kobayashi et al., 2000; de vriese et al., 2000; Hink et al., 2001). Thus, endothelial dysfunction is likely to play a key role in the pathogenesis of diabetic vascular disease (de vriese et al., 2000). Since the plasma ET-1 level is increased in diabetic rats, the impairment of the endothelium may conceivably be related to this level. If this is indeed the case, chronic administration of an endothelin antagonist to diabetic rats should improve endothelial function. However, another possible explanation for the endothelial dysfunction seen in the diabetic state is that it is a consequence of the stress placed on endothelial cells by such diabetes-related phenomena as the increased plasma glucose, oxidative stress and/or the increased plasma levels of low-density lipoprotein (LDL), oxidized LDL and triglyceride. If this is indeed the case, use of an endothelin antagonist will not improve endothelial function in diabetic rats. By chronically administering the endothelin antagonist J-104132, a potent orally active mixed antagonist of endothelin A and B (ETA/ETB) receptors (Nishikibe et al., 1999; Ikeda et al., 1999), we tested the idea that the increase in ET-1 seen in streptozotocin-induced diabetic rats is a cause, rather than a consequence, of the endothelial dysfunction reported in these animals.
Recently, we reported that the impairment of the ACh-induced relaxation seen in aortic strips obtained from diabetic rats may be due to the presence of an abnormal oxidative metabolism of NO, rather than to decreases in eNOS mRNA and NO production (Kobayashi & Kamata, 2001). For this reason, when we examined the effects of J-104132 on endothelial dysfunction, we focused on the superoxide-anion system.
Methods
Animals and experimental design
Male Wistar rats, 7 weeks old and 220 – 300 g in weight, received a single injection via the tail vein of STZ 75 mg kg−1, dissolved in a citrate buffer. Age-matched control rats were injected with the buffer alone. Food and water were given ad libitum. This study was conducted in accordance with the Guide for the Care and Use of Laboratory Animals adopted by the Committee on the Care and Use of Laboratory Animals of Hoshi University (which is accredited by the Ministry of Education, Science, Sports and Culture, Japan).
J-104132 treatment
Starting 7 weeks after the STZ injection, the STZ-induced diabetic rats were given J-104132 (10 m kg−1, p.o., daily) for 4 weeks. Eleven weeks after the STZ injection, the rats were killed by decapitation under diethyl ether anaesthesia.
Measurement of plasma glucose, cholesterol, HDL and triglycerides
Eleven weeks after the STZ injection, plasma total cholesterol and triglyceride were determined using a commercially available enzyme kit (Wako Chemical Company, Osaka, Japan). Cholesterol ester mass was calculated as (total cholesterol −free cholesterol) ×1.68 (Tsai et al., 1992).
Preparation of VLDL and LDL
Lipoproteins (VLDL, d=0.96 – 1.019; LDL, d=1.019 – 1.063) were isolated by density-gradient ultracentrifugation (Havel et al., 1955) from pooled plasma (EDTA, 1 mM) obtained from age-matched controls, diabetic rats, or J-104132-treated diabetic rats. The lipoproteins were extensively dialyzed against phosphate-buffered saline (PBS). The concentration of glucose in the plasma was determined by the O-toluidine method.
Measurement of isometric force
Rats were anaesthetized with diethyl ether and killed by decapitation 11 weeks after treatment with STZ or buffer. A section of the aorta from between the aortic arch and the diaphragm was then removed and placed in oxygenated, modified Krebs – Henseleit solution. The solution consisted of (mM): NaCl 118.0, KCl 4.7, NaHCO3 25.0, CaCl2 1.8, NaH2PO4, MgSO4 1.2, dextrose 11.0. The aorta was cleaned of loosely adhering fat and connective tissue and cut into helical strips 2 mm in width and 20 mm in length. The tissue was then placed in a bath containing 10 ml of well-oxygenated (95% O2, 5% CO2) KHS at 37°C. With one end of the tissue connected to a tissue holder and other to a force-displacement transducer (Nihon Kohden, TB611T), it was allowed to equilibrate for 60 min under a resting tension of 1.0 g (determined to be optimal in preliminary experiments). During this period, the Krebs – Henseleit solution in the tissue bath was replaced every 20 min. After equilibration, each aortic strip was contracted with 10−6 M noradrenaline (NA) and the presence of functional endothelial cells was confirmed by demonstrating relaxations in response to 10−5 M ACh. For the relaxation studies, the aortic strips, which were weighed at the end of each experiment, were precontracted with an equieffective concentration of NA (5×10−8∼3×10−7 M). When the NA-induced contraction had reached a plateau level, ACh (10−9∼10−5 M) or sodium nitroprusside (SNP) (10−10∼10−5 M) was added in a cumulative manner.
Measurement of the expression of the mRNA for endothelial nitric oxide synthase (eNOS)
Oligonucleotides
Oligonucleotides (ON) for rat endothelial nitric oxide synthase (eNOS) were used with primers, as described previously. The primers (with the respective Gen-Bank data library accession numbers and the coding sequence of the PCR-amplified product given in brackets) were:rat glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (X02231, position 492 – 799, amplification of a 308 bp sequence) ON 1: 5′-TCCCTCAAGATTGTCAGCAA-3′, ON 2: 5′-AGATCCACAACGGATACATT-3′: rat eNOS (RNU02534, amplification of a 693 bp sequence) ON 3: 5′-TCCAGAAACACAGACAGTGCA-3′, ON 4: 5′-CAGGAAGTAAGTGAGAGC-3′; rat p22phox (according to Zalba et al., 2000, amplification of a 435 bp) ON 5: 5′ GCTCATCTGTCTGCTGGAGTA-3′, ON 6: 5′-ACGACCTCATCTGTCACTGGA-3′.
RNA isolation and RT – PCR
RNA was isolated using the guanidinium method (Chomczynski & Sacchi, 1987). Aortae were carefully isolated and cleaned of adhering parenchyma and connective tissue, then homogenized in RNA buffer. The RNA was quantified by ultraviolet absorbance spectrophotometry. For the RT – PCR analysis, first-standard cDNA was synthesized from total RNA using Oligo (dT) and a cDNA Synthesis Kit (Life Science). Twenty-four (GAPDH) or thirty (eNOS) PCR cycles (94°C for 1 min, 62°C for 1 min, 72°C for 1 min) and p22phox PCR cycles (94°C for 1 min, 57°C for 1 min, 72°C for 1 min) were performed using one half of the reverse-transcription (RT) mixture. The PCR products so obtained were analysed on ethidium-bromide-stained agarose (1.5%) gel. The eNOS, p22phox and GAPDH products were quantified by scanning densitometry, the amount of eNOS and p22phox being normalized with respect to the amount of GAPDH product.
Quantification of superoxide anion by measurement of the amount of NBT reduced
Aortic rings were incubated with nitro blue tetrazolium (NBT) to allow the O2− generated by the tissue to reduce the NBT to blue formazan. The details of this assay have been published previously (Wang et al., 1998). Briefly, aortae were cut into transverse rings 10 mm in length. These were placed for 1.5 h in 5 ml buffer at 37°C containing NBT (100 μmol l−1) in the presence or absence of ACh (10−7 M). NBT reduction was stopped by addition of 0.5 N HCl (5 ml). After this incubation, the rings were minced and homogenized in a mixture of 0.1 N NaOH and 0.1% SDS in water containing 40 mg l−1 diethylentriaminepentaacetic acid. The mixture was centrifuged at 20,000×g for 20 min and the resultant pellet resuspended in 1.5 ml of pyridine kept at 80°C for 1.5 h to extract formazan. The mixture was then subjected to a second centrifugation at 10,000×g for 10 min. The absorbance of the formazan was determined spectrophotometrically at 540 nm. The amount of NBT reduced (=quantity of formazan), was calculated as follows: amount of NBT reduced=A•V/(T•Wt•ε•l), where A is the absorbance, V is the volume of pyridine, T is the time for which the rings were incubated with NBT, Wt is the blotted wet weight of the aortic rings, ε is the extinction coefficient (0.7 l mmol−1 per mm) and l is the length of the light path. The results are reported in pmol min−1 per Wt mg.
Drugs
Streptozotocin, nitro blue tetrazolium (NBT), diethylentriaminepentaacetic acid, (−) noradrenaline hydrochloride and sodium nitroprusside were all purchased from Sigma Chemical Co. (St. Louis, MO, U.S.A.) and acetylcholine chloride from Daiichi Pharmaceutical Co. Ltd. (Tokyo, Japan). J-104132 ((+)-(5S,6R,7R)-2-butyl-7-[2-(2S)-2-carboxypropyl]-5-(3,4-methyenedioxyphenyl) cycloperteno[1,2-b] pyridine-6-carboxylic acid) was gifted by Banyu Co. Ltd. (Tsukuba, Japan). All drugs were dissolved in distilled water. All concentrations are expressed as the final molar concentration of the base in the organ bath.
Statistical analysis
Data are expressed as the mean±s.e.mean. Where appropriate, statistical differences were determined by Dunnett's test for multiple comparisons after a one-way analysis of variance, a probability level of P<0.05 being regarded as significant. Statistical comparisons between concentration-response curves were made by a two-way ANOVA, with Bonferroni's correction for multiple comparisons being performed post hoc; P<0.05 was considered significant.
Results
Plasma glucose, cholesterol and triglyceride levels
As shown in Table 1, the plasma glucose level was significantly higher in STZ-induced diabetes than in controls. Treatment with J-104132 (10 mg kg−1, p.o. for 4 weeks) did not alter plasma glucose, total cholesterol, HDL, VLDL, LDL or triglyceride in our established diabetic rats.
Table 1.
Levels of various plasma parameters in age-matched controls, STZ-diabetic rats and J-104132-treated diabetic rats
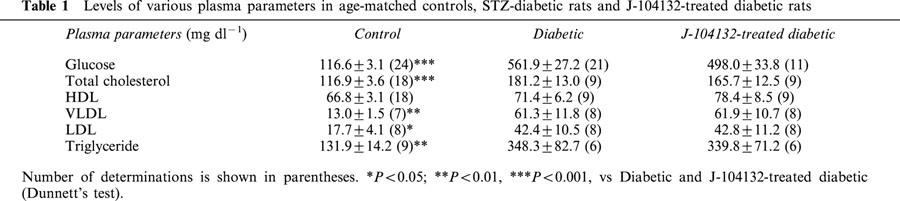
Relaxation responses to ACh and SNP
When the NA (5×10−8∼3×10−7 M)-induced contraction had reached a plateau, ACh (1×10−9∼1×10−5 M) or SNP (1×10−10∼1×10−5 M) was added cumulatively (Figure 1). In aortic strips from age-matched control rats, ACh (1×10−9∼1×10−5 M) caused a concentration-dependent relaxation, with the maximum response at 10−5 M. This relaxation was significantly weaker in strips from STZ-induced diabetic rats (P<0.05). Aortic strips from STZ-induced diabetic rats chronically treated with J-104132 (10 mg kg−1, p.o. daily for 4 weeks) relaxed in a normal way to ACh (Figure 1a). The relaxation responses caused by SNP (1×10−10∼1×10−5 M) did not differ significantly among the three groups (Figure 1b).
Figure 1.
Concentration-response curves for ACh-induced (a) and SNP-induced (b) relaxations of aortic strips obtained from age-matched controls, untreated diabetic rats and chronically J-104132-treated diabetic rats. Ordinate shows relaxation of aortic strips as a percentage of the contraction induced by an equieffective concentration of noradrenaline (5×10−8∼3×10−7 M). Each data point represents the mean±s.e.mean of 6∼8 experiments; the s.e. is included only when it exceeds the dimension of the symbol used. *P<0.05, diabetic vs control; #P<0.05, diabetic vs J-104132-treated diabetic (two-way ANOVA).
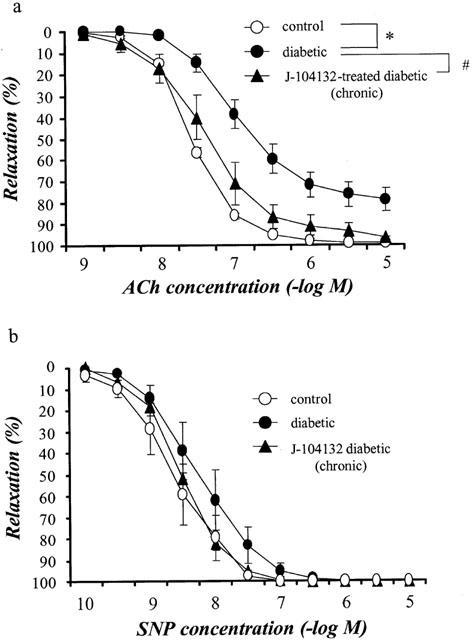
In marked contrast, following incubation of aortic strips in vitro with J-104132 (3×10−9 M), the impaired ACh-induced relaxation seen in strips from STZ-induced diabetic rats showed no improvement (Figure 2).
Figure 2.
Concentration-response curves for ACh-induced relaxation of aortic strips obtained from age-matched controls and from untreated diabetic rats. Strips either treated in vitro with 3×10−9 M J-104132 for 20 min or left untreated. Ordinate shows relaxation of aortic strips as a percentage of the contraction induced by an equieffective concentration of noradrenaline (5×10−8∼3×10−7 M). Each data point represents the mean±s.e.mean of 6∼8 experiments; the s.e. is included only when it exceeds the dimension of the symbol used. ***P<0.001, diabetic vs control (two-way ANOVA).
Expression of the mRNA for eNOS
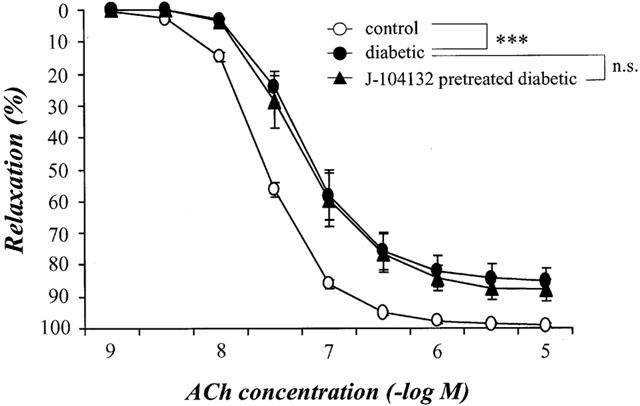
To investigate the possible mechanisms underlying the impaired ACh-induced relaxation seen in STZ-induced diabetic rats and its normalization in chronically J-104132-treated individuals, we examined whether the expression of the mRNA for eNOS is changed by chronic J-104132 treatment. Use of RT – PCR on the total RNA isolated from the aortae of age-matched controls, untreated diabetic and chronically J-104132-treated diabetic rats revealed that the expression ratio eNOS/GAPDH did not differ among the three groups (Figure 3).
Figure 3.
RT – PCR assay of the expression of the mRNA for endothelial NO synthase (eNOS) in aortae from controls, STZ-diabetic and chronically J-104132-treated STZ-diabetic rats. (a) Expression of the mRNA for eNOS assayed by RT – PCR. (b) Quantitative analysis of expression of the mRNA for eNOS (by scanning densitometry). Control rats (n=7, open column); STZ-induced diabetic rats (n=5, closed column); J-104132-treated diabetic rats (n=6, hatched column). Each column represents the mean±s.e.mean of six determinations (eNOS/GAPDH). The RT – PCR assay was performed as described in Methods. Each total RNA preparation (2.0 μg) was reverse transcribed and half of the cDNA product was PCR-amplified using the appropriate primers, 24 cycles (GAPDH) and 28 cycles (eNOS) being employed. A portion of the PCR reaction product was electrophoresed on a 1.5% agarose gel containing ethiodium bromide.
Quantification of superoxide anion by measurement of amount of NBT reduced
To judge from our measurements of the amount of NBT reduced by O2−, the basal O2− level was greater in aortic rings from diabetic rats than in those from the controls (P<0.05). Chronic administration of J-104132 normalized the increased basal O2−, the difference from the non-treated diabetic group being significant at P<0.01 (Figure 4).
Figure 4.
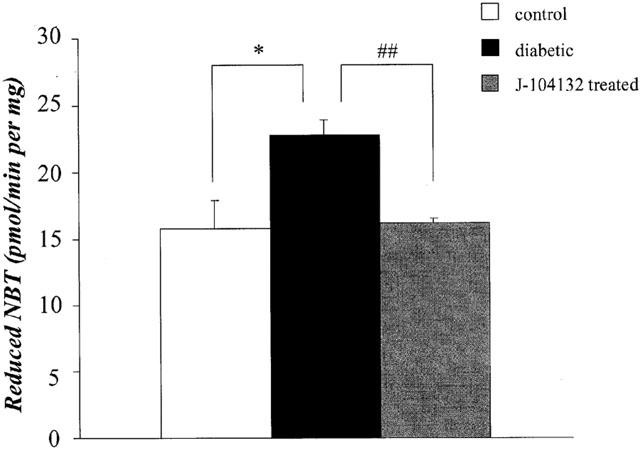
Quantification of superoxide-anion production by measurement of amount of reduced nitro blue tetrazolium (NBT) in age-matched controls, STZ-diabetic and chronically J-104132-treated STZ-diabetic rats. Each column represents the mean±s.e.mean of six experiments.*P<0.05, diabetic vs control; ##P<0.01, diabetic vs J-104132-treated diabetic (Dunnett's test).
Expression of the mRNA for p22phox subunit
To investigate the mechanism underlying the increase in superoxide anion in aortae from diabetic rats and its normalization in J-104132-treated aorta from diabetic rats, we examined whether the expression of the mRNA for the p22phox subunit might have been changed by the chronic J-104132 treatment. Application of RT – PCR to the total RNA isolated from the aortae of age-matched controls, untreated diabetic and chronically J-104132-treated diabetic rats revealed the following. The expression of p22phox mRNA was higher in diabetic rats than in control rats (P<0.05). After treatment with J-104132, the level was lower than in non-treated diabetic rats (P<0.05); in fact, it was similar to that seen in the controls (Figure 5).
Figure 5.
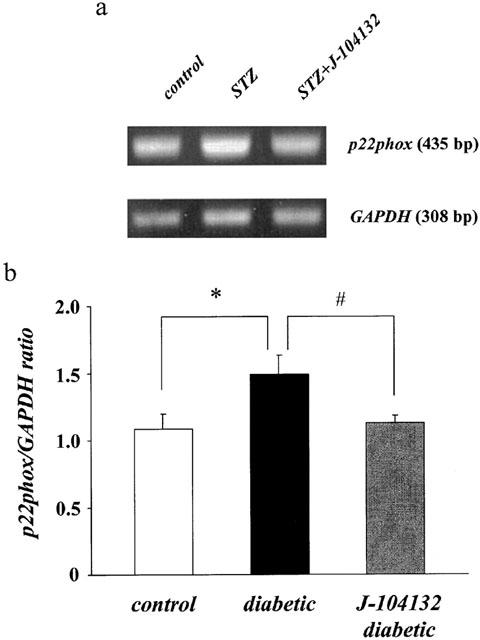
RT – PCR assay of expression of the mRNA for p22phox in aortae from controls, STZ-diabetic and chronically J-104132-treated STZ-diabetic rats. (a) Expression of the mRNA for p22phox assayed by RT – PCR. (b) Quantitative analysis of expression of the mRNA for p22phox (by scanning densitometry). Control rats (n=7, open column); STZ-induced diabetic rats (n=5, closed column); J-104132-treated diabetic rats (n=5, hatched column). Each column represents the mean±s.e.mean of six determinations (p22phox/GAPDH). The RT – PCR assay was performed as described in Methods. Each total RNA preparation (2.0 μg) was reverse transcribed and half of the cDNA product was PCR-amplified using the appropriate primers, 24 cycles (GAPDH) and 28 cycles (p22phox) being employed. A portion of the PCR reaction product was electrophoresed on a 1.5% agarose gel containing ethiodium bromide. *P<0.05, diabetic vs control; #P<0.05, diabetic vs J-104132-treated diabetic (Dunnett's test).
Discussion
The main conclusion from the present study is that in rats with established STZ-induced diabetes, chronic administration of the endothelin antagonist J-104132 improves endothelial function, which is impaired in these animals, and that this effect of J-104132 may be due to a decrease in the aortic superoxide anion, rather than to an increase in NO production.
Because the endothelium can be easily damaged in the preparation of helical strips (e.g. Zonta et al., 1998), we are always very careful to avoid such damage. Our success in this regard is evidenced in the present study by the finding that a relaxation of over 80% of the noradrenaline-induced contraction was induced by as little as 10−7 M ACh (Figure 1a). The reduced endothelium-dependent relaxation seen in diabetic rats in the present study is in agreement with the results of numerous other studies on aortae from STZ-induced diabetic rats (Oyama et al., 1986; Kamata et al., 1989; Poston & Taylor, 1995; Pieper, 1998; Kamata & Kobayashi, 1996; Kobayashi & Kamata, 1999a, 1999b; 2001; Kobayashi et al., 2000; de vriese et al., 2000; Hink et al., 2001).
A considerable body of evidence now suggests that the impairment of endothelium-dependent relaxation seen in diabetes and atherosclerosis may involve inactivation of NO by oxygen-derived free radicals (Kamata & Kobayashi, 1996; Kobayashi & Kamata, 1999a; 2001; Meraji et al., 1987; Pieper et al., 1992; 1996; Hattori et al., 1991; Ooboshi et al., 1997; Lund et al., 1999; Pagano et al., 1998). Production of superoxide anion inactivates NO (Marshall et al., 1988; Rubanyi & Vanhoutte, 1986; Kobayashi & Kamata, 2001) and dismutation of free radicals has generally (Pieper et al., 1996; Hattori et al., 1991; Kamata & Kobayashi, 1996) but not always (Heygate et al., 1995) been found to improve impaired endothelium-dependent relaxation in experimental models of diabetes. Indeed, we recently reported that NO is metabolized by O2− to NO3−, not just to NO2−, and that the resulting rapid inactivation of NO may be responsible for the impairment of endothelium-dependent relaxation seen in aortic strips from diabetic rats (Kobayashi & Kamata, 2001). In the present study, chronic administration of J-104132 effectively restored the ACh-induced endothelium-dependent relaxation, suggesting that NO metabolism may be improved by such treatment. This conclusion is supported by findings that the basal O2− level was greater in aortic rings from diabetic rats than in those from the controls and that the chronic administration of J-104132 to diabetic rats normalized the basal O2− level. In contrast, following incubation of aortic strips in vitro with J-104132, the impaired ACh-induced relaxation seen in strips from STZ-induced diabetic rats showed no improvement, suggesting that J-104132 did not itself produce an antioxidant effect.
Recent studies have underscored the importance of NADPH-oxidase-derived reactive oxygen species in vascular biology. Many components of the leukocyte- NADPH-oxidase complex – including p22phox, p47phox, p67phox and gp91phox (or a related homologue) – have been identified in endothelial cells or vascular smooth muscle cells (Jones et al., 1996; Bayraktutan et al., 1998; Gorlach et al., 2000; Patterson et al., 1999; Ushio-Fukai et al., 1996). Reactive oxygen species from sources other than NADPH oxidase, such as xanthine oxidase (Adkins & Taylor, 1990) or cytochrome P-450 (Bysani et al., 1990), may also play a role. Here, we focused on NADPH oxidase as a source of reactive oxygen species because (i) the gp91phox mRNA for the NADH/NADPH oxidase subunit is upregulated in the steady-state in the aorta in STZ-induced diabetic rats (Hink et al., 2001) and (ii) ET-1 increases the expression of gp91phox mRNA in human endothelial cells (Duerrschmidt et al., 2000). In the present study, we showed that in STZ-induced diabetic rats, the p22phox mRNA for the NADH/NADPH oxidase subunit was significantly increased too, an increase that was completely antagonized by the chronic administration of J-104132. These results are consistent with there being an increase in O2− in STZ-diabetic rats and a normalization of the O2− level by chronic J-104132-treatment. We have previously reported that the plasma ET-1 concentration is increased in STZ-induced diabetic rats and that this increase may be due to an overexpression of the mRNA for prepro ET-1 (Makino & Kamata, 1998; Makino et al., 2001). We have also reported that the overproduction of ET-1 seen in STZ-induced diabetes is a result of hyperglycaemia, not of an increase in LDL cholesterol or triglyceride (Makino & Kamata, 2000). Taken together, the above evidence suggests that the finding that J-104132 induces an improvement in the endothelial dysfunction seen in STZ-induced diabetic rats may be explained by the following sequence of events: (i) the plasma ET-1 concentration is increased in STZ-induced diabetic rats; (ii) the increased ET-1 may stimulate NADPH oxidase, which produces O2−; (iii) the additional O2− may not be metabolized to H2O2 because superoxide dismutase activity is decreased in STZ-induced diabetic rats (Kamata & Kobayashi, 1996; Kobayashi & Kamata, 1999b), a disorder that may lead to an abnormal NO metabolism and a subsequent impairment of endothelium-dependent relaxation; (iv) when J-104132 is chronically administered to STZ-induced diabetic rats, it may have a long-term antagonistic effect on the ET-1-induced stimulation of NADPH oxidase. As a consequence, the enhanced O2− level seen in these animals would be normalized, as indeed we found in the present study, thereby resulting in an improvement in the endothelial dysfunction.
We also demonstrated in the present study that ET-1 may induce NADPH oxidase, because chronic administration of the ET-1-receptor antagonist J-104132 normalized the level of this enzyme.
We found in the present study that chronic administration of J-104132 produces an improvement in the endothelial dysfunction exhibited by aortae from diabetic rats without altering the expression of eNOS mRNA. This suggests that the mechanism underlying the altered endothelial function seen in J-104132-treated diabetics (compared to untreated diabetics) does not depend on a change in eNOS expression.
The elevation in the circulating LDL level seen in diabetes is one of the factors responsible for the endothelial dysfunction. Indeed, STZ-diabetic mice are improved by the chronic administration of cholestyramine, an effect due to a lowering of the plasma LDL level (Kamata et al., 1996). Chronic administration of pravastatin produces an improvement in the endothelial dysfunction shown by aortae from STZ-induced diabetic rats without lowering plasma cholesterol, an effect of pravastatin that may be due to decreased LDL oxidation (Kobayashi et al., 2000). In the present study, we found that chronic administration of J-104132 for 4 weeks had no effect on plasma glucose, total cholesterol, VLDL cholesterol, LDL cholesterol or triglyceride, suggesting that the improvement effect we observed with J-104132 is not related to a lowering of plasma VLDL, LDL or glucose. We recently reported that following incubation of aortae from control rats with LDL isolated from diabetic rats, endothelium-dependent relaxation was impaired and that this inhibitory effect was prevented by superoxide dismutase, a scavenger of superoxide anions (Kobayashi et al., 2000). Furthermore, we have also reported that the basal O2− level is greater in aortic rings from diabetic rats than in those from the controls (Kobayashi & Kamata, 2001). These results strongly suggest that the increased LDL in diabetic rats may cause a greater production of O2−. As mentioned above, the expression of the p22phox mRNA for the NADH/NADPH oxidase subunit was significantly increased in STZ-induced diabetic rats and this increase was completely prevented by chronic administration of J-104132. We are left with the conclusion that J-104132 improves the impaired endothelium-dependent relaxation seen in the aorta of the diabetic rat by a mechanism that involves a lowering of O2−, but this is not secondary to a lowering of the plasma LDL level. This implies that the elevated plasma LDL level seen in diabetes is not the only causal factor responsible for endothelial dysfunction in the aorta.
In conclusion, we found that chronic administration of the endothelin antagonist J-104132 leads to an improvement in the endothelial dysfunction seen in the aorta in rats with established STZ-induced diabetes. This effect of J-104132 may be due to a decrease in the aortic superoxide anion via an inhibitory effect of this agent on the induction of NADPH oxidase.
Acknowledgments
This work was supported in part by the Ministry of Education, Science, Sports, and Culture, Japan.
Abbreviations
- ACh
acetylcholine
- eNOS
endothelial nitric oxide synthase
- ET-1
endothelin-1
- ETA/ETB
endothelin A/endothelin B receptor
- GAPDH
glyceraldehydes-3-phosphate dehydrogenase
- HDL
high-density lipoprotein
- KHS
Krebs – Henseleit solution
- LDL
low-density lipoprotein
- NA
noradrenaline
- NADH/NADPH
nicotinamide adenine dinucleotide/nicotinamide dinucleotide phosphate
- NBT
nitro blue tetrazolium
- O2−
superoxide anion
- RT – PCR
reverse transcription polymerase chain reaction
- SDS
sodium dodecyl sulphate
- SNP
sodium nitroprusside
- SOD
superoxide dismutase
- STZ
streptozotocin
- VLDL
very low-density lipoprotein
References
- ADKINS W.K., TAYLOR A.E. Role of xanthine oxidase and neutrophils in ischemia-reperfusion injury in rabbit lung. J. Appl. Physiol. 1990;69:2012–2018. doi: 10.1152/jappl.1990.69.6.2012. [DOI] [PubMed] [Google Scholar]
- BAYRAKTUTAN U., DRAPER N., LANG D., SHAH A.M. Expression of functional neutrophil-type NADPH oxidase in cultured rat coronary microvascular endothelial cells. Cardiovasc. Res. 1998;38:256–262. doi: 10.1016/s0008-6363(98)00003-0. [DOI] [PubMed] [Google Scholar]
- BYSANI G.K. , KENNEDY T.P., KY N, , RAO N.V., BLAZE C.A., HOIDAL J.R. Role of cytochrome P-450 in reperfusion injury of the rabbit lung. J. Clin. Invest. 1990;86:1434–1441. doi: 10.1172/JCI114859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CHOMCZYNSKI P., SACCHI N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-choloroform extraction. Anal. Biochem. 1987;162:156–159. doi: 10.1006/abio.1987.9999. [DOI] [PubMed] [Google Scholar]
- DE VRIESE A.S., VERBEUREN T.J., VAN DE VOORDE J., LAMEIRE N.H., VANHOUTTE P.M. Endothelial dysfunction in diabetes. Br. J. Pharmacol. 2000;130:963–974. doi: 10.1038/sj.bjp.0703393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DUERRSCHMIDT N., WIPPICH N., GOETTSH W., BROEMME H.J., MORAWIETZ H. Endothelin-1 induces NAD(P)H oxidase in human endothelial cells. Biochem. Biophys. Res. Commun. 2000;269:713–717. doi: 10.1006/bbrc.2000.2354. [DOI] [PubMed] [Google Scholar]
- GORLACH A., BRANDES R.P., NGUYEN K., AMIDI M., DEHGHANI F., BUSSE R. A gp91phox containing NADPH oxidase selectively expressed in endothelial cells is a major source of oxygen radical generation in the arterial wall. Circ. Res. 2000;87:26–32. doi: 10.1161/01.res.87.1.26. [DOI] [PubMed] [Google Scholar]
- GOTO K., HAMA H., KASUYA Y. Molecular pharmacology and pathophysiological significance of endothelin. Jpn. J. Pharmacol. 1996;72:261–290. doi: 10.1254/jjp.72.261. [DOI] [PubMed] [Google Scholar]
- HAAK T., JUNGMANN E., FELBER A., HILLMANN U., USADEL K.H. Increased plasma levels of endothelin in diabetic patients with hypertension. Am. J. Hypertens. 1992;5:161–166. doi: 10.1093/ajh/5.3.161. [DOI] [PubMed] [Google Scholar]
- HATTORI Y., KAWASAKI H., ABE K., KANNO M. Superoxide dismutase recovers altered endothelium-dependent relaxation in diabetic rat aorta. Am. J. Physiol. 1991;261:H1086–H1094. doi: 10.1152/ajpheart.1991.261.4.H1086. [DOI] [PubMed] [Google Scholar]
- HAVEL R.J., EDER H.A., BRAGSON J.H. The distribution and chemical composition of ultracentrifugally separated lipoproteins in human serum. J. Clin. Invest. 1955;34:1345–1353. doi: 10.1172/JCI103182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HEYGATE K.M., LAWRENCE I.G., BENNETT M.A., THURSTON H. Impaired endothelium-dependent relaxation in isolated resistance arteries of spontaneously diabetic rats. Br. J. Pharmacol. 1995;116:3251–3259. doi: 10.1111/j.1476-5381.1995.tb15132.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HINK U., LI H., MOLLNAU H., OELZE M., MATHEIS E., HARTMANN M., SKATCHKOV M., THAISS F., STAHL R.A., WARNHOLTZ A., MEINERTZ T., GRIENDLING K., HARRISON D.G., FORSTERMANN U., MUNZEL T. Mechanisms underlying endothelial dysfunction in diabetes mellitus. Circ. Res. 2001;88:e14–e22. doi: 10.1161/01.res.88.2.e14. [DOI] [PubMed] [Google Scholar]
- IKEDA T., OHTA H., OKADA M., KAWAI N., NAKAO R., SIEGEL P.K.S., KOBAYASHI T., MAEDA S., MIYAUCHI T., NISHIKIBE M. Pathophysiological roles of endothelin-1 in Dahl salt-sensitive hypertension. Hypertension. 1999;34:514–519. doi: 10.1161/01.hyp.34.3.514. [DOI] [PubMed] [Google Scholar]
- JONES S.A., O'DONNELL V.B., WOOD J.D., BROUGHTON J.P., HUGHES E.J., JONES O.T. Expression of phagocyte NADPH oxidase components in human endothelial cells. Am. J. Physiol. 1996;271:H1626–H1634. doi: 10.1152/ajpheart.1996.271.4.H1626. [DOI] [PubMed] [Google Scholar]
- KAMATA K., KOBAYASHI T. Changes in superoxide dismutase mRNA expression by streptozotocin-induced diabetes. Br. J. Pharmacol. 1996;119:583–589. doi: 10.1111/j.1476-5381.1996.tb15712.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KAMATA K., MIYATA N., KASUYA Y. Impairment of endothelium-dependent relaxation and changes in levels of cyclic GMP in aorta from streptozotocin-induced diabetic rats. Br. J. Pharmacol. 1989;97:614–618. doi: 10.1111/j.1476-5381.1989.tb11993.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KAMATA K., SUGIURA M., KOJIMA S., KASUYA Y. Preservation of endothelium-dependent relaxation in cholesterol-fed and streptozotocin-induced diabetic mice by the chronic administration of cholestyramine. Br. J. Pharmacol. 1996;118:385–391. doi: 10.1111/j.1476-5381.1996.tb15414.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KOBAYASHI T., KAMATA K. Effect of insulin treatment on smooth muscle contractility and endothelium-dependent relaxation in rat aortae from established STZ-induced diabetes. Br. J. Pharmacol. 1999a;127:835–842. doi: 10.1038/sj.bjp.0702554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KOBAYASHI T., KAMATA K. Relationship among cholesterol, superoxide anion and endothelium-dependent relaxation in diabetic rats. Eur. J. Pharmacol. 1999b;367:213–222. doi: 10.1016/s0014-2999(98)00971-6. [DOI] [PubMed] [Google Scholar]
- KOBAYASHI T., KAMATA K. Effect of chronic insulin treatment on NO production and endothelium-dependent relaxation in aortae from established STZ-induced diabetic rats. Atherosclerosis. 2001;155:313–320. doi: 10.1016/s0021-9150(00)00583-9. [DOI] [PubMed] [Google Scholar]
- KOBAYASHI T., MATSUMOTO T., KAMATA K. Mechanisms underlying the chronic pravastatin treatment-induced improvement in the impaired endothelium-dependent aortic relaxation seen in streptozotocin-induced diabetic rats. Br. J. Pharmacol. 2000;131:231–238. doi: 10.1038/sj.bjp.0703572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LUND D.D., FARACI F.M., OOBOSHI H., DAVIDSON B.L., HEISTAD D.D. Adenovirus-mediated gene transfer is augmented in basilar and carotid arteries of heritable hyperlipidemic rabbits. Stroke. 1999;30:120–125. doi: 10.1161/01.str.30.1.120. [DOI] [PubMed] [Google Scholar]
- MAKINO A., KAMATA K. Elevated plasma endothelin-1 level in streptozotocin-induced diabetic rats and responsiveness of the mesenteric arterial bed to endothelin-1. Br. J. Pharmacol. 1998;123:1065–1072. doi: 10.1038/sj.bjp.0701704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MAKINO A., KAMATA K. Time-course changes in plasma endothelin-1 and its effects on the mesenteric arterial bed in streptozotocin-induced diabetic rats. Diabetes Obes. Metab. 2000;2:47–55. doi: 10.1046/j.1463-1326.2000.00024.x. [DOI] [PubMed] [Google Scholar]
- MAKINO A., ODA S., KAMATA K. Mechanisms underlying increased release of endothelin-1 from aorta in diabetic rats. Peptides. 2001;22:639–645. doi: 10.1016/s0196-9781(01)00374-6. [DOI] [PubMed] [Google Scholar]
- MARSHALL J.J., WEI E.P., KONTOS H.A. Independent blockade of cerebral vasodilation from acetylcholine and nitric oxide. Am. J. Physiol. 1988;255:H847–H854. doi: 10.1152/ajpheart.1988.255.4.H847. [DOI] [PubMed] [Google Scholar]
- MERAJI S., JAYAKODY L., SENARATNE M.P., THOMSON A.B., KAPPAGODA T. Endothelium-dependent relaxation in aorta of BB rat. Diabetes. 1987;36:978–981. doi: 10.2337/diab.36.8.978. [DOI] [PubMed] [Google Scholar]
- NISHIKIBE M., OHTA H., OKADA M., ISHIKAWA K., HAYAMA T., FUKURODA T., NOGUCHI K., SAITO M., KANOH T., OZAKI S., KAMEI T., HARA K., WILLIAM D., KIVLIGHN S., KRAUSE S., GABEL R., ZINGARO G., NOLAN N., O'BRIEN J., CLAYTON F., LYNCH J., PETTIBONE D., SIEGL P. Pharmacological properties of J-104132 (L-753,037), a potent, orally active, mixed ETA/ETB endothelin receptor antagonist. J. Pharmacol. Exp. Ther. 1999;289:1262–1270. [PubMed] [Google Scholar]
- OOBOSHI H., RIOS C.D., CHU Y., CHRISTENSEN S.D., FARACI F.M., DAVIDSON B.L., HEISTAD D.D. Augmented adenovirus-mediated gene transfer in atherosclerotic vessels. Arterioscler. Thromb. Vasc. Biol. 1997;17:1786–1792. doi: 10.1161/01.atv.17.9.1786. [DOI] [PubMed] [Google Scholar]
- OYAMA Y., KAWASAKI H., HATTORI Y., KANNO M. Attenuation of endothelium-dependent relaxation in aorta from diabetic rats. Eur. J. Pharmacol. 1986;132:75–78. doi: 10.1016/0014-2999(86)90013-0. [DOI] [PubMed] [Google Scholar]
- PAGANO P.J., GRISWOLD M.C., RAVEL D., COHEN R.A. Vascular action of the hypoglycaemic agent gliclazide in diabetic rabbits. Diabetologia. 1998;41:9–15. doi: 10.1007/s001250050860. [DOI] [PubMed] [Google Scholar]
- PATTERSON C., RUEF J., MADAMANCHI N.R., BARRY-LANE P., HU Z., HORAIST C., BALLINGER C.A., BRASIER A.R., BODE C., RUNGE M.S. Stimulation of a vascular smooth muscle cell NAD(P)H oxidase by thrombin. Evidence that p47(phox) may participate in forming this oxidase in vitro and in vivo. J. Biol. Chem. 1999;274:19814–19822. doi: 10.1074/jbc.274.28.19814. [DOI] [PubMed] [Google Scholar]
- PIEPER G.M. Review of alterations in endothelial nitric oxide production in diabetes: protective role of arginine on endothelial dysfunction. Hypertension. 1998;31:1047–1060. doi: 10.1161/01.hyp.31.5.1047. [DOI] [PubMed] [Google Scholar]
- PIEPER G.M., MEI D.A., LANGENSTROER P., O'ROURKE S.T. Bioassay of endothelium-derived relaxing factor in diabetic rat aorta. Am. J. Physiol. 1992;263:H676–H680. doi: 10.1152/ajpheart.1992.263.3.H676. [DOI] [PubMed] [Google Scholar]
- PIEPER G.M., SIEBENEICH W., ROZA A.M., JORDAN M., ADAMS M.B. Chronic treatment in vivo with dimethylthiourea, a hydroxyl radical scavenger, prevents diabetes-induced endothelial dysfunction. J. Cardiovasc. Pharmacol. 1996;28:741–745. doi: 10.1097/00005344-199612000-00002. [DOI] [PubMed] [Google Scholar]
- POSTON L., TAYLOR P.D. Glaxo/MRS Young Investigator Prize. Endothelium-mediated vascular function in insulin-dependent diabetes mellitus. Clin. Sci. 1995;88:245–255. doi: 10.1042/cs0880245. [DOI] [PubMed] [Google Scholar]
- RUBANYI G.M., VANHOUTTE P.M. Oxygen-derived free radicals, endothelium and responsiveness of vascular smooth muscle. Am. J. Physiol. 1986;250:H815–H821. doi: 10.1152/ajpheart.1986.250.5.H815. [DOI] [PubMed] [Google Scholar]
- TAKAHASHI K., GHATEI M.A., LAM H.C., O'HALLORAN D.J., BLOOM S.R. Elevated plasma endothelin in patient with diabetes mellitus. Diabetologia. 1990;33:306–310. doi: 10.1007/BF00403325. [DOI] [PubMed] [Google Scholar]
- TSAI M.Y., YUAN J., HUNNINGHAKE D.B. Effect of gemfibrozil on composition of lipoproteins and distribution of LDL subspecies. Atherosclerosis. 1992;95:35–42. doi: 10.1016/0021-9150(92)90173-e. [DOI] [PubMed] [Google Scholar]
- TSUNODA K., ABE K., SATO T., YOKOSAWA S., YOSHINAGA K. Decreased conversion of big endothelin-1 to endothelin-1 in patients with diabetes mellitus. Clin. Exp. Pharmacol. Physiol. 1991;18:731–732. doi: 10.1111/j.1440-1681.1991.tb01388.x. [DOI] [PubMed] [Google Scholar]
- USHIO-FUKAI M., ZAFARI A.M., FUKUI T., ISHIZAKA N., GRIENDLING K.K. p22phox is a critical component of the superoxide-generating NADH/NADPH oxidase system and regulates angiotensin II-induced hypertrophy in vascular smooth muscle cells. J. Biol. Chem. 1996;271:23317–23321. doi: 10.1074/jbc.271.38.23317. [DOI] [PubMed] [Google Scholar]
- WANG H.D., PAGANO P.J., DU Y., CAYATTE A.J., QUINN M.T., BRECHER P., COHEN R.A. Superoxide anion from the adventitia of the rat thoracic aorta inactivates nitric oxide. Circ. Res. 1998;82:810–818. doi: 10.1161/01.res.82.7.810. [DOI] [PubMed] [Google Scholar]
- ZALBA G., BEAUMONT F.J., SAN JOSE G.A., FORTUNO A., FORTUNO M.A., ETAYO J.C., DIEZ J. Vascular NADH/NADPH oxidase is involved in enhanced superoxide production in spontaneously hypertensive rats. Hypertension. 2000;35:1055–1061. doi: 10.1161/01.hyp.35.5.1055. [DOI] [PubMed] [Google Scholar]
- ZONTA F., BARBLERI A., REGUZZONI M., CALLIGARO A. Quantitative changes in pharmacodynamic parameters of noradrenaline in different rat aorta preparations: influence of endogenous EDRF. J. Autono. Pharmacol. 1998;18:129–138. doi: 10.1046/j.1365-2680.1998.1830129.x. [DOI] [PubMed] [Google Scholar]


