Abstract
The present study aims to investigate whether phenotypic changes, reported to occur in cultured isolated airway smooth muscle (ASM) cells, are of relevance to intact ASM. Moreover, we aimed to gain insight into the signalling pathways involved.
Culturing of bovine tracheal smooth muscle (BTSM) strips for up to 8 days in the presence of 10% foetal bovine serum caused a time-dependent (t1/2=2.8 days) decrease in maximal contraction (Emax) to methacholine compared to serum-deprived controls (Emax=74±4% at day 8). A reduced Emax was also found using insulin-like growth factor-1 (30 ng ml−1) and platelet-derived growth factor (30 ng ml−1), but not using epidermal growth factor (10 ng ml−1) (Emax=83±3, 67±8, 100±4%, respectively). Similar serum and growth factor-induced changes in Emax were found for KCl-induced contraction (65±9, 80±7, 64±11% and 107±2%, respectively).
Strong correlations were found between the growth factor-induced reductions in Emax and their proliferative responses, assessed by [3H]-thymidine-incorporation, in BTSM cells. (r=0.97, P=0.002 for methacholine and r=0.93, P=0.007 for KCl).
The PDGF-induced reduction in Emax was inhibited completely by combined treatment with either PD 98059 (30 μM) or LY 294002 (10 μM).
These results indicate that serum and growth factors may cause a functional shift towards a less contractile phenotype in intact BTSM, which is associated with their proliferative response and dependent on signalling pathways involving the mitogen-activated protein kinase pathway and the phosphatidylinositol-3-kinase pathway.
Keywords: Airway smooth muscle; contractility; phenotype; bovine; platelet-derived growth factor; insulin-like growth factor-1; epidermal growth factor; foetal bovine serum; phosphatidylinositol-3-kinase, extracellular signal regulated kinase 1/2
Introduction
Studies using primary cultures of airway smooth muscle (ASM) cells have revealed that airway myocytes develop an immature phenotype, also referred to as a synthetic phenotype, upon culturing in serum-rich culture media (Halayko et al., 1996; 1999). Characteristics of this synthetic phenotype include an increased proliferative and synthetic potency (Johnson & Knox, 1997), but a decreased contractile responsiveness, due to a decreased expression of contractile proteins and cell surface receptors, such as the muscarinic M3-receptor (Halayko et al., 1996; Halayko & Solway, 2001; Mitchell et al., 2000; Stephens et al., 1998). Changes toward the synthetic phenotype are, however, reversible: long-term treatment with serum-deprived media re-induces a contractile phenotype in canine ASM cells, characterized by its elongated morphology and re-expression of contractile proteins and muscarinic M3-receptors (Halayko & Solway, 2001; Mitchell et al., 2000). Moreover, serum deprivation-induced transition towards a hypercontractile phenotype has been reported in canine ASM cells (Ma et al., 1998).
Isolated ASM preparations obtained from asthmatics may respond with an increased maximal contractility and/or sensitivity to contractile agonists (de jongste et al., 1987; Stephens et al., 1998). Also, passively sensitized human airway smooth muscle exhibits an increased contractile responsiveness (Schmidt et al., 2000) and an increased myosin light chain kinase (MLCK) content (Ammit et al., 2000). Similar results have been obtained using preparations from ragweed sensitized dogs (Antonissen et al., 1979; Jiang et al., 1992; Stephens et al., 1998). Although there is no evidence linking allergic sensitization or asthma-induced changes in contractility to phenotypic changes, it has been postulated that switching towards a synthetic airway smooth muscle phenotype may be involved in airway remodelling in patients with chronic asthma, characterized by increased smooth muscle mass and irreversible airflow obstruction (Hirst, 1996). Transition from the synthetic to the (hyper)-contractile phenotype could be involved in enhanced airway hyperresponsiveness in these patients (Hirst et al., 2000b).
The mechanisms involved in smooth muscle phenotypic changes are poorly understood. Although multiple signalling pathways have been associated with phenotypic changes, the exact roles of these signalling pathways are not yet clear. The mitogen-activated protein kinase pathway, also referred to as extracellular signal regulated kinase pathway (MAPK/ERK pathway) has been associated with smooth muscle cell de-differentiation towards the synthetic phenotype (Roy et al., 2001; Hayashi et al., 1999). Less clear is the role of phosphatidylinositol-3-kinase (PI-3 kinase) activity in this process, because both maintenance of the contractile phenotype (Hayashi et al., 1998) and transition towards a synthetic phenotype (Ramos et al., 1996; Reusch et al., 2001) have been associated with PI-3 kinase activity. Also, activation of RhoA has been shown to be involved in the upregulation of smooth muscle specific gene expression through Rho-kinase-dependent translocation of serum response factor to the nucleus (Camoretti-Mercado et al., 2000) and through RhoA-mediated actin polymerization (Mack et al., 2001).
A role for extracellular matrix components has also been implicated in regulating airway smooth muscle phenotype, showing that fibronectin and collagen type I favoured progression towards a synthetic phenotype, whereas laminin and matrigel (a solubilized basement membrane matrix) strongly inhibited the progression towards a synthetic phenotype (Hirst et al., 2000a). Knowing that cell to cell contacts and extracellular matrix components are preserved in intact smooth muscle, we were interested to learn about the occurrence of functional changes in intact bovine tracheal smooth muscle (BTSM). We evaluated the contractile properties of BTSM strips exposed to serum and to different growth factors (platelet-derived growth factor (PDGF), insulin-like growth factor-1 (IGF-1) and epidermal growth facor (EGF)) in relation to their mitogenic potencies. In addition, we investigated the role of PI-3 kinase and ERK1/2 signalling pathways in the phenotypic changes in contractility in response to stimulation with PDGF.
Methods
Tissue preparation and organ culture procedure
Bovine tracheae were obtained from local slaughterhouses and rapidly transported to the laboratory in Krebs-Henseleit (KH) buffer of the following composition (mM): NaCl 117.5, KCl 5.60, MgSO4 1.18, CaCl2 2.50, NaH2PO4 1.28, NaHCO3 25.00 and glucose 5.50, pregassed with 5% CO2 and 95% O2; pH 7.4. After dissection of the smooth muscle layer and careful removal of mucosa and connective tissue, tracheal smooth muscle strips were prepared while incubated in gassed KH-buffer at room temperature. Care was taken to cut tissue strips with macroscopically identical length (1 cm) and width (2 mm). Tissue strips were washed once in sterile Dulbecco's modification of Eagle's medium (DMEM), supplemented with NaHCO3 (7 mM), HEPES (10 mM), sodium pyruvate (1 mM), nonessential amino acid mixture (1 : 100), gentamicin (45 μg ml−1), penicillin (100 u ml−1), streptomycin (100 μg ml−1) and amphotericin B (1.5 μg ml−1). Next, tissue strips were transferred into suspension culture flasks and a volume of 7.5 ml medium, with or without 10% foetal bovine serum (FBS), was added per tissue strip. Strips were maintained in culture in an incubator shaker (37°C, 55 r.p.m.) for a maximum of 8 days, refreshing the medium on day 4. When applied, growth factors (PDGF, IGF-1, EGF) or kinase inhibitors (PD 98059, LY 294002, given 30 min prior to the growth factors) were added in a small volume (7.5 μl per tissue strip). Culture flasks containing kinase inhibitors were protected from light during the whole experiment. Occasionally, some strips were used for contraction experiments immediately after preparation.
Isometric tension measurements
Tissue strips, collected from suspension culture flasks, were washed with several volumes of KH-buffer pregassed with 5% CO2 and 95% O2, pH 7.4 at 37°C. Subsequently, strips were mounted for isometric recording (Grass force-displacement transducer FT03) in 20 ml water-jacked organ baths, containing KH-buffer at 37°C, continuously gassed with 5% CO2 and 95% O2, pH 7.4. During a 90 min equilibration period, with washouts every 30 min, resting tension was graduallly adjusted to 3 g. Subsequently, muscle strips were precontracted with 20 and 30 mM isotonic KCl solutions. Following two wash-outs, basal smooth muscle tone was established by the addition of 0.1 μM isoprenaline and tension was re-adjusted to 3 g, immediately followed by two changes of fresh KH-buffer. After another equilibration period of 30 min cumulative concentration response curves (CRCs) were constructed to stepwise increasing concentrations of isotonic KCl (5.6–50 mM) or methacholine (1 nM–100 μM). When maximal KCl or methacholine-induced tension was obtained, the strips were washed several times and basal tone was established using isoprenaline (10 μM).
Isolation of bovine tracheal smooth muscle cells
After the removal of mucosa and connective tissue, tracheal smooth muscle was chopped using a McIlwain tissue chopper, three times at a setting of 500 μm and three times at a setting of 100 μm. Tissue particles were washed two times with the medium mentioned above, supplemented with 0.5% FBS. Enzymatic digestion was performed in the same medium, supplemented with collagenase P (0.75 mg ml−1), papain (1 mg ml−1) and soybean trypsin inhibitor (1 mg ml−1). During digestion, the suspension was incubated in an incubator shaker (Innova 4000) at 37°C, 55 r.p.m. for 20 min, followed by a 10 min period of shaking at 70 r.p.m. After filtration of the obtained suspension over 50 μm gauze, cells were washed three times in medium supplemented with 10% FBS.
[3H]-thymidine-incorporation
BTSM cells were plated in 24-well cluster plates at a density of 50,000 cells per well directly after isolation and were allowed to attach overnight in 10% FBS containing medium. Cells were washed twice with sterile phosphate buffered saline (PBS, composition (mM): NaCl 140.0; KCl 2.6; KH2PO4 1.4; Na2HPO4.2H2O 8.1; pH 7.4) and made quiescent by incubation in FBS-free medium, supplemented with apo-transferrin (5 μg ml−1), ascorbate (100 μM) and insulin (1 μM) for 72 h. Cells were then washed with PBS and stimulated with mitogens in FBS- and insulin-free medium for 28 h, the last 24 h in the presence of [3H]-thymidine (0.25 μCi ml−1). After incubation the cells were washed twice with PBS at room temperature and once with ice-cold 5% trichloroacetic acid (TCA). Cells were treated with this TCA-solution on ice for 30 min and subsequently the acid-insoluble fraction was dissolved in 1 ml NaOH (1 M). Incorporated [3H]-thymidine was quantified by liquid-scintillation counting using a Beckman LS1701 β-counter.
Data analysis
All data represent means±s.e.mean from n separate experiments. The statistical significance of differences between data was determined by the Student's t-test for paired observations or one-way ANOVA, where appropiate. Differences were considered to be statistically significant when P<0.05.
Materials
Dulbecco's modification of Eagle's Medium (DMEM) and methacholine hydrochloride were obtained from ICN Biomedicals (Costa Mesa, CA, U.S.A.). Foetal bovine serum, NaHCO3 solution (7.5%), HEPES solution (1 M), sodium pyruvate solution (100 mM), non-essential amino acid mixture, gentamycin solution (10 mg ml−1), penicillin/streptomycin solution (5000 u ml−1/5000 μg ml−1) and amphotericin B solution (250 μg ml−1) (Fungizone) were obtained from Gibco BRL Life Technologies (Paisley, U.K.). Platelet-derived growth factor AB (PDGF-AB, human recombinant), epidermal growth factor (EGF, human recombinant), insulin-like growth factor 1 (IGF-1, human recombinant), insulin (from bovine pancreas), apotransferrin (human), soybean trypsin inhibitor and (−)isoprenaline hydrochloride were from Sigma Chemical Co. (St. Louis, MO, U.S.A.). PD 98059 and LY 294002 were obtained from Tocris Cookson Ltd. (Bristol, U.K.). L(+)ascorbic acid was from Merck (Darmstadt, Germany). [methyl-3H]-thymidine (specific activity 25 Ci mmol−1) was obtained from Amersham (Buckinghamshire, U.K.) Papain and Collagenase P were from Boehringer (Mannheim, Germany). All other chemicals were of analytical grade.
Results
Effects of treatment with FBS-enriched or FBS-free media on methacholine-induced contraction
Maximal methacholine-induced contractile force (Emax) of BTSM strips cultured for up to 8 days in the presence of 10% FBS was significantly lower compared to strips cultured in FBS-free medium (Emax=17.2±0.8 and 22.2±2.2 g, respectively; P<0.05; Figure 1). No difference in the sensitivity to methacholine was found (pEC50=6.99±0.10 and 6.99±0.05 for FBS-treated and FBS-deprived BTSM strips, respectively).
Figure 1.
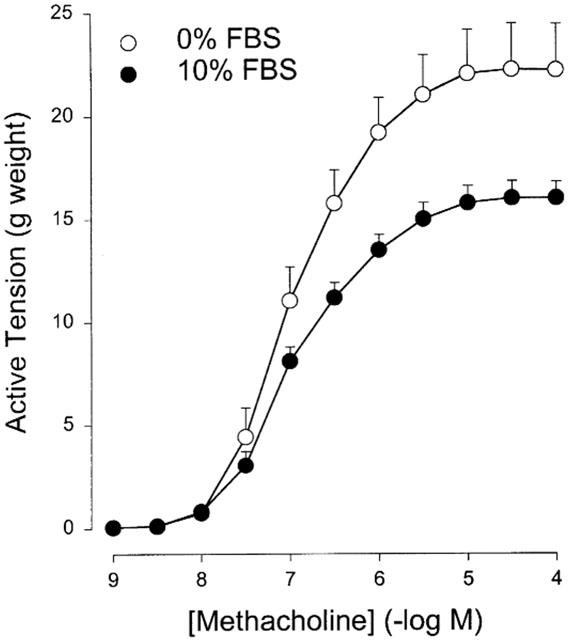
Methacholine-induced contraction of BTSM strips, after exposure to 10% FBS-enriched or FBS-free culture media for 8 days. Data represent the means±s.e.mean from six experiments.
The development of the differences in maximal methacholine-induced contraction between BTSM strips cultured in FBS-enriched and in FBS-free media was time-dependent (Figure 2a). The difference in Emax between FBS-treated and FBS-free conditions was maximal at day 8, with a half-maximal effect after 2.8 days (Figure 2b). Comparison with results obtained with freshly used strips shows that the Emax of strips maintained in FBS-free and FBS-enriched media diverged in time (Figure 2a).
Figure 2.
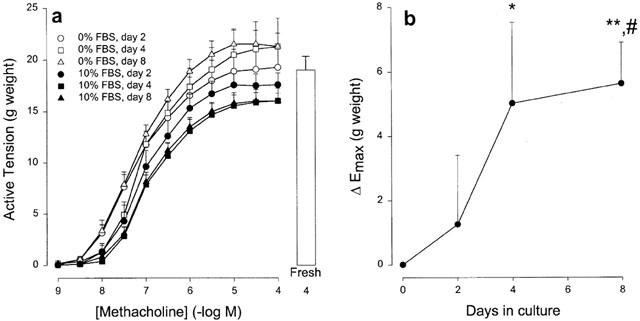
(a) Cumulative concentration response curves of methacholine-induced contraction of BTSM strips, cultured in FBS-enriched (10% FBS) or FBS-free (0% FBS) medium for the different periods of time indicated. Data represent the means±s.e.mean from six experiments. For comparison, Emax to methacholine (100 μM) in freshly used BTSM strips is included, depicted by the white bar. (b) Time-dependent differences in maximal methacholine-induced contraction (ΔEmax) between FBS-treated and FBS-deprived BTSM strips. Data are taken from (a) and represent means±s.e.mean from six experiments. *P<0.05, **P<0.001 compared to FBS-deprived strips, #P<0.05 compared to day 2 in FBS-treated strips (Student's t-test for paired observations).
Effects of treatment with growth factors on methacholine-induced contraction
The ability to alter contractile behaviour was not limited to FBS treatment. Treatment for 8 days with IGF-1 (30 ng ml−1) and PDGF (30 ng ml−1) were also effective in inducing a statistically significant depression of the Emax for methacholine-induced contraction compared to vehicle-treated BTSM strips (Figure 3). No effect was observed for EGF (10 ng ml−1), however. The PDGF-induced decrease in Emax was concentration-dependent (Table 1). As with FBS-treated BTSM strips, the pEC50 of methacholine-induced contraction was unaltered upon treatment with the above mentioned growth factors (Table 1).
Figure 3.
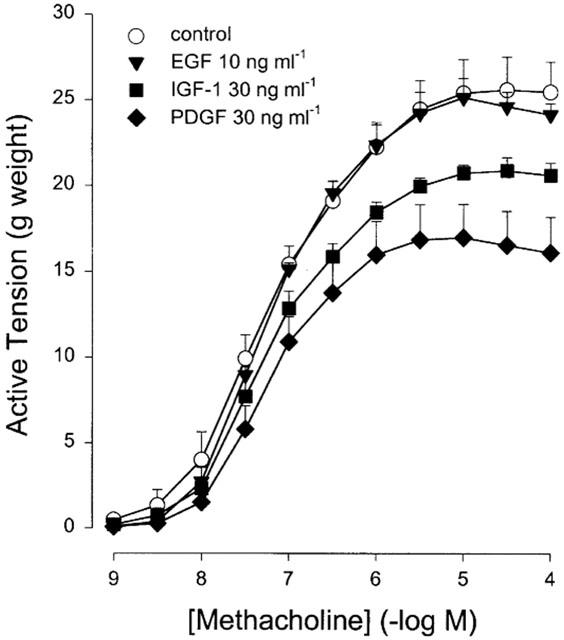
Methacholine-induced contraction of BTSM strips, cultured for 8 days in FBS-free medium containing EGF (10 ng ml−1), IGF-1 (30 ng ml−1), or PDGF (30 ng ml−1). Vehicle-treated strips served as controls. Data represent means±s.e.mean from five experiments.
Table 1.
Contractile properties to methacholine and KCl of BTSM strips, cultured for 8 days in the presence of growth factors and the proliferative responses of these growth factors in BTSM cells
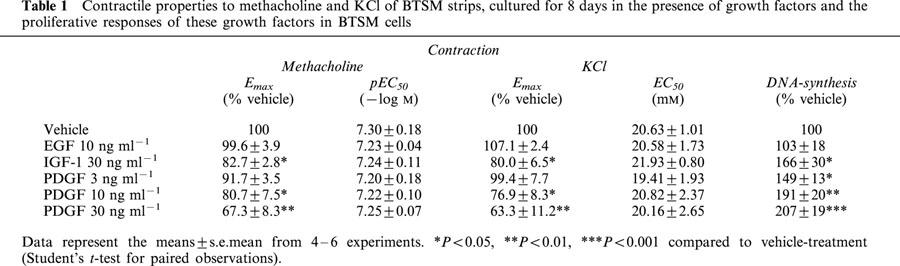
Effects on KCl-induced contraction
To establish whether the growth factor-induced effects on BTSM contractility were limited to methacholine-induced contraction, contraction to KCl was also determined in BTSM strips pre-treated with growth factors for 8 days. As observed for methacholine, maximal KCl-induced contraction was sensitive to pretreatment with IGF-1 (30 ng ml−1) and PDGF (30 ng ml−1), but not EGF (10 ng ml−1). Moreover, changes in maximal KCl-induced contraction depended on the concentration of PDGF used. The potency (EC50) of KCl was unchanged after treatment with growth-factors, irrespective of the growth factor applied. Interestingly, changes in KCl and methacholine-induced contraction were similar, both qualitatively and quantitatively (Table 1).
Relationship between growth factor-induced proliferation and inhibition of maximal contraction
As indicated in Figure 3 and Table 1, the effects of growth factors on maximal contraction of BTSM strips to methacholine or KCl depended on the nature and the concentration of the growth factor used. The proliferative responses of isolated BTSM cells, measured as [3H]-thymidine incorporation, were also dependent on the nature and concentration of the growth factors under investigation, with a reciprocal rank order of potency (Table 1). A strong correlation was observed between the efficacy of growth factors to induce depression of maximal methacholine or KCl-induced contraction of BTSM strips and the proliferative potency of these factors in BTSM cells (r=0.97, P=0.002 and r=0.93, P=0.007 for methacholine and KCl-induced contraction, respectively; Figure 4a, b)
Figure 4.

Relationships between depression of maximal methacholine (a) and KCl (b) induced contraction of BTSM strips and proliferative responses of BTSM cells induced by treatment with vehicle (1), EGF 10 ng ml−1 (2), PDGF 3 ng ml−1 (3), IGF-1 30 ng ml−1 (4), PDGF 10 ng ml−1 (5) or PDGF 30 ng ml−1 (6). Data represent means from 4–6 experiments. Significance level was obtained using one-way analysis of variance (ANOVA).
Effects of kinase inhibitors on PDGF-induced depression of maximal methacholine-evoked contraction
Since the growth factor-induced depression of BTSM contraction depended on the proliferative effect of the growth factors used, we explored the involvement of proliferation-associated signalling pathways in growth factor-induced depression of maximal contractility to methacholine. For this purpose, PD 98059 (30 μM), an inhibitor of the MAPK/ERK pathway or LY 294002 (10 μM), an inhibitor of PI-3 kinase were added to the culture medium 30 min before the addition of PDGF (10 ng ml−1). In agreement with the above observations, PDGF alone reduced the maximum methacholine-induced contraction (Figure 5a). However, in the presence of PD 98059, no measurable change in contractility was induced by PDGF (Figure 5b). PD 98059 by itself did not significantly affect maximal methacholine-induced contraction or sensitivity (Emax=20.5±3.0 and 22.1±2.1 g and pEC50=7.29±0.08 and 7.15±0.10 for PD 98059 and vehicle treated strips, respectively). Also in the presence of LY 294002, PDGF did not induce a measurable change in methacholine-evoked contraction (Figure 5c). However, pretreatment with LY 294002 by itself induced a significant depression of maximum contraction compared to vehicle-treated strips (Emax=18.9±2.0 and 22.1±2.1 g for LY294002 and vehicle treated strips, respectively, P<0.05). The potency of methacholine-induced contraction remained unaltered (pEC50=7.29±0.07 and 7.15±0.10 for LY294002 and vehicle treated strips, respectively).
Figure 5.
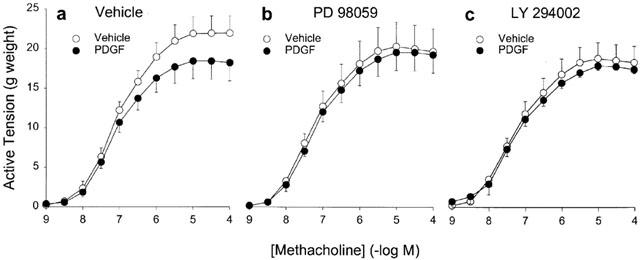
Methacholine-induced contraction of BTSM strips, cultured for 8 days in FBS-free medium. Strips were treated with vehicle (a), 30 μM PD 98059 (b) or 10 μM LY 294002 (c) in the absence or presence of PDGF 10 ng ml−1. Data represent means±s.e.mean from 5–6 simultaneously performed experiments.
Discussion
FBS treatment-induced decrease of contractile function has been demonstrated recently in organ-cultured vascular tissue. In cultured rat tail arterial smooth muscle, contractile responses to both noradrenaline and 60 mM K+ were diminished upon FBS-treatment for 4 days (Lindqvist et al., 1997). A similar effect has also been observed in guinea-pig ileum smooth muscle, which showed a decreased contractility to carbachol and 60 mM K+ after treatment with FBS-rich medium (Gomez & Sward, 1997). This loss of contractile function was explained by a continuously elevated intracellular [Ca2+]i upon culturing in FBS, thus leading to a decreased function of voltage-operated calcium channels (VOC) and/or a decreased sensitivity of the contractile apparatus for Ca2+. Effects of long-term exposure to FBS on contraction of organ cultured airway smooth muscle have not yet been described. FBS contains a variety of mitogenic stimuli and it causes contraction of canine ASM (Abdullah et al., 1994). Therefore, long-term effects of FBS are not necessarily the consequence of proliferative stimulation, but may also be caused by continuously elevated [Ca2+]i levels or stimulation of the contractile apparatus.
Our data show that pretreatment with both 10% FBS and the growth factors PDGF and IGF-1, followed by a washout period of over 120 min, induce a depression in maximal response to methacholine. The observed effects for 10% FBS are time-dependent. The diverging change of Emax in time (Figure 2) implies that the differences in Emax on day 8 are the consequence of both deprivation-induced increases in Emax and FBS-induced decreases in Emax. It is crucial for the interpretation of our results, that KCl-induced contraction and methacholine-induced contraction were influenced similarly by long-term exposure to growth factors, both quantitatively and qualitatively. These results imply that KCl and methacholine-induced contraction were affected by a common mechanism. However, methacholine induces contraction through stimulation of PI-turnover and subsequent release of Ca2+ from intracellular stores and influx through receptor operated Ca2+-channels (Zaagsma et al., 1997). In contrast, KCl-induced contraction is fully dependent on VOC mediated Ca2+-influx and does not require receptor mediated stimulation of PI-turnover. Therefore, quantitative similarities between KCl and methacholine-induced contraction can be achieved only by affecting contraction downstream intracellular Ca2+-increases. Further evidence that changes in [Ca2+]i or in the sensitivity of the contractile apparatus to Ca2+ cannot explain the 10% FBS or growth factor induced effects, comes from the observation that the potency (pEC50) of methacholine is not affected by culturing in the presence of 10% FBS or growth factors. Methacholine requires only fractional stimulation of PI-turnover and subsequent elevations of [Ca2+]i for BTSM contraction (Hoiting et al., 1996; Meurs et al., 1988). Altering events upstream of changes in [Ca2+]i or in the sensitivity of the contractile apparatus for Ca2+, should therefore be accompanied by a shift in the concentration-response curve to methacholine.
Modulation towards a less contractile phenotype is associated with a decrease in contractile protein expression (Mitchell et al., 2000), although changes in cytoskeletal organization should also be considered as an explanation for the altered maximal contraction (Halayko & Solway, 2001). However, agonist-induced cytoskeletal reorganization occurs rapidly (within 5 min) in human ASM cells (Togashi et al., 1998). Considering the long-term nature of the 10% FBS-induced decline in maximal contractile response, growth-related changes towards a less contractile phenotype become the most likely explanation for the observed effects. Western analysis of proteins harvested from canine ASM cells in primary culture shows a decline in the expression of contractile proteins (e.g. smooth muscle myosin heavy chain, smooth muscle α-actin) in the period prior to proliferation (Halayko et al., 1996). We therefore hypothesized that the degree of decline in contractile response should be reciprocally related to the degree by which proliferation is stimulated as a consequence of phenotypic modulation. Indeed, a strong correlation exists between the proliferative responses of the applied growth factors on BTSM cells and the degree of depression in contractile response, both for KCl and methacholine. Since our proliferation data are consistent with earlier findings in BTSM cells (Kelleher et al., 1995), a similar relationship could be constructed using our contraction data and the proliferative data published by Kelleher et al. (1995) (r=0.98 and r=0.97 for methacholine and KCl, respectively). The relative inactivity of EGF, applied in its maximally mitogenic concentration in BTSM cells, is in agreement with the low proliferative efficacy as obtained by us and others (Kelleher et al., 1995).
The MAPK/ERK pathway and the PI 3-kinase pathway were examined in order to gain insight in the mechanisms involved in the growth factor-induced change in phenotype. The inhibition of the MAPK/ERK-pathway using PD 98059 inhibited the development of contractile depression at a concentration that has been shown to fully inhibit PDGF-induced proliferation in BTSM cells (Karpova et al., 1997). This supports the hypothesis, that the development of a decreased contractile function is the consequence of proliferation-associated phenotypic modulation to a less contractile state. Interestingly, modulation of arterial smooth muscle cells towards a synthetic phenotype induced by FBS could also be inhibited by PD 98059, though only partially (Roy et al., 2001).
Inhibition of the PI 3-kinase pathway by LY 294002, applied during organ culture at a concentration known to reduce PDGF-mediated proliferation of BTSM cells to basal levels (Walker et al., 1998), inhibited the PDGF induced decrease of contractile response, suggestive for inhibition of modulation to a less contractile phenotype. In the absence of PDGF, LY 294002 treatment itself lowered maximal contraction to methacholine. This effect might be attributed to a diminished PI-3-kinase mediated maintenance of the cytoskeleton (Aplin et al., 1998; Rodriguez-Viciana et al., 1997). In addition, the effects of PI-3-kinase inhibition may also suggest an increase in apoptosis. This would imply apoptosis to be a significant regulatory mechanism in the intact BTSM strip. However, growth factors inhibit apoptosis, and this would induce enhancement rather than a depression of contractility. Although we do not know the exact nature of the LY 294002-induced effect, the inhibitory effect on PDGF-induced contractile depression is in agreement with the concept that proliferation and modulation to a less contractile state are stimulated simultaneously. Also, it implies a role for PI 3-kinase in functional modulation of BTSM.
Activation of the MAPK/ERK cascade is achieved via the PI 3-kinase pathway, but a redundant pathway stimulates MAPK/ERK independent of PI 3-kinase when a large number of receptors are activated (Duckworth & Cantley, 1997). Since LY 294002 and PD 98059 both totally inhibited the PDGF-induced change in phenotype, one might conclude that the pathway stimulated through PI-3-kinase, rather than the redundant MAPK/ERK-pathway, is responsible for the observed effects in phenotypic switching. Separate, parallel functioning pathways opposed to serially functioning pathways can, however, not be excluded: proliferation of BTSM cells induced by high concentrations of PDGF-BB has been reported to be inhibited completely by either PD 98059 or LY 294002, whereas PD 98059 does not inhibit PDGF-stimulated PI 3-kinase activity, nor does LY 294002 inhibit PDGF-stimulated MAPK activity (Walker et al., 1998).
Changes in phenotype may be relevant in asthma. Growth factors can be released in asthmatic airways from epithelial cells, inflammatory cells and ASM cells, or leaked into the airways as a consequence of extravasation, contributing to the formation of phenotypically altered myocytes with increased proliferative and synthetic capabilities. When extravasation ceases and growth factor concentrations return to basal levels redifferentiation to the contractile phenotype or even to a hypercontractile phenotype may occur as has been shown in cultured canine ASM cells (Halayko et al., 1999). Induction of synthetic smooth muscle phenotypes as postulated to occur in asthma (Hirst, 1996) is therefore not favourable and may contribute to typical features of asthmatic airways, such as hyperreactivity, irreversible airflow obstruction and the progressive increase in the severity of the disease.
In conclusion, BTSM contractility is sensitive to treatment with FBS or growth factors in vitro. Both receptor-dependent (methacholine) and receptor-independent (KCl) stimulation of BTSM, treated with FBS or growth factors, led to a decrease in maximal contraction to these agonists. This implies that changes occurred at the level of the contractile machinery. Moreover, the decline in contractile response correlates with the proliferative conditions of the culture medium and can be totally inhibited by PD 98059 and LY 294002. As a consequence, it seems that intact BTSM is sensitive to phenotypic changes and that these changes are regulated through signalling pathways involving both the MAPK/ERK and the PI-3 kinase-pathway.
Acknowledgments
This work was supported by the Netherlands Asthma Foundation (NAF grant 99.53).
Abbreviations
- BTSM
bovine tracheal smooth muscle
- DMEM
Dulbecco's modification of Eagle's medium
- EGF
epidermal growth factor
- ERK
extracellular signal regulated kinase
- FBS
foetal bovine serum
- IGF-1
insulin-like growth factor-1
- KH
Krebs-Henseleit
- MAPK
mitogen-activated protein kinase
- MLCK
myosin light chain kinase
- PBS
phosphate buffered saline
- PDGF
platelet-derived growth factor
- PI-3 kinase
phosphatidylinositol-3 kinase
- PI-turnover
phosphoinositide turnover
- VOC
voltage operated calcium channel
References
- ABDULLAH N.A., HIRATA M., MATSUMOTO H., AIZAWA H., INOUE R., HAMANO S., IKEDA S., XIE Z., HARA N., ITO Y. Contraction and depolarization induced by fetal bovine serum in airway smooth muscle. Am. J. Physiol. 1994;266:L528–L535. doi: 10.1152/ajplung.1994.266.5.L528. [DOI] [PubMed] [Google Scholar]
- AMMIT A.J., ARMOUR C.L., BLACK J.L. Smooth-muscle myosin light-chain kinase content is increased in human sensitized airways. Am. J. Respir. Crit. Care Med. 2000;161:257–263. doi: 10.1164/ajrccm.161.1.9901005. [DOI] [PubMed] [Google Scholar]
- ANTONISSEN L.A., MITCHELL R.W., KROEGER E.A., KEPRON W., TSE K.S., STEPHENS N.L. Mechanical alterations of airway smooth muscle in a canine asthmatic model. J. Appl. Physiol. 1979;46:681–687. doi: 10.1152/jappl.1979.46.4.681. [DOI] [PubMed] [Google Scholar]
- APLIN A.E., HOWE A., ALAHARI S.K., JULIANO R.L. Signal transduction and signal modulation by cell adhesion receptors: the role of integrins, cadherins, immunoglobulin-cell adhesion molecules, and selectins. Pharmacol. Rev. 1998;50:197–263. [PubMed] [Google Scholar]
- CAMORETTI-MERCADO B., LIU H.W., HALAYKO A.J., FORSYTHE S.M., KYLE J.W., LI B., FU Y., MCCONVILLE J., KOGUT P., VIEIRA J.E., PATEL N.M., HERSHENSON M.B., FUCHS E., SINHA S., MIANO J.M., PARMACEK M.S., BURKHARDT J.K., SOLWAY J. Physiological control of smooth muscle-specific gene expression through regulated nuclear translocation of serum response factor. J. Biol. Chem. 2000;275:30387–30393. doi: 10.1074/jbc.M000840200. [DOI] [PubMed] [Google Scholar]
- DE JONGSTE J.C., MONS H., BONTA I.L., KERREBIJN K.F. In vitro responses of airways from an asthmatic patient. Eur. J. Respir. Dis. 1987;71:23–29. [PubMed] [Google Scholar]
- DUCKWORTH B.C., CANTLEY L.C. Conditional inhibition of the mitogen-activated protein kinase cascade by wortmannin. J. Biol. Chem. 1997;272:27665–27670. doi: 10.1074/jbc.272.44.27665. [DOI] [PubMed] [Google Scholar]
- GOMEZ M., SWARD K. Long-term regulation of contractility and calcium current in smooth muscle. Am. J. Physiol. 1997;273:C1714–C1720. doi: 10.1152/ajpcell.1997.273.5.C1714. [DOI] [PubMed] [Google Scholar]
- HALAYKO A.J., CAMORETTI-MERCADO B., FORSYTHE S.M., VIEIRA J.E., MITCHELL R.W., WYLAM M.E., HERSHENSON M.B., SOLWAY J. Divergent differentiation paths in airway smooth muscle culture: Induction of functionally contractile myocytes. Am. J. Physiol. 1999;276:L197–L206. doi: 10.1152/ajplung.1999.276.1.L197. [DOI] [PubMed] [Google Scholar]
- HALAYKO A.J., SALARI H., MA X., STEPHENS N.L. Markers of airway smooth muscle cell phenotype. Am. J. Physiol. 1996;270:L1040–L1051. doi: 10.1152/ajplung.1996.270.6.L1040. [DOI] [PubMed] [Google Scholar]
- HALAYKO A.J., SOLWAY J. Molecular mechanisms of phenotypic plasticity in smooth muscle cells. J. Appl. Physiol. 2001;90:358–368. doi: 10.1152/jappl.2001.90.1.358. [DOI] [PubMed] [Google Scholar]
- HAYASHI K., SAGA H., CHIMORI Y., KIMURA K., YAMANAKA Y., SOBUE K. Differentiated phenotype of smooth muscle cells depends on signaling pathways through insulin-like growth factors and phosphatidylinositol 3-kinase. J. Biol. Chem. 1998;273:28860–28867. doi: 10.1074/jbc.273.44.28860. [DOI] [PubMed] [Google Scholar]
- HAYASHI K., TAKAHASHI M., KIMURA K., NISHIDA W., SAGA H., SOBUE K. Changes in the balance of phosphoinositide 3-kinase/protein kinase B (Akt) and the mitogen-activated protein kinases (ERK/p38MAPK) determine a phenotype of visceral and vascular smooth muscle cells. J. Cell. Biol. 1999;145:727–740. doi: 10.1083/jcb.145.4.727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HIRST S.J. Airway smooth muscle cell culture: application to studies of airway wall remodelling and phenotype plasticity in asthma. Eur. Respir. J. 1996;9:808–820. doi: 10.1183/09031936.96.09040808. [DOI] [PubMed] [Google Scholar]
- HIRST S.J., TWORT C.H., LEE T.H. Differential effects of extracellular matrix proteins on human airway smooth muscle cell proliferation and phenotype. Am. J. Repir. Cell. Mol. Biol. 2000a;23:335–344. doi: 10.1165/ajrcmb.23.3.3990. [DOI] [PubMed] [Google Scholar]
- HIRST S.J., WALKER T.R., CHILVERS E.R. Phenotypic diversity and molecular mechanisms of airway smooth muscle proliferation in asthma. Eur. Respir. J. 2000b;16:159–177. doi: 10.1034/j.1399-3003.2000.16a28.x. [DOI] [PubMed] [Google Scholar]
- HOITING B.H., MEURS H., SCHUILING M., KUIPERS R., ELZINGA C.R., ZAAGSMA J. Modulation of agonist-induced phosphoinositide metabolism, Ca2+ signalling and contraction of airway smooth muscle by cyclic AMP- dependent mechanisms. Br. J. Pharmacol. 1996;117:419–426. doi: 10.1111/j.1476-5381.1996.tb15207.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- JIANG H., RAO K., HALAYKO A.J., LIU X., STEPHENS N.L. Ragweed sensitization-induced increase of myosin light chain kinase content in canine airway smooth muscle. Am. J. Respir. Cell. Mol. Biol. 1992;7:567–573. doi: 10.1165/ajrcmb/7.6.567. [DOI] [PubMed] [Google Scholar]
- JOHNSON S.R., KNOX A.J. Synthetic functions of airway smooth muscle in asthma. Trends Pharmacol. Sci. 1997;18:288–292. doi: 10.1016/s0165-6147(97)01092-4. [DOI] [PubMed] [Google Scholar]
- KARPOVA A.Y., ABE M.K., LI J., LIU P.T., RHEE J.M., KUO W.L., HERSHENSON M.B. MEK1 is required for PDGF-induced ERK activation and DNA synthesis in tracheal myocytes. Am. J. Physiol. 1997;272:L558–L565. doi: 10.1152/ajplung.1997.272.3.L558. [DOI] [PubMed] [Google Scholar]
- KELLEHER M.D., ABE M.K., CHAO T.S.O., JAIN M., GREEN J.M., SOLWAY J., ROSNER M.R., HERSHENSON M.B. Role of MAP kinase activation in bovine tracheal smooth muscle mitogenesis. Am. J. Physiol. 1995;268:L894–L901. doi: 10.1152/ajplung.1995.268.6.L894. [DOI] [PubMed] [Google Scholar]
- LINDQVIST A., NILSSON B.O., HELLSTRAND P. Inhibition of calcium entry preserves contractility of arterial smooth muscle in culture. J. Vasc. Res. 1997;34:103–108. doi: 10.1159/000159207. [DOI] [PubMed] [Google Scholar]
- MA X., WANG Y., STEPHENS N.L. Serum deprivation induces a unique hypercontractile phenotype of cultured smooth muscle cells. Am. J. Physiol. 1998;274:C1206–C1214. doi: 10.1152/ajpcell.1998.274.5.C1206. [DOI] [PubMed] [Google Scholar]
- MACK C.P., SOMLYO A.V., HAUTMANN M., SOMLYO A.P., OWENS G.K. Smooth muscle differentiation marker gene expression is regulated by RhoA-mediated actin polymerization. J. Biol. Chem. 2001;276:341–347. doi: 10.1074/jbc.M005505200. [DOI] [PubMed] [Google Scholar]
- MEURS H., ROFFEL A.F., POSTEMA J.B., TIMMERMANS A., ELZINGA C.R.S., KAUFFMAN H., ZAAGSMA J. Evidence for a direct relationship between phosphoinositide metabolism and airway smooth muscle contraction induced by muscarinic agonists. Eur. J. Pharmacol. 1988;156:271–274. doi: 10.1016/0014-2999(88)90331-7. [DOI] [PubMed] [Google Scholar]
- MITCHELL R.W., HALAYKO A.J., KAHRAMAN S., SOLWAY J., WYLAM M.E. Selective restoration of calcium coupling to muscarinic M(3) receptors in contractile cultured airway myocytes. Am. J. Physiol. Lung. Cell. Mol. Physiol. 2000;278:L1091–L1100. doi: 10.1152/ajplung.2000.278.5.L1091. [DOI] [PubMed] [Google Scholar]
- RAMOS K.S., ZHANG Y., SADHU D.N., CHAPKIN R.S. The induction of proliferative vascular smooth muscle cell phenotypes by benzo(a)pyrene is characterized by up-regulation of inositol phospholipid metabolism and c-Ha-ras gene expression. Arch. Biochem. Biophys. 1996;332:213–222. doi: 10.1006/abbi.1996.0335. [DOI] [PubMed] [Google Scholar]
- REUSCH H.P., ZIMMERMANN S., SCHAEFER M., PAUL M., MOELLING K. Regulation of Raf by Akt controls growth and differentiation in vascular smooth muscle cells. J. Biol. Chem. 2001;276:33630–33637. doi: 10.1074/jbc.M105322200. [DOI] [PubMed] [Google Scholar]
- RODRIGUEZ-VICIANA P., WARNE P.H., KHWAJA A., MARTE B.M., PAPPIN D., DAS P., WATERFIELD M.D., RIDLEY A., DOWNWARD J. Role of phosphoinositide 3-OH kinase in cell transformation and control of the actin cytoskeleton by Ras. Cell. 1997;89:457–467. doi: 10.1016/s0092-8674(00)80226-3. [DOI] [PubMed] [Google Scholar]
- ROY J., KAZI J.M., HEDIN U., THYBERG J. Phenotypic modulation of arterial smooth muscle cells is associated with prolonged activation of ERK 1/2. Differentiation. 2001;67:50–58. doi: 10.1046/j.1432-0436.2001.067001050.x. [DOI] [PubMed] [Google Scholar]
- SCHMIDT D., WATSON N., RUEHLMANN E., MAGNUSSEN H., RABE K.F. Serum immunoglobulin E levels predict human airway reactivity in vitro. Clin. Exp. Allergy. 2000;30:233–241. doi: 10.1046/j.1365-2222.2000.00767.x. [DOI] [PubMed] [Google Scholar]
- STEPHENS N.L., LI W., WANG Y., MA X. The contractile apparatus of airway smooth muscle. Biophysics and biochemistry. Am. J. Respir. Crit. Care Med. 1998;158:S80–S94. doi: 10.1164/ajrccm.158.supplement_2.13tac300. [DOI] [PubMed] [Google Scholar]
- TOGASHI H., EMALA C.W., HALL I.P., HIRSHMAN C.A. Carbachol-induced actin reorganization involves Gi activation of Rho in human airway smooth muscle cells. Am. J. Physiol. 1998;274:L803–L809. doi: 10.1152/ajplung.1998.274.5.L803. [DOI] [PubMed] [Google Scholar]
- WALKER T.R., MOORE S.M., LAWSON M.F., PANETTIERI R.A., , JR, CHILVERS E.R. Platelet-derived growth factor-BB and thrombin activate phosphoinositide 3-kinase and protein kinase B: role in mediating airway smooth muscle proliferation. Mol. Pharmacol. 1998;54:1007–1015. doi: 10.1124/mol.54.6.1007. [DOI] [PubMed] [Google Scholar]
- ZAAGSMA J., ROFFEL A.F., MEURS H. Muscarinic control of airway function. Life Sci. 1997;60:1061–1068. doi: 10.1016/s0024-3205(97)00048-9. [DOI] [PubMed] [Google Scholar]


