Abstract
Microdialysis was used to study the acute and chronic effects of escitalopram (S-citalopram; ESCIT) and chronic citalopram (CIT), together with the 5-HT1A receptor antagonist WAY100,635 (N-[2-[methoxyphenyl)-1-piperazinyl]ethyl]-N-(2-pyridinyl) cyclohexane carboxamide trihydrochloride) and the 5-HT1A receptor agonist 8-hydroxy-2-(di-n-propylamino)tetralin (8-OH-DPAT), on extracellular 5-hydroxytryptamine (5-HT) levels in the rat prefrontal cortex.
Extracellular 5-HT rose to 234 and 298% of basal values after subcutaneous (s.c.) acute doses of 0.15 and 0.63 mg kg−1 ESCIT. No further increase was observed at 2.5 mg kg−1 ESCIT (290%).
The effect of 13-day s.c. infusion of 10 mg kg−1day−1 ESCIT on extracellular 5-HT (422% of baseline) was greater than after 2 days (257% of baseline), whereas exposure to ESCIT was similar. In contrast, the increase in extracellular 5-HT induced by the infusion of CIT for 2 (306%) and 13 days (302%) was similar. However, brain and plasma levels of S-citalopram in rats infused with CIT for 13 days were lower than after 2 days.
Acute treatment with 2.5 mg kg−1 ESCIT or 5 mg kg−1 CIT raised extracellular 5-HT by 243 and 276%, respectively, in rats given chronic vehicle but had no effect in rats given ESCIT (10 mg kg−1 day−1) or CIT (20 mg kg−1 day−1) for 2 or 13 days, suggesting that the infused doses had maximally increased extracellular 5-HT. WAY100,635 (0.1 mg kg−1 s.c.) increased extracellular 5-HT levels by 168, 174 and 169% of prechallenge values in rats infused with vehicle or ESCIT for 2 or 13 days, respectively. WAY100,635 enhanced extracellular 5-HT levels to 226, 153 and 164% of prechallenge values in rats infused with vehicle or CIT for 2 and 13 days, respectively.
8-OH-DPAT (0.025 mg kg−1) reduced extracellular 5-HT by 54% in control rats, but had no effect in those given ESCIT and CIT for 13 days.
This series of experiments led to the conclusion that chronic treatment with ESCIT desensitizes the 5-HT1A receptors, regulating the release of 5-HT in the prefrontal cortex and enhances the effect of the drug on extracellular 5-HT. They also indicate that chronic treatment with ESCIT and CIT did not prevent WAY100,635 from raising extracellular 5-HT.
Keywords: 5-HT1A autoreceptor sensitivity; 5-HT1A autoreceptor blockade; 5-HTreuptake inhibition; antidepressants; citalopram; escitalopram; microdialysis; pharmacokinetics; prefrontal cortex; WAY100,635
Introduction
Citalopram (CIT) is a clinically effective antidepressant drug (Milne & Goa, 1991; Pollock, 2001), whose selective effects on the serotonergic system are documented in vitro and in vivo (Hyttel, 1977; Pozzi et al., 1999). CIT is a racemic compound and in rat brain synaptosomes, S-citalopram (escitalopram, ESCIT) is 130 times more potent than R-citalopram in inhibiting serotonin (5-HT, 5-hydroxytryptamine) reuptake (Hyttel et al., 1992). In mice, ESCIT but not the R-enantiomer potentiated the behavioural syndrome induced by 5-hydroxytryptophan, an indirect indicator of 5-HT reuptake inhibition in vivo (Hyttel et al., 1992). The selectivity of ESCIT in inhibiting 5-HT reuptake has been confirmed in human embryonic kidney cells transfected with mRNA encoding the human 5-HT transporter (Owens et al., 2001). These findings showed that the CIT-induced inhibition of 5-HT reuptake and antidepressant activity resides in the S-enantiomer. Randomized clinical trials have shown that ESCIT is effective in patients with major depression (Montgomery et al., 2001; Burke et al., 2002; Wade et al., 2002; Lepola et al., 2003).
Enhancement of brain serotonergic transmission by selective serotonin reuptake inhibitors (SSRIs) is important for their therapeutic effect (Salomon et al., 1993). However, the fact that chronic treatment is required to alleviate depression (Garattini & Samanin, 1988) suggests that adaptive neuronal changes are necessary. In functional neuroimaging studies, depression was associated with abnormalities of cerebral blood flow and metabolism in various regions of the prefrontal cortex (Drevets, 1998). The reversal of such changes by antidepressant therapies suggests that the prefrontal cortex may be important in the action of these drugs. Microdialysis studies in rats showed that extracellular 5-HT in the prefrontal cortex did not increase much in response to acute SSRI treatment (Invernizzi et al., 1992; 1996; Fuller, 1994; Gardier et al., 1996). This is at least partly because their effect is limited by the simultaneous activation of autoinhibitory somatodendritic 5-HT1A autoreceptors caused by increased endogenous 5-HT levels in the raphe nuclei (Adell & Artigas, 1991; Invernizzi et al., 1992; Gartside et al., 1995; Romero & Artigas, 1997).
The effect of SSRI on cortical extracellular 5-HT is enhanced by chronic treatment (Bel & Artigas, 1993; Invernizzi et al., 1994; 1996; Dawson et al., 2000) and although some inconsistencies have been reported (Hjorth & Auerbach, 1994; Dremencov et al., 2000), electrophysiological and microdialysis studies indicate that somatodendritic 5-HT1A autoreceptors become desensitized after prolonged administration of an SSRI (Blier & De Montigny, 1983; Blier et al., 1984; Chaput et al., 1986; Invernizzi et al., 1994; 1996; Rutter et al., 1994; Dawson et al., 2000). These results suggest that the desensitization of 5-HT1A receptors is involved in the slow onset of the antidepressant effect of SSRIs. Thus, the more rapid improvement in depressed patients given an SSRI with pindolol, a 5-HT1A/1B/β-adrenoceptor antagonist, suggests that the therapeutic effect of SSRIs may be accelerated by reducing 5-HT1A receptor function (Artigas et al., 1994; Blier & Bergeron, 1995; Zanardi et al., 1997; 1998; Bordet et al., 1998). However, other studies have failed to confirm these results (Berman et al., 1997; Tome et al., 1997), and it remains to be established whether selective blockade of 5-HT1A receptors in depressed patients improves the antidepressant effect of SSRIs.
Mork et al. (2003) recently reported that acute treatment of rats with ESCIT raised extracellular 5-HT, and that this effect was inhibited by the R-enantiomer. However, there is no published information on the effects of chronic ESCIT treatment on brain 5-HT levels. In the present study, we compared the effect of chronic ESCIT and CIT on extracellular 5-HT levels in the prefrontal cortex using intracerebral microdialysis in conscious rats. Firstly, we examined the acute effect of ESCIT on extracellular 5-HT levels in the prefrontal cortex; then, using subcutaneous (s.c.) osmotic pumps to deliver the drug continuously, we examined the effect of 2 and 13-days treatment with ESCIT and CIT on basal extracellular 5-HT levels and the response to an acute challenge dose of ESCIT, CIT or the selective 5-HT1A receptor antagonist WAY 100,635 (N-[2-[methoxyphenyl)-1-piperazinyl]ethyl]-N-(2-pyridinyl) cyclohexane carboxamide trihydrochloride) (Forster et al., 1995). To investigate whether the sensitivity of 5-HT1A receptors regulating 5-HT release was reduced after chronic treatment, we examined the inhibitory effect of the 5-HT1A receptor agonist 8-hydroxy-2-(di-n-propylamino)tetralin (8-OH-DPAT) (Sharp & Hjorth, 1992) on cortical extracellular 5-HT levels in rats infused with ESCIT and CIT for 13 days.
Methods
Animals and drug treatment
Male rats (CD-COBS, Charles River, Italy), weighing about 350 g (about 250 g at the beginning of chronic treatment), were used. The animals were housed at constant room temperature (21±1°C) and relative humidity (60±5%) under a regular light/dark schedule (light 07:00–19:00). Food and water were freely available.
Procedures involving animals and their care were conducted in conformity with the institutional guidelines that are in compliance with national (D.L. n. 116, G.U., suppl. 40, 18 Febbraio 1992, Circolare No. 8, G.U., 14 luglio 1994) and international laws and policies (EEC Council Directive 86/609, OJ L 358,1, Dec.12, 1987; Guide for the Care and Use of Laboratory Animals, U.S. National Research Council, 1996).
For acute administration, 0.02, 0.04, 0.15, 0.63 and 2.5 mg kg−1 S-citalopram oxalate (H. Lundbeck A/S, Denmark) was dissolved in phosphate-buffered saline (PBS) and injected subcutaneously (s.c.) in a volume of 2 ml kg−1. Osmotic pumps (Alzet, model 2ML2) were implanted s.c. for chronic treatment. The pumps were filled with PBS, 32 mg ml−1 S-citalopram oxalate or 67 mg ml−1 CIT HBr, corresponding, respectively, to 10 and 20 mg kg−1 day−1 of the free base. The doses were calculated for rats with a mean body weight of 300 g, which were infused for 2 or 13 days. Preliminary experiments showed that these doses of ESCIT and CIT maximally increased extracellular 5-HT in the prefrontal cortex. Continuous infusion of ESCIT and CIT for 13 days had no significant effects on the weight gain of the rats that otherwise appeared healthy, with no gross behavioural changes.
On the day of the experiment, 24 h after implantation of the microdialysis probe, once basal levels of 5-HT in the dialysate were stable (no more than 15% difference between three consecutive samples), rats were injected with 2.5 mg kg−1 s.c. ESCIT or 5 mg kg−1 s.c. CIT. After 1 h, rats were given 0.1 mg kg−1 s.c. WAY100,635 (N-[2-[methoxyphenyl)-1-piperazinyl]ethyl]-N-(2-pyridinyl) cyclohexane carboxamide trihydrochloride) (Pharmacia, Nerviano, Italy) dissolved in saline. The output of 5-HT in the prefrontal cortex was measured for 60 min after challenge. About 24 h later, rats were killed and blood and brain samples were collected for the determination of drug levels and probe placement. ESCIT, CIT and WAY100,635 challenge studies and drug measurements were conducted in animals implanted with the minipump.
Another group of rats was given PBS, 10 mg kg−1 day−1 ESCIT or 20 mg kg−1 day−1 CIT, as above. On day 13, they were anaesthetized with 3.5 mL kg−1 equithesin (composition: 1.2 g pentobarbital, 5.3 g chloral hydrate, 2.7 g MgSO4·7 H2O, 49.5 ml propylene glycol, 12.5 ml ethanol and 58 ml distilled water) and the osmotic pump was removed to allow clearance of the drug, and a dialysis probe was implanted into the prefrontal cortex. At 24 h after removal of the pump, rats were injected with 0.025 mg kg−1 s.c. 8-OH-DPAT (Sigma-RBI, Milan, Italy) or saline, and extracellular 5-HT was measured for 2 h. Drug levels in the brain and plasma were determined in a separate group of rats given ESCIT or CIT for 13 days, 24 h after removal of the osmotic pump.
Dialysis procedure
Rats were anaesthetized with 3.5 ml kg−1 equithesin and placed on a stereotaxic apparatus (David Kopf Instruments, Tujunga, CA, U.S.A.). A hole was drilled in the frontal bone and a small incision was made in the dura with a bent needle tip. The probe, perfused with artificial cerebrospinal fluid (aCSF; see below for composition), was lowered slowly into the prefrontal cortex and fixed vertically to the skull using two or three stainless-steel anchorage screws and acrylic cement. Stereotaxic coordinates relative to the probe tip were as follows: AP=+12.7, L=±0.6, V=+4.6 from the interaural line according to the Paxinos and Watson atlas (1982). The dialysis probes were of the concentric type and were prepared essentially as described by Robinson & Whishaw (1988), except that the dialysis membrane was made of cuprophan (Sorin Biomedica, Italy). The exposed membrane was 4 mm long. Each rat was implanted with a single probe. Rats were allowed to recover from anaesthesia, one in each cage, with free access to food and water. About 24 h after surgery, the inlet cannula was connected by polyethylene tubing to a 2.5 ml syringe containing aCSF (composition: 145 mM NaCl, 3 mM KCl, 1.26 mM CaCl2.2·H2O, 1 mM MgCl2·6·H2O in distilled water, buffered at pH 7.4 with 2 mM sodium phosphate buffer). Each probe was perfused at a constant flow rate of 1 μl min−1 using a microinfusion pump (CMA/Microdialysis, Stockholm, Sweden). After 30–60 min washout, consecutive 20-min samples of perfusate were collected in minivials and immediately injected into the high-performance liquid chromatograph (HPLC) with electrochemical detection (HPLC-ED).
Osmotic pumps
Osmotic pumps (Alzet, model 2ML2, Charles River, Italy) delivering about 5 μl h−1 during 14 days were used. Rats were anaesthetized with 3.5 ml kg−1 i.p. equithesin. The skin was shaved, washed with antiseptic solution, and an incision was made between the scapulae and a hemostat inserted to create a pocket. The filled pump was inserted into the pocket and the wound was closed with 2–3 clips.
5-HT and ESCIT assays
5-HT concentrations in the dialysate were determined by HPLC-ED, as previously described (Invernizzi et al., 1992). Concentrations of ESCIT in the plasma and brain were determined using minor modifications of the HPLC procedure described by Kosel et al. (1998) (i.e., plasma extraction was with ether and detection was UV absorption; (Grignaschi et al., 1998). Brain tissue was homogenized in distilled water (10 ml g−1) and 2 ml of homogenate was processed as described for the plasma.
The lower limit for quantification of ESCIT in the plasma was 10 ng ml−1 using 1 ml; in the brain the limit was about 50 ng g−1, using approximately 200 mg of tissue. At these concentrations, the coefficients of variation (C.V.) for the precision and reproducibility of the assay were between 10 and 15%. Higher concentrations gave a C.V. of less than 5%.
Histological procedure
At the end of the experiments, rats were anaesthetized with 400 mg kg−1 i.p. chloral hydrate and killed by decapitation; their brains were immediately removed and the correct placement of the probes was checked by examining the probe tracks. Only rats with correct probe placement were included in the results.
Statistics
The effects of chronic infusion of PBS, ESCIT or CIT on basal 5-HT levels before challenge with ESCIT, CIT or 8-OH-DPAT were compared by one-way analysis of variance (ANOVA) followed by Tukey–Kramer's test. All time-course data were analysed by two-way ANOVA for repeated measures with treatment and time as between and within factors, respectively. Post hoc comparisons were made by Tukey–Kramer's test. The effect of the antagonist WAY100,635 on extracellular 5-HT after acute challenge with ESCIT or CIT in rats receiving chronic vehicle or drug was compared by the Kruskal–Wallis test after normalization of 5-HT levels as a percentage of preinjection values. The number of rats in each group varied from three to 10 and is reported in the tables and figure legends. Statistical analysis was carried out using the Stat View 5.0 statistical package for Apple-Macintosh computer (SAS Institute Inc., Cary, NC, U.S.A.). The values missing, because of occasional problems in sample collection or analysis, were replaced by the mean of the samples immediately before and after.
Brain and plasma concentrations of S-citalopram measured after acute ESCIT and CIT or at different times after infusion were compared by Student's t-test.
Results
Extracellular 5-HT in the prefrontal cortex after acute ESCIT
The basal extracellular concentration of 5-HT in the prefrontal cortex was 4.6±0.3 fmol 20 μl−1 (mean±s.e.m., n=28), not corrected for probe recovery. No significant differences were found between basal 5-HT (preinjection) levels in different experimental groups (F5,22=1.3, P>0.05). Basal 5-HT levels in rats receiving PBS were stable during the 2 h observation period.
As shown in Figure 1, ESCIT dose dependently raised extracellular 5-HT in the prefrontal cortex (Ftreatment 5,22=13.7, P<0.001; Ftime 6,132=16.7, P<0.001; Ftreatment × time 30,132=3.3, P<0.001). Extracellular 5-HT levels in rats given 0.15, 0.63 and 2.5 mg kg−1 s.c. were significantly higher than in rats given PBS (all P<0.05; Tukey–Kramer's test). Extracellular 5-HT levels were maximally increased 40–60 min after injection and reached 234 and 298% of preinjection levels at 0.15 and 0.63 mg kg−1. No further increase was observed at the dose of 2.5 mg kg−1 (290% of preinjection levels). The extracellular levels of 5-HT in rats given 0.15 mg kg−1 ESCIT increased maximally at 40 min, and then gradually returned to baseline. Extracellular 5-HT was still significantly higher than preinjection levels 2 h after 0.63 and 2.5 mg kg−1 ESCIT (P<0.05, Tukey–Kramer's test). No significant increase in extracellular 5-HT was observed in the prefrontal cortex of rats given 0.02 and 0.04 mg kg−1 ESCIT (P>0.05 vs. PBS; Tukey–Kramer's test) (Figure 1).
Figure 1.
Extracellular 5-HT levels in the prefrontal cortex of rats injected subcutaneously with PBS, 0.02, 0.04, 0.15, 0.63 and 2.5 mg kg−1 ESCIT. Once the extracellular concentration of 5-HT was stable, rats were injected s.c. with ESCIT or vehicle (arrow). Data are means±s.e.m. and are percentages of preinjection values. Basal levels of 5-HT in fmol 20 μl−1 (mean±s.e.m.) were: PBS, 4.5±0.4 (n=4); 0.02 mg kg−1, 4.1±0.4 (n=3); 0.04 mg kg−1, 4.6±0.6 (n=5); 0.15 mg kg−1, 3.7±0.5 (n=4); 0.63 mg kg−1, 6.3±1.1 (n=4); 2.5 mg kg−1, 4.7±0.6 (n=8). Solid symbols indicate P<0.05 vs PBS (Tukey–Kramer's test).
Extracellular 5-HT in the prefrontal cortex of rats given chronic ESCIT: effect of ESCIT and WAY 100 635 challenges
Table 1 shows the effect of chronic ESCIT on extracellular 5-HT in the prefrontal cortex before ESCIT challenge. Extracellular levels of 5-HT in rats given PBS for 2 and 13 days were not significantly different (F1,8=0.9, P>0.05) and were therefore pooled. The infusion of 10 mg kg−1 day−1 ESCIT significantly increased cortical extracellular 5-HT (F2,19=22.8, P<0.001). Extracellular 5-HT levels in rats given a 2-day infusion of ESCIT were 257% higher than in PBS-treated controls (P<0.05; Tukey–Kramer's test). Infusion of 10 mg kg−1 day−1 ESCIT for 13 days increased extracellular 5-HT to 422%, and the effect was significantly greater than in rats given the drug for 2 days, or PBS (both P<0.05; Tukey–Kramer's test).
Table 1.
Effects of chronic escitalopram (ESCIT) or citalopram (CIT) treatment on basal extracellular 5-HT in the prefrontal cortex of rats and concentrations of S-citalopram in the brain and plasma during infusion and after washout
| 5-HT (fmol 20 μl−1) | S-citalopram concentrations | ||
|---|---|---|---|
| Brain (ng g−1) | Plasma (ng mL−1) | ||
| During infusion | |||
| PBS | 4.6±0.4 (10) | — | — |
| ESCIT 2 days | 11.8±1.6* (6) | 228±29 (9) | 59±12 (9) |
| ESCIT 13 days | 19.4±2.9*,** (6) | 231±44 (6) | 52±12 (6) |
| PBS | 5.2±0.8 (10) | — | — |
| CIT 2 days | 15.9±1.6* (9) | 331±52 (9) | 62±8.1 (9) |
| CIT 13 days | 15.7±1.6* (10) | 185±32*** (10) | 26.3±4.4*** (10) |
| After 24 h washout | |||
| PBS (+saline) | 6.2±0.5 (5) | — | — |
| PBS (+8-OH-DPAT) | 5.4±0.5 (6) | — | — |
| ESCIT 13 days | 6.9±0.8 (7) | <50 | <10 |
| CIT 13 days | 6.2±0.8 (8) | <50 | <10 |
Rats received PBS, 10 mg kg−1 day−1 ESCIT or 20 mg kg−1 day−1 CIT from a subcutaneous osmotic pump for 2 or 13 days. Cortical extracellular 5-HT and S-citalopram in the brain and plasma were measured at the end of infusion and 24 h after the removal of the osmotic pump
Data are mean±s.e.m. The number of rats is shown in parentheses.
P<0.05 vs PBS;
P<0.05 vs 2 days (Tukey–Kramer's test);
P<0.05 vs 2 days (Student's t-test).
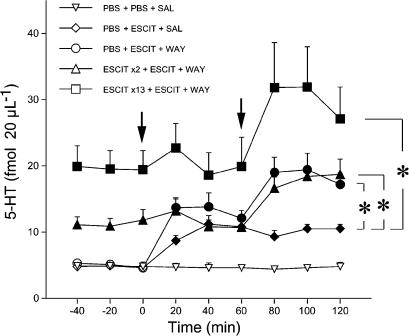
Figure 2 shows the effect of a challenge dose of ESCIT followed by WAY100,635 on extracellular 5-HT in the prefrontal cortex of rats given 10 mg kg−1 day−1 ESCIT or PBS for 2 and 13 days. ANOVA indicated a significant effect of treatment (F4,26=10.6, P<0.001), time (F6,156=16, P<0.001) and treatment by time interaction (F24,156=3.3, P<0.001). The challenge dose of ESCIT significantly increased extracellular 5-HT in rats infused chronically with PBS (P<0.05 vs vehicle, Tukey–Kramer's test). 5-HT reached 243% of basal levels at 40 min and remained high. In contrast, the ESCIT challenge had no significant effect in rats infused with the drug for 2 or 13 days (P>0.05; Tukey–Kramer's test). WAY100,635 challenge, 60 min later, significantly enhanced the increase of extracellular 5-HT caused by ESCIT. Extracellular 5-HT in rats given WAY100,635 reached 168, 174 and 169% of prechallenge values in rats infused chronically with PBS or ESCIT for 2 and 13 days (all P<0.05 vs prechallenge values; Tukey–Kramer's test). The effect of WAY100,635 expressed as percentage increase over prechallenge values in the three experimental groups was not significantly different (P>0.05, Kruskall–Wallis' test).
Figure 2.
Effect of a challenge dose of ESCIT and the selective 5-HT1A receptor antagonist WAY100,635 (WAY) on extracellular 5-HT levels in the prefrontal cortex in rats given chronic ESCIT. Rats were infused with PBS or 10 mg kg−1 day−1 ESCIT for 2 and 13 days. On the last day, with the osmotic pump in place, rats were injected s.c. with two consecutive drug challenges: 2.5 mg kg−1 ESCIT (first arrow), followed by 0.1 mg kg−1 WAY100,635 or saline (SAL) 60 min apart (second arrow). Data are means±s.e.m. Basal levels of 5-HT in fmol 20 μl−1 (mean±s.e.m.) were: PBS+PBS+SAL, 4.8±0.3 (n=5); PBS+ESCIT+ SAL, 4.6±0.6 (n=4); PBS+ ESCIT+WAY, 4.6±0.4 (n=10); ESCIT × 2+ESCIT+WAY, 11.8±1.6 (n=6); ESCIT × 13+ESCIT+WAY, 19.4±2.9 (n=6). Solid symbols indicate P<0.05 vs PBS+PBS+SAL (Tukey–Kramer's test). Asterisks indicate a significant increase of extracellular 5-HT in rats given WAY100,635 compared to those receiving ESCIT alone (P<0.05; Tukey–Kramer's test).
Extracellular 5-HT in the prefrontal cortex of rats given chronic CIT: effect of CIT and WAY 100 635 challenges
CIT 20 mg kg−1 day−1 via subcutaneous osmotic pump significantly increased extracellular 5-HT in the prefrontal cortex (F2,26=20.8, P<0.001) (Table 1). Extracellular 5-HT increased to a similar extent (P>0.05, Tukey–Kramer's test) in rats infused for two (306%) or 13 days (302%).
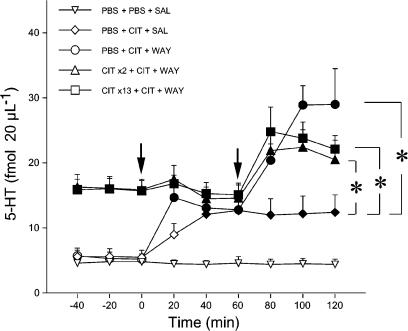
Figure 3 shows the effect of a challenge dose of CIT (5 mg kg−1 s.c.) followed by WAY100,635 60 min later on extracellular 5-HT in the prefrontal cortex of rats given PBS or CIT for 2 and 13 days. ANOVA indicated a significant effect of treatment (F4,34=8.5, P<0.001), time (F6,204=15.9, P<0.001) and treatment by time interaction (F24,204=4.1, P<0.001). Extracellular levels of 5-HT in rats given the CIT challenge were significantly higher than in PBS-injected controls (P<0.05, Tukey–Kramer's test). 5-HT reached 230% of prechallenge levels and did not return to basal levels until the end of the observation period. In contrast, CIT had no effect on extracellular 5-HT in rats receiving the drug for 2 or 13 days (P>0.05; Tukey–Kramer's test). WAY100,635 significantly enhanced the increase of extracellular 5-HT caused by CIT. Extracellular 5-HT reached 226, 153 and 158% of prechallenge values in the prefrontal cortex of rats infused chronically with PBS, or CIT for 2 or 13 days, respectively (all P<0.05 vs prechallenge values; Tukey–Kramer's test). WAY100,635 tended to have more effect in rats infused chronically with PBS than in those receiving CIT for 2 and 13 days, but the differences were not significant (P>0.05, Kruskall–Wallis' test).
Figure 3.
Effect of a challenge dose of CIT and the selective 5-HT1A receptor antagonist WAY100,635 (WAY) on extracellular 5-HT levels in the prefrontal cortex in rats given chronic CIT. Rats were infused with PBS or 20 mg kg−1 day−1 CIT for 2 and 13 days. On the last day, with the osmotic pump in place, rats were injected s.c. with two consecutive drug challenges: 5 mg kg−1 CIT (first arrow), followed by 0.1 mg kg−1 WAY100,635 or saline (SAL) 60 min apart (second arrow). Data are means±s.e.m. Basal levels of 5-HT in fmol 20 μl−1 (mean±s.e.m.) were: PBS+PBS+SAL, 4.8±0.3 (n=5); PBS+CIT+SAL, 5.5±1.1 (n=5); PBS+CIT+WAY, 5.2±0.8 (n=10); CIT × 2+CIT+WAY, 15.9±1.6 (n=9); CIT × 13+CIT+WAY, 15.7±1.6 (n=10). Solid symbols indicate P<0.05 vs PBS+PBS+SAL (Tukey–Kramer's test). Asterisks indicate a significant increase of extracellular 5-HT in rats given WAY100,635 compared to those receiving CIT alone (P<0.05; Tukey–Kramer's test).
Effect of 8-OH-DPAT on extracellular 5-HT in rats given chronic ESCIT or CIT
At 24 h after the removal of the osmotic pump, no differences were found in extracellular 5-HT levels in the prefrontal cortex of rats given PBS, ESCIT or CIT for 13 days (F3,22=0.8, P>0.05) (Table 1).
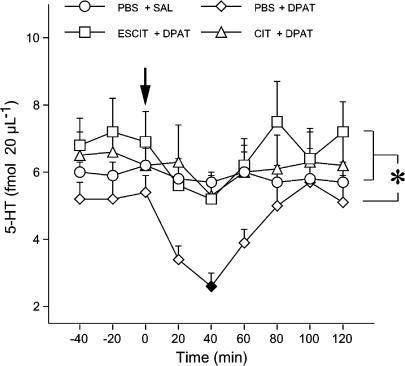
Figure 4 shows the effect of 0.025 mg kg−1 of 8-OH-DPAT or saline on extracellular 5-HT in rats infused with PBS, 10 mg kg−1 day−1 s.c. ESCIT or 20 mg kg−1 day−1 s.c. CIT for 13 days. ANOVA applied to the period between 20 and 60 min after 8-OH-DPAT indicated a significant effect of treatment (F3,22=9.8, P<0.001) and time (F2,44=6.4, P<0.01), but no treatment by time interaction (F6,44=1, P>0.05). The injection of saline in rats infused with PBS for 13 days had no effects on extracellular 5-HT (P>0.05 vs prechallenge values; Tukey–Kramer's test). 8-OH-DPAT reduced extracellular 5-HT levels by 54% in rats infused with PBS (P<0.05 vs PBS+saline; Tukey–Kramer's test), but had no effect in those given chronic ESCIT and CIT (9 and 7% reduction of extracellular 5-HT levels, respectively; both P>0.05 vs PBS+saline, Tukey–Kramer's test).
Figure 4.
Effect of chronic ESCIT and CIT on 8-OH-DPAT-induced decrease of extracellular 5-HT in the prefrontal cortex. Rats were infused with PBS, 10 mg kg−1 day−1 ESCIT or 20 mg/kg CIT for 13 days. On day 14, about 24 h after removal of the osmotic pump, rats were injected with a challenge dose of the selective 5-HT1A receptor agonist 8-OH-DPAT (DPAT; 0.025 mg kg−1 s.c.) or saline (SAL) (arrow). Data are means±s.e.m. Basal levels of 5-HT in fmol 20 μl−1 were: PBS+SAL, 6.2±0.5 (n=5); PBS+DPAT, 5.4±0.5 (n=6); ESCIT+DPAT, 6.9±0.9 (n=7); CIT+DPAT, 6.2±0.8 (n=8). Solid symbol indicates P<0.05 vs PBS+SAL (Tukey–Kramer's test). The asterisk indicates a significant difference between extracellular 5-HT levels in rats given chronic ESCIT or CIT and those infused with PBS (P<0.05 for both comparisons; Tukey–Kramer's test).
S-citalopram in the plasma and brain
Table 1 shows plasma and brain concentrations of S-citalopram in rats infused with 10 mg kg−1 day−1 ESCIT or 20 mg kg−1 day−1 CIT. The mean plasma and brain concentrations of ESCIT were not significantly different in rats given ESCIT for 2 or 13 days. In contrast, the plasma and brain concentrations of ESCIT in rats given CIT for 13 days were significantly lower than in rats given the drug for 2 days (both P<0.05, Student's t-test). S-citalopram plasma and brain concentrations were below the limit of quantification 24 h after removal of the osmotic pump in rats given CIT or ESCIT (Table 1).
Table 2 shows plasma and brain concentrations of S-citalopram in rats 40 min after injection of 0.63 and 2.5 mg kg−1 s.c. ESCIT or 1.25 and 5 mg kg−1 s.c. CIT. Plasma and brain concentrations of S-citalopram increased proportionally to the dose of ESCIT and CIT. Plasma and brain concentrations of S-citalopram after s.c. CIT were not different from those with half doses of ESCIT (P>0.05 for all comparisons; Student's t-test).
Table 2.
Effects of acute escitalopram (ESCIT) or citalopram (CIT) treatment on the concentrations of S-citalopram in the brain and plasma 40 min after s.c. injection
| S-citalopram concentrations | ||
|---|---|---|
| Drug (mg kg−1) | Brain (ng g−1) | Plasma (ng ml−1) |
| ESCIT (0.63) | 190±41 | 22±6 |
| ESCIT (2.5) | 699±108 | 96±24 |
| CIT (1.25) | 229±41 | 39±7 |
| CIT (5) | 528±25 | 96±13 |
Data are mean±s.e.m. of four rats per group.
Discussion
ESCIT dose dependently raised extracellular 5-HT levels in the prefrontal cortex, the threshold dose (0. 15 mg kg−1) more than doubling them. ESCIT 0.63 mg kg−1 increased cortical extracellular 5-HT levels to three times the basal value, and 2.5 mg kg−1 did not raise it further. These results confirm a previous report that ESCIT increased extracellular 5-HT in the prefrontal cortex of rats (Mork et al., 2003) and suggest that doses of ESCIT above 0.63 mg kg−1 maximally inhibited brain 5-HT reuptake. Accordingly, 0.8 mg kg−1 s.c. ESCIT potentiated 5-hydroxytryptophan-induced behaviour in rats and mice (Hyttel et al., 1992; Sanchez et al., 2003), and the same dose of CIT reversed H75/12-induced depletion of brain 5-HT, an indirect measure of 5-HT reuptake inhibition in vivo (Hyttel, 1977). In line with these results, we found that the brain concentrations of S-citalopram 40 min after injection of 0.63 mg kg−1 (586 nM) or an equivalent dose of S-citalopram contained in the racemate (706 nM) were well above the IC50 for inhibition of [3H]5-HT uptake in rat brain synaptosomes (1.5–2.1 nM) (Hyttel et al., 1992; Sanchez et al., 2003) and in cells expressing the human 5-HT transporter (2.5–6.6 nM) (Owens et al., 2001; Sanchez et al., 2003).
Short-term (2-day) infusion of 10 mg kg−1 day−1 ESCIT increased extracellular 5-HT in the prefrontal cortex to 257% of the basal value. This effect is similar to that observed in the prefrontal cortex of rats infused with 20 mg kg−1 day−1 CIT (306%) and comparable to that in rats acutely injected with 0.63 mg kg−1 ESCIT (298%), a dose that maximally increased extracellular 5-HT (present study). In addition, a challenge dose of 2.5 mg kg−1 ESCIT or 5 mg kg−1 CIT significantly raised extracellular 5-HT in the prefrontal cortex of rats given chronic PBS, but had no effect in rats given the drugs for 2 days. These results indicate that both doses of ESCIT and CIT infused through the osmotic pump maximally increased extracellular 5-HT levels, so the transporter sites are presumably saturated by the brain concentrations of S-citalopram achieved after the infusion of 10 mg kg−1 day−1 ESCIT and 20 mg kg−1 day−1 CIT (228 and 331 ng g−1, respectively). Plasma levels of S-citalopram in rats infused with ESCIT and CIT for 2 days (ca. 60 ng ml−1) are close to plasma concentrations achieved in patients taking therapeutic doses of ESCIT (Rochat et al., 1995; Sidhu et al., 1997; Zheng et al., 2000; Waugh & Goa, 2003). In the rat, brain concentrations of S-citalopram greatly exceed those in the plasma and it is reasonable to assume that the same occurs in humans. Thus, concentrations of the active enantiomer of CIT at the site of action are presumably close to those saturating the 5-HT transporter in both species. A recent positron emission tomography imaging study showed that the binding potential of [11C]DASB, a selective ligand for the 5-HT transporter, was reduced by about 80% in the striatum of patients treated with 20 mg day−1 CIT (Meyer et al., 2001).
Cortical extracellular 5-HT levels increased much more in rats infused with 10 mg kg−1 day−1 ESCIT for 13 days than for 2 days. In line with these results, chronic treatment enhanced the effect of fluoxetine, fluvoxamine and CIT on extracellular 5-HT levels (Bel & Artigas, 1993; Invernizzi et al., 1994; 1996; Rutter et al., 1994; Kreiss & Lucki, 1995). However, other studies did not confirm that chronic treatment enhanced the effect of CIT on extracellular 5-HT (Hjorth & Auerbach, 1994; Gundlah et al., 1997). The greater effect of ESCIT was apparently not due to pharmacokinetic reasons because plasma and brain concentrations of S-citalopram were comparable after short- and long-term infusions, indicating that the steady state was achieved in 2 days, in line with the short half-life of ESCIT in the rat (about 1.5 h) (Mork et al., 2003), and then maintained for the duration of the study.
Surprisingly, the effect of CIT on extracellular 5-HT was not enhanced after a 13-days continuous infusion. This apparently contradicts the results with ESCIT and those obtained in our previous study, showing that a low dose of CIT (1 mg/kg i.p.), which had no effect in rats repeatedly injected with saline, significantly increased extracellular 5-HT in those receiving 10 mg/kg CIT twice daily for 2 weeks (Invernizzi et al., 1994). ESCIT and CIT differ in their ability to raise extracellular 5-HT in the rat frontal cortex (Mork et al., 2003). R-citalopram had no effect by itself, but attenuated ESCIT-induced increase of extracellular 5-HT (Mork et al., 2003). A 2-day infusion of 10 mg kg−1 ESCIT and 20 mg kg−1 CIT increased extracellular 5-HT to a similar extent, whereas only the effect of ESCIT was enhanced by 13 days infusion. Thus, it is conceivable that in rats given chronic CIT, R-citalopram must be continuously present to attenuate the effect of S-citalopram on extracellular 5-HT. We found that brain and plasma levels of ESCIT in rats given CIT for 13 days were approximately 40–60% lower than at 2 days. Similar findings were reported by Cremers et al (2000b), who measured plasma levels of CIT at various times after the continuous infusion of 20 mg kg−1 day−1 CIT through subcutaneous osmotic pumps. Although no statistical analysis was presented, their data indicated that plasma levels of the drug after 15 day's infusion were about 50% lower than at 3 days. These findings raise the possibility that chronic infusion did not enhance the effect of CIT on extracellular 5-HT due to the decrease in brain levels of S-citalopram. However, further studies are necessary to clarify the role of R-citalopram in the long-term effects of CIT on extracellular 5-HT.
The effects of ESCIT and CIT on extracellular 5-HT were enhanced by the selective 5-HT1A receptor antagonist WAY100,635. It is unlikely that pharmacokinetic interactions accounted for the greater effect of these drugs in rats receiving WAY100,635 because it did not affect brain availability of CIT (Grignaschi et al., 1998; Cremers et al., 2000a). In addition, 2.5 mg kg−1 ESCIT and 5 mg kg−1 CIT evoked a maximal increase in extracellular 5-HT, so under these experimental conditions, the contribution of increased ESCIT levels to the drug response would have been minimal. Thus, in accordance with previous studies (Hjorth, 1993; Gartside et al., 1995; Invernizzi et al., 1996; 1997; Gobert et al., 1997; Dawson et al., 2000; Hervas et al., 2000), WAY100,635 facilitates the effect of ESCIT and CIT on extracellular 5-HT, presumably by preventing the activation of 5-HT1A receptors by SSRIs through an increased endogenous 5-HT tone in the dorsal raphe. The effect of WAY100,635 was similar in rats infused with vehicle and in those receiving ESCIT or CIT for 2 or 13 days. Thus, the indirect activation of 5-HT1A autoreceptors, which restrain the effect of ESCIT and CIT on 5-HT release, is maintained after chronic treatment. Previous studies have in fact shown that blockade of 5-HT1A receptors enhances the effect of chronic CIT treatment on extracellular 5-HT (Arborelius et al., 1996; Gundlah et al., 1997).
In previous studies (Hjorth & Sharp, 1991; Invernizzi et al., 1994; 1996), 8-OH-DPAT (0.025 mg kg−1) reduced extracellular 5-HT levels in the prefrontal cortex of rats infused chronically with saline. This inhibitory effect of 8-OH-DPAT on 5-HT release was significantly attenuated in rats infused with ESCIT and CIT for 13 days. Similar results were obtained in rats injected with 10 mg kg−1 i.p. CIT twice daily for 14 days (Invernizzi et al., 1994) and challenged with 0.025 mg kg−1 8-OH-DPAT 24 h after the last dose of the chronic schedule. Studies with other SSRIs, in particular fluoxetine, confirmed that 5-HT1A receptors are desensitized by chronic treatment and coadministration of the 5-HT1A receptor antagonists WAY100,635 or pindolol prevented the desensitization of 5-HT1A receptors induced by the SSRI alone (Dawson et al., 2000; 2002; Hervas et al., 2001). These results are consistent with electrophysiological evidence that the function of 5-HT1A receptors of the dorsal raphe is reduced by chronic treatment with CIT and other SSRI (Blier & De Montigny, 1983; Blier et al., 1984; Chaput et al., 1986), and suggest that chronic treatment with ESCIT and CIT desensitizes 5-HT1A receptors controlling the release of 5-HT in the prefrontal cortex. Other microdialysis studies failed to confirm the desensitization of 5-HT1A receptors in rats given chronic CIT (Auerbach & Hjorth, 1995). As already discussed by Auerbach & Hjorth (1995), the contrasting results may be related to the use of different experimental conditions, and in particular to anaesthesia. In fact, the studies reporting the desensitization of 5-HT1A autoreceptors after chronic SSRIs were conducted in awake rats (Invernizzi et al., 1994; 1996; Dawson et al., 2000; Hervas et al., 2001), whereas failure to detect desensitization was reported in anaesthetized rats (Hjorth & Auerbach, 1994; Auerbach & Hjorth, 1995).
In line with previous studies (Invernizzi et al., 1994; Arborelius et al., 1996), 24 h after removal of the osmotic pump, when plasma and brain concentrations of S-citalopram were below the limit of quantification of the analytical procedure, the basal extracellular levels of 5-HT in the prefrontal cortex of rats given ESCIT for 13 days were not significantly different from PBS-treated rats. The fact that 5-HT1A autoreceptors were desensitized at this same interval suggests that the hypofunction of 5-HT1A autoreceptors is not sufficient to increase extracellular 5-HT. Accordingly, blockade of 5-HT1A receptors with WAY100,635 had no effect on extracellular 5-HT (Gobert et al., 1997; Dawson & Nguyen, 1998).
It may appear surprising that the selective 5-HT1A receptor antagonist WAY100,635 is able to enhance the effect of ESCIT and CIT on extracellular 5-HT in spite of the desensitization of 5-HT1A receptors. This raises the question of the relevance of 5-HT1A receptor desensitization for the enhancement of extracellular 5-HT after chronic treatment with SSRIs. It may be expected that WAY100,635 would reduce or have no effect on the SSRI-induced increase of extracellular 5-HT if the 5-HT1A receptors were desensitized. In actual fact, WAY100,635 enhanced extracellular 5-HT in the prefrontal cortex of rats receiving chronic ESCIT or CIT. The persistent effect of WAY100,635 confirms previous data (Arborelius et al., 1996; Gundlah et al., 1997; Hervas et al., 2001), and indicates that chronic SSRIs only partially desensitizes 5-HT1A receptors. In accordance with this interpretation, the blunted response to low doses of 8-OH-DPAT on 5-HT release, synthesis and firing activity of 5-HT neurons in rats given chronic SSRI is reinstated by increasing the dose of the agonist (Chaput et al., 1986; Bohmaker et al., 1993; Invernizzi et al., 1994; Jolas et al., 1994; Rutter et al., 1994; Cremers et al., 2000b). Taken together, these findings indicate that although 5-HT1A receptors controlling 5-HT release in the prefrontal cortex are desensitized, they can still limit the increase of extracellular 5-HT after chronic administration of ESCIT and CIT. Therefore, it appears that 5-HT1A autoreceptor desensitization plays a minor role in enhancing the effect of chronic SSRI on extracellular 5-HT. It remains to be established whether other adaptive changes occurring after chronic treatment with SSRIs such as the desensitization of terminal 5-HT1B receptors or of the NMDA receptors controlling 5-HT release (Nowak et al., 1996; Pallotta et al., 2001) contribute to the enhancement of extracellular 5-HT.
In conclusion, the present results show that chronic ESCIT treatment desensitizes 5-HT1A receptors, controlling the release of 5-HT in the prefrontal cortex and enhances the effect of the drug on extracellular 5-HT. They also indicate that R-citalopram may have negative effects on the ability of chronic ESCIT to enhance extracellular 5-HT. Given the importance of the increase in serotonergic neurotransmission for the antidepressant effect of SSRIs (Salomon et al., 1993), these results suggest that the addition of 5-HT1A antagonists may help to improve the antidepressant effect of ESCIT.
Acknowledgments
IC and SA contributed equally to this work. This work was supported by H. Lundbeck A/S (Denmark). We are grateful to Pharmacia (Nerviano, Italy) for the generous gift of WAY100,635 and to J. Baggott for language editing.
Abbreviations
- ANOVA
analysis of variance
- CIT
citalopram
- CSF
cerebrospinal fluid
- [11C]DASB, [11C]N,N
dimethyl-2-(2-amino-4-cyanophenylthio)benzylamine
- ESCIT
escitalopram
- HPLC
high-performance liquid chromatography
- 5-HT
5-hydroxytryptamine
- 8-OH-DPAT
8-hydroxy-2-(di-n-propylamino)tetralin
- PBS
phosphate-buffered saline
- SAL
saline
- SSRIs
selective serotonin reuptake inhibitors
- WAY100,635
N-[2-[methoxyphenyl)-1-piperazinyl]ethyl]-N-(2-pyridinyl) cyclohexane carboxamide trihydrochloride
References
- ADELL A., ARTIGAS F. Differential effects of clomipramine given locally or systemically on extracellular 5-hydroxytryptamine in raphe nuclei and frontal cortex. An in vivo brain microdialysis study. Naunyn Schmiedebergs Arch. Pharmacol. 1991;343:237–244. doi: 10.1007/BF00251121. [DOI] [PubMed] [Google Scholar]
- ARBORELIUS L., NOMIKOS G.G., HERTEL P., SALMI P., GRILLNER P., HOOK B.B., HACKSELL U., SVENSSON T.H. The 5-HT1A receptor antagonist (S)-UH-301 augments the increase in extracellular concentrations of 5-HT in the frontal cortex produced by both acute and chronic treatment with citalopram. Naunyn Schmiedebergs Arch. Pharmacol. 1996;353:630–640. doi: 10.1007/BF00167182. [DOI] [PubMed] [Google Scholar]
- ARTIGAS F., PEREZ V., ALVAREZ E. Pindolol induces a rapid improvement of depressed patients treated with serotonin reuptake inhibitors. Arch. Gen. Psychiatry. 1994;51:248–251. doi: 10.1001/archpsyc.1994.03950030084009. [DOI] [PubMed] [Google Scholar]
- AUERBACH S.B., HJORTH S. Effect of chronic administration of the selective serotonin (5-HT) uptake inhibitor citalopram on extracellular 5-HT and apparent autoreceptor sensitivity in rat forebrain in vivo. Naunyn Schmiedebergs Arch. Pharmacol. 1995;352:597–606. doi: 10.1007/BF00171317. [DOI] [PubMed] [Google Scholar]
- BEL N., ARTIGAS F. Chronic treatment with fluvoxamine increases extracellular serotonin in frontal cortex but not in raphe nuclei. Synapse. 1993;15:243–245. doi: 10.1002/syn.890150310. [DOI] [PubMed] [Google Scholar]
- BERMAN R.M., DARNELL A.M., MILLER H.L., ANAND A., CHARNEY D.S. Effect of pindolol in hastening response to fluoxetine in the treatment of major depression: a double-blind, placebo-controlled trial. Am. J. Psychiatry. 1997;154:37–43. doi: 10.1176/ajp.154.1.37. [DOI] [PubMed] [Google Scholar]
- BLIER P., BERGERON R. Effectiveness of pindolol with selected antidepressant drugs in the treatment of major depression. J. Clin. Psychopharmacol. 1995;15:217–222. doi: 10.1097/00004714-199506000-00011. [DOI] [PubMed] [Google Scholar]
- BLIER P., DE MONTIGNY C. Electrophysiological investigations on the effect of repeated zimelidine administration on serotonergic neurotransmission in the rat. J. Neurosci. 1983;3:1270–1278. doi: 10.1523/JNEUROSCI.03-06-01270.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BLIER P., DE MONTIGNY C., TARDIF D. Effects of the two antidepressant drugs mianserin and indalpine on the serotonergic system: single-cell studies in the rat. Psychopharmacology (Berl) 1984;84:242–249. doi: 10.1007/BF00427453. [DOI] [PubMed] [Google Scholar]
- BOHMAKER K., EISON A.S., YOCCA F.D., MELLER E. Comparative effects of chronic 8-OH-DPAT, gepirone and ipsapirone treatment on the sensitivity of somatodendritic 5-HT1A autoreceptors. Neuropharmacology. 1993;32:527–534. doi: 10.1016/0028-3908(93)90048-8. [DOI] [PubMed] [Google Scholar]
- BORDET R., THOMAS P., DUPUIS B. Effect of pindolol on onset of action of paroxetine in the treatment of major depression: intermediate analysis of a double-blind, placebo-controlled trial. Reseau de Recherche et d'Experimentation Psychopharmacologique. Am. J. Psychiatry. 1998;155:1346–1351. doi: 10.1176/ajp.155.10.1346. [DOI] [PubMed] [Google Scholar]
- BURKE W.J., GERGEL I., BOSE A. Fixed-dose trial of the single isomer SSRI escitalopram in depressed outpatients. J. Clin. Psychiatry. 2002;63:331–336. doi: 10.4088/jcp.v63n0410. [DOI] [PubMed] [Google Scholar]
- CHAPUT Y., DE MONTIGNY C., BLIER P. Effects of a selective 5-HT reuptake blocker, citalopram, on the sensitivity of 5-HT autoreceptors: electrophysiological studies in the rat brain. Naunyn Schmiedebergs Arch. Pharmacol. 1986;333:342–348. doi: 10.1007/BF00500007. [DOI] [PubMed] [Google Scholar]
- CREMERS T.I., DE BOER P., LIAO Y., BOSKER F.J., DEN BOER J.A., WESTERINK B.H., WIKSTROM H.V. Augmentation with a 5-HT(1A), but not a 5-HT(1B) receptor antagonist critically depends on the dose of citalopram. Eur. J. Pharmacol. 2000a;397:63–74. doi: 10.1016/s0014-2999(00)00247-8. [DOI] [PubMed] [Google Scholar]
- CREMERS T.I., SPOELSTRA E.N., DE BOER P., BOSKER F.J., MORK A., DEN BOER J.A., WESTERINK B.H., WIKSTROM H.V. Desensitisation of 5-HT autoreceptors upon pharmacokinetically monitored chronic treatment with citalopram. Eur. J. Pharmacol. 2000b;397:351–357. doi: 10.1016/s0014-2999(00)00308-3. [DOI] [PubMed] [Google Scholar]
- DAWSON L.A., NGUYEN H.Q. Effects of 5-HT1A receptor antagonists on fluoxetine-induced changes in extracellular serotonin concentrations in rat frontal cortex. Eur. J. Pharmacol. 1998;345:41–46. doi: 10.1016/s0014-2999(97)01580-x. [DOI] [PubMed] [Google Scholar]
- DAWSON L.A., NGUYEN H.Q., SMITH D.I., SCHECHTER L.E. Effects of chronic fluoxetine treatment in the presence and absence of (+/−)pindolol: a microdialysis study. Br. J. Pharmacol. 2000;130:797–804. doi: 10.1038/sj.bjp.0703378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DAWSON L.A., NGUYEN H.Q., SMITH D.L., SCHECHTER L.E. Effect of chronic fluoxetine and WAY-100635 treatment on serotonergic neurotransmission in the frontal cortex. J. Psychopharmacol. 2002;16:145–152. doi: 10.1177/026988110201600205. [DOI] [PubMed] [Google Scholar]
- DREMENCOV E., GUR E., LERER B., NEWMAN M.E. Subchronic fluoxetine administration to rats: effects on 5-HT autoreceptor activity as measured by in vivo microdialysis. Eur. Neuropsychopharmacol. 2000;10:229–236. doi: 10.1016/s0924-977x(00)00078-x. [DOI] [PubMed] [Google Scholar]
- DREVETS W.C. Functional neuroimaging studies of depression: the anatomy of melancholia. Annu. Rev. Med. 1998;49:341–361. doi: 10.1146/annurev.med.49.1.341. [DOI] [PubMed] [Google Scholar]
- FORSTER E.A., CLIFFE I.A., BILL D.J., DOVER G.M., JONES D., REILLY Y., FLETCHER A. A pharmacological profile of the selective silent 5-HT1A receptor antagonist WAY-100635. Eur. J. Pharmacol. 1995;281:81–88. doi: 10.1016/0014-2999(95)00234-c. [DOI] [PubMed] [Google Scholar]
- FULLER R.W. Uptake inhibitors increase extracellular serotonin concentration measured by brain microdialysis. Life Sci. 1994;55:163–167. doi: 10.1016/0024-3205(94)00876-0. [DOI] [PubMed] [Google Scholar]
- GARATTINI S., SAMANIN R. Biochemical hypotheses on antidepressant drugs: a guide for clinicians or a toy for pharmacologists. Psychol. Med. 1988;18:287–304. doi: 10.1017/s0033291700007844. [DOI] [PubMed] [Google Scholar]
- GARDIER A.M., MALAGIE I., TRILLAT A.C., JACQUOT C., ARTIGAS F. Role of 5-HT1A autoreceptors in the mechanism of action of serotoninergic antidepressant drugs: recent findings from in vivo microdialysis studies. Fundam. Clin. Pharmacol. 1996;10:16–27. doi: 10.1111/j.1472-8206.1996.tb00145.x. [DOI] [PubMed] [Google Scholar]
- GARTSIDE S.E., UMBERS V., HAJOS M., SHARP T. Interaction between a selective 5-HT1A receptor antagonist and an SSRI in vivo: effects on 5-HT cell firing and extracellular 5-HT. Br. J. Pharmacol. 1995;115:1064–1070. doi: 10.1111/j.1476-5381.1995.tb15919.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GOBERT A., RIVET J.M., CISTARELLI L., MILLAN M.J. Potentiation of the fluoxetine-induced increase in dialysate levels of serotonin (5-HT) in the frontal cortex of freely moving rats by combined blockade of 5-HT1A and 5-HT1B receptors with WAY 100,635 and GR 127,935. J. Neurochem. 1997;68:1159–1163. doi: 10.1046/j.1471-4159.1997.68031159.x. [DOI] [PubMed] [Google Scholar]
- GRIGNASCHI G., INVERNIZZI R.W., FANELLI E., FRACASSO C., CACCIA S., SAMANIN R. Citalopram-induced hypophagia is enhanced by blockade of 5-HT(1A) receptors: role of 5-HT(2C) receptors. Br. J. Pharmacol. 1998;124:1781–1787. doi: 10.1038/sj.bjp.0702028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GUNDLAH C., HJORTH S., AUERBACH S.B. Autoreceptor antagonists enhance the effect of the reuptake inhibitor citalopram on extracellular 5-HT: this effect persists after repeated citalopram treatment. Neuropharmacology. 1997;36:475–482. doi: 10.1016/s0028-3908(97)00052-x. [DOI] [PubMed] [Google Scholar]
- HERVAS I., QUEIROZ C.M., ADELL A., ARTIGAS F. Role of uptake inhibition and autoreceptor activation in the control of 5-HT release in the frontal cortex and dorsal hippocampus of the rat. Br. J. Pharmacol. 2000;130:160–166. doi: 10.1038/sj.bjp.0703297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HERVAS I., VILARO M.T., ROMERO L., SCORZA M.C., MENGOD G., ARTIGAS F. Desensitization of 5-HT(1A) autoreceptors by a low chronic fluoxetine dose effect of the concurrent administration of WAY-100635. Neuropsychopharmacology. 2001;24:11–20. doi: 10.1016/S0893-133X(00)00175-5. [DOI] [PubMed] [Google Scholar]
- HJORTH S. Serotonin 5-HT1A autoreceptor blockade potentiates the ability of the 5-HT reuptake inhibitor citalopram to increase nerve terminal output of 5-HT in vivo: a microdialysis study. J. Neurochem. 1993;60:776–779. doi: 10.1111/j.1471-4159.1993.tb03217.x. [DOI] [PubMed] [Google Scholar]
- HJORTH S., AUERBACH S.B. Lack of 5-HT1A autoreceptor desensitization following chronic citalopram treatment, as determined by in vivo microdialysis. Neuropharmacology. 1994;33:331–334. doi: 10.1016/0028-3908(94)90062-0. [DOI] [PubMed] [Google Scholar]
- HJORTH S., SHARP T. Effect of the 5-HT1A receptor agonist 8-OH-DPAT on the release of 5-HT in dorsal and median raphe-innervated rat brain regions as measured by in vivo microdialysis. Life Sci. 1991;48:1779–1786. doi: 10.1016/0024-3205(91)90216-x. [DOI] [PubMed] [Google Scholar]
- HYTTEL J. Neurochemical characterization of a new potent and selective serotonin uptake inhibitor: Lu 10-171. Psychopharmacology (Berl) 1977;51:225–233. doi: 10.1007/BF00431629. [DOI] [PubMed] [Google Scholar]
- HYTTEL J., BOGESO K.P., PERREGAARD J., SANCHEZ C. The pharmacological effect of citalopram residues in the (S)-(+)-enantiomer. J. Neural. Transm. Gen. Sect. 1992;88:157–160. doi: 10.1007/BF01244820. [DOI] [PubMed] [Google Scholar]
- INVERNIZZI R., BELLI S., SAMANIN R. Citalopram's ability to increase the extracellular concentrations of serotonin in the dorsal raphe prevents the drug's effect in the frontal cortex. Brain Res. 1992;584:322–324. doi: 10.1016/0006-8993(92)90914-u. [DOI] [PubMed] [Google Scholar]
- INVERNIZZI R., BRAMANTE M., SAMANIN R. Chronic treatment with citalopram facilitates the effect of a challenge dose on cortical serotonin output: role of presynaptic 5-HT1A receptors. Eur. J. Pharmacol. 1994;260:243–246. doi: 10.1016/0014-2999(94)90344-1. [DOI] [PubMed] [Google Scholar]
- INVERNIZZI R., BRAMANTE M., SAMANIN R. Role of 5-HT1A receptors in the effects of acute chronic fluoxetine on extracellular serotonin in the frontal cortex. Pharmacol. Biochem. Behav. 1996;54:143–147. doi: 10.1016/0091-3057(95)02159-0. [DOI] [PubMed] [Google Scholar]
- INVERNIZZI R., VELASCO C., BRAMANTE M., LONGO A., SAMANIN R. Effect of 5-HT1A receptor antagonists on citalopram-induced increase in extracellular serotonin in the frontal cortex, striatum and dorsal hippocampus. Neuropharmacology. 1997;36:467–473. doi: 10.1016/s0028-3908(97)00060-9. [DOI] [PubMed] [Google Scholar]
- JOLAS T., HAJ-DAHMANE S., KIDD E.J., LANGLOIS X., LANFUMEY L., FATTACCINI C.M., VANTALON V., LAPORTE A.M., ADRIEN J., GOZLAN H. Central pre- and postsynaptic 5-HT1A receptors in rats treated chronically with a novel antidepressant, cericlamine. J. Pharmacol. Exp. Ther. 1994;268:1432–1443. [PubMed] [Google Scholar]
- KOSEL M., EAP C.B., AMEY M., BAUMANN P. Analysis of the enantiomers of citalopram and its demethylated metabolites using chiral liquid chromatography. J. Chromatogr. B. Biomed. Sci. Appl. 1998;719:234–238. doi: 10.1016/s0378-4347(98)00384-3. [DOI] [PubMed] [Google Scholar]
- KREISS D.S., LUCKI I. Effects of acute and repeated administration of antidepressant drugs on extracellular levels of 5-hydroxytryptamine measured in vivo. J. Pharmacol. Exp. Ther. 1995;274:866–876. [PubMed] [Google Scholar]
- LEPOLA U.M., LOFT H., REINES E.H. Escitalopram (10-20 mg/day) is effective and well tolerated in a placebo-controlled study in depression in primary care. Int. Clin. Psychopharmacol. 2003;18:211–217. doi: 10.1097/00004850-200307000-00003. [DOI] [PubMed] [Google Scholar]
- MEYER J.H., WILSON A.A., GINOVART N., GOULDING V., HUSSEY D., HOOD K., HOULE S. Occupancy of serotonin transporters by paroxetine and citalopram during treatment of depression: a [(11)C]DASB PET imaging study. Am. J. Psychiatry. 2001;158:1843–1849. doi: 10.1176/appi.ajp.158.11.1843. [DOI] [PubMed] [Google Scholar]
- MILNE R.J., GOA K.L. Citalopram. A review of its pharmacodynamic and pharmacokinetic properties, and therapeutic potential in depressive illness. Drugs. 1991;41:450–477. doi: 10.2165/00003495-199141030-00008. [DOI] [PubMed] [Google Scholar]
- MONTGOMERY S.A., LOFT H., SANCHEZ C., REINES E.H., PAPP M. Escitalopram (S-enantiomer of citalopram): clinical efficacy and onset of action predicted from a rat model. Pharmacol. Toxicol. 2001;88:282–286. doi: 10.1034/j.1600-0773.2001.d01-118.x. [DOI] [PubMed] [Google Scholar]
- MORK A., KREILGAARD M., SANCHEZ C. The R-enantiomer of citalopram counteracts escitalopram-induced increase in extracellular 5-HT in the frontal cortex of freely moving rats. Neuropharmacology. 2003;45:167–173. doi: 10.1016/s0028-3908(03)00138-2. [DOI] [PubMed] [Google Scholar]
- NOWAK G., LI Y., PAUL I.A. Adaptation of cortical but not hippocampal NMDA receptors after chronic citalopram treatment. Eur. J. Pharmacol. 1996;295:75–85. doi: 10.1016/0014-2999(95)00585-4. [DOI] [PubMed] [Google Scholar]
- OWENS M.J., KNIGHT D.L., NEMEROFF C.B. Second-generation SSRIs: human monoamine transporter binding profile of escitalopram and R-fluoxetine. Biol. Psychiatry. 2001;50:345–350. doi: 10.1016/s0006-3223(01)01145-3. [DOI] [PubMed] [Google Scholar]
- PALLOTTA M., SEGIETH J., SADIDEEN F., WHITTON P.S. Repeated but not acute clomipramine decreases the effect of N-methyl-D-aspartate receptor activation on serotonergic transmission between the raphe nuclei and frontal cortex. Neuropharmacology. 2001;41:294–300. doi: 10.1016/s0028-3908(01)00078-8. [DOI] [PubMed] [Google Scholar]
- PAXINOS G., WATSON C. The Rat Brain in Stereotaxic Coordinates. Sidney: Academic Press; 1982. [Google Scholar]
- POLLOCK B.G. Citalopram: a comprehensive review. Expert Opin. Pharmacother. 2001;2:681–698. doi: 10.1517/14656566.2.4.681. [DOI] [PubMed] [Google Scholar]
- POZZI L., INVERNIZZI R., GARAVAGLIA C., SAMANIN R. Fluoxetine increases extracellular dopamine in the prefrontal cortex by a mechanism not dependent on serotonin: a comparison with citalopram. J. Neurochem. 1999;73:1051–1057. doi: 10.1046/j.1471-4159.1999.0731051.x. [DOI] [PubMed] [Google Scholar]
- ROBINSON T.E., WHISHAW I.Q. Normalization of extracellular dopamine in striatum following recovery from a partial unilateral 6-OHDA lesion of the substantia nigra: a microdialysis study in freely moving rats. Brain Res. 1988;450:209–224. doi: 10.1016/0006-8993(88)91560-0. [DOI] [PubMed] [Google Scholar]
- ROCHAT B., AMEY M., BAUMANN P. Analysis of enantiomers of citalopram and its demethylated metabolites in plasma of depressive patients using chiral reverse-phase liquid chromatography. Ther. Drug. Monit. 1995;17:273–279. doi: 10.1097/00007691-199506000-00011. [DOI] [PubMed] [Google Scholar]
- ROMERO L., ARTIGAS F. Preferential potentiation of the effects of serotonin uptake inhibitors by 5-HT1A receptor antagonists in the dorsal raphe pathway: role of somatodendritic autoreceptors. J. Neurochem. 1997;68:2593–2603. doi: 10.1046/j.1471-4159.1997.68062593.x. [DOI] [PubMed] [Google Scholar]
- RUTTER J.J., GUNDLAH C., AUERBACH S.B. Increase in extracellular serotonin produced by uptake inhibitors is enhanced after chronic treatment with fluoxetine. Neurosci. Lett. 1994;171:183–186. doi: 10.1016/0304-3940(94)90635-1. [DOI] [PubMed] [Google Scholar]
- SALOMON R.M., MILLER H.L., DELGADO P.L., CHARNEY D. The use of tryptophan depletion to evaluate central serotonin function in depression and other neuropsychiatric disorders. Int. Clin. Psychopharmacol. 1993;8 Suppl 2:41–46. doi: 10.1097/00004850-199311002-00006. [DOI] [PubMed] [Google Scholar]
- SANCHEZ C., BERGQVIST P.B., BRENNUM L.T., GUPTA S., HOGG S., LARSEN A., WIBORG O. Escitalopram, the S-(+)-enantiomer of citalopram, is a selective serotonin reuptake inhibitor with potent effects in animal models predictive of antidepressant and anxiolytic activities. Psychopharmacology (Berl) 2003;167:353–362. doi: 10.1007/s00213-002-1364-z. [DOI] [PubMed] [Google Scholar]
- SHARP T., HJORTH S.In vivo neurochemical studies of 5-HT1A autoreceptor function 5-Hydroxytryptamine, CNS Receptors and Brain Function 1992New York: Pergamon Press; 297–323.ed., Bradley, P.B., Handley, S.L., Coopez, S.J., Key, B.J., Barnes, N.M. & Coote, J.H. pp [Google Scholar]
- SIDHU J., PRISKORN M., POULSEN M., SEGONZAC A., GROLLIER G., LARSEN F. Steady-state pharmacokinetics of the enantiomers of citalopram and its metabolites in humans. Chirality. 1997;9:686–692. doi: 10.1002/(SICI)1520-636X(1997)9:7<686::AID-CHIR9>3.0.CO;2-5. [DOI] [PubMed] [Google Scholar]
- TOME M.B., ISAAC M.T., HARTE R., HOLLAND C. Paroxetine and pindolol: a randomized trial of serotonergic autoreceptor blockade in the reduction of antidepressant latency. Int. Clin. Psychopharmacol. 1997;12:81–89. [PubMed] [Google Scholar]
- WADE A., MICHAEL LEMMING O., BANG HEDEGAARD K. Escitalopram 10 mg/day is effective and well tolerated in a placebo-controlled study in depression in primary care. Int. Clin. Psychopharmacol. 2002;17:95–102. doi: 10.1097/00004850-200205000-00001. [DOI] [PubMed] [Google Scholar]
- WAUGH J., GOA K.L. Escitalopram: a review of its use in the management of major depressive and anxiety disorders. CNS Drugs. 2003;17:343–362. doi: 10.2165/00023210-200317050-00004. [DOI] [PubMed] [Google Scholar]
- ZANARDI R., ARTIGAS F., FRANCHINI L., SFORZINI L., GASPERINI M., SMERALDI E., PEREZ J. How long should pindolol be associated with paroxetine to improve the antidepressant response. J. Clin. Psychopharmacol. 1997;17:446–450. doi: 10.1097/00004714-199712000-00002. [DOI] [PubMed] [Google Scholar]
- ZANARDI R., FRANCHINI L., GASPERINI M., LUCCA A., SMERALDI E., PEREZ J. Faster onset of action of fluvoxamine in combination with pindolol in the treatment of delusional depression: a controlled study. J. Clin. Psychopharmacol. 1998;18:441–446. doi: 10.1097/00004714-199812000-00004. [DOI] [PubMed] [Google Scholar]
- ZHENG Z., JAMOUR M., KLOTZ U. Stereoselective HPLC-assay for citalopram and its metabolites. Ther. Drug Monit. 2000;22:219–224. doi: 10.1097/00007691-200004000-00014. [DOI] [PubMed] [Google Scholar]