Abstract
Two isoforms of the rat prostaglandin E2 receptor, rEP3α-R and rEP3β-R, differ only in their C-terminal domain. To analyze the function of the rEP3-R C-terminal domain in agonist induced desensitization, a cluster of Ser/Thr residues in the C-terminal domain of the rEP3α-R was mutated to Ala and both isoforms and the receptor mutant (rEP3α-ST341-349A-R) were stably expressed in HEK293 cells.
All rEP3-R receptors showed a similar ligand-binding profile. They were functionally coupled to Gi and reduced forskolin-induced cAMP-formation.
Repeated exposure of cells expressing the rEP3α-R isoform to PGE2 reduced the agonist induced inhibition of forskolin-stimulated cAMP-formation by 50% and led to internalization of the receptor to intracellular endocytotic vesicles. By contrast, Gi-response as well as plasma membrane localization of the rEP3β-R and the rEP3α-ST341-349A-R were not affected by prior agonist-stimulation.
Agonist-stimulation of HEK293-rEP3α-R cells induced a time- and dose-dependent phosphorylation of the receptor most likely by G protein-coupled receptor kinases and not by protein kinase A or protein kinase C. By contrast, upon agonist-stimulation the rEP3β-R was not phosphorylated and the rEP3α-ST341-349A-R was phosphorylated only weakly.
These results led to the hypothesis that agonist-induced desensitization of the rEP3α-R isoform is mediated most likely by a GRK-dependent phosphorylation of Ser/Thr residues 341–349. Phosphorylation then initiates uncoupling of the receptor from Gi protein and receptor internalization.
Keywords: Prostaglandin receptor, receptor isoform, homologous desensitization, arrestin, internalization, G protein-coupled receptor kinases
Introduction
Prostaglandin E2 (PGE2) regulates a variety of physiological and pathophysiological processes (Narumiya et al., 1999; Kobayashi & Narumiya, 2002 and references within). It binds to specific PGE2-receptors (EP-R), which belong to the class of G protein-coupled ectoreceptors (GPCR) with seven transmembrane domains. Four subtypes of EP-R have been described, which differ in their affinity to synthetic ligands and their G protein-coupling specificity: EP1-R is linked to Gq and to an as yet not identified G protein and mediates an increase in cytosolic Ca2+ concentration most likely by activation of Ca2+-channels; EP2-R and EP4-R couple to Gs and increase intracellular cAMP, while EP3-R couples to Gi and decreases hormone-stimulated cAMP formation (Coleman et al., 1994). These receptors display an overall sequence identity of about 40% with the putative transmembrane domains being most conserved (Coleman et al., 1994).
EP3-R is expressed in a wide range of tissues. It has been shown to be involved in acid-induced duodenal bicarbonate secretion and maintenance of mucosal integrity (Takeuchi et al., 1999), PGE2-regulated tumor-associated angiogenesis and tumor growth (Amano et al., 2003) and it is the primary receptor conferring LPS- and IL-1β-stimulated fever generation (Ushikubi et al., 1998).
There exist EP3-R isoforms, which differ only in their C-terminal domain and are generated by alternative splicing (Pierce & Regan, 1998). The expression pattern of EP3-R isoforms differs between various cell types. In rat sensory neurons, which release neuropeptides upon PGE2-stimulation via EP3-R, only the EP3γ-R but not the EP3α-R or the EP3β-R isoform is expressed (Southall & Vasko, 2001). In human tissues the EP3V-R and EP3RVI-R isoforms were found to be highly expressed in the uterus only (Kotani et al., 2000).
It has been reported that EP3-R isoforms differ in their ability to activate Gq and Gs in addition to Gi (Namba et al., 1993) and in their degree of constitutive, agonist-independent Gi activation (Jin et al., 1997). In addition, the different C-terminal ends of EP3-R isoforms are likely to confer different agonist-induced desensitization characteristics. Of the two mouse EP3-R isoforms, mEP3α-R and mEP3β-R, only the mEP3α-R underwent agonist-induced desensitization and internalization (Negishi et al., 1993). The importance of the C-terminal domain for EP3-R desensitization was also shown in studies using a receptor hybrid consisting of the N-terminal main portion of the nondesensitizable rEP3β-R up to the end of the seventh transmembrane domain and the C-terminal portion of the Gs-coupled, desensitizable human EP4-R (hEP4-R) (Neuschäfer-Rube et al., 1997a). For this receptor hybrid, which was exclusively coupled to Gi (Neuschäfer-Rube et al., 1997b), the C-terminal domain of EP4-R was necessary and sufficient to confer agonist-induced receptor desensitization.
Based on a model developed from studies of the β2-adrenergic receptor, desensitization of G protein-coupled receptors is initiated by receptor phosphorylation, which facilitates binding of the adaptor protein β-arrestin to the ligand-activated receptor. β-Arrestin binding to the receptor physically prevents further activation of G proteins by the ligand-occupied receptor, a phenomenon called ‘uncoupling' (Pitcher et al., 1992). Receptor phosphorylation can be catalyzed either by second messenger-activated kinases like protein kinase A (PKA) or protein kinase C (PKC) or by G protein coupled receptor kinases (GRK) (Lohse, 1993; Premont et al., 1995).
Besides uncoupling, ligand-induced receptor endocytosis, also called receptor internalization or sequestration, may further contribute to receptor desensitization. Receptor sequestration is believed to result from specific interactions of the agonist-occupied receptor with components of the endocytotic machinery (Ferguson, 2002 and references within). For many, GPCRs receptor sequestration is a phosphorylation dependent process. In these cases, binding of β-arrestin targets the agonist-activated and phosphorylated receptor to clathrin coated pits (Laporte et al., 1999). However, uncoupling and sequestration are not always associated. For example, mutation of phosphorylation sites in the N-formyl peptide receptor (Maestes et al., 1999) or suppression of m2-muscarinic acetylcholine receptor phosphorylation by dominant-negative GRK2K220R suppressed uncoupling but left sequestration unaffected (Pals-Rylaarsdam et al., 1995). On the other hand, mutation of defined receptor phosphorylation sites of the lutropin/chorion gonadotropin receptor hindered receptor internalization but had no effect on uncoupling from the G protein (Lazari et al., 1998).
In contrast to the rEP3β-R, the C-terminal domain of the rEP3α-R contains a cluster of five Ser/Thr residues that may serve as a target for agonist-induced phosphorylation. The current study analyzes the function of this Ser/Thr cluster in agonist-induced phosphorylation, uncoupling and internalization of rEP3α-R. For this purpose, Ser/Thr residues within this cluster were mutated to alanines. rEP3α-R, rEP3β-R and the rEP3α-ST341-349A-R mutant with or without a C-terminal GFP/CFP-tag were stably expressed in HEK293 cells and agonist-induced receptor phosphorylation, uncoupling and internalization were analyzed biochemically and by in vivo confocal laser scanning microscopy.
We found that upon agonist-stimulation only the rEP3α-R was phosphorylated, desensitized and internalized. Both uncoupling and internalization of the rEP3α-R were dependent on the presence of the Ser/Thr cluster in its C-terminal domain. Therefore, agonist-stimulated desensitization of rEP3α-R is most likely secondary to a GRK-mediated phosphorylation of Ser/Thr residues 341–349.
Methods
Plasmid construction
The cDNAs for rEP3α-R and rEP3β-R were cloned from rat hepatocytes (Neuschäfer-Rube et al., 1994; Boie et al., 1997) and inserted into the pcDNAI vector (Invitrogen, Karlsruhe). A FLAG-octapeptide sequence was inserted after the initiator methionine of both receptors as described previously (Neuschäfer-Rube et al., 1999), using pcDNAI-rEP3α-R as template for epitope tagging of rEP3α-R. cDNAs encoding FLAG-tagged receptors were subcloned into the expression vector pcDNA3 (Invitrogen). Mutations of Ser and Thr in the 341–349 cluster to Ala in rEP3α were generated by PCR-based site-directed mutagenesis with proof-reading Powerscript-Polymerase (PAN-Systems, Aichenbach; Germany) and the following program: 3′ 95°, 35 × (1′ 95°, 1′ 55°, 2′ 72°), 10′ 72°. A 420 bp 5′ fragment was generated from pcDNA3/FLAG-rEP3α-R using rEP3-f as forward primer and rEP3αmut-r as reverse primer (Table 1). A 190 bp 3′ fragment was produced with primers rEP3αmut-f and SP6-r (Table 1). The PCR products were fused by PCR with both overlapping fragments as template and the primers rEP3-f and SP6-r. The PCR product was digested with BamHI and XbaI and ligated into pcDNA3/FLAG-rEP3α-R, which was digested with the same enzymes generating plasmid pcDNA3/FLAG-rEP3α-ST341-349A-R. For generation of the cDNA for a rEP3β-R-GFP fusion protein, an AgeI site was inserted at the 3′ site of the FLAG-rEP3β-R cDNA by PCR using FLAG-rEP3-3-Eco-f as forward primer and rEP3β-Age-r as reverse primer. The resulting PCR-product was digested with EcoRI and AgeI and inserted in the EcoRI/AgeI site of pEGFP-N1 (BD Clontech, Heidelberg, Germany). For generation of the cDNAs for FLAG-rEP3α-R-CFP and FLAG-rEP3α-ST341-349A-R-CFP fusion proteins, an in-frame XbaI site was created 3′ of the respective cDNA by PCR as described above using rEP3α-XbaI-r as a reverse primer. The resulting PCR-product was digested with EcoRI and XbaI and inserted in frame in the EcoRI/XbaI site of pcDNA3-CFP. For generation of the cDNAs coding for FLAG-rEP3α-S355-361A-R-CFP or FLAG-rEP3α-ST341-361A-R fusion proteins, 468 bp PCR products were generated using the primer combination rEP3-f/rEP3αmut2-r and the plasmids pcDNA3/FLAG-rEP3α-R-CFP or pcDNA3/FLAG-rEP3α-ST341-349A-R-CFP as templates. PCR products were digested with BstEII and XbaI and cloned into the plasmid pcDNA3/FLAG-rEP3α-R-CFP, which was digested with the same enzymes. All cDNAs were sequenced to exclude that mutations other than the ones intended were introduced by PCR artifacts.
Table 1.
Sequence of the oligonucleotides employed for epitope-tagging and site-directed mutagenesis
| Primer | Sequence | Position |
|---|---|---|
| rEP3-f | 5′-TCTGGTGGTGACCTTTGCCTGCAACCTGGC | 769–798 |
| rEP3αmut-f | 5′-GACCACGCCAACTATGCTGCCGCCGCTGCCGCCT | 1130–1177 |
| TGCCCTGCCCAGGC | ||
| rEP3αmut-r | 5′-GCCTGGGCAGGGCAAGGCGGCAGCGGCGGCAGC | 1177–1130 |
| ATAGTTGGCGTGGTC | ||
| rEP3αmut2-r | CGGCGGTCTAGAGCTCTTTCTAGCTGGTCAGCCCACATC | 1213–1168 |
| AGGACTGCGGCGCCT | ||
| rEP3α-XbaI-r | 5′-CGGCGGTCTAGAGCTCTTTCTAGCTGGTCACTCCA | 1213–1190 |
| CAT | ||
| rEP3β-Age-r | 5′-CGGACCGGTGCTCCTTCCCTGGGGGAAGAAATTC | 1290–1268 |
| SP6-r | 5′-GCGACGTCTAGCATTTAGGTGACAC | pcDNA3: 1028–1004 |
Sequence is given in the 5′ to 3′ direction. Recognition sites for restriction enzymes are underlined. Mutations are shown in bold. Position refers to the position in the published sequence GenBank AccNo. E06014.
Cell culture and transfection
HEK293 cells were cultured in DMEM containing 10% (v v−1) FCS, penicillin (100 U ml−1) and streptomycin (100 μg ml−1). For generation of receptor expressing cell lines, HEK293 cells were seeded at a density of 1.5 × 106 cells 10 cm−1 diameter plate. After 24 h, cells were transfected with expression vectors for rEP3-R or rEP3-R-GFP/CFP fusion proteins using a modified calcium-phosphate method, including 5% bovine serum and 2.5 μg ml−1 25-hydroxycholesterol (Sigma, Heidelberg, Germany) in the transfection medium. Cells were selected with G-418 at 0.5 mg ml−1 in complete DMEM. Clonal cell lines were isolated by single-cell cloning and tested for receptor expression by PGE2 binding.
Membrane isolation and PGE2 binding assay
Transfected cells were scraped into a hypotonic homogenization buffer containing 50 mM Tris-HCl pH 7.5, 5 mM EDTA and 0.2 mM Pefabloc SC (Biomol, Hamburg, Germany), 10 μg ml−1 leupeptin and 10 μg ml−1 soybean trypsin-inhibitor as protease inhibitors. Cells were incubated on ice for 10 min and disrupted by vigorous vortexing. A crude membrane fraction was prepared by centrifugation of the homogenate at 35,000 × g for 20 min. The resulting pellet was suspended in binding buffer containing 25 mM MES/NaOH pH 6.2, 10 mM MgCl2 and 1 mM EDTA. Membranes were stored at −70°C. PGE2 binding was assayed by incubating membranes (20–50 μg protein) with 0.5–5 nM [3H]PGE2 and various concentrations of unlabeled PGE2 in 100 μl binding buffer for 1 h at 20°C. Nonspecific binding was determined in the presence of 25 μM PGE2. Free ligand was removed by rapid vacuum filtration through GF 52 filters (Schleicher & Schüll, Dassel, Germany). Filters were washed five times with 4 ml ice-cold binding buffer. Radioactivity retained on the filter was counted in 5 ml Rotiszint 22 (Roth, Karlsruhe, Germany). Binding constants were calculated by nonlinear regression analysis (LIGAND, Munson & Rodbard, 1980).
Cell surface ligand binding
Cells in 24-well plates (1 × 105 cells/well) were washed once with incubation buffer (HEPES buffered salt solution pH 7.4 containing 15 mM HEPES, 4.7 mM KCl, 140 mM NaCl, 1.2 mM KH2PO4, 11 mM glucose and 2.2 mM CaCl2) and then incubated for 2 h at 4°C with 100 μl 5 nM [3H]PGE2±10 μM PGE2 to determine nonspecific binding in the same buffer. Binding was performed at 4°C to inhibit ligand-induced internalization during the incubation period. Plates were washed three times with ice-cold incubation buffer and cell associated radioactivity was released by lysing cells in 400 μl 0.3 M NaOH, 1% (w v−1) SDS. The radioactivity in the cell lysates was counted in 5 ml Rotiszint 22.
cAMP-formation (desensitization assay)
Stably transfected HEK293 cells were cultured in 3.5 cm diameter plates to a density of 5 × 105 cells/plate. Cells were washed three times with 1 ml incubation buffer and then preincubated in 1 ml of the same buffer with or without 100 nM M&B28767 for 10 min. The agonist was removed by two washes with incubation buffer, followed by an acid wash with 50 mM glycine, 150 mM NaCl pH 3 for 1 min and two additional washes with incubation buffer. Cells were preincubated with 1 ml incubation buffer containing 1 mM IBMX at 37°C for 10 min. Then M&B28767 (1 μM) and forskolin (100 μM) were added in a volume of 10 μl buffer to the final concentration indicated. After incubation for 10 min, the reaction was stopped by removing the buffer and snap-freezing the cells in liquid nitrogen. Cells were lysed in 500 μl of 10 mM HCl containing 1 mM IBMX for 1 h at 4°C. The lysate was centrifuged at 25,000 × g and cAMP in the supernatant was quantified using a 125IcAMP assay kit by Amersham (Braunschweig, Germany).
Ligand internalization assay
Receptor expressing HEK293 cells grown in 24-well plates (1 × 105 cells/well) were washed three times with incubation buffer and preincubated in the same buffer for 10 min at 37°C with or without 100 nM M&B28767. The agonist was removed as described for the desensitization assay. Cells were then incubated for 2 h at 4°C with 100 μl 5 nM [3H]PGE2±10 μM PGE2 to determine non-specific binding in the same buffer. Cells were washed three times with ice-cold incubation buffer and cell-associated radioactivity was released by lysing cells in 150 μl/well 5% (w v−1) Triton X-100. The radioactivity in the cell lysates was counted in 5 ml Rotiszint 22.
Intact cell phosphorylation
Receptor expressing HEK293 cells in six-well plates were washed two times with phosphate-free DMEM and prelabeled for 60 min with 150 μCi ml−1 (32P)orthophosphate in 500 μl of the same medium containing 10% (v v−1) FCS, which had been dialyzed extensively against 10 mM HEPES buffer pH 7.5 containing 150 mM sodium chloride (cutoff 10–20 kDa). After cell labeling, various reagents in a volume of 500 μl in phosphate-free DMEM containing 10% (v v−1) dialyzed FCS were added for 10 min at 37°C unless otherwise indicated. Where indicated, cells were treated with 400 nM staurosporine 20 min before stimulation. After stimulation, medium was removed and the cells were washed once with ice cold 10 mM HEPES buffer pH 7.5 containing 150 mM sodium chloride before immunoprecipitation.
Immunoprecipitation
Cells grown in six-well plates at 5 × 105 well were scraped into 800 μl lysis buffer/well (1% (v v−1) Triton X-100, 0.05% (w v−1) SDS, 50 mM HEPES pH 7.5, 150 mM sodium chloride, 5 mM EDTA, 10 mM sodium fluoride, 10 mM sodium pyrophosphate and 0.2 mM Pefabloc SC, 10 μg ml−1 leupeptin and 10 μg ml−1 soybean trypsin-inhibitor) transferred to microfuge tubes and centrifuged at 4°C for 30 min at 35,000 × g to remove insoluble material. The supernatant was precleared for 60 min at 4°C with 100 μl 10% (v v−1) Sepharose 4B in lysis buffer containing 0.1% (w v−1) bovine serum albumin. The precleared supernatant was incubated for 4 h at 4°C with an immune complex of 1 μg of mAb α-GFP and 100 μl 10% (v v−1) Protein-G Sepharose FF in lysis buffer containing 0.2% (w v−1) bovine serum albumin, which was preformed by incubation for 60 min at 4°C. Immune complexes were collected by centrifugation and washed four times with ice cold lysis buffer. Bound receptor proteins were displaced from the immune complexes by incubating for 60 min at 37°C and 10 min at 60°C under vigorous shaking with Lämmli sample buffer containing 5% (v v−1) β-mercaptoethanol. The amount of receptor proteins loaded onto SDS–PAGE was equalized by parallel determination of cell surface ligand binding of receptor expressing cells as described above. Proteins were either transferred to PVDF membrane for Western blotting or gels were stained with Coomassie-blue and dried for PhosphorImager analysis. Phosphorylated bands were quantified using Quantity One (Bio-Rad, Madison, U.S.A.).
Western blotting
Membrane proteins of transfected (normalized for receptor content by PGE2-binding assay) or control cells treated with 100 nM M&B28767 for 0 or 5 min were solubilized in Lämmli sample buffer under reducing conditions for 30 min at 37°C and 15 min at 60°C with vigorous shaking, separated on 7.5% SDS–polyacrylamide gels and transferred onto PVDF membrane (Millipore, Bedford, U.S.A.). After blocking with 5% (w v−1) skim milk in TBS, 0.1% (v v−1) Tween-20 (TBS-T) GFP/CFP-tagged receptor proteins were detected by incubating immunoblots overnight with 0.4 μg ml−1 mAb α-GFP antibodies in 5% (w v−1) skim milk in TBS-T at 4°C followed by horseradish peroxidase-conjugated anti-mouse IgG (1 : 20,000 dilution, Dianova, Hamburg, Germany). Antigen–antibody complexes were visualized with the Super-Signal System (Pierce, Rockford, U.S.A.) according to the manufacturer's specifications.
Monitoring of agonist-induced rEP3-R-GFP/CFP translocation in living cells by confocal laser scanning microscopy (receptor internalization)
Confocal microscopy was performed using a Zeiss laser scanning microscope (LSM 510). FLAG-tagged rEP3α-R-CFP, rEP3α-ST341-349A-R-CFP and rEP3β-R-GFP expressing cells were grown on glass coverslips for 24 h and then transferred into a heated (37°C) microscope stage filled with 1 ml Krebs–HEPES buffer pH 7.4 and treated with 100 nM M&B28767 for 0–30 min. Images were collected sequentially using a single line excitation with optical sections less than 0.9 μM (GFP, lexc: 488 nm, LP filter: 505 nm; CFP, lexc: 458 nm, BP filter: 462–505 nm).
Materials
All materials were of analytical grade and from commercial sources. [3H]PGE2, 32Pi, Sepharose Cl-4B and Protein-G Sepharose FF were obtained from Amersham/Pharmacia (Freiburg, Germany), unlabeled PGE2 and IBMX were purchased from Calbiochem-Novabiochem (Bad Soden, Germany). M&B28767 was a generous gift from Rhone-Poulenc Rorer (Dagenham, GB). Cell culture media were obtained from Biochrom (Berlin, Germany). Primers (Table 1) were synthesized by NAPS (Göttingen, Germany) and MWG (Ebersberg, Germany). The antibody to GFP was from Roche (Mannheim, Germany). The plasmid pcDNA3-CFP was kindly supplied by Dr M. Schäfer. The sources of other materials are given in the text.
Results
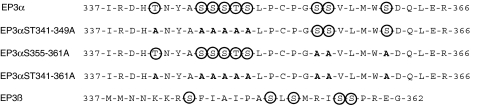
To study the function of the Ser/Thr cluster 341–349 within the rEP3α-R C-terminal domain in agonist-induced rEP3-R desensitization, we mutated these Ser/Thr residues to alanine (Figure 1) and generated HEK293 cell lines stably expressing rEP-3-R isoforms and the mutated receptor.
Figure 1.
C-terminal domain of rEP3-R isoforms and Ser/Thr to Ala mutations. C-terminal peptides, which differ between the two isoforms, are shown. Potential phosphorylation sites are marked by circles. Ser/Thr to Ala mutations are highlighted in bold.
Characterization of the ligand binding and signaling properties of rEP3-R isoforms and the rEP3α-R receptor mutant
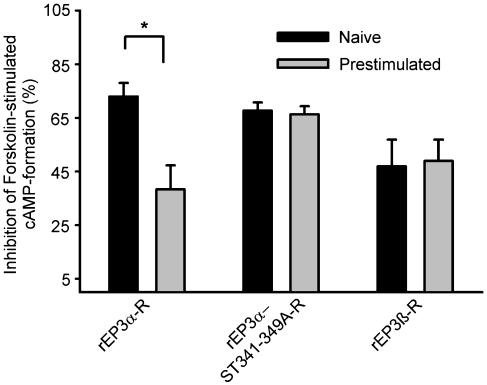
Binding properties of the receptor proteins were characterized by saturation binding assays in membranes of cells expressing rEP-3-R isoforms or the mutated receptors. The Kd-values for PGE2 binding were in the range of 1.3–5 nM and did not differ significantly between the receptors (Table 2). The Bmax values for PGE2-binding ranged from 0.3 pmol mg−1 membrane protein in cells expressing the rEP3α-ST341-349A-R-CFP to 4 pmol mg−1 membrane protein in cells expressing the rEPα-R-CFP (Table 2). rEP3-R isoforms and the receptor mutant were coupled to Gi. Treatment of receptor expressing cells with 100 nM of the EP3-R specific agonist M&B28767 inhibited cAMP-formation induced by 1 μM forskolin by 70% (rEP3α-R and rEP3α-ST341-349A-R) or 50% (rEP3β-R) (Figure 2). Stimulation with 100 nM M&B28767 alone had no effect on cAMP-formation (not shown). Therefore the different C-terminal ends or mutation of the Ser/Thr cluster in rEP3α-R had no apparent influence on the binding and signaling properties of the receptor.
Table 2.
Binding characteristics of rEP-R isoforms and the rEP3α-R mutant
| Receptor | Kd (nM) | Bmax (pmol mg−1) |
|---|---|---|
| rEP3α-R | 5.0±1.0 (3) | 0.6±0.06 (3) |
| rEP3β-R | 3.9±0.2 (3) | 2.5±0.57 (3) |
| rEP3α-ST341-349A-R | 1.3±0.3 (3) | 0.4±0.03 (3) |
| rEP3α-R-CFP | 3.4±0.2 (3) | 4.2±0.35 (3) |
| rEP3β-R-GFP | 2.1±0.8 (3) | 2.0±0.49 (3) |
| rEP3α-ST341-349A-R-CFP | 1.6±0.9 (4) | 0.3±0.06 (4) |
| rEP3α-S355-361A-R-CFP | 3.2±0.5 (3) | 0.6±0.01 (3) |
| rEP3α-ST341-361A-R-CFP | 3.1±0.1 (3) | 0.6±0.07 (3) |
Membranes from HEK293 cells stably expressing EP3-R isoforms or the receptor mutant were prepared and examined for [3H]PGE2-binding. Kd and Bmax values were determined by nonlinear regression analysis of saturation binding assays. Details are described in the Methods section. Values are means±s.e.m. from the numbers of independent experiments (indicated in parentheses).
Figure 2.
M&B28767-induced uncoupling of rEP3-R isoforms and the rEP3α-ST341-349A-R mutant. HEK293 cells stably expressing rEP3α-R, rEP3β-R or the rEP3α-ST341-349A-R mutant were exposed where indicated to 100 nM of the EP3-R agonist M&B28767 for 10 min. The agonist was then removed by an acid wash followed by extensive washing with incubation medium (see Methods). cAMP formation was determined by radioimmunoassay in cell cultures after stimulation for 10 min with 1 μM forskolin and 10 nM M&B28767. Values are means±s.e.m. and are expressed as a percent of control cells tested in parallel that were not exposed to the agonist in the preincubation period. Student's t-test for paired samples: *P<0.05.
Agonist-induced uncoupling from Gi
Cells, stably expressing either the wild-type rEP3α-R or rEP3β-R isoform or the rEP3α-ST341-349A-R mutant were preincubated with a saturating concentration of the EP3-R agonist M&B28767 (100 nM) for 10 min. The agonist was then completely removed (see Methods). In cells expressing the rEP3β-R M&B28767-dependent inhibition of forskolin-induced cAMP formation in a second stimulation phase was not attenuated by prior agonist treatment. However, in cells expressing rEP3α-R agonist-dependent inhibition of forskolin-stimulated cAMP-formation was reduced to about 50% in a second, compared to a first stimulation phase, which reflects uncoupling of the rEP3α-R from Gi protein (Figure 2). Mutation of Ser/Thr residues 341–349 in rEP3α-R completely abolished agonist-induced uncoupling (Figure 2) underscoring the importance of this Ser/Thr cluster for agonist-induced uncoupling of EP3-R from the Gi signaling chain.
Agonist-induced internalization of the rEP3-R
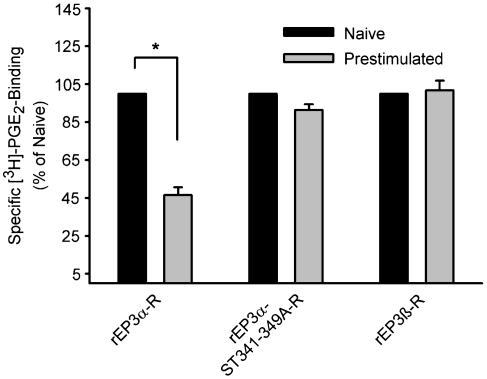
Agonist-induced rEP3-R internalization was assayed biochemically by analyzing the reduction of cell surface-accessible PGE2-binding sites and by laser scanning microscopy. Preincubation with 100 nM M&B28767 for 10 min did not affect cell surface PGE2-binding of rEP3β-R expressing cells, but reduced the number of binding sites on rEP3α-R expressing cells by 50% (Figure 3). This receptor internalization was abolished in cells expressing rEP3α-ST341-349A-R, which lacks the cluster of Ser/Thr residues in the C-terminal domain. Apparently, Ser/Thr residues in this cluster are essential determinants for agonist-induced receptor internalization.
Figure 3.
M&B28767-induced internalization of rEP3-R isoforms and the rEP3α-ST341-349A-R mutant. HEK293 cells stably expressing rEP3α-R, rEP3β-R or the rEP3α-ST341-349A-R mutant were exposed where indicated to 100 nM of the EP3-R agonist M&B28767 for 10 min. The agonist was then removed by an acid wash followed by extensive washing with incubation medium (see Methods). Cells were then incubated with 5 nM [3H]PGE2 for 2 h at 4°C. Unspecific cell surface binding was determined in the presence of 10 μM PGE2. Unbound ligand was removed, cells were lysed and radioactivity in the cell lysate was counted. Values are means±s.e.m. and are expressed as a percent of control cells tested in parallel that were not exposed to the agonist in the preincubation period. Student's t-test for paired samples: *P<0.05.
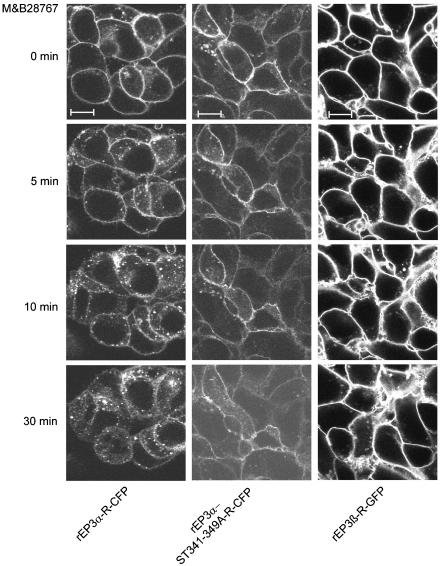
In addition to the biochemical rEP3-R internalization, agonist-induced translocation of GFP/CFP-tagged rEP3-R from the plasma membrane into intracellular compartments was monitored for 30 min in living cells by laser scanning microscopy. In the absence of agonist, all receptors were expressed predominantly at the plasma membrane (Figure 4). Agonist treatment of rEP3β-R expressing cells did not change the distribution of the receptor neither for short (5 min) nor for long (30 min) periods of agonist exposure. In contrast, cells expressing rEP3α-R-CFP exhibited receptor translocation from the plasma membrane to intracellular endocytotic vesicles within 5 min of M&B28767 addition (Figure 4). After prolonged agonist exposure, these endocytotic vesicles grew in size and number with a maximum after 10 min and were still observed 30 min after agonist treatment. Receptor internalization was completely abolished by the mutation of Ser/Thr residues 341–349. The receptor remained located predominantly at the plasma membrane despite stimulation of cells expressing this receptor mutant with M&B28767 for 30 min. These results are consistent with those obtained by the biochemical internalization studies and emphasize the importance of the Ser/Thr cluster in the C-terminal domain of rEP3α-R for agonist-induced receptor internalization.
Figure 4.
M&B28767-induced subcellular redistribution of rEP3-R isoforms and the rEP3α-ST341-349A-R mutant. HEK293 cells stably expressing GFP- or CFP-tagged rEP3α-R, rEP3β-R or rEP3α-ST341-349A-R were transferred to a heated (37°C) microscope stage filled with 1 ml Krebs–HEPES buffer pH 7.4 and the distribution of the receptors was visualized before and after treatment with 100 nM M&B28767 on a Zeiss 510 inverted LSM (lexc=488 nm for GFP and 462 nm for CFP, lem=515 nm for GFP and 505 nm for CFP). Representative confocal microscopic images of receptor fluorescence in the cells treated with M&B28767 for 0, 5, 10 and 30 min at 37°C are shown. Scale bars=10 μM.
The possibility would be envisaged that the C-terminal extension of the receptor protein with the GFP-tag might affect the agonist-dependent internalization. The results, however, indicate that the C-terminal CFP-tag neither inhibited the agonist-induced internalization of the wild-type rEP3α-R nor caused a subcellular redistribution of the receptor proteins in the absence of the ligand.
Immunological detection of rEP3-R-GFP fusion proteins
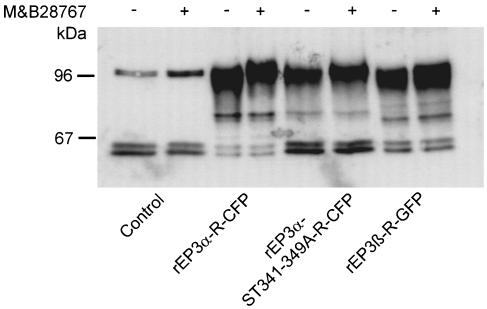
rEP3-R-GFP/CFP fusion-proteins were immunprecipitated with α-GFP antibody and visualized in Western blots with the same antibody (Figure 5). All three receptor proteins appeared as a broad band with an apparent molecular weight of 90–100 kDa. This band corresponds to the predicted molecular weight of the fusion protein of complex-glycosylated rEP3-R and the 27 kDa GFP/CFP protein fused to the C-terminal end of rEP3-R (Böer et al., 2000). In untransfected HEK293 cells, a band with a similar molecular weight was also detected. However, this band was by far less intense and might represent a fragment of the α-GFP antibody used for immunoprecipitation, as this band was absent in Western blots of membrane proteins derived from nontransfected HEK293 cells (not shown). The intensity of the receptor bands was quantified densitometrically. Apparently, equal amounts of the receptor protein were present in all lanes. As the amount of total protein applied to the lanes was normalized to the cell surface PGE2-binding on the cells from which the samples were obtained, the close similarity in quantity of immunoreactive protein in the different lanes indicates that the determination of cell surface binding on intact cells is a valid approach for the quantification for the subsequent phosphorylation studies.
Figure 5.
Immunological detection of rEP3α-CFP-R, rEP3β-GFP-R and rEP3α-ST341-349A-CFP-R. Receptor proteins of transfected cells were immunoprecipitated with α-GFP antibodies, resolved by SDS–PAGE under reducing conditions and transferred to PVDF membrane. Receptor proteins were detected with a sandwich of monoclonal α-GFP and horseradish-peroxidase-conjugated anti-mouse antiserum visualized with enhanced chemiluminescence as described in Methods.
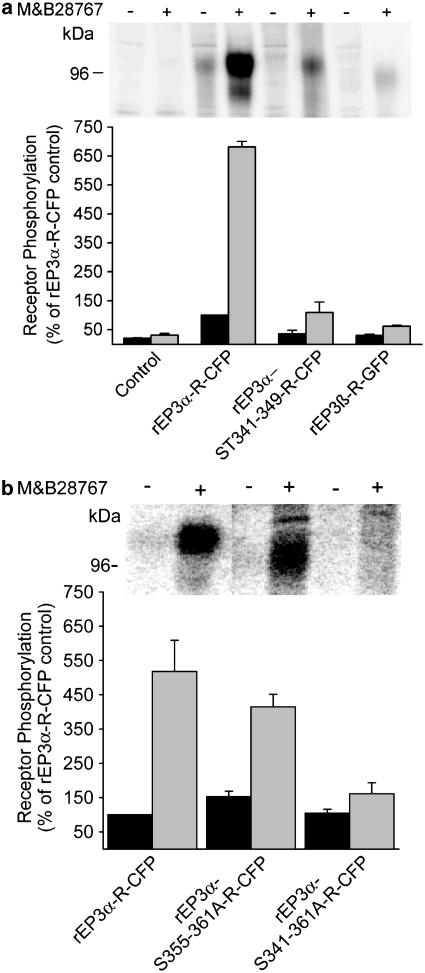
Agonist-induced EP3-R phosphorylation
To study agonist-induced phosphorylation of expressed receptors, cells expressing GFP/CFP-tagged receptors were labeled with 32Pi and stimulated with 100 nM of the EP3-R agonist M&B28767 for 10 min. A phosphorylated protein was immunoprecipitated with the α-GFP antibody from solubilized proteins of cells expressing the rEP3α-R-CFP stimulated with M&B28767 (Figure 6). This protein comigrated with the rEP3α-R-CFP detected by Western blot and was absent in nontransfected control cells. In contrast, no phosphorylated protein was immunoprecipitated from rEP3β-R-GFP expressing cells with or without agonist-stimulation (Figure 6a). Elimination of Ser/Thr residues 341–349 in the rEP3α-R C-terminal domain led to a dramatic reduction of basal and agonist-induced phosphorylation of the rEP3α-R-CFP (Figure 6a). In contrast, elimination of Ser residues 355–361 reduced M&B28767-induced phosphorylation slightly but not significantly (Figure 6b). Substitution of all Ser/Thr residues in the rEP3α-R C-terminal domain by alanine (rEP3α-ST341-361A-R) completely blocked receptor phosphorylation (Figure 6b). Thus, Ser/Thr residues within the cluster Ser/Thr 341–349 appear to be the main target site for agonist-induced phosphorylation of the receptor and phosphorylation of Ser residues 355–361 contribute only to a minor extent.
Figure 6.
M&B28767-induced phosphorylation of rEP3-R isoforms and receptor mutants. 32Pi-labeled HEK293 control cells or cells stably expressing GFP/CFP-tagged rEP3R isoforms or the rEP3α-ST341-349A-R-CFP (a) or rEP3α-S355-361A-R-CFP or rEP3α-ST341-361A-R-CFP (b) mutants were stimulated for 10 min at 37°C with 100 nM M&B28767. Receptors were immunoprecipitated with α-GFP antibodies and normalized for cell surface receptor expression. Receptor proteins were resolved under reducing conditions by SDS–PAGE and phosphorylation was determined by PhosphorImager analysis after 2d. Agonist-induced receptor phosphorylation was expressed relative to that obtained in rEP3α-R-CFP expressing cells not exposed to M&B28767. The upper panel shows a representative PhosphorImager analysis and the lower panel the quantitative analysis from pooled data from at least three independent experiments. Data are means±s.e.m.
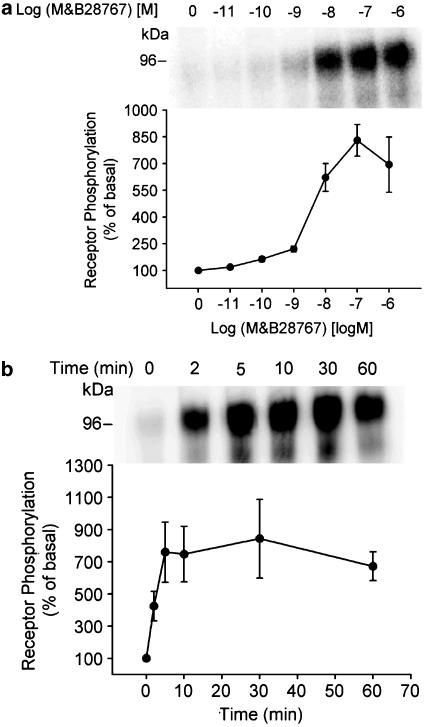
M&B28767-induced phosphorylation of the rEP3α-R-CFP was dose-dependent with an apparent EC50 between 1 and 10 nM and a maximum at 100 nM M&B28767 (Figure 7a). Phosphorylation of rEP3α-R-CFP thus occurred in an agonist-concentration range similar to the Kd value of the receptor revealing that only the agonist-occupied receptor was a kinase substrate. rEP3α-R-CFP phosphorylation was rapid, and was detected as early as 2 min after agonist treatment (Figure 7b) with maximum at 5 min and was sustained for at least 60 min in the presence of agonist (Figure 7b).
Figure 7.
Dose and time dependence of M&B28767-stimulated rEP3α-R-CFP phosphorylation. (a) 32Pi-labeled HEK293 cells expressing the rEP3α-R-CFP were stimulated for 10 min at 37°C with the concentrations of M&B28767 indicated. Receptor phosphorylation was analyzed as described in the legend to Figure 6. (b) 32Pi-labeled HEK293 cells expressing the rEP3α-R-CFP were stimulated for the times indicated at 37°C with 100 nM M&B28767. The upper panels show a representative PhosphorImager analysis and the lower panel the quantitative analysis from pooled data from three independent experiments. Receptor phosphorylation is expressed as percent of phosphorylation in unstimulated rEP3α-R-CFP cells. Data are means±s.e.m.
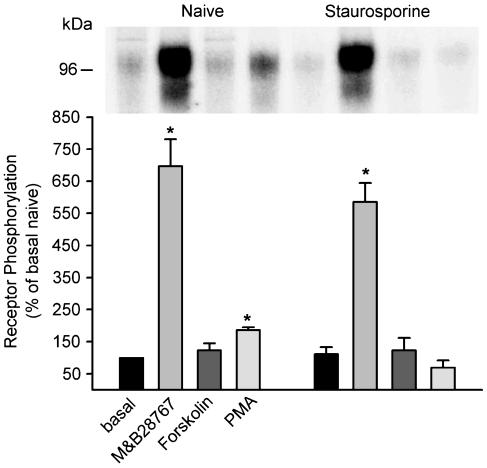
Inhibition by staurosporine of second messenger kinase but not agonist-induced phosphorylation of the rEP3α-R-CFP
To investigate which kinases affect agonist-induced rEP3α-R-CFP phosphorylation, receptor expressing cells were treated with agonist or with both specific activators and inhibitors of the second messenger-dependent kinases PKC or camp-dependent PKA. Forskolin, which increases intracellular cAMP concentration by activating adenylate cyclase leading to activation of PKA, had no significant effect on rEP3α-R-CFP phosphorylation (Figure 8). In contrast, the phorbolester PMA, a potent PKC activator, led to a slight but significant increase in rEP3α-R-CFP phosphorylation (Figure 8) but to a much lesser extent than the agonist M&B28767.
Figure 8.
Phosphorylation of the rEP3α-R-CFP by M&B28767 and activators and inhibitors of second-messenger-dependent kinases. 32Pi-labeled HEK293 cells expressing the rEP3α-R-CFP were preincubated without or with 400 nM staurosporine for 20 min at 37°C prior to cellular stimulation. Cells were then stimulated for 10 min at 37°C with 100 nM M&B28767, 2 μM PMA or 50 μM forskolin+1 mM IBMX. Receptors were immunoprecipitated with α-GFP antibody, resolved by SDS–PAGE and phosphorylation determined by PhosphorImager analysis after 2d. The upper panels show a representative PhosphorImager analysis and the lower panel the quantitative analysis from pooled data from three independent experiments. Receptor phosphorylation is expressed as percent of phosphorylation of untreated cells in the absence of staurosporine. Data are means±s.e.m.; *P<0.05 compared to basal levels.
Staurosporine at a concentration of 400 nM, which blocks PKA and PKC activity, inhibited PMA-induced rEP3α-R-CFP phosphorylation and decreased basal receptor phosphorylation. In contrast, staurosporine had no effect on M&B28767-induced phosphorylation, indicating that neither PKC nor PKA are involved in agonist-induced rEP3α-R-CFP phosphorylation (Figure 8).
Discussion
The biological effects elicited by prostanoid receptors, like those of many other GPCRs, are regulated by an attenuation of their intracellular signal transduction in response to short- or long-term ligand exposure. Recent studies showed that the C-terminal domain of prostanoid receptors plays an important role in this desensitization process. Variations of the mEP3-R C-terminal domain generated by alternative splicing resulted in differences in rapid agonist induced receptor desensitization (Negishi et al., 1993). The role of the C-terminal domain in EP-R desensitization is also emphasized by the finding that the C-terminal domain of the desensitizable EP4-R was sufficient to confer a rapid agonist-induced GRK-mediated phosphorylation and desensitization in a hybrid receptor in which it was substituted for the homologous domain of the non-desensitizable rEP3β-R (Neuschäfer-Rube et al., 1997a). In contrast, the third intracellular loop of the EP4-R was neither necessary nor sufficient to mediate agonist-induced desensitization of this hybrid receptor (Neuschäfer-Rube et al., 1997a). The current study aimed to analyze the structures and mechanism that are involved in agonist-induced desensitization of rat EP3-R isoforms.
rEP3-R C-terminal domain and desensitization
In a previous study, it was shown that the mouse EP3α-R underwent rapid agonist-induced desensitization in contrast to the C-terminal splice variant EP3β-R (Negishi et al., 1993). Supporting this previous finding, the rat EP3α-R also underwent agonist-induced uncoupling from Gi (Figure 2) and was rapidly internalized after agonist-treatment (Figures 3 and 4). In contrast, signaling by the rat EP3β-R was not affected by prior agonist-stimulation. One main structural difference between the two C-terminal splice variants is the number and the pattern of Ser/Thr residues in the part of the C-terminal domain that is encoded by the differentially spliced region of the mRNAs. The rEP3α-R contains nine Ser/Thr residues, which are all conserved between mouse and rat EP3α-R, five of which are clustered in the proximal half. In contrast, rEP3β-R contains only five Ser/Thr residues that are less conserved between rat and mouse, that is, S344T and S353G, and are not clustered. Clusters of Ser/Thr residues in the C-terminal domain of many GPCR were shown to be important determinants for receptor desensitization, acting as targets for agonist-induced receptor phosphorylation and/or as binding sites for the adaptor protein β-arrestin (Oakley et al., 2001). For the angiotensin II receptor and the substance P receptor to name but a few it was shown that Ser/Thr cluster in the C-terminal domain was essential for the formation of stable complexes with the adaptor protein β-arrestin, leading to a sustained sequestration of the receptor (Oakley et al., 2001). The cluster Ser/Thr 345–349 might fulfill such a function in the C-terminal domain of the EP3α-R. In support of this hypothesis, elimination of all Ser/Thr residues in this cluster in the rEP3α-ST341-349A-R blocked both agonist-induced uncoupling from Gi and redistribution of the receptor into intracellular compartments.
rEP3-R desensitization by phosphorylation
For many GPCRs agonist-induced uncoupling and internalization depend on the phosphorylation of the receptor either by second messenger-dependent kinases or GRKs (Lohse, 1993; Premont et al., 1995). For the regulation of prostanoid receptor signaling this is not always the case. Of the two C-terminal isoforms of the human thromboxane A2 receptor (TPα-R and TPβ-R), which were both phosphorylated and uncoupled from Gq protein upon agonist-stimulation, only the TPβ-R isoform was internalized (Parent et al., 1999). For the human IP-R, phosphorylation of the receptor by PKC was a prerequisite for agonist-induced uncoupling whereas PKC was not essential for IP-R internalization (Smyth et al., 2000). In the case of the human EP4-R maximal agonist-induced phosphorylation of the receptor greatly enhanced the stability of receptor/β-arrestin complexes but was not essential for receptor internalization (Neuschäfer-Rube et al., 2004). The current study provides evidence that agonist-stimulated phosphorylation may be essential for both uncoupling and internalization of the rEP3-R. Stimulation of cells expressing the rEP3α-R with the agonist M&B28767 led to a dose- and time-dependent receptor phosphorylation (Figure 7a and b) and induced both its uncoupling and internalization. By contrast, M&B28767 neither induced phosphorylation nor uncoupling or internalization of the rEP3β-R, stably expressed in HEK293 cells (Figures 2, 3, 4 and 6). Mutation of Ser/Thr residues 341–349, which may act as potential phosphorylation sites, attenuated M&B28767-induced phosphorylation of the rEP3α-R and completely blocked uncoupling and internalization. Interestingly, mutation of Ser/Thr residues 341–349 also blocked basal phosphorylation of the rEP3α-R, indicating a small fraction of constitutive active receptor as described for the mouse rEP3α-R (Hasegawa et al., 1996).
In contrast, mutation of Ser residues 355–361 in the distal part of the C-terminal did not affect M&B28767-induced phosphorylation of the rEP3α-R. This result ruled out that loss of agonist-induced phosphorylation by mutation of Ser/Thr residues 341–349 was due to affecting the secondary structure of the C-terminal domain disturbing access to Ser 355–366.
Therefore, phosphorylation of the rEP3α-R at Ser/Thr residues between 341 and 349 seemed to be essential for agonist-induced desensitization of the rEP3-R. However, additional phosphorylation that depends on the presence of Ser/Thr residues in the rEP3α-R C-terminal domain, or other parts of the receptor, that is, Ser or Thr residues in the first, second or third intracellular loop, cannot be ruled out.
Kinases involved in rEP3α-R phosphorylation
Agonist-dependent receptor phosphorylation could be mediated either through second messenger-dependent kinases or GRK. Examples exist for both mechanisms. The human IP-R, which increases cAMP-formation at low iloprost concentrations and InsP3-formation at high iloprost concentrations, was phosphorylated and desensitized by high iloprost concentrations only (Smyth et al., 1998). Phosphorylation and desensitization were inhibited by staurosporine, thus inferring a PKC-dependent mechanism. In HEK293 cells cotransfected with the Gs-coupled DP-R and either the human TPα-R or TPβ-R isoform stimulation with PGD2 led to a heterologous PKA-dependent, but PKC-independent phosphorylation and desensitization of the TPα-R (Foley et al., 2001). Conversely, GRK-dependent phosphorylation of Ser and Thr residues in the C-terminal domain has been shown to mediate receptor desensitization of the β1-adrenergic receptor (Freedman et al., 1995), the endothelin receptor (Freedman et al., 1997), the delta opioid receptor (Pei et al., 1995) and the angiotensin II receptor (Oppermann et al., 1996) to name but a few. A function of GRKs in phosphorylation and desensitization of prostanoid receptors has been shown for the human thromboxane receptor and the human EP4-R (Neuschäfer-Rube et al., 1999; Zhou et al., 2001). The involvement of GRKs in agonist-stimulated rEP3α-R phosphorylation and desensitization is supported by several lines of evidence.
First, a PKA-dependent phosphorylation of rEP3α-R as the sole mechanism of desensitization seems to be unlikely, since in HEK293 cells stably transfected with either one of the rEP3-R isoforms, cAMP formation did not increase after agonist exposure and therefore PKA was not activated. By contrast, agonist-induced stimulation of cAMP-formation was decreased.
Second, PKA activation by forskolin, which led to a massive increase in cAMP formation in HEK293 cells (not shown), had no effect on rEP3α-R or rEP3β-R phosphorylation (Figure 8; Neuschäfer-Rube et al., 1999). On the other hand, activation of PKC by a high dose (2 μM) of the phorbolester PMA led to a slight phosphorylation of the rEP3α-R (Figure 8) and the rEP3β-R (Neuschäfer-Rube et al., 1999). The PMA-dependent receptor phosphorylations were suppressed by the PKC-inhibitor staurosporine, which, however, had no effect on the agonist-induced phosphorylation of the rEP3α-R (Figure 8). Thus, apparently PKC was not involved in M&B28767-stimulated phosphorylation and desensitization of the rEP3α-R.
Third, substitution of Ser/Thr residues 341–349, which do not include a consensus motif for phosphorylation by PKA (RRXS) or PKC (SXK/R) blocked agonist-induced phosphorylation and desensitization of the rEP3α-R.
Conclusion
Alternative splicing of rEP3-R RNA generates C-terminal isoforms, which differ in their ability to undergo agonist-induced desensitization. This might differentially regulate the strength and persistence of downstream rEP3-R signaling, depending on the rEP3-R isoform predominantly expressed on different cell types. Agonist-induced desensitization of the rEP3α-R isoform is most likely initiated by phosphorylation at its C-terminal domain, where a cluster of Ser/Thr residues appears to be a substrate for GRKs when the receptor is activated by a bound agonist. Phosphorylation of Ser/Thr residues within this cluster will initiate the uncoupling of the receptor from Gi and internalization of the receptor into intracellular compartments.
Acknowledgments
This work was supported by Grant Pu 100/1-1 from the Deutsche Forschungsgemeinschaft and by the Fonds der Chemischen Industrie. We thank Dagmar Michel for their excellent technical assistance.
Abbreviations
- DMEM
Dulbecco's modified Eagle's medium
- FCS
fetal calf serum
- GFP
green fluorescent protein
- Gx
heterotrimeric Gx protein
- HEPES
N-(2-hydroxyethyl) piperazin-N′-2-ethanesulfonic acid
- HEK
human embryonal kidney
- IBMX
isobutyl-methyl-xanthine
- mAb
monoclonal antibody
- MES
2-(N-morpholino)ethanesulfonic acid
- PAGE
polyacrylamide gel electrophoresis
- PBS
phosphate-buffered saline
- PKA
protein kinase A
- PKC
protein kinase C
- PVDF
polyvinylidenfluoride
References
- AMANO H., HAYASHI I., ENDO H., KITASATO H., YAMASHINA S., MARUYAMA T., KOBAYASHI M., SATOH K., NARITA M., SUGIMOTO Y., MURATA T., YOSHIMURA H., NARUMIYA S., MAJIMA M. Host prostaglandin E2 EP3 signaling regulates tumor-associated angiogenesis and tumor growth. J. Exp. Med. 2003;197:221–232. doi: 10.1084/jem.20021408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BÖER U., NEUSCHÄFER-RUBE F., MÖLLER U., PÜSCHEL G.P. Requirement of N-glycosylation of the prostaglandin E2 receptor EP3β for correct sorting to the plasma membrane but not for correct folding. Biochem. J. 2000;350:839–847. [PMC free article] [PubMed] [Google Scholar]
- BOIE Y., STOCCO R., SAWYER N., SLIPETZ D.M., UNGRIN M.D., NEUSCHÄFER-RUBE F., PÜSCHEL G.P., METTERS K.M., ABRAMOVITZ M. Molecular cloning and characterization of the four rat prostaglandin E2 prostanoid receptor subtypes. Eur. J. Pharmacol. 1997;340:227–241. doi: 10.1016/s0014-2999(97)01383-6. [DOI] [PubMed] [Google Scholar]
- COLEMAN R.A., SMITH W.L., NARUMIYA S. International Union of Pharmacology classification of prostanoid receptors: properties, distribution, and structure of the receptors and their subtypes. Pharmacol. Rev. 1994;46:205–229. [PubMed] [Google Scholar]
- FERGUSON S.S. Evolving concepts in G protein-coupled receptor endocytosis: the role in receptor desensitization and signaling. Pharmacol. Rev. 2002;53:1–24. [PubMed] [Google Scholar]
- FOLEY J.F., KELLEY L.P., KINSELLA B.T. Prostaglandin D2 receptormediated desensitization of the alpha isoform of the human thromboxane A2 receptor. Biochem. Pharmacol. 2001;62:229–239. doi: 10.1016/s0006-2952(01)00661-x. [DOI] [PubMed] [Google Scholar]
- FREEDMAN N.J., AMENT A.S., OPPERMANN M., STOFFEL R.H., EXUM S.T., LEFKOWITZ R.J. Phosphorylation and desensitization of human endothelin A and B receptors. Evidence for G protein-coupled receptor kinase specificity. J. Biol. Chem. 1997;272:17734–17743. doi: 10.1074/jbc.272.28.17734. [DOI] [PubMed] [Google Scholar]
- FREEDMAN N.J., LIGGETT S.B., DRACHMAN D.E., PEI G., CARON M.G., LEFKOWITZ R.J. Phosphorylation and desensitization of the human beta 1adrenergic receptor. Involvement of G protein-coupled receptor kinases and cAMPdependent protein kinase. J. Biol. Chem. 1995;270:17953–17961. doi: 10.1074/jbc.270.30.17953. [DOI] [PubMed] [Google Scholar]
- HASEGAWA H., NEGISHI M., ICHIKAWA A. Two isoforms of the prostaglandin E receptor EP3 subtype different in agonist-independent constitutive activity. J. Biol. Chem. 1996;271:1857–1860. doi: 10.1074/jbc.271.4.1857. [DOI] [PubMed] [Google Scholar]
- JIN J., MAO G.F., ASHBY B. Constitutive activity of human prostaglandin E receptor EP3 isoforms. Br. J. Pharmacol. 1997;121:317–323. doi: 10.1038/sj.bjp.0701121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KOBAYASHI T., NARUMIYA S. Prostanoids in health and disease; lessons from receptor-knockout mice. Adv. Exp. Med. Biol. 2002;507:593–597. doi: 10.1007/978-1-4615-0193-0_90. [DOI] [PubMed] [Google Scholar]
- KOTANI M., TANAKA I., OGAWA Y., SUGANAMI T., MATSUMOTO T., MURO S., YAMAMOTO Y., SUGAWARA A., YOSHIMASA Y., SAGAWA N., NARUMIYA S., NAKAO K. Multiple signal transduction pathways through two prostaglandin E receptor EP3 subtype isoforms expressed in human uterus. J. Clin. Endocrinol. Metab. 2000;85:4315–4322. doi: 10.1210/jcem.85.11.6989. [DOI] [PubMed] [Google Scholar]
- LAPORTE S.A., OAKLEY R.H., ZHANG J., HOLT J.A., FERGUSON S.S., CARON M.G., BARAK L.S. The beta2-adrenergic receptor/beta arrestin complex recruits the clathrin adaptor AP-2 during endocytosis. Proc. Natl. Acad. Sci. U.S.A. 1999;96:3712–3717. doi: 10.1073/pnas.96.7.3712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LAZARI M.F., BERTRAND J.E., NAKAMURA K., LIU X., KRUPNICK J.G., BENOVIC J.L., ASCOLI M. Mutation of individual serine residues in the C-terminal tail of the lutropin/choriogonadotropin receptor reveal distinct structural requirements for agonist-induced uncoupling and agonist-induced internalization. J. Biol. Chem. 1998;273:18316–18324. doi: 10.1074/jbc.273.29.18316. [DOI] [PubMed] [Google Scholar]
- LOHSE M.J. Molecular mechanisms of membrane receptor desensitization. Biochim. Biophys. Acta. 1993;1179:171–188. doi: 10.1016/0167-4889(93)90139-g. [DOI] [PubMed] [Google Scholar]
- MAESTES D.C., POTTER R.M., PROSSNITZ E.R. Differential phosphorylation paradigms dictate desensitization and internalization of the N-formyl peptide receptor. J. Biol. Chem. 1999;274:29791–29795. doi: 10.1074/jbc.274.42.29791. [DOI] [PubMed] [Google Scholar]
- MUNSON P.J., RODBARD D. Ligand: a versatile computerized approach for characterization of ligand-binding systems. Anal. Biochem. 1980;107:220–239. doi: 10.1016/0003-2697(80)90515-1. [DOI] [PubMed] [Google Scholar]
- NAMBA T., SUGIMOTO Y., NEGISHI M., IRIE A., USHIKUBI F., KAKIZUKA A., ITO S., ICHIKAWA A., NARUMIYA S. Alternative splicing of C-terminal tail of prostaglandin E receptor subtype EP3 determines G-protein specificity. Nature. 1993;365:166–170. doi: 10.1038/365166a0. [DOI] [PubMed] [Google Scholar]
- NARUMIYA S., SUGIMOTO Y., USHIKUBI F. Prostanoid receptors: structures, properties, and functions. Physiol. Rev. 1999;79:1193–1226. doi: 10.1152/physrev.1999.79.4.1193. [DOI] [PubMed] [Google Scholar]
- NEGISHI M., SUGIMOTO Y., IRIE A., NARUMIYA S., ICHIKAWA A. Two isoforms of prostaglandin E receptor EP3 subtype. Different COOH-terminal domains determine sensitivity to agonist-induced desensitization. J. Biol. Chem. 1993;268:9517–9521. [PubMed] [Google Scholar]
- NEUSCHÄFER-RUBE F., DEVRIES C., HÄNECKE K., JUNGERMANN K., PÜSCHEL G.P. Molecular cloning and expression of a prostaglandin E2 receptor of the EP3β subtype from rat hepatocytes. FEBS Lett. 1994;351:119–122. doi: 10.1016/0014-5793(94)00837-x. [DOI] [PubMed] [Google Scholar]
- NEUSCHÄFER-RUBE F., HÄNECKE K., BLASCHKE V., JUNGERMANN K., PÜSCHEL G.P. The C-terminal domain of the Gs-coupled EP4 receptor confers agonist-dependent coupling control to Gi but no coupling to Gs in a receptor hybrid with the Gi-coupled EP3 receptor. FEBS Lett. 1997a;401:185–190. doi: 10.1016/s0014-5793(96)01468-8. [DOI] [PubMed] [Google Scholar]
- NEUSCHÄFER-RUBE F., HÄNECKE K., PÜSCHEL G.P. The C-terminal domain of the human EP4 receptor confers agonist-induced receptor desensitization in a receptor hybrid with the rat EP3beta receptor. FEBS Lett. 1997b;415:119–124. doi: 10.1016/s0014-5793(97)01105-8. [DOI] [PubMed] [Google Scholar]
- NEUSCHÄFER-RUBE F., HERMOSILLA R., REHWALD M., RÖNNSTRAND L., SCHÜLEIN R., WERNSTEDT C, PÜSCHEL G.P. Identification of a Ser/Thr cluster in the C-terminal domain of the human prostaglandin receptor EP4 that is essential for agonist-induced beta-arrestin1 recruitment but differs from the apparent principal phosphorylation site. Biochem J. 2004;379:573–585. doi: 10.1042/BJ20031820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- NEUSCHÄFER-RUBE F., OPPERMANN M., MÖLLER U., BÖER U., PÜSCHEL G.P. Agonist-induced phosphorylation by G protein-coupled receptor kinases of the EP4 receptor carboxyl-terminal domain in an EP3/EP4 prostaglandin E2 receptor hybrid. Mol. Pharmacol. 1999;56:419–428. doi: 10.1124/mol.56.2.419. [DOI] [PubMed] [Google Scholar]
- OAKLEY R.H., LAPORTE S.A., HOLT J.A., BARAK L.S., CARON M.G. Molecular determinants underlying the formation of stable intracellular G proteincoupled receptor-beta-arrestin complexes after receptor endocytosis. J. Biol. Chem. 2001;276:19452–19460. doi: 10.1074/jbc.M101450200. [DOI] [PubMed] [Google Scholar]
- OPPERMANN M., FREEDMAN N.J., ALEXANDER R.W., LEFKOWITZ R.J. Phosphorylation of the type 1A angiotensin II receptor by G protein-coupled receptor kinases and protein kinase C. J. Biol. Chem. 1996;271:13266–13272. doi: 10.1074/jbc.271.22.13266. [DOI] [PubMed] [Google Scholar]
- PALS-RYLAARSDAM R., XU Y., WITT-ENDERBY P., BENOVIC J.L., HOSEY M.M. Desensitization and internalization of the m2 muscarinic acetylcholine receptor are directed by independent mechanisms. J. Biol. Chem. 1995;270:29004–29011. doi: 10.1074/jbc.270.48.29004. [DOI] [PubMed] [Google Scholar]
- PARENT J.L., LABRECQUE P., ORSINI M.J., BENOVIC J.L. Internalization of the TXA2 receptor alpha and beta isoforms. Role of the differentially spliced COOH terminus in agonist-promoted receptor internalization. J. Biol. Chem. 1999;274:8941–8948. doi: 10.1074/jbc.274.13.8941. [DOI] [PubMed] [Google Scholar]
- PEI G., KIEFFER B.L., LEFKOWITZ R.J., FREEDMAN N.J. Agonist dependent phosphorylation of the mouse delta-opioid receptor: involvement of G protein-coupled receptor kinases but not protein kinase C. Mol. Pharmacol. 1995;48:173–177. [PubMed] [Google Scholar]
- PIERCE K.L., REGAN J.W. Prostanoid receptor heterogeneity through alternative mRNA splicing. Life Sci. 1998;62:1479–1483. doi: 10.1016/s0024-3205(98)00093-9. [DOI] [PubMed] [Google Scholar]
- PITCHER J., LOHSE M.J., CODINA J., CARON M.G., LEFKOWITZ R.J. Desensitization of the isolated beta 2-adrenergic receptor by beta-adrenergic receptor kinase, cAMP-dependent protein kinase, and protein kinase C occurs via distinct molecular mechanisms. Biochemistry. 1992;31:3193–3197. doi: 10.1021/bi00127a021. [DOI] [PubMed] [Google Scholar]
- PREMONT R.T., INGLESE J., LEFKOWITZ R.J. Protein kinases that phosphorylate activated G protein-coupled receptors. FASEB J. 1995;9:175–182. doi: 10.1096/fasebj.9.2.7781920. [DOI] [PubMed] [Google Scholar]
- SMYTH E.M., AUSTIN S.C., REILLY M.P., FITZGERALD G.A. Internalization and sequestration of the human prostacyclin receptor. J. Biol. Chem. 2000;275:32037–32045. doi: 10.1074/jbc.M003873200. [DOI] [PubMed] [Google Scholar]
- SMYTH E.M., LI W.H., FITZGERALD G.A. Phosphorylation of the prostacyclin receptor during homologous desensitization. A critical role for protein kinase C. J. Biol. Chem. 1998;273:23258–23266. doi: 10.1074/jbc.273.36.23258. [DOI] [PubMed] [Google Scholar]
- SOUTHALL M.D., VASKO M.R. Prostaglandin receptor subtypes, EP3C and EP4, mediate the prostaglandin E2-induced cAMP production and sensitization of sensory neurons. J. Biol. Chem. 2001;276:6083–6091. doi: 10.1074/jbc.M011408200. [DOI] [PubMed] [Google Scholar]
- TAKEUCHI K., UKAWA H., KATO S., FURUKAWA O., ARAKI H., SUGIMOTO Y., ICHIKAWA A., USHIKUBI F., NARUMIYA S. Impaired duodenal bicarbonate secretion and mucosal integrity in mice lacking prostaglandin E-receptor subtype EP3. Gastroenterology. 1999;117:128–135. doi: 10.1016/s0016-5085(99)70398-7. [DOI] [PubMed] [Google Scholar]
- USHIKUBI F., SEGI E., SUGIMOTO Y., MURATA T., MATSUOKA T., KOBAYASHI T., HIZAKI H., TUBOI K., KATSUYAMA M., ICHIKAWA A., TANAKA T., YOSHIDA N., NARUMIYA S. Impaired febrile response in mice lacking the prostaglandin E receptor subtype EP3. Nature. 1998;395:281–284. doi: 10.1038/26233. [DOI] [PubMed] [Google Scholar]
- ZHOU H, YAN F., TAI H.H. Phosphorylation and desensitization of the human thromboxane receptor-alpha by G protein-coupled receptor kinases. J. Pharmacol. Exp. Ther. 2001;298:1243–1251. [PubMed] [Google Scholar]