Abstract
Carcinoma of the prostate continues to be a major health problem in the United States. Beginning in 1988, a marked increase in detection of prostate cancer occurred due to the development of a test for prostate-specific antigen (PSA). Controversy exists, however, about the value of PSA as a tumor marker. Although it has prognostic significance both before and after definitive therapy for prostate cancer, it is unclear whether routine PSA screening will translate into a survival advantage for patients. Because of its limitations, PSA may not ultimately be a good enough marker to be used as a screening tool. However, molecular biology has led to a rapid rise in the number of potential new prostate tumor markers, which may eventually overcome the weaknesses of PSA. Considerable progress has occurred in the diagnosis and management of prostate cancer: more is understood about the risk factors for the disease, possible ways to prevent it, and new ways to diagnose and monitor it. These developments have already translated into better patient care, while also identifying where further improvements are needed.
Key words: Prostate cancer, Biomarkers, Prostate-specific antigen, Prostate-specific antigen doubling time, Prostate-specific membrane antigen
Carcinoma of the prostate continues to be a major health problem in the United States. Of the more than 700,000 new cases of cancer occurring in men each year, approximately one third or 230,000 new cases were expected to be cancer of the prostate in 2005.1 In fact, the rates of this disease in US African American and Caucasian men are the highest in the world. Beginning in 1988, a marked increase in detection of prostate cancer occurred owing to the development of a test for prostate-specific antigen (PSA). The detection rate began to decline in 1997 but began to increase again in 2000.2 Although carcinoma of the prostate represents a high proportion of all new cancers in men in the United States, it accounts for only 10% of cancer deaths in men. Interestingly, between 1993 and 1997, the annual mortality from this disease declined and then leveled off in subsequent years.3 Controversy exists over the explanation for the decline in mortality. Some attribute the change to the increasing use of PSA screening, whereas others argue that the long natural history of the disease precludes a change in mortality beginning in 1993, given that extensive screening began only around 1989. Four years of testing is simply too short a period to yield such a drop in mortality. Another argument against attributing this change to PSA screening is that prostate cancer mortality also declined during the same period in countries such as England, where routine screening was not performed.
Two other explanations may explain the drop in mortality. One is the ability of PSA monitoring to identify progressive cancer after local therapy much earlier than was possible previously. As a result, subsequent therapies, primarily androgen ablation, although infrequently curative, slowed the course of the disease, thereby prolonging survival. Another excellent explanation is that prospective randomized studies demonstrated that earlier use of hormone therapy in combination with radiation or surgery improved survival compared with radiation or surgery alone.4–6
One thing is clear: clinicians are becoming more knowledgeable about this disease and the risk factors contributing to its development and progression. The most significant risk factor is age. Autopsy studies show that even by the age of 20, approximately 10% of men have prostate cancer cells in the prostate.7 This increases to 30% of men by age 50 and over 50% by age 80. Race is another risk factor; African American men appear to have a worse prognosis than Caucasian men.8 Diet also appears to play an important role in the development and possibly the progression of the disease. If one plots the mortality rate from prostate cancer according to the average dietary fat intake of countries, a direct correlation is seen (Figure 1).9 Also, some prostate cancers have been the result of genetic changes, including changes to the HPC1 gene.
Figure 1.
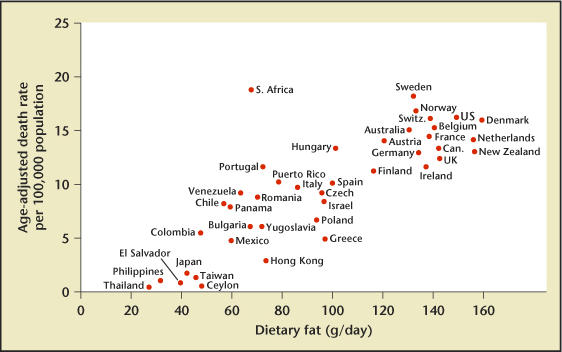
Relationship of dietary fat intake and death rate from prostatic cancer. Reproduced from Carroll and Khor,9 with permission from the publisher, S. Karger AG, Basel, Switzerland.
Protective factors, including tomatoes and tomato products, broccoli, and vitamin D, have also been suggested to play a role. Men with high levels of selenium appear to be at lower risk of the disease.8
One of the more important questions being asked is whether prostate cancer can actually be prevented. A recent study found a significantly lower incidence of the disease after 7 years of daily finasteride.10 Other chemoprevention studies are underway, including one comparing vitamin E and selenium with placebo.
Biomarkers
Few areas of oncology have a tumor marker as valuable for cancer detection as urology does in PSA. After definitive therapy for prostate cancer, a rise in PSA almost invariably is indicative of progressive disease. Furthermore, PSA has some prognostic significance prior to therapy: the lower the PSA level, the less likely the disease will recur; the higher the level, the less likely the cancer will be curable. One of the current debates is whether patients should have a supersensitive PSA test after surgery or radiation to detect very early recurrence. At the present time, only approximately 30% of men whose PSA rises above 0.4 ng/mL will eventually develop metastatic disease. Thus the significance of a supersensitive test value of 0.06 ng/mL has uncertain clinical significance but most certainly will add to patient anxiety.
One area of debate is the PSA value that is indicative of treatment failure after radiation therapy. The American Society for Therapeutic Radiology and Oncology Consensus Panel has defined PSA failure as 3 consecutive increases in a patient’s PSA level.11 More recently, however, this definition has come under criticism because it underestimates the true failure rate. Regardless of the value, a persistently rising PSA level means the patient is experiencing disease progression.
Data are also accruing regarding the prognostic importance of PSA. One recent study found that men whose PSA increased by 2 ng/mL or more in the year before diagnosis have a significantly higher mortality from prostate cancer despite attempted curative therapy.12 The time to a rise in PSA after surgery also has important prognostic value. Men whose PSA doubles in less than 3 months have a much lower overall survival than those with longer doubling times (Figure 2).13
Figure 2.
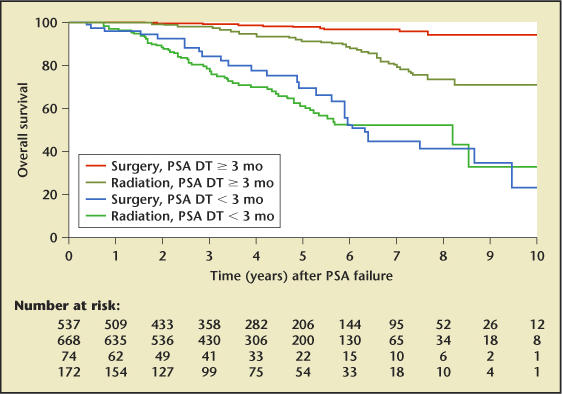
Prostate-specific antigen doubling time (PSA DT) for predicting overall survival. Reproduced, with permission, from D’Amico et al.13
Another prognostic factor is the time to the first increase in PSA. A PSA that rises above 0.4 ng/mL within 3 years of surgery carries a significantly higher mortality risk than a rise that occurs beyond that time.14 What remains unclear with relation to these prognostic markers, however, is whether there is a survival benefit to offering these high-risk men adjuvant therapy and, if so, when the optimal time to initiate that therapy is.
Routine Screening for Prostate Cancer: Is It Worthwhile?
The value of PSA as a tumor marker has led to extensive studies into its use for early diagnosis and screening. The pendulum swings back and forth regarding its value as a screening tool. Despite the large volume of published papers, the true value of PSA for routine screening remains unknown. Most people do not understand, however, why there is still uncertainty because there is no question that early detection has improved and more curable cancers are being diagnosed. Also, the proportion of men diagnosed with metastatic disease has dropped to almost zero. Are not these accomplishments sufficient proof that screening is worthwhile? Unfortunately, the answer is no because both of those findings could be due to lead and length time biases and overdetection rather than a true benefit of screening.15 Lead time bias means that cancers are diagnosed at an earlier point in time during the course of the disease without leading to a change in the timing of the eventual outcome. In other words, those men with aggressive cancers still go on to die of their disease without living any longer than if they had been detected later without early PSA testing. This can occur because very aggressive, life-threatening cancers may develop micrometastases very early in the course of disease before a PSA test would become abnormal. These metastases are not detectable when the cancer is diagnosed and will eventually lead to that person’s death.
A second reason existing data do not prove screening saves lives is length time bias, which means that there are essentially 2 types of cancers: those that grow slowly and those that grow and spread rapidly. Screening is more likely to detect the slow-growing cancers because the faster-growing cancers progress and cause symptoms that lead a patient to seek care before he can be diagnosed by screening. When the results of these 2 groups are compared, the survival of patients whose cancers were diagnosed by screening looks better, but it does not mean that the screening is saving lives.
A third reason screening may not lower mortality despite improving early detection is overdetection. It is widely known that approximately 12 million men have prostate cancer cells in their bodies, yet only a small fraction of them will die of this disease even if not treated. Although screening may detect many of these cancers at an early stage, and the cancers are then cured by treatment, the treatment is unnecessary because the cancer is not life threatening. Only a randomized study comparing a screened group with an unscreened group will make it possible to determine whether screening saves lives. Two such studies are underway, one in the United States and the other in Europe, but no results are yet available.
Until those studies are completed, some message must be provided to the public. Should men be told to get screened now despite no proof of benefit, or should they wait perhaps years before being tested and possibly miss out on the chance for early detection? A responsible approach is to present a balanced explanation of the uncertainty of screening that includes the risks and benefits so each man can choose for himself whether to be screened. Everyone should be told that the message about screening is mixed; screening will be good for some but not for most men. It is quite clear that screening increases the chances of finding a potentially curable cancer and offers a man peace of mind if he is found not to have cancer. However, the chances of benefitting from screening are likely to be quite small. On average, the odds of finding cancer in asymptomatic men who undergo a PSA test is approximately 4%. A recent Scandinavian randomized study comparing surgery with watchful waiting in men with localized disease found that the death rate was 4.8% lower in 10 years in men undergoing surgery.16 If these results were applied in the United States, they would translate into approximately 1.9 men avoiding death in 10 years per 1000 men who undergo a screening test (0.04 × 0.048). If avoiding metastatic disease is included, then approximately 4 men per 1000 tested could be better off, but at what price? Approximately 150 men out of 1000 will undergo at least 1 biopsy, and at least 10 out of 150 will have a second biopsy and at least 40 out of 1000 will undergo treatment, with 20% to 60% of these men becoming impotent and 20% of them suffering from some degree of urinary incontinence.
When presented with this information, men’s reactions will be mixed. Most may feel that avoiding death is worthwhile regardless of the risk of side effects, whereas others may feel that the benefits do not outweigh the risks. Ultimately, this is an individual’s decision. For a patient to make an informed choice, clinicians need to provide each man with enough data to be able to choose. Without appropriate information, men are forced to “place” a bet without knowing the odds of winning or losing. Unfortunately for patients, few doctors are capable of providing the true odds to their patients. When presenting the side effects of radical prostatectomy or radiation therapy to patients, most urologists do not provide statistics on the actual risks, and if they do they quote data from published reports by high-volume specialists rather than relating their own results. Studies have shown that complications are directly related to the number of radical prostatectomies performed by a surgeon; low-volume surgeons have higher complication rates than high-volume surgeons.17 Although validated written surveys are available for measuring outcomes, physicians need to be encouraged to use them. In addition, methods are needed to distinguish men who have comorbid diseases or worse cancers so that not all cases are put into the same group. Increasing use of these surveys will enable patients to make a more informed choice.
There are other unresolved issues about using this marker for early detection. At what PSA level should a biopsy be performed? How many cores should be taken? If a biopsy is negative, should it ever be repeated and, if so, under what conditions? When PSA first came into use, a value greater than 4.0 ng/mL was thought to represent an abnormality warranting ultrasound and biopsy. In the past few years, however, some clinicians have suggested that recommending a biopsy for men whose PSA is greater than 2.5 ng/mL will significantly increase sensitivity without causing too much overtreatment.18 More recently, however, the Prostate Cancer Prevention Trial (PCPT), which evaluated the ability of finasteride to prevent cancer, found that even at the very low PSA levels of less than 0.5 ng/mL or 0.5–1.0 ng/mL, 6.6% and 10% of men, respectively, will have a positive prostate biopsy (Table 1).10 This finding means that no man can be told definitively that he does not have prostate cancer, regardless of his PSA level. The use of PSA for detecting cancer has been heavily criticized by Stamey and colleagues, who believe that elevations in PSA from 4 ng/mL to 10 ng/mL are caused more by benign prostatic hyperplasia than by cancer.19 Thus, the problem with PSA as a screening tool is that the overall sensitivity may be too low to effectively reduce mortality. Of course, increasing the sensitivity of PSA may result in more overdetection of the disease, causing greater harm than good because the overwhelming majority of men with prostate cancer cells will die of something other than this disease without ever suffering any morbidity from it.
Table 1.
Prostate Cancer (CaP) in Men with Low Prostate-Specific Antigen (PSA)
| PSA level (ng/mL) | Men with CaP (%) | High-grade CaP (%) |
|---|---|---|
| < 0.5 | 6.6 | 12.5 |
| 0.6–1.0 | 10.1 | 10.0 |
| 1.1–2.0 | 17.0 | 11.8 |
| 2.1–3.0 | 23.9 | 19.1 |
| 3.1–4.0 | 26.9 | 25.0 |
Reprinted from Thompson IM, Pauler DK, Goodman PJ, et al. Prevalence of prostate cancer among men with a prostate-specific antigen level ≤ 4.0 ng per milliliter. N Engl J Med. 2004;350:2239–2246. Copyright © 2004 Massachusetts Medical Society. All rights reserved.
Rather than setting a cutoff PSA level at which to recommend biopsy, perhaps the rate of change could be a useful indicator. Studies have found that a PSA velocity greater than 0.75 ng/mL/year for approximately 18 months is a good surrogate for the presence of cancer.
When to repeat the biopsy is another uncertainty. Measuring free and bound PSA may be helpful, but fluctuations in PSA could lead to many unnecessary biopsies. Sustained increases in PSA are also a good indicator, but this issue is far from resolved. More information is needed to guide clinicians in making good decisions about repeat biopsies.
Other Tumor Markers
Ultimately, PSA may not be a good enough marker to be used as a routine screening tool. Molecular biology has led to a rapid rise in the number of potential new prostate tumor markers, which may eventually overcome the weaknesses of PSA. Unfortunately, discovering new markers is a much easier task than proving their value as screening tools. The tests required to validate them are extensive and expensive, and no new marker is close to having enough data to qualify it as a PSA replacement. The requirements for a new tumor marker are clear. It should distinguish between benign and malignant disease; it should have a high sensitivity, specificity, and positive and negative predictive value; it should diagnose potentially life-threatening tumors rather than slow-growing ones; and it should be inexpensive and easy to use. Some of the more promising markers are prostate-specific membrane antigen, early prostate cancer antigen, genetic markers including PCA3, hypermethylation, and a fused gene product of TMPRSS2 and ERG or ERTV1 genes.20,23 Each of these has respectable characteristics in very limited testing but probably not adequate sensitivity and specificity for screening. They may, however, have advantages as tumor markers but only time will tell.
Summary
Considerable progress has occurred in the diagnosis and management of prostate cancer. More is understood about the risk factors for the disease, possible ways to prevent it, and new ways to diagnose and monitor it. These developments have already translated into better patient care while also identifying where further improvements are needed.
Main Points.
Beginning in 1988, a marked increase in detection of prostate cancer occurred owing to the development of a test for prostate-specific antigen (PSA). The detection rate began to decline in 1997 but began to increase again in 2000.
Clinicians are becoming more knowledgeable about prostate cancer and the risk factors contributing to its development and progression, the most significant of which is age. Other risk factors include race, diet, and genetic changes.
After definitive therapy for prostate cancer, a rise in PSA level almost invariably is indicative of progressive disease. Furthermore, PSA has some prognostic significance before therapy: the lower the PSA level, the less likely the disease will recur; the higher the level, the less likely the cancer will be curable.
The problem with PSA as a screening tool is that the overall sensitivity may be too low to effectively reduce mortality. Of course, increasing the sensitivity of PSA may result in more overdetection of the disease, causing greater harm than good because the overwhelming majority of men with prostate cancer cells will die of something other than this disease without ever suffering any morbidity from it.
The requirements for a new prostate cancer marker are clear. It should distinguish between benign and malignant disease; it should have a high sensitivity, specificity, and positive and negative predictive value; it should diagnose potentially life-threatening tumors rather than slow-growing ones; and it should be inexpensive and easy to use.
References
- 1.Cancer Facts and Figures 2005. Atlanta: American Cancer Society, Inc.; 2005. [Google Scholar]
- 2.Hsing AW, Tsao L, Devesa SS. International trends and patterns of prostate cancer incidence and mortality. Int J Cancer. 2000;85:60–67. doi: 10.1002/(sici)1097-0215(20000101)85:1<60::aid-ijc11>3.0.co;2-b. [DOI] [PubMed] [Google Scholar]
- 3.Hankey BF, Feuer EJ, Clegg LX, et al. Cancer surveillance series: interpreting trends in prostate cancer-part I: evidence of the effects of screening in recent prostate cancer incidence, mortality, and survival rates. J Natl Cancer Inst. 1999;91:1017–1024. doi: 10.1093/jnci/91.12.1017. [DOI] [PubMed] [Google Scholar]
- 4.Messing EM, Manola J, Sarosdy M, et al. Immediate hormonal therapy compared with observation after radical prostatectomy and pelvic lymphadenectomy in men with node-positive prostate cancer. N Engl J Med. 1999;341:1781–1788. doi: 10.1056/NEJM199912093412401. [DOI] [PubMed] [Google Scholar]
- 5.Bolla M, Collette L, Blank L, et al. Long-term results with immediate androgen suppression and external irradiation in patients with locally advanced prostate cancer (an EORTC study): a phase III randomised trial. Lancet. 2002;360:103–108. doi: 10.1016/s0140-6736(02)09408-4. [DOI] [PubMed] [Google Scholar]
- 6.D’Amico AV, Manola J, Loffredo M, et al. 6-month androgen suppression plus radiation therapy vs radiation therapy alone for patients with clinically localized prostate cancer: a randomized controlled trial. JAMA. 2004;292:821–827. doi: 10.1001/jama.292.7.821. [DOI] [PubMed] [Google Scholar]
- 7.Sakr WA, Haas GP, Cassin BF, et al. The frequency of carcinoma and intraepithelial neoplasia of the prostate in young male patients. J Urol. 1993;150:379–385. doi: 10.1016/s0022-5347(17)35487-3. [DOI] [PubMed] [Google Scholar]
- 8.Haas GP, Sakr WA. Epidemiology of prostate cancer. CA Cancer J Clin. 1997;47:273–287. doi: 10.3322/canjclin.47.5.273. [DOI] [PubMed] [Google Scholar]
- 9.Carroll KK, Khor HT. Dietary fat in relation to tumorigenesis. Prog Biochem Pharmacol. 1975;10:308–353. [PubMed] [Google Scholar]
- 10.Thompson IM, Pauler DK, Goodman PJ, et al. Prevalence of prostate cancer among men with a prostate-specific antigen level ≤ 4.0 ng per milliliter. N Engl J Med. 2004;350:2239–2246. doi: 10.1056/NEJMoa031918. [DOI] [PubMed] [Google Scholar]
- 11.Consensus statement: guidelines for PSA following radiation therapy, authors. American Society for Therapeutic Radiology and Oncology Consensus Panel. Int J Radiat Oncol Biol Phys. 1997;37:1035–1041. [PubMed] [Google Scholar]
- 12.D’Amico AV, Chen MH, Roehl KA, Catalona WJ. Preoperative PSA velocity and the risk of death from prostate cancer after radical prostatectomy. N Engl J Med. 2004;351:125–135. doi: 10.1056/NEJMoa032975. [DOI] [PubMed] [Google Scholar]
- 13.D’Amico AV, Moul JW, Carroll PR, et al. Surrogate end point for prostate cancer-specific mortality after radical prostatectomy or radiation therapy. J Natl Cancer Inst. 2003;95:1376–1383. doi: 10.1093/jnci/djg043. [DOI] [PubMed] [Google Scholar]
- 14.Pound CR, Partin AW, Eisenberger M, et al. Natural history of progression after PSA elevation following radical prostatectomy. JAMA. 1999;281:1591–1597. doi: 10.1001/jama.281.17.1591. [DOI] [PubMed] [Google Scholar]
- 15.Cookson MS. Prostate cancer: screening and early detection. Cancer Control. 2000;8:133–140. doi: 10.1177/107327480100800203. [DOI] [PubMed] [Google Scholar]
- 16.Bill-Axelson A, Holmberg L, Ruutu M, et al. Radical prostatectomy versus watchful waiting in early prostate cancer. N Engl J Med. 2005;352:1977–1984. doi: 10.1056/NEJMoa043739. [DOI] [PubMed] [Google Scholar]
- 17.Hu JC, Gold KF, Pashos CL, et al. Role of surgeon volume in radical prostatectomy outcomes. J Clin Oncol. 2003;21:401–405. doi: 10.1200/JCO.2003.05.169. [DOI] [PubMed] [Google Scholar]
- 18.Punglia R, D’Amico AV, Catalona W, et al. Effect of verification bias on screening for prostate cancer by measurement of prostate-specific antigen. N Engl J Med. 2003;349:335–342. doi: 10.1056/NEJMoa021659. [DOI] [PubMed] [Google Scholar]
- 19.Stamey TA, Caldwell M, McNeal JE, et al. The prostate specific antigen era in the United States is over for prostate cancer: what happened in the last 20 years? J Urol. 2004;172:1297–1301. doi: 10.1097/01.ju.0000139993.51181.5d. [DOI] [PubMed] [Google Scholar]
- 20.Carter HB, Morrell CH, Pearson JD, et al. Estimation of prostatic growth using serial prostate-specific antigen measurements in men with and without prostate disease. Cancer Res. 1992;52:3323–3328. [PubMed] [Google Scholar]
- 21.Paul B, Dhir R, Landsittel D, et al. Detection of prostate cancer with a blood-based assay for early prostate cancer antigen. Cancer Res. 2005;65:4097–4100. doi: 10.1158/0008-5472.CAN-04-4523. [DOI] [PubMed] [Google Scholar]
- 22.Bussemakers MJ, van Bokhoven A, Verhaegh GW, et al. DD3: a new prostate-specific gene, highly overexpressed in prostate cancer. Cancer Res. 1999;59:5975–5979. [PubMed] [Google Scholar]
- 23.Tomlins SA, Rhodes DR, Perner S, et al. Recurrent fusion of TMPRSS2 and ETS transcription factor genes in prostate cancer. Science. 2005;310:644–648. doi: 10.1126/science.1117679. [DOI] [PubMed] [Google Scholar]


