Abstract
Background
Radon is a ubiquitous noble gas in the environment and a primary source of harmful radiation exposure for humans; it decays in a cascade of daughters (RAD) by releasing the cell damaging high energy alpha particles.
Results
We studied natural distribution of RAD 210Po and 210Bi in the different parts of the postmortem brain of 86-year-old woman who had suffered from Alzheimer's disease (AD). A distinct brain map emerged, since RAD distribution was different among the analyzed brain areas. The highest RAD irradiation (mSv·year-1) occurred in the decreasing order of magnitude: amygdale (Amy) >> hippocampus (Hip) > temporal lobe (Tem) ~ frontal lobe (Fro) > occipital lobe (Occ) ~ parietal lobe (Par) > substantia nigra (SN) >> locus ceruleus (LC) ~ nucleus basalis (NB); generally more RAD accumulated in the proteins than lipids of gray and white (gray > white) brain matter. Amy and Hip are particularly vulnerable brain structure targets to significant RAD internal radiation damage in AD (5.98 and 1.82 mSv·year-1, respectively). Next, naturally occurring RAD radiation for Tem and Fro, then Occ and Par, and SN was an order of magnitude higher than that in LC and NB; the later was within RAD we observed previously in the healthy control brains.
Conclusion
Naturally occurring environmental RAD exposure may dramatically enhance AD deterioration by selectively targeting brain areas of emotions (Amy) and memory (Hip).
Background
Alzheimer's Disease (AD), the most common cause of dementia in the elderly, is a progressive neurodegenerative disease of unknown origin that gradually robs the patient of cognitive function and eventually causes death [1]. Recently, we showed that radon daughters 210Po and 210Bi (RAD), accrue selectively in the brain proteins and lipids of men and women who suffered from AD and Parkinson's Disease, respectively [2,3]. We proposed that AD is a systemic brain-cell disease which selectively involves the cell membrane protein structures of ion gates, pores, and channels, with consequent chlorine leaking into the cells, collapse of the cell membrane gradient, and functional cell death. Other authors proposed calcium and potassium channel impairment, respectively [4,5]; the latter authors proposed AD to be a general systemic disease of the body as somatic fibroblasts showed the same Ca-channel defect as that of neurons. Thus, the pathological substrate of AD may well be described as "channelopathy", a condition where the impaired cell membrane protein structures lead to the deregulation of the ionic influx by the brain cells [6-9].
Most of the AD studies are limited to the brain cortex and limbic system, notably the hippocampus, since these are the well recognized brain areas involved in the AD memory loss [10]; the role of other brain structures in AD is poorly understood [11-13]. In this case report, we studied the distribution of naturally occurring environmental RAD in different brain areas in AD. Radon is a ubiquitous noble gas in the air we breath [14], it is lipid soluble and (in spite of being a noble gas) capable of forming weak chemical bonds [15], and tends to accumulate in high-carbon body fat tissue including the brain [16]. Radon and RAD are the source of four cell destructive high energy alpha particles which may significantly contribute to the internal radiation dose of the brain and play a role in AD etiology and pathology [17,18].
Subject and methods
We studied the distribution of naturally occurring environmental RAD polonium-210 (210Po; alpha particle emitter) and bismuth-210 (210Bi; beta particle emitter) in nine different brain regions of an 86-year-old deceased woman. She was a resident of North Dakota who suffered from AD at old age and with otherwise uneventful medical history. Post-mortem samples were obtained from all four brain lobes, i.e., frontal (Fro). parietal (Par), occipital (Occ), and temporal (Tem), and from the five well defined inside the brain structures, i.e., the hippocampus (Hip), amygdale (Amy), substantia nigra (SN), locus ceruleus (LC), and nucleus basalis (NB). The pathological diagnosis of AD was based on the presence of an age-adjusted moderate to a severe number of plaques in the neocortex [19]. We separated cortical gray and subcortical white matter from each brain lobe and separate gray and adjacent white matter for every subcortical ganglion. One gram of each sample was fractionated into protein and lipid content before assessing 210Bi and 210Po activity separately.
Quantitative determination of the proteins and lipids from selected brain regions and their 210Po and 210Bi radioactivity was performed as described previously [2,3]. In brief, the proteins and lipids were extracted from the gray and white matter of the brain and ganglia by following the respective methods of Bradford [20] and Folch et al. [21]. The protein fraction was passed through a polymembrane under a negative pressure gradient [22]. The 210Po from the samples and 208Po from a spiked solution were plated on a silver disc [23]. Alpha and beta particle activities were determined in an Alpha Spectrometer System supplied with a radionuclide library software package (EG&G ORTEC, Oak Ridge, TN) and a Beckman scintillation spectrometer (Beckman Co., Fullerton, CA), respectively.
Lead-210 (210Pb) decays to 210Bi, which in turn decays to 210Po; each decays at a different rate. After 600 days (1.64 years) a "secular equilibrium" is reached; the activities of the 210Bi and 210Po in the sample are then equal to the 210Pb activity. The standard Bateman differential equation of growth and decay of radio-nuclides in a decay chain was used to correct for 210Po formation from 210Bi directly and from 210Pb indirectly via 210Bi [24].
The radioactivity of 210Bi and 210Po was assessed in replicates of every studied brain area and expressed in μBq·g-1 tissue (1 μBq equals 1 disintegration per 106 second, i.e., 31 disintegrations per year). The difference between the two replicates of the same sample and for the same radionuclide didn't exceed 5%. We consider the difference in the RAD radionuclide retention between any two brain areas of >20% to be significant (>>), that of 10–20% to be probably significant (>), and that below 10% as non-significant (~). The normal range of the biological variability is ± 20%, and the maximal acceptable difference between the 210 Po and 210Bi in the same brain area sample was set at 10%.
The particular brain area cell death radiation risk to the high energy RAD alpha particles was calculated on the assumption of the brain cell density of 6.4·106·g-1 (90% glia, 10% neurons) and the average weight of the adult female brain of 1250 g [25,26]; the brain weight of our subject was in that category. The absorbed physical energy was first expressed in micro Grays (μGy) and then transformed to mili Sieverts (mSi) to provide for the assessment of the biological effective dose of radiation [27]. It should be noted that every single 210Po disintegration means an instant death to a minimum of three cells along the path of its high energy (5.305MeV) decaying alpha particle [28]; altogether, there are four such "killing" alpha particles in the radon decay chain (Fig 1)[15].
Figure 1.
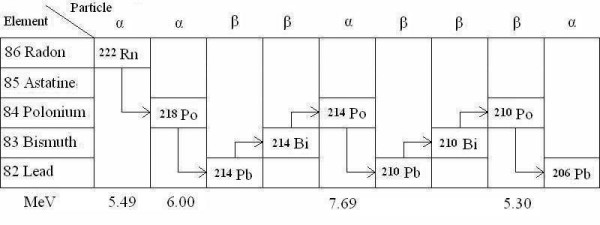
Radon (222Rn) radiation decay.
This research was approved by the University of North Dakota Institutional Review Board, Grand Forks, ND (IRB-9509-027), and carried out in full compliance with Helsinki Declaration.
Results
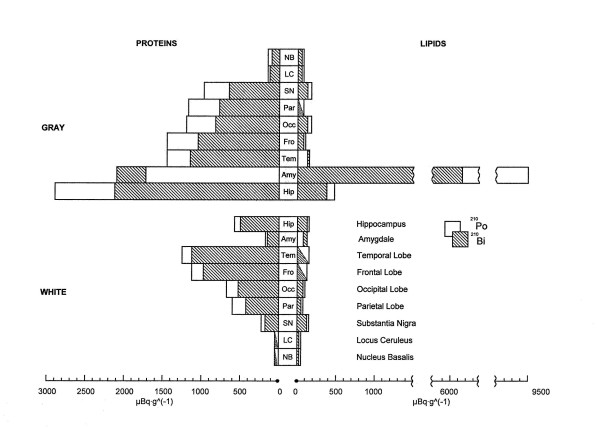
The results showed a highly selective distribution of 210Po and 210Bi in the proteins and lipids from the gray and white matter of the brain and ganglia in AD; indeed, RAD accumulation differs significantly between the brain areas (Fig 2). We observed a very good congruence of 210Po and 210Bi in all the duplicate samples from the same brain areas, indicating reliability of the results obtained by the two different analytical methods of alpha and beta particle counting; values for 210Po tend to be somewhat higher than that of 210Bi but still within the accepted limit of accuracy. As a rule, RAD accumulation was higher in the proteins than lipids of various brain structures, and higher in the gray than white brain matter proteins, respectively.
Figure 2.
Brain structure distribution of polonium-210 (□) and bismuth-210 (■) in the proteins and lipids of the gray and white brain matter in an Alzheimer disease victim (μBq g-1 tissue).
The retention of RAD in the gray brain matter proteins was, in the decreasing order of magnitude:
Hip >> Amy >> Tem ~ Fro > Occ ~ Par > SN >> LC ~ NB. (A)
RAD retention was generally lower in the white matter brain and cerebral ganglia proteins than that of the gray brain matter. The comparable sequence for both RAD (210Po and 210Bi) in the white brain matter proteins was somewhat different from that in the grey matter:
Tem > Fro>> Occ ~ Par ~ Hip >> SN > Amy >> LC ~ NB (B)
It should be noted that the range of the 210Po activity in the gray matter proteins of the different brain structures (A) may be as low as 150 μBq for NB and as high as 2906 μBq in the Hip [or 100 vs. 2138 μBq (per gram) of 210Bi for the same brain structures], a factor of 20 difference! A similar range was also observed in the proteins from the brain white matter (B), although the actual sequence was somewhat different (50 vs. 1152 μBq·g-1of 210Po and 50 vs. 1119 μBq·g-1of 210Bi for NB and Tem, respectively).
The notable exception to the uniformly higher RAD accumulation in the gray (A) and white (B) brain matter proteins than lipids in AD was the accentuated RAD retention in the amygdale lipids. Indeed, the retention of 210Po and 210Bi in the Amy lipids reached astonishing 9285 and 6162 μBq·g-1, respectively, well above anything we have observed of RAD in any other brain area. The retention of 468 μBq·g-1 of 210Po and 362 μBq·g-1 of 210Bi in the Hip was the next highest for the RAD retention in the lipids. Although increased in relation to the other lipid RAD, the Hip lipid RAD retention was at the lower end of the RAD activities seen in the proteins.
To better assess the radiation risk from 210Po "killer" alpha particles over the last year of the subject's life, we combined together 210Po activity in the gray brain matter proteins and lipids, since proteins and lipids from the same anatomical structure are not naturally separated. Apparently, a substantial brain cell loss should have occurred in the Amy and Hip as a result of the cell killing potential of the high energy RAD alpha particles (Table 1) [29]. The decreasing order of magnitude sequence of RAD 210Po in proteins and lipids from different brain areas showed the radiation risk to be:
Table 1.
Estimated annual regional brain cell loss and cell dose per gram tissue from 210Po high energy alpha particles (5.305 MeV) in the proteins and lipids.*
| Brain structure | Emissions | Estimated cell loss | Cell dosimetry Energy absorbed | Biolgical effectiveness | |
| Bq•y•g-1 | Number | %•y-1 | βGy•yr-1 | mSv•y-1 | |
| Amygdale | 347 828 | 1 043 484 | 16.65 | 295 | 5.90 |
| Hippocampus | 106 475 | 319 426 | 4.99 | 90 | 1.80 |
| Temporal lobe | 50 078 | 150 233 | 2.35 | 42 | 0.85 0.79a |
| Frontal lobe | 49 545 | 148 635 | 2.32 | 42 | 0.84 0.75a |
| Occipital lobe | 43 739 | 131 217 | 2.05 | 37 | 0.74 |
| Parietal lobe | 39 005 | 117 016 | 1.83 | 33 | 0.66 |
| Substantia nigra | 30 855 | 92 564 | 1.45 | 27 | 0.54 |
| Locus ceruleus | 6 848 | 20 544 | 0.30 | 6 | 0.12 |
| Nucleus basalis | 6 848 | 20 544 | 0.30 | 6 | 0.12 |
*The average person absorbs ≈450 βGy of cosmic radiation in a year. Since all 210Po radiation is composed solely of alpha particles then a relative effective factor is 20 (1 Sv = 20 Gy) [29].
aMomčilović et al.[2,3].
Amy >> Hip > Tem ~ Fro > Occ ~ Par > SN >> LC ~ NB (C)
We estimated that a minimum of 15% of amygdale cell population per gram tissue (>106 cells) was destroyed over a single year, about 5% of hippocampus cell population was also destroyed over the same time, and as much as 1–2% of that in the four brain lobe cell population. Since the brain lobes have a total cell mass considerably greater than that of Amy and Hip, the actual cell loss from brain lobes would be also substantial. Evidently, the different areas of the AD brain are exposed to a different radiation risk and consequent cell loss.
Discussion
The major finding of this case report is that the explored areas of the AD brain are specifically and selectively targeted by RAD, so that there is a different radiation risk to the various brain structures at the same environmental radon and RAD exposure. It has never before been observed that naturally occurring environmental RAD can reach dangerous levels of radiation exposure in certain brain areas such as Amy and Hip. These findings also confirmed our previous observations about brain proteins as a targeted biochemical compartment in the AD brain. Indeed, the RAD deposition in the gray and white brain matter proteins and lipids from the frontal and temporal brain lobes of this single subject were within the average values we reported for the same brain structures in a group of people who suffered from AD (see Table 1) [2,3]. Consequently, if the RAD deposition to the proteins and lipids of the two identical brain regions in these two separate studies is approximately equal, it is reasonable to assume that RAD distribution in all the other areas of the AD brain would also have the same pattern of distribution as found here. North Dakota is known for it's high RAD [31], but the RAD in the frontal and temporal region of this case is very well within the average RAD from the same brain region samples obtained from the Alzheimer's Foundation [2,3]. Since the Alzheimer's Foundation brains came from the different parts of the USA, it appears that, according to our instrument limits, the regional environmental exposure to RAD did not affect the RAD brain distribution.
Our study identified the proteins in the hippocampus and amygdale as a two primary brain area targets for RAD in AD. Further, we noted that there was two times more RAD deposited in hippocampus than in the cortex per unit mass of the protein. This is an indicative ratio since the Hip is assembled of three cellular layers identical with three out of six cell layers of the brain cortex [25]. Thus, if three Hip layers yield two times more RAD than the six layers of the brain cortex, and three of the cortex layers are identical with those of Hip, it is evident that the presence of three more (but different) cortical layers did not contributed to the RAD; therefore we think that our finding supports the concept of laminar specificity of cortical pathology in AD [10] We also noted biochemical similarity of RAD retention in the parietal and occipital lobe proteins vs. that in respective frontal and temporal lobes; the later was higher (C).
The lowest RAD retention was observed in the proteins of locus ceruleus and nucleus basalis, otherwise an area where a great neuronal loss was reported for LC, NB, and SN in AD subjects [13,31]. Since neurons are also composed of proteins, this observation implies how proteins from these brain areas may have either different affinity for RAD or, perhaps, the fact that we analyzed proteins in both neurons and glia cells of the brain; the later is much more abundant (90%). Events like impaired conformational changes in protein post-synaptic scaffolding [32], the fall in number of neuron synaptic contacts [33] the failing support of astrocytes which are especially vulnerable to the ionic radiation [34], and cyto-architectonic collapse of functional neurons [35], may all precede the neuronal loss in AD brain.
We predicted correctly that, as in the previous AD study, RAD would as well selectively accrue in the brain cell proteins of Amy and Hip [2,3]; what we didn't know at that time was that different brain areas would had quite distinct RAD affinity. Essentially, our finding of high protein RAD affinity in AD credits the importance of mal variant AD proteins in the neurons [6-8], with a caveat that actual mal variant protein biochemical structure may be quite different for various brain areas and their cell population. The reason for the repeatedly observed high RAD protein affinity and respective brain area specific protein affinity in AD remains obscure. We only know that the biochemical structure of the AD protein could be changed such that more carbon bonds would be available to moderate radon movement before radon decays in RAD; it may be even some variety of a prion protein of a chronic disease [9]. It has been shown that metals Al, Cu, Fe, Pb, Si, and Zn acts like a potent "seeding" factors inducing excessive amyloid Aβ peptide formation [36-39]. Indeed, Aβ is a major protein component of the senile plaque and what is the hallmark of AD [40]; the degradation of these excess proteins in the AD brain is further reduced by the lack of the proteosomes, a large protein complex responsible for intracellular degradation of misfolded, oxidized, or aggregated proteins [41]. Since radon is a radioactive noble gas it transfers freely across the blood-brain barrier in and out of the brain; when radon decays to RAD both the high energy of cell killing potential is released and the heavy metals generated, the later would act as a potent seeding agents for Aβ generation. Thus, radon can be both a direct cause of AD via the imunogenic debris of the killed cells of already changed proteins in the AD brain, and by also enhancing Aβ synthesis.
The only exception to the rule that AD specifically targets the brain matter proteins was that in this subject the highest RAD deposition was observed in the amygdale gray matter lipids. Since we already saw selective RAD accumulation to the brain lipids in Parkinson's disease [2,3], we concluded that an unfortunate event had occurred to this study's subject. i.e., the combined protein and lipid cell membrane chanellopathy. This failure of AD brain cell lipids may be secondary to the failures of protein folding, their conformation change, appearance of false ionic channels, and consequent failure in the Ca-channel ionic cell influx [3,42,43].
We are impressed that the tiny amygdale alone received an equivalent of 2/3 of the respective total yearly human body physical energy dose from cosmic rays (299 vs. 450 μG·y-1) [29]. The biological quality of different types of radiation is different (alpha being the most adverse to the biological tissue), and when the results are corrected for the high biological quality factor of alpha radiation, Amy will receive a fifteen-times greater biological effective radiation dose than the whole human body over the entire year (5980 vs. 450 μSi·y-1, respectively). Approximately one million of Amy cells will be killed in a year, an equivalent of one gram of that 10 gram heavy brain structure. This estimate is a very conservative approximation, based on only three direct cell kills per decayed high energy alpha particle, since as much as fifty cells may be irreparably injured and die after some delay by a single 210Po alpha particle as a result of the "bystander effect" [44]. What we observed in this AD brain is the internal radiation "amygdalectomy" and how that might explain some of the respective emotional torpidity and insensitivity associated with AD and old age. This case appears to fit the usual pattern of events in AD; on average, person spend several years in the mild or minimal stages, between 4 and 5 years in the moderate disease stages, and depending on the quality of care in the depending stages, a year or more requiring full nursing care [45].
Conclusion
In conclusion, AD is a complex and progressive brain disease characterized by the failing ability to cope with environmental xenobiotic hazards [2,3], excessive free radical injury, inflammation and immunity deficiency [46], cell repair impairment [47], and the protein synthesis [48]. The ubiquitous environmental RAD exposure, and high RAD accumulation in the sensitive brain structures may either induce or hasten or both the irreversible "shut down" process of the ailing human brain in AD.
Competing interests
The author(s) declare that they have no competing interests.
Authors' contributions
BM planned and designed the study, did data analysis and interpretation, and drafted the manuscript. GIL conceived the study, coordinated the radio analytical work, did dose calculations and helped to draft the manuscript. MC did pathology, pathological diagnosis, disease classification, brain dissection, and helped towards clinical data interpretation.
Acknowledgments
Acknowledgements
This study was supported in part by the U.S. Environmental Protection Agency contract ND92-257, the Technical Training Foundation, North Andover, MA, U.S.A., the Ministry of Science, Education and Sport of the Republic of Croatia grant 0022013, and the generous philanthropic support of RCS Trading Co. Ltd., Isle of Man, UK. We wish to thank Hassaan A. Alkhatib, PhD and John Duerre, Prof. Emeritus Microbiology and Immunology for their help.
Contributor Information
Berislav Momčilović, Email: momcilovic@mimi.imi.hr.
Glenn I Lykken, Email: glenn_lykken@und.nodak.edu.
Marvin Cooley, Email: marvin_cooley@und.nodak.edu.
References
- Cummings JL, Cole G. Alzheimer disease. JAMA. 2002;287:2335–2338. doi: 10.1001/jama.287.18.2335. [DOI] [PubMed] [Google Scholar]
- Momčilović B, Alkhatib HA, Duerre JA, Cooley MA, Long WM, Harris TR, Lykken GI. Environmental radon daughters reveal pathognomonic changes in the brain proteins and lipids in patients with Alzheimer's disease and Parkinson's disease and cigarette smokers. Arh hig rada tokiskol. 1999;50:347–369. [PubMed] [Google Scholar]
- Momčilović B, Alkhatib HA, Duerre JA, Cooley MA, Long WM, Harris TR, Lykken GI. Environmental lead-210 and bismuth-210 accrue selectively in the brain proteins in Alzheimer disease and brain lipids in Parkinson disease. Alzheimer Dis Assoc Disord. 2001;15:106–115. doi: 10.1097/00002093-200104000-00012. [DOI] [PubMed] [Google Scholar]
- Arsipe N, Pollard HB, Rojas E. Giant multilevel channels formed by Alzheimer disease amyloid β-protein [AβP-(1–40)] in bilayer membranes. Proc Natl Acad Sci USA. 1993;90:10573–10577. doi: 10.1073/pnas.90.22.10573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Etcheberrigaray R, Ito E, Oka K, Tofel-Grehl B, Gibson GE, Alkon DL. Potassium channel dysfunction in fibroblasts identifies patients with Alzheimer disease. Proc Natl Acad Sci USA. 1993;90:8209–8213. doi: 10.1073/pnas.90.17.8209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Etcheberrigaray R, Ibarreta D. Alteraciones de canales ionicos y segundos mensajeros en la enfermedad de Alzheimer. Relevancia de estudios en celulas extraneurales. Rev Neurol. 2001;33:740–749. [PubMed] [Google Scholar]
- Trojanowski JQ. Tauists, baptists, syners, apostates, and new data. Ann Neurol. 2002;51:263–265. doi: 10.1002/ana.10281. [DOI] [PubMed] [Google Scholar]
- Ingram V. Alzheimer's disease. Am Sci. 2003;91:312–321. [Google Scholar]
- Quist A, Doudevski I, Lin H, Azimova R, Ng D, Frangione B, Kagan B, Ghiso J, Lal R. Amyloid ion channels; a common structural link for protein-misfolding disease. Proc Natl Acad Sci USA. 2005;102:10427–10432. doi: 10.1073/pnas.0502066102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Hoesen GW, Solodkin A. Cellular and systems neuroanatomical changes in Alzheimer's disease. Ann New York Acad Sci. 1994;747:12–35. doi: 10.1111/j.1749-6632.1994.tb44399.x. [DOI] [PubMed] [Google Scholar]
- DeKosky ST, Ikonomovic MD, Styren SD, Beckett L, Wisniewski S, Bennett DA, Cochran EJ, Kordower JH, Mufson EJ. Upregulation of choline acetyltransferase activity in hippocampus and frontal cortex of elderly subjects with mild cognitive impairment. Ann Neurol. 2002;51:145–155. doi: 10.1002/ana.10069. [DOI] [PubMed] [Google Scholar]
- Ishunina TA, Fisser B, Swaab DF. Sex difference in androgen receptor immunoreactivity in Basal forbrain nuclei of elderly and Alzheimer patients. Exp Neurol. 2002;176:122–132. doi: 10.1006/exnr.2002.7907. [DOI] [PubMed] [Google Scholar]
- Zarow C, Lyness SA, Mortimer JA, Chui HC. Neuronal loss is greater in the locus coeruleus than nucleus basalis and substantia nigra in Alzheimer and Parkinson disease. Arch Neurol. 2003;60:337–341. doi: 10.1001/archneur.60.3.337. [DOI] [PubMed] [Google Scholar]
- Eicholz GG. Human exposure. In: Cothern CR, Smith JE Jr, editor. Environmental radon. New York, Plenum Press; 1987. pp. 131–213. [Google Scholar]
- Cothern CR, Smith JE., Jr . Environmental radon. New York, Plenum Press; 1987. Radioactive decay; pp. 307–315. [Google Scholar]
- Nussbaum E. Research and evelopment reports UR503. Rochester NY, University of Rochester; 1957. Radon solubility in body tissues and in fatty acids. [Google Scholar]
- Momčilović B, Lykken GI. On men and radon – A noble gas of many disguises. In: Garaj-Vrhovac V, Kopjar N, Miljanić S, Zagreb, Croatia, editor. Part I, Proceedings of the VI Symposium of the Croatian Radiation Protection Society. Croatian Radiation Protection Association; 2005. pp. 235–239. [Google Scholar]
- Momčilović B, Lykken GI. On men and radon – A noble gas of many disguises. In: Garaj-Vrhovac V, Kopjar N, Miljanić S, Zagreb, Croatia, editor. Part II, Proceedings of the VI Symposium of the Croatian Radiation Protection Society. Croatian Radiation Protection Association; 2005. pp. 240–247. [Google Scholar]
- Mirra SS, Heyman A, McKeel D. The consortium to establish a registry for Alzheimer's disease (CERAD). II. Standardization of the neuropathologic assessment of Alzheimer's disease. Neurology. 1991;41:479–486. doi: 10.1212/wnl.41.4.479. [DOI] [PubMed] [Google Scholar]
- Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:34–38. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Folch T, Lees MT, Sloan-Stanley GH. A simple method for the isolation and purification of total lipids from animal tissue. J Boil Chem. 1957;226:497–509. [PubMed] [Google Scholar]
- Gaffney JS, Orlandini KA, Marley NA. Measurements of 7Be and 210Pb in rain, snow, and hail. J Appl Meteorol. 1994;33:869–873. [Google Scholar]
- Laul JC, Smith MR, Thomas CW, Jackson PO, Hubbard N. PNL-SA-12851. Richland WA, Pacific Northwest Laboratory; 1985. Analysis of natural radio nuclides from uranium and thorium series in briny ground. [Google Scholar]
- Bateman H. The solution of a system of differential equations occurring in the theory of radioactive transformation. Proc Cambridge Philosoph Soc. 1910;15:423–427. [Google Scholar]
- Barr ML, Kiernan JA. The human nervous system. 6. Philadelphia: JB Lippincott; 1993. pp. 278–292. [Google Scholar]
- Takahashi T. Atlas of the human body. New York, Harper Perennial; 1994. [Google Scholar]
- Ronto G, Tarjan I, eds An introduction to biophysics with medical orientation. Budapest Akademiai Kiado. 1991. pp. 85–180.
- Day C. Alpha radiation can damage DNA even when it misses the cell nucleus. Phys Today. 1999;52:19–20. [Google Scholar]
- The American Heritage® Book of English Usage & 37. rad/rem/roentgen/gray/sievert. 1999. http://www.bartleby.com/64/C004/037.html
- Brekke DW. North Dakota Geological Survey Letter. University Station, Grand Forks, ND; 1987. The occurrence of radon in North Dakota; pp. 24–28. [Google Scholar]
- Whitehouse PJ, Price DL, Struble RG, Clark AW, Coyle JT, DeLong MR. Alzheimer's disease and senile dementia: Loss of neurons in the basal forebrain. Science. 1982;215:1237–1239. doi: 10.1126/science.7058341. [DOI] [PubMed] [Google Scholar]
- Conroy WG, Lin Z, Nai Q, Coggan JS, Berg DK. PDZ-containing proteins provide a functional postsynaptic scaffold for nicotinic receptors in neurons. Neuron. 2003;38:759–771. doi: 10.1016/s0896-6273(03)00324-6. [DOI] [PubMed] [Google Scholar]
- Geinsman Y, de Tolledo-Morrell, Morrell F. Comparison of structural synaptic modifications induced by long-term potentiation in the hippocampal dental gyrus of young and adult aged rats. Ann New York Acad Sci. 1994;747:452–466. doi: 10.1111/j.1749-6632.1994.tb44428.x. [DOI] [PubMed] [Google Scholar]
- Campbell B, Novick R. Effects of beta rays on central nervous tissue. Proc Soc Exp Biol Med. 1949;72:34–38. doi: 10.3181/00379727-72-17323. [DOI] [PubMed] [Google Scholar]
- Eyupoglu IY, Bechmann I, Nitsch R. Modification of microglia function protects from lesion-induced neuronal alterations and promotes sprouting in the hippocampus. FASEB J. 2003;17:110–110. doi: 10.1096/fj.02-0825fje. [DOI] [PubMed] [Google Scholar]
- Agrimi U, Gijardo Gdi. Amyloid, amyloid-inducers, cytokines and heavy metals in scarpie and other human and animal subacute spongiform encephalopathies: Some hypotheses. Medical Hypothesis. 1993;40:113–116. doi: 10.1016/0306-9877(93)90139-h. [DOI] [PubMed] [Google Scholar]
- Cherny RA, Legg JT, McLean CA, Fairlie DP, Uang X, Atwood CS, Beyreuther K, Tanzi RE, Masters CL, Bush AI. Aqueous dissolution of Alzheimer's Disease Aβ amyloid deposits by biometal depletion. J Biological Chem. 1999;274:23223–23228. doi: 10.1074/jbc.274.33.23223. [DOI] [PubMed] [Google Scholar]
- Maynard CJ, Cappai R, Volitakis I, Cherny RA, White AR, Beyreuther K, Masters CL, Bush AI, Li Q-X. Overexpression of Alzheimer's Disease amyloid-β oposes the age- dependent elevation of brain copper and iron. J Biological Chem. 2002;277:44670–44676. doi: 10.1074/jbc.M204379200. [DOI] [PubMed] [Google Scholar]
- Ritchie CW, Bush AJ, Mackinnon A, Macfarlane S, Mastwyk M, MacGregor L, Kiers L, Cherny R, Li OX, Tammer A, Carrington D, Mavros C, Volitakis I, Xilinas M, Ames D, Davis S, Beyreuther K, Tanzi RE, Masters CL. Metal-protein attenuation with iodochlorhydroxyquin (clioquinol) targeting Abeta amyloid deposition and toxicity in Alzheimer's Disease: a pilot phase 2 clinical trail. Arch Neurol. 2003;60:1685–1691. doi: 10.1001/archneur.60.12.1685. [DOI] [PubMed] [Google Scholar]
- Li R, Lindholm K, Yang L-B, Yue X, Citron M, Yan R, Beah T, Sue L, Sabbagh M, Cai H, Wong P, Price D, Shen Y. Amyloid β peptide load is correlated with increased β- secretase activity in sporadic Alzheimer's Disease patients. Proc Natl Acad Sci USA. 2004;101:3632–3637. doi: 10.1073/pnas.0205689101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flood F, Murphy S, Cowburn RF, Lannfelt L, Walker B, Johnston JA. Proteosome- mediated effects on amyloid precusor protein processing at the γ-secretase site. Biochem J. 2005;385:545–550. doi: 10.1042/BJ20041145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mattison MP. Calcium and neuronal injury in Alzheimer's disease. Ann New York Acad Sci. 1994;747:50–76. [PubMed] [Google Scholar]
- McClure RJ, Kanfer JN, Panchalingam K, Klunk WE, Pettegrew JW. Alzheimer's disease: Membrane associated metabolic changes. Ann New York Acad Sci. 1994;747:110–125. doi: 10.1111/j.1749-6632.1994.tb44404.x. [DOI] [PubMed] [Google Scholar]
- Hall EJ. The bystander effect. Health Phys. 2003;85:31–35. doi: 10.1097/00004032-200307000-00008. [DOI] [PubMed] [Google Scholar]
- Galton CJ, Hodges JG. Oxford Textbook of Medicine Warrell DA, Cox TM, Firth JD. 4. Vol. 3. Oxford Univ Press, Oxford; 2003. Alzheimer's Disease and other dementias; p. 24.13.8. [Google Scholar]
- Huang J, Kim LJ, Mealey R, Marsh HC, Jr, Zhang Y, Tenner AJ, Sander Connolly E, Jr, Pinsky DJ. Neuronal protection in stroke by an sLex-glycosylated complement inhibitory protein. Science. 1999;285:595–599. doi: 10.1126/science.285.5427.595. [DOI] [PubMed] [Google Scholar]
- Kruk PA, Rampino NJ, Bohr VA. DNA damage and repair in telomeres: Relation to aging. Proc Natl Acad Sci USA. 1995;92:258–262. doi: 10.1073/pnas.92.1.258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee C-K, Weindruch R, Prolla TA. Gene-expression profile of the ageing brain in mice. Nature Genetics. 2000;25:294–297. doi: 10.1038/77046. [DOI] [PubMed] [Google Scholar]