Abstract
UV exposure suppresses the immune response to a variety of microbial, fungal, and viral Ags. In addition, UV radiation is a complete carcinogen and the immune suppression induced by UV radiation is a major risk factor for skin cancer induction. In this study, we examined the mechanisms underlying the induction of immune suppression and tolerance induction by UV radiation. Transferring lymph nodes cells from UV-irradiated, FITC-sensitized mice into normal recipients transferred immune tolerance. Contrary to expectations, the cell responsible was an FITC+, IL-10-secreting, CD19+, B220+ B cell. Because the lipid mediator of inflammation, platelet-activating factor (PAF) is released by UV-irradiated keratinocytes and is essential for the induction of immune suppression, we determined its role in tolerance induction. When UV-irradiated mice were injected with PCA 4248, a selective PAF receptor (PAFR) antagonist, transfer of tolerance was suppressed. However, immune suppression was not transferred when FITC+ cells from the draining lymph nodes of UV-irradiated, PAFR-deficient donor mice were injected into the recipients. Because PCA 4248 also blocks serotonin receptor binding, we measured the effect that blocking both serotonin and PAFR binding has on the transfer of immune suppression. Only when both PAF and serotonin binding were blocked could we inhibit tolerance induction. These data identify a novel function for PAF and serotonin in modulating immune function, the activation of immunoregulatory B cells.
The UV radiation in sunlight, an environmental factor that humans come into contact with on a daily basis, can adversely affect health and well-being. Sunlight-induced nonmelanoma skin cancer is the most prevalent type of cancer diagnosed in the industrialized world (1). In addition to being a complete carcinogen, UV radiation is also immune suppressive, and compelling evidence generated with experimental animals, biopsy-proven skin cancer patients, and cancer-prone immunosuppressed transplant patients indicate that the immune suppression induced by UV radiation is a major risk factor for skin cancer induction (2). In addition to tumor immunity, UV exposure suppresses a wide variety of immune reactions including contact hypersensitivity (CHS)4 to chemical haptens (3), and delayed type hypersensitivity to allogeneic histocompatibility Ags (4), viral (5), bacterial (6), and fungal Ags (7). In many of the studies mentioned above, significant and substantial immune suppression was achieved after a single exposure to UV radiation, using doses that are easily obtained during normal human occupational and/or recreational exposure (8, 9). Because UV-induced immune suppression contributes to skin cancer induction, and in view of the fact that a single exposure to sunlight can suppress the immune response to microbial Ags, it is important to study the mechanisms underlying UV-induced systemic immune suppression.
A considerable amount of evidence supports a role for UV-induced biological response modifiers and cytokines in activating systemic immune suppression (2). Almost immediately after UV exposure, keratinocytes secrete the lipid mediator of inflammation, platelet-activating factor (PAF) (10). Binding of PAF to its receptor induces a number of downstream effects, including the synthesis of cytokines and eicosanoids (11). In earlier studies, we demonstrated that treating keratinocytes with PAF up-regulated the transcription of COX-2 and IL-10 and activated keratinocytes to secrete PGE2. Both cytokine gene transcription and PGE2 synthesis was blocked by treating the keratinocytes with a selective PAF receptor (PAFR) antagonist (12, 13). Moreover, treating UV-irradiated mice with a series of selective PAFR antagonists blocked UV-induced immune suppression (13), presumably by blocking the induction of PGE2, which we know from previous work activates a cytokine cascade that results in systemic immune suppression (14). These studies suggest that the release of PAF by UV-irradiated keratinocytes is one of the first steps in the cascade of events leading to immune suppression.
After hapten sensitization of UV-irradiated mice, immune tolerance develops, which is specific for the hapten used to sensitize the UV-irradiated animal, and mediated in part, by UV-induced cytokines (15). One model system that has been used to study UV-induced tolerance induction was originally described by Okamoto and Kripke (16). They observed that hapten unresponsiveness could be induced in recipient mice injected with draining lymph node cells isolated from UV-irradiated FITC-sensitized mice. The distinct advantage of using FITC as the contact sensitizer is that one can follow the fate of the Ag-bearing cells. Using this model system, Kripke and colleagues (16–18) found that the cells that transferred immune tolerance were UV-damaged, pyrimidine dimer-positive, Ia-positive, FITC-positive APC.
The purpose of the studies presented here was to investigate the role of PAF in UV-induced immune suppression and tolerance induction. We asked two questions: 1) Does PAF play a role in inducing tolerance in UV-irradiated mice? 2) What are the mechanisms involved? Although we originally expected to find that the cell that transferred immune suppression and activated immune tolerance in the recipient mice was an Ag-positive APC, our data indicate that the cell responsible for immune suppression and tolerance induction is an IL-10-secreting B cell. Furthermore, we made the unexpected finding that both PAF and serotonin (5-HT) receptor binding must be blocked to abrogate the induction of immune suppression. These findings illustrate a novel function for PAF and serotonin in the immune response, the activation of immune regulatory B cells that suppress CHS.
Materials and Methods
Abs and reagents
Abs (FITC, PE, allophycocyanin, and/or CyChrome conjugated) specific for B220, CD4, CD8α, CD11b, CD11c, CD40, CD80, CD86, Ia, and Thy-1 were purchased from BD Pharmingen. Abs specific for CD19, CD24, Gr-1 (Ly6G), and OX 40L (CD134L) were purchased from eBiosciences. FITC isomer I, LPS (Escherichia coli serotype 026:B6), and the PAFR antagonist PCA 4248, were purchased from Sigma-Aldrich. Ketanserin and CV3988 were purchased from Biomol. Complete RPMI 1640 supplemented with 10% newborn calf serum (HyClone) was purchased from Invitrogen Life Technologies.
Mice
Specific pathogen-free, IL-10-deficient mice (Ref. 19; stock no. 002250) and B cell-deficient mice (Ref. 20; stock no. 002288), backcrossed onto a C57BL/6 background, and the wild-type (WT) controls, were purchased from The Jackson Laboratory. PAFR-deficient mice, backcrossed onto a C57BL/6 background (21), were obtained from Dr. J. Travers (University of Indiana Medical School, Indianapolis, IN). Within each experiment, all mice were age and sex matched. All procedures were reviewed and approved by the University of Texas M. D. Anderson Cancer Center Animal Care and Use Committee.
Isolation of cell subsets
FITC-positive cells were isolated by incubating 107 draining lymph node cells with 10 μl of anti-FITC microbeads (Miltenyi Biotec) in PBS containing 5% FBS at 4°C for 30 min (22). The cell/bead suspension was washed three times and then passed over a strong magnet. Both the flow-through fraction and the adherent fraction were collected. To isolate B220+ cells, draining lymph node cells from hapten-sensitized mice were cultured with 10 μg of anti-B220 in PBS containing 10% FBS at 4°C for 30 min. The cells were then mixed with mouse anti-rat-coated magnetic beads (4:1 ratio; Dynal Biotech) and the B220+ cells were separated from the B220− cells by passing the cells over a magnetic column. Cell purity was determined by flow cytometry.
Flow cytometry
Cells were resuspended in PBS containing 5% rat serum and 0.5% BSA (Sigma-Aldrich), and incubated on ice with mAb for 30 min. The cells were then washed in PBS containing 0.5% BSA and fixed in 2% paraformaldehyde. Fluorescent intensity was determined with a FACSCalibur (BD Biosciences).
Suppression of CHS and tolerance induction by UV exposure
A modification of the procedure described by Okamoto and Kripke (16) was used to induce immune suppression (see Fig. 1A). UV radiation (270–390 nm) was supplied by a bank of 6 FS-40 sunlamps (National Biologic). The dorsal hair of the mice was removed with electric clippers and the mice were exposed to 15 kJ/m2 of UVB (290–320 nm) radiation as measured with an IL-1700 research radiometer (International Light). Four days later, the mice were sensitized on the shaved abdominal skin by applying 400 μl of a 5 mg/ml solution of FITC diluted in acetone/dibutylphthalate (1/1 v/v). Eighteen hours later, the draining inguinal lymph nodes were removed, single-cell suspensions were prepared, and 1 × 106 cells were injected into each hind footpad of 10 normal syngeneic recipient mice. Six days later, the induction of CHS in these animals was measured. The mice were sedated, and the thickness of each right ear was measured with an engineer’s micrometer (Mitutoyo). The mice were then challenged with hapten by applying 10 μl of a 5 mg/ml solution of FITC to each ear surface. Eighteen hours later, the mice were sedated, the thickness of each right ear was remeasured, and the change in ear thickness was calculated. To determine whether transferring cells from UV-irradiated, hapten-sensitized mice induces tolerance, the animals were allowed to recover and 3 wk later, they were shaved and sensitized with FITC by applying 400 μl of a 5 mg/ml solution of FITC to the abdominal skin. Six days later, the mice were sedated, the thickness of each left ear was measured, and 10 μl of FITC was applied to each ear surface. Eighteen hours later, the thickness of each left ear was remeasured, and the change in ear thickness was calculated. The negative control for the CHS experiment consisted of measuring the change in ear thickness in recipient mice that did not receive any cells. The negative control for the tolerance experiment consisted of measuring ear swelling in mice that did not receive any cells, were not sensitized, but were challenged. The positive control consisted of transferring cells from mice that were shaved and sensitized with hapten but were not exposed to UV. The data are expressed as the mean Δ ear swelling ± SEM (n = 10). Statistical differences between the positive control and the experimental groups were determined by use of a one-way ANOVA followed by the Tukey-Kramer multiple comparison test (GraphPad Software). Probabilities <0.05 were considered to be significant.
FIGURE 1.
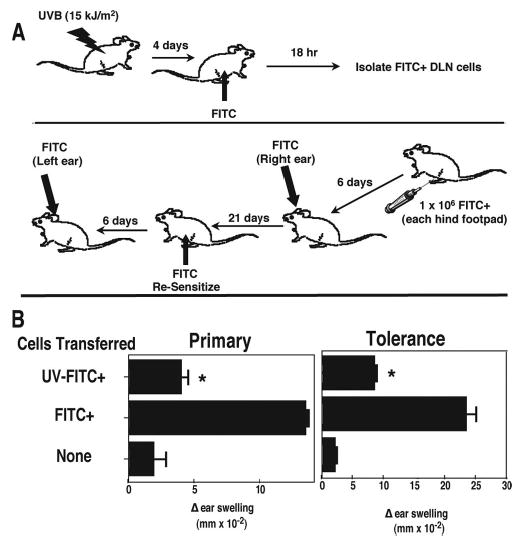
Transferring lymph node cells from UV-irradiated, FITC-sensitized mice to normal recipients induces tolerance. A, Mice are exposed to 15 kJ/m2 of UVB radiation and 4 days later were painted with FITC on the unirradiated ventral skin. Eighteen hours later, the draining lymph nodes were collected and injected into naive recipient mice. The right ear of each mouse was challenged 6 days later. CHS was read the next day, and the mice were rested for 21 days. They were then sensitized with FITC, and the left ear was challenged 6 days later. The induction of tolerance was measured 1 day later. B, The recipient mice received cells from hapten-sensitized mice (FITC+), or cells from mice that were exposed to UV and then sensitized with hapten (UV-FITC+), or were not injected (None).*, Statistically significant difference (p < 0.001) from the FITC+ control.
Cytokine production by draining lymph node cells isolated from UV-irradiated mice
FITC-positive draining lymph node cells were isolated from UV-irradiated or control mice using anti-FITC magnetic beads as described above. The cells were resuspended in complete RPMI 1640 and cultured at 37°C with and without 1 μg/ml LPS and 5 μg/ml anti-CD40 mAb. Twenty-four hours later, the supernatant fluid was removed and cytokine production was determined by ELISA, as described previously (14). Statistical differences between the positive control and the experimental groups (n = 3) were determined by use of a two-tailed Student’s t test; probabilities <0.05 were considered to be significant.
Results
Transfer of immune suppression and tolerance induction by FITC+ draining lymph node cells from UV-irradiated mice
The initial focus of these experiments was to test the hypothesis that PAF plays a role in tolerance induction. To do this, we chose to modify a procedure previously published by Okamoto and Kripke (16). In their experiments, mice were first exposed to UV and then the fluorescent hapten FITC was applied directly to the UV-irradiated skin. Eighteen hours later, draining lymph node cells from the UV-irradiated mice were transferred into normal mice where they failed to induce a CHS reaction and instead induced tolerance. They observed that FITC+ APC were responsible for transferring immune suppression in their experiments. Our modification consisted of first exposing the mice to UV radiation but then sensitizing the mice at a distant nonirradiated site. Others have shown previously that this modification will result in a suppressed CHS reaction (23), suggesting that it would serve as an ideal model system to determine whether PAFR binding plays a role in the induction of immune tolerance. The protocol used is shown in Fig. 1A. Mice were first exposed to UV radiation and then the fluorescent hapten FITC was applied to a nonirradiated site 4 days later. Draining lymph node cells from these mice were then transferred into normal syngeneic recipient where their ability to induce CHS and induce immune tolerance were examined (Fig. 1B). In the left panel of Fig. 1B, the ability of FITC+ cells from normal and UV-irradiated mice to induce CHS in the recipient mice is shown. The background response is measured in mice that did not receive any cells (hence no immunization) but were challenged. The Δ ear swelling response in these mice was minimal. The positive control for this experiment consisted of measuring the Δ ear swelling response found in animals that were injected with FITC+ draining lymph node cells isolated from unirradiated mice. As expected, a vigorous CHS reaction was noted when these cells were transferred. When, however, draining lymph node cells were transferred from UV-irradiated, FITC-sensitized mice, the resulting CHS reaction was significantly depressed compared with the positive control (p < 0.001).
The mice were allowed to recover, and 21 days later, they were sensitized by applying FITC to the abdominal skin (Fig. 1B, right panel). Although a very good ear swelling reaction was found in mice that were resensitized after first receiving draining lymph node cells from normal FITC-sensitized mice, the mice that received draining lymph node cells from UV-irradiated FITC-sensitized mice were tolerant, in that they were not able to generate an immune reaction upon sensitization (p < 0.001). If the mice that received draining lymph nodes cells from UV-irradiated FITC-sensitized animals were sensitized with an irrelevant hapten (i.e., dinitrofluorobenzene), no tolerance was noted indicating that the tolerance induced was hapten specific (data not shown). These data indicate that immune tolerance can be induced by transferring draining lymph node cells from mice exposed to UV at one site and painted with hapten at a distant nonirradiated site.
Characterization of the cells that induce immune tolerance
Draining lymph node cells were isolated from normal or UV-irradiated, FITC-sensitized mice and analyzed by flow cytometry. Initially, we measured MHC class II expression, CD80, CD86, OX40L, and B220 expression on FITC+ cells and compared the profiles found on cells from normal FITC-sensitized mice (Fig. 2A, left panel) with that found on cells isolated from UV-irradiated FITC-sensitized mice (Fig. 2A, right panel). For the most part, the expression of MHC class II, CD80, CD86, and OX40L on FITC+ cells was similar, regardless of whether the cells were isolated from UV-irradiated or normal mice. One notable difference was the up-regulation of a FITCdim, B220bright subpopulation of cells found in the draining lymph node cells isolated from the UV-irradiated mice.
FIGURE 2.
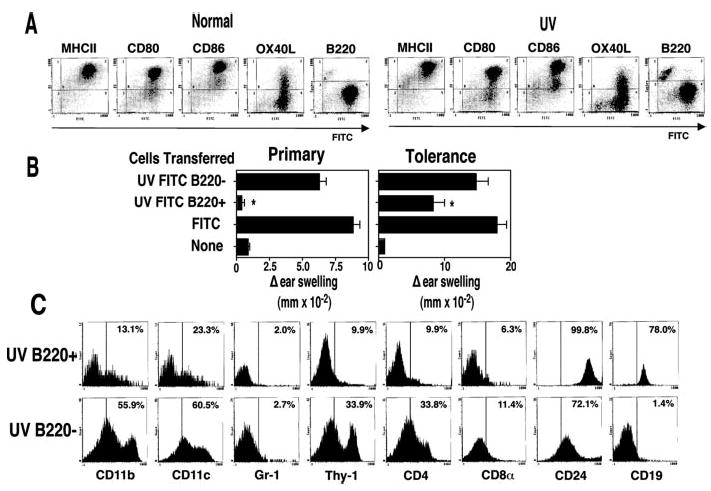
FITC+B220+ cell transfer tolerance. A, FITC-positive cells from hapten-sensitized mice (Normal) or UV-irradiated hapten-sensitized mice (UV) were stained with PE-labeled Abs to various cell surface markers. The data are expressed as fluorescence intensity on a log scale. B, The recipient mice received cells from FITC-sensitized mice (Normal) or cells from mice that were exposed to UV and then sensitized with FITC. Magnetic beads were used to separate the B220-positive cells (UV-B220+) from the B220−cells (UV-B220−), negative controls (None) were not injected.*, A statistically significant difference (p < 0.05) from the normal control. C, Cell surface phenotype of UV B220+ vs UV B220− cells. Data are expressed as relative cell count vs mean fluorescence intensity (MFI) on a log scale. Numbers indicate the percentage of cells in each population that is positive for each marker.
To determine whether the B220+ cells from UV-irradiated mice play a role in tolerance induction, Ab-coated magnetic beads were used to isolate the B220+ and B220− subpopulations from the draining lymph nodes of UV-irradiated, FITC-sensitized mice. Recipient mice received 1 × 106 UV B220+ or 1 × 106 UV B220+ cells in each hind footpad, and the ability of these cells to induce CHS or induce tolerance is found in Fig. 2B. A vigorous CHS reaction was observed in mice injected with UV B220− cells, one that was statistically indistinguishable (p > 0.05) from the positive control (mice that received 1 × 106 unseparated draining lymph node cells from normal FITC-sensitized mice). In contrast, the CHS reaction found in mice injected with UV B220+ cells was significantly different (p < 0.05) from the response found in the positive control and not statistically different from the response found in the negative control (p > 0.05). A similar situation was found when tolerance induction was measured (Fig. 2B, right panel). Sensitization of mice originally injected with UV B220+ cells resulted in tolerance (p < 0.05 vs positive control). When the mice that first received UV B220− cells were sensitized with hapten, the immune response generated was not statistically different from that found in the positive control (p > 0.05). These data indicate that the B220+ cells found in the draining lymph nodes of UV-irradiated FITC-sensitized mice fail to transfer CHS, and induce tolerance in recipient mice.
To further characterize the cells from the UV-irradiated mice that induce tolerance, we first isolated the FITC-positive cells using anti-FITC-coated microbeads and then used anti-B220 beads to separate the FITC+ population into B220+ and B220− subpopulations. These cells were then stained with PE-conjugated mAb to yield further information regarding their cell surface phenotype (Fig. 2C). The B220+ cells expressed CD19 and CD24 on their cells surface with minimal staining for Gr-1, Thy-1, CD8α, or CD4. A small percentage of the UV B220+ cells expressed CD11b and CD11c, suggesting that the cells within the UV B220+ subpopulation are primarily B cells with some dendritic cell contamination. For the sake of comparison the UV B220− subpopulation is also shown. This subpopulation of cells appears to contain a mixture of dendritic cells, T cells, and B cells.
B lymphocytes are responsible for transfer of immune suppression and tolerance induction in UV-irradiated mice
To determine whether the B cells or the dendritic cells found in the UV+, FITC+, B220+ draining lymph nodes transfer immune suppression and induce tolerance, two approaches were taken. Based on the observation that one of the major differences between the draining lymph node cells isolated from normal and UV-irradiated mice was the up-regulation of a FITCdimB220bright population, we gated on this population (subpopulation R5 in Fig. 3, left panel) and used flow cytometry to determine its cell surface phenotype. Because we hypothesized that the tolerance-inducing cells may be either a B220+, CD19+, B cell, or a B220+ dendritic cell (24), we measured CD11c and CD11b expression on the FITCdimB220bright population (Fig. 3A, right panel). Our data indicate that the B220+ cells are not dendritic cells because they are CD11c and CD11b negative. Further support for the hypothesis that the tolerance-inducing cells are B lymphocytes is presented in Fig. 3B. In this experiment, draining lymph node cells were isolated from UV-irradiated WT mice, or B cell-deficient (μMT), FITC-sensitized mice. These cells were then transferred into normal WT animals, where CHS and tolerance induction was measured. As before, a vigorous CHS reaction was noted in mice injected with WT FITC-sensitized draining lymph node cells, and CHS was suppressed when the recipient mice were injected with cells from donors that were exposed to UV radiation before FITC sensitization (p < 0.01). However, when the draining lymph node cells were isolated from UV-irradiated, FITC-sensitized, B cell-deficient mice, no immune suppression was noted (p > 0.05 vs FITC WT).
FIGURE 3.
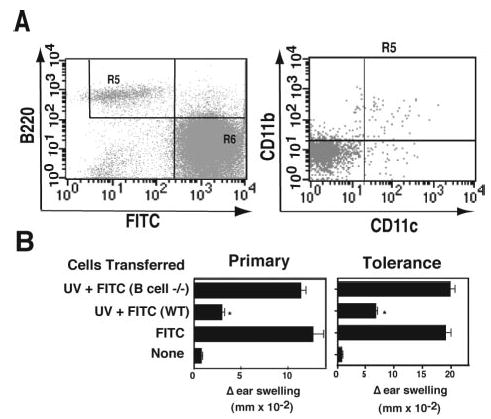
Tolerance-inducing cells are B cells. A, FITC-bearing cells were stained with PE-B220, allophycocyanin-CD11b, and CyChrome-CD11c. The FITCdim, B220bright population was selected (left panel, R5), and CD11c vs CD11b staining was measured (right panel). The data are expressed as mean MFI on a log scale. B, Cells were transferred from UV-irradiated-hapten-sensitized B cell-deficient mice (UV + FITC B cell−/−), UV-irradiated hapten-sensitized WT mice (UV + FITC WT) or hapten-sensitized WT mice (FITC WT). Negative control mice were not injected (None).*, A statistically significant difference (p < 0.01) from the FITC+ control.
Similarly, B lymphocytes were required for tolerance induction (Fig. 3B, right panel). Whereas sensitization of mice originally injected with WT cells isolated from UV-irradiated FITC-sensitized mice induced tolerance (p < 0. 001 vs FITC WT), no tolerance induction was noted when the donor cells were isolated from UV-irradiated B cell-deficient mice (p > 0.05 vs FITC WT). These findings indicate that UV-induced, B220+, FITC+, B cells transfer immune suppression and mediate tolerance induction.
IL-10 is secreted by FITC+ cells isolated from the draining lymph nodes of UV-irradiated mice
To determine the role, if any, that cytokines play in immune suppression and tolerance induction, we first measured cytokine release by draining lymph node cells from UV-irradiated, FITC-sensitized mice. We were particularly interested in IL-10 and IL-12, because previous studies indicated that increased IL-10 production and decreased secretion of IL-12 plays a role in UV-induced immune suppression (25, 26). Lymph node cells were isolated from control mice (FITC-sensitized) or mice exposed to UV and then sensitized with FITC. The cells were stimulated with LPS and anti-CD40, and cytokine secretion was measured by ELISA (Table I). A marginal increase in IL-12p70 secretion was observed when the cells were stimulated with LPS/anti-CD40 compared with IL-12p70 production by cells cultured in medium only. Prior UV exposure had no real effect on IL-12p70 secretion. A different situation was observed when IL-10 secretion was measured. The amount of IL-10 found in the supernatant of unstimulated cells was below the limit of detection (15 pg/ml). When cells from normal FITC-sensitized mice were stimulated with LPS/anti-CD40, IL-10 was found in the supernatant. Treating lymph node cells from UV-irradiated, FITC-sensitized cells with LPS/anti-CD40 caused a significant increase (p < 0.01) in IL-10 secretion when compared with the amount of IL-10 secreted by the normal cells.
Table I.
Cytokine secretion by draining lymph node cells isolated from UV-irradiated hapten-sensitized micea
| pg/ml/106 Cells
|
||
|---|---|---|
| Treatmentb | IL-12p70 | IL-10 |
| FITC | 15.2 ± 0.7 | BLDc |
| FITC + LPS/anti-CD40 | 25.2 ± 4.9 | 72.0 ± 17.0 |
| UV + FITC | 11.2 ± 1.3 | BLD |
| UV + FITC + LPS/anti-CD40 | 20.6 ± 3.0 | 166 ± 21.0d |
Mice were exposed to UV (15 kJ/m2; dorsal skin) and 4 days later were sensitized with hapten (0.5% FITC, abdominal skin). Eighteen hours later, FITC+ cells were isolated with magnetic beads.
The cells were cultured in complete RPMI 1640 with or without LPS (1 μg/ml) and anti-CD40 mAb (5 μg/ml) for 24 h. Cytokines present in the supernatant were determined by ELISA.
Below the limit of detection (15 pg/ml).
Value of p < 0.01 vs FITC + LPS/anti-CD40; two-tailed Student’s t test.
These findings suggest that increased IL-10 secretion may play a role in the induction of tolerance in our experiments. To determine whether this was the case, draining lymph node cells were isolated from UV-irradiated FITC-sensitized WT or IL-10-deficient mice (Fig. 4). As expected, a vigorous CHS reaction was noted in recipient mice injected with WT FITC-sensitized draining lymph node cells; and CHS was suppressed when the recipient mice were injected with cells from donors that were exposed to UV radiation before FITC sensitization (p < 0.0001). However, when the draining lymph node cells were isolated from UV-irradiated, FITC-sensitized IL-10−/− mice, no immune suppression was noted (p > 0.62 vs FITC WT).
FIGURE 4.
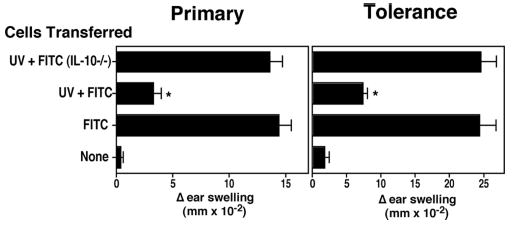
Failure to induce tolerance when cells are transferred from IL-10−/− UV-irradiated donor mice. Lymph node cells were isolated from hapten-sensitized mice (FITC), UV-irradiated hapten-sensitized WT mice (UV + FITC), or UV-irradiated hapten-sensitized IL-10-deficient mice (UV + FITC IL-10−/−). CHS and tolerance were determined as described previously.*, A statistically significant difference (p < 0.0001) from the FITC+ WT control.
A similar situation was found when tolerance induction was measured (Fig. 4, right panel). Whereas sensitization of mice originally injected with WT cells isolated from UV-irradiated FITC-sensitized mice induced tolerance (p < 0.0001 vs FITC WT), no tolerance induction was noted when the donor cells were isolated from UV-irradiated, IL-10−/− mice (p > 0.95 vs FITC WT). These findings indicate that IL-10 plays a critical role in tolerance induction when UV-induced, B220+, FITC+ cells are transferred into normal recipient mice.
A role for PAF and serotonin receptor binding in immune suppression and tolerance induction
To determine whether PAF plays a role in the induction of immune tolerance, mice were injected with the selective PAFR antagonist, PCA 4248, immediately before UV exposure, as described previously (13). The mice were sensitized with FITC, and the draining lymph node cells were transferred into recipient mice as described above (Fig. 5A). We found a depressed CHS reaction in recipient mice that received draining lymph node cells from UV-irradiated, FITC-sensitized donors (p < 0.0001; UV + FITC vs FITC). The ability to generate CHS was restored by treating UV-irradiated donor mice with the selective PAFR antagonist (p > 0.05; FITC vs UV + FITC + PCA 4248). Similarly, tolerance induction was reversed by injecting the donor mice with PCA 4248 (p > 0.4; FITC vs UV + FITC + PCA 4248).
FIGURE 5.
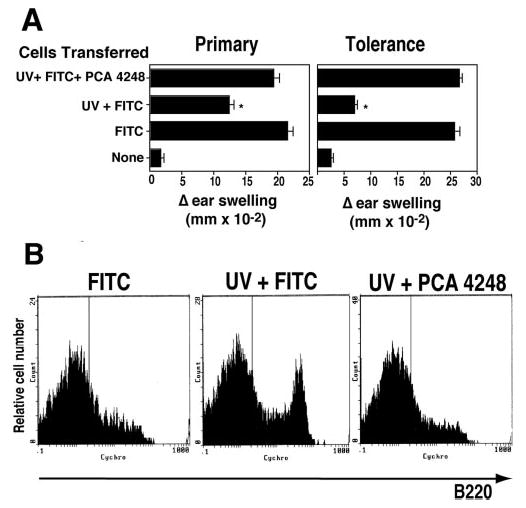
Treating UV-irradiated mice with a selective PAFR antagonist blocks tolerance induction. A, Lymph node cells were isolated from donor mice sensitized with hapten (FITC), exposed to UV and sensitized with hapten (UV + FITC) or exposed to UV, injected with a PAFR antagonist (PCA 4248) and then sensitized with hapten (UV + FITC + PCA 4248). CHS and tolerance were determined as described previously.*, A statistically significant difference (p < 0.0001) from the FITC+ control. B, Anti-FITC-coated magnetic beads were used to isolate draining lymph node cells from hapten-sensitized mice (FITC), UV-irradiated hapten-sensitized mice (FITC), and UV-irradiated, hapten-sensitized, PCA 4248-injected mice (UV + PCA 4248). The cells were stained with CyChrome-labeled anti-B220, and the data are expressed as MFI on a log scale vs cell count.
We also examined the effect that injecting PCA 4248 had on the induction of the FITC+, B220+ subpopulation of cells found in the draining lymph nodes of UV-irradiated, FITC-sensitized mice (Fig. 5B). Anti-FITC-coated magnetic beads were used to isolate draining lymph node cells from FITC-sensitized mice, UV-irradiated FITC-sensitized mice, and UV-irradiated FITC-sensitized, PCA 4248-injected mice. The cells were then stained with CyChrome-labeled anti-B220. As described above, we saw an up-regulation of B220bright cells after UV exposure (Fig. 5B; FITC vs UV + FITC). This population was not present in draining lymph node cells isolated from mice exposed to UV, sensitized with FITC and injected with PCA 4248 (Fig. 5B; UV + FITC vs UV + PCA 4248).
Because receptor antagonists, such as PCA 4248 are selective, but not specific, inhibitors of receptor ligand binding, we repeated the experiments described above using PAFR-deficient mice. Draining lymph node cells were isolated from UV-irradiated FITC-sensitized PAFR-deficient mice (21) and transferred into age- and sex-matched controls. Much to our surprise, lymph node cells from PAFR−/− did transfer immune suppression (Fig. 6). This unexpected result prompted a further review of the inhibitory activity of PCA 4248. PCA 4248 has dual activities. In addition to blocking the binding of PAF to its receptor (27), it can also block serotonin-induced inflammation (28). This suggests that to block tolerance induction in our system, both PAF and serotonin binding must be inhibited. This was tested in two ways. First, we exposed PAFR knockout mice to UV and then injected them with ketanserin, a serotonin receptor antagonist (Fig. 6A). Alternatively, we exposed WT mice to UV radiation, and injected them with a CV3988 (a PAFR antagonist) only, ketanserin only, or a mixture of the two (Fig. 6B). As can be seen in Fig. 6, transfer of immune suppression occurred only when both PAF and serotonin receptor binding was blocked. Lymphocytes from UV-irradiated, FITC-sensitized, PAFR-deficient mice could transfer immune suppression, and immune suppression was blocked when these mice were injected with ketanserin. Similarly, injecting UV-irradiated FITC-sensitized WT mice with ketanserin only, or CV3988 only, did not reverse the transfer of tolerance. However, when both were used in concert, the transfer of suppression was inhibited.
FIGURE 6.
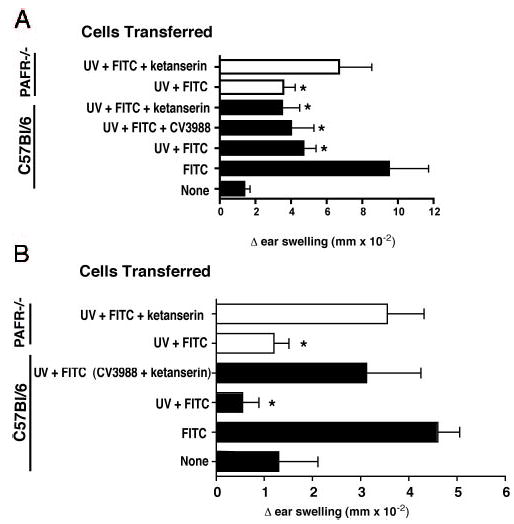
PAF and serotonin receptor binding is required to induce immune suppression. Recipient mice received cells from WT mice (□) or PAFR−/− mice (▪) exposed to UV and sensitized with hapten (UV + FITC). A, Transfer of cells from WT or PAFR−/− mice induced immune suppression (*, p < 0.05 vs FITC only control). Injecting a serotonin (ketanserin) or a PAFR (CV 3988) antagonist into WT donors did not reverse the transfer of immune suppression. Injecting ketanserin into PAFR−/− mice reverses (p > 0.05 vs FITC control) the induction of immune suppression. B, Injecting both a PAF and serotonin receptor antagonist into WT recipients, but not either alone, blocks the transfer of immune suppression (p < 0.05 vs UV + FITC; p > 0.05 vs FITC only).
Discussion
The initial focus of the experiments presented here was to determine whether PAF, a lipid mediator of inflammation that plays a critical role in UV-induced immune suppression, could suppress APC function in vivo. We made two unexpected observations. First, we found that we could transfer UV-induced immune suppression and induce tolerance in normal recipients by transferring IL-10-secreting, B220+, CD19+ B cells. Second, we observed that we could block the transfer of immune suppression only when we simultaneously blocked PAF and serotonin receptor engagement.
Although not as widely studied as CD4+CD25+ T regulatory cells, there is a growing appreciation for the role of B lymphocytes in regulating T cell immunity (29). Ag-pulsed B cells can present a tolerogenic signal for both CD4+ (30) and CD8+ (31) T cells, in some cases regardless of high expression of costimulatory molecules on the Ag-pulsed B cells (31). Naive and not memory T cells appear to be more susceptible to tolerance induction by Ag-bearing B cells (29, 30). A common mechanism to explain the induction of T cell tolerance by B cells is IL-10 secretion. Examples include chronic intestinal inflammation (32), arthritis (33), autoimmunity (34), and ileitis (35). The IL-10-secreting B cell that we find in the draining lymph node of UV-irradiated mice is yet one more example that can be added to a growing list of immunoregulatory B cells.
A second unexpected observation was the requirement for PAF and serotonin receptor binding for the transfer of immune suppression. The role of PAF in inflammation is well-appreciated (36). The role of serotonin in inflammation is known but perhaps not as well-appreciated. Serotonin functions as a neurotransmitter in the brain, induces peristalsis in the gut, and can function as an inflammatory mediator in peripheral tissues (37, 38). Our findings suggest that serotonin receptor engagement is essential for immune suppression in vivo. This appears to contradict data published by Matsuda et al. (39) who showed that the vasoactive effects of serotonin were critically important for the efflux of immune cells into the site of Ag deposition during the challenge phase of delayed-type hypersensitivity. Note, however, we could only block the induction of immune suppression when both PAF and serotonin receptor binding was inhibited. This may suggest that serotonin receptor binding by itself sends one signal to the immune system, but when serotonin and PAFRs are simultaneously engaged, an immune-suppressive signal is sent. The exact source of the “UV-induced” serotonin in our system is not entirely clear. It is interesting to note that recent studies indicate that serotonin is produced by the skin (40). Whether its production is up-regulated following total body UV exposure remains to be seen.
Because a common feature shared by many of the diseases mentioned above (i.e., Crohn’s, arthritis, autoimmunity) is chronic inflammation, we suggest that inflammatory mediators such as PAF and serotonin may be inducing/activating IL-10 secretion by B cells. Normal B lymphocytes express both the PAFR and the serotonin receptor on their surface (37, 41). In addition, PAF-treated B cells secrete a variety of cytokines, including IL-10 (42, 43). In our studies, we find that PCA 4248, a receptor antagonist that blocks binding of both PAF and serotonin to their receptors, inhibits immune suppression and the induction of tolerance, in part by blocking the up-regulation of the tolerance-inducing FITC+, B220+ B cells. It is interesting to note that using an arthritis model, Mauri et al. (33) could only transfer tolerance when the B cells were isolated from mice first injected with Ag emulsified in CFA, and then stimulated with anti-CD40. Injecting B cells that were stimulated in vitro with anti-CD40 and Ag but without the inflammatory signal provided by the adjuvant, was not protective. They propose that a complex pathway of cell-cell communication is required to drive the differentiation of the regulatory B cells in vivo. Based on our findings, we suggest that PAF and serotonin receptor binding may be critical steps in activating the complex cellular interactions required to activate regulatory B cells.
Our findings also provide new insight into the mechanisms underlying UV-induced immune suppression. Although there are many examples of UV exposure activating immune regulatory T cells (22, 44–46), and evidence exists to indicate UV-damaged dendritic cells (16, 17) can induce tolerance, a role for UV-activated B cells in tolerance induction is novel. This somewhat unexpected result may reflect the modification we used to induce tolerance in our system. Because we are interested in studying the mechanisms involved in the induction of systemic immune suppression by UV radiation, and specifically the role of UV-induced epidermal cytokines in systemic tolerance induction, we applied that hapten to unirradiated skin. In previous studies, the contact allergen was placed directly unto UV-irradiated skin, where UV-damaged Langerhans cells acquired the Ag, migrated to the draining lymph node (47), and activated the suppressive pathway (16). It has been known for some time that the mechanisms underlying the immune suppression that results after the hapten is applied directly to the UV-irradiated site (local immune suppression) differs from that observed when the hapten is applied at a distant nonirradiated site (systemic immune suppression) (2). Our findings expand on previous observations by showing that at least two separate cellular pathways lead to tolerance, one (local) mediated by Langerhans cells and the other (systemic) mediated by B cells.
Although the cells that activate immune tolerance may differ, a common feature shared by both pathways is IL-10. Alard et al. (48) report that treating mice with Abs to IL-10 will reverse the induction of tolerance in mice where the hapten is applied directly to the irradiated skin. This compares favorably with our data showing no tolerance induction when draining lymph node cells are isolated from UV-irradiated IL-10-deficient mice. We suspect that the IL-10 ultimately targets dendritic cells, based on the well-known ability of IL-10-treated dendritic cells to induce immune tolerance (49). Alternatively, PAF and its downstream mediator, PGE2 may be directly affecting dendritic cells. Activation of dendritic cells in the presence of PGE2 inhibits the production of biologically active IL-12p70 and promotes the secretion of immunosuppressive IL-12p40 homodimers (50, 51), similar to what was found when IL-12 secretion by dendritic cells isolated from UV-irradiated mice was measured (26). Studies are in progress to determine whether PAF and/or serotonin can directly suppress the function of dendritic cells, but at the present time we favor the view that the cytokines and biological response modifiers up-regulated in response to UV exposure may work in parallel to target dendritic cell function and induce tolerance.
Finally, the data presented here confirm a recent study by Byrne and Halliday (52) who report that UV exposure activates a B cell that suppresses APC function. The data presented here expand on Byrne’s observation by demonstrating that UV-induced inflammatory mediators are responsible for activating the UV-induced regulatory B cells. These findings may have broad implications besides providing a better insight into the mechanisms involved in UV-induced tolerance induction. Secretion of inflammatory mediators such as PAF and serotonin are up-regulated in response to chronic inflammation, oxidative stress, sepsis, cigarette smoking, and chemotherapy (36, 53, 54). Some of these events are associated with immune suppression suggesting that inflammatory mediators may play an important role. A good example is the induction of immune suppression that results after dermal exposure to volatile organic chemicals. We find that Th1-type immune reactions are suppressed following dermal application of jet fuel (55, 56), and that injecting PAFR antagonists into jet fuel-treated mice blocks the induction of immune suppression (12). It is not clear whether mechanisms similar to those described here are involved, but these observations do suggest that the role of inflammatory mediators in activating immune suppression may be widespread.
Acknowledgments
We thank Nasser Kazimi for assistance with the animal experiments, and Karen Ramirez for help with the flow cytometry.
Footnotes
This work was supported by grants from the National Cancer Institute (CA75575, CA88943, and CA112660). S.N.B. was supported by a C. J. Martin Fellowship (No. 307726) from the National Health and Medical Research Council of Australia. The animal and flow cytometry facilities at the M. D. Anderson Cancer Center were supported in part by a core grant from the National Cancer Institute (CA16672).
Abbreviations used in this paper: CHS, contact hypersensitivity; PAF, platelet-activating factor; PAFR, PAF receptor; WT, wild type; MFI, mean fluorescence intensity.
Disclosures
The authors have no financial conflict of interest.
References
- 1.Boring CC, Squires TS, Tong T. Cancer statistics. CA Cancer J Clin. 1992;42:19–38. doi: 10.3322/canjclin.42.1.19. [DOI] [PubMed] [Google Scholar]
- 2.Ullrich SE. Photoimmune suppression and photocarcinogenesis. Frontiers Biosci. 2002;7:D684–D703. doi: 10.2741/A804. [DOI] [PubMed] [Google Scholar]
- 3.Noonan FP, De Fabo EC, Kripke ML. Suppression of contact hypersensitivity by ultraviolet radiation: an experimental model. Springer Semin Immunopathol. 1981;4:293–304. doi: 10.1007/BF01892183. [DOI] [PubMed] [Google Scholar]
- 4.Ullrich SE. Suppression of the immune response to allogeneic histocompatibility antigen by a single exposure to UV radiation. Transplantation. 1986;42:287–291. doi: 10.1097/00007890-198609000-00012. [DOI] [PubMed] [Google Scholar]
- 5.Howie SEM, Norval M, Maingay J. Exposure to low dose UVB light suppresses delayed type hypersensitivity to herpes simplex virus in mice. J Invest Dermatol. 1986;86:125–128. doi: 10.1111/1523-1747.ep12284128. [DOI] [PubMed] [Google Scholar]
- 6.Jeevan A, Kripke ML. Effect of a single exposure to ultraviolet radiation on Mycobacterium bovis Bacillus Calmette-Guerin infection in mice. J Immunol. 1989;143:2837–2843. [PubMed] [Google Scholar]
- 7.Denkins Y, I, Fidler J, Kripke ML. Exposure of mice to UVB radiation suppresses delayed hypersensitivity to. Candida albicans Photochem Photobiol. 1989;49:615–619. doi: 10.1111/j.1751-1097.1989.tb08432.x. [DOI] [PubMed] [Google Scholar]
- 8.Garssen J, Goettsch W, de Gruijl F, Slob W, van Loveren H. Risk assessment of UVB effects on resistance to infectious diseases. Photochem Photobiol. 1996;64:269–274. doi: 10.1111/j.1751-1097.1996.tb02457.x. [DOI] [PubMed] [Google Scholar]
- 9.Nghiem DX, Kazimi N, Clydesdale G, Ananthaswamy HN, Kripke ML, Ullrich SE. Ultraviolet A radiation suppresses an established immune response: implications for sunscreen design. J Invest Dermatol. 2001;117:1193–1199. doi: 10.1046/j.0022-202x.2001.01503.x. [DOI] [PubMed] [Google Scholar]
- 10.Barber LA, Spandau DF, Rathman SC, Murphy RC, Johnson CA, Kelley SW, Hurwitz SA, Travers JB. Expression of the platelet-activating factor receptor results in enhanced ultraviolet B radiation-induced apoptosis in a human epidermal cell line. J Biol Chem. 1998;273:18891–18897. doi: 10.1074/jbc.273.30.18891. [DOI] [PubMed] [Google Scholar]
- 11.Pei Y, Barber LA, Murphy RC, Johnson CA, Kelley SW, Dy LC, Fertel RH, Nguyen TM, Williams DA, Travers JB. Activation of the epidermal platelet-activating factor receptor results in cytokine and cyclo-oxygenase-2 biosynthesis. J Immunol. 1998;161:1954–1961. [PubMed] [Google Scholar]
- 12.Ramos G, Kazimi N, Nghiem DX, Walterscheid JP, Ullrich SE. Platelet activating factor receptor binding plays a critical role in jet fuel-induced immune suppression. Toxicol Appl Pharmacol. 2004;195:331–338. doi: 10.1016/j.taap.2003.07.014. [DOI] [PubMed] [Google Scholar]
- 13.Walterscheid JP, Ullrich SE, Nghiem DX. Platelet-activating factor, a molecular sensor for cellular damage, activates systemic immune suppression. J Exp Med. 2002;195:171–179. doi: 10.1084/jem.20011450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Shreedhar V, Giese T, Sung VW, Ullrich SE. A cytokine cascade including prostaglandin E2, interleukin-4, and interleukin-10 is responsible for UV-induced systemic immune suppression. J Immunol. 1998;160:3783–3789. [PubMed] [Google Scholar]
- 15.Niizeki H, Streilein JW. Hapten-specific tolerance induced by acute, low-dose ultraviolet B radiation of skin is mediated via interleukin-10. J Invest Dermatol. 1997;109:25–30. doi: 10.1111/1523-1747.ep12276415. [DOI] [PubMed] [Google Scholar]
- 16.Okamoto H, Kripke ML. Effector and suppressor circuits of the immune response are activated in vivo by different mechanisms. Proc Natl Acad Sci USA. 1987;84:3841–3845. doi: 10.1073/pnas.84.11.3841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Vink AA, Moodycliffe AM, Shreedhar V, Ullrich SE, Roza L, Yarosh DB, Kripke ML. The inhibition of antigen-presenting activity of dendritic cells resulting from UV irradiation of murine skin is restored by in vitro photorepair of cyclobutane pyrimidine dimers. Proc Natl Acad Sci USA. 1997;94:5255–5260. doi: 10.1073/pnas.94.10.5255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Vink AA, Strickland FM, Bucana C, Cox PA, Roza L, Yarosh DB, Kripke ML. Localization of DNA damage and its role in altered antigen-presenting cell function in ultraviolet-irradiated mice. J Exp Med. 1996;183:1491–1500. doi: 10.1084/jem.183.4.1491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kuhn R, Lohler J, Rennick D, Rajewsky K, Muller W. Interleukin-10-deficient mice develop chronic enterocolitis. Cell. 1993;75:263–274. doi: 10.1016/0092-8674(93)80068-p. [DOI] [PubMed] [Google Scholar]
- 20.Kitamura D, Roes J, Kuhn R, Rajewsky K. A B cell-deficient mouse by targeted disruption of the membrane exon of the immunoglobulin μ chain gene. Nature. 1991;350:423–426. doi: 10.1038/350423a0. [DOI] [PubMed] [Google Scholar]
- 21.Ishii S, Kuwaki T, Nagase T, Maki K, Tashiro F, Sunaga S, Cao WH, Kume K, Fukuchi Y, Ikuta K, et al. Impaired anaphylactic responses with intact sensitivity to endotoxin in mice lacking a platelet-activating factor receptor. J Exp Med. 1998;187:1779–1788. doi: 10.1084/jem.187.11.1779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Moodycliffe AM, Nghiem D, Clydesdale G, Ullrich SE. Immune suppression and skin cancer development: regulation by NKT cells. Nat Immunol. 2000;1:521–525. doi: 10.1038/82782. [DOI] [PubMed] [Google Scholar]
- 23.Kripke ML, McClendon E. Studies on the role of antigen-presenting cells in the systemic suppression of contact hypersensitivity by UVB radiation. J Immunol. 1986;137:443–447. [PubMed] [Google Scholar]
- 24.Martin P, Del Hoyo GM, Anjuere F, Arias CF, Vargas HH, Fernandez LA, Parrillas V, Ardavin C. Characterization of a new subpopulation of mouse CD8α+B220+ dendritic cells endowed with type 1 interferon production capacity and tolerogenic potential. Blood. 2002;100:383–390. doi: 10.1182/blood.v100.2.383. [DOI] [PubMed] [Google Scholar]
- 25.Rivas JM, Ullrich SE. Systemic suppression of DTH by supernatants from UV-irradiated keratinocytes: an essential role for keratinocyte-derived interleukin 10. J Immunol. 1992;149:3865–3871. [PubMed] [Google Scholar]
- 26.Schmitt DA, Ullrich SE. Exposure to ultraviolet radiation causes dendritic cells/macrophages to secrete immune suppressive IL-12p40 homodimers. J Immunol. 2000;165:3162–3167. doi: 10.4049/jimmunol.165.6.3162. [DOI] [PubMed] [Google Scholar]
- 27.Fernandez-Gallardo S, Ortega MP, Priego JG, de Casa-Juana MF, Sunkel C, Sanchez Crespo M. Pharmacological actions of PCA 4248, a new platelet-activating factor receptor antagonist: in vivo studies. J Pharmacol Exp Ther. 1990;255:34–39. [PubMed] [Google Scholar]
- 28.Martins MA, Lima MC, Bozza PT, Faria Neto HC, Silva PM, Sunkel CE, Cordeiro RS. Interference of the PAF receptor antagonist, PCA 4248, with the rat pleurisy evoked by inflammatory mediators or allergen. Eur J Pharmacol. 1993;237:17–22. doi: 10.1016/0014-2999(93)90087-x. [DOI] [PubMed] [Google Scholar]
- 29.Fuchs EJ, Matzinger P. B cells turn off virgin but not memory T cells. Science. 1992;258:1156–1159. doi: 10.1126/science.1439825. [DOI] [PubMed] [Google Scholar]
- 30.Eynon EE, Parker DC. Parameters of tolerance induction by antigen targeted to B lymphocytes. J Immunol. 1993;151:2958–2964. [PubMed] [Google Scholar]
- 31.Hollsberg P, Batra V, Dressel A, Hafler DA. Induction of anergy in CD8 T cells by B cell presentation of antigen. J Immunol. 1996;157:5269–5276. [PubMed] [Google Scholar]
- 32.Mizoguchi A, Mizoguchi E, Takedatsu H, Blumberg RS, Bhan AK. Chronic intestinal inflammatory condition generates IL-10-producing regulatory B cell subset characterized by CD1d upregulation. Immunity. 2002;16:219–230. doi: 10.1016/s1074-7613(02)00274-1. [DOI] [PubMed] [Google Scholar]
- 33.Mauri C, Gray D, Mushtaq N, Londei M. Prevention of arthritis by interleukin 10-producing B cells. J Exp Med. 2003;197:489–501. doi: 10.1084/jem.20021293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Fillatreau S, Sweenie CH, McGeachy MJ, Gray D, Anderton SM. B cells regulate autoimmunity by provision of IL-10. Nat Immunol. 2002;3:944–950. doi: 10.1038/ni833. [DOI] [PubMed] [Google Scholar]
- 35.Olson TS, Bamias G, Naganuma M, Rivera-Nieves J, Burcin TL, Ross W, Morris MA, Pizarro TT, Ernst PB, Cominelli F, Ley K. Expanded B cell population blocks regulatory T cells and exacerbates ileitis in a murine model of Crohn disease. J Clin Invest. 2004;114:389–398. doi: 10.1172/JCI20855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Prescott SM, Zimmerman GA, Stafforini DM, McIntyre TM. Platelet-activating factor and related lipid mediators. Annu Rev Biochem. 2000;69:419–445. doi: 10.1146/annurev.biochem.69.1.419. [DOI] [PubMed] [Google Scholar]
- 37.Mossner R, Lesch KP. Role of serotonin in the immune system and in neuroimmune interactions. Brain Behav Immun. 1998;12:249–271. doi: 10.1006/brbi.1998.0532. [DOI] [PubMed] [Google Scholar]
- 38.Lesch KP, Mossner R. Genetically driven variation in serotonin uptake: is there a link to affective spectrum, neurodevelopmental, and neurode-generative disorders? Biol Psychiatry. 1998;44:179–192. doi: 10.1016/s0006-3223(98)00121-8. [DOI] [PubMed] [Google Scholar]
- 39.Matsuda H, Ushio H, Geba GP, Askenase PW. Human platelets can initiate T cell-dependent contact sensitivity through local serotonin release mediated by IgE antibodies. J Immunol. 1997;158:2891–2897. [PubMed] [Google Scholar]
- 40.Slominski A, Wortsman J, Tobin DJ. The cutaneous serotoninergic/melatoninergic system: securing a place under the sun. FASEB J. 2005;19:176–194. doi: 10.1096/fj.04-2079rev. [DOI] [PubMed] [Google Scholar]
- 41.Nguer CM, Pellegrini O, Galanaud P, Benveniste J, Thomas Y, Richard Y. Regulation of pafacether receptor expression in human B cells. J Immunol. 1992;149:2742–2748. [PubMed] [Google Scholar]
- 42.Nguer CM, Treton D, Rola-Pleszczynski M, Mishal Z, Thomas Y, Galanaud P, Richard Y. Regulation of platelet-activating factor receptor expression in human B cells and B cell lines. Lipids. 1996;31:1051–1058. doi: 10.1007/BF02522462. [DOI] [PubMed] [Google Scholar]
- 43.Bastien Y, Toledano BJ, Mehio N, Cameron L, Lamoukhaid B, Renzi P, Hamid Q, Mazer BD. Detection of functional platelet-activating factor receptors on human tonsillar B lymphocytes. J Immunol. 1999;162:5498–5505. [PubMed] [Google Scholar]
- 44.Fisher MS, Kripke ML. Suppressor T lymphocytes control the development of primary skin cancers in UV-irradiated mice. Science. 1982;216:1133–1134. doi: 10.1126/science.6210958. [DOI] [PubMed] [Google Scholar]
- 45.Schwarz A, Beissert S, Grosse-Heitmeyer K, Gunzer M, Bluestone JA, Grabbe S, Schwarz T. Evidence for functional relevance of CTLA-4 in ultraviolet-radiation-induced tolerance. J Immunol. 2000;165:1824–1831. doi: 10.4049/jimmunol.165.4.1824. [DOI] [PubMed] [Google Scholar]
- 46.Shreedhar VK, Pride MW, Sun Y, Kripke ML, Strickland FM. Origin and characteristics of ultraviolet-B radiation-induced suppressor T lymphocytes. J Immunol. 1998;161:1327–1335. [PubMed] [Google Scholar]
- 47.Kripke ML, Munn CG, Jeevan A, Tang JM, Bucana C. Evidence that cutaneous antigen-presenting cells migrate to regional lymph nodes during contact sensitization. J Immunol. 1990;145:2833–2838. [PubMed] [Google Scholar]
- 48.Alard P, Kurimoto I, Niizeki H, Doherty JM, Streilein JW. Hapten-specific tolerance induced by acute, low-dose ultraviolet B radiation of skin requires mast cell degranulation. Eur J Immunol. 2001;31:1736–1746. doi: 10.1002/1521-4141(200106)31:6<1736::aid-immu1736>3.0.co;2-t. [DOI] [PubMed] [Google Scholar]
- 49.Wakkach A, Fournier N, Brun V, Breittmayer JP, Cottrez F, Groux H. Characterization of dendritic cells that induce tolerance and T regulatory 1 cell differentiation in vivo. Immunity. 2003;18:605– 617. doi: 10.1016/s1074-7613(03)00113-4. [DOI] [PubMed] [Google Scholar]
- 50.Kalinski P, Hilkens CMU, Snijders A, Snijdewint FGM, Kapsenberg ML. IL-12-deficient dendritic cells, generated in the presence of prostaglandin E2, promote type 2 cytokine production in maturing human naive T helper cells. J Immunol. 1997;159:28–35. [PubMed] [Google Scholar]
- 51.Kalinski P, Vieira PL, Schuitemaker JH, de Jong EC, Kapsenberg ML. Prostaglandin E2 is a selective inducer of interleukin-12 p40 (IL- 12p40) production and an inhibitor of bioactive IL-12p70 heterodimer. Blood. 2001;97:3466–3469. doi: 10.1182/blood.v97.11.3466. [DOI] [PubMed] [Google Scholar]
- 52.Byrne SN, Halliday GM. B cells activated in lymph nodes in response to ultraviolet irradiation or by interleukin-10 inhibit dendritic cell induction of immunity. J Invest Dermatol. 2005;124:570–578. doi: 10.1111/j.0022-202X.2005.23615.x. [DOI] [PubMed] [Google Scholar]
- 53.Ishii S, Shimizu T. Platelet-activating factor (PAF) receptor and genetically engineered PAF receptor mutant mice. Prog Lipid Res. 2000;39:41–82. doi: 10.1016/s0163-7827(99)00016-8. [DOI] [PubMed] [Google Scholar]
- 54.Darst M, Al-Hassani M, Li T, Yi Q, Travers JM, Lewis DA, Travers JB. Augmentation of chemotherapy-induced cytokine production by expression of the platelet-activating factor receptor in a human epithelial carcinoma cell line. J Immunol. 2004;172:6330–6335. doi: 10.4049/jimmunol.172.10.6330. [DOI] [PubMed] [Google Scholar]
- 55.Ullrich SE. Dermal application of JP-8 jet fuel induces immune suppression. Toxicol Sci. 1999;52:61–67. doi: 10.1093/toxsci/52.1.61. [DOI] [PubMed] [Google Scholar]
- 56.Ullrich SE, Lyons HJ. Mechanisms involved in the immunotoxicity induced by dermal application of JP-8 jet fuel. Toxicol Sci. 2000;58:290–298. doi: 10.1093/toxsci/58.2.290. [DOI] [PubMed] [Google Scholar]


