Abstract
In this research we examined the mechanisms by which ethanol (EtOH) inhibits luteinizing hormone-releasing hormone (LHRH) release from incubated medial basal hypothalamic explants. EtOH (100 mM) stimulated the release of two inhibitory neurotransmitters: γ-aminobutyric acid (GABA) and β-endorphin. EtOH also inhibited NO production, indicative of a suppression of nitric oxide synthase (NOS) activity. This inhibition was reversed by naltroxone (10−8 M), a μ-opioid receptor blocker, indicating that the inhibition of NOS by EtOH is mediated by β-endorphin. EtOH also blocked N-methyl-d-aspartic acid-induced LHRH release, but the blockade could not be reversed by either the GABA receptor blocker, bicuculline (10−5 M), naltroxone (10−8 M), or both inhibitors added together. However, increasing the concentration of naltrexone (10−6 M) but not bicuculline (10−4 M) reversed the inhibition. When we lowered the concentration of EtOH (50 mM), the EtOH-induced blockade of LHRH release could be reversed by either bicuculline (10−5 M), naltroxone (10−8 M), or the combination of the two blockers. Therefore, GABA is partially responsible for the blockade of N-methyl-d-aspartic acid-induced LHRH release. The block by GABA was exerted by inhibiting the activation of cyclooxygenase by NO, because it was reversed by prostaglandin E2, the product of activation of cyclooxygenase. Because the inhibition caused by the higher concentration of EtOH could not be reduced by bicuculline (10−4 M) but was blocked by naltroxone (10−6 M), the action of alcohol can be accounted for by stimulation of β-endorphin neurons that inhibit LHRH release by inhibition of activation of NOS and stimulation of GABA release.
Keywords: nitric oxide synthase, cyclooxygenase, prostaglandin E2, naltrexone, bicuculline
Ethanol (EtOH) can suppress reproductive function in humans, monkeys, and small rodents, such as the rat (1–4). In conscious animals, administration of EtOH via an indwelling gastric cannula in doses that produce mild intoxication inhibits pulsatile release of luteinizing hormone (LH), but not follicle-stimulating hormone (4). In this situation, the responsiveness of the pituitary gland to acute injection of LH-releasing hormone (LHRH) is unaffected, which indicates that the mechanism of this effect is via suppression of pulsatile LHRH release into the hypophyseal portal vessels that triggers release of LH from gonadotropes of the adenohypophysis.
Nitric oxide synthase (NOS)-containing neurons occur in various regions of the hypothalamus, including the median eminence-arcuate region (5). Previous research has indicated that NO stimulates the release of LHRH both in vivo and in vitro (6). On the basis of in vitro experiments using incubation of medial basal hypothalamus explants (MBH) in a static incubation system, it has been determined that norepinephrine (NE) activates constitutive NOS in this region (5–7). The NO released from these neurons diffuses to LHRH terminals, where it induces the release of LHRH. It has been shown that NO not only activates guanylate cyclase followed by increased cGMP release but also activates cyclooxygenase (COX) that increases release of prostanoids (7). Prostaglandin E2 (PGE2) by activating adenylate cyclase (8) with consequent increase in cAMP evokes exocytosis of LHRH granules by activation of protein kinase A. The LHRH released diffuses into the hypophyseal portal vessels that deliver it to the anterior pituitary gland where it acts on gonadotropes to release LH (9). Support for this theoretical pathway stems from the ability of inhibitors of NOS, such as NG-monomethyl-l-arginine, to inhibit LHRH release, whereas releasers of NO, such as sodium nitroprusside (NP), induce LHRH release as well as that of PGE2 from MBH (6). The action of NP is caused by the combined activation of soluble guanylate cyclase that increases cGMP and COX that liberates PGE2, which also can stimulate LHRH release (6).
The release of LHRH is not only under the control of stimulatory neurotransmitters such as NE (9, 10) and glutamic acid (11) but is also under the control of inhibitory neurotransmiters such as γ-amino butyric acid (GABA) and β-endorphin, both of which inhibit LHRH release in female (12) and male rats (13). The inhibitory action of GABA on LHRH release also is mediated by NO, because NP-stimulated GABA release was prevented by hemoglobin (a scavenger of NO). It is possible that the increase in GABA release during LHRH secretion induced by NO could be a mechanism to terminate the pulses of LHRH (14).
β-endorphin also can inhibit LHRH release, probably by stimulating μ-opiate receptors on NOergic neurons because we have shown that β-endorphin inhibits the activity of NOS in MBH, whereas naltrexone, a μ-opiate receptor antagonist, increased the activity of NOS in this tissue. Furthermore, β-endorphin also blocked the action of NP on PGE2 release and consequently LHRH secretion (15).
We showed previously that EtOH blocks the release of PGE2 and LHRH induced by NP (5). Because EtOH has been shown to alter GABA (16) and β-endorphin release (17, 18), we hypothesized that the inhibition of LHRH release by EtOH could be caused by increased release of these inhibitory neurotransmitters with consequent decrease in NO generation that would decrease LHRH secretion. The present experiments were designed to test this hypothesis to elucidate the complex mechanisms by which EtOH inhibits LHRH release.
Materials and Methods
Male rats of the Wistar strain (200–250 g) from our colony were used. All rats were kept in group cages in a light-controlled (0500–1900 hr) and temperature-controlled (23–25°C) room with free access to laboratory chow and water.
In Vitro Studies.
After decapitation and removal of the brain, the MBH was dissected by making frontal cuts just behind the optic chiasm, extending dorsally 1.0 mm, a horizontal cut extended from this point caudally to just behind the pituitary stalk, where another frontal cut was made. Longitudinal cuts were made 1 mm lateral to the midline bilaterally. The hypothalami were preincubated in Krebs–Ringer bicarbonate-buffered (KRB) medium (pH 7.4) containing 0.1% glucose-KRB for 30 min before replacement with fresh medium or medium containing the substances to be tested. The incubation was continued for 30 min followed by removal of the medium and storage of samples at −20°C before assay for LHRH or β-endorphin. All incubations were carried out in a Dubnoff metabolic shaker (50 cycles per min; 95% O2/5% CO2) at 37°C.
LHRH was measured by RIA with highly specific LHRH antiserum kindly provided by Ayala Barnea (University of Texas Southwestern Medical Center, Dallas). The sensitivity of the assay was 0.2 pg per tube, and the curve was linear up to 100 pg of LHRH. β-endorphin also was measured by RIA using a highly specific antiserum kindly provided by George Chrousos (National Institute of Child Health and Human Development, Bethesda, MD).
For the determination of GABA release, two MBH were used for each determination. They were incubated in 0.1% glucose KRB medium with addition of 20 mM Hepes, 1 mM ascorbic acid, and 0.1% BSA. The preincubation and incubation times were the same as for LHRH, as described above. At the end of the incubation period, the media were heated for 10 min at 100°C and centrifuged at 10,000 × g for 10 min. The supernatants were frozen at −70°C until determination of GABA by the [3H]muscimol radioreceptor assay as described by Bernasconi et al. (19).
Determination of NOS activity was performed by a modification (5) of the [14C]citrulline method of Bredt and Snyder (20). Briefly, after preincubation of the MBH (one per tube) for 15 min in 0.1% glucose KRB medium, they were exposed to different compounds and 0.1% glucose KRB medium (pH 7.4) for another 15 min. At the end of the incubation period the medium was discarded and the MBH were homogenized in 20 mM Hepes (pH 7.4) with addition of 1.25 mM CaCl2 and 1 mM DTT. After the MBH was homogenized, 120 μM NADPH and 200,000 dpm of [14C]arginine (297 mCi/mmol) were added to each tube and incubated for an additional 10 min under the same conditions as described above. At the end of this 10 min the tubes were transferred immediately to a refrigerated centrifuge and spun at 10,000 × g for 10 min, and the supernatants were immediately applied to individual columns of Dowex AG 50W-X8 200 mesh sodium form and washed with 2 ml of double-distilled water. All collected fluid from each column was counted for [14C]citrulline activity in a scintillation counter. Because NOS converts arginine into equimolar quantities of NO and citrulline, the data were expressed as NO produced per MBH per 10 min.
Chemicals.
LHRH and β-endorphin for iodination and standards were purchased from Peninsula Laboratories. Iodine-125 for iodination was purchased from DuPont/New England Nuclear. Naltrexone, GABA, EtOH, sodium NP, bicuculline, N-methyl-d-aspartic acid (NMDA), Hepes, NADPH, and DTT were from Sigma. Dowex AG-50W-X8 Na+ form mesh 200–400 was from Bio-Rad, and (U-14C)-arginine-monohydrochloride 296 mCi/mmol was from Amersham Pharmacia.
Statistics.
All data are expressed as means plus one SEM. Comparisons between groups were performed by using a one-way ANOVA followed by the Student-Newman-Keuls multiple comparison test for unequal replicates. Dunnett's test was used when comparisons were made versus the control group. Student's t test was used when comparing two groups. Differences with P values <0.05 were considered significant.
Results
Effect of EtOH and β-Endorphin on GABA Release.
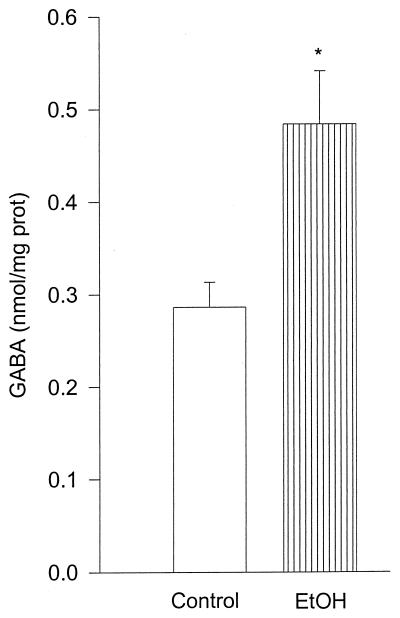
Previous experiments demonstrated that incubation of MBH with β-endorphin (10 nM) significantly increased GABA release (15) and also that NO had a stimulatory effect (14). In the present experiments we found that EtOH (100 mM) significantly stimulated (P < 0.05) GABA release (Fig. 1).
Figure 1.
The effect of EtOH (100 mM) on GABA release from MBH incubated in vitro. In this and subsequent figures, the height of the column represents the mean and the vertical line represents 1 SEM. Significantly different from control, *, P < 0.05. n = 7 from each column (Student-Neuman-Keuls test).
Effect of EtOH, GABA, and NP on β-Endorphin Release.
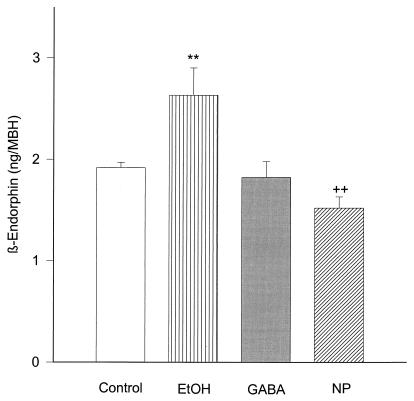
As shown previously the addition of GABA (10 mM) did not affect β-endorphin release from MBH (15). This result was confirmed in the current experiments (Fig. 2). EtOH (100 mM) significantly increased (P < 0.01) the release of β-endorphin from MBH, and the addition of NP, an NO donor (600 μM), significantly inhibited (P < 0.01) β-endorphin release (Fig. 2).
Figure 2.
The effect of EtOH (100 mM), GABA (10 mM), and NP (600 μM) on β-endorphin release from MBH incubated in vitro. **, P < 0.01 as compared with Control column. n = 7 for each column (Dunnet test). EtOH stimulated, GABA did not modify, and NP inhibited β-endorphin release.
Effect of EtOH on NMDA-Stimulated NOS Activity.
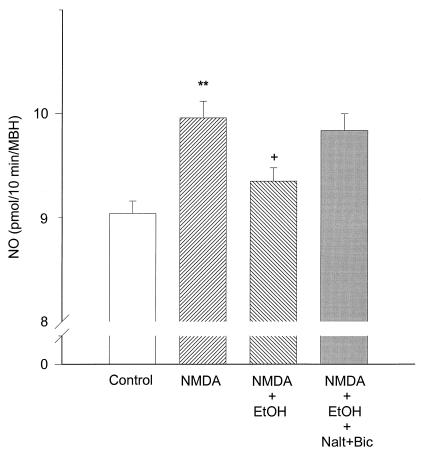
The activity of NOS in MBH in the presence of NMDA (20 mM) was significantly (P < 0.01) increased as measured by the citrulline method (Fig. 3). This increase was significantly (P < 0.05) diminished by EtOH (Fig. 3), and the inhibition was reversed by the addition of naltrexone (10−8 M) (a μ-opiod receptor blocker) and bicuculline (10−5 M) (a GABAA receptor antagonist) that were added together (P < 0.05) (Fig. 3).
Figure 3.
The effect of NMDA (20 mM), EtOH (100 mM), bicuculline (Bic) (10 μM), naltrexone (Nalt) (10nM), and their combinations on NOS activity measured by the 14C-citrulline method. Because activation of the enzyme yields equimolar release of NO and citrulline, the results are plotted as NO release. **, P < 0.01, as compared versus control column; +, P < 0.05 as compared versus NMDA column and NMDA+EtOH+Nalt+Bic column. n = 9 for each column (Student-Neuman-Keul test). NMDA increased NOS activity, EtOH blocked it, and Nalt+Bic reverted this inhibition.
Effect of EtOH on NMDA-Stimulated LHRH Release.
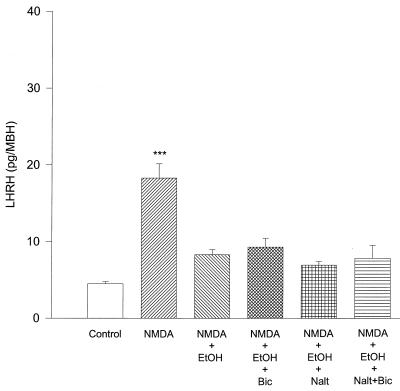
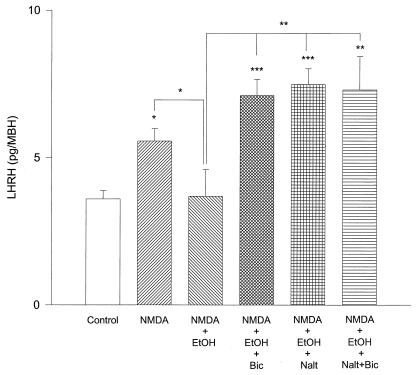
LHRH release was dramatically increased by NMDA (20 mM) (P < 0.001) and highly significantly (P < 0.001) blocked by EtOH (100 mM) (Fig. 4). To investigate whether this inhibition was caused by β-endorphin and/or GABA, naltrexone (10−8 M), bicuculline (10−5 M), or both inhibitors together were added to the media. The addition of these substances separately or in combination failed to reverse the inhibition produced by EtOH on NMDA-stimulated LHRH release (Fig. 4).
Figure 4.
The effect of NMDA (20 mM), EtOH (100 mM), bicuculline (Bic) (10 μM), naltrexone (Nalt) (10 nM), and their combinations on LHRH release from MBH incubated in vitro. ***, P < 0.001 versus all columns. n = 9 for each column (Student-Neuman-Keuls test). NMDA stimulated highly significant LHRH release. EtOH significantly blocked this stimulation, and neither Bic, Nalt, nor a combination of both would revert this inhibition.
When the dose of alcohol was reduced to 50 mM in contrast to the results with the higher concentration of EtOH (100 mM), bicuculline, naltrexone, or the two inhibitors together completely reversed the EtOH blockade of NMDA-stimulated LHRH release. All three of these interventions produced a similar increase in LHRH release that was significantly greater than that induced by NMDA alone (Fig. 5).
Figure 5.
Effect of EtOH (50 mM) on NMDA (20 mM)-induced LHRH. The same combination of drugs were used as in Fig. 4. In contrast to the results shown in Fig. 4, bicuculline (Bic) alone, naltrexone (Nalt) alone, or their combination all not only reversed the EtOH block of LHRH, but actually significantly increased the values of above those in the NMDA-stimulated controls. *, P < 0.05; **, P < 0.01; ***, P < 0.001.
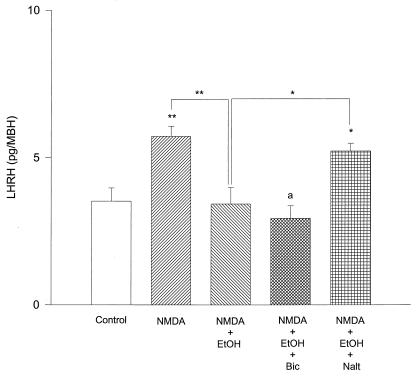
Because bicuculline and naltrexone were ineffective to block inhibition of LHRH release at the 100 mM concentration, increased concentrations of bicuculline (10−4 M) and naltrexone (10−6 M) were used. In that situation the increased concentration of bicuculline (10−4 M) had no effect on the inhibition of NMDA-induced LHRH obtained with the higher concentration of alcohol (100 mM), but the higher concentration of naltrexone (10−6 M) completely blocked the inhibition (Fig. 6).
Figure 6.
Effect of EtOH on NMDA-stimulated LHRH release of EtOH (100 mM) on NMDA-induced stimulation of LHRH release. The increased concentration of bicuculline (Bic) (10−4 M) did not reverse the EtOH-induced inhibition; however, naltrexone (Nalt) (10−6 M) completely reversed it. *, P < 0.05; **, P < 0.01; a, P < 0.05 vs control.
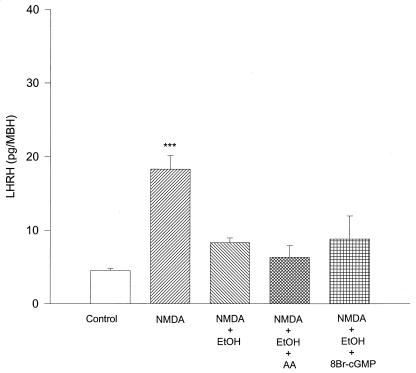
Effect of Arachidonic Acid (AA) and cGMP on the Inhibitory Action of EtOH on LHRH Release Stimulated by NMDA.
In previous studies we have shown that cGMP can increase LHRH release (5). In the present experiments we found that 8-bromo-cGMP (1 mM), a nonhydrolysable and soluble analogue of cGMP that induces LHRH release, had no effect on the inhibitory action of EtOH (100 mM) on NMDA (20 mM)-stimulated LHRH release (Fig. 7). Because previous experiments have shown that EtOH inhibits NP-stimulated PGE2 release from MBH (5), in these experiments we examined the possibility that EtOH inhibition of phospholipase A2 decreased availability of AA for conversion to PGE2 by COX. Therefore we added AA (50 μM) to MBH in the presence of NMDA and EtOH as described above and found no reversal of the inhibition of LHRH induced by EtOH (Fig. 7).
Figure 7.
Effect of NMDA (20 mM), EtOH (100 mM), AA (50 μM), 8-bromo-cGMP (nonhydrolizable analogue of cGMP) (1 mM), and their combinations on LHRH release from MBH in vitro. ***, P < 0.001 versus all columns. n = 5 per column (Student-Newman-Keuls test). NMDA highly significantly stimulated LHRH release that was blocked by EtOH, and AA or 8-bromo-cGMP failed to reverse this inhibition.
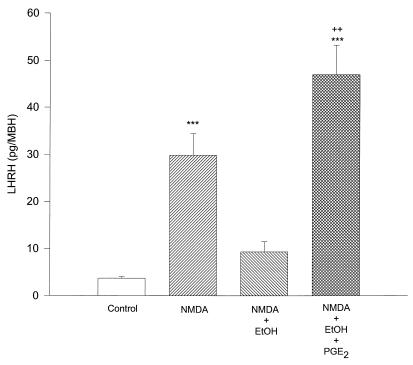
Effect of PGE2 on the Inhibitory Action of EtOH on Stimulated Release of LHRH by NMDA.
Because PGE2 is involved in the pathway of LHRH secretion and was inhibited by EtOH as shown in previous studies (5), in the present experiments we determined whether the addition of exogenous PGE2 could reverse the inhibitory action of EtOH on NMDA-stimulated release of LHRH. The addition of PGE2 (100 nM) not only was able to highly significantly overcome (P < 0.001) the inhibitory action of EtOH (100 mM) but even significantly increased (P < 0.01) the release of LHRH as compared with the release of LHRH stimulated by NMDA alone (20 mM) (Fig. 8).
Figure 8.
Effect of NMDA (20 mM) EtOH (100 mM), PGE2 (100 nM), and their combinations on LHRH release from MBH incubated in vitro. ***, P < 0.001 versus control and NMDA+EtOH columns. ++, P < 0.01 versus NMDA column. n = 6 for each column (Student-Neuman-Keuls test). NMDA highly significantly stimulated LHRH release that was blocked by EtOH, and the addition of PGE2 reversed this inhibition and further significantly increased LHRH release as compared with NMDA alone.
Discussion
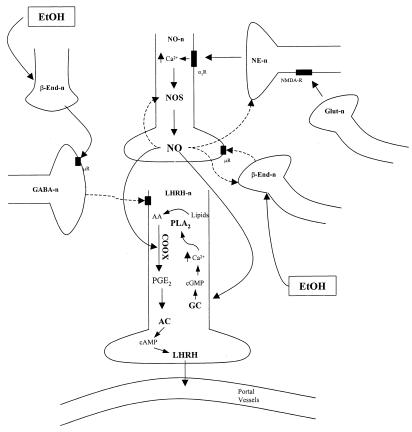
To understand the mechanism of action of alcohol it is necessary to put it into the context of our previous work on the NOergic control of LHRH release (Fig. 9). These experiments indicated that glutamergic neurons act on NMDA receptors on noradrenergic axons. The noradrenergic axons activate α1 adrenergic receptors on NOergic neurons to increase intracellular Ca2+ that combines with calmodulin to activate NOS. The released NO diffuses into LHRH neuronal terminals and stimulates LHRH release (9). This mechanism is confirmed here because NMDA, an excitatory amino acid that mimics the actions of glutamic acid, stimulated not only an increase in NOS activity, but also LHRH release. We previously have shown that β-endorphin inhibits LHRH release. Although β-endorphin activates GABA release, the released GABA appears to have no inhibitory effect on β-endorphin release, and GABA also does not directly affect the alteration of NOS activity (15). In the present experiments, we also have shown that NO released from NP appears to act back on the β-endorphin neurons to reduce secretion of β-endorphin.
Figure 9.
Diagrammatic representation of the postulated mechanism of action of EtOH to suppress NMDA-stimulated LHRH release. For explanation, see Discussion. β-end, β-endorphin; μR, μ-opioid receptor; GABA-n, GABA neuron; NO-n, NO-ergic neuron; NE-n, noradrenergic neuron; α1r, α1 adrenergic receptor; NMDA-r, NMDA receptor; Glut-n, glutamergic neuron; LHRH-n, LHRH neuronal terminal; lipids, membrane phospholipids; PLA2, phospholipase A2; GC, guanylate cyclase; AC, adenylate cyclase. Solid arrow indicates stimulation. Dashed arrow indicates inhibition.
Alcohol in concentrations that would be expected to produce moderate intoxication in vivo in rats (5) blocked the NMDA-induced increase in NOS activity and LHRH release. Our data indicate that the inhibitory action of EtOH on LHRH release is caused by its stimulation of β-endorphin release. As demonstrated previously (15), β-endorphin also stimulates GABA release by activation of mμ opioid receptors because its stimulatory effect was blocked by the mμ opioid receptor antagonist naltrexone. As shown here EtOH similarly stimulated GABA release.
Because the NMDA-induced stimulation of LHRH release was blocked by alcohol, one could argue that the inhibitory effect was mediated by GABA. Indeed, at the lower (50 mM) concentration of EtOH bicuculline (10−5 M), a GABAA receptor blocker blocked the inhibitory action of EtOH. However, even the higher dose of bicuculline (10−4 M) could not block the inhibitory effect of 100 mM EtOH, indicating that the inhibitory effect of alcohol is mediated only in part by GABA.
The inhibitory action of β-endorphin on LHRH is mediated at least in part by suppression of the activity of NOS. The action of alcohol to lower NOS activity was blocked by the combined action of bicuculline (10−5 M) and naltrexone (10−8 M). Because GABA by itself has no effect on NOS activity, this result probably is caused by the inhibitory action of naltrexone on the inhibitory activity of β-endorphin on NOS. Furthermore, in previous experiments, naltrexone (10−8 M) alone blocked the suppression of NOS induced by β-endorphin (15). Not only did GABA not act in earlier experiments to inhibit NOS (14), but also in these experiments, it did not have any effect on β-endorphin release. On the other hand, NP that releases NO inhibited β-endorphin release, suggesting a negative feedback of NO to suppress β-endorphin release.
EtOH also acts after the generation of NO because it blocks NP-induced LHRH and PGE2 release (5). Although the lower dose of bicuculline (10−5 M) blocked the LHRH release that followed the lower concentration of EtOH (50 mM), increasing the concentration of bicuculline to 10−4 M failed to block the inhibitory effect of the higher concentration of EtOH (100 mM) on the response to NMDA. At the lower EtOH concentration (50 mM), bicuculline (10−5 M) not only reversed the block but even increased LHRH release above that to NMDA alone, indicating that this action of GABA to block the response of the LHRH terminals to NO is probably important in mediating the action of alcohol. However, the release of GABA probably is caused by the EtOH-induced release of β-endorphin, because alcohol caused an increase in β-endorphin and GABA release.
The primary action of GABA appears to be to inhibit the NOergic activation of COX because addition of effective concentrations of 8-bromo cGMP to correct for a possible blockade of guanylyl cyclase did not reverse the action of GABA (14) and had no effect on the suppressive action of alcohol. Provision of arachidonate to provide more substrate for COX also failed to reverse the block, suggesting that the primary action of both GABA and EtOH was by suppression of COX. This conclusion was supported by the finding that the addition of PGE2 reversed the EtOH block.
Although the higher dose of bicuculline (10−4 M) failed to reverse the inhibitory effect of alcohol (100 mM) on LHRH release, naltrexone (10−6 M) completely blocked it. This finding suggests that the primary effect of alcohol was to stimulate β-endorphin release, which then in turn stimulated GABA release. However, even when the action of GABA was not blocked, the inhibition of LHRH release could be reversed by naltrexone because it reversed the suppression of NOS activity induced by β-endorphin.
At the lower concentration of EtOH (50 mM), either the lower dose of bicuculline (10−5 M), naltrexone (10−8 M), or the two together blocked the inhibitory effect of alcohol completely. In fact, not only was the inhibitory action blocked, but the release of LHRH was increased above the levels obtained with NMDA alone. This finding suggests that not only is β-endorphin being released spontaneously from the preparation in vitro but that it is in turn releasing GABA. Both of these are acting together to suppress “basal” LHRH release in vitro by their actions to suppress the response to the release of NO in the case of GABA, and by the combined action of β-endorphin to not only suppress NOS, but also to release GABA, which then blocks the response to residual NO that might be released.
The results reported here indicate that the suppression of LHRH release from MBH in vitro by EtOH can be explained by its stimulation of the release of β-endorphin. β-endorphin not only inhibits NOS, thereby decreasing NO release that activates LHRH release, but also stimulates GABA release that accounts in part for the inhibitory effect of EtOH on LHRH release by blocking the response of the LHRH neuronal terminals to NO. We hypothesize that alcohol acts on putative EtOH receptors on the cell bodies of β-endorphinergic neurons located in the arcuate nucleus that are resident within the MBH to stimulate β-endorphin release. The β-endorphin released inhibits LHRH release by the pathway described. Naltrexone has been found to be effective in reducing the craving for alcohol of alcoholics (21), possibly because naltrexone blocks alcohol-induced β-endorphin release, which not only blocks LHRH release but also contributes to the euphoric action of alcohol through actions in the limbic system.
The action of alcohol to block LHRH release is shared by the proinflammatory cytokines, granulocyte/macrophage colony-stimulating factor (GM-CSF) and IL-1α. GM-CSF has been shown to block LHRH release by stimulation of GABA release that blocks the response of LHRH neuronal terminals to NO (22). The response to NO also is blocked by IL-1α (23), and we have shown that IL-1α (10−11 M) stimulates β-endorphin release as well (V. R., unpublished data). Therefore, we hypothesize that the actions of proinflammatory cytokines to inhibit LHRH release also are mediated by the β-endorphin pathway described here.
Acknowledgments
We thank Ana Ines Casella for her administrative assistance and Judy Scott for preparation of the manuscript. This work was supported by Grants PIA 6649 and BID 802-OC-AR-PICT 00353 from Consejo Nacional de Investigaciones Científicas y Tecnicas (CONICET) (V.R.) and National Institutes of Health Grant MH51853 (S.M.M.).
Abbreviations
- LH
luteinizing hormone
- LHRH
LH-releasing hormone
- GABA
γ-aminobutyric acid
- NOS
nitric oxide synthase
- NMDA
N-methyl-d-aspartic acid
- MBH
medial basal hypothalamus explants
- NE
norepinephrine
- PGE2
prostaglandin E2
- NP
nitroprusside
- KRB
Krebs–Ringer bicarbonate-buffered
- EtOH
ethanol
- AA
arachidonic acid
- COX
cyclooxygenase
Footnotes
Article published online before print: Proc. Natl. Acad. Sci. USA, 10.1073/pnas.040569597.
Article and publication date are at www.pnas.org/cgi/doi/10.1073/pnas.040569597
References
- 1.Cicero T J, Badger T. J Pharmacol Exp Ther. 1977;201:427–433. [PubMed] [Google Scholar]
- 2.Cicero T J, Newman K S, Gerrity M, Scumoker P F, Bell R D. Life Sci. 1982;31:1587–1596. doi: 10.1016/0024-3205(82)90050-9. [DOI] [PubMed] [Google Scholar]
- 3.Mendelson J H, Mello N K, Ellingboe J. Exp Ther. 1977;202:676–682. [PubMed] [Google Scholar]
- 4.Dees W L, Rettori V, Kozlowski G P, McCann S M. Alcohol. 1985;2:641–646. doi: 10.1016/0741-8329(85)90139-9. [DOI] [PubMed] [Google Scholar]
- 5.Canteros G, Rettori V, Franchi A, Genaro A, Cebral E, Faletti A, Gimeno M, McCann S M. Proc Natl Acad Sci USA. 1995;92:3416–3420. doi: 10.1073/pnas.92.8.3416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rettori V, Belova N, Dees W L, Nyberg C L, Gimeno M, McCann S M. Proc Natl Acad Sci USA. 1993;90:10130–10134. doi: 10.1073/pnas.90.21.10130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rettori V, Gimeno M, Lyson K, McCann S M. Proc Natl Acad Sci USA. 1992;89:11543–11546. doi: 10.1073/pnas.89.23.11543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Negro-Vilar A, Ojeda S M, McCann S M. Endocrinology. 1979;104:1749–1757. doi: 10.1210/endo-104-6-1749. [DOI] [PubMed] [Google Scholar]
- 9.McCann S M, Rettori V. Proc Soc Exp Biol Med. 1996;211:7–15. doi: 10.3181/00379727-211-43950b. [DOI] [PubMed] [Google Scholar]
- 10.Negro-Vilar A, Advis J P, Ojeda S R, McCann S M. Endocrinology. 1982;111:932–938. doi: 10.1210/endo-111-3-932. [DOI] [PubMed] [Google Scholar]
- 11.Rettori V, Kamat A, McCann S M. Brain Res Bull. 1994;33:501–503. doi: 10.1016/0361-9230(94)90074-4. [DOI] [PubMed] [Google Scholar]
- 12.McCann S M, Rettori V. Adv Biochem Psychopharmacol. 1986;42:173–179. [PubMed] [Google Scholar]
- 13.Masotto C, Wisnieski G, Negro-Vilar A. Endocrinology. 1989;125:548–553. doi: 10.1210/endo-125-1-548. [DOI] [PubMed] [Google Scholar]
- 14.Seilicovich A, Duvilanski B H, Pizera D, Theas S, Gimeno M, Rettori V, McCann S M. Proc Natl Acad Sci USA. 1995;92:3421–3424. doi: 10.1073/pnas.92.8.3421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Faletti A G, Mastronardi C A, Lomniczi A, Seilicovich A, Gimeno M, McCann S M, Rettori V. Proc Natl Acad Sci USA. 1999;96:1722–1726. doi: 10.1073/pnas.96.4.1722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Seilicovich A, Duvilanski B, Gonzalez N N, Rettori V, Mitridate de Novara A, Muñoz Maines V, Fitzer de Plazas S. Eur J Pharmacol. 1985;111:365–368. doi: 10.1016/0014-2999(85)90643-0. [DOI] [PubMed] [Google Scholar]
- 17.Seizinger B, Boverman K, Hollt V, Herz A. J Pharmacol Exp Ther. 1984;230:455–460. [PubMed] [Google Scholar]
- 18.Herz A. Psychopharmacology. 1997;129:99–111. doi: 10.1007/s002130050169. [DOI] [PubMed] [Google Scholar]
- 19.Bernasconi R, Bittinger H, Hied J, Martin P. J Neurochem. 1980;34:614–618. doi: 10.1111/j.1471-4159.1980.tb11188.x. [DOI] [PubMed] [Google Scholar]
- 20.Bredt D S, Snyder S H. Proc Natl Acad Sci USA. 1989;86:9030–9033. doi: 10.1073/pnas.86.22.9030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Croop R S, Faulkner E B, Labriola D F. Arch Gen Psychiatry. 1997;54:1130–1135. doi: 10.1001/archpsyc.1997.01830240090013. [DOI] [PubMed] [Google Scholar]
- 22.Kimura M, Yu W H, Rettori V, McCann S M. Neuroimmunomodulation. 1997;4:237–243. doi: 10.1159/000097342. [DOI] [PubMed] [Google Scholar]
- 23.Rettori V, Belova N, Kamat A, Lyson K, McCann S M. Neuroimmunomodulation. 1994;1:86–91. doi: 10.1159/000097095. [DOI] [PubMed] [Google Scholar]