Abstract
The limited current understanding of megakaryocyte-lineage development and megakaryocyte biology is in large part because of a paucity of useful systems in which to conduct experiments. To overcome this problem, we have developed a transgenic mouse that uses the GP-Ibα regulatory sequences to achieve megakaryocyte-lineage restricted expression of an avian retroviral receptor. Through the transgenic avian receptor, avian retroviruses can efficiently and selectively infect megakaryocyte-lineage cells in vitro and in vivo. Serial infections can be performed to introduce and express multiple genes in the same cell. We have used this system to generate and characterize a pure population of primary CD41-positive megakaryocyte progenitors.
Despite the biological importance of platelets in both health and disease, remarkably little is known about the molecular mechanisms involved in megakaryocytopoiesis and platelet formation. Many factors have contributed to the limited current understanding of this basic biological process. The minimal representation of megakaryocytes in the bone marrow, estimated at <0.5% of the bone marrow cells (1), has hindered the procurement of purified primary cell populations. Human cell lines with megakaryocyte-related characteristics or markers have many shortcomings, including outgrowth from leukemic bone marrow, expression of multiple nonmegakaryocyte lineage markers, and abnormal karyotypes (2). Murine megakaryocyte cell lines are essentially nonexistent. In addition, the limited number of megakaryocyte lineage markers and the lack of identifiable developmental stages have prevented an understanding of the sequential steps of megakaryocyte lineage development. To improve on the available reagents, we have developed a murine model in which to study megakaryocytopoiesis and the biological properties of megakaryocyte-lineage cells.
Retroviral vectors are excellent tools for introducing genetic material into cells of interest (3), allowing one to study the biological effect(s) of gene products expressed by the incoming virus. The commonly used murine vectors infect a broad range of murine tissue types, including hematopoietic cells. In contrast, avian retroviral vectors cannot replicate in mammalian cells because of a block to viral entry (lack of the needed receptor) and a block in intracellular virus assembly. The expression of the subgroup A avian leukosis virus (ALV-A) receptor, TVA, on the surface of mammalian cells confers susceptibility to ALV-A infection and to infection by retroviruses pseudotyped with the ALV-A envelope protein (4, 5). Furthermore, TVA expression in muscle (6) or glial cells (7) allows for tissue-restricted infection in transgenic mice. We reasoned that transgenic mice with TVA expression limited to cells of the megakaryocyte lineage would allow us to selectively infect megakaryocyte lineage cells with avian retroviruses, providing a valuable system for studying the least understood hematopoietic lineage.
Megakaryocyte lineage markers are few in number and have been identified largely by their expression on platelets. They include, but are not limited to, GP-IIb, GP-IX, PF4, and GP-Ibα. Regulatory regions from the PF4 gene (8–10) and the GP-Ibα gene (11, 12) have been used to express heterologous proteins in transgenic mice, and transgene expression appears to be restricted to the megakaryocyte lineage within the hematopoietic tissues. Herein we describe a transgenic mouse that uses the GP-Ibα regulatory sequences to express TVA in a megakaryocyte-lineage restricted fashion. The TVA-expressing hematopoietic cells can be selectively infected with avian retroviruses in culture and in live animals. The inefficient expression of the ALV-A env gene and its associated failure to prevent superinfection by additional viral particles (13) allows for the independent and sequential introduction of multiple genetic sequences. We have used this model system to generate and characterize a population of primary CD41-positive megakaryocyte progenitors.
MATERIALS AND METHODS
Cell Lines.
293T cells and DF1 cells (a chicken fibroblast cell line developed by Doug Foster, University of Minnesota, and Steve Hughes, National Cancer Institute, Frederick, MD) were grown in DMEM-21 with 10% fetal calf serum (GIBCO/BRL), 100 units of penicillin G sodium per ml, and 0.1 mg of streptomycin sulfate per ml.
Plasmids and Viral Vectors.
The 2738 bp of sequence immediately upstream of the human GP-Ibα transcription start site came from plasmid p-2738/LUC (14). The tv-a cDNA sequence is from pΔ72KpnI-RI-tva950 (a gift of Paul Bates, University of Pennsylvania, Philadelphia). β-Galactosidase (β-gal)-expressing murine leukemia virus retroviral vectors pseudotyped with ALV-A envelope protein were generated by CaPO4 transfection of 293T cells with: pHIT111 (packageable sequence expressing the LacZ gene), pHIT60 (murine leukemia virus gag-pol expression plasmid), and pCB6-envA (encodes the ALV subgroup A env gene) (15). Supernatants were harvested 48–72 hr later, filtered (0.45 μm), and used as fresh virus stock. The RCAS avian retroviral vector system produces replication competent avian retrovirus and was generated in DF1 cells as described (16–18). DF1 cells, RCAS-PURO (expresses puromycin-resistance gene), and RCAS-AP (expresses human placental alkaline phosphatase; HPAP) were kindly provided by Eric Holland (National Institutes of Health, Bethesda, MD) and Stephen Hughes (National Cancer Institute).
Generation of Transgenic Mice.
The tv-a cDNA sequence contained in a HindIII–ClaI fragment from pΔ72Kpn-RI-tva950 was ligated into p-2738/LUC. The resulting plasmid (pGP-Ibα-TVA) positioned the tv-a translation initiation codon at the site of the normal initiation codon in exon 2 of GP-Ibα (Fig. 1), immediately downstream of 3086 bp of GP-Ibα sequence that includes 330 bp from the first exon, the first intron, and the proximal end of the second exon. A 4.1-kb NsiI–ClaI fragment containing the GP-Ibα regulatory sequence and tv-a cDNA was used to inject oocytes by using standard procedures (19).
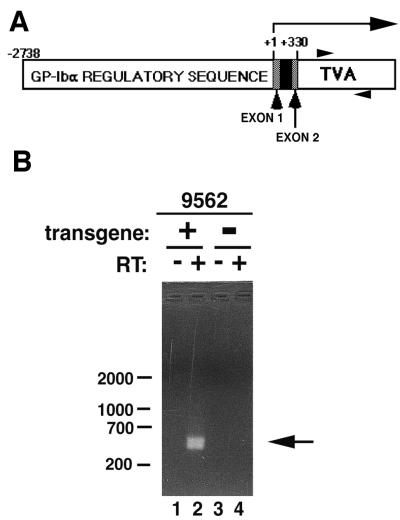
Figure 1.
Generation of transgenic mice expressing the ALV receptor. (A) A schematic of the transgene cassette used to express TVA indicates the positions of the 5′ (−2738) and 3′ (+330) ends of the GP-Ibα regulatory sequence ligated to 950 bp of tv-a cDNA. Numbering is assigned relative to “0” for the native GP-Ibα transcription start site. The 330-bp of untranslated GP-Ibα sequence includes the first exon and part of the second exon (░⃞) separated by the first intron (■). The horizontal arrow indicates the transcribed message. Translation begins with a methionine at the 5′ end of the tv-a cDNA. Arrowheads indicate the positions of primers used for RT-PCR. (B) A founder mouse positive for the transgene by Southern blot analysis, no. 9562, was bred with nontransgenic littermates. RNA was isolated from the bone marrow of a transgene-containing offspring (9562 “+”) and a nontransgenic littermate (9562 “−”) and was subjected to RT-PCR without (−) and with (+) reverse transcriptase in the reaction. The 420-bp band indicative of TVA mRNA (arrow) is seen in lane 2. The 420-bp signal (lane 2) is absent when reverse transcriptase is not included in the RT-PCR reaction (lane 1). Bone marrow from mice lacking the transgene do not express TVA mRNA (lanes 3 and 4). DNA size markers (bp) are indicated on the left.
Screening Transgenic Mice.
Distal tail segments were dissolved overnight at 55°C, and precipitated DNA was resuspended in 200 μl of sterile ddH2O. NcoI-digested tail DNA (15 μl) was used in Southern blot analyses as described (20). The tv-a probe, a 620-bp EagI–EcoRI fragment from pΔ72Kpn-RI-tva-950, was radiolabeled as described (20). Tv-a mRNA expression in bone marrow was identified by reverse transcription (RT)–PCR: cDNA copies of total mRNA (TRIzol reagent; BRL) were generated by using the SuperScript reverse transcription system (GIBCO/BRL) according to the manufacturer’s recommendations, followed by 30 cycles of PCR (30 sec at 95°C, 30 sec at 58°C, and 30 sec at 72°C) by using primers TVA#1 (5′-TGCTGCCCGGTAACGTGA-3′) and TVA#2 (5′-GGCAGAGCAGTTCAGTCC-3′) to generate a 420-bp product.
Cell Staining.
Hematopoietic cells were washed in PBS and applied to glass slides by using a Stat-Spin Technologies cytofuge (Norwood, MA). Wright-Giemsa (Baxter) stains were performed according to the manufacturer. For immunohistochemical staining, bone marrow cells were air dried for 5 min and then fixed in methanol or Diff-Quick Fixative (Baxter). Endogenous peroxidase activity was quenched by a 30-min room-temperature incubation in 0.3% H2O2. Cells were then soaked in Ca2+-free, Mg2+-free PBS for 5 min at room temperature, followed by a 20-min room-temperature incubation in normal rabbit (for anti-CD41) or normal goat (for anti-TVA) serum. Excess liquid was removed by blotting. To detect CD41 expression, fixed and blocked cells were then incubated for 30 min at room temperature in a 1:100 dilution of a rat mAb against murine CD41 (PharMingen), followed by a 5-min soak in Ca2+-free, Mg2+-free PBS. A rabbit polyclonal directed against murine P-selectin was used at 1:500 (PharMingen), a rabbit polyclonal that reacts with human and murine von Willebrand factor (vWF) was used at 1:1,000 (Dako), and a 1:1,000 dilution of an antigen-affinity purified rabbit polyclonal antibody directed against TVA, each followed by a 5-min soak in Ca2+-free Mg2+-free PBS. Biotinylated secondary antibodies and developing reagents were used according to the Vectastain Elite ABC kit (Vector Laboratories). β-gal (21) and alkaline phosphatase (22) stains were performed as described.
Tissue Staining.
Mouse tissue was fixed in formalin and embedded in paraffin. Tissue sections were applied to glass slides, deparaffinized by serial soaking in Xylene, 100% ethanol, 95% ethanol, 70% ethanol, ddH20, and PBS, and stained for TVA expression as described above.
Bone Marrow Cultures.
Freshly harvested femurs and tibias were flushed with Hanks’ buffered salt solution containing 1% BSA. Mature red blood cells were lysed by a 5 min room temperature incubation in 5 ml of ACK lysis buffer (23), and the cells were then resuspended in specific growth medium. All conditions used Iscove’s modified Dulbecco’s medium (IMDM) supplemented with 10% horse serum (GIBCO/BRL), 100 units of penicillin G sodium per ml, and 0.1 mg of streptomycin sulfate per ml. Cytokine supplements included stem cell factor (SCF; Peprotech) at 10 or 20 ng/ml, thrombopoietin (TPO; R&D Systems) at 50 ng/ml, and IL-3 (Peprotech) at 10 ng/ml.
Fluorescence-Activated Cell Sorter (FACS) Analysis.
After 3 days of culture in IMDM supplemented with SCF, IL-3, and TPO, 0.5–1.0 × 106 bone marrow cells were washed in PBS, resuspended in 100 μl of FACS buffer (0.3% BSA and 0.1% NaN3 in PBS), then incubated with 1 μl of Fc block (anti-CD16/32; PharMingen) for 5 min followed by 1 hr in primary antibody [fluoroscein isothiocyanate-conjugated anti-CD41 mAb (PharMingen; 1:250 dilution); or antigen-affinity purified rabbit polyclonal anti-TVA antibody (1:1,000)], washed three times in FACS buffer, and then resuspended in 100 μl (anti-TVA) or 500 μl (anti-CD41) of FACS buffer. For TVA detection, cells were incubated for 45 min with a Fab fragment of an anti-rabbit-phycoerythrin secondary antibody (Boehringer Mannheim; 1:250 dilution) followed by three washes in FACS buffer and resuspension in 500 μl of FACS buffer.
In Vitro Infections.
In vitro bone marrow infections were performed with 0.5–1.0 × 106 cells in 250 μl of growth medium in the well of a 12-well dish and 250 μl of virus stock with 8 μg/ml polybrene. The cells were incubated at 37°C for 3 hr with rocking every 20–30 min and then diluted with 2.5 ml of growth medium.
In Vivo Infections.
Two- to three-month-old transgenic mice and their nontransgenic littermates received 50 ng of TPO via i.p. injection on days 1, 2, 3, and 4. Infectious particles (0.25–0.5 × 106) of a β-gal-expressing virus coated with avian subgroup A envelope protein were injected i.p. in 250 μl on days 3 and 4. Mice were killed on day 5 and fresh bone marrow was analyzed.
RESULTS
Generation of transgenic mice expressing TVA. Of eight pups born from fertilized oocytes injected with the GP-Ibα-TVA-950 expression cassette (Fig. 1A), one mouse (no. 9562) contained the tv-a transgene as evidenced by Southern blot analysis. RT-PCR of bone marrow RNA from transgenic and nontransgenic 9562 offspring demonstrated a 420-bp fragment indicative of tv-a mRNA only in mice carrying the transgene (Fig. 1B, lanes 1 and 2 vs. lanes 3 and 4). The 420-bp fragment is not caused by DNA contaminating the RNA preparation because the 420-bp band is absent when reverse transcriptase is not included in the RT-PCR cDNA synthesis reaction (Fig. 1B, lane 1 vs. lane 2). Peripheral blood counts from 8- to 10-month-old transgenic mice and their nontransgenic littermates show no gross differences (data not shown), and transgenic mice followed up to 1 year have no overt health problems.
ALV Receptor Protein Is Selectively Expressed in Murine Hematopoietic Tissue.
Hematoxylin/eosin staining of decalcified femur sections demonstrates normal murine bone marrow (Fig. 2A). TVA expression is readily seen on megakaryocytes in the femur sections from transgenic mice (Fig. 2B), whereas no TVA expression was seen in nontransgenic littermates (Fig. 2C). TVA was not detected in mature red and white blood cells or their readily identifiable precursors, but we consistently found limited numbers of small bone marrow cells expressing low levels of TVA (Fig. 2B). TVA expression is also readily apparent on the megakaryocytes in the splenic red pulp, a normal site for murine hematopoiesis, but not on the lymphoid cells in or around the follicles (Fig. 2 D and E). TVA expression was not detected in splenic tissue from nontransgenic littermates (data not shown). Immunohistochemical staining of liver, small intestine, heart, and kidney from the transgenic mice failed to detect TVA protein (data not shown).
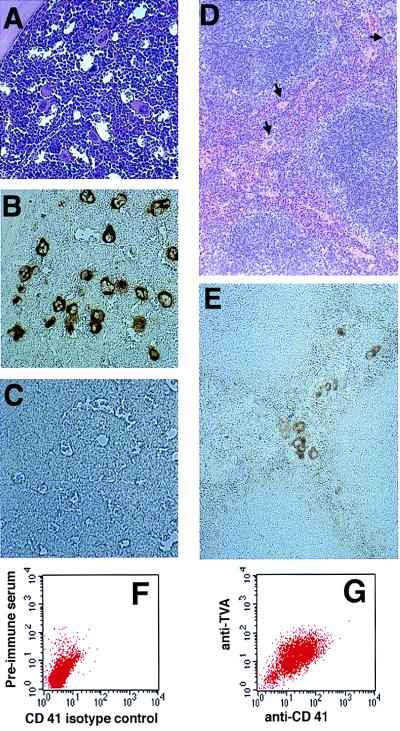
Figure 2.
ALV receptor protein is expressed in murine hematopoietic tissue. Hematoxylin/eosin stains of bone marrow (A) and spleen (D) from a TVA-expressing transgenic mouse show grossly normal tissue. Immunohistochemical staining using an anti-TVA antibody demonstrates TVA expression on megakaryocytes from transgenic mice (B) but not from nontransgenic littermates (C). TVA expression is also seen in splenic megakaryocytes from transgenic mice (E). Independent FACS analysis using isotype controls for anti-CD41 and anti-TVA plotted against forward scatter was used to identify a generous gate that encompasses all cells stained by either antibody. Two-dimensional FACS analysis using isotype controls for anti-CD41 and anti-TVA demonstrates cells congregated in the left lower quadrant of the FACS dot plot (F), with a shift of the population up and to the right after incubation with anti-TVA and anti-CD41 antibodies (G).
GP-IIb (αIIb integrin) is a specific marker for megakaryocyte lineage cells and platelets (24, 25). FACS analysis for coexpression of GP-IIb (CD41) and TVA on fresh bone marrow cells grown 3 days in liquid culture demonstrated that ≈20% of the cells double stained for CD41 and TVA, 45% stained only for CD41, and 35% stained for neither (Fig. 2 F and G). We could not convincingly detect TVA expression in the absence of CD41 staining. The combined immunohistochemical and FACS data provide strong evidence that TVA expression is limited to megakaryocyte lineage cells.
TVA-Expressing Megakaryocyte-Lineage Cells Can Be Serially Infected with Avian Retroviruses.
Fresh bone marrow cells were infected with RCAS-PURO to determine whether TVA expression conferred susceptibility to subgroup A, avian retroviruses. After 7 days in puromycin selection, uninfected cells and infected cells from nontransgenic littermates had all died, but infected bone marrow cells from transgenic animals were proliferating. As the sequential introduction of genes of interest would be useful in studies of megakaryocyte development and biology, the puromycin-resistant cells were infected with RCAS-AP, an avian retrovirus that expresses HPAP. HPAP staining 48 hr later demonstrated that nearly all of the puromycin-resistant cells express HPAP (Fig. 3A), whereas mock-infected puromycin-resistant cells showed no HPAP staining (Fig. 3B). The HPAP-expressing cells were therefore sequentially infected by two avian retroviruses, RCAS-PURO and RCAS-AP, and expressed functional gene products derived from each virus.
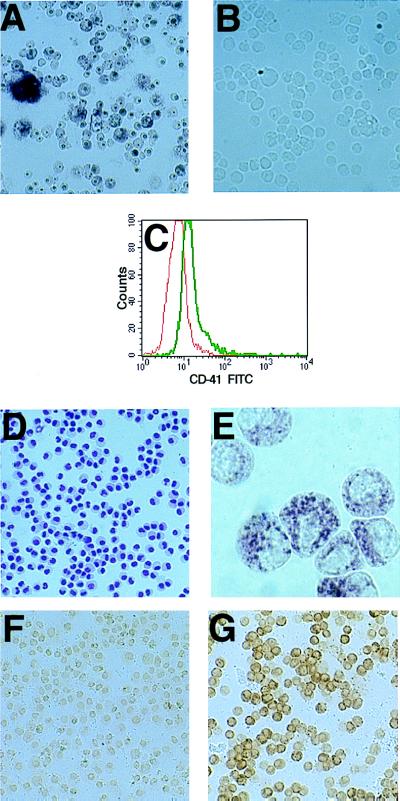
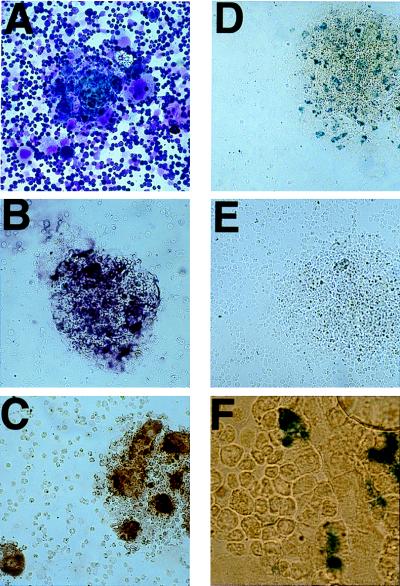
Figure 3.
TVA-expressing megakaryocyte-lineage cells can be serially infected with avian retroviruses. Bone marrow cells from TVA-expressing mice were cultured in the presence of IL-3, SCF, and TPO, infected with RCAS-PURO, and then subjected to puromycin selection. HPAP staining 48 hr after the puromycin-resistant cells were infected with RCAS-AP demonstrates that essentially all cells express HPAP (A), whereas no HPAP expression is seen in the mock-infected puromycin-resistant cells (B). FACS analysis of the puromycin-resistant cells using anti-CD41 (green) or an isotype control (red), and gating on live cells by use of propidium iodide staining, demonstrates that essentially the entire cell population expresses CD41. Wright-Giemsa (D) and immunohistochemical stains for vWF (E), P-selectin (F), and TVA (G) show the CD41-expressing, puromycin-resistant cells to be quite homogeneous.
FACS analysis revealed that the entire puromycin-resistant population expresses CD41, the defining marker for all megakaryocyte-lineage cells (Fig. 3C). The cells grow in culture for 30–35 days and then senesce. During this time, they remain very homogeneous in appearance with a folded nucleus and a moderate nuclear-cytoplasmic ratio (Fig. 3D). Only a rare cell develops into a mature megakaryocyte. In addition to CD41, the cells uniformly express vWF (Fig. 3E), a protein known to be expressed in megakaryocyte lineage cells and not in cells of other hematopoietic lineages. The puromycin-resistant cells do not express P-selectin (CD62; Fig. 3F) or acetylcholinesterase (data not shown), two marks of late megakaryocyte-lineage maturation. As expected, the cells all express the TVA transgene (Fig. 3G). The phenotype of the puromycin-resistant cells provides additional evidence that TVA expression is limited to megakaryocyte-lineage cells, and it demonstrates that puromycin selection after RCAS-PURO infection is an excellent method for obtaining a large, pure population of phenotypically immature cells committed to the megakaryocyte lineage.
In vitro infectivity studies were first performed by using bone marrow cells grown in the presence of SCF, TPO, and IL-3, but we subsequently saw far more mature megakaryocytes when IL-3 was not in the culture media (Fig. 4 A and B). When bone marrow cells are cultured for 5 days, infected with RCAS-AP, and stained 48 hr later for HPAP expression, small TVA-expressing cells from early in the megakaryocyte lineage are the predominant cell infected in the presence of IL-3 (Fig. 4C). In contrast, HPAP-expressing, mature megakaryocytes are readily seen when IL-3 is not in the culture media (Fig. 4D). The type of megakaryocyte lineage cell infected reflects IL-3’s effect on expanding the number of megakaryocyte-lineage cells while simultaneously inhibiting maturation along the megakaryocyte developmental pathway (26–29).
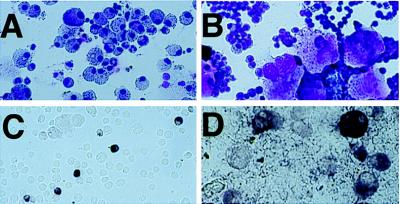
Figure 4.
IL-3 promotes infection of early megakaryocyte-lineage cells. Bone marrow cells from transgenic mice were grown in media supplemented with SCF, TPO, and IL-3 (A and C) or with SCF and TPO (B and D). Cells were infected with RCAS-AP after 5 days in culture and stained for HPAP expression 48 hr after infection. Wright-Giemsa stains demonstrate numerous mature megakaryocytes when IL-3 is not in the growth media (B), whereas mature megakaryocytes are rare when IL-3 is present (A). Likewise, infected, HPAP-expressing mature megakaryocytes are readily apparent only when IL-3 is not in the growth media (C and D).
Retroviral Infection of ALV Receptor-Expressing Cells in Live Animals.
TVA-expressing transgenic mice and nontransgenic littermates were killed 2 days after receiving serial i.p. injections of TPO and infectious particles encoding β-gal. Freshly harvested bone marrow appeared normal by Wright-Giemsa stain (Fig. 5A). At the cell concentrations used in the cyto-spin, the megakaryocyte-lineage cells tended to clump (Fig. 5 A–C). Immunohistochemical staining of fresh samples suggests colocalization of CD41 (Fig. 5B) and TVA (Fig. 5C) in the cells from the TVA-expressing transgenic mice. TVA was not detected in the nontransgenic littermates (data not shown).
Figure 5.
Retroviral infection of TVA-expressing cells in live animals. After i.p. injection of 0.25–0.5 × 106 env-A coated, β-gal-expressing infectious particles on each of 2 consecutive days, mice were killed on the next day. Wright-Giemsa staining (A) of freshly harvested bone marrow demonstrated the megakaryocytes to be mostly in clumps. Cells that stained positive for CD41 (B) and for TVA (C) were predominantly located in the megakaryocyte-containing clumps. β-gal staining demonstrated positive cells limited to the small cells within the megakaryocyte-containing clumps (D). No staining was seen in bone marrow from nontransgenic control mice subjected to the identical infections (E). A region with preserved bone marrow architecture from a transgenic mouse (F) demonstrates β-gal expressing cells adjacent to a bone marrow blood vessel.
β-gal staining of the marrow from the infected mice produced blue cells in the megakaryocyte clumps from the transgenic mice (Fig. 5D), but blue cells were not seen after β-gal staining of infected bone marrow taken from nontransgenic littermate controls (Fig. 5E). The majority of the β-gal expressing cells are the small CD41-positive cells (Fig. 5 B and D), the same cells that predominate when in vitro infections are performed in the presence of IL-3. These findings further suggest that the small TVA-positive cells seen in bone marrow sections are of megakaryocyte lineage. Interestingly, β-gal expressing cells were found immediately adjacent to a bone marrow blood vessel that survived the harvest and cytospin preparation (Fig. 5F).
DISCUSSION
In this paper we describe a transgenic mouse model for the study of megakaryocytopoiesis and megakaryocyte biology. The model uses a human GP-Ibα 5′-regulatory sequence to achieve TVA expression restricted to megakaryocyte lineage cells. The TVA-expressing cells can be selectively infected by avian retroviruses in vitro and in vivo. Sequential infections can be performed to introduce multiple genes of interest into the TVA-expressing cells, making these mice a powerful tool for studying megakaryocyte function and development. We have used these mice to generate a pure population of primary CD41-positive megakaryocyte progenitors, and to demonstrate that the onset of vWF expression occurs before the onset of P-selectin and acetylcholinesterase expression during megakaryocytopoiesis.
TVA Expression Is Restricted to Cells of the Megakaryocyte Lineage.
Immunohistochemical staining of bone marrow, spleen, and other tissues for TVA expression (Fig. 2), bone marrow FACS analysis for the coexpression of TVA and CD41 (Fig. 2), and the phenotype of cells infected with avian retroviruses (Figs. 3 and 5) all support the conclusion that TVA expression is limited to megakaryocyte-lineage cells in hematopoietic tissue. In addition, we have not detected TVA expression on mature cells of the white or red blood cell series, and we see no TVA expression in the splenic lymphoid tissue. Furthermore, after infection of total bone marrow with RCAS-PURO, all uromycin-resistant cells are CD41- and vWF-positive (Fig. 3 C and D), a powerful argument for TVA expression being limited to megakaryocyte lineage cells. Although other areas of TVA expression below the detection limits of our anti-TVA antibody cannot be formally excluded, the retrovirus infection experiments demonstrate that megakaryocyte-lineage cells from the transgenic mice can be selectively infected in vitro and in vivo by using avian retroviruses.
TVA-Expressing Cells Are Susceptible to Avian Retroviral Infection in Vitro and in Vivo.
Small megakaryocyte precursors, not mature megakaryocytes, are the predominant target cell when the transgenic mouse bone marrow is infected in vivo. Because mature megakaryocytes have more surface TVA than the smaller, less mature megakaryocyte-lineage cells (Fig. 2B), greater numbers of surface TVA molecules do not appear to provide an advantage for efficient infection. In fact, some infections may occur in cells with TVA surface expression below the detection limits of our antibody (Fig. 5 C and D). Nuclear membrane breakdown occurs during megakaryocyte endomitosis precluding an intact nuclear membrane as a barrier to efficient infection of mature, polyploid megakaryocytes (30). The apparent preference for early megakaryocyte-lineage cells in our in vivo experiments could simply reflect some combination of the relative number of megakaryocyte precursors and megakaryocytes, the degree of endomitosis occurring at the time of infection, and the short interval between infection and analysis. Efforts are underway to determine the percentage of megakaryocyte-lineage cells we can infect in vivo. Attempts using virus expressing the green fluorescent protein have been hindered by the extensive endogenous fluorescence of fresh murine bone marrow.
Unlike in vivo infections, we can readily control the cytokine milieu influencing hematopoietic cells in vitro. We observed that IL-3, a stimulatory cytokine of multilineage hematopoietic progenitor cells, inhibits the development of mature megakaryocytes and promotes the infection of small early megakaryocyte-lineage cells (Fig. 4). Conversely, when bone marrow is cultured and infected in the absence of IL-3, large mature megakaryocytes are readily seen and a significant number are productively infected (Fig. 4). These findings are compatible with earlier work demonstrating that IL-3 expands the number of megakaryocyte-lineage cells as it impedes progress along the megakaryocyte developmental pathway (26–29). We are pursuing experiments to determine whether mature polyploid megakaryocytes can be directly infected with simple retroviruses, or whether the infected megakaryocytes matured from infected early megakaryocyte-lineage cells.
Bone marrow analysis of the IP-infected mice did not demonstrate an obvious increase in megakaryocytes, and subsequent dose response experiments (data not shown) demonstrated that 50 ng of TPO daily for 5 days fails to increase platelet counts or noticeably increase bone marrow megakaryocytes. The administered TPO was therefore unlikely to have promoted viral infectivity in the experiments presented (Fig. 5). We are currently optimizing in vivo infection efficiency by modifying our use of hematopoietic cytokines, changing the route (i.p. vs. i.v.) and the number of viral particles used per infection, and testing the effect of performing infections during the myelopoietic “rebound” after 5-fluorouracil administration.
The TVA-expressing mice provide an excellent system for obtaining a large pure population of primary megakaryocyte precursors that are committed to the megakaryocyte developmental pathway. The mice can be crossed with mice carrying specific genetic defects, i.e., MPL−/− or NF-E2−/− mice, thereby providing a null genetic background in which to study the role of specific gene products. Selected gene products can then be introduced into megakaryocyte-lineage cells by using avian retroviruses, providing a powerful means to study the effect of specific mutations on megakaryocyte-lineage cell development and function. Furthermore, given the critical yet poorly understood role of the bone marrow microenvironment in hematopoietic development, hypotheses derived from experiments using cultured cells can be tested in the context of the whole animal by way of in vivo infections. To extend the usefulness of this system, we are currently developing new transgenic mice that express TVA under the control of 5′-regulatory sequences from PF4, GP-IIb, GP-IX, and MPL. Unlike prior transgenic mice with lineage-restricted TVA expression, we have expressed TVA on cells that normally continue to divide throughout life. This feature provides a distinct advantage when using simple retroviruses to introduce genetic material in vivo and in vitro, and the general model used should be applicable to other hematopoietic lineages.
Acknowledgments
We thank Dan R. Littman (New York University, New York) for unyielding support that allowed the development of this project; John Scarborough (Oregon Health Sciences University, Portland) for performing the oocyte injections and for his patience in teaching us how to work with mice; Jerry Ware (Scripps Research Institute, La Jolla, CA), Steve Hughes (National Cancer Institute, Frederick, MD), Eric Holland (National Institutes of Health, Bethesda, MD), and Paul Bates (University of Pennsylvania, Philadelphia) for plasmids; Kurt Ziegler (University of California, San Francisco) for DF1 cells; Paul Bates and John Young (Harvard Medical School, Cambridge, MA) for rabbit polyclonal anti-TVA antisera; and Thanh K. Tran (University of California, San Francisco) for antigen-affinity purification of the anti-TVA antisera. This work was supported by National Institutes of Health Transfusion Medicine Specialized Centers of Research Grant P50 HL54476.
ABBREVIATIONS
- ALV
avian leukosis virus
- ALV-A
subgroup A ALV
- TVA
ALV-A receptor
- β-gal
β-galactosidase
- FACS
fluorescence-activated cell sorter
- HPAP
human placental alkaline phosphatase
- RT
reverse transcription
- TPO
thrombopoietin
- vWF
von Willebrand factor
- SCF
stem cell factor
References
- 1.Levine R F. Br J Haematol. 1980;45:487–497. doi: 10.1111/j.1365-2141.1980.tb07168.x. [DOI] [PubMed] [Google Scholar]
- 2.Hoffman R. Blood. 1989;74:1196–1212. [PubMed] [Google Scholar]
- 3.Miller A D. In: Development and Applications of Retroviral Vectors. Coffin J, Hughes S H, Varmus H E, editors. Plainview, NY: Cold Spring Harbor Lab. Press; 1997. pp. 437–474. [PubMed] [Google Scholar]
- 4.Bates P, Young J A, Varmus H E. Cell. 1993;74:1043–1051. doi: 10.1016/0092-8674(93)90726-7. [DOI] [PubMed] [Google Scholar]
- 5.Young J A, Bates P, Varmus H E. J Virol. 1993;67:1811–1816. doi: 10.1128/jvi.67.4.1811-1816.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Federspiel M J, Bates P, Young J A, Varmus H E, Hughes S H. Proc Natl Acad Sci USA. 1994;91:11241–11245. doi: 10.1073/pnas.91.23.11241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Holland E C, Varmus H E. Proc Natl Acad Sci USA. 1998;95:1218–1223. doi: 10.1073/pnas.95.3.1218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cui Z, Reilly M P, Surrey S, Schwartz E, McKenzie S E. Blood. 1998;91:2326–2333. [PubMed] [Google Scholar]
- 9.Ravid K, Li Y C, Rayburn H B, Rosenberg R D. J Cell Biol. 1993;123:1545–1553. doi: 10.1083/jcb.123.6.1545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ravid K, Beeler D L, Rabin M S, Ruley H E, Rosenberg R D. Proc Natl Acad Sci USA. 1991;88:1521–1525. doi: 10.1073/pnas.88.4.1521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ware J, Russell S R, Marchese P, Ruggeri Z M. J Biol Chem. 1993;268:8376–8382. [PubMed] [Google Scholar]
- 12.Ware J, Hashimoto Y, Zieger B, Russell S. C R Acad Sci III. 1996;319:811–817. [PubMed] [Google Scholar]
- 13.Quintrell N, Hughes S H, Varmus H E, Bishop J M. J Mol Biol. 1980;143:363–393. doi: 10.1016/0022-2836(80)90218-1. [DOI] [PubMed] [Google Scholar]
- 14.Hashimoto Y, Ware J. J Biol Chem. 1995;270:24532–24539. doi: 10.1074/jbc.270.41.24532. [DOI] [PubMed] [Google Scholar]
- 15. Soneoka Y, Cannon P M, Ramsdale E E, Griffiths J C, Romano G, Kingsman S M, Kingsman A J. Nucleic Acids Res. 1995;23:628–633. doi: 10.1093/nar/23.4.628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hughes S H, Greenhouse J J, Petropoulos C J, Sutrave P. J Virol. 1987;61:3004–3012. doi: 10.1128/jvi.61.10.3004-3012.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Greenhouse J J, Petropoulos C J, Crittenden L B, Hughes S H. J Virol. 1988;62:4809–4812. doi: 10.1128/jvi.62.12.4809-4812.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Federspiel M J, Hughes S H. Methods Cell Biol. 1997;52:179–214. [PubMed] [Google Scholar]
- 19.Hogan B, Costantini F, Lacy E. Manipulating the Mouse Embryo: A Laboratory Manual. Plainview, NY: Cold Spring Harbor Lab. Press; 1986. [Google Scholar]
- 20.Gaur M, Leavitt A D. J Virol. 1998;72:4678–4685. doi: 10.1128/jvi.72.6.4678-4685.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Norton P A, Coffin J M. Mol Cell Biol. 1985;5:281–290. doi: 10.1128/mcb.5.2.281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bates P, Malim M H. Methods Mol Biol. 1997;63:41–48. doi: 10.1385/0-89603-481-X:41. [DOI] [PubMed] [Google Scholar]
- 23.Coligan J E, Krujsbeek A M, Margulies D H, Shevach E M, Strober W, editors. Current Protocols in Immunology. New York: Wiley; 1995. [Google Scholar]
- 24.Lemarchandel V, Ghysdael J, Mignotte V, Rahuel C, Romeo P H. Mol Cell Biol. 1993;13:668–676. doi: 10.1128/mcb.13.1.668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Uzan G, Prenant M, Prandini M H, Martin F, Marguerie G. J Biol Chem. 1991;266:8932–8939. [PubMed] [Google Scholar]
- 26.Segal G M, Stueve T, Adamson J W. J Cell Physiol. 1988;137:537–544. doi: 10.1002/jcp.1041370320. [DOI] [PubMed] [Google Scholar]
- 27.Mazur E M, Cohen J L, Bogart L, Mufson R A, Gesner T G, Yang Y C, Clark S C. J Cell Physiol. 1988;136:439–446. doi: 10.1002/jcp.1041360307. [DOI] [PubMed] [Google Scholar]
- 28.Dolzhanskiy A, Basch R S, Karpatkin S. Blood. 1997;89:426–434. [PubMed] [Google Scholar]
- 29.Dolzhanskiy A, Hirst J, Basch R S, Karpatkin S. Br J Haematol. 1998;100:415–426. doi: 10.1046/j.1365-2141.1998.00579.x. [DOI] [PubMed] [Google Scholar]
- 30.Vitrat N, Cohen-Solal K, Pique C, Le Couedic J P, Norol F, Larsen A K, Katz A, Vainchenker W, Debili N. Blood. 1998;91:3711–3723. [PubMed] [Google Scholar]