Abstract
An unresolved question in the study of the polyglutamine neurodegenerative disorders is the extent to which partial loss of normal function of the mutant protein contributes to the disease phenotype. To address this, we studied Kennedy disease, a degenerative disorder of lower motor neurons caused by a CAG/glutamine expansion in the androgen receptor (Ar) gene. Signs of partial androgen insensitivity, including testicular atrophy and decreased fertility, are common in affected males, although the underlying mechanisms are not well understood. Here, we describe a knock-in mouse model that reproduces the testicular atrophy, diminished fertility, and systemic signs of partial androgen insensitivity that occur in Kennedy disease patients. Using this model, we demonstrate that the testicular pathology in this disorder is distinct from that mediated by loss of AR function. Testes pathology in 113 CAG knock-in mice was characterized by morphological abnormalities of germ cell maturation, decreased solubility of the mutant AR protein, and alterations of the Sertoli cell cytoskeleton, changes that are distinct from those produced by AR loss-of-function mutation in testicular feminization mutant mice. Our data demonstrate that toxic effects of the mutant protein mediate aspects of the Kennedy disease phenotype previously attributed to a loss of AR function.
Kennedy disease is one of nine neurodegenerative disorders caused by expansions of CAG/glutamine (Q) tracts in the coding regions of otherwise unrelated genes.1 The mutation in Kennedy disease occurs in the first exon of the androgen receptor (Ar) gene2 and causes hormone-dependent dysfunction and degeneration of lower motor neurons in males.3–5 The mutant AR protein misfolds and aggregates and, like other proteins with expanded glutamine tracts, may exert toxic effects by disrupting the ubiquitin-proteasome pathway and inducing a stress response, by altering the activity of transcriptional co-regulators, or by impairing synaptic function and axonal transport.6–10 These shared features likely reflect common mechanisms underlying the toxicity of these mutant proteins.
It has been suggested that a partial loss of the normal function of the protein may also contribute to the pathogenesis of several CAG repeat disorders, including Kennedy and Huntington diseases.11,12 In Kennedy disease, in which the normal function of the disease-causing protein is well understood, patients may exhibit signs of partial androgen insensitivity, including testicular atrophy and decreased fertility.13 These clinical signs correlate with cell culture models demonstrating a partial loss of AR function that is mediated by the expanded glutamine tract and is made manifest by decreased expression of androgen-responsive genes or reporter constructs.14–17 However, it has not been established whether the features of partial androgen insensitivity in Kennedy disease are in fact due to loss of AR function in vivo or whether other mechanisms are responsible for these clinical manifestations.
To explore the pathogenesis of Kennedy disease, we developed a knock-in mouse model in which 113 CAG repeats are targeted to the first exon of the mouse Ar gene (AR113Q mice). Mutant male mice develop hormone-dependent neuromuscular pathology, which will be described elsewhere. Here, we demonstrate that these mice also model the testicular atrophy and decreased fertility that commonly occur in Kennedy disease patients. Surprisingly, we found that the testicular pathology in AR113Q mice is distinct from that which occurs in testicular feminization mutant (tfm) mice, with a loss-of-function mutation in the Ar gene, and is characterized by marked morphological abnormalities of germ cell maturation and alterations in the Sertoli cell cytoskeleton. These observations indicate that features of Kennedy disease previously attributed to a partial loss of AR function are in actuality mediated by toxicity of the mutant protein.
Materials and Methods
Generation of Ar Knock-In Mice
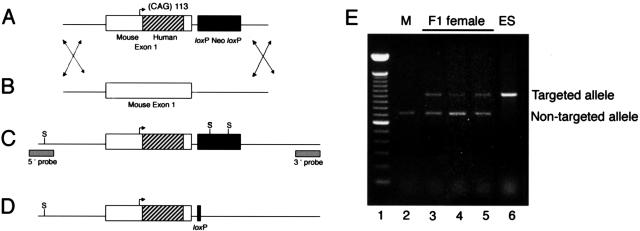
A targeting vector containing 6 kb of the mouse Ar locus was constructed using the backbone targeting vector pLP118 and genomic sequence isolated from a J1 mouse embryonic stem cell library constructed in phage EMBL3. Ar exon 1 was immediately followed by the phosphoglycerol kinase promoter driving expression of the neomycin resistance gene. This cassette, flanked by loxP sites, was in the opposite orientation of the Ar gene. A portion of the coding region of mouse exon 1 from the SmaI to BstEII sites (Pro37 to Gly423) was exchanged for the same region in human Ar exon 1. During this exchange, CAG repeats of 21, 41, or 113 were introduced. The obtained targeting plasmids were checked by sequencing across the CAG repeat and cloning sites and were electroporated into CJ7 ES cells derived from 129S1/SvlmJ mice.19 Homologous recombination was confirmed by Southern blot using 5′ and 3′ probes that fell outside the targeting construct and that detected an SphI restriction polymorphism introduced during targeted insertion of the neomycin resistance cassette. Cre recombinase was then transiently expressed in correctly targeted ES cell clones using pMC-Cre,20 and subclones were identified in which the neomycin cassette was successfully removed. Chimeric male mice derived from injection of these clones were crossed with C57Bl/6J females. F1 females with the targeted allele were identified by polymerase chain reaction (PCR) analysis using primers that span the CAG repeat. F1 heterozygous females were crossed with C57Bl/6J males to derive the F2 animals used in these studies. All procedures involving mice were approved by the University of Michigan Committee on Use and Care of Animals, in accordance with the NIH Guidelines for the Care and Use of Experimental Animals.
Immunohistochemistry and Immunofluorescence
Tissue was fixed in formalin, embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin. For immunohistochemistry, antigen retrieval was achieved by boiling in glycine buffer (pH 3.5) for 10 minutes. Tissue sections were stained with polyclonal antibodies against AR or β-tubulin (Santa Cruz Biotechnology, Santa Cruz, CA). Proteins were visualized using the Vectastain ABC kit (Vector Laboratories, Burlingame, CA) or with an Alexa Fluor 594 secondary antibody (Molecular Probes, Eugene, OR). Light images were captured using an Olympus BX41 microscope, 40× lens, and Insight digital camera. Confocal images were captured using a Zeiss LSM 510 microscope and a 63× water immersion lens.
Protein Expression Analysis
Tissue homogenates were prepared in radioimmunoprecipitation assay buffer containing Complete Protease Inhibitor (Roche, Indianapolis, IN) using a PowerGen 125 (Fisher, Pittsburgh, PA). After homogenization, samples were spun at 15,000 × g for 15 minutes, and both supernatants and pellets were analyzed by 7% sodium dodecyl sulfate-polyacrylamide gel electrophoresis. Proteins were transferred to Immunobilon-P membranes using a semidry transfer apparatus and detected by chemiluminescence. Blots were probed with polyclonal anti-AR antibody N-20 (Santa Cruz) or monoclonal anti-polyglutamine antibody 1C2 (Chemicon, Temecula, CA). Major urinary proteins (MUPs) in urine were detected by electrophoresis of 1 μl of urine on 10% SDS-PAGE followed by Coomassie blue staining.
Gene Expression Analysis
Total RNA was isolated from tissues with Trizol (Invitrogen, Carlsbad, CA) and served as a template for cDNA synthesis using the High Capacity cDNA Archive kit from Applied Biosystems (Foster City, CA). Gene-specific primers and probes labeled with the fluorescent reporter dye 6-carboxyfluorescein and the quencher 6-carboxytetramethyl-rhodamine were purchased from Applied Biosystems. TaqMan assays were performed using 5-ng aliquots of cDNA. Replicate tubes were analyzed for the expression of 18S rRNA using a VIC-labeled probe. Threshold cycle values were determined by an ABI Prism 7900HT sequence detection system, and relative expression levels were calculated using the standard curve method of analysis.
Measurement of Serum Hormone Levels
Serum testosterone levels were determined by radioimmunoassay (Diagnostic Systems, Webster, TX). The reference preparation of testosterone, obtained from Steraloids, Inc., was purified by high-performance liquid chromatography and verified by thin layer chromatography and immunoassay. The testosterone radioimmunoassay had a sensitivity of 0.05 ng/ml. Luteinizing hormone (LH) and follicle-stimulating hormone (FSH) levels were determined by sandwich assay and radioimmunoassay, respectively, performed by the University of Virginia Center for Research in Reproduction Ligand Assay and Analysis Core Facility. The mouse LH and FSH reference preparations were provided by Dr. A.F. Parlow and the National Hormone and Peptide Program. The LH assay had a sensitivity of 0.07 ng/ml, and the FSH assay had a sensitivity of 2.0 ng/ml.
Sperm Count
The epididymides were removed, minced in 4 ml of phosphate buffered saline (pH 7.4), and gently rocked at room temperature for 10 minutes. After centrifugation at 500 × g for 1 minute, sperm count was determined using a hemacytometer.
Terminal Deoxynucleotide Transferase End Labeling (TUNEL) Assay
The presence of TUNEL-positive cells was determined using formalin-fixed, paraffin-embedded sections of testes and the ApopTag Peroxidase In Situ Apoptosis Detection kit (Chemicon). The number of labeled cells in five randomly selected 200× high-power fields was determined for each slide.
Results
Generation of Ar 113 CAG Knock-In Mice
AR113Q knock-in mice were created by recombining a SmaI to BstEII fragment of human Ar exon 1 containing 113 CAG repeats with the mouse Ar gene (Figure 1, A–D). Chimeric mice generated by injection of correctly targeted ES cells into C57BL/6J blastocysts were mated to C57BL/6J females. Germline transmission of the targeted allele to female agouti pups was confirmed by PCR (Figure 1E). These mice were crossed with C57BL/6J males and yielded the expected proportion of male and female mice with the anticipated transmission of the targeted Ar allele (data not shown). Because the Ar gene is on the X chromosome, male mice hemizygous for either the wild-type or mutant allele were generated from these matings. The phenotype reported here is based on the characterization of F2 male mice derived from two indistinguishable, independent lines of 113 CAG repeat Ar knock-in mice.
Figure 1.
Generation of an expanded CAG allele at the androgen receptor locus. A: Schematic representation of targeting construct. Arrow indicates initiation methionine. The insertion of a SmaI to BstEII fragment of human exon 1 is indicated by stripes. B: Endogenous androgen receptor (Ar) allele. C: Predicted structure of the mutant CAG expansion allele generated by homologous recombination. S, SphI sites. Location of probes used for Southern blot analysis is indicated. D: Representation of targeted mouse allele after a Cre-mediated excision event. E: Detection of targeted and non-targeted Ar alleles in F1 female mice. PCR primers that span the CAG repeat were used to amplify sequences from a wild-type C57BL/6 male (lane 2), three F1 agouti females (lanes 3 to 5), and the ES cell clone used to generate the chimeric male that sired the F1 females (lane 6). Lane 1 = 100-bp ladder.
AR113Q Males Display Partial Androgen Insensitivity
In our initial effort to assess the effectiveness of the expanded glutamine AR to masculinize AR113Q males, we examined the appearance of the external genitalia. No mice with ambiguous genitalia were identified, and a strict genotype-phenotype correlation was observed when sex was confirmed by PCR detection of the Sry gene (data not shown). Similarly, gross and microscopic inspection revealed an absence of mammary tissue in mutant males, an expected outcome because this tissue is known to undergo perinatal, androgen-dependent regression in male mice. These data confirmed that the expanded glutamine AR functioned sufficiently well to direct appropriate development of certain male secondary sex characteristics.
Despite these findings, our attempts to breed mutant males were largely unsuccessful. Multiple attempted matings yielded only two litters, sired by two AR113Q males younger than 10 weeks of age mated with wild-type females. Additional attempted breeding of these two males and of five other AR113Q males with either wild-type (for up to 2 months) or AR113Q females (for 2 to 6 months) failed to yield any litters. Because Kennedy disease patients often exhibit decreased fertility and testicular atrophy, we sought to further explore these systemic effects of the mutant allele. As an additional control, we used knock-in mice generated with the identical targeting construct except for the inclusion of only 21 or 41 CAG repeats (AR21Q or AR41Q, respectively). A CAG repeat of 21 in the Ar gene is in the normal range in humans, whereas a repeat of 41 is at the lower end of the pathogenic range for Kennedy disease. We found that a glutamine tract of either 21 or 41 amino acids in the AR protein had no detectable pathogenic effects in mice. Male AR21Q and AR41Q mice bred as vigorously as wild-type males, had normal weight testes (Figure 2A; data not shown), and had normal levels of circulating testosterone (Figure 2B). These mice also confirmed that recombination of 1050 nucleotides of human Ar exon 1 into the mouse gene, encoding a region of approximately 85% amino acid identity, resulted in no detectable physiological effects. In contrast, testes of AR113Q mice were markedly atrophic, weighing only about one-half that of wild-type testes (Figure 2A). Despite this testicular atrophy, AR113Q males had a similar range of serum testosterone values compared with wild-type males, although the mean value was slightly elevated (Figure 2B). Similarly elevated serum testosterone levels have been observed in some patients with Kennedy disease.21
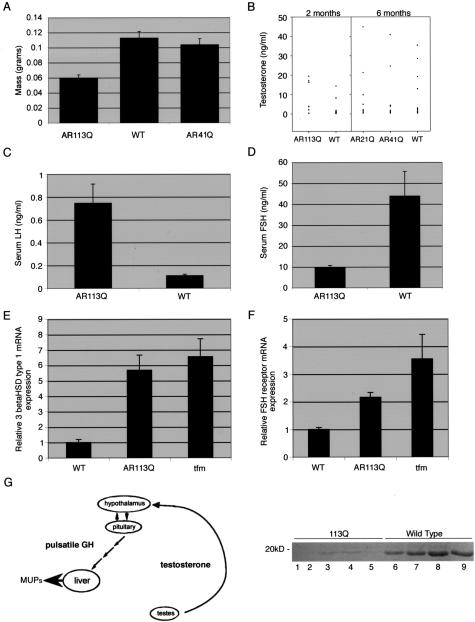
Figure 2.
Partial androgen insensitivity in AR113Q knock-in mice. A: Testes of AR113Q knock-in males (n = 7) were significantly smaller than those of wild-type littermates (n = 10) (P < 0.0001 by unpaired Student’s t-test) or AR41Q knock-in males (n = 4) (P < 0.0002). There was no significant difference in testes weight between AR41Q knock-in and wild-type males (P = 0.4). B: Circulating testosterone levels in AR113Q males (n = 7) and wild-type littermates (n = 12) fell in a similar range, although the mean value for AR113Q males was slightly elevated (P < 0.035). No significant difference in testosterone levels was observed among AR21Q (n = 11), AR41Q males (n = 12), and wild-type littermates (n = 15) (P > 0.2). In C, serum LH levels were significantly higher in AR113Q males (n = 6) than in wild-type controls (n = 7) (P < 0.0005), whereas in D, serum FSH levels were significantly lower in AR113Q males (n = 6) than in wild-type controls (n = 8) (P < 0.005). E and F: Relative mRNA expression levels of 3βHSD type 1 (E) and FSH receptor (F) in testes of wild-type (n = 5), AR113Q (n = 3 for E; n = 7 for F), and tfm males (n = 4) at 3 to 4 months of age. Data shown are means ± SD relative to the expression in wild-type mice. G: Diagram of testosterone’s indirect regulation of MUPs production in the liver mediated through action on the hypothalamic-pituitary axis (modified from 23) (left panel). Lower levels of urinary MUPs were detected in AR113Q knock-in males (lanes 1 to 5) than in wild-type littermates (lanes 6 to 9) (right panel).
We next sought to determine whether AR113Q males exhibited systemic effects that modeled the partial loss of AR function that is manifest in Kennedy disease patients. To accomplish this, we examined several parameters that reflect androgen function. AR113Q males had significantly higher levels of serum LH than did wild-type mice (Figure 2C). LH binds receptors on Leydig cells to stimulate testosterone synthesis and secretion, and elevated LH levels in AR113Q mice are consistent with a partial loss of AR function. In support of this interpretation, 3β-hydroxysteroid dehydrogenase type 1 (3βHSD type 1), a Leydig cell transcript encoding a regulator of steroidogenesis,22 was expressed at higher levels in AR113Q males with morphologically abnormal testes and in tfm males compared with wild-type controls (Figure 2E). Although this increase in 3βHSD type 1 mRNA expression may reflect partial loss of AR function, it is also possible that differences in the relative cellular contribution of wild-type and mutant testes account for some of this apparent increase. In contrast to LH, serum FSH levels were significantly lower in AR113Q males than in wild-type mice (Figure 2D), a factor that may contribute to a failure in spermatogenesis. Along with lower levels of circulating hormone, FSH receptor mRNA levels were twofold higher in testes of AR113Q males than in wild-type controls (Figure 2F). Expression of other androgen-responsive targets was also altered in AR113Q males, including the levels of MUPs. MUPs are pheromone-binding proteins that contribute to normal mating behavior and whose production in the liver is indirectly regulated by circulating androgens23 (Figure 2G, left panel). Testosterone produced by the testes acts on the hypothalamic-pituitary axis to induce pulsatile release of growth hormone. This pattern of growth hormone release in males leads to elevated production of MUPs by the liver. MUPs excreted in the urine are readily detected by SDS-PAGE. Analysis revealed markedly lower levels of urinary MUPs in AR113Q compared with wild-type males (Figure 2G, right panel), consistent with the notion that these mutant mice exhibited signs of partial androgen insensitivity. It is noteworthy that not all androgen-responsive genes were differentially expressed in mutant and wild-type males. Similar levels of expression of the Leydig cell transcript 17β-hydroxysteroid dehydrogenase type III and the renal transcripts ornithine decarboxylase 1 and the cytochrome P450 Cyp2d9 were found in wild-type and AR113Q mice (data not shown). Taken together, these data confirm that AR113Q males exhibited signs of partial androgen insensitivity that modeled those of Kennedy disease patients.
Testes Pathology Reveals Toxic Effects of the Expanded Glutamine AR
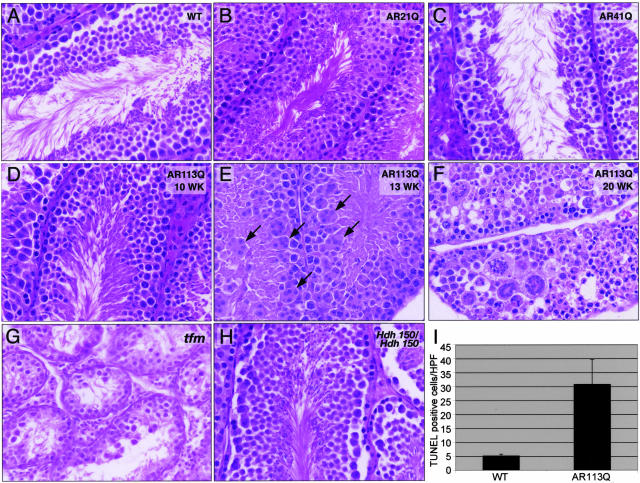
The observation that AR113Q males exhibited decreased fertility and systemic manifestations of partial androgen insensitivity prompted us to further explore the basis of this phenotype. Because both testicular atrophy and decreased fertility were present in this mouse model, we examined the testes of wild-type and AR113Q mice. Histological analysis revealed striking age-dependent testicular pathology in AR113Q knock-ins. Testes of AR113Q young adults (10 weeks or less) were morphologically similar to those of wild-type males and showed apparently normal germ cell maturation (Figure 3, A and D). With age, testes of AR113Q mice exhibited the frequent appearance of large multinucleated cells in virtually all tubules (Figure 3E), an approximate sevenfold decrease of epididymal sperm (5.99 × 106± 4.7 × 105 per ml in wild-type versus 8.47 × 105± 2.29 × 105 per ml in AR113Q, P = 0.001), and an approximate sixfold increase of TUNEL-positive cells (Figure 3I). This testicular pathology tended to progress with increasing age, resulting in extremely abnormal germ cell maturation in a subset of 20-week-old mice (Figure 3F). That this testicular pathology was progressive was supported by the fact that it was observed in none of seven mice aged 8 to 11 weeks but in all seven males aged 12 to 20 weeks. Furthermore, of the two mutant males that sired litters at less than 10 weeks, one had severely abnormal testes at 20 weeks, pictured in Figure 3F. Most strikingly, this testicular pathology was distinct from that which occurs in mice that lack a functional AR. Genetic males with the tfm mutation24 have a nonfunctional AR, are infertile, and have atrophic testes. Tfm testes exhibit a complete block of germ cell maturation yet showed none of the abnormal cytology that is so prominent in AR113Q testes (Figure 3G). We conclude that the testicular pathology in AR113Q mice is not solely mediated by a partial loss of AR function, as previously thought, but rather reflects toxic effects of the mutant protein conferred by the expanded glutamine tract. That this testicular pathology occurred in a glutamine length-dependent manner was confirmed by examination of testes from AR21Q and AR41Q mice, both of which were morphologically indistinguishable from those of wild-type mice (Figure 3, B and C). Although this testes pathology was glutamine length dependent, it was also AR specific. Male mice homozygous for a mutant Huntington disease gene homolog with the targeted insertion of 150 CAG repeats (Huntington disease homolog (Hdh) 150/Hdh150)25 exhibited morphologically normal testes (Figure 3H) that were of normal weight (data not shown).
Figure 3.
Age-dependent testicular pathology in AR113Q knock-in males. Testes from 14-week-old wild-type (A), 18-month-old AR21Q (B), and AR41Q knock-ins (C); 10-, 13-, and 20-week-old AR113Q knock-ins (D–F); and 12-week-old tfm (G) and 100-week-old Hdh150/Hdh150 knock-in males (H) were evaluated by hematoxylin and eosin staining. Arrows in E point to some of the multinucleated cells. Original magnification, ×400. I: The number of TUNEL-positive cells was determined in testes of wild-type (n = 4) and AR113Q males (n = 4) at 3 to 4 months of age. Data shown are mean number of labeled cells per 200× high-power field ± SD (P < 0.03 by Student’s t-test).
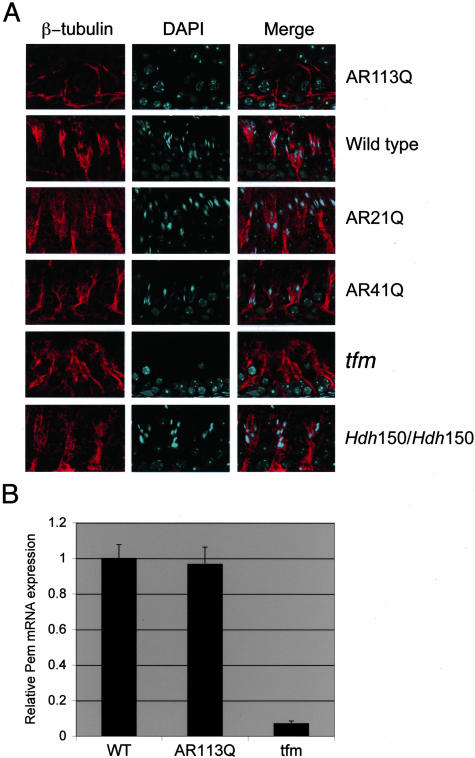
The predominant expression of AR protein by Sertoli cells led us to examine whether this cell type contributes to the testicular pathology in AR113Q mice. Sertoli cells are large supporting cells of the seminiferous tubules. Although their nuclei reside at the base of the tubule, their cytoplasm extends to its lumen. Along the way, microtubule-rich cell processes extend around syncytia of developing germ cells, pinching their cytoplasm so that they appear as individual germ cells by light microscopy. We hypothesized that abnormalities of the Sertoli cell cytoskeleton in AR113Q mice might lead to the appearance of multinucleated cells arising from these syncytia and to abnormal germ cell maturation. To test this hypothesis, we visualized the Sertoli cell cytoskeleton by β-tubulin immunofluorescence. We observed decreased staining for β-tubulin and diminished branching of the cytoskeleton in Sertoli cells of AR113Q mice compared with controls, including wild-type, AR21Q, AR41Q, and tfm mice (Figure 4A). These abnormalities in β-tubulin staining coincided with or preceded the development of testicular pathology in all of the AR113Q mice examined. No abnormalities of the Sertoli cell cytoskeleton were observed in Hdh150/Hdh150 mice, indicating that these cytoskeletal changes were reflective of toxicity specific to the mutant AR. Interestingly, expression of mRNA encoding the homeobox protein Pem, also known as reproductive homeobox on X, was similar in AR113Q and wild-type males (Figure 4B). Because Pem is an androgen-regulated gene expressed by Sertoli cells, these data suggested that Sertoli cell number was similar in AR113Q and wild-type testes. These data also suggested that changes in the cytoskeleton rather than Sertoli cell number were important contributors to the testicular pathology of AR113Q mice. However, the possibility that cytoskeletal abnormalities occurred predominantly as a consequence of disrupted germ cell maturation could not be excluded.
Figure 4.
Abnormalities of the Sertoli cell cytoskeleton in AR113Q knock-in males. Α: β-tubulin immunofluorescence highlights the Sertoli cell cytoskeleton in adult AR113Q, wild-type, AR21Q, AR41Q, tfm, and Hdh150/Hdh150 mice. Original magnification, ×630. B: Relative Pem mRNA expression levels in testes of wild-type (n = 5), AR113Q (n = 7), and tfm males (n = 4) at 3 to 4 months of age. Data shown are means ± SD relative to the expression in wild-type mice.
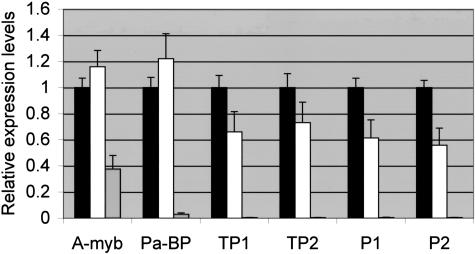
The striking morphological abnormalities in testes of AR113Q males prompted us to examine the expression of several genes that are markers of normal spermatogenesis and to compare their expression in wild-type and tfm mutant males. Quantitative real-time RT-PCR revealed that the expression of transition protein 1 and protamines 1 and 2 was significantly lower in AR113Q males than in wild-type controls (Figure 5). The basic proteins encoded by these genes replace histones in mature spermatids and are required for proper chromatin condensation.26 When the AR113Q males were separated into those with morphologically normal and abnormal testes, no statistically significant differences were detected between these subsets (data not shown), suggesting that changes in gene expression preceded the appearance of histological alterations. In contrast to the expression of the transition proteins and protamines, the expression of A-myb, a transcriptional regulator active in early meiotic prophase,27 and pro-acrosin binding protein, a gene expressed in primary spermatocytes,28 was not significantly different in AR113Q and wild-type males. Tfm mutant males expressed markedly lower levels of all genes examined, a reflection of the arrest of spermatogenesis that is evident in these mice.
Figure 5.
Altered expression of markers of spermatogenesis in AR113Q males. Relative mRNA expression levels of markers of spermatogenesis were determined in testes of wild-type (n = 5, black bars), AR113Q (n = 7, white bars), and tfm males (n = 4, striped bars) at 3 to 4 months of age. Data shown are means ± SD relative to the expression in wild-type mice. Pa-BP, pro-acrosin binding protein; TP1, transition protein 1; TP2, transition protein 2; P1, protamine 1; P2, protamine 2. Expression of TP1, P1, and P2 were significantly less in AR113Q compared with wild-type males (P < 0.05 by analysis of variance with Newman-Keuls post test).
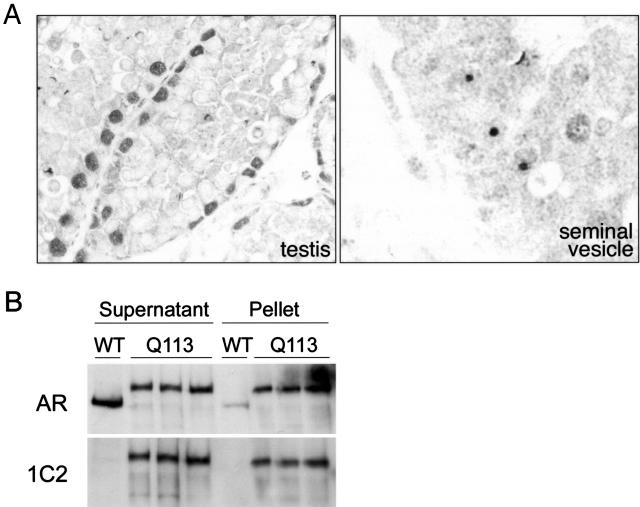
The testicular pathology in AR113Q mice occurred in the absence of detectable intranuclear inclusions of AR protein (Figure 6A, left panel). Using a polyclonal antibody against the AR amino terminus, we detected strong diffuse nuclear staining in Sertoli cells, whose large nuclei reside at the base of the seminiferous tubules. The presence of diffuse staining of Sertoli cell nuclei and the absence of intranuclear inclusions were consistent in all animals examined, including those with the most disordered germ cell maturation. In contrast, intranuclear inclusions were detected elsewhere in the genitourinary tract of these same animals, including the lining of the seminal vesicle (Figure 6A, right panel). Despite the absence of morphologically detectable AR aggregates in testes, biochemical analysis revealed decreased solubility of the mutant AR. Separation of protein lysates into soluble and pelleted fractions after high-speed centrifugation revealed a tendency of the expanded glutamine AR to reside in the pellet while the wild-type receptor remained in the soluble fraction (Figure 6B). This analysis confirmed similar overall expression of the wild-type and expanded glutamine AR and demonstrated that the mutant protein formed protein complexes in testes that were not detected by conventional morphological methods. Because AR protein was detected only in the nucleus by immunohistochemistry, our data suggest that complexes formed by the expanded glutamine AR were predominantly localized to this subcellular compartment.
Figure 6.
Androgen receptor protein expression in AR113Q and wild-type testes. A: Androgen receptor immunohistochemistry revealed diffuse nuclear staining in Sertoli cells of the testes of AR113Q knock-in male at 20 weeks (left panel) and intranuclear inclusions elsewhere in the genitourinary tract, including the lining of the seminal vesicle (right panel). Original magnification, ×400. B: Protein lysates from testes of one wild-type (WT) and three AR113Q knock-in adult male mice were separated into soluble and pellet fractions by centrifugation at 15,000 × g for 15 minutes and then resolved by 7% SDS-PAGE. AR protein was visualized by Western blot using an antibody against the amino terminus of the receptor (top) or against the expanded glutamine tract (1C2, bottom).
Discussion
We have generated a knock-in mouse model of Kennedy disease and used it to explore the pathogenesis of the systemic manifestations of this disorder. Mutant male mice have decreased fertility, signs of partial androgen insensitivity, and testicular atrophy, all of which are also seen in Kennedy disease patients. Our analyses demonstrate that AR113Q mice exhibit striking, age-dependent testicular pathology characterized by progressive abnormalities of germ cell maturation and the Sertoli cell cytoskeleton. This pathology is novel and contrasts with the complete block of spermatogenesis that is detected by morphological and genetic analyses of testes from tfm mutant mice (Figures 3 and 5) and from humans with testicular feminization syndrome. Ar conditional null mutant mice, which lack a functional receptor only in Sertoli cells, similarly exhibit an arrest of spermatogenesis and fail to develop the abnormalities of germ cell maturation or Sertoli cell cytoskeleton seen in AR113Q mice.29–31 We conclude that toxic effects of the expanded glutamine protein mediate aspects of the Kennedy disease phenotype previously attributed to a loss of AR function.
In support of this conclusion is the fact that similar testicular pathology has been reported in a yeast artificial chromosome transgenic model of Huntington disease.32 The observation that the expanded glutamine huntingtin protein expressed as a transgene also causes abnormalities of germ cell maturation and the Sertoli cell cytoskeleton suggests that common mechanisms mediate toxicity in both of these models. In contrast, we detected no testicular pathology in a knock-in mouse model of Huntington’s disease. Although the basis of this discrepancy is currently unclear, it is possible that either genetic background or huntingtin expression levels vary between the homozygous knock-in mice evaluated here and the YAC transgenic mice that were previously described. This latter possibility is in line with the observation that expression levels of expanded glutamine proteins significantly influence the onset and severity of phenotype in several transgenic mouse models of polyglutamine disease.
The toxic mechanisms that mediate testicular pathology in AR113Q mice do not exclude possible contributions by other factors, including a partial loss of normal AR function. It is well established that an expanded glutamine tract results in a partial loss of function in cell culture models of Kennedy disease.14–17 Elevated levels of serum LH and lower levels of urinary MUPs indicate that the expanded glutamine AR has also undergone a partial loss of function in AR113Q mice. Diminished activity of the mutant receptor as a regulator of gene expression may impact the development of testicular pathology through pathways that are not activated in the complete absence of a functional receptor, such as exists in tfm and Sertoli cell-specific Ar null mutants. Additionally, the detection of decreased levels of serum FSH, a hormone that binds to receptors on Sertoli cells to stimulate spermatogenesis, suggests that hypothalamic dysfunction or altered levels of other hormones, such as estrogens or inhibin, may also contribute to the phenotype in AR113Q mice.
The cytoskeletal abnormalities identified in Sertoli cells of AR113Q mice have implications beyond mechanisms leading to infertility. Among neurons, the extremely long axonal processes of motor neurons appear to make them particularly vulnerable to disruptions of the cytoskeleton and the transport machinery that uses this scaffold. This vulnerability is reflected by the set of recently identified mutations in mice and humans that affect components of this pathway and result in neuron dysfunction or death.33–38 Disruption of axonal transport has been observed in several models of polyglutamine disease, including Kennedy disease.8,9,39–41 Our data raise the possibility that disruption of axonal transport may be mediated by alterations in the cytoskeleton itself. Aggresome-forming proteins, including those that contain long glutamine tracts, cause a similar collapse of the cytoskeleton in cell culture.42–44 Here, we have observed such cytoskeletal alterations in vivo in the absence of morphologically detectable aggregates but in tissues where soluble, biochemically detectable protein complexes predominate.
The observed formation of androgen receptor protein complexes in vivo and the likelihood that they predominate in the cell nucleus suggest that a similar mechanism may lead to both loss of AR function and toxicity of the expanded glutamine receptor. Prior studies have shown that transcriptional co-regulators, including cyclic AMP response element-binding protein, SRC-1, and GRIP1, which interact with the wild-type AR, are sequestered into intranuclear inclusions of the expanded glutamine receptor.45,46 We propose that similar interactions occur between transcriptional co-regulators and the soluble microaggregates of the expanded glutamine AR identified here. The resulting misassortment of transcriptional co-regulators may lead to decreased activation of AR target genes, accounting for a partial loss of function, while simultaneously exerting broad effects on gene expression that mediate some aspects of AR113Q toxicity.
Acknowledgments
We thank Drs. Gary Hammer, Monzy Thomas, and Christopher Krebs for helpful discussions; Dr. Kenneth Fischbeck for review of our manuscript; Elizabeth Horn for preparation of figures; and the University of Michigan Transgenic Animal Core, Microscopy and Image Analysis Lab, and DNA Sequencing Core for assistance with this project.
Footnotes
Address reprint requests to Andrew P. Lieberman, Department of Pathology, University of Michigan Medical School, 1301 Catherine, 4233 Medical Science 1, Ann Arbor, Michigan 48109. E-mail: liebermn@umich.edu.
Supported by the Kennedy’s Disease Association, the University of Michigan Nathan Shock Center, the Muscular Dystrophy Association (grant 3816), a Beeson Career Development Award from the National Institutes of Health and the American Federation for Aging Research (K08 AG024758 to A.P.L.), a V.A. Merit Review grant to R.L.A., the D.O.D. (DAMD17-02-1-0099 to D.M.R.), University of Michigan Cancer Center Support grant (5 P30 CA46592), and the MDRTC Cell and Molecular Biology Core NIH grant (DK-20572). The University of Virginia Center for Research in Reproduction Ligand Assay and Analysis Core was supported by NICHD (SCCPRR) Grant U54-HD28934.
References
- Zoghbi HY, Orr HT. Glutamine repeats and neurodegeneration. Annu Rev Neurosci. 2000;23:217–247. doi: 10.1146/annurev.neuro.23.1.217. [DOI] [PubMed] [Google Scholar]
- La Spada AR, Wilson EM, Lubahn DB, Harding AE, Fischbeck KH. Androgen receptor gene mutations in X-linked spinal and bulbar muscular atrophy. Nature. 1991;352:77–79. doi: 10.1038/352077a0. [DOI] [PubMed] [Google Scholar]
- Chevalier-Larsen ES, O’Brien CJ, Wang H, Jenkins SC, Holder L, Lieberman AP, Merry DE. Castration restores function and neurofilament alterations of aged symptomatic males in a transgenic mouse model of spinal and bulbar muscular atrophy. J Neurosci. 2004;24:4778–4786. doi: 10.1523/JNEUROSCI.0808-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katsuno M, Adachi H, Kume A, Li M, Nakagomi Y, Niwa H, Sang C, Kobayashi Y, Doyu M, Sobue G. Testosterone reduction prevents phenotypic expression in a transgenic mouse model of spinal and bulbar muscular atrophy. Neuron. 2002;35:843–854. doi: 10.1016/s0896-6273(02)00834-6. [DOI] [PubMed] [Google Scholar]
- McManamny P, Chy HS, Finkelstein DI, Craythorn RG, Crack PJ, Kola I, Cheema SS, Horne MK, Wreford NG, O’Bryan MK, De Kretser DM, Morrison JR. A mouse model of spinal and bulbar muscular atrophy. Hum Mol Genet. 2002;11:2103–2111. doi: 10.1093/hmg/11.18.2103. [DOI] [PubMed] [Google Scholar]
- Thomas M, Dadgar N, Aphale A, Harrell JM, Kunkel R, Pratt WB, Lieberman AP. Androgen receptor acetylation site mutations cause trafficking defects, misfolding, and aggregation similar to expanded glutamine tracts. J Biol Chem. 2004;279:8389–8395. doi: 10.1074/jbc.M311761200. [DOI] [PubMed] [Google Scholar]
- McCampbell A, Taylor JP, Taye AA, Robitschek J, Li M, Walcott J, Merry D, Chai Y, Paulson H, Sobue G, Fischbeck KH. CREB-binding protein sequestration by expanded polyglutamine. Hum Mol Genet. 2000;9:2197–2202. doi: 10.1093/hmg/9.14.2197. [DOI] [PubMed] [Google Scholar]
- Szebenyi G, Morfini GA, Babcock A, Gould M, Selkoe K, Stenoien DL, Young M, Faber PW, MacDonald ME, McPhaul MJ, Brady ST. Neuropathogenic forms of huntingtin and androgen receptor inhibit fast axonal transport. Neuron. 2003;40:41–52. doi: 10.1016/s0896-6273(03)00569-5. [DOI] [PubMed] [Google Scholar]
- Piccioni F, Pinton P, Simeoni S, Pozzi P, Fascio U, Vismara G, Martini L, Rizzuto R, Poletti A. Androgen receptor with elongated polyglutamine tract forms aggregates that alter axonal trafficking and mitochondrial distribution in motor neuronal processes. FASEB J. 2002;16:1418–1420. doi: 10.1096/fj.01-1035fje. [DOI] [PubMed] [Google Scholar]
- Thomas M, Yu Z, Dadgar N, Varambally S, Yu J, Chinnaiyan AM, Lieberman AP. The unfolded protein response modulates toxicity of the expanded glutamine androgen receptor. J Biol Chem. 2005;280:21264–21271. doi: 10.1074/jbc.M500144200. [DOI] [PubMed] [Google Scholar]
- Zuccato C, Tartari M, Crotti A, Goffredo D, Valenza M, Conti L, Cataudella T, Leavitt BR, Hayden MR, Timmusk T, Rigamonti D, Cattaneo E. Huntingtin interacts with REST/NRSF to modulate the transcription of NRSE-controlled neuronal genes. Nat Genet. 2003;35:76–83. doi: 10.1038/ng1219. [DOI] [PubMed] [Google Scholar]
- Lieberman AP, Fischbeck KH. Triplet repeat expansion in neuromuscular disease. Muscle Nerve. 2000;23:843–850. doi: 10.1002/(sici)1097-4598(200006)23:6<843::aid-mus2>3.0.co;2-8. [DOI] [PubMed] [Google Scholar]
- Fischbeck KH, Lieberman A, Bailey CK, Abel A, Merry DE. Androgen receptor mutation in Kennedy’s disease. Philos Trans R Soc Lond B Biol Sci. 1999;354:1075–1078. doi: 10.1098/rstb.1999.0461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kazemi-Esfarjani P, Trifiro MA, Pinsky L. Evidence for a repressive function of the long polyglutamine tract in the human androgen receptor: possible pathogenetic relevance for the (CAG)n-expanded neuronopathies. Hum Mol Genet. 1995;4:523–527. doi: 10.1093/hmg/4.4.523. [DOI] [PubMed] [Google Scholar]
- Lieberman AP, Harmison G, Strand AD, Olson JM, Fischbeck KH. Altered transcriptional regulation in cells expressing the expanded polyglutamine androgen receptor. Hum Mol Genet. 2002;11:1967–1976. doi: 10.1093/hmg/11.17.1967. [DOI] [PubMed] [Google Scholar]
- Mhatre AN, Trifiro MA, Kaufman M, Kazemi-Esfarjani P, Figlewicz D, Rouleau G, Pinsky L. Reduced transcriptional regulatory competence of the androgen receptor in X-linked spinal and bulbar muscular atrophy. Nat Genet. 1993;5:184–188. doi: 10.1038/ng1093-184. [DOI] [PubMed] [Google Scholar]
- Chamberlain NL, Driver ED, Miesfeld RL. The length and location of CAG trinucleotide repeats in the androgen receptor N-terminal domain affect transactivation function. Nucleic Acids Res. 1994;22:3181–3186. doi: 10.1093/nar/22.15.3181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu RN, Ito M, Saunders TL, Camper SA, Jameson JL. Role of Ahch in gonadal development and gametogenesis. Nat Genet. 1998;20:353–357. doi: 10.1038/3822. [DOI] [PubMed] [Google Scholar]
- Swiatek PJ, Gridley T. Perinatal lethality and defects in hindbrain development in mice homozygous for a targeted mutation of the zinc finger gene Krox20. Genes Dev. 1993;7:2071–2084. doi: 10.1101/gad.7.11.2071. [DOI] [PubMed] [Google Scholar]
- Gu H, Zou YR, Rajewsky K. Independent control of immunoglobulin switch recombination at individual switch regions evidenced through Cre-loxP-mediated gene targeting. Cell. 1993;73:1155–1164. doi: 10.1016/0092-8674(93)90644-6. [DOI] [PubMed] [Google Scholar]
- Dejager S, Bry-Gauillard H, Bruckert E, Eymard B, Salachas F, LeGuern E, Tardieu S, Chadarevian R, Giral P, Turpin G. A comprehensive endocrine description of Kennedy’s disease revealing androgen insensitivity linked to CAG repeat length. J Clin Endocrinol Metab. 2002;87:3893–3901. doi: 10.1210/jcem.87.8.8780. [DOI] [PubMed] [Google Scholar]
- O’Shaughnessy PJ, Johnston H, Willerton L, Baker PJ. Failure of normal adult Leydig cell development in androgen-receptor-deficient mice. J Cell Sci. 2002;115:3491–3496. doi: 10.1242/jcs.115.17.3491. [DOI] [PubMed] [Google Scholar]
- Tullis KM, Krebs CJ, Leung JY, Robins DM. The regulator of sex-limitation gene, rsl, enforces male-specific liver gene expression by negative regulation. Endocrinology. 2003;144:1854–1860. doi: 10.1210/en.2002-0190. [DOI] [PubMed] [Google Scholar]
- Gaspar ML, Meo T, Bourgarel P, Guenet JL, Tosi M. A single base deletion in the Tfm androgen receptor gene creates a short-lived messenger RNA that directs internal translation initiation. Proc Natl Acad Sci USA. 1991;88:8606–8610. doi: 10.1073/pnas.88.19.8606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin CH, Tallaksen-Greene S, Chien WM, Cearley JA, Jackson WS, Crouse AB, Ren S, Li XJ, Albin RL, Detloff PJ. Neurological abnormalities in a knock-in mouse model of Huntington’s disease. Hum Mol Genet. 2001;10:137–144. doi: 10.1093/hmg/10.2.137. [DOI] [PubMed] [Google Scholar]
- Wouters-Tyrou D, Martinage A, Chevaillier P, Sautiere P. Nuclear basic proteins in spermiogenesis. Biochimie. 1998;80:117–128. doi: 10.1016/s0300-9084(98)80018-7. [DOI] [PubMed] [Google Scholar]
- Toscani A, Mettus RV, Coupland R, Simpkins H, Litvin J, Orth J, Hatton KS, Reddy EP. Arrest of spermatogenesis and defective breast development in mice lacking A-myb. Nature. 1997;386:713–717. doi: 10.1038/386713a0. [DOI] [PubMed] [Google Scholar]
- Baba T, Niida Y, Michikawa Y, Kashiwabara S, Kodaira K, Takenaka M, Kohno N, Gerton GL, Arai Y. An acrosomal protein, sp32, in mammalian sperm is a binding protein specific for two proacrosins and an acrosin intermediate. J Biol Chem. 1994;269:10133–10140. [PubMed] [Google Scholar]
- Chang C, Chen YT, Yeh SD, Xu Q, Wang RS, Guillou F, Lardy H, Yeh S. Infertility with defective spermatogenesis and hypotestosteronemia in male mice lacking the androgen receptor in Sertoli cells. Proc Natl Acad Sci USA. 2004;101:6876–6881. doi: 10.1073/pnas.0307306101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Gendt K, Swinnen JV, Saunders PT, Schoonjans L, Dewerchin M, Devos A, Tan K, Atanassova N, Claessens F, Lecureuil C, Heyns W, Carmeliet P, Guillou F, Sharpe RM, Verhoeven G. A Sertoli cell-selective knockout of the androgen receptor causes spermatogenic arrest in meiosis. Proc Natl Acad Sci USA. 2004;101:1327–1332. doi: 10.1073/pnas.0308114100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holdcraft RW, Braun RE. Androgen receptor function is required in Sertoli cells for the terminal differentiation of haploid spermatids. Development. 2004;131:459–467. doi: 10.1242/dev.00957. [DOI] [PubMed] [Google Scholar]
- Leavitt BR, Guttman JA, Hodgson JG, Kimel GH, Singaraja R, Vogl AW, Hayden MR. Wild-type huntingtin reduces the cellular toxicity of mutant huntingtin in vivo. Am J Hum Genet. 2001;68:313–324. doi: 10.1086/318207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xia CH, Roberts EA, Her LS, Liu X, Williams DS, Cleveland DW, Goldstein LS. Abnormal neurofilament transport caused by targeted disruption of neuronal kinesin heavy chain KIF5A. J Cell Biol. 2003;161:55–66. doi: 10.1083/jcb.200301026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hafezparast M, Klocke R, Ruhrberg C, Marquardt A, Ahmad-Annuar A, Bowen S, Lalli G, Witherden AS, Hummerich H, Nicholson S, Morgan PJ, Oozageer R, Priestley JV, Averill S, King VR, Ball S, Peters J, Toda T, Yamamoto A, Hiraoka Y, Augustin M, Korthaus D, Wattler S, Wabnitz P, Dickneite C, Lampel S, Boehme F, Peraus G, Popp A, Rudelius M, Schlegel J, Fuchs H, Hrabe de Angelis M, Schiavo G, Shima DT, Russ AP, Stumm G, Martin JE, Fisher EM. Mutations in dynein link motor neuron degeneration to defects in retrograde transport. Science. 2003;300:808–812. doi: 10.1126/science.1083129. [DOI] [PubMed] [Google Scholar]
- Zhao C, Takita J, Tanaka Y, Setou M, Nakagawa T, Takeda S, Yang HW, Terada S, Nakata T, Takei Y, Saito M, Tsuji S, Hayashi Y, Hirokawa N. Charcot-Marie-Tooth disease type 2A caused by mutation in a microtubule motor KIF1Bbeta. Cell. 2001;105:587–597. doi: 10.1016/s0092-8674(01)00363-4. [DOI] [PubMed] [Google Scholar]
- Reid E, Kloos M, Ashley-Koch A, Hughes L, Bevan S, Svenson IK, Graham FL, Gaskell PC, Dearlove A, Pericak-Vance MA, Rubinsztein DC, Marchuk DA. A kinesin heavy chain (KIF5A) mutation in hereditary spastic paraplegia (SPG10). Am J Hum Genet. 2002;71:1189–1194. doi: 10.1086/344210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LaMonte BH, Wallace KE, Holloway BA, Shelly SS, Ascano J, Tokito M, Van Winkle T, Howland DS, Holzbaur EL. Disruption of dynein/dynactin inhibits axonal transport in motor neurons causing late-onset progressive degeneration. Neuron. 2002;34:715–727. doi: 10.1016/s0896-6273(02)00696-7. [DOI] [PubMed] [Google Scholar]
- Puls I, Jonnakuty C, LaMonte BH, Holzbaur EL, Tokito M, Mann E, Floeter MK, Bidus K, Drayna D, Oh SJ, Brown RH, Jr, Ludlow CL, Fischbeck KH. Mutant dynactin in motor neuron disease. Nat Genet. 2003;33:455–456. doi: 10.1038/ng1123. [DOI] [PubMed] [Google Scholar]
- Gunawardena S, Her LS, Brusch RG, Laymon RA, Niesman IR, Gordesky-Gold B, Sintasath L, Bonini NM, Goldstein LS. Disruption of axonal transport by loss of huntingtin or expression of pathogenic polyQ proteins in Drosophila. Neuron. 2003;40:25–40. doi: 10.1016/s0896-6273(03)00594-4. [DOI] [PubMed] [Google Scholar]
- Lee WC, Yoshihara M, Littleton JT. Cytoplasmic aggregates trap polyglutamine-containing proteins and block axonal transport in a Drosophila model of Huntington’s disease. Proc Natl Acad Sci USA. 2004;101:3224–3229. doi: 10.1073/pnas.0400243101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trushina E, Dyer RB, Badger JD, II, Ure D, Eide L, Tran DD, Vrieze BT, Legendre-Guillemin V, McPherson PS, Mandavilli BS, Van Houten B, Zeitlin S, McNiven M, Aebersold R, Hayden M, Parisi JE, Seeberg E, Dragatsis I, Doyle K, Bender A, Chacko C, McMurray CT. Mutant huntingtin impairs axonal trafficking in mammalian neurons in vivo and in vitro. Mol Cell Biol. 2004;24:8195–8209. doi: 10.1128/MCB.24.18.8195-8209.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bence NF, Sampat RM, Kopito RR. Impairment of the ubiquitin-proteasome system by protein aggregation. Science. 2001;292:1552–1555. doi: 10.1126/science.292.5521.1552. [DOI] [PubMed] [Google Scholar]
- Johnston JA, Ward CL, Kopito RR. Aggresomes: a cellular response to misfolded proteins. J Cell Biol. 1998;143:1883–1898. doi: 10.1083/jcb.143.7.1883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia-Mata R, Bebok Z, Sorscher EJ, Sztul ES. Characterization and dynamics of aggresome formation by a cytosolic GFP-chimera. J Cell Biol. 1999;146:1239–1254. doi: 10.1083/jcb.146.6.1239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCampbell A, Taye AA, Whitty L, Penney E, Steffan JS, Fischbeck KH. Histone deacetylase inhibitors reduce polyglutamine toxicity. Proc Natl Acad Sci USA. 2001;98:15179–15184. doi: 10.1073/pnas.261400698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stenoien DL, Cummings CJ, Adams HP, Mancini MG, Patel K, DeMartino GN, Marcelli M, Weigel NL, Mancini MA. Polyglutamine-expanded androgen receptors form aggregates that sequester heat shock proteins, proteasome components and SRC-1, and are suppressed by the HDJ-2 chaperone. Hum Mol Genet. 1999;8:731–741. doi: 10.1093/hmg/8.5.731. [DOI] [PubMed] [Google Scholar]