Abstract
We developed a useful method for the detection of rubella virus genome RNA by reverse transcription loop-mediated isothermal amplification (RT-LAMP) and compared the sensitivity of RT-LAMP with that of other virological tests: reverse transcription-PCR (RT-PCR) and virus isolation. The rubella virus genome was amplified by RT-LAMP from clinical isolates obtained between 1987 and 2004 with similar sensitivities to the Takahashi vaccine strain. The detection limit of RT-LAMP was compared with that of RT-PCR using the Takahashi vaccine strain. We detected rubella virus genome material corresponding to 30 PFU/ml in a culture fluid sample by RT-LAMP within 60 min after the extraction of RNA with equal sensitivity to RT-nested PCR. The positive result rates of RT-LAMP, RT-PCR, and virus isolation were also compared using throat swabs obtained from patients who were clinically diagnosed with acute rubella virus infection in 2004 in Tochigi, Japan. Among nine patients with clinical rubella, the positive result rates were three/nine (33.3%) for virus isolation, six/nine (66.7%) for RT-PCR, and seven/nine (77.8%) for RT-LAMP. Consequently, RT-LAMP for rubella virus would be expected to be a reliable rapid diagnostic tool in the clinical setting.
Rubella virus is an enveloped positive-strand RNA virus that is the sole member of the Rubivirus genus of the Togaviridae family. The rubella virus genome consists of less than 10,000 nucleotides and encodes five proteins, including three structural proteins, E1, E2, and the capsid protein. E1 is a structural glycoprotein with neutralizing and hemagglutinating epitopes (5, 22, 31). According to a report on the meeting organized by the World Health Organization (WHO) in 2004 to discuss the standardization of nomenclature for describing the genetic characteristics of wild-type rubella virus, the E1 gene sequence between genome positions 8731 and 9469 is recommended as a target site for routine genotyping analysis for molecular epidemiology (30).
Rubella virus infection is one of the communicable diseases in infants and children. Most patients with rubella develop mild symptoms and recover without any complications or sequelae. However, infection among young women who have no immunity against rubella virus during early pregnancy, especially within the first trimester, may cause fetal death or congenital rubella virus infection/syndrome (CRS). CRS is characterized by multiple malformations: deafness, eye abnormalities, congenital cardiac disease, neurological abnormalities, and so on (2, 31). Although the burden of CRS is not well characterized in all countries, WHO estimated that more than 100,000 cases of CRS occur every year in developing countries and 43% (91/214) of countries did not have a national immunization program for rubella in 2002 (2, 31). However, in the United States and several European countries, the indigenous circulation of rubella has been interrupted and CRS has been eliminated. This achievement of the elimination of CRS largely depends on vaccination programs with effective surveillance (2).
In Japan, nationwide outbreaks of rubella have not occurred since 1992-1993. The number of reported cases of clinical rubella decreased from 2,795 in 2003 and 4,248 in 2004 to 895 in 2005 from approximately 3,000 pediatric sentinel sites without requiring any laboratory confirmation (national surveillance data from the National Institute of Infectious Disease, Japan). Although only one case of CRS was reported annually during 1999 to 2003, 10 patients with CRS were reported to the national surveillance program in 2004 and two were reported in 2005. The issue of rubella and CRS still remains important in Japan for the elimination of CRS with the accelerated control of rubella. A single dose of monovalent rubella vaccine targeted to children aged between 12 and 90 months was introduced in a national immunization program in Japan in 1994. From April 2006, Japan has launched a two-dose immunization program with combined measles and rubella vaccine given to children aged 1 year and aged 5 or 6 years, before primary school entry.
There are some problems with diagnosing rubella and CRS from clinical information alone, because some infected individuals are asymptomatic or have mild symptoms. Different viruses such as measles, human parvovirus B19, and enterovirus cause symptoms similar to those of rubella (2, 25, 32). Also, misclassification of cases of CRS may occur, since some patients with CRS may have a single symptom or defect or may have a positive result for immunoglobulin M (IgM) enzyme immunosorbent assays (EIAs) without clinical symptoms (4, 5, 33). The development of efficient laboratory tests will enable the accurate diagnosis of rubella and CRS.
Virus isolation is traditionally performed for the laboratory confirmation of rubella virus infection, but the sensitivity of virus isolation is low because of poor conditions of sample transport and inadequate timing of the sample collection. It takes 3 to 4 weeks to obtain the results of virus isolation after three blind passages in RK13 or Vero cells (5, 22). Serological tests such as the hemagglutination inhibition method are performed principally with paired sera from the acute and convalescent phases (5). Although IgM EIA is employed for diagnosis using a single serum sample, false-positive results of serum rubella IgM tests may occur in people with human parvovirus B19 infection, with a positive heterophile test for infectious mononucleosis, or with positive rheumatoid factor (1, 24, 25, 28, 29).
Recently, several molecular biological laboratory tests for the detection of rubella virus have been developed, such as reverse transcription-PCR (RT-PCR) (3, 6, 8, 13, 15, 22). Loop-mediated isothermal amplification (LAMP) was originally developed for DNA amplification, as reported by Notomi et al. (19) in 2000. This method was also applied for the detection of RNA genomes. LAMP amplifies targeted nucleotides with six primers within 1 hour under isothermal conditions without any temperature shifts such as those used for denaturation, annealing, and extension in the PCR cycling reactions (19). In this study, we developed an RT-LAMP method for the detection of the rubella virus genome and compared the sensitivity of RT-LAMP with those of RT-PCR and virus isolation using clinical samples.
(We presented this study at the plenary session of the 46th Annual Meeting of the Japanese Society of Clinical Virology, Fukuoka, Japan, June 2005.)
MATERIALS AND METHODS
Clinical samples and virus isolation.
We obtained nine samples from patients diagnosed with acute rubella virus infection by some of the authors in our collaborating hospital between April and May 2004 in Tochigi prefecture, Japan, during a rubella community outbreak. Some of them were serologically diagnosed by IgM antibodies against rubella virus that were assayed using the Rubella IgM EIA kit (Denka Seiken, Niigata, Japan) per the instruction manual. Clinical samples of throat swabs were obtained from patients with acute rubella virus infection, and 0.1 ml of samples was inoculated on monolayers of RK13 cells in minimum essential medium containing 2% calf serum and adequate antibiotics. After three blind passages, the plate was stained with avidin-biotinylated enzyme complex, using polyclonal rabbit antibodies against rubella virus, monoclonal antibodies against rabbit IgG, and avidin-biotinylated enzyme complex staining kits (Vector Laboratories, Inc., California). The name of the isolated rubella virus was specified according to the WHO nomenclature (30).
The Takahashi vaccine strain developed at the Kitasato Institute, Japan (RV/Takahashi: AB03338), was used as the reference strain in this study. Also, we used four isolates as wild circulating strains: two strains (RVi/Tokyo.JPN/87CRS-w: AB238916 and RVi/Tokyo.JPN/87CRS-o: AB238917) isolated in 1987 in Tokyo, Japan; one strain (RVi/Tokyo.JPN/95CRS-n: AB238918) isolated in 1995 in Tokyo, Japan; and one strain (RVi/Kanagawa.JPN/04-s: AB238915) isolated from a patient with acute rubella virus infection in 2004 in Kanagawa, Japan. The E1 sequence data were registered with the DNA Data Bank of Japan under the accession numbers in parentheses.
RT-LAMP.
The genomic RNA was extracted from 200 μl of virus culture fluids of rubella virus or clinical samples using a magnetic bead RNA purification kit (TOYOBO Co. Ltd., Osaka, Japan). RNA was resuspended in 30 μl of distilled water, and 5 μl was subjected to RT-LAMP and RT-PCR. RT-LAMP was carried out using autocycling strand displacement DNA synthesis performed using Bst DNA polymerase (New England Biolabs). The RT-LAMP primers were designed using the software program for LAMP primer design (Eiken Chemical Co. Ltd., Japan). The RT-LAMP primers were designed in the E1 region between genome positions 8476 and 8680. A diagram of the RT-LAMP primers is shown in Fig. 1. We synthesized six primers: outer primers (F3 and B3), inner primers (FIP and BIP), and two additional loop primers (F loop and B loop). FIP contained a sequence complementary to F1 linked with the F2 sequence, and BIP contained a B1 sequence linked with a sequence complementary to B2. These four primers amplified the targeted DNA. We synthesized the F loop primer between F1 and F2, and the B loop primer between B1 and B2, to enhance the sensitivity and reactivity (17, 18, 19).
FIG. 1.
Diagram of RT-LAMP primers for the detection of the rubella virus genome. The eight primer sites are shown in the upper panel. Sequence alignments of six LAMP primers are shown in the lower panel. Positive-sense F3 and complementary B3 were used as outer primers. FIP contains the sequence complementary to F1 linked with the F2 sequence. BIP contains the B1 sequence linked with the sequence complementary to B2. Two additional loop primers (F and B) are synthesized between F1 and F2 and between B1 and B2, respectively. The arrows show the direction of DNA synthesis.
For the RT-LAMP reaction, an RT-LAMP mixture was made up to a total of 25 μl, containing 40 pmol each of FIP and BIP, 5 pmol each of F3 and B3, 20 pmol each of F loop and B loop, 1.4 mM of each deoxynucleoside triphosphate, 0.8 mM betaine, 20 mM Tris-HCl, 10 mM KCl, 10 mM (NH4)2SO4, 8 M MgSO4, 0.1% Tween 20, 0.5 U of avian myeloblastosis virus reverse transcriptase, 8 U of Bst DNA polymerase (New England Biolabs), and 5 μl of sample RNA. The reaction mixture was incubated in an LA200 real-time turbidimeter (Teramecs, Japan) at 63°C for 60 min (16, 17, 18, 19).
As the RT-LAMP reaction progresses, the reaction by-products, pyrophosphate ions, bind to magnesium ions and form a white precipitate of magnesium pyrophosphate. Thus, the value of the turbidity is closely related to the amplification of targeted DNA (16).
RT-PCR.
The purified RNA genome was converted to cDNA with avian myeloblastosis virus reverse transcriptase (Life Science Inc.) at 50°C for 1 h, using primer 9407-9426 (−) (5′-AGGGCGGCGGTGACGAACTT-3′). Sets of primers in the E1 region for the detection of rubella virus genes were designed as follows: 8110-8129 (+) (5′-GTCTCTTGATCACGCCCTCG-3′) and 8797-8816 (−) (5′-GTGTTGCAGAACGGGTGTTCA-3′) for the first PCR and 8476-8494 (+) (same as F3 RT-LAMP primer) and 8663-8680 (−) (same as B3 RT-LAMP primer) for the nested PCR. The RT-PCR was performed with 1.25 U of Taq DNA polymerase (TaKaRa BioMedicals, Tokyo, Japan) in a thermal cycler (TaKaRa BioMedicals, Tokyo, Japan) with 35 rounds of the following thermal cycling conditions; denaturation at 95°C for 30 seconds, annealing at 58°C for 30 seconds, and extension at 72°C for 1 min 40 seconds. The PCR products were confirmed by electrophoresis through a 1.5% agarose gel and stained with ethidium bromide (10, 20, 27).
To conduct the sequence analysis using the PCR products from virus isolates, we made different sets of PCR primers designed as follows: 7981-8000 (+) (5′-CGAAGACGGCTGGACTTGCC-3′) and 9743-9762 (−) (5′-CTATGCAGCAACAGGTGCGG-3′) for the first PCR and 8110-8129 (+) and 9743-9762 (−) for the nested PCR. The nested RT-PCR products including the target site between genome positions 8731 and 9469 were used for routine genotyping analysis (30).
RESULTS
Sensitivity of RT-LAMP.
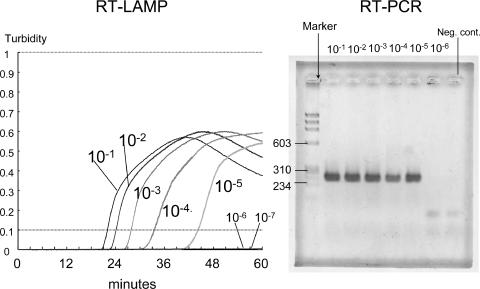
The rubella virus genomic RNA was extracted from 200 μl of culture fluid containing 105.5 PFU/0.1 ml of the Takahashi vaccine strain. The genome was serially diluted by 10-fold, and each dilution was subjected to RT-PCR and RT-LAMP. The results are shown in Fig. 2. The genome was amplified at least until the 10−5 dilution by both RT-LAMP and RT-PCR. The detection limit was calculated as 30 PFU/ml in culture fluid for both RT-LAMP and RT-PCR and estimated as 1 PFU of infectious particles in 5 μl of RNA materials in a single test.
FIG. 2.
Detection limit of RT-LAMP and RT-PCR. The Takahashi vaccine strain containing 105.5 PFU/0.1 ml was used. RNA was serially diluted by 1:10, and each dilution was subjected to RT-LAMP and RT-PCR. A result with a turbidity of ≥0.1 was considered to be LAMP positive. Numbers at left of the right panel are sizes in base pairs of the DNA marker.
As for the specificity of RT-LAMP for rubella, RNA samples extracted from other viruses (measles virus, mumps virus, respiratory syncytial virus [RSV], and influenza virus) were examined by RT-LAMP for rubella. These viruses were found to be negative for rubella RT-LAMP, demonstrating the lack of cross-reaction (data are not shown).
Detection of rubella virus genome by RT-LAMP.
Nine patients who were clinically diagnosed with acute rubella virus infection were examined. They were 0 to 8 years old, and six (66.7%) were male. The results of virus isolation, RT-PCR, and RT-LAMP are summarized in Table 1. Rubella viruses were isolated from three patients (33.3%) and designated RVi/Tochigi.JPN/04-s, RVi/Tochigi.JPN/04-h, and RVi/Tochigi.JPN/04-i, their E1 region genome sequences having the accession numbers AB238919, AB238920, and AB238921, respectively. They were classified into genotype 1D. Rubella virus genome was detected in six patients (66.7%) by RT-PCR and seven (77.8%) by RT-LAMP. The rubella virus genome was detected by RT-PCR and RT-LAMP in all three patients positive for virus isolation. Among six patients negative for virus isolation, the virus genome was detected in three by RT-PCR and in four by RT-LAMP. The backgrounds of the patients are shown in Table 2, together with the results of virological examinations. Eight patients did not have a history of immunization against rubella, and the rubella immunization history of case 1 was unknown. The rubella IgM EIA was performed in four (cases 5, 6, 7, and 9) out of nine patients and was found to be positive in all. Of two patients negative for all three tests, one patient (case 5) was positive for IgM EIA antibodies and another patient (case 3) had family contact with a laboratory-confirmed patient (case 6). Among four patients (cases 5, 6, 7, and 9) who were serologically confirmed to have rubella virus infection by IgM EIA, the rubella virus genome was detected in three by RT-LAMP and RT-PCR. Among five patients who were clinically diagnosed without serological confirmation, RT-PCR or RT-LAMP detected rubella virus genome in four patients.
TABLE 1.
Results of virological examinations: virus isolation, RT-PCR, and RT-LAMP, using nine clinical samples obtained from nine patients suspected of rubella virus infection
| Method and type of result (n) | No. of results by method and type (n)
|
|||
|---|---|---|---|---|
| RT-PCR
|
RT-LAMP
|
|||
| Positive (6) | Negative (3) | Positive (7) | Negative (2) | |
| Virus isolation | ||||
| Positive (3) | 3 | 0 | 3 | 0 |
| Negative (6) | 3 | 3 | 4 | 2 |
| RT-PCR | ||||
| Positive (6) | 6 | 0 | ||
| Negative (3) | 1 | 2 | ||
TABLE 2.
Immunization history and the results of serology, virus isolation, RT-PCR, and RT-LAMP for nine patients
| Patient | Immunization history | Result by methoda
|
|||
|---|---|---|---|---|---|
| Rubella IgM EIA | Virus isolation | RT-PCR | RT-LAMP | ||
| Case 1 | Unknown | ND | − | + | + |
| Case 2 | No | ND | − | + | + |
| Case 3 | No | ND | − | − | − |
| Case 4 | No | ND | − | + | + |
| Case 5 | No | + | − | − | − |
| Case 6 | No | + | − | − | + |
| Case 7 | No | + | + | + | + |
| Case 8 | No | ND | + | + | + |
| Case 9 | No | + | + | + | + |
+, positive; −, negative; ND, not done.
RT-LAMP detected the genome of different genotypes, 1E (RVi/Tokyo.JPN/87CRS-w, RVi/Tokyo.JPN/87CRS-o, and RVi/Tokyo.JPN/95CRS-n) and 1a (RVi/Kanagawa.JPN/04-s). The difference between the Takahashi vaccine strain and the wild strains in this study was 2.8 to 5.8% of nucleotides and 1.0 to 2.4% of amino acids in the E1 region. There was no significant difference in the sensitivity of rubella RT-LAMP among the circulating wild strains and vaccine strain in Japan (data not shown).
DISCUSSION
The rubella virus genomes from the Takahashi vaccine strain and the current wild strains were successfully amplified by RT-LAMP. The detection limit was calculated as 30 PFU/ml in culture fluid for both RT-LAMP and RT-PCR and estimated as 1 PFU of infectious particles in 5 μl of RNA materials in a single test. Actually, RT-LAMP detected the rubella virus genome in all six samples positive for RT-PCR and also in one of three samples negative for RT-PCR. The detection limit of RT-LAMP for rubella (30 PFU/ml in culture fluids) showed the slightly lower sensitivity of this method compared with that for other viruses: 0.15 to 0.4 50% tissue culture infective doses (TCID50)/ml of measles virus (10), 3 PFU/ml of mumps virus (20), and 0.5 to 1.5 TCID50/ml of RSV (27). RT-LAMP for measles virus, mumps virus, and RSV has been used for routine clinical examinations in our laboratory.
The RT-LAMP primers were designed based on the Takahashi vaccine strain, since the wild strains of rubella (clades 1 and 2) were found to differ by 8 to 10% at the nucleotide level (5). The genetic differences in the E1 region between the wild strains and Takahashi vaccine strain were 2.8 to 5.8% in our study. Although RT-LAMP was not performed for clade 2, we suppose that RT-LAMP would detect other genotypes because the RT-LAMP primers were designed in the conserved region. RT-LAMP amplifies 200 to 250 bp of nucleotides with high sensitivity and specificity. The products of RT-LAMP are not appropriate for sequence analysis because most of the region amplified by RT-LAMP is occupied by the RT-LAMP primers as shown in Fig. 1. WHO recommended the E1 coding region for molecular epidemiology or a window of 739 nucleotides, 8731 to 9469, for routine molecular epidemiological analysis (30, 32, 33). As for genotyping, E1 gene should be amplified by conventional RT-PCR.
RT-LAMP for rubella would be expected to be a reliable and rapid diagnostic method in the clinical setting, because RT-LAMP showed an equivalent or higher detection rate compared with RT-PCR and virus isolation for nine samples obtained from clinically diagnosed patients. The RT-LAMP procedure has clinical advantages of simplicity and rapidity, in comparison with virus isolation and RT-PCR. Virus isolation requires complex procedures for cell culture, is not always successful, and is not appropriate for a clinical laboratory diagnostic tool. The genome amplification method always has the possibility of false positives due to cross-contamination. Since RT-LAMP is performed as a simple procedure in a single tube with sensitivity similar to that of nested PCR, cross-reaction would less likely occur in RT-LAMP than in RT-PCR.
The detection limit of RT-PCR primers used in our study (30 PFU/ml) was similar to that in previous studies, and the RT-PCR primers were designed to have the same target as the RT-LAMP primers in order to compare the sensitivities of RT-LAMP and RT-PCR. In previous reports, RT-PCR for rubella targeting the E1 region detected up to 8 infectious units of WHO international standard/ml in amniotic fluid (8, 15) and a 10−6 dilution of culture fluid containing 106.8 TCID50/ml of rubella virus, reported by Bosma et al. (3). Cooray et al. (6) also reported that the rubella genome RNA was detected in oral fluid containing 100 PFU/ml.
In several laboratories, real-time PCR has been developed for the detection of measles virus (21, 23), mumps virus (14), and RSV (9, 12), but real-time PCR for rubella virus has not been reported. This method is based on PCR temperature shifts and shows higher sensitivity than RT-PCR with a reduction of the risk of cross-contamination. As it takes 2 to 3 h to obtain the results by real-time PCR, RT-LAMP has the distinct benefit of a faster reaction, within 60 min. Also, Tzeng et al. (26) improved the methodology of virus isolation developed using a replicon-based reporter gene assay for the detection of rubella virus. LAMP is a simpler, more sensitive, and more rapid method than virus isolation.
Misclassification of the clinical diagnosis of the nine patients from whom we collected samples in our study might have occurred, since we did not require a serological confirmation for the definition of the cases. Among the nine patients in this study, IgM EIA was performed in four patients. The remaining five did not receive an IgM test, but they were strongly suspected to have rubella virus infection. These five samples from patients with suspected rubella were appropriate for use in our study for the following two reasons. The outbreak of rubella in this area in April 2004 was confirmed by isolating rubella virus from patients. Also, the positive predictive value of case definition is considered to be high during outbreaks (11).
This study had some limitations, including the different criteria for the clinical diagnosis of rubella, the differences in timing and procedure for sample collection, and the limited number of clinical samples. However, at least 100 samples should be tested for the assessment of the procedure according to the review on the validation of nucleic acid-based tests published by Dimech et al. (7). The purpose of this study was to establish a rapid, sensitive, and simple method of RT-LAMP for the detection of rubella virus. The detection of the rubella virus genome by RT-LAMP can be confirmed using clinical samples from patients with rubella or CRS. We should further evaluate RT-LAMP for rubella diagnosis in the field.
During the progress of rubella control toward CRS elimination, the clinical application of RT-LAMP would be helpful for rapid diagnosis when managing patients or determining the response to an outbreak. A rapid response should be made to cope with outbreaks of rubella or CRS. RT-LAMP would contribute to the rapid diagnosis of index cases for the control of outbreaks. Methods for laboratory-confirmed diagnosis such as RT-LAMP will improve the surveillance of rubella and CRS by enabling the early detection of outbreaks and decreasing the risk of misclassification of cases that may occur with clinical diagnoses.
Acknowledgments
This study was supported in part by a grant from the 21st Century COE Program of the Ministry of Education, Culture, Sports, Science and Technology of Japan.
REFERENCES
- 1.Atkison, W., J. Hamborsky, and S. Wolfe. 2004. Rubella, p. 145-158. In W. Atkinson, J. Hamborsky, and S. Wolfe (ed.), Centers for Disease Control and Prevention. Epidemiology and prevention of vaccine-preventable diseases, 8th ed. Public Health Foundation, Washington, D.C.
- 2.Banatvala, J. E., and D. W. G. Brown. 2004. Rubella. Lancet 363:1127-1137. [DOI] [PubMed] [Google Scholar]
- 3.Bosma, T. J., K. M. Corbett, S. O'Shea, J. E. Banatvala, and J. M. Best. 1995. PCR for detection of rubella virus RNA in clinical samples. J. Clin. Microbiol. 33:1075-1079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Centers for Disease Control and Prevention. 2001. Control and prevention of rubella: evaluation and management of suspected outbreaks, rubella in pregnant women, and surveillance for congenital rubella syndrome. MMWR Recomm. Rep. 50(RR-12):1-23. [PubMed] [Google Scholar]
- 5.Chantler, J., J. S. Wolinsky, and A. Tingle. 2001. Rubella virus, p. 963-990. In D. M. Knipe and P. M. Howley (ed.), Fields virology, 4th ed. Lippincott Willians & Wilkins, Philadelphia, Pa.
- 6.Cooray, S., L. Warrener, and L. Jin. 2006. Improved RT-PCR for diagnosis and epidemiological surveillance of rubella. J. Clin. Virol. 35:73-80. [DOI] [PubMed] [Google Scholar]
- 7.Dimech, W., D. S. Bowden, B. Brestovac, K. Byron, G. James, D. Jardine, T. Sloots, and E. M. Dax. 2004. Validation of assembled nucleic acid-based tests in diagnostic microbiology laboratories. Pathology 36:45-50. [DOI] [PubMed] [Google Scholar]
- 8.Eggerding, F. A., J. Peters, R. K. Lee, and C. B. Inderlied. 1991. Detection of rubella virus gene sequences by enzymatic amplification and direct sequencing of amplified DNA. J. Clin. Microbiol. 29:945-952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Falsey, A. R., M. A. Formica, J. J. Treanor, and E. E. Walsh. 2003. Comparison of quantitative reverse transcription-PCR to viral culture for assessment of respiratory syncytial virus shedding. J. Clin. Microbiol. 41:4160-4165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fujino, M., N. Yoshida, S. Yamaguchi, N. Hosaka, Y. Ota, T. Notomi, and T. Nakayama. 2005. A simple method for the detection of measles virus genome by loop-mediated isothermal amplification (LAMP). J. Med. Virol. 76:406-413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gordis, L. 2004. Assessing the validity and reliability of diagnostic and screening tests, p. 71-940. In L. Gordis (ed.), Epidemiology, 3rd ed. Elsevier Saunders, Philadelphia, Pa.14712149
- 12.Gueudin, M., A. Vabret, J. Petitjean, S. Gouarin, J. Brouard, and F. Freymuth. 2003. Quantitation of respiratory syncytial virus RNA in nasal aspirates of children by real-time RT-PCR assay. J. Virol. Methods 109:39-45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ho-Terry, L., G. M. Terry, and P. Londesborough. 1990. Diagnosis of foetal rubella virus infection by polymerase chain reaction. J. Gen. Virol. 71:1607-1611. [DOI] [PubMed] [Google Scholar]
- 14.Kubar, A., M. Yapar, B. Besirbellioglu, I. Y. Avci, and C. Guney. 2004. Rapid and quantitative detection of mumps virus RNA by one-step real-time RT-PCR. Diagn. Microbiol. Infect. Dis. 49:83-88. [DOI] [PubMed] [Google Scholar]
- 15.Macé, M., D. Cointe, C. Six, D. Levy-Bruhl, I. P. du Châtelet, D. Ingrand, and L. Grangeot-Keros. 2004. Diagnostic value of reverse transcription-PCR of amniotic fluid for prenatal diagnosis of congenital rubella infection in pregnant women with confirmed primary rubella infection. J. Clin. Microbiol. 42:4818-4820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mori, Y., M. Kitao, N. Tomita, and T. Notomi. 2004. Real-time turbidimetry of LAMP reaction for quantifying template DNA. J. Biochem. Biophys. Methods 59:145-157. [DOI] [PubMed] [Google Scholar]
- 17.Nagamine, K., K. Watanabe, K. Ohtsuka, T. Hase, and T. Notomi. 2001. Loop-mediated isothermal amplification reaction using a nondenatured template. Clin. Chem. 47:1742-1743. [PubMed] [Google Scholar]
- 18.Nagamine, K., T. Hase, and T. Notomi. 2002. Accelerated reaction by loop-mediated isothermal amplification using loop primers. Mol. Cell. Probes 16:223-229. [DOI] [PubMed] [Google Scholar]
- 19.Notomi, T., H. Okayama, H. Masubuchi, T. Yonekawa, K. Watanabe, N. Amino, and T. Hase. 2000. Loop-mediated isothermal amplification of DNA. Nucleic Acids Res. 28:e63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Okafuji, T., N. Yoshida, Y. Motegi, T. Ihara, Y. Ota, and T. Notomi, and T. Nakayama. 2005. Rapid diagnostic method for detection of mumps virus genome by loop-mediated isothermal amplification. J. Clin. Microbiol. 43:1625-1631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ozoenmena, L. C., P. D. Minor, and M. A. Afzal. 2004. Comparative evaluation of measles virus specific TaqMan PCR and conventional PCR using synthetic and natural RNA templates. J. Med. Virol. 73:79-84. [DOI] [PubMed] [Google Scholar]
- 22.Plotkin, S. A., and S. Reef. 2004. Rubella vaccine, p. 707-743. In S. A. Plotkin and W. A. Orenstein (ed.), Vaccine, 4th ed. W. B. Saunders, Philadelphia, Pa.
- 23.Schalk, J. A. C., C. van den Elzen, H. Ovelgönne, C. Baas, and P. M. J. Jongen. 2004. Estimation of the number of infectious measles viruses in live virus vaccines using quantitative real-time PCR. J. Virol. Methods 117:179-187. [DOI] [PubMed] [Google Scholar]
- 24.Thomas, H. I. J., E. Barrett, L. M. Hesketh, A. Wynne, and P. Morgan-Capner. 1999. Simultaneous IgM reactivity by EIA against more than one virus in measles, parvovirus B19 and rubella infection. J. Clin. Virol. 14:107-118. [DOI] [PubMed] [Google Scholar]
- 25.Tipple, G. A., R. Hamkar, T. Mohktari-Azad, M. Gray, J., Ball, C. Head, and S. Ratnam. 2004. Evaluation of rubella IgM enzyme immunoassays. J. Clin. Virol. 30:233-238. [DOI] [PubMed] [Google Scholar]
- 26.Tzeng, W.-P., Y. Zhou, J. Icenogle, and T. K. Frey. 2005. Novel replicon-based reporter gene assay for detection of rubella virus in clinical specimens. J. Clin. Microbiol. 43:879-885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ushio, M., I. Yui, N. Yoshida, M. Fujino, T. Yonekawa, Y. Ota, T. Notomi, and T. Nakayama. 2005. Detection of respiratory syncytial virus genome by subgroups-A, B specific reverse transcription loop-mediated isothermal amplification (RT-LAMP). J. Med. Virol. 77:121-127. [DOI] [PubMed] [Google Scholar]
- 28.Watson, J. C., S. C. Hadler, C. A. Dykewicz, S. Reef, and L. Phillips. 1998. Measles, mumps, and rubella—vaccine use and strategies for elimination of measles, rubella, and congenital rubella syndrome and control of mumps:recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm. Rep. 47(RR-8):1-57. [PubMed] [Google Scholar]
- 29.World Health Organization. 1999. Guidelines for surveillance of congenital rubella syndrome and rubella. WHO/V&B/99.22. [Online.] http://www.who.int/gpv-documents/.
- 30.World Health Organization. 2005. Standardization of the nomenclature for genetic characteristics of wild-type rubella viruses. Wkly. Epidemiol. Rec. 80:125-132. [PubMed] [Google Scholar]
- 31.World Health Organization. 2000. Rubella vaccines. WHO position paper. Wkly. Epidemiol. Rec. 75:161-172. [Google Scholar]
- 32.World Health Organization. 2005. Global measles and rubella laboratory network—update. Wkly. Epidemiol. Rec. 80:384-388. [PubMed] [Google Scholar]
- 33.Zheng, D., T. K. Frey, J. Icenogle, S. Katow, S. Abernathy, K. Song, W. Xu, V. Yarullin, R. G. Desjats, Y. Aboudy, G. Enders, and M. Croxson. 2003. Global distribution of rubella virus genotypes. Emerg. Infect. Dis. 9:1523-1530. [DOI] [PMC free article] [PubMed] [Google Scholar]