Abstract
Previously, we established an intranasal mouse model of Klebsiella pneumoniae infection and validated its utility using a highly virulent wild-type strain and an avirulent capsular polysaccharide mutant. In the present study we compare the host responses to both infections by examining cytokine production, cellular infiltration, pulmonary histology, and intranasal immunization.
Klebsiella pneumoniae has been known as an important cause of gram-negative bacterial pneumonia for more than 100 years. The best-characterized virulence factor of this species is capsular polysaccharide, a voluminous layer of acidic sugar polymers responsible for a variety of K. pneumoniae phenotypes, including protection against complement-mediated killing and inhibition of macrophage phagocytosis (6, 11). Capsule has also been investigated using a number of different animal models for its importance in K. pneumoniae pathogenesis. Isogenic capsule mutant strains are deficient for causing disease in mice via intraurethral and intratracheal inoculation routes (6, 24).
A substantial amount of research over the last 20 years has focused on the importance of cytokine production in the host defense against K. pneumoniae pulmonary infection. Nearly all of these studies have examined infection of a wild-type strain after intratracheal inoculation and compared outcomes in both wild-type and immunodeficient mice (23, 26). Among the host factors that have been investigated include tumor necrosis factor alpha (TNF-α), interleukin-12 (IL-12), IL-17, and macrophage inflammatory protein 1α (MIP-1α) (12, 29). These experiments have helped to reinforce the importance of the inflammatory response in clearing K. pneumoniae infection and have provided substantial evidence for the protective role of a TH1-mediated response. Any interference with a rapid host response (e.g., suppression of the proinflammatory cytokines) leads to a more severe disease process (16). Conversely, augmenting the immune response with exogenous inflammatory mediators decreases the morbidity and mortality associated with infection (23).
Despite the importance of capsular polysaccharide for the pathogenesis of K. pneumoniae, few studies have compared the host responses against wild-type and well-defined capsule mutant strains. In examining the previous literature for in vivo studies of capsule mutant infections, only a few of these have included immunological assays as part of their comparison (Table 1) . Many of these studies feature one or more deficiencies, including the use of spontaneous capsule mutant strains, choosing few time points for comparison, or using atypical inoculation routes. Two studies in particular have detailed the difference in cytokine production between a wild-type infection and one caused by a spontaneous capsule-deficient strain and examined the production of a number of cytokines during each infection (30, 32). Similar levels of production of TNF-α, IL-1β, and IL-6 were seen in both bronchoalveolar lavage (BAL) and serum samples at most time points in both infected groups. Interestingly, the level of IL-6 production was significantly higher in BAL fluid from capsule mutant infected mice than from wild-type infected mice (32). Further investigation demonstrated that the capsule-deficient strain induced an early peak of IFN-γ production that was lacking in the wild-type infected mice (30). Instead, the wild-type infection induced higher levels of IL-10 production, and it was postulated that capsule serves to induce the production of IL-10, which helps to suppress the host inflammatory response and allow the bacteria to grow in a more permissive environment.
TABLE 1.
Comparison of in vivo studies with capsule mutants
| Type of cps mutationa | Capsule productionb | Infection routec | Tissue(s); time point(s)d | LD50e | Immunological assay(s)f | Reference |
|---|---|---|---|---|---|---|
| UV, HS | Precipitin | i.p. | None | Y | None | 1 |
| Sp | Cell vol | i.p. | None | Y | None | 25 |
| Sp | India ink | i.t. | Lung; 1-28 days | Y | Lung histology | 10 |
| Sp | None | Burn | Skin, blood, liver; 12 h-6 days | Y | None | 8 |
| Sp | Swelling | s.c. | Skin abscess; 24 h | N | None | 22 |
| UV | ELISA | i.u. | Bladder, urine, kidney; 5 days | N | None | 2 |
| Tn | Immunoblot | i.p. | None | Y | None | 3 |
| In | Uronic acid | i.g. | Intestine; 1-20 days | N | Intestine histology | 11 |
| Sp | India ink | i.n. | Lung, serum; 6-72 h | Y | Lung histology, BAL, and serum cytokines | 32 |
| Sp | India ink | i.n. | None | N | BAL and serum cytokines | 31 |
| In | ELISA | i.t. | Lung; 7 days | N | Lung histology | 6 |
| Sp | Hydrophobicity | i.t. | Lung; 4-14 days | N | Lung histology | 4 |
| Sp | Swelling | i.g., i.u. | Intestine, bladder; 1-10 days | N | None | 24 |
| Tn | Uronic acid | i.n. | Trachea, lung, spleen; 12-72 h | Y | Lung histology | 18 |
The type of capsule mutation used in the study. UV, induced by UV light; Sp, spontaneous mutation; Tn, transposon insertion mutant; In, insertional mutation; HS, induced by high salt concentrations.
The method used to monitor capsule production. ELISA, enzyme-linked immunosorbent assay.
The route of inoculation for the animal studies. i.p., intraperitoneal; s.c., subcutaneous; i.u., intraurethral; i.g., intragastric; i.n., intranasal; i.t., intratracheal.
The types of tissues examined and the time frame of the study.
Whether the LD50 was either determined in the study. Y, yes; N, no.
The type of immunological assay(s) (if any) performed in the study.
Our initial experiments in the establishment of this intranasal model demonstrated that a cpsB mutant of K. pneumoniae was severely attenuated in its ability to cause disease (18). This cpsB mutant (VK20) appears to be wild type in its production of lipopolysaccharide (LPS) and enterobacterial common antigen, two other surface polysaccharides of K. pneumoniae (18). All infected mice survived intranasal challenge with as many as 9 × 108 CFU of the cpsB mutant strain. Furthermore, when inoculated at a dose equivalent to the wild-type 50% lethal dose (LD50), 104 CFU, the cpsB mutant bacteria were cleared from all tissues by 48 h. All future infections were performed with doses that would result in similar concentrations of bacteria in the lungs of infected mice at the 12-h time point, allowing for a more relevant comparison between infections (∼107 CFU of the cpsB mutant strain or ∼104 CFU of the wild-type KPPR1 strain) (18). However, bacterial concentrations in the lungs increasingly diverge after the 12-h time point, with the wild-type infection peaking at 72 h postinfection with ensuing lethality. In contrast, the cpsB mutant is unable to grow in either the trachea or lungs and fails to disseminate to the spleen. However, small numbers of this attenuated strain can persist in the lungs for several days (18).
Despite past achievements in elucidating the host response to K. pneumoniae infection, we believe that our comprehensive model can be used in concert with our defined capsule mutant to bring further insight to this interesting area of research. In the present study we describe the use of the intranasal mouse model to study the host response to K. pneumoniae infection by either a wild-type or a capsule mutant strain. Comparing the weights of infected tissues provided a broad comparison of inflammation during each infection. Cytokine quantitation using both RNA and protein measurements allowed a more detailed examination of differences in the host responses to these infections. The use of fluorescence-activated cell sorting (FACS) analysis and histology to examine infected lungs provides an insight into the different cell types recruited during each infection. Finally, we evaluated the efficacy of the capsule mutant strain in generating protective immunity against a wild-type infection.
Gross measures of inflammation.
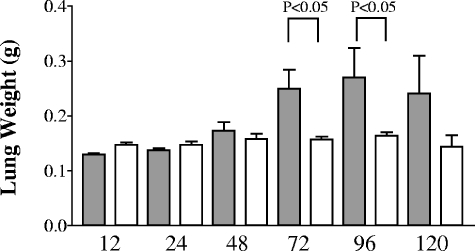
One of the most common methods to determine gross inflammation is to examine the weight of infected tissues. At early time points after intranasal inoculation, no differences were seen when whole lung and spleen weights were compared between wild-type- and cpsB mutant-infected mice (Fig. 1). However, at later time points the wild-type bacteria induced a substantial inflammatory response that was clearly lacking in the mutant infected tissue, and this was reflected in significant differences in lung weights at the 72- and 96-h time points. Interestingly, there were no differences in spleen weights at any time during infection (data not shown).
FIG. 1.
Weights of lungs infected by K. pneumoniae. C57BL/6 mice were intranasally infected with either 104 CFU of the wild-type KPPR1 strain or 107 CFU of the cpsB mutant strain as described previously (18). Lungs were harvested from five mice per group per time point, with wild-type results indicated in gray and cpsB mutant results indicated in white. Mean weights with the standard deviations are shown. P values were determined by using a one-way analysis of variance statistical test.
Inflammatory cytokine production.
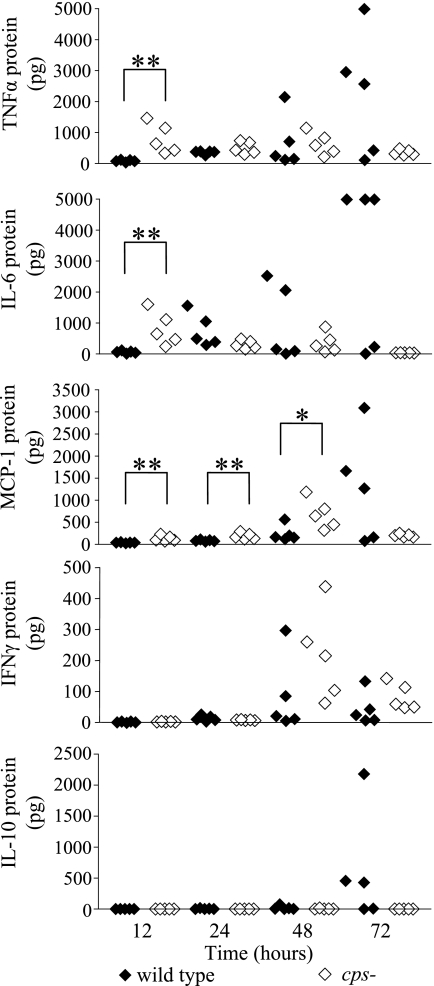
To further characterize the host response to Klebsiella-induced pneumonia, we measured the production of several cytokines in both lungs and serum during infection by using a mouse inflammation cytometric bead array kit (BD Biosciences, San Diego, CA). Several interesting trends were noted. High levels of cytokine production were not apparent until at least 48 h after inoculation, even among wild-type-infected lung samples (Fig. 2 and data not shown). The intensity of this inflammatory response tightly correlated with the severity of infection, since those tissues with the highest bacterial counts also had the most significant cytokine production (Table 2). Examinations of cytokine production during Streptococcus pneumoniae infection show a similar trend, where only mice that become visibly sick show an increase of inflammatory cytokines in the lung, including MCP-1, MIP-2, and IL-6 (9). The production of these cytokines remains relatively low until 48 h postinfection, and only reach high levels at 72 h and later. However, since most studies evaluating cytokine responses in the lung use different methods of quantitation, it is difficult to directly compare data between publications.
FIG. 2.
Cytokine production in K. pneumoniae-infected mice. Mice were infected with either wild-type or cpsB mutant bacteria as described in Fig. 1. At each time point mice from each group were sacrificed, and whole lung homogenates and blood samples were quantified for cytokine production by using a mouse inflammation cytometric bead array kit (BD Biosciences, San Diego, CA). The data are shown for five mice per time point. The upper limit of detection for this assay is 5,000 pg per sample. *, P < 0.05; **, P < 0.001 (unpaired Mann-Whitney t test).
TABLE 2.
High levels of proinflammatory cytokines in the lungs correlate directly with the bacterial concentrationa
| Mouse | Bacterial load in wild type (log10 CFU/g of tissue) | Bacterial concn (pg/sample)b
|
||
|---|---|---|---|---|
| TNF-α | MCP-1 | IL-6 | ||
| 1 | 5.50 | 430.2 | 163.4 | 231.1 |
| 2 | 4.59 | 128.8 | 87.8 | 25.3 |
| 3 | 9.78 | 2,961.3 | 1,666.8 | 5,000.0 |
| 4 | 9.72 | 2,576.4 | 1,268.1 | 5,000.0 |
| 5 | 10.62 | 5,000.0 | 3,093.2 | 5,000.0 |
| Mean ± SD | 8.04 ± 2.78 | 2,219.3 ± 1,998.8 | 1,255.9 ± 1,235.2 | 3,051.3 ± 2,669.4 |
Mice were infected with 104 CFU of wild-type bacteria, and lungs were harvested at 72 h. Data are taken from the experiment described in Fig. 2.
Correlations: TNF-α, r2 = 0.8791 and P = 0.0185; MCP-1, r2 = 0.8008 and P = 0.0403; IL-6, r2 = 0.9757 and P = 0.0016.
Previous reports suggested that the K. pneumoniae capsule serves to suppress the innate inflammatory response, and our data support this model (31, 32). At early time points the cytokines examined were all expressed at very low levels until the 48-h time point, and IL-10 was not observed until 72 h (Fig. 2). However, the lack of an early cytokine response against a wild-type infection may be a consequence of the ability of the capsule to inhibit phagocytosis and therefore inhibit subsequent processing by antigen-presenting cells. This reduction in antigen presentation would lead to a relative reduction in T- and B-cell stimulation and an overall reduction in the number of activated immune cells that could produce proinflammatory cytokines; the capsule may be working indirectly in this case to dampen the intensity of the inflammatory response. A complementary function of the capsule appears to lie in protecting the bacterium from this attack; even when the host mounts an intense inflammatory response late in infection, wild-type K. pneumoniae continues to multiply in the lungs. This confluence of capsule-mediated phenotypes only exacerbates the destructive impact of this infection to the host and further retards the effectiveness of the immune response.
The most interesting aspect of the cpsB mediated cytokine response was an altered production of IFN-γ. At the 48- and 72-h time points, there was a higher level of IFN-γ production in cpsB-infected lungs than during wild-type infection (Fig. 2). IFN-γ production in wild-type-infected mice did not reach the quantities found in cpsB mutant-infected tissues at any time point. IFN-γ has been shown to play different roles when K. pneumoniae is inoculated by different routes (20). IFN-γ knockout mice suffered greater mortality after intratracheal infection, with a higher bacterial burden in the lungs than wild-type mice. However, the knockout mice showed rates of survival at least as high if not higher than wild-type mice after intravenous K. pneumoniae inoculation. The authors of that study concluded that localized pulmonary infection requires IFN-γ whereas systemic infection does not. Intranasal studies have also demonstrated that mice deficient in IFN-γ production show a larger bacterial burden and suffer greater mortality from wild-type Klebsiella infection (30). The higher levels of IFN-γ that are produced during the cpsB mutant infection are likely a result of the high rate of phagocytosis and clearance of the cpsB mutant strain.
Between 12 and 48 h postinfection, significantly higher concentrations of TNF-α, MCP-1, and IL-6 were produced in cpsB mutant infected lungs compared to the wild-type infection (Fig. 2). This difference may be attributable to the 1,000-fold difference between these bacterial inocula (Fig. 2). Additional experiments with the KPPR1 strain inoculated at 107 CFU demonstrated a concomitant increase in TNF-α and MCP-1 production to the levels seen in cpsB mutant-infected lungs (data not shown). Mice lacking MCP-1α (CCL3−/−) have been shown to survive infection less well than wild-type mice and have increased bacterial burden in their lungs (19). These mice do not appear to be defective for recruiting monocytes, macrophages, or neutrophils, and they produce normal levels of cytokines in response to Klebsiella infection. However, using in vitro assays alveolar macrophages from the CCL3−/− mice were found to be defective for the phagocytosis of opsonized Klebsiella, which may account for the uncontrolled growth observed in the lungs and decreased survival (19). In addition to directly measuring cytokine levels in tissue, we also monitored the transcript level of these same cytokines from the lung tissue by using quantitative reverse transcription-PCR (qRT-PCR) (Table 3). In general, similar trends were observed.
TABLE 3.
Fold change in cytokine and chemokine transcript level as measured by qRT-PCR from whole lungsa
| Time postinfection (h) | Avg fold change
|
|||||||
|---|---|---|---|---|---|---|---|---|
| IL-10
|
MCP-1
|
TNF-α
|
IFN-γ
|
|||||
| WT | cps | WT | cps | WT | cps | WT | cps | |
| 12 | 18.6 | 2.8 | 1.0 | 0.0 | 5.7 | −1.3 | 1.8 | 1.4 |
| 24 | 7.9 | 1.8 | 0.0 | 23.8 | 6.9 | −2.3 | 1.3 | 1.8 |
| 48 | 172.4 | 5.0 | 5.8 | −2.8 | 24.6 | 1.0 | 4.0 | 3.5 |
| 72 | 1,379.6 | 3.2 | −3.8 | −4.1 | 38.6 | −1.4 | 4.4 | 1.7 |
Mice were infected with either wild-type (WT) or cps mutant K. pneumoniae as described in Fig. 1, and samples were obtained at 12, 24, 48, and 72 h postinfection. At each time point, mice were euthanized, and both lungs were removed and stored in RNAlater (Ambion, Austin, TX). Total nucleic acid was extracted by using TRIzol reagent (Invitrogen, Carlsbad, CA) according to the manufacturer's instructions. The total nucleic acid was treated twice with DNase (Fisher Scientific, Pittsburgh, PA) at 37°C to remove contaminating DNA. cDNA synthesis and qRT-PCRs were performed as described previously (13) Each value represents the average fold change obtained from three independent reactions performed on three individual mice relative to samples from uninfected mice. A “0” indicates no change, and a negative value indicates a reduction in transcript compared to the baseline. The primers used for each reaction are shown in Table 4. Reactions were also run for IL-6 and IL-17; however, because there was no background signal for the uninfected control the fold change could not be calculated. Nevertheless, from these reactions it was clear that for IL-6 there was an induction in mice infected with the wild type but not the cps mutant, and for IL-17 there was an induction in mice infected with either strain.
Cellular response to infection.
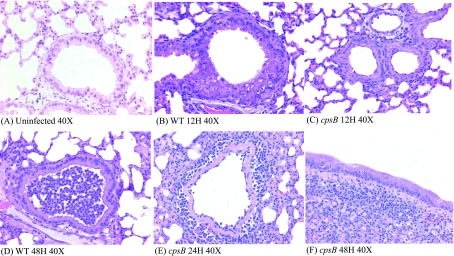
Earlier histological studies showed some dramatic differences in the inflammatory cell response over the course of wild-type infection (18). We followed up on these experiments by comparing histological samples from both wild-type- and cpsB mutant-infected lungs. The differences in pulmonary epithelial cell responses are dramatic even at an early 12-h time point (Fig. 3A to C). Although capsule mutant-infected lungs show some hypertrophy of epithelial cells compared to an uninfected sample, the wild-type-infected lungs show dramatic cellular changes in the epithelium, including hypertrophy and cytoplasmic clearance, that is reflective of mucus production by these cells. Interestingly, histological analysis of cpsB mutant-infected lungs indicates large areas of lymphocyte infiltration that are absent in wild-type-infected lungs (Fig. 3E and F). The higher concentration of lymphocytes in cpsB mutant-infected lungs could also be a consequence of either a loss of lymphocytes during wild-type infection or an influx of lymphocytes induced by the high rate of clearance of the cpsB mutant bacteria. The incredibly high concentration of neutrophils in wild-type lungs are evidenced by histology, with neutrophils present in many infected airways (Fig. 3D). By comparison few neutrophils can be seen in the airways of cpsB-infected mice, but in these infections localized populations of lymphocytes can be seen (Fig. 3E and F).
FIG. 3.
Histological examination of K. pneumoniae-infected lungs. Mice were infected with either wild-type or cpsB mutant bacteria as described in Fig. 1. At various time points after infection, mice were sacrificed, and whole lungs were removed and processed for paraffin embedding and sectioning as described previously (18). Samples were stained with hematoxylin and eosin, and all pictures shown were taken at ×40 magnification. (A) Uninfected lung; (B) wild-type infection at 12 h; (C) cpsB mutant infection at 12 h; (D) wild-type infection at 48 h; (E) cpsB mutant infection at 24 h; (F) cpsB mutant infection at 48 h.
After these experiments FACS analysis of whole lungs was performed to determine in a more quantitative fashion what types of cells are responding to K. pneumoniae intranasal infection (Table 5). FACS assays of whole lung tissues were performed by using previously established protocols (14, 21). As expected, at 48 h postinfection wild-type-infected lungs showed dramatically higher levels of Gr-1-positive cells (granulocytes) and Mac-1-positive cells (neutrophils and myeloid cells) than the cpsB mutant-infected lungs. At this time point wild-type-infected lungs were comprised of more than 70% neutrophils, whereas cpsB mutant-infected lungs contained only ca. 4%. Although the histological results show an apparent increase in lymphocyte populations in cpsB mutant-infected lungs, the FACS data suggest that the absolute number of B and T lymphocytes in cpsB mutant-infected lungs is very similar to that from uninfected mice. However, there may still be a redistribution of these cells during cpsB infection that could impact bacterial clearance. In addition, it is unclear whether the difference seen via FACS is a result of the large influx of neutrophils during wild-type infection that may skew these percentage results.
TABLE 5.
FACS analysis of whole lung samples at 48 h postinfectiona
| Antibody marker | Cell type | Uninfected animal (%) | Mean % ± SD
|
|
|---|---|---|---|---|
| Wild-type mice | cpsB mutant mice | |||
| Gr-1 | Granulocytes | 2.42 | 71.55 ± 8.1 | 3.87 ± 1.72 |
| Mac-1 | Neutrophils/ myeloid cells | 10.64 | 73.58 ± 8.1 | 10.96 ± 2.8 |
| CD19 | B cells | 8.93 | 4.75 ± 4.1 | 9.37 ± 1.69 |
| TCR/CD4 | CD4+ T cells | 18.91 | 4.45 ± 1.2 | 21.7 ± 4.1 |
| TCR/CD8 | CD8+ T cells | 21.69 | 5.48 ± 2.4 | 22.39 ± 4.1 |
Three mice each were infected with either 104 CFU of the wild-type strain or 107 CFU of the cpsB mutant strain as described previously (18). At 48 h postinfection, whole lungs were taken, processed into single cell suspensions, and evaluated via FACS analysis with several different antibodies to identify different immune cell populations. Antibody markers are listed, along with predominant immune cell type that was targeted. Mean percentages of total cell populations are presented with the standard deviations. The data from the lungs of one uninfected mouse are presented for comparison.
Immunization studies.
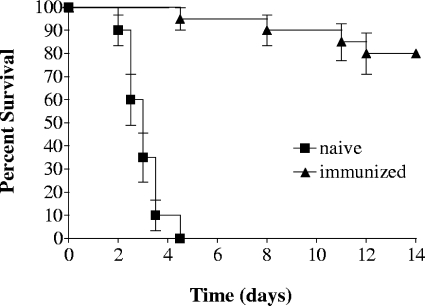
To explore whether immunization with the cpsB mutant can provide protection against a wild-type infection, mice were intranasally inoculated with 9 × 107 CFU of the cpsB mutant strain. Six weeks later, both immunized and age-matched naive mice were challenged with 1.3 × 106 CFU of KPPR1; this dose is ∼500-fold greater than the wild-type LD50 value (18). All naive mice succumbed to infection by 5 days postinoculation, whereas 80% of the immunized mice survived (Fig. 4). This result indicates that noncapsular Klebsiella antigens can stimulate a protective memory immune response. Moreover, it also suggests that noncapsular antigens can serve as targets to generate an effective defense against infection by a capsule-positive strain.
FIG. 4.
Protection against KPPR1 infection by immunization with the cpsB mutant strain. n = 20 mice per group; P < 0.0001 (log-rank test). The data are representative of two independent experiments.
As an immunizing agent, purified K. pneumoniae capsule has been shown to protect against subsequent infection with wild-type bacteria (7). In addition, other noncapsular epitopes have been used to immunize against K. pneumoniae infection, including LPS and type 3 fimbriae (5, 17, 28). In our immunization study we found that an acapsular K. pneumoniae mutant can also protect against wild-type infection. This finding correlates with results from other pathogens, including a porcine model of Actinobacillus pleuropneumoniae infection, wherein inoculation with an isogenic acapsular mutant was shown to be protective against subsequent wild-type infection (15). Our results suggest that noncapsular epitopes of wild-type K. pneumoniae are still accessible to the immune system as effective targets despite the presence of capsule and can be targeted by the adaptive immune response in immunized mice. Alternatively, there may be a time during infection when capsule production is suppressed, allowing access to noncapsular epitopes. Future work to examine the expression of bacterial genes during each stage of infection is an important future goal, and analyzing sera from cpsB immunized mice may aid in discovering the accessible epitopes. One likely target is the LPS O antigen, which has been shown through previous microscopic studies to protrude through some K. pneumoniae capsular types, including the serotype K2 capsule that this wild-type strain expresses (27). If the O antigen is the target of the memory response, it would be interesting to immunize mice with a bacterium deficient in both components and observe whether other bacterial surface components will initiate an effective memory response. However, earlier studies indicate that a capsule-negative, O antigen-negative strain is highly susceptible to rapid clearance after intranasal inoculation, and it may prove difficult to maintain a sufficient bacterial dose in vivo to stimulate a memory response (18).
TABLE 4.
Primer sequences used for qRT-PCR analysis
| Product | Primer sequence (5′-3′)
|
|
|---|---|---|
| Forward | Reverse | |
| GAPDHa | TGG CAA AGT GGA GAT TGT TGC C | AAG ATG GTG ATG GGC TTC CCG |
| IL-6 | CTG ATG CTG GTG ACA ACC AC | AGC CTC CGA CTT GTG AAC TG |
| IL-10 | TGG GTG AGA AGC TGA AGA CC | TGG CCT TGT AGA CAC CTT GG |
| MCP-1 | GGA CAG ATG TGG TGG GTT TC | GGG TGA CAG TGA TTT CTC TTC C |
| IL-12 | CAG TAC ACC TGC CAC AAA GG | TTG GTG CTT CAC ACT TCA GG |
| TNF-α | GTC CCC AAA GGG ATG AGA AG | TTT GCT ACG ACG TGG GCT AC |
| IFN-γ | CAC GGC ACA GTC ATT GAA AG | CAT CCT TTT GCC AGT TCC TC |
| IL-17 | TAC CTC AAC CGT TCC ACG TC | AGC TTC CCA GAT CAC AGA GG |
GAPDH, glyceraldehyde-3-phosphate dehydrogenase.
Editor: J. B. Bliska
REFERENCES
- 1.Baer, H., and L. Ehrenworth. 1956. The pathogenicity of Klebsiella pneumoniae for mice: the relationship to the quantity and rate of production of type-specific capsular polysaccharide. J. Bacteriol. 72:713-717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Camprubi, S., S. Merino, V. J. Benedi, and J. M. Tomas. 1993. The role of the O-antigen lipopolysaccharide and capsule on an experimental Klebsiella pneumoniae infection of the rat urinary tract. FEMS Microbiol. Lett. 111:9-13. [DOI] [PubMed] [Google Scholar]
- 3.Chang, H.-Y., J. H. Lee, W. L. Deng, T. F. Fu, and H. L. Peng. 1996. Virulence and outer membrane properties of a galU mutant of Klebsiella pneumoniae CG43. Microb. Pathog. 20:255-261. [DOI] [PubMed] [Google Scholar]
- 4.Chhibber, S., S. Aggarwal, and V. Yadav. 2003. Contribution of capsular and lipopolysaccharide antigens to the pathogenesis of Klebsiella pneumoniae respiratory tract infection. Folia Microbiol. 48:699-702. [DOI] [PubMed] [Google Scholar]
- 5.Chhibber, S., S. Wadhwa, and V. Yadav. 2004. Protective role of liposome incorporated lipopolysaccharide antigen of Klebsiella pneumoniae in a rat model of lobar pneumonia. Jpn. J. Infect. Dis. 57:150-155. [PubMed] [Google Scholar]
- 6.Cortes, G., N. Borrell, B. de Astorza, C. Gomez, J. Sauleda, and S. Alberti. 2002. Molecular analysis of the contribution of the capsular polysaccharide and the lipopolysaccharide O side chain to the virulence of Klebsiella pneumoniae in a murine model of pneumonia. Infect. Immun. 70:2583-2590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cryz, S. J., Jr., E. Furer, and R. Germanier. 1986. Immunization against fatal experimental Klebsiella pneumoniae pneumonia. Infect. Immun. 54:403-407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cryz, S. J., Jr., E. Furer, and R. Germanier. 1984. Prevention of fatal experimental burn-wound sepsis due to Klebsiella pneumoniae KP1-O by immunization with homologous capsular polysaccharide. J. Infect. Dis. 150:817-822. [DOI] [PubMed] [Google Scholar]
- 9.Dallaire, F., N. Ouellet, Y. Bergeron, V. Turmel, M. C. Gauthier, M. Simard, and M. G. Bergeron. 2001. Microbiological and inflammatory factors associated with the development of pneumococcal pneumonia. J. Infect. Dis. 184:292-300. [DOI] [PubMed] [Google Scholar]
- 10.Domenico, P., W. G. Johanson, Jr., and D. C. Straus. 1982. Lobar pneumonia in rats produced by clinical isolates of Klebsiella pneumoniae. Infect. Immun. 37:327-335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Favre-Bonte, S., T. R. Licht, C. Forestier, and K. A. Krogfelt. 1999. Klebsiella pneumoniae capsule expression is necessary for colonization of large intestines of streptomycin-treated mice. Infect. Immun. 67:6152-6156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Greenberger, M. J., S. L. Kunkel, R. M. Strieter, N. W. Lukacs, J. Bramson, J. Gauldie, F. L. Graham, M. Hitt, J. M. Danforth, and T. J. Standiford. 1996. IL-12 gene therapy protects mice in lethal Klebsiella pneumonia. J. Immunol. 157:3006-3012. [PubMed] [Google Scholar]
- 13.Handley, S. A., P. H. Dube, and V. L. Miller. 2006. Histamine signaling through the H2 receptor in the Peyer's patch is important for controlling Yersinia enterocolitica infection. Proc. Natl. Acad. Sci. USA 103:9268-9273. [DOI] [PMC free article] [PubMed]
- 14.Handley, S. A., P. H. Dube, P. A. Revell, and V. L. Miller. 2004. Characterization of oral Yersinia enterocolitica infection in three different strains of inbred mice. Infect. Immun. 72:1645-1656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Inzana, T., J. Todd, and H. Veit. 1993. Safety, stability, and efficacy of noncapsulated mutants of Actinobacillus pleuropneumoniae for use in live vaccines. Infect. Immun. 61:1682-1686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Laichalk, L. L., S. L. Kunkel, R. M. Strieter, J. M. Danforth, M. B. Bailie, and T. J. Standiford. 1996. Tumor necrosis factor mediates lung antibacterial host defense in murine Klebsiella pneumonia. Infect. Immun. 64:5211-5218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lavender, H. F., J. R. Jagnow, and S. Clegg. 2004. Biofilm formation in vitro and virulence in vivo of mutants of Klebsiella pneumoniae. Infect. Immun. 72:4888-4890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lawlor, M. S., J. Hsu, P. D. Rick, and V. L. Miller. 2005. Identification of Klebsiella pneumoniae virulence determinants using an intranasal infection model. Mol. Microbiol. 58:1054-1073. [DOI] [PubMed] [Google Scholar]
- 19.Lindell, D. M., T. J. Standiford, P. Mancuso, Z. Leshen, and G. B. Huffnagle. 2001. Macrophage inflammatory protein 1a/CCL3 is required for clearance of an acute Klebsiella pneumoniae pulmonary infection. Infect. Immun. 69:6364-6369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Moore, T. A., M. L. Perry, A. G. Getsoian, M. W. Newstead, and T. J. Standiford. 2002. Divergent role of gamma interferon in a murine model of pulmonary versus systemic Klebsiella pneumoniae infection. Infect. Immun. 70:6310-6318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Scott, H. M., and J. L. Flynn. 2002. Mycobacterium tuberculosis in chemokine receptor 2-deficient mice: influence of dose on disease progression. Infect. Immun. 70:5946-5954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Simoons-Smit, A. M., A. M. Verweij-van Vught, and D. M. MacLaren. 1986. The role of K antigens as virulence factors in Klebsiella. J. Med. Microbiol. 21:133-137. [DOI] [PubMed] [Google Scholar]
- 23.Standiford, T. J., J. M. Wilkowski, T. H. Sisson, N. Hattori, B. Mehrad, K. A. Bucknell, and T. A. Moore. 1999. Intrapulmonary tumor necrosis factor gene therapy increases bacterial clearance and survival in murine gram-negative pneumonia. Hum. Gene Ther. 10:899-909. [DOI] [PubMed] [Google Scholar]
- 24.Struve, C., and K. A. Krogfelt. 2003. Role of capsule in Klebsiella pneumoniae virulence: lack of correlation between in vitro and in vivo studies. FEMS Microbiol. Lett. 218:149-154. [DOI] [PubMed] [Google Scholar]
- 25.Takahashi, M., K. Yoshida, and C. L. San Clemente. 1977. Relation of colonial morphologies in soft agar to morphologies and biological properties of the K-9 strain of Klebsiella pneumoniae and is variants. Can. J. Microbiol. 23:448-451. [DOI] [PubMed] [Google Scholar]
- 26.Tanabe, M., T. Matsumoto, K. Shibuya, K. Tateda, S. Miyazaki, A. Nakane, Y. Iwakura, and K. Yamaguchi. 2005. Compensatory response of IL-1 gene knockout mice after pulmonary infection with Klebsiella pneumoniae. J. Med. Microbiol. 54:7-13. [DOI] [PubMed] [Google Scholar]
- 27.Tomas, J. M., S. Camprubi, S. Merino, M. R. Davey, and P. Williams. 1991. Surface exposure of O1 serotype lipopolysaccharide in Klebsiella pneumoniae strains expressing different K antigens. Infect. Immun. 59:2006-2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Yadav, V., S. Sharma, K. Harjai, H. Mohan, and S. Chibber. 2005. Lipopolysaccharide-mediated protection against Klebsiella pneumoniae-induced lobar pneumonia: intranasal versus intramuscular route of immunization. Folia Microbiol. 50:83-86. [DOI] [PubMed] [Google Scholar]
- 29.Ye, P., P. B. Garvey, P. Zhang, S. Nelson, G. Bagby, W. R. Summer, P. Schwarzenberger, J. E. Shellito, and J. K. Kolls. 2001. Interleukin-17 and lung host defense against Klebsiella pneumoniae infection. Am. J. Respir. Cell. Mol. Biol. 25:335-340. [DOI] [PubMed] [Google Scholar]
- 30.Yoshida, K., T. Matsumoto, K. Tateda, K. Uchida, S. Tsujimoto, Y. Iwakura, and K. Yamaguchi. 2001. Protection against pulmonary infection with Klebsiella pneumoniae in mice by interferon-γ through activation of phagocytic cells and stimulation of production of other cytokines. J. Med. Microbiol. 50:959-964. [DOI] [PubMed] [Google Scholar]
- 31.Yoshida, K., T. Matsumoto, K. Tateda, K. Uchida, S. Tsujimoto, and K. Yamaguchi. 2001. Induction of interleukin-10 and down-regulation of cytokine production by Klebsiella pneumoniae capsule in mice with pulmonary infection. J. Med. Microbiol. 50:456-461. [DOI] [PubMed] [Google Scholar]
- 32.Yoshida, K., T. Matsumoto, K. Tateda, K. Uchida, S. Tsujimoto, and K. Yamaguchi. 2000. Role of bacterial capsule in local and systemic inflammatory responses of mice during pulmonary infection with Klebsiella pneumoniae. J. Med. Microbiol. 49:1003-1010. [DOI] [PubMed] [Google Scholar]