medical sciences. For the article “Estrogen receptor (ER)-β isoforms: A key to understanding ER-β signaling,” by Yuet-Kin Leung, Paul Mak, Sazzad Hassan, and Shuk-Mei Ho, which appeared in issue 35, August 29, 2006, of Proc Natl Acad Sci USA (103:13162–13167; first published August 22, 2006; 10.1073/pnas.0605676103), the authors note that in Fig. 2C, the x axis is labeled incorrectly, due to a printer's error. The corrected figure and its legend appear below. This error does not affect the conclusions of the article.
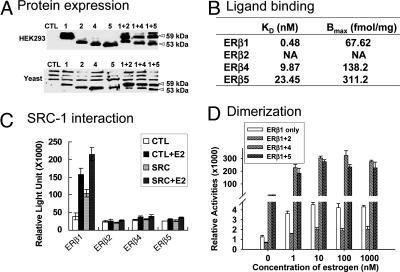
Fig. 2.
Characterization of ER-β isoforms. (A) Western blot analysis of ER-β isoforms overexpressed in HEK293 cells and yeast. N-terminal-specific polyclonal ER-β antibody (H150 from Santa Cruz Biotechnology), which would recognize all ER-β isoforms, was used in this study. An equal amount (50 μg) of protein was loaded to each lane. Mock-transfected cells or an untransformed yeast strain were set up as a control experiment (CTL). Samples expressing ER-β1, -β2, -β4, and -β5 were labeled as 1, 2, 4, and 5, respectively. The size of the ER-β isoforms was consistent with the predicted molecular size, ranging from 53 to 59 kDa. Coexpression of ER-β isoforms with ER-β1 was also performed in both cell line and yeast. Lanes 1 and 2, 1 and 4, and 1 and 5 represent the samples overexpressing ER-β1 and -β2, ER-β1 and -β4, and ER-β1 and -β5, respectively. (B) Tabulated results of in vitro estrogen receptor binding assay. Four hundred micrograms of total yeast lysate expressing ER-β isoforms was applied to each binding reaction as described in Materials and Methods. Binding data were calculated and analyzed with GraphPad Prism 4.0 software to determine the Bmax and Kd of each isoform. (C) Effects of SRC-1 on the transactivation activities of ER-β isoforms. SRC-1 expression vector was transfected into HEK293 cells carrying different ER-β isoform expression vectors with the reporter plasmid. Transactivation assays were performed as described in Materials and Methods in the presence or absence of 1 nM E2. Three independent experiments were performed and averaged. The standard deviation was calculated. (D) Dimerization of ER-β isoforms by Y2H experiment. E2 at two different concentrations (1 nM to 1 μM) was incubated overnight with different yeast strains. A Beta-Glo assay was performed to quantify the reporter (β-gal) activity. The higher the reporter activity, the stronger the interaction between two of the same ER-β (homodimer) or different (heterodimer) isoforms. Four types of homodimers (β1 + β1, β2 + β2, β4 + β4, and β5 + β5) and three kinds of heterodimers (β1 + β2, β1 + β4, and β1 + β5) were subjected to Y2H analyses. The background value was subtracted during data analyses. Experiments were performed in triplicate, and the standard deviation was calculated. All results were summarized in this figure, except for β2, β4, and β5 homodimers, in which activities were undetectable.