Abstract
In the Apc1638+/− mouse model of intestinal tumorigenesis, targeted inactivation of the cyclin-dependent kinase inhibitor p21WAF1/cip1 is highly effective in enhancing Apc-initiated tumor formation in the intestine. Because p21WAF1/cip1 plays a critical role in regulating intestinal cell proliferation, maturation, and tumorigenesis, we examined whether its inactivation would enhance tumor formation in a different mouse model of colon cancer. Therefore, we mated p21−/− mice with mice carrying a genetic deficiency of the Muc2 gene, which encodes the major gastrointestinal mucin. Muc2−/− mice develop tumors in the small and large intestine and the rectum, but in contrast to tumors in Apc1638+/− mice, this does not involve increased expression or nuclear localization of β-catenin. We found that inactivation of p21WAF1/cip1 significantly increased the frequency and size of intestinal tumors in Muc2 knockout mice and also led to development of more invasive adenocarcinomas. This enhanced tumorigenesis significantly decreased mouse life span. Further, inactivation of p21WAF1/cip1 increased cell proliferation, decreased apoptosis, and decreased intestinal trefoil factor expression in the mucosa of both the small and large intestine. Surprisingly, reduced expression of p27kip1 was also observed in the Muc2−/−, p21+/−, and p21−/− mice. In contrast, the expression of c-myc was significantly elevated. Thus, p21 modulates the formation of tumors whose initiation does (Apc) or does not (Muc2) involve altered β-catenin-Tcf4 signaling, but which may converge on common elements downstream of this signaling pathway.
p21WAF1/cip1 is an inhibitor of cyclin-dependent kinase (cdk) activity and is therefore an important regulator of cell cycle progression, and overall cell maturation, including differentiation and apoptosis.1–4 Despite this apparently central role of p21, mice with a targeted, homozygous inactivation of the p21 gene are essentially normal, although embryonic fibroblasts from such mice are defective in cell-cycle arrest at the G1 checkpoint.5 Thus, either p21 is not critical in cell maturation or its loss can be compensated for by other alterations that preserve pathways necessary for normal development and tissue homeostasis.
In the intestinal tract, p21 appears to play a key role in the maturation of cells as they migrate from the crypt toward the lumen.6 p21 is expressed as cells exit the proliferative compartment. This pattern of expression, as well as the known functions of p21, suggested to us that p21 levels would modulate tumor formation in the intestine, and indeed, the introduction of a targeted inactivation of p21 into the Apc1638+/− mouse, in which intestinal tumors form when the wild-type Apc gene is lost or inactivated, substantially increased intestinal tumor formation.3 More recently, the importance of p21 in homeostasis of the intestinal mucosa has been emphasized and clarified by the finding that expression of p21 is repressed by c-MYC protein,7–9 a target of APC-β-catenin-Tcf signaling.8,10–12 c-MYC sequesters the transcription factor MIZ in a MIZ/MYC complex,7,9,13,14 and data have been presented that the down-regulation of c-myc gene expression, and consequent activation of p21 expression by MIZ, is a critical event in triggering intestinal cell differentiation.8
Interestingly, analysis of gene expression profiles in colonic tumor cells in which β-catenin-Tcf signaling is abrogated by expression of a dominant-negative Tcf -4 demonstrated up-regulation of gene markers that are characteristic of either the mucosecretory or the absorptive cell lineages in the intestinal mucosa,15–18 consistent with our report that down-regulation of β-catenin-Tcf signaling accompanied, and was mechanistically linked to, colonic cell differentiation in tissue culture.12 This therefore suggests that loss of normal cell differentiation patterns in the intestinal mucosa, possibly attributed to the regulation of p21 expression through effects of β-catenin-Tcf signaling and c-myc expression, is a key event in tumor formation, and indeed, introduction of the targeted inactivation of p21 in the Apc1638+/− mouse, which increased tumor formation, decreased the number of mucin-expressing goblet cells in the intestinal mucosa.3
We also showed that genetic inactivation of the Muc2 gene, which encodes the principal colonic mucin,19–23 was sufficient to cause tumor formation.24 However, unlike the loss of Apc function, this loss of Muc2 expression was not accompanied by elevated β-catenin expression, or the relocalization of β-catenin to the nucleus.24 Thus, there was no evidence that β-catenin-Tcf signaling was aberrant in this model, although c-myc expression was elevated in the tumors.24 Therefore, although the mechanism of tumorigenesis in the Muc2 and the Apc models may converge, the initial events seem to be distinct, and differ in their overt affects on β-catenin-Tcf signaling. The question therefore arises whether the targeted inactivation of p21 would also be effective in augmenting tumor formation in the Muc2−/− mouse model. Such data are essential for interpretation of the role that loss of p21 plays in the central pathways involved in intestinal tumorigenesis. We here report that introduction of a targeted inactivation of p21 increased tumor formation in the small intestine, colon, and rectum, of mice with a targeted inactivation of Muc2, accompanied by increased cell proliferation, decreased apoptosis, and decreased differentiation in the intestinal mucosa, which was associated with down-regulation of p27kip and up-regulation of c-myc.
Materials and Methods
The p21 and Muc2 mouse models, and methods for genotyping, have been reported.3,24 Muc2−/− mice (mixed C57BL/6J and 129/SvOla background) were mated with p21−/− mice (mixed 129S6/SvEvTac and NIH Black Swiss background) to generate Muc2+/−, p21+/− offspring (F1). F1 mice were mated to produce desired genotypes: Muc2−/−, p21+/+, Muc2−/−, p21+/−, or Muc2−/−, p21−/−. At weaning (∼3 to 4 weeks), littermates were fed AIN-76A diet (Teklad, Madison, WI), ad libitum.
Mice were weighed weekly and maintained on diet for 36 weeks, or until they exhibited significant weight loss or other signs of extensive tumor formation. Mice were killed by CO2 overdose and cervical dislocation, and then rapidly dissected for evaluation of tumors and fixation of tissues, as described previously.3,4,24,25 Proliferation and apoptosis were evaluated by staining for proliferating cell nuclear antigen (Zymed, South San Francisco, CA) or terminal dUTP nick-end labeling (TUNEL) assay (Trevigen, Gaithersburg, MD), as described.3,26
Total RNA and protein were isolated from the frozen tissues using TRIzol reagent (Invitrogen Life Technology, Carlsbad, CA), as described.25 The quantity of RNA and protein were measured spectrophotometrically. As we described,25 cDNA was synthesized from DNase-treated total RNA using TaqMan Multiscribe reverse transcriptase (Applied Biosystems, Inc., Foster City, CA). Quantitative polymerase chain reaction analysis was done using the ABI Prism 7900-HT sequence detection system (96-well, Applied Biosystems, Inc., Foster City, CA). The primers for p21, p27, c-myc, and β-actin, the amplification conditions for the quantitative real-time polymerase chain reaction, and data analysis, were reported previously.25
Western blot analyses of steady-state levels of specific proteins were done by standard methods, as described,25 using the following primary antibodies for detection: anti-p21, anti-p27, anti-c-myc (Santa Cruz Biotechnology, Santa Cruz, CA); and anti-β-actin (Sigma, St. Louis, MO). Signal was detected by the enhanced chemiluminescence technique (Amersham Life Science, Piscataway, NJ). Immunohistochemical staining for intestinal trefoil factor (ITF) was previously reported in detail.24 Briefly, 4-μm formalin-fixed and paraffin-embedded sections were deparaffinized and rehydrated, quenched with 1.5% H2O2, blocked with 10% normal goat serum, and probed with rabbit anti-ITF polyclonal antibody (kindly provided by Catherine Tomasetto, Strasbourg, France). Detection was with biotinylated anti-rabbit IgG (Santa Cruz Biotechnology), followed by incubation with avidin-biotin complex (Vector Labs, Burlingame, CA) and the substrate 3′,5′-diaminobenzidine, combined with hematoxylin counterstaining.
Results
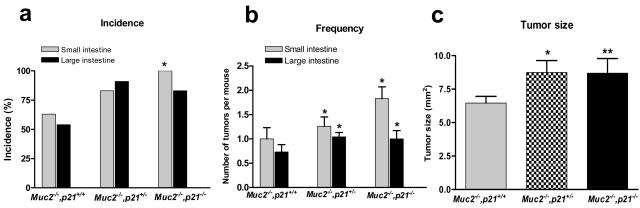
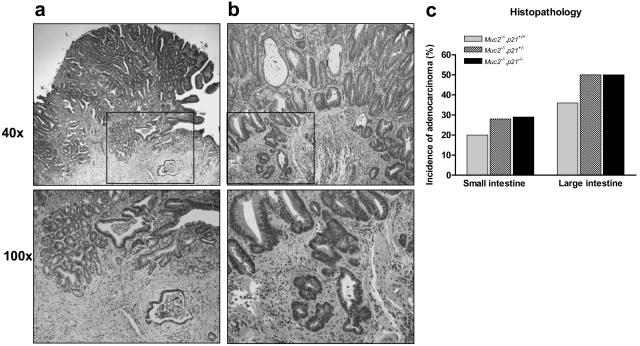
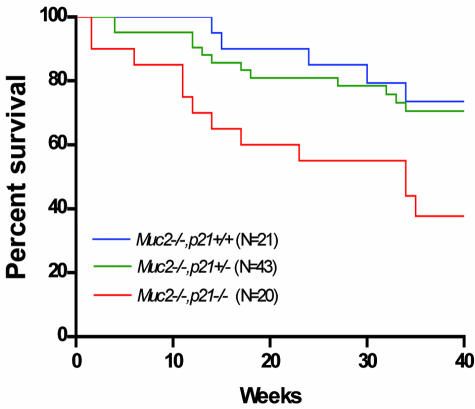
Tumors developed throughout the intestinal tract in the Muc2−/−, p21 wild-type mice. Small intestinal tumors developed in ∼60% of the Muc2−/−, p21 wild-type mice at a frequency of 1.0 tumor per mouse at an age of 36 weeks (Figure 1, a and b). This is similar to the incidence and frequency of tumors we previously reported for the Muc2−/− mice.24 However, littermates that were Muc2−/− and either p21+/− or −/− had a significantly higher tumor incidence of 83% and 100% in small intestinal tumors, respectively (P = 0.025) (Figure 1a). In addition, the small intestinal tumor frequency per mouse was increased by 30% in the Muc2−/−, p21+/− mice and by >80% in the Muc2−/−, p21−/− mice (Figure 1b) (P = 0.029). The effect on tumor size was also striking: small intestinal tumors in Muc2−/−, p21−/− or Muc2−/−, p21+/− mice were 35% larger than the tumors in Muc2−/−, p21+/+ mice (P = 0.02 and P = 0.03, respectively, compared to the Muc2−/−, p21+/+ mice) (Figure 1c). Most important, 28% of the small intestinal tumors in the Muc2−/−, p21+/− and Muc2−/−, p21−/− mice (8 of 29 and 8 of 28, respectively) were invasive adenocarcinomas (Figure 2a), more than the 20% in the Muc2−/−, p21 wild-type mice (3 of 15) (Figure 2, a and c). Because Muc2−/−, p21−/− mice died earlier (Figure 3), the Muc2−/−, p21−/− mice that were sacrificed at 36 weeks, which provided the histopathological data, may overrepresent those with less aggressive phenotype, accounting for the similarity in the incidence, frequency, size, and pathology of these mice to the Muc2−/−, p21+/− mice.
Figure 1.
The incidence (a), frequency (b), and size (c) of intestinal tumors in the Muc2−/−, p21+/+, Muc2−/−, p21+/−, or Muc2−/−, p21−/− mice. a: *P = 0.025, in comparison to Muc2−/−, p21+/+ mice by Fisher’s exact test. b: *P = 0.02. c: *P = 0.03 and **P = 0.02 in comparison to Muc2−/−, p21+/+ mice by Mann-Whitney test.
Figure 2.
Adenocarcinomas of the duodenum (a) and colon (b) in Muc2−/−, p21−/− mice. c: Incidence of adenocarcinoma in the small and large intestine of Muc2−/−, p21+/+, Muc2−/−, p21+/−, or Muc2−/−, p21−/− mice.
Figure 3.
Survival of Muc2−/−, p21+/+, Muc2−/−, p21+/−, or Muc2−/−, p21−/− mice.
The inactivation of p21 also increased tumor formation in the colon and rectum of the mice. Although 53% of Muc2−/−, p21 wild-type mice developed large intestinal tumors, in the Muc2−/−, p21+/− or Muc2−/−, p21−/− mice, this increased to 91% and 83%, respectively (Figure 1a), and again the frequency was also significantly increased from 0.63 large intestinal tumors per mouse in the Muc2−/−, p21+/+ mice to 1.04 and 1.00 in the Muc2−/−, p21+/− or Muc2−/−, p21−/− mice (Figure 1b). Analysis of histopathology revealed that 50% of the large intestinal tumors were early or advanced invasive adenocarcinomas (Figure 2b) in the Muc2−/−, p21+/− or Muc2−/−, p21−/− mice (13 of 26 and 8 of 16, respectively) (Figure 2c), whereas 36% (4 of 11) of large intestinal tumors in the Muc2−/−, p21+/− mice were invasive adenocarcinoma. More interesting, 33% (5/15) of Muc2−/−, p21+/+ mice developed rectal tumors. The incidence of this pathology was somewhat higher in the p21 heterozygous mice (43%, 10 of 23), and increased further, to 60% (9 of 15), in the Muc2−/−, p21−/− mice. All of the rectal tumors were either adenomas or invasive adenocarcinomas. As for the small intestinal tumors, mice with less aggressive disease may be underrepresented in the data because of early death of the Muc2−/−, p21−/− mice (Figure 3).
In the Apc mouse model of tumorigenesis, the increased tumor formation caused by the introduction of the targeted inactivation of p21 was accompanied by a disruption in homeostasis of the intestinal mucosa, demonstrated by increased proliferation, decreased apoptosis, and a decrease in recognizable goblet cells in the intestinal mucosa.3 All of these changes were already present in the Muc2−/− model that was wild-type for p21.24 However, because inactivation of p21 was effective in increasing tumor formation in the Muc2−/− mice, we investigated whether there were further perturbations in these three aspects of cell maturation.
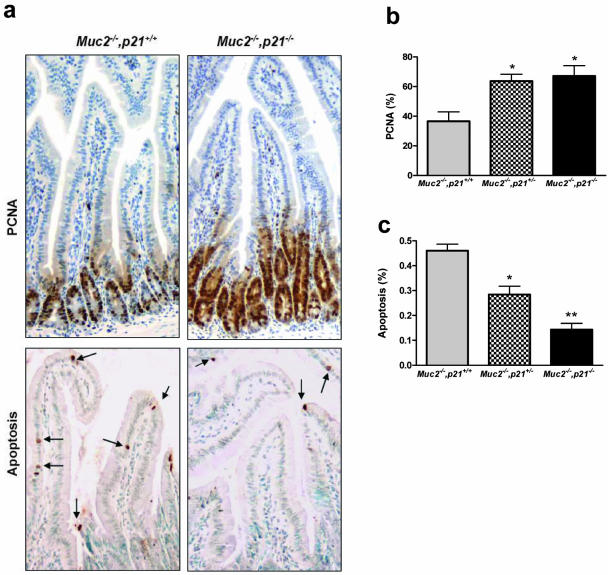
Examples of the assays for proliferation [proliferating cell nuclear antigen (PCNA) staining[ and apoptosis (TUNEL staining) are shown in Figure 4a. In the duodenum, the proliferation index (percent PCNA-positive cells) of Muc2−/−, p21+/+ mice was 38% (Figure 4b). Using the same assay, with mice wild-type at both loci and of similar age maintained on the same diet, we reported that the proliferation index was 19%.24 The twofold increase is consistent with the increase we have reported for the Muc2−/− mice.24 However, the introduction of one inactivated p21 allele increased the proliferation index even further to ∼60% (Figure 4b; P < 0.01), and inactivation of the second p21 allele resulted in an increased proliferation index of ∼70%. Similarly, the Muc2−/−, p21+/+ mice had an apoptotic index in the duodenum of 0.45 (Figure 4c), lower than the 0.90 level that we reported for mice wild-type at both loci, and consistent with the reduced apoptosis we reported in the Muc2−/− mice.24 Again, however, introduction of the targeted inactivation of p21 further reduced the apoptotic index, in this case more clearly in a p21 gene dose-dependent manner: by 38% in the Muc2−/−, p21+/− mice and by 60% in the Muc2−/−,p21−/− mice (Figure 4c, P = 0.06 and <0.01, respectively). The increased proliferation and decreased apoptosis were also seen in the flat mucosa of the colons from the Muc2−/−, p21+/− or Muc2−/−, p21−/− mice compared with Muc2−/− littermates wild-type for p21 (data not shown). Therefore, changes in proliferation and apoptosis were consistently associated with the increased tumor formation because of p21 inactivation.
Figure 4.
a: Examples of PCNA and TUNEL staining (for apoptosis, arrows) in the duodenum of Muc2−/−, p21+/+ or Muc2−/−, p21−/− mice. Proliferation index (b) and apoptotic index (c) in the duodenum of Muc2−/−, p21+/+, Muc2−/−, p21+/−, or Muc2−/−, p21−/− mice were measured by PCNA and TUNEL staining, respectively. The value shown in the bars represents the mean ± SE of 10 different mice per group. b: *P < 0.01 in comparison to Muc2−/−, p21+/+ mice. c: *P = 0.06 and **P < 0.01 in comparison to Muc2−/−, p21+/+ mice by Mann-Whitney test. Original magnification, 20 × 10 (a).
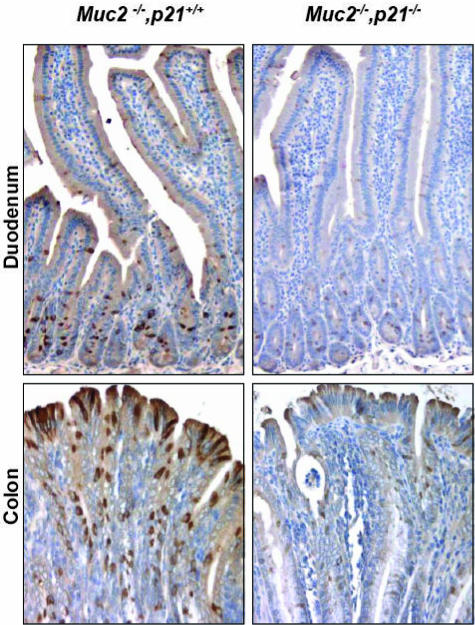
The most interesting finding regarded differentiation of the goblet cell lineage. The intestinal mucosa of the Muc2−/− mouse is characterized by the absence of recognizable goblet cells.24 However, this cell lineage is not completely ablated, because cells in the mucosa still stain immunohistochemically for ITF, another principal secreted product of goblet cells and component of intestinal mucus.27,28 This can be clearly seen in the duodenum and colon of the Muc2−/−, p21+/+ mice (Figure 5). However, on homozygous inactivation of the p21 gene, the goblet cell lineage is further perturbed as indicated by the significant reduction in ITF staining (Figure 5). Thus, the loss of p21 expression in the intestinal mucosa of the Muc2−/− mice, which accelerates and enhances tumor formation, alters cell maturation—increasing cell proliferation, decreasing cell differentiation and apoptosis—in the intestinal mucosa.
Figure 5.
ITF expression in the duodenum and colon of Muc2−/−, p21+/+ or Muc2−/−, p21−/− mice. Original magnification, 20 × 10.
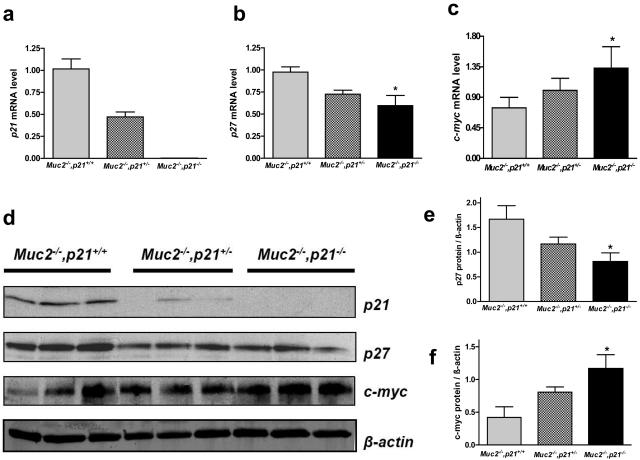
Gene expression was investigated in the mucosa of these mice. As expected, the levels of p21 mRNA decreased in a gene dosage-dependent manner in the Muc2−/−, p21+/− and Muc2−/−, p21−/− mice compared to the Muc2−/−, p21 wild-type mice (Figure 6a), and this was also reflected in the protein levels of p21 (Figure 6d). We have previously shown that the inactivation of p27kip1 in mice maintained on the AIN-76A diet was sufficient to cause tumor formation (W. Yang, et al, manuscript in preparation).25 It was therefore of interest that the inactivation of p21 in the Muc2−/− mice led to a concurrent reduction in p27 mRNA by 30% (Figure 6b) and protein expression by 50% (Figure 6, d and e).
Figure 6.
p21, p27 and c-myc expression in the colon of Muc2−/−, p21+/+, Muc2−/−, p21+/−, or Muc2−/−, p21−/− mice. a–c: Relative level of mRNA of p21 (a), p27(b), and c-myc (c) in the colon, assayed by quantitative real-time reverse transcriptase-polymerase chain reaction. The value shown in the bars represents the mean ± SE of five different mice per group. d: Protein expression of p21, p27, and c-myc, assayed by Western blot. The quantification of p27 (e) and c-myc (f) protein were normalized to β-actin. *P < 0.05 in comparison to Muc2−/−, p21+/+ mice by Mann-Whitney test.
As regards the reduction in p27 expression, it may be specific for the compound knockout (ie, Muc2−/−, p21−/−) mice, because preliminary data indicated that there is no reduction in p27 expression in the Muc2−/− mice compared to Muc2+/+ mice when both are wild-type for p21 (N. Popova and A. Velcich, personal communication), and it has been reported that p27 expression is not reduced in p21−/− mice compared to wild-type mice.29–31
Finally, as outlined in the Introduction, p21 is under negative control of myc expression in the intestinal mucosa. We therefore investigated whether there is a feedback, and thus whether the decreased p21 expression alters c-myc expression. Figure 6c illustrates that c-myc mRNA levels indeed rose in the intestinal mucosa of the Muc2−/− mice in conjunction with the targeted inactivation of p21. This increase was ∼60% in the Muc2−/−, p21−/− mice compared to the Muc2−/−, p21+/+ mice (P < 0.05) (Figure 6c). Further quantifying Western blot signals demonstrated that c-myc protein level in Muc2−/−, p21−/− mice was elevated by 1.78-fold greater than the Muc2−/−, p21+/+ mice (P < 0.05) (Figure 6, d and f).
Discussion
There is considerable evidence that p21WAF1/cip1 plays a fundamental role in pathways that regulate intestinal cell maturation and homeostasis of the intestinal mucosa.1,3,4,6,32 In the intestinal tract, p21 is expressed as cells exit the proliferative compartment, and loss of both expression and topological regulation is detected early in colon tumor formation.1,6 Absence of p21 is linked to inability of colon tumor cells to arrest in the G1 phase of the cell cycle,13,33,34 when stimulated by the nonsteroidal anti-inflammatory drug sulindac,4,34,35 or by radiation.36 However, despite this evidence for a key role of p21, its targeted inactivation does not grossly perturb the mucosa and does not cause tumor formation.5
Loss of p21 in mice that have an initiating mutation in Apc does cause marked enhancement of tumor formation.3 This report extends this observation to another initiator of intestinal tumorigenesis: the inactivation of Muc2, the gene that encodes the major intestinal mucin. Inactivation of Muc2 initiates tumor formation by a pathway that is distinct from that of loss of APC function, in that loss of Muc2 expression does not involve elevation of β-catenin expression or accumulation in the nucleus, both of which are characteristic of APC-initiated tumors.24 The difference in mechanism is confirmed by more recent evidence that combining the Apc and Muc2 mutations in the mouse is synergistic in terms of tumor formation initiated by either mutation alone (A. Velcich, manuscript in preparation). Thus, the fact that inactivation of p21 enhances tumor formation in these two different mouse models underlines the importance of p21 in intestinal homeostasis and tumorigenesis.
As in the Apc1638+/− model, the targeted inactivation of p21 causes a disruption in cell maturation pathways that includes further elevation in proliferation, depressed apoptosis, and perturbation of differentiation lineages. In the Apc1638+/− model, the perturbation of differentiation by inactivation of p21 was manifest as a decrease in the number of mucin-containing goblet cells.3 However, in the Muc2−/− mouse, this cell phenotype is already not detectable in the intestinal mucosa, but the loss of p21 appears to further perturb development of the lineage because it results in decreased expression of ITF, another marker of this cell type, which instead persists in the Muc2−/−, p21+/+ mice (Figure 5).24 In this regard, it is of interest that in the Muc2−/−, p21−/− mice, there is down-regulation of another cdk inhibitor, p27kip1. Although the inactivation of p27 is able to initiate tumor formation in mice maintained on the AIN-76A diet,25 like Muc2−/− mice, the p27−/− mice still express ITF, albeit assayed at the mRNA level, while the mucin-expressing goblet cells are reduced.25 Therefore, it may be that in the Muc2−/−, p21−/− mice, the combined inactivation of p21, and down-regulation of p27, has a more extensive affect on the development of the goblet cell lineage than does inactivation and/or down-regulation of only one of these cdk inhibitors.
In the intestinal mucosa, tumor initiation by loss of function of APC, either through mutation, deletion, or epigenetic events, is most often because of the effects of APC on β-catenin/Tcf signaling, and indeed, evidence that altered β-catenin-Tcf signaling is sufficient to initiate tumor formation is very strong.15,18,37–39 One of the direct, key targets of β-catenin-Tcf signaling is c-myc. Brief inactivation of c-myc is sufficient to induce a sustained loss of the transformed phenotype.40 The importance of c-myc is also supported by the fact that Muc2−/− initiated tumor formation, although not targeting β-catenin-Tcf signaling, still caused elevation of c-myc expression,24 and that loss of p21, which increases tumor initiation and progression, further elevates c-myc expression (Figure 6). Thus, these data reinforce a key role for c-myc in intestinal tumor formation. This is consistent with our recent report that c-myc likely plays a key role in regulating the maturation pathway as cells migrate from the proliferative compartment toward the lumen in the intestinal mucosa.41
In summary, our data demonstrated that the loss of p21 in the Muc2−/− mouse model of intestinal tumor formation is linked to further perturbation in cellular mechanisms of intestinal homeostasis. This is coincident with alterations of expression of at least two molecules critical in the maturation of intestinal epithelial cells: down-regulation of p27 and up-regulation of c-myc. Each has multiple targets and pathways that it can modulate. The alterations of these targets and pathways by gene and proteomic profiling of cells as they migrate along the crypt-villus axis of the intestinal tract in mouse genetic models, with and without modulation of tumorigenesis by other loci and environmental factors, will be fundamental in defining the key events in establishing probability of tumor formation and progression.
Acknowledgments
We thank Dr. Philip Leder for providing the p21−/− mice and Dr. Maomi Li for reading the manuscript.
Footnotes
Address reprint requests to WanCai Yang, M.D., Department of Oncology, Montefiore Medical Center, Albert Einstein Cancer Center, 111 East 210th St., Bronx, NY 10467. E-mail: wyang@montefiore.org.
Supported in part by the National Cancer Institute (grants CA96605, CA100926, CA87559, and P01 13330).
References
- Polyak K, Hamilton SR, Vogelstein B, Kinzler KW. Early alteration of cell-cycle-regulated gene expression in colorectal neoplasia. Am J Pathol. 1996;149:381–387. [PMC free article] [PubMed] [Google Scholar]
- Gartel AL, Tyner AL. Transcriptional regulation of the p21((WAF1/CIP1)) gene. Exp Cell Res. 1999;246:280–289. doi: 10.1006/excr.1998.4319. [DOI] [PubMed] [Google Scholar]
- Yang WC, Mathew J, Velcich A, Edelmann W, Kucherlapati R, Lipkin M, Yang K, Augenlicht LH. Targeted inactivation of the p21(WAF1/cip1) gene enhances Apc-initiated tumor formation and the tumor-promoting activity of a Western-style high-risk diet by altering cell maturation in the intestinal mucosal. Cancer Res. 2001;61:565–569. [PubMed] [Google Scholar]
- Yang WC, Velcich A, Mariadason J, Nicholas C, Corner G, Houston M, Edelmann W, Kucherlapati R, Holt PR, Augenlicht LH. p21(WAF1/cip1) is an important determinant of intestinal cell response to sulindac in vitro and in vivo. Cancer Res. 2001;61:6297–6302. [PubMed] [Google Scholar]
- Deng C, Zhang P, Harper JW, Elledge SJ, Leder P. Mice lacking p21CIP1/WAF1 undergo normal development, but are defective in G1 checkpoint control. Cell. 1995;82:675–684. doi: 10.1016/0092-8674(95)90039-x. [DOI] [PubMed] [Google Scholar]
- el-Deiry WS, Tokino T, Waldman T, Oliner JD, Velculescu VE, Burrell M, Hill DE, Healy E, Rees JL, Hamilton SR. Topological control of p21WAF1/CIP1 expression in normal and neoplastic tissues. Cancer Res. 1995;55:2910–2919. [PubMed] [Google Scholar]
- Wu S, Cetinkaya C, Munoz-Alonso MJ, von der Lehr N, Bahram F, Beuger V, Eilers M, Leon J, Larsson LG. Myc represses differentiation-induced p21CIP1 expression via Miz-1-dependent interaction with the p21 core promoter. Oncogene. 2003;22:351–360. doi: 10.1038/sj.onc.1206145. [DOI] [PubMed] [Google Scholar]
- van de Wetering M, Sancho E, Verweij C, de Lau W, Oving I, Hurlstone A, van der Horn K, Batlle E, Coudreuse D, Haramis AP, Tjon-Pon-Fong M, Moerer P, van den Born M, Soete G, Pals S, Eilers M, Medema R, Clevers H. The beta-catenin/TCF-4 complex imposes a crypt progenitor phenotype on colorectal cancer cells. Cell. 2002;111:241–250. doi: 10.1016/s0092-8674(02)01014-0. [DOI] [PubMed] [Google Scholar]
- Gartel AL, Shchors K. Mechanisms of c-myc-mediated transcriptional repression of growth arrest genes. Exp Cell Res. 2003;283:17–21. doi: 10.1016/s0014-4827(02)00020-4. [DOI] [PubMed] [Google Scholar]
- He TC, Sparks AB, Rago C, Hermeking H, Zawel L, da Costa LT, Morin PJ, Vogelstein B, Kinzler KW. Identification of c-MYC as a target of the APC pathway. Science. 1998;281:1509–1512. doi: 10.1126/science.281.5382.1509. [DOI] [PubMed] [Google Scholar]
- Korinek V, Barker N, Morin PJ, van Wichen D, de Weger R, Kinzler KW, Vogelstein B, Clevers H. Constitutive transcriptional activation by a beta-catenin-Tcf complex in APC−/− colon carcinoma. Science. 1997;275:1784–1787. doi: 10.1126/science.275.5307.1784. [DOI] [PubMed] [Google Scholar]
- Mariadason JM, Bordonaro M, Aslam F, Shi L, Kuraguchi M, Velcich A, Augenlicht LH. Down-regulation of beta-catenin TCF signaling is linked to colonic epithelial cell differentiation. Cancer Res. 2001;61:3465–3471. [PubMed] [Google Scholar]
- Seoane J, Le HV, Massague J. Myc suppression of the p21(Cip1) Cdk inhibitor influences the outcome of the p53 response to DNA damage. Nature. 2002;419:729–734. doi: 10.1038/nature01119. [DOI] [PubMed] [Google Scholar]
- Schneider A, Peukert K, Eilers M, Hanel F. Association of Myc with the zinc-finger protein Miz-1 defines a novel pathway for gene regulation by Myc. Curr Top Microbiol Immunol. 1997;224:137–146. doi: 10.1007/978-3-642-60801-8_14. [DOI] [PubMed] [Google Scholar]
- Kolligs FT, Hu G, Dang CV, Fearon ER. Neoplastic transformation of RK3E by mutant beta-catenin requires deregulation of Tcf/Lef transcription but not activation of c-myc expression. Mol Cell Biol. 1999;19:5696–5706. doi: 10.1128/mcb.19.8.5696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen S, Guttridge DC, You Z, Zhang Z, Fribley A, Mayo MW, Kitajewski J, Wang CY. Wnt-1 signaling inhibits apoptosis by activating beta-catenin/T cell factor-mediated transcription. J Cell Biol. 2001;152:87–96. doi: 10.1083/jcb.152.1.87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kishida M, Koyama S, Kishida S, Matsubara K, Nakashima S, Higano K, Takada R, Takada S, Kikuchi A. Axin prevents Wnt-3a-induced accumulation of beta-catenin. Oncogene. 1999;18:979–985. doi: 10.1038/sj.onc.1202388. [DOI] [PubMed] [Google Scholar]
- Morin PJ, Sparks AB, Korinek V, Barker N, Clevers H, Vogelstein B, Kinzler KW. Activation of beta-catenin-Tcf signaling in colon cancer by mutations in beta-catenin or APC. Science. 1997;275:1787–1790. doi: 10.1126/science.275.5307.1787. [DOI] [PubMed] [Google Scholar]
- Kim YS, Gum JR., Jr Diversity of mucin genes, structure, function, and expression. Gastroenterology. 1995;109:999–1001. doi: 10.1016/0016-5085(95)90412-3. [DOI] [PubMed] [Google Scholar]
- van Klinken BJ, Einerhand AW, Duits LA, Makkink MK, Tytgat KM, Renes IB, Verburg M, Buller HA, Dekker J. Gastrointestinal expression and partial cDNA cloning of murine Muc2. Am J Physiol. 1999;276:G115–G124. doi: 10.1152/ajpgi.1999.276.1.G115. [DOI] [PubMed] [Google Scholar]
- Aslam F, Palumbo L, Augenlicht LH, Velcich A. The Sp family of transcription factors in the regulation of the human and mouse MUC2 gene promoters. Cancer Res. 2001;61:570–576. [PubMed] [Google Scholar]
- Velcich A, Palumbo L, Selleri L, Evans G, Augenlicht L. Organization and regulatory aspects of the human intestinal mucin gene (MUC2) locus. J Biol Chem. 1997;272:7968–7976. doi: 10.1074/jbc.272.12.7968. [DOI] [PubMed] [Google Scholar]
- Velcich A, Palumbo L, Jarry A, Laboisse C, Racevskis J, Augenlicht L. Patterns of expression of lineage-specific markers during the in vitro-induced differentiation of HT29 colon carcinoma cells. Cell Growth Differ. 1995;6:749–757. [PubMed] [Google Scholar]
- Velcich A, Yang W, Heyer J, Fragale A, Nicholas C, Viani S, Kucherlapati R, Lipkin M, Yang K, Augenlicht L. Colorectal cancer in mice genetically deficient in the mucin Muc2. Science. 2002;295:1726–1729. doi: 10.1126/science.1069094. [DOI] [PubMed] [Google Scholar]
- Yang WC, Bancroft L, Nicholas C, Lozonschi I, Augenlicht LH. Targeted inactivation of p27kip1 is sufficient for large and small intestinal tumorigenesis in the mouse, which can be augmented by a Western-style high-risk diet. Cancer Res. 2003;63:4990–4996. [PubMed] [Google Scholar]
- Augenlicht LH, Anthony GM, Church TL, Edelmann W, Kucherlapati R, Yang K, Lipkin M, Heerdt BG. Short-chain fatty acid metabolism, apoptosis, and Apc-initiated tumorigenesis in the mouse gastrointestinal mucosa. Cancer Res. 1999;59:6005–6009. [PubMed] [Google Scholar]
- Sands BE, Podolsky DK. The trefoil peptide family. Annu Rev Physiol. 1996;58:253–273. doi: 10.1146/annurev.ph.58.030196.001345. [DOI] [PubMed] [Google Scholar]
- Kindon H, Pothoulakis C, Thim L, Lynch-Devaney K, Podolsky DK. Trefoil peptide protection of intestinal epithelial barrier function: cooperative interaction with mucin glycoprotein. Gastroenterology. 1995;109:516–523. doi: 10.1016/0016-5085(95)90340-2. [DOI] [PubMed] [Google Scholar]
- Albrecht JH, Poon RY, Ahonen CL, Rieland BM, Deng C, Crary GS. Involvement of p21 and p27 in the regulation of CDK activity and cell cycle progression in the regenerating liver. Oncogene. 1998;16:2141–2150. doi: 10.1038/sj.onc.1201728. [DOI] [PubMed] [Google Scholar]
- Kwon YH, Jovanovic A, Serfas MS, Kiyokawa H, Tyner AL. P21 functions to maintain quiescence of p27-deficient hepatocytes. J Biol Chem. 2002;277:41417–41422. doi: 10.1074/jbc.M203388200. [DOI] [PubMed] [Google Scholar]
- Kwon YH, Jovanovic A, Serfas MS, Tyner AL. The Cdk inhibitor p21 is required for necrosis, but it inhibits apoptosis following toxin-induced liver injury. J Biol Chem. 2003;278:30348–30355. doi: 10.1074/jbc.M300996200. [DOI] [PubMed] [Google Scholar]
- Mahyar-Roemer M, Roemer K. p21 Waf1/Cip1 can protect human colon carcinoma cells against p53-dependent and p53-independent apoptosis induced by natural chemopreventive and therapeutic agents. Oncogene. 2001;20:3387–3398. doi: 10.1038/sj.onc.1204440. [DOI] [PubMed] [Google Scholar]
- Archer S, Meng S, Wu J, Johnson J, Tang R, Hodin R. Butyrate inhibits colon carcinoma cell growth through two distinct pathways. Surgery. 1998;124:248–253. [PubMed] [Google Scholar]
- Goldberg Y, Nassif II, Pittas A, Tsai LL, Dynlacht BD, Rigas B, Shiff SJ. The anti-proliferative effect of sulindac and sulindac sulfide on HT-29 colon cancer cells: alterations in tumor suppressor and cell cycle-regulatory proteins. Oncogene. 1996;12:893–901. [PubMed] [Google Scholar]
- Augenlicht LH, Mariadason JM, Wilson A, Arango D, Yang WC, Heerdt BG, Velcich A. Short chain fatty acids and colon cancer. J Nutr. 2002;132:3804S–3808S. doi: 10.1093/jn/132.12.3804S. [DOI] [PubMed] [Google Scholar]
- McDonald ER, III, Wu GS, Waldman T, El-Deiry WS. Repair defect in p21 WAF1/CIP1 −/− human cancer cells. Cancer Res. 1996;56:2250–2255. [PubMed] [Google Scholar]
- Harada N, Tamai Y, Ishikawa T, Sauer B, Takaku K, Oshima M, Taketo MM. Intestinal polyposis in mice with a dominant stable mutation of the beta-catenin gene. EMBO J. 1999;18:5931–5942. doi: 10.1093/emboj/18.21.5931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morin PJ. Beta-catenin signaling and cancer. Bioessays. 1999;21:1021–1030. doi: 10.1002/(SICI)1521-1878(199912)22:1<1021::AID-BIES6>3.0.CO;2-P. [DOI] [PubMed] [Google Scholar]
- Munemitsu S, Albert I, Rubinfeld B, Polakis P. Deletion of an amino-terminal sequence beta-catenin in vivo and promotes hyperphosporylation of the adenomatous polyposis coli tumor suppressor protein. Mol Cell Biol. 1996;16:4088–4094. doi: 10.1128/mcb.16.8.4088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jain M, Arvanitis C, Chu K, Dewey W, Leonhardt E, Trinh M, Sundberg CD, Bishop JM, Felsher DW. Sustained loss of a neoplastic phenotype by brief inactivation of MYC. Science. 2002;297:102–104. doi: 10.1126/science.1071489. [DOI] [PubMed] [Google Scholar]
- Mariadason J, Nicholas C, L’Italien K, Zhuang M, Smartt H, Heerdt BG, Yang WC, Corner C, Wilson AJ, Klampfer L, Arango D, Augenlicht LH: The transcriptional reprogramming that defines intestinal epithelial cell maturation along the crypt-villus axis. Gastroenterology, 2005 (in press) [DOI] [PubMed] [Google Scholar]