Abstract
The vanilloid receptor-1 (VR1, or transient receptor potential vanilloid-1 receptor, TRPV1) is activated by capsaicin, the key ingredient of hot peppers. TRPV1 was originally described on sensory neurons as a central integrator of various nociceptive stimuli. However, several human skin cell populations are also now recognized to express TRPV1, but with unknown function. Exploiting the human hair follicle (HF) as a prototypic epithelial-mesenchymal interaction system, we have characterized the HF expression of TRPV1 in situ and have examined TRPV1 signaling in organ-cultured human scalp HF and outer root sheath (ORS) keratinocytes in vitro. TRPV1 immunoreactivity was confined to distinct epithelial compartments of the human HF, mainly to the ORS and hair matrix. In organ culture, TRPV1 activation by capsaicin resulted in a dose-dependent and TRPV1-specific inhibition of hair shaft elongation, suppression of proliferation, induction of apoptosis, premature HF regression (catagen), and up-regulation of intrafollicular transforming growth factor-β2. Cultured human ORS keratinocytes also expressed functional TRPV1, whose stimulation inhibited proliferation, induced apoptosis, elevated intracellular calcium concentration, up-regulated known endogenous hair growth inhibitors (interleukin-1β, transforming growth factor-β2), and down-regulated known hair growth promoters (hepatocyte growth factor, insulin-like growth factor-I, stem cell factor). These findings strongly support TRPV1 as a significant novel player in human hair growth control, underscore the physiological importance of TRPV1 in human skin beyond nociception, and identify TRPV1 as a promising, novel target for pharmacological manipulations of epithelial growth disorders.
The tingling or burning sensation that comes along with the consumption of hot peppers arises from capsaicin.1 The molecular target of this agent is the vanilloid (capsaicin) receptor-1 (VR1/TRPV1), which functions as a calcium-permeable nonspecific cation channel.1,2 In addition to capsaicin, as the best-investigated (exogenous) TRPV1 ligand, this receptor can also be activated and/or sensitized by endogenous endovanilloids such as heat, acidosis, arachidonic acid derivatives, lipid peroxidation metabolites, and endocannabinoids (such as anandamide), suggesting that TRPV1 operates as a central integrator molecule of various nociceptive stimuli.3,4 In fact, a subset of sensory neurons can be defined by their exquisite susceptibility to neuropeptide-depletion and ultimately induction of neuronal degeneration by capsaicin and other vanilloids such as resiniferatoxin (RTX).1,5 Clinically, this has long been exploited for the management of numerous chronic pain (such as postherpetic neuralgia) and pruritic syndromes.6
However, beginning with our discovery that mast cells also express functional TRPV1,7 we and others have subsequently described that TRPV1 expression is much more widespread than previously thought suggesting that TRPV1 functions are not limited to sensory ones. It is now recognized that the activation of TRPV1 on several neuroectoderm- or mesoderm-derived cell populations, such as mast cells,7 glial cells,8 bronchial epithelial cells,9 uroepithelial cells,10 and keratinocytes11–13 in vitro results in changes in proliferation, apoptosis, differentiation, and/or cytokine release. Most recently, we and others14,15 found by immunohistology that human skin and its appendages prominently express TRPV1 immunoreactivity (TRPV1-ir) in vivo not only on sensory nerve fibers, but also in the epidermis, hair follicle (HF), sebaceous gland, and several dermal cell populations. Although this supports the concept that the functional roles of TRPV1 in human skin biology reach well beyond nociception, the full range of the functional properties of TRPV1 signaling in cutaneous physiology and pathology remains to be explored and defined.
Because human HF prominently express TRPV1-ir,14,15 and because the HF represents a prototypic, easily manipulated and abundantly available neuroectodermal-mesodermal interaction system that allows to exemplarily dissect the effects of test agents on well-defined epithelial, neural crest-derived, and mesenchymal cell functions under physiologically relevant conditions,16–20 we chose to explore TRPV1 expression and functions in this microcosmic tissue interaction system in situ and in HF organ culture.21
Specifically, we wished to characterize the expression of TRPV1 in human HFs in situ to assess the effects of TRPV1 stimulation on hair shaft elongation, pigmentation, HF keratinocyte proliferation, and apoptosis as well as on HF cycling and the expression patterns of selected major hair growth-regulatory genes in organ-cultured (ie, denervated) human scalp HFs in vitro. This was complemented with studies on the effect of capsaicin on cultured human outer root sheath (ORS) and HaCaT keratinocytes. With the help of these studies in a physiologically and clinically highly relevant human model system, we hoped to obtain new hot insight into the functional role of TRPV1 signaling in epithelial-mesenchymal interactions, as an essential basis for the design of novel therapeutic strategies in the management of clinically relevant epithelial growth disorders via the pharmacological targeting of TRPV1.
Materials and Methods
Isolation and Maintenance of HFs
The study was approved by the Institutional Research Ethics Committee and adhered to Declaration of Helsinki guidelines. Human anagen HFs were isolated from skin obtained from females undergoing face-lift surgery.21 Isolated HFs were maintained in 24-multiwell plates in supplemented Williams E medium (Biochrom, Cambridge, UK) supplemented with 2 mmol/L l-glutamine (Invitrogen, Paisley, UK), 10 ng/ml hydrocortisone (Sigma-Aldrich, Taufkirchen, Germany), 10 μg/ml insulin (Sigma), and antibiotics. Length measurements were performed on individual HFs using a light microscope with an eyepiece measuring graticule.
ORS and HaCaT Keratinocyte Cultures
Anagen HFs were digested using trypsin to obtain ORS keratinocytes.22 Similarly, human dermal fibroblasts (HDFs) were obtained from de-epidermized dermis using enzymatic digestion. ORS cultures were kept on feeder layer of mitomycin-treated HDFs23 in a 1:3 mixture of Ham’s F12 (Biochrom) and in serum-free medium (SFM, Invitrogen) supplemented with 0.1 nmol/L cholera toxin, 5 μg/ml insulin, 0.4 μg/ml hydrocortisone, 2.43 μg/ml adenine, 2 nmol/L triiodothyronine, 10 ng/ml epidermal growth factor, 1 mmol/L ascorbyl-2-phosphate, and antibiotics (all from Sigma). HaCaT keratinocytes were cultured in Dulbecco’s modified Eagle’s medium (Sigma) supplemented with 10% fetal calf serum (Sigma), 2 mmol/L l-glutamine, and antibiotics.
Histology
Cryostat sections (8 μm thick) of scalp skin and cultured HFs were fixed in acetone, air-dried, and processed for histochemistry. Hematoxylin and eosin (H&E, Sigma) staining was used for studying HF morphology whereas melanin pigment was visualized by the Masson-Fontana histochemistry.24
Immunohistochemistry and Immunocytochemistry
For the detection of TRPV1 in human skin, a peroxidase-anti-peroxidase technique (Linaris, Wertheim, Germany), using diaminobenzidine as a chromogen, was used.25 After incubation with a polyclonal goat anti-TRPV1 antibody (1:40; Santa Cruz Biotechnology, Santa Cruz, CA), sections were stained with biotinylated multilink swine anti-goat/mouse/rabbit IgG (1:200; DAKO, Glostrup, Denmark) as secondary antibody and then with an avidin-biotin kit (Linaris). As negative controls, the appropriate TRPV1 antibody was either omitted from the procedure or was preincubated with a synthetic blocking peptide (Santa Cruz). In addition, similarly to as we described before,14 the specificity of TRPV1 staining was also measured on tissues recognized to be TRPV1-positive (rat spinal cord) or TRPV1-negative (spinal cord and skin samples from TRPV1 knockout mice) (data not shown).
For the detection of TRPV1 on isolated HFs, two complementary techniques, the tyramide-substrate amplification (TSA)26,27 and the alkaline-phosphatase (AP) activity-based25,28 methods were used. For the TSA technique, sections were first incubated by the TRPV1 antibody (1:400), then with biotinylated multilink swine anti-goat/mouse/rabbit IgG (1:200), and finally with streptavidin-horseradish peroxidase (TSA kit; Perkin-Elmer, Boston, MA) followed by an application of tetramethyl-rhodamine isothiocyanate-tyramide (1:50, TSA kit). Sections were counterstained by 4,6-diamidino-2-phenylindole (DAPI) (1 μg/ml; Boehringer Mannheim, Mannheim, Germany) for visualization of cell nuclei. For the AP-based method, after staining with the TRPV1-antibody (1:40) and the biotinylated multilink swine anti-goat/mouse/rabbit IgG (1:200), sections were labeled by a streptavidin-AP conjugate (1% reagent mixture; Vector Laboratories, Burlingame, CA). Immunoreactions were finally visualized using Fast Red (Sigma) and the sections were counterstained by hematoxylin (Sigma). The intensity of TRPV1-ir was measured at four previously defined reference areas of interest of the distal ORS layers at a 0 to 255 U/pixel intensity range using the Image Pro Plus 4.5.0 software (Media Cybernetics, Silver Spring, MD), and the average TRPV1-ir was calculated (n = 20 to 30 HFs in each group). For the detection of TRPV1 in ORS keratinocytes, acetone-fixed cells were incubated with the TRPV1-antibody (1:40) and then a fluorescein isothiocyanate (FITC)-conjugated secondary antibody (1:400, Vector) was used to visualize the immunosignal.
To evaluate apoptotic cells in co-localization with a proliferation marker Ki-67, a Ki-67/TUNEL (terminal dUTP nick-end labeling) double-staining method was used.29,30 Cryostat sections were fixed in formalin, ethanol, and acetic acid and labeled with a digoxigenin-deoxyUTP (ApopTag Fluorescein In Situ Apoptosis detection kit; Intergen, Purchase, NY) in the presence of terminal deoxynucleotidyl transferase (TdT), followed by incubation with a mouse anti-Ki-67 antiserum (DAKO). TUNEL+ cells were visualized by an anti-digoxigenin FITC-conjugated antibody (ApopTag kit), whereas Ki-67 was detected by a rhodamine-labeled goat anti-mouse antibody (Jackson ImmunoResearch, West Grove, PA). Negative controls were performed by omitting TdT and the Ki-67 antibody.
For simultaneous immunodetection of TRPV1 and NKI/beteb, a sensitive marker of skin melanocytes,31 acetone-fixed cryostat sections were incubated with a goat anti-TRPV1 antibody (1:40) and then with a rhodamine-labeled rabbit anti-goat IgG (1:200, Jackson ImmunoResearch). Samples were then stained with a mouse anti-NKI/beteb antibody (1:20; Cell Systems, St. Katharinen, Germany) and finally with a FITC-conjugated goat anti-mouse secondary antibody (1:200, Jackson ImmunoResearch).
Staining for transforming growth factor (TGF)-β2 was performed with a rabbit anti-TGF-β2 antibody (1:50, Santa Cruz)32 followed by a FITC-labeled goat anti-rabbit antibody (1:200, Jackson ImmunoResearch). Filaggrin immunostaining was performed using a rabbit anti-filaggrin antibody (1:100; Covance, Richmond, CA) and a FITC-labeled goat anti-rabbit secondary antibody (1:200, Jackson ImmunoResearch). The mean fluorescence intensity of TGF-β2 and filaggrin was measured at four previously defined reference areas of interest in the hair matrix of the hair bulb (for TGF-β2) or in the isthmus region (for filaggrin) using the Image Pro Plus 4.5.0 software (Media Cybernetics), and the average values were calculated (n = 20 to 30 HFs in each group).
Quantitative Real-Time PCR (Q-PCR)
Q-PCR was performed on an ABI Prism 7000 sequence detection system (Applied Biosystems, Foster City, CA) by using the 5′ nuclease assay. Total RNA was isolated using either TRIzol (for cell cultures, Invitrogen) or the RNA easy kit (for HFs; Qiagen, Hilden, Germany). Three μg of total RNA were then reverse-transcribed into cDNA by using 15 U of AMV reverse transcriptase (RT) (Promega, Madison, WI) and 0.025 μg/μl random primers (Promega). PCR amplification was performed by using the TaqMan primers and probes (assay ID, Hs00218912_m1 for human TRPV1; assay ID, Rn00583117_m1 for rat TRPV1) using the TaqMan universal PCR master mix protocol (Applied Biosystems). As internal controls, transcripts of glyceraldehyde 3-phosphate dehydrogenase (GAPDH) were determined (assay ID, Hs99999905_m1 for human GAPDH; assay ID, Rn00576699_m1 for rat GAPDH).
Semiquantitative RT-PCR
The expression of mRNA for TGF-β2 in cultured HFs (15 per group) was determined by semiquantitive RT-PCR.33 The total RNA was extracted using the RNA easy kit (Qiagen) and then was reverse-transcribed with random primers and RT provided in the First Strand cDNA Synthesis kit for RT-PCR (Boehringer). Subsequent PCR amplification [94°C for 5 minutes; 30 cycles of 94°C for 30 seconds, 57°C (TGF-β2) or 55°C (β-actin) for 60 seconds, 72°C for 60 seconds; 72°C for 10 minutes] was performed on the UNO-Thermoblock (Biometra, Göttingen, Germany) with the following primers (all from Sigma): TGF-β2, 5′-ATC CCG CCC ACT TTC TAC AGA C-3′ and 5′-CAT CCA AAG CAC GCT TCT TCC-3′ (GenBank accession number, Y00083); β-actin, 5′-CGA CAA CGG CTC CGG CAT GTG C-3′ and 5′-CGT CAC CGG AGT CCA TCA CGA TGC-3′ (GenBank accession number, NM001101). The PCR products were visualized on a 2% agarose gel with ethidium bromide and the photographed bands were quantified by an Image Pro Plus 4.5.0 software (Media Cybernetics).
Western Blotting
To determine the expression of TRPV1 in cultured cells, the Western blot technique was applied.34 Cells were harvested in homogenization buffer [20 mmol/L Tris-Cl, pH 7.4, 5 mmol/L EGTA, 1 mmol/L 4-(2-aminoethyl)benzenesulfonyl fluoride, 20 μmol/L leupeptin, all from Sigma] and the protein content of samples was measured by a modified BCA protein assay (Pierce, Rockford, IL). The samples were subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis (8% gels were loaded with 20 to 30 μg protein per lane), transferred to nitrocellulose membranes (Bio-Rad, Vienna, Austria), and then probed with the above anti-TRPV1 antibody (1:100). A horseradish peroxidase-conjugated rabbit anti-goat IgG antibody (1:1000, Bio-Rad) was used as a secondary antibody, and the immunoreactive bands were visualized by enhanced chemiluminescence (Amersham, Little Chalfont, UK) on light-sensitive films (AGFA, Brussels, Belgium).
Calcium Measurement
Changes in intracellular calcium concentration ([Ca2+]i) on TRPV1 activation were detected as described before.34 Cells were cultured on coverslips and a calcium-sensitive probe fura-2 AM (5 μmol/L; Molecular Probes, Eugene, OR) was introduced into the intracellular space (1 hour at 37°C). The coverslips, containing the fura-2-loaded cells, were then placed on the stage of an inverted fluorescence microscope (Diaphot; Nikon, Tokyo, Japan). Excitation was alternated between 340 and 380 nm using a dual-wavelength monochromator (Deltascan; Photon Technology Int., New Brunswick, NJ). The emission was monitored at 510 nm with a photomultiplier at an acquisition rate of 10 Hz per ratio, and the fluorescence ratio (F340:F380) values were determined. Cells were continuously washed by Tyrode’s solution (in mmol/L, 137 NaCl, 5.4 KCl, 0.5 MgCl2, 1.8 CaCl2, 11.8 Hepes-NaOH, 1 g/L glucose, pH 7.4, all from Sigma) using a slow background perfusion system, whereas the agents investigated were applied through a rapid perfusion system positioned in close proximity to the cell measured.
Proliferation Assay
Proliferation was measured by a colorimetric bromodeoxyuridine (BrdU) assay kit (Boehringer).8 Cells were plated in 96-well multititer plates (for the HaCaT cells, 5000 cells/well; for the ORS keratinocytes, 10,000 cells/well layered on 5000 cells/well mitomycin-treated HDFs) in quadruplicates and 4 hours later were treated with various compounds for 72 hours. Cells were then incubated with 10 μmol/L BrdU for 4 hours, and the cellular incorporation of BrdU was determined colorimetrically according to the manufacturer’s protocol. During the proliferation experiments, the following media were used (see above for detailed compositions): low-Ca SFM (low Ca2+ concentration, 0.4 mmol/L); high-Ca SFM (Ca2+ concentration was set to 2 mmol/L using CaCl2).
Flow Cytometry
The effect of TRPV1 activation on cellular proliferation, differentiation, apoptosis, and the expressions of various regulators of HF growth and development were assessed by flow cytometry. Control and capsaicin-treated ORS keratinocytes were immunostained with the following primary antibodies: Ki67 (mouse, 1:100; DAKO), proliferating cell nuclear antigen (mouse, 1:50; Novocastra, Newcastle on Tyne, UK), filaggrin (rabbit, 1:100; Covance), cytokeratin-14 (CK-14, mouse, 1:20; Novocastra), CK-17 (mouse, 1:20; Novocastra), involucrin (mouse, 1:50; Sigma), insulin-like growth factor-I (rabbit, 1:100; Santa Cruz), hepatic growth factor (1:20, rabbit; Santa Cruz), c-kit/stem cell factor (SCF, mouse, 1:50; Santa Cruz), TGF-β2 (mouse, 1:50; Santa Cruz), interferon-γ (IFN-γ) (goat, 1:20; Santa Cruz), interleukin (IL)-1β (goat, 1:50; Santa Cruz), tumor necrosis factor-α (rabbit, 1:20; BD Pharmingen, San Diego, CA), and fibroblast growth factor-5 (goat, 1:20; Santa Cruz). FITC-conjugated appropriate secondary antibodies (1:100, Vector) were then applied and, after fixation, cells were analyzed by a flow cytometer (EPICS XL-4; Coulter, Miami, FL). After isotype control calibration, the immunopositive signals were determined (at least 50,000 cells per antigen) and expressed as the percentage of total cell number. For the detection of apoptosis, cells were incubated with a FITC-conjugated annexin-V antibody (Sigma) and analyzed similarly.
Statistical Analysis
When applicable, data were analyzed using a two-tailed unpaired t-test and P < 0.05 values were regarded as significant differences.
Results and Discussion
Human Scalp HFs Express TRPV1 Mainly in the ORS and in the Hair Matrix
Recent reports suggest the expression of TRPV1 on human epidermal keratinocytes.11–15 However, given the substantial differences between epidermal and HF keratinocytes17,18,35 and the multiple changes in their gene expression patterns that keratinocytes are known to undergo after being placed in culture, we began by characterizing TRPV1 expression in the human HF in vivo, using full-thickness normal scalp skin obtained during routine plastic surgery.
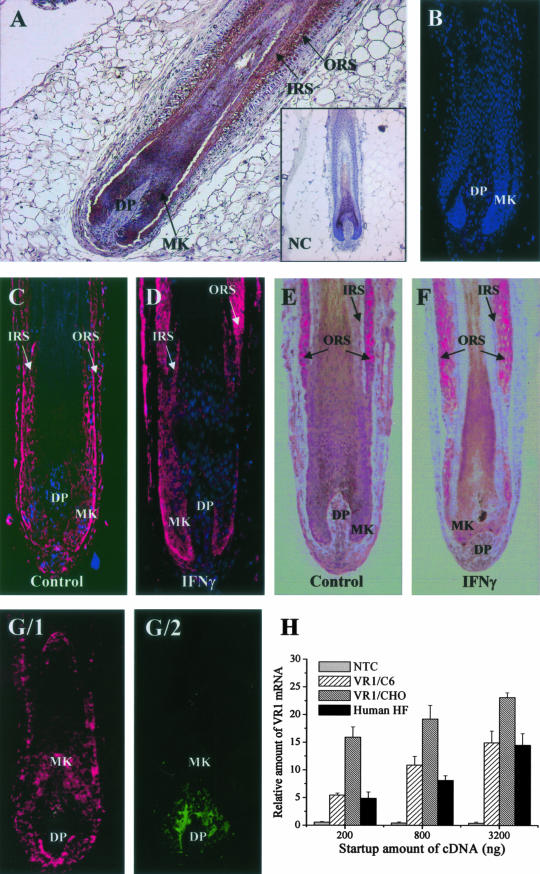
As revealed by immunohistochemistry, TRPV1-ir in human scalp HFs in anagen VI stage of the hair cycle17,18 was restricted to the follicle epithelium, where it was most prominent in the ORS and less pronounced in the hair matrix (Figure 1A). This specific (as assessed by numerous positive and negative controls, see Materials and Methods section; Figure 1A, inset)14 and marked TRPV1-ir was comparably high to that previously seen on epidermal keratinocytes of human skin.14 TRPV1-ir, however, was absent from the inductive, specialized HF mesenchyme, the fibroblasts of the dermal papilla (DP) (Figure 1A). An identical expression pattern of TRPV1-ir, as assessed by complementary TSA and AP techniques, was found on anagen VI HFs cultivated in organ cultures (Figure 1, C and E); ie, most intense signals were localized to the ORS and matrix keratinocytes and (yet much less intensively) to inner root sheath keratinocytes.
Figure 1.
TRPV1-ir on human skin and cultured human HFs. A: TRPV1-ir (with diaminobenzidine as a chromogen) on inner root sheath, ORS, and matrix (MK) keratinocytes (DP). Inset, negative control of TRPV1 staining. B: Negative control of TRPV1 staining. TSA (C, D) and AP (E, F) TRPV1-ir on cultured anagen (C, E) and catagen (IFN-γ-induced; D, F) human HFs. G/1, G/2: Double-fluorescence immunolabeling of TRPV1+ (red, G/1) and NKI/beteb+ (green, G/2) cells in the human cultured HFs. Note the lack of co-localization of TRPV1-ir on HF melanocytes. Nuclei were counterstained by DAPI (blue fluorescence in B–D; also in G, but not shown for clarity). H: Q-PCR analysis of TRPV1 expression in the human HF. Data of TRPV1 expression were normalized to the expression of GAPDH of the same sample and are expressed as relative mRNA amounts as a function of startup cDNA (relative amount of 1 was defined as the detection threshold). NTC, nontemplate (negative) control; TRPV1/C6 and TRPV1/CHO, C6 and CHO cells stably expressing the recombinant rat TRPV1 (positive controls). Values are mean ± SEM of three independent determinations. Original magnifications: ×100 (A), ×200 (B–G).
It has long been appreciated in the field that organ-cultured human HFs usually do not achieve more than early catagen development, before the follicle begins to degenerate.21,36 However, using IFN-γ as the strongest in vitro catagen inducer for human scalp HFs described so far, we have recently even been able induce mid-catagen stages.27,37 Using these results, it was intriguing to observe that TRPV1 expression significantly increased on cultured HFs (especially on the ORS keratinocytes) that were experimentally induced to undergo the early to mid stages of HF involution (catagen) phase by 1000 IU/ml IFN-γ (Figure 1, D and F) (catagen I/II was evident by light microscopy evaluation of morphological signs, up-regulation of TUNEL+ and down-regulation of Ki67+ keratinocytes of the hair matrix, along with termination of melanogenesis in the HF pigmentary unit, see below and in Stenn and Paus18 and Müller-Röver et al38). Image analysis revealed that the mean staining intensity of TRPV1 was by 32.3 ± 6.8% (mean ± SEM, P < 0.05, n = 20 to 30 HFs in each group) higher in the IFN-γ-treated (hence catagen I/II) HFs than in the control (anagen) ones (data not shown) suggesting that the expression of TRPV1 in these cells is in fact regulated by the cycling machinery of the HF.
To clarify whether some of the TRPV1-ir in the hair matrix reflected TRPV1 expression by the specialized melanocytes of the HF pigmentary unit,39 double-immunolabeling was performed with antibodies against TRPV1 and NKI/beteb, a very sensitive melanocyte marker (Figure 1G).31 Using this method, we found no co-localization of the two immunosignals suggesting that HF melanocytes, similar to our previous demonstration on human epidermal melanocytes in situ,14 also failed to express TRPV1. Therefore, both the mesoderm-derived DP fibroblast, and the neural crest-derived HF melanocytes did not express TRPV1 immunoreactivity.
To control whether intraepithelial TRPV1-ir corresponded to the presence of TRPV1 transcripts, RNA was rapidly extracted from freshly microdissected proximal human HF in the anagen VI stage of the hair cycle17–19 and subjected to Q-PCR. As shown in Figure 1H, mRNA transcripts for TRPV1 were unambiguously identified in normal human scalp HFs. Remarkably, the HFs expressed TRPV1 at very high levels, which were comparable to those of the two positive controls; ie, Chinese hamster ovary (CHO) and C6 cells recombinantly overexpressing the rat TRPV1.34 Therefore, in vivo the human HF epithelium indeed transcribes the TRPV1 gene at high levels, similarly to other nonneuronal epithelial and mesenchymal cell types.7–14
TRPV1 Stimulation Inhibits Hair Shaft Elongation and Hair Matrix Keratinocyte Proliferation
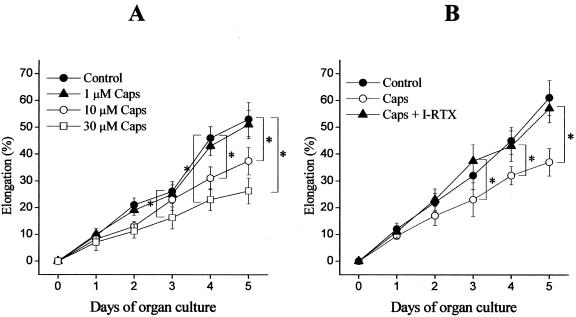
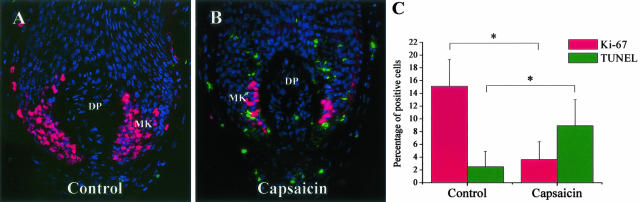
To explore the functional consequences of TRPV1 stimulation in vitro under as physiological conditions as possible, microdissected, organ-cultured normal anagen VI scalp HFs were exposed to the prototypic, most extensively studied, and clinically used TRPV1 agonist, capsaicin (0.1 to 30 μmol/L).1,5,6,15 Capsaicin significantly inhibited hair shaft elongation in a time- and dose-dependent manner (Figure 2A), as an indication of hair growth-inhibitory properties of TRPV1 signaling. Hair growth inhibition by capsaicin was further confirmed by the finding that treatment of cultured HFs by capsaicin for 5 days significantly decreased the number of Ki67+ keratinocytes in the anagen hair bulb (Figure 3).
Figure 2.
The activation of TRPV1 inhibits HF elongation. A: Cultured HFs (18 per group) were treated with either the vehicle (DMSO, control) or with various concentrations of capsaicin (Caps) for the time indicated. B: Cultures were treated with the vehicle (control), 10 μmol/L capsaicin (Caps), or 10 μmol/L capsaicin and 100 nmol/L iodo-RTX (Caps + I-RTX). Length measurements were made on individual HFs using a light microscope with an eyepiece measuring graticule. All data were compared with those of the control group, and analyzed by two-tailed unpaired t-test. Results are expressed as mean ± SD. *, Significant (P < 0.05) differences. Two to three additional experiments yielded similar results.
Figure 3.
The activation of TRPV1 inhibits proliferation and induces apoptosis in cultured HFs. Co-immunolabeling of proliferating (Ki-67+, red fluorescence) and apoptotic (TUNEL+, green fluorescence) cells on control (A) and capsaicin-treated (10 μmol/L for 5 days, B) cultured HFs. Nuclei were counterstained by DAPI (blue fluorescence). MK, matrix keratinocytes. C: Statistical analysis of number of Ki-67 and TUNEL+ cells as compared to the number of DAPI+ cells on several HFs per group. Data are expressed as mean ± SD. *, Significant (P < 0.05) differences. Original magnifications, ×400.
The inhibitory effect of capsaicin on hair shaft elongation was completely abolished by the specific TRPV1 antagonist iodo-resiniferatoxin (I-RTX, 100 nmol/L),40 demonstrating a TRPV1 specificity of the observed hair growth-inhibitory effect of capsaicin (Figure 2B). The antagonist alone only exerted a minor, statistically insignificant modulation of HF elongation (data not shown). These data unambiguously argue for the functional expression of TRPV1 in human organ-cultured scalp HFs.
TRPV1 Stimulation Up-Regulates HF Keratinocyte Apoptosis and Induces Premature Catagen Onset in Vitro
These findings were further supported by the observation that the number of TUNEL+ keratinocytes in the hair bulb significantly increased after capsaicin stimulation, suggesting the induction of apoptosis by TRPV1 stimulation (Figure 3). This inhibition of proliferation and stimulation of apoptosis by TRPV1 signaling in normal epithelial cells of the human HF in situ are perfectly in line with previous reports on the proliferation-inhibitory and apoptosis-stimulatory effects of TRPV1 activation for example in glial cells,8 and in airway9,41 and breast epithelial cells42 in vitro.
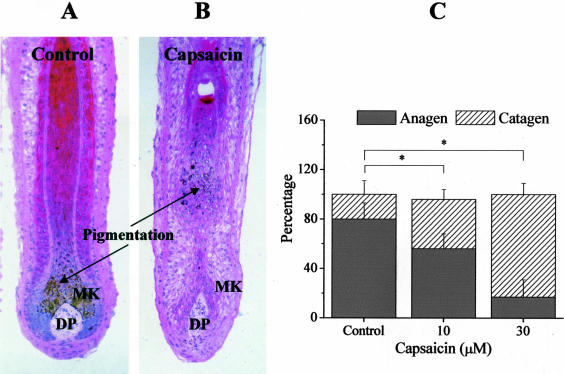
Using quantitative histomorphometry of H&E-staining section of HFs and analyzing such staining based on well-defined morphological criteria,18,38 we next assessed the effect of the TRPV1 agonist on HF cycling in vitro, namely on the anagen-catagen transition, which is characterized by massive up-regulation of keratinocyte apoptosis and down-regulation of keratinocyte proliferation in the anagen hair bulb.18,38,43 As shown in Figure 4, capsaicin stimulated the onset of catagen transformation of the HF; whereas at day 0, most of the HFs (∼80%) were in the anagen VI phase, after 5 days of treatment with 10 to 30 μmol/L capsaicin, ∼50 to 80% of the HFs had entered catagen (Figure 4C). It should be noted, however, that capsaicin was primarily capable of inducing only early catagen stages, and that we very rarely found that HF regression in vitro had progressed into late stages of the catagen transformation after application of the TRPV1 agonist.
Figure 4.
The activation of TRPV1 induces catagen transformation in cultured HFs. Cultured HFs were treated with either the vehicle (A) or with 10 μmol/L capsaicin for 5 days (B), then processed for H&E staining, and the percentage of HFs in anagen or catagen state, according to previously well-defined criteria (narrower hair bulb and depigmentation in catagen HFs), was determined (C). MK, matrix keratinocytes. Results are expressed as mean ± SEM. *, Significant (P < 0.05) differences. Two to three additional experiments yielded similar results. Original magnifications, ×100.
Characteristically, the onset of catagen is accompanied by an interruption of all pigmentary activity of HF melanocytes and by a gradual depigmentation of the lower HF,18,39 which was also evident in capsaicin-treated HFs (Figure 4, A and B). However, we were interested in learning whether TRPV1 activation affected pigmentation on HFs, which were not yet transformed to catagen. Since the first visible signs of catagen induction were seen at days 3 to 4 of capsaicin treatment, in the next experiments, the application of the TRPV1 agonist was terminated at day 3. Using Masson-Fontana histochemistry, we found that the activation of TRPV1 by capsaicin did not modify the pigmentation (data not shown), which was in accordance with our finding that the melanocytes of the HF pigmentary unit do not express TRPV1 (Figure 1F).
Terminal Differentiation of Human HF Keratinocytes in Situ Appears To Be Independent of TRPV1 Stimulation
Next, we investigated whether capsaicin also affected differentiation in the HF epithelium. Therefore, the expression of the keratinocyte differentiation marker filaggrin44 was studied. As before, cultured HFs were treated with capsaicin only for 3 days, and then immunohistochemistry was performed to detect filaggrin expression. As seen Figure 5, A and B, vanilloid application modified filaggrin expression only insignificantly: the mean fluorescence intensity values in the isthmus region were 76.8 ± 6.3 for the control and 80.3 ± 4.8 for the capsaicin-treated HFs (mean ± SEM, P > 0.5, n = 20 to 30 HFs in each group, data not shown). These data suggest that, in contrast to its effect on HF elongation (Figure 2), proliferation (Figure 3), cycling (Figure 4), and apoptosis (Figure 3), capsaicin, similarly to its in vitro action on cultured ORS keratinocytes (see below and Table 1), does not affect in situ differentiation of HF keratinocyte in a major way. Our findings, therefore, indicate that the hair growth-inhibitory effects of TRPV1 stimulation are chiefly based on a stimulation of hair matrix keratinocyte apoptosis and on inhibiting proliferation (Figure 3) of these cells. These results were in contrast with our previous findings on glial cells8 where the growth-inhibitory effect of capsaicin was accompanied by the modification of differentiation, suggesting that the characteristics of TRPV1 signaling show strong cell-type dependence (see also Concluding Remarks section).
Figure 5.
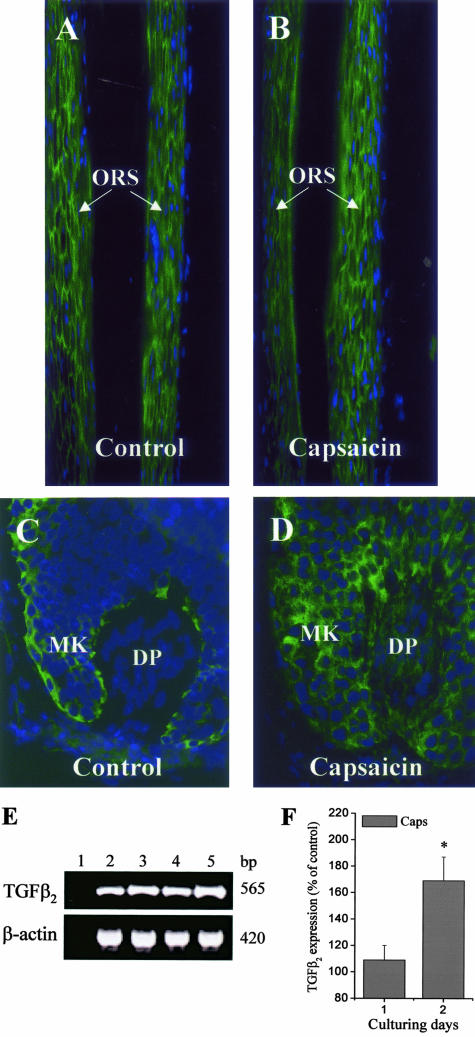
The activation of TRPV1 does not affect terminal differentiation but up-regulates TGF-β2 expression in cultured HFs. Fluorescence immunolabeling of differentiation marker filaggrin on control (A) and capsaicin-treated (10 μmol/L for 3 days, B) HFs (the isthmus region exhibiting the most intense staining is shown). Note the lack of effect of capsaicin on filaggrin expression (see text for statistics). Fluorescence immunolabeling of TGF-β2 on control (C) and capsaicin-treated (10 μmol/L for 2 days, D) HFs. MK, matrix keratinocytes. Note the up-regulation of TGF-β2 by capsaicin (see text for statistics). Nuclei were counterstained by DAPI (blue fluorescence). E: RT-PCR analysis of TGF-β2 (and β-actin, used as an internal control) mRNA expression in cultured HFs. Lane 1, reaction without template (negative control); lane 2, control at day 1; lane 3, 10 μmol/L capsaicin treated at day 1; lane 4, control at day 2; lane 5, 10 μmol/L capsaicin treated at day 2. bp, predicted product sizes. F: mRNA of transcripts were quantified by densitometry and the values of TGF-β2 were normalized to those of β-actin. Data of the capsaicin-treated groups, obtained in three independent experiments, are expressed as mean ± SEM values as a percentage of the daily matched control samples regarded as 100%. *, Significant (P < 0.05) differences. Original magnifications: ×100 (A, B); ×400 (C, D).
Table 1.
Effect of Capsaicin Treatment on the Expressions of Different Markers on ORS Keratinocytes
| Marker | Capsaicin (% of control) |
|---|---|
| Proliferation | |
| Ki-67 | 73.8 ± 7* (↓) |
| PCNA (proliferating cell nuclear antigen) | 71.6 ± 9* (↓) |
| Apoptosis | |
| Annexin-V | 143 ± 8* (↑) |
| Differentiation | |
| CK-14 (cytokeratin-14) | 94.2 ± 11 |
| CK-17 (cytokeratin-17) | 98.3 ± 6 |
| Filaggrin | 102.3 ± 8 |
| Involucrin | 95.1 ± 13 |
| Hair growth-promoting factors | |
| HGF (hepatic growth factor) | 63.5 ± 81* (↓) |
| IGF-I (insulin-like growth factor-I) | 53 ± 9* (↓) |
| SCF (c-kit/stem cell factor) | 74.9 ± 6* (↓) |
| Hair growth-inhibitory factors | |
| FGF-5 (fibroblast growth factor-5) | 95 ± 5 |
| IFN-γ (interferon-γ) | 91.3 ± 9 |
| IL-1β (interleukin-1β) | 143.7 ± 5* (↑) |
| TGF-β2 (transforming growth factor-β2) | 128.6 ± 6* (↑) |
| TNF-α (tumor necrosis factor-α) | 98.6 ± 4 |
Flow cytometry analyses were performed, as described under Materials and Methods, on control and capsaicin-treated (10 μmol/L for 2 days in high-Ca2+ SFM) ORS keratinocytes, and values of the treated samples were normalized as percentage of the control regarded as 100%. Data are expressed as mean ± SEM of three determinations.
Significant (P < 0.05) alterations.
The Hair Growth-Inhibitory Effects of TRPV1 Signaling May in Part Be Mediated via TGF-β2
The capsaicin-induced changes on cultured HFs were very similar to those described for TGF-β2, a well-known inhibitor of hair shaft elongation and inducer of both apoptosis and catagen in human HFs.21,29,45 Therefore, we investigated a possible involvement of TGF-β2 in the hair growth-inhibitory effect of TRPV1 stimulation. Capsaicin treatment for 2 to 3 days indeed markedly up-regulated the HF expression of TGF-β2, both at the protein (as revealed by immunohistochemistry, Figure 5, C and D) (at day 2, the mean fluorescence intensity values in the hair bulb region of the capsaicin-treated HFs were by 132.8 ± 13% higher than the control ones; mean ± SEM, P < 0.001, n = 20 to 30 HFs in each group, data not shown) and the mRNA level (as assessed by RT-PCR; Figure 5, E and F). In addition, co-culture of anagen HFs with capsaicin and TGF-β2-neutralizing antibodies partially abrogated the capsaicin-induced inhibition of hair shaft elongation (35.2 ± 6% suppression of the effect of capsaicin to inhibit hair shaft elongation; mean ± SEM, P < 0.05, n = 6, data not shown). These findings, ie, TRPV1 activation up-regulated the level of TGF-β2 in HFs that were not yet induced to transform to catagen and TGF-β2-neutralizing antibodies partially abrogated the TRPV1-mediated effects indicate that at least some of the hair growth-inhibitory effects of TRPV1 signaling in epithelial biology may be indirectly mediated by stimulation of the expression and secretion of TGF-β2. The activation of TRPV1 very often results in the release of various cytokines and growth modulatory agents (eg, interleukins, prostaglandins) on numerous cell types such as mast cells,7 bronchial epithelial cells,9 and keratinocytes.13 However, according to our knowledge, this is the first demonstration of the link between the TRPV1 and TGF-β2 signaling mechanisms. We should emphasize, however, the TRPV1-mediated alterations in organ-cultured human anagen VI HFs may indeed recruit other signaling pathways in addition to TGF-β2 and signaling via its cognate receptor (see also Concluding Remarks section).
Human Cultured ORS Keratinocytes, but Not DP Fibroblasts, Express TRPV1
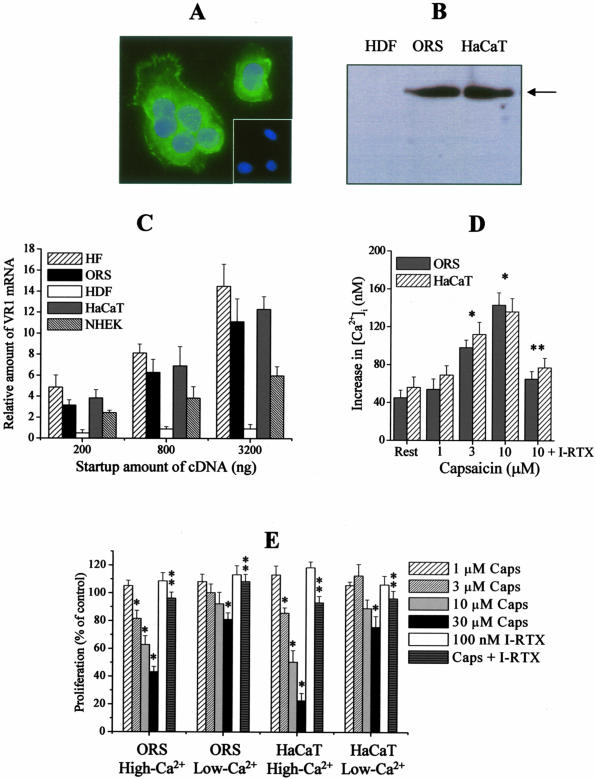
To double-check and further analyze the data obtained with intact, microdissected anagen HFs, primary cultures of human ORS keratinocytes were used, whereas HaCaT keratinocytes were used as positive controls, since the latter have previously been reported to express functional TRPV1.13 By immunocytochemistry (Figure 6A) and Western blotting (Figure 6B), we were able to present the first evidence that the TRPV1 protein is indeed expressed in normal ORS keratinocytes, and this at a level comparable to that of HaCaT keratinocytes (Figure 6B). Using Q-PCR, we could also detect TRPV1 mRNA transcripts in ORS keratinocytes and, as expected,11–13 in HaCaT cells and normal human epidermal keratinocytes (Figure 6C). In perfect agreement with our findings obtained with cultured HFs (Figure 1) and on human skin in situ,14 TRPV1 was undetectable in cultured HDFs, neither at the protein (Figure 6B) nor at the mRNA (Figure 6C) level.
Figure 6.
Identification of TRPV1 in cultured ORS and HaCaT keratinocytes, the activation of which elevates [Ca2+]i and inhibits proliferation. A: TRPV1-ir (green fluorescence) in cultured ORS keratinocytes. Nuclei were counterstained by DAPI (blue fluorescence). Inset, negative control of TRPV1 staining. B: Western blot analysis of TRPV1 expression on cell lysates of ORS and HaCaT keratinocytes, and of HDFs. Arrow indicates predicted molecule size (95 kd). C: Q-PCR analysis of TRPV1 expression in cultured cells and in the human HFs (for the human HFs, data are identical to those shown in Figure 1H). Data of TRPV1 expression were normalized to the expression of GAPDH of the same sample and are expressed as relative mRNA amounts as a function of startup cDNA (relative amount of 1 was defined as the detection threshold). NHEK, normal human epidermal keratinocytes. Values are mean ± SEM of three independent determinations. D: Fura-2 AM-loaded cells were challenged with various concentrations of capsaicin or with 10 μmol/L capsaicin and 100 nmol/L iodo-RTX (I-RTX) together in Tyrode’s solution containing 1.8 mmol/L calcium, and [Ca2+]i was determined as described in Materials and Methods. Data are expressed as mean ± SEM obtained on 15 to 20 cells per group. *, Significant (P < 0.05) differences compared to the resting [Ca2+]i. **, Significant (P < 0.05) effect of I-RTX to prevent the action of 10 μmol/L capsaicin to increase [Ca2+]i. E: A BrdU proliferation assay. Cells were plated in 96-well multititer plates in quadruplicates and were treated with various concentrations of capsaicin (Caps) or with 30 μmol/L capsaicin and 100 nmol/L iodo-RTX (I-RTX) together for 72 hours. For determining the extracellular calcium dependence, experiments were performed in low-Ca2+ SFM (0.4 mmol/L) and high-Ca2+ SFM (2 mmol/L). Data are expressed as mean ± SEM as a percentage of the matched control values regarded as 100%. *, Significant (P < 0.05) differences compared to control (vehicle-treated) proliferation. **, Significant (P < 0.05) effect of I-RTX to prevent the action of 30 μmol/L capsaicin to inhibit proliferation. Original magnification, ×630 (A).
TRPV1 Stimulation Inhibits the Proliferation, Elevates [Ca2+]i, and Induces Apoptosis in Cultured ORS and HaCaT Keratinocytes
On sensory neurons and several nonneuronal cell types (including HaCaT cells), TRPV1 functions as a Ca2+-permeable cation channel.1,2,5,7–9,12,13 Hence, we also tested whether the application of capsaicin affects the intracellular Ca2+ homeostasis in ORS keratinocytes. As revealed by optical Ca2+ imaging, capsaicin dose-dependently increased [Ca2+]i in ORS keratinocytes (similarly to HaCaT cells) in Tyrode’s solution containing 1.8 mmol/L Ca2+ (Figure 6D). In contrast, the TRPV1 antagonist I-RTX prevented the [Ca2+]i-increasing action of capsaicin, demonstrating that this is a TRPV1-mediated event. It is important to note that none of the studied concentrations of capsaicin was able to significantly modify [Ca2+]i of ORS or HaCaT keratinocytes in vitro when calcium was removed from the Tyrode’s solution (data not shown). This supports the concept that the capsaicin-induced elevation in [Ca2+]i originated chiefly from the extracellular space via TRPV1.
We also measured the effect of TRPV1 activation on ORS and HaCaT keratinocyte proliferation using BrdU enzyme-linked immunosorbent assays. In addition, because TRPV1-induced cellular events on sensory neurons (eg, excitation, desensitization, and neurotoxicity) and on nonneuronal cell types (eg, cytokine release) are chiefly initiated by the influx of Ca2+ via TRPV1,1,2,5,7–9,13 we also investigated the extracellular Ca2+ dependence of TRPV1 activation (this was impossible to measure in the case of organ-cultured HFs that did not grow and eventually died in low-Ca2+ solutions, data not shown). As seen in (Figure 6E), in high-Ca2+ (2 mmol/L) SFM, capsaicin dose-dependently inhibited proliferation of ORS and HaCaT keratinocytes which effect was prevented by I-RTX. However, as a marked contrast, the TRPV1 agonist exerted a much less growth-inhibitory effect in low-Ca2+ (0.4 mmol/L) SFM. These data strongly argued for that the TRPV1-evoked cellular responses in cultured keratinocytes (and presumably in cultured HFs) are mediated by the concomitant elevation of [Ca2+]i.
These assays were complemented with a wide-spectrum flow cytometry analysis of alterations in the levels of selected proliferation and apoptosis markers on capsaicin treatment of cultured ORS keratinocytes (Table 1). Consistent with the above findings (Figures 3 and 6), capsaicin (10 μmol/L for 2 days in high-Ca2+ SFM) significantly suppressed the expression of Ki67 and proliferating cell nuclear antigen also suggesting inhibition of proliferation. Moreover, the TRPV1 agonist elevated the number of annexin-V+ cells reflecting stimulation of apoptosis (similarly to that seen in the HF, Figure 3). However, also similarly to data obtained with the cultured HFs (Figure 5, A and B), capsaicin did not modify the expression of numerous differentiation markers (Table 1).45–47 These data indicate that the activation of TRPV1 signaling both on organ-cultured HFs and on primary ORS keratinocytes results in the inhibition of proliferation and induction of apoptosis, with an insignificant effect on the process of differentiation.
TRPV1 Stimulation in ORS Keratinocytes Down-Regulates the Expression of Hair Growth-Stimulatory Factors but Up-Regulates the Expression of Hair Growth Inhibitors
Finally, we investigated whether and how TRPV1 stimulation by capsaicin application altered the protein expressions of various cytokines and growth factors that are well appreciated to positively or negatively regulate HF growth (Table 1). Flow cytometry analysis revealed that vanilloid treatment significantly down-regulated the ORS keratinocyte expressions of key promoters of HF growth such as hepatocyte growth factor,18,30,48 insulin-like growth factor-I,18,49 and stem cell factor.18,50 In contrast, capsaicin up-regulated the levels of IL-1β and TGF-β2, well-known potent hair growth inhibitors18,21,29,45,51 whereas the expressions of other hair growth inhibitory agents such as fibroblast growth factor-5, IFN-γ, and tumor necrosis factor-α18,27,52,53 were not affected.
Concluding Remarks
Taken together, the data reported here introduce vanilloid receptors as significant new players in human hair growth control, with TRPV1-mediated signaling exerting profound effects on HF keratinocyte proliferation and apoptosis, and directly and/or indirectly manipulating a number of well-recognized hair growth modulators. This underscores that the physiological functions of TRPV1 and its elusive endogenous ligands, in human skin and likely other mammalian tissues as well, far extend beyond sensory neuron-coupled nociception. Thus TRPV1 joins the group of other receptors (eg, nicotinic and muscarinic acetylcholine receptors, tachykinin, or glutamate receptors) that were originally described as neuron-specific signal transducers, but later were identified to be functionally active on numerous nonneuronal cell types, including those present in human skin and its appendages.54–56
The concept that TRPV1 signaling targets previously unappreciated cellular mechanisms is supported by most recent preliminary data that we have obtained in a pilot MicroArray assay (using a commercially available focus chip that contains 1300 genes with recognized relevance in investigative dermatology). Namely, capsaicin treatment (10 μmol/L for 2 days) of organ-cultured human scalp HFs resulted in the induction of such genes of agents inhibiting HF growth as insulin-like growth factor-I-binding protein-3,57 growth/differentiation factor-15/macrophage inhibitory cytokine-1/placental TGF-β,58 and activin βA,59 whereas TRPV1 activation down-regulated the gene expression of matrix GLA-protein precursor (MGP) that is an inhibitor of bone morphogenic protein-2/TGF-β signaling in the HF.59 Although these preliminary results remain to be repeated and confirmed by Q-PCR, they already point to an even wider spectrum of direct or indirect target genes of TRPV1 signaling that is apparent from the current study.
Interestingly, although the cellular actions affected by TRPV1 activation showed clear calcium dependence (ie, capsaicin was much less effective in inhibiting ORS keratinocyte proliferation in low-Ca2+ SFM, Figure 6E), capsaicin did not significantly affect terminal differentiation of cultured ORS keratinocytes, a process that is also dependent on extracellular [Ca2+] in the skin.60 These data might be explained by several arguments, such as: 1) the induction of catagen and, therefore, the processes of apoptosis (along with the inhibition of proliferation) were initiated earlier by the activation of TRPV1 than possible alterations in expression of differentiation markers; 2) the high calcium concentration of the culturing medium (close to 2 mmol/L), that was required to maintain the HF in organ-culture, alone induced increased expression of the differentiation markers filaggrin (Figure 5A); hence, the TRPV1-mediated calcium influx was unable to further modify (presumably increase) this elevated level (Figure 5B); 3) the effect of capsaicin of various cellular mechanisms possesses a strong dose and cell-type dependence, as reflected by differential action, eg, on stimulation of cytokine release (in keratinocytes, IL-8;13 in mast cells, IL-4 but not tumor necrosis factor-α7), cell death (apoptosis at small concentrations in thymocytes;61 apoptosis in glial cells,8 and in airway9,41 and breast epithelial cells;42 necrotic cell death1 in neurons and, at high doses, on thymocytes61), and, as revealed by current findings, on differentiation (modification of differentiation on glial cells8 but not of ORS keratinocytes, Figure 5); 4) finally, given that capsaicin treatment up-regulates the expression of hair growth inhibitors (such as TGF-β2 and IL-1β) whereas down-modulates the levels of the hair growth stimulators hepatocyte growth factor, insulin-like growth factor-I, and stem cell factor (Figure 5, Table 1), it appears that TRPV1 stimulation results in a complex alterations of the cytokine network of human HFs, the net effects of which eventually coalesce in the cellular HF changes described in Figures 2 to 6.
The well-defined, close physical association of TRPV1-expressing sensory nerve fibers with the TRPV1-expressing ORS keratinocytes of the HF,62,63 along with other TRPV1-positive nonneuronal cell types such as, eg, mast cells and Langerhans cells,7,14,15 invite an intriguing hypothesis with potential therapeutic implication. Namely, the dual activation of TRPV1 by exogenous capsaicin or endovanilloids (such as eicosanoids and prostaglandins that are also found in the HF64) on neuronal and nonneuronal cell types of the skin likely results in the simultaneous release of neuropeptides from sensory axons and of other mediators (eg, histamine, proinflammatory cytokines) from HF cells, presumably from ORS keratinocytes. Given the recognized hair growth-modulatory properties of such sensory neuron-derived neuropeptides as substance P and calcitonin gene-related peptides16,65,66 and the involvement of TRPV1-expressing mast cells7,14 in hair growth,67,68 this could activate complex, multidirectional signaling cascades including augmentation of the action of the TRPV1 agonists and mast cell degranulation both via TRPV1 and certain neuropeptide receptors such as NK1. The activation of such complex cascades may also explain why TRPV1 stimulation by capsaicin, under defined circumstances (various applied concentrations, telogen versus anagen HF stages, species differences), can even stimulate hair growth in mice,65 ie, can induce anagen in telogen HF in vivo (note that in the current in vitro study the hair growth-inhibitory effects of capsaicin were seen on human scalp HFs that exhibited maximal growth activity, ie, anagen VI). Furthermore, preliminary observations from our currently ongoing studies in TRPV1 knockout mice14 (such as alterations in HF cycling, effects on HF keratinocytes proliferation and perifollicular mast cells) appears to be also consistent with the concept that TRPV1 signaling is indeed a key molecular mechanism in hair biology (Bodó and colleagues, study in progress).
Taken together, the current study not only gives a hot new twist to human hair growth control by introducing TRPV1 signaling as a potent, physiologically relevant hair growth-inhibitory force that may be clinically exploited, eg, for the treatment of unwanted hair growth (hirsutism) by the topical application of TRPV1 agonists, and of hair loss (effluvium, alopecia) by administering TRPV1 antagonists. Our study also highlights that TRPV1 signaling is a major, newly recognized player in epithelial biology in general—both by its direct effect on epithelial tissues (eg, epidermal and ORS keratinocytes) and its complex indirect effects on neuroectodermal-mesenchymal interaction (eg, via the modification of neuropeptide release from sensory skin nerves and of skin mast cell activation). This invites one to systemically explore in future studies how the anti-proliferative TRPV1 signaling can be manipulated in a clinically desired manner by endogenous and exogenous ligands in the management of hyperproliferative epithelial growth disorders of the skin (eg, psoriasis, actinic keratosis, keratoacanthoma, and squamous cell carcinoma) and elsewhere. Therefore, clinicians who apply agents that activate and/or sensitize TRPV1 (eg, capsaicin or resiniferatoxin1,5,6,14,15,69) now need to take the above into account.
Acknowledgments
We thank S. Wegerich, G. Pilnitz-Stolze, and M. Dietrich for technical assistance; Dr. B. Gerstmayer (Memorec Biotec GmbH, Cologne, Germany) for performance of the MicroArray assay; and P. Pertile (Cutech Srl, Venice, Italy) for continuous consultation.
Footnotes
Address reprint requests to Tamás Bíró, M.D., Ph.D., Department of Physiology, University of Debrecen, MHSC, 4012 Debrecen, Nagyerdei krt. 98., PO Box 22, Hungary. E-mail: biro@phys.dote.hu.
Supported in part by grants from the Deutsche Forschungsgemeinschaft (Pa 345/11-1 to R.P.), Cutech Srl (to R.P.), Hungarian research grants (OTKA F035036, OTKA TS040773, NKFP 00088/2001, OMFB 00200/2002, ETT 365/2003 to T.B.), the European Union (Erasmus fellowship to E.B.), the Hungarian Ministry of Education (György Békésy postdoctoral scholarship to T.B.), and NATO (science fellowship to T.B.).
E.B. and T.B. contributed equally to this work.
References
- Szallasi A, Blumberg PM. Vanilloid (capsaicin) receptors and mechanisms. Pharmacol Rev. 1999;51:159–212. [PubMed] [Google Scholar]
- Caterina MJ, Schumacher MA, Tominaga M, Rosen TA, Levine JD, Julius D. The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature. 1997;389:816–824. doi: 10.1038/39807. [DOI] [PubMed] [Google Scholar]
- Ugawa S, Ueda T, Ishida Y, Nishigaki M, Shibata Y, Shimada S. Amiloride-blockable acid-sensing ion channels are leading acid sensors expressed in human nociceptors. J Clin Invest. 2002;110:1185–1190. doi: 10.1172/JCI15709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Marzo V, Blumberg PM, Szallasi A. Endovanilloid signaling in pain. Curr Opin Neurobiol. 2002;12:372–379. doi: 10.1016/s0959-4388(02)00340-9. [DOI] [PubMed] [Google Scholar]
- Karai L, Brown DC, Mannes AJ, Connelly ST, Brown J, Gandal M, Wellisch OM, Neubert JK, Olah Z, Iadarola MJ. Deletion of vanilloid receptor 1-expressing primary afferent neurons for pain control. J Clin Invest. 2004;113:1344–1352. doi: 10.1172/JCI20449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bíró T, Ács G, Ács P, Modarres S, Blumberg PM. Recent advances in understanding of vanilloid receptors: a therapeutic target for treatment of pain and inflammation in skin. J Invest Dermatol Symp Proc. 1997;2:56–60. doi: 10.1038/jidsymp.1997.12. [DOI] [PubMed] [Google Scholar]
- Bíró T, Maurer M, Modarres S, Lewin NE, Brodie C, Ács G, Ács P, Paus R, Blumberg PM. Characterization of functional vanilloid receptors expressed by mast cells. Blood. 1998;91:1332–1340. [PubMed] [Google Scholar]
- Bíró T, Brodie C, Modarres S, Lewin NE, Ács P, Blumberg PM. Specific vanilloid responses in C6 rat glioma cells. Mol Brain Res. 1998;56:89–98. doi: 10.1016/s0169-328x(98)00033-3. [DOI] [PubMed] [Google Scholar]
- Veronesi B, Oortgiesen M, Carter JD, Devlin RB. Particulate matter initiates inflammatory cytokine release by activation of capsaicin and acid receptors in a human bronchial epithelial cell line. Toxicol Appl Pharmacol. 1999;154:106–115. doi: 10.1006/taap.1998.8567. [DOI] [PubMed] [Google Scholar]
- Birder LA, Kanai AJ, de Groat WC, Kiss S, Nealen ML, Burke NE, Dineley KE, Watkins S, Reynolds IJ, Caterina MJ. Vanilloid receptor expression suggests a sensory role for urinary bladder epithelial cells. Proc Natl Acad Sci USA. 2001;98:13396–13401. doi: 10.1073/pnas.231243698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Denda M, Fuziwara S, Inoue K, Denda S, Akamatsu H, Tomitaka A, Matsunaga H. Immunoreactivity of TRPV1 on epidermal keratinocyte of human skin. Biochem Biophys Res Commun. 2001;285:1250–1252. doi: 10.1006/bbrc.2001.5299. [DOI] [PubMed] [Google Scholar]
- Inoue K, Koizumi S, Fuziwara S, Denda S, Inoue K, Denda M. Functional vanilloid receptors in cultured normal human epidermal keratinocytes. Biochem Biophys Res Commun. 2002;291:124–129. doi: 10.1006/bbrc.2002.6393. [DOI] [PubMed] [Google Scholar]
- Southall MD, Li T, Gharibova LS, Pei Y, Nicol GD, Travers JB. Activation of epidermal vanilloid receptor-1 induces release of proinflammatory mediators in human keratinocytes. J Pharmacol Exp Ther. 2003;30:217–222. doi: 10.1124/jpet.102.040675. [DOI] [PubMed] [Google Scholar]
- Bodó E, Kovács I, Telek A, Varga A, Paus R, Kovács L, Bíró T. Vanilloid receptor-1 is widely expressed on various epithelial and mesenchymal cell types of human skin. J Invest Dermatol. 2004;123:410–413. doi: 10.1111/j.0022-202X.2004.23209.x. [DOI] [PubMed] [Google Scholar]
- Ständer S, Moormann C, Schumacher M, Buddenkotte J, Artuc M, Shpacovitch V, Brzoska T, Lippert U, Henz BM, Luger TA, Metze D, Steinhoff M. Expression of vanilloid receptor subtype 1 in cutaneous sensory fibers, mast cells, and epithelial cells of appendage structures. Exp Dermatol. 2004;13:129–139. doi: 10.1111/j.0906-6705.2004.0178.x. [DOI] [PubMed] [Google Scholar]
- Paus R, Peters EM, Eichmüller S, Botchkarev VA. Neural mechanisms of hair growth control. J Invest Dermatol Symp Proc. 1997;2:61–68. doi: 10.1038/jidsymp.1997.13. [DOI] [PubMed] [Google Scholar]
- Paus R, Cotsarelis G. The biology of hair follicles. N Engl J Med. 1999;341:491–497. doi: 10.1056/NEJM199908123410706. [DOI] [PubMed] [Google Scholar]
- Stenn KS, Paus R. Controls of hair follicle cycling. Physiol Rev. 2001;81:449–494. doi: 10.1152/physrev.2001.81.1.449. [DOI] [PubMed] [Google Scholar]
- Cotsarelis G, Millar SE. Towards a molecular understanding of hair loss and its treatment. Trends Mol Med. 2001;7:293–301. doi: 10.1016/s1471-4914(01)02027-5. [DOI] [PubMed] [Google Scholar]
- Botchkarev VA, Botchkareva NV, Peters EM, Paus R. Epithelial growth control by neurotrophins: leads and lessons from the hair follicle. Prog Brain Res. 2004;146:493–513. doi: 10.1016/S0079-6123(03)46031-7. [DOI] [PubMed] [Google Scholar]
- Philpott MP, Green MR, Kealey T. Human hair growth in vitro. J Cell Sci. 1990;3:463–471. doi: 10.1242/jcs.97.3.463. [DOI] [PubMed] [Google Scholar]
- Limat A, Noser F. Serial cultivation of single keratinocytes from the outer root sheath of human scalp hair follicles. J Invest Dermatol. 1986;87:485–488. doi: 10.1111/1523-1747.ep12455548. [DOI] [PubMed] [Google Scholar]
- Limat A, Hunziker T, Biollat C, Bayreuther K, Noser F. Post-mitotic human dermal fibroblasts efficiently support the growth of human follicular keratinocytes. J Invest Dermatol. 1989;92:758–762. doi: 10.1111/1523-1747.ep12722530. [DOI] [PubMed] [Google Scholar]
- Barbosa AJ, Castro LP, Margarida A, Nogueira MF. A simple and economical modification of the Masson-Fontana method for staining melanin granules and enterochromaffin cells. Stain Technol. 1984;59:193–196. doi: 10.3109/10520298409113855. [DOI] [PubMed] [Google Scholar]
- Paus R, van der Veen C, Eichmüller S, Kopp T, Hagen E, Müller-Röver S, Hofmann U. Generation and cyclic remodeling of the hair follicle immune system in mice. J Invest Dermatol. 1998;111:7–18. doi: 10.1046/j.1523-1747.1998.00243.x. [DOI] [PubMed] [Google Scholar]
- Botchkarev VA, Botchkareva NV, Albers KM, Chen LH, Welker P, Paus R. A role for p75 neurotrophin receptor in the control of apoptosis-driven hair follicle regression. FASEB J. 2000;14:1931–1942. doi: 10.1096/fj.99-0930com. [DOI] [PubMed] [Google Scholar]
- Ito T, Ito N, Bettermann A, Tokura Y, Takigawa M, Paus R. Collapse and restoration of MHC class-I-dependent immune privilege: exploiting the human hair follicle as a model. Am J Pathol. 2004;164:623–634. doi: 10.1016/S0002-9440(10)63151-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Panteleyev AA, Botchkareva NV, Sundberg JP, Christiano AM, Paus R. The role of the hairless (hr) gene in the regulation of hair follicle catagen transformation. Am J Pathol. 1999;155:159–171. doi: 10.1016/S0002-9440(10)65110-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Foitzik K, Lindner G, Müller-Röver S, Maurer M, Botchkareva N, Botchkarev V, Handjiski B, Metz M, Hibino T, Soma T, Dotto GP, Paus R. Control of murine hair follicle regression (catagen) by TGF-beta1 in vivo. FASEB J. 2000;14:752–760. doi: 10.1096/fasebj.14.5.752. [DOI] [PubMed] [Google Scholar]
- Lindner G, Menrad A, Gharadi E, Merlino G, Welkes P, Handjinski B, Roloff B, Paus R. Involvement of hepatocyte growth factor/scatter factor and met receptor signaling in hair follicle morphogenesis and cycling. FASEB J. 2000;14:319–332. doi: 10.1096/fasebj.14.2.319. [DOI] [PubMed] [Google Scholar]
- Vennegoor C, Hageman P, Van Nouhuijs H, Ruiter DJ, Calafat J, Ringens PJ, Rumke P. A monoclonal antibody specific for cells of the melanocyte lineage. Am J Pathol. 1988;130:179–192. [PMC free article] [PubMed] [Google Scholar]
- Botchkareva NV, Botchkarev VA, Chen LH, Lindner G, Paus R. A role for p75 neurotrophin receptor in the control of hair follicle morphogenesis. Dev Biol. 1999;216:135–153. doi: 10.1006/dbio.1999.9464. [DOI] [PubMed] [Google Scholar]
- Kjellman C, Olofsson SP, Hansson O, Von Schantz T, Lindvall M, Nilsson I, Salford LG, Sjogren HO, Widegren B. Expression of TGF-beta isoforms, TGF-beta receptors, and SMAD molecules at different stages of human glioma. Int J Cancer. 2000;89:251–258. doi: 10.1002/1097-0215(20000520)89:3<251::aid-ijc7>3.0.co;2-5. [DOI] [PubMed] [Google Scholar]
- Lázár J, Szabó T, Kovács L, Blumberg PM, Bíró T. Distinct features of recombinant rat vanilloid receptor-1 expressed in various expression systems. Cell Mol Life Sci. 2003;60:2228–2240. doi: 10.1007/s00018-003-3230-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Panteleyev AA, Jahoda CA, Christiano AM. Hair follicle predetermination. J Cell Sci. 2001;114:3419–3431. doi: 10.1242/jcs.114.19.3419. [DOI] [PubMed] [Google Scholar]
- Philpott MP, Sanders DA, Kealey T. Whole hair follicle organ culture. Dermatol Clin. 1996;14:595–608. doi: 10.1016/s0733-8635(05)70387-9. [DOI] [PubMed] [Google Scholar]
- Ito T, Ito N, Saathoff M, Bettermann A, Takigawa M, Paus R: IFN-γ is a potent inducer of catagen-like changes in cultured human anagen hair follicles. Br J Dermatol (in press) [DOI] [PubMed] [Google Scholar]
- Müller-Röver S, Handjiski B, van der Veen C, Eichmüller S, Foitzik K, McKay IA, Stenn KS, Paus R. A comprehensive guide for the accurate classification of murine hair follicles in distinct hair cycle stages. J Invest Dermatol. 2001;117:3–15. doi: 10.1046/j.0022-202x.2001.01377.x. [DOI] [PubMed] [Google Scholar]
- Tobin DJ, Paus R. Graying: gerontobiology of the hair follicle pigmentary unit. Exp Gerontol. 2001;36:29–54. doi: 10.1016/s0531-5565(00)00210-2. [DOI] [PubMed] [Google Scholar]
- Wahl P, Foged C, Tullin S, Thomsen C. Iodo-resiniferatoxin, a new potent vanilloid receptor antagonist. Mol Pharmacol. 2001;59:9–15. doi: 10.1124/mol.59.1.9. [DOI] [PubMed] [Google Scholar]
- Agopyan N, Head J, Yu S, Simon SA. TRPV1 receptors mediate particulate matter-induced apoptosis. Am J Physiol. 2004;286:L563–L572. doi: 10.1152/ajplung.00299.2003. [DOI] [PubMed] [Google Scholar]
- Kang HJ, Soh Y, Kim MS, Lee EJ, Surh YJ, Kim HR, Kim SH, Moon A. Roles of JNK-1 and p38 in selective induction of apoptosis by capsaicin in ras-transformed human breast epithelial cells. Int J Cancer. 2003;103:475–482. doi: 10.1002/ijc.10855. [DOI] [PubMed] [Google Scholar]
- Lindner G, Botchkarev VA, Botchkareva NV, Ling G, van der Veen C, Paus R. Analysis of apoptosis during hair follicle regression (catagen). Am J Pathol. 1997;151:1601–1617. [PMC free article] [PubMed] [Google Scholar]
- Lenoir MC, Bernard BA, Pautrat G, Darmon M, Shroot B. Outer root sheath cells of human hair follicle are able to regenerate a fully differentiated epidermis in vitro. Dev Biol. 1988;130:610–620. doi: 10.1016/0012-1606(88)90356-9. [DOI] [PubMed] [Google Scholar]
- Soma T, Tsuji Y, Hibino T. Involvement of transforming growth factor-beta2 in catagen induction during the human hair cycle. J Invest Dermatol. 2002;118:993–997. doi: 10.1046/j.1523-1747.2002.01746.x. [DOI] [PubMed] [Google Scholar]
- Langbein L, Rogers MA, Winter H, Praetzel S, Beckhaus U, Rackwitz HR, Schweizer J. The catalog of human hair keratins. I. Expression of the nine type I members in the hair follicle. J Biol Chem. 1999;28:19874–19884. doi: 10.1074/jbc.274.28.19874. [DOI] [PubMed] [Google Scholar]
- Langbein L, Rogers MA, Winter H, Praetzel S, Schweizer J. The catalog of human hair keratins. II. Expression of the six type II members in the hair follicle and the combined catalog of human type I and II keratins. J Biol Chem. 2001;37:35123–35132. doi: 10.1074/jbc.M103305200. [DOI] [PubMed] [Google Scholar]
- Shimaoka S, Tsuboi R, Jindo T, Imai R, Takamori K, Rubin JS, Ogawa H. Hepatocyte growth factor/scatter factor expressed in follicular papilla cells stimulates human hair growth in vitro. J Cell Physiol. 1995;165:333–338. doi: 10.1002/jcp.1041650214. [DOI] [PubMed] [Google Scholar]
- Philpott MP, Sanders DA, Kealey T. Effects of insulin and insulin-like growth factors on cultured human hair follicles: IGF-I at physiologic concentrations is an important regulator of hair follicle growth in vitro. J Invest Dermatol. 1994;102:857–861. doi: 10.1111/1523-1747.ep12382494. [DOI] [PubMed] [Google Scholar]
- Peters EM, Maurer M, Botchkarev VA, Jensen K, Welker P, Scott GA, Paus R. Kit is expressed by epithelial cells in vivo. J Invest Dermatol. 2003;121:976–984. doi: 10.1046/j.1523-1747.2003.12478.x. [DOI] [PubMed] [Google Scholar]
- Hoffmann R, Eicheler W, Wenzel E, Happle R. Interleukin-1beta-induced inhibition of hair growth in vitro is mediated by cyclic AMP. J Invest Dermatol. 1997;108:40–42. doi: 10.1111/1523-1747.ep12285625. [DOI] [PubMed] [Google Scholar]
- Hebert JM, Rosenquist T, Gotz J, Martin GR. FGF5 as a regulator of the hair growth cycle: evidence from targeted and spontaneous mutations. Cell. 1994;78:1017–1025. doi: 10.1016/0092-8674(94)90276-3. [DOI] [PubMed] [Google Scholar]
- Paus R, Ito N, Takigawa M, Ito T. The hair follicle and immune privilege. J Invest Dermatol Symp Proc. 2003;8:188–194. doi: 10.1046/j.1087-0024.2003.00807.x. [DOI] [PubMed] [Google Scholar]
- Nguyen VT, Ndoye A, Hall LL, Zia S, Arredondo J, Chernyavsky AI, Kist DA, Zelickson BD, Lawry MA, Grando SA. Programmed cell death of keratinocytes culminates in apoptotic secretion of a humectant upon secretagogue action of acetylcholine. J Cell Sci. 2001;114:1189–1204. doi: 10.1242/jcs.114.6.1189. [DOI] [PubMed] [Google Scholar]
- Kanda N, Watanabe S. Substance P enhances the production of interferon-induced protein of 10 kDa by human keratinocytes in synergy with interferon-gamma. J Invest Dermatol. 2002;119:1290–1297. doi: 10.1046/j.1523-1747.2002.19626.x. [DOI] [PubMed] [Google Scholar]
- Genever PG, Maxfield SJ, Kennovin GD, Maltman J, Bowgen CJ, Raxworthy MJ, Skerry TM. Evidence for a novel glutamate-mediated signaling pathway in keratinocytes. J Invest Dermatol. 1999;112:337–342. doi: 10.1046/j.1523-1747.1999.00509.x. [DOI] [PubMed] [Google Scholar]
- Hembree JR, Harmon CS, Nevins TD, Eckert RL. Regulation of human dermal papilla cell production of insulin-like growth factor binding protein-3 by retinoic acid, glucocorticoids, and insulin-like growth factor-1. J Cell Physiol. 1996;167:556–561. doi: 10.1002/(SICI)1097-4652(199606)167:3<556::AID-JCP19>3.0.CO;2-1. [DOI] [PubMed] [Google Scholar]
- Welsh JB, Sapinoso LM, Kern SG, Brown DA, Liu T, Bauskin AR, Ward RL, Hawkins NJ, Quinn DI, Russell PJ, Sutherland RL, Breit SN, Moskaluk LA, Frierson HF, Jr, Hampton GM. Large-scale delineation of secreted protein biomarkers overexpressed in cancer tissue and serum. Proc Natl Acad Sci USA. 2003;100:3410–3415. doi: 10.1073/pnas.0530278100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakamura M, Matzuk MM, Gerstmayer B, Bosio A, Lauster R, Miyachi Y, Werner S, Paus R. Control of pelage hair follicle development and cycling by complex interactions between follistatin and activin. FASEB J. 2003;17:497–499. doi: 10.1096/fj.02-0247fje. [DOI] [PubMed] [Google Scholar]
- Bikle DD, Ratnam A, Mauro T, Harris J, Pillai S. Changes in calcium responsiveness and handling during keratinocyte differentiation. Potential role of the calcium receptor. J Clin Invest. 1996;97:1085–1093. doi: 10.1172/JCI118501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amantini C, Mosca M, Luccarini R, Perfumi M, Morrone S, Piccoli M, Santoni G. Distinct thymocyte subsets express the vanillioid receptor VR1 that mediates capsaicin-induced apoptotic cell death. Cell Death Differ. 2004;11:1342–1356. doi: 10.1038/sj.cdd.4401506. [DOI] [PubMed] [Google Scholar]
- Kaidoh T, Inoue T. Intercellular junctions between palisade nerve endings and outer root sheath cells of rat vellus hairs. J Comp Neurol. 2000;420:419–427. doi: 10.1002/(sici)1096-9861(20000515)420:4<419::aid-cne1>3.0.co;2-3. [DOI] [PubMed] [Google Scholar]
- Peters EM, Botchkarev VA, Müller-Röver S, Moll I, Rice FL, Paus R. Developmental timing of hair follicle and dorsal skin innervation in mice. J Comp Neurol. 2002;448:28–52. doi: 10.1002/cne.10212. [DOI] [PubMed] [Google Scholar]
- Müller-Decker K, Leder C, Neumann M, Neufang G, Bayerl C, Schweizer J, Marks F, Fürstenberger G. Expression of cyclooxygenase isozymes during morphogenesis and cycling of pelage hair follicles in mouse skin: precocious onset of the first catagen phase and alopecia upon cyclooxygenase-2 overexpression. J Invest Dermatol. 2003;121:661–668. doi: 10.1046/j.1523-1747.2003.12473.x. [DOI] [PubMed] [Google Scholar]
- Paus R, Heinzelmann T, Schultz KD, Furkert J, Fechner K, Czarnetzki BM. Hair growth induction by substance P. Lab Invest. 1994;71:134–140. [PubMed] [Google Scholar]
- Peters EM, Botchkarev VA, Botchkareva NV, Tobin DJ, Paus R. Hair-cycle-associated remodeling of the peptidergic innervation of murine skin, and hair growth modulation by neuropeptides. J Invest Dermatol. 2001;116:236–245. doi: 10.1046/j.1523-1747.2001.01232.x. [DOI] [PubMed] [Google Scholar]
- Paus R, Maurer M, Slominski A, Czarnetzki BM. Mast cell involvement in murine hair growth. Dev Biol. 1994;163:230–240. doi: 10.1006/dbio.1994.1139. [DOI] [PubMed] [Google Scholar]
- Maurer M, Fischer E, Handjiski B, von Stebut E, Algermissen B, Bavandi A, Paus R. Activated skin mast cells are involved in murine hair follicle regression (catagen). Lab Invest. 1997;77:319–332. [PubMed] [Google Scholar]
- Stander S, Luger T, Metze D. Treatment of prurigo nodularis with topical capsaicin. J Am Acad Dermatol. 2001;44:471–478. doi: 10.1067/mjd.2001.110059. [DOI] [PubMed] [Google Scholar]