Abstract
Type IV collagen, a major component of the basement membrane (BM), is composed of six genetically distinct α(IV) chains, α1(IV) to α6(IV). Their genes are paired on three different chromosomes in a head-to-head arrangement. The α5(IV) gene (COL4A5) and the α6(IV) gene (COL4A6) are on chromosome Xq22 and are regulated by a bidirectional promoter. Loss of the α5(IV)/α6(IV) chains in epithelial BM occur in the early stage of cancer invasion. However, the regulatory mechanism of the specific loss of the α5(IV)/α6(IV) chains during cancer cell invasion is still undetermined. In the present study, we examined the expression of the α5(IV)/α6(IV) chains and the methylation profiles of the bidirectional promoter region of COL4A5/COL4A6 in colon cancer cell lines and colorectal tumor tissues. The expression of the α5(IV)/α6(IV) chains was down-regulated in colorectal cancer, and the loss of expression of the α5(IV)/α6(IV) chains was associated with the hypermethylation of their promoter region. In conclusion, the hypermethylation of the bidirectional promoter region of COL4A5/COL4A6 is one of the events that is responsible for the loss of expression of the α5(IV)/α6(IV) chains and the remodeling of the epithelial BM during cancer cell invasion.
Type IV collagen is a major component of the basement membrane (BM) that separates epithelial cells from the underlying stroma. Type IV collagen is composed of six genetically different α(IV) chains, α1(IV) to α6(IV). Their genes are paired on three different chromosomes in a head-to-head arrangement. The α1(IV) gene (COL4A1) and the α2(IV) gene (COL4A2) are localized on chromosome 13q34,1 the α3(IV) gene (COL4A3) and the α4(IV) gene (COL4A4) are on chromosome 2q36,2 and the α5(IV) gene (COL4A5) and the α6(IV) gene (COL4A6) are on chromosome Xq22.3 Three molecular forms of type IV collagen, composed of [α1(IV)]2/α2(IV), α3(IV)/α4(IV)/α5(IV) and [α5(IV)]2/α6(IV), have been characterized. They form meshworks in which the molecules are cross-linked at their C- and N-terminal ends.4 The [α1(IV)]2/α2(IV) chains exist in all BMs of the whole body. However, the α3(IV)-α6(IV) chains are distributed in the BM in a tissue-specific manner. For instance, the α3(IV)/α4(IV)/α5(IV) chains are distributed in the BM of the glomerulus of the kidney and in the BM of the alveoli of the lung,5–7 whereas the [α5(IV)]2/α6(IV) chains localize in the BM of epidermis, smooth muscle cells,6 mammary glands,8 and epithelium of the alimentary tract.6,9
We have reported the remodeling of type IV collagen α chains in the BM of several invasive cancers, including basal cell carcinoma of the skin,10 breast cancer,8 adenocarcinoma of the lung,11 and adenoid cystic carcinoma of the salivary glands.12 In the normal colonic epithelium, the BM consists of both the [α1(IV)]2/α2(IV) and the [α5(IV)]2/α6(IV) chains, and the loss of the [α5(IV)]2/α6(IV) chains in the early invasive stage of colon cancer was observed by immunohistochemical examination.13,14 However, the regulatory mechanism of the specific loss of the α5(IV)/α6(IV) chains is still unknown. Type IV collagen, for a long time, has been considered only as a scaffold for the binding of other BM components (laminin, heparan sulfate proteoglycan, and nidogen). Recently, many studies have demonstrated the potentially active roles of type IV collagen in cell behavior. Their active role in angiogenesis,15–17 cell growth, adhesion,15 and tumor cell invasion18,19 has also been reported. Therefore, the loss of the tissue-specific α(IV) chains in the epithelial BM may be closely related to biologically significant events in the early invasive stage of cancer.
Aberrant methylation of the CpG islands of the promoter region was identified as an epigenetic mechanism for the silencing of multiple genes. In malignant tumors, the hypermethylation of multiple genes was reported. The methylation of tumor suppressor genes, mismatch repair genes, adhesion molecules, and extracellular matrix genes has been described.20 In addition, in colorectal cancer, many types of gene are silenced by the hypermethylation of their promoters.21 Burbelo and colleagues22 demonstrated that the expression of the α1(IV)/α2(IV) chains was inhibited by the hypermethylation of their promoters in teratocarcinoma cells. However, there are no reports, to our knowledge, that have investigated the regulatory relationship between the expression of the α3(IV) to α6(IV) chains and the hypermethylation of their promoters. In this study, we investigated the expression of type IV collagen α chains in several human colon cancer cell lines and human colorectal tumor tissues and analyzed the relationship between the loss of expression of the α5(IV)/α6(IV) chains and the presence of the hypermethylation of the promoter region of COL4A5 and COL4A6.
Materials and Methods
Tissue Samples
Tissue samples for this study were obtained by surgical resection at the Kumamoto University School of Medicine between 1999 and 2004. We selected cases in which frozen tissues had been preserved for genomic analysis. We selected 12 cases of colorectal adenocarcinoma and 2 cases of colon tubular adenoma. Informed consent was obtained from each patient. In 12 patients with colorectal cancer, 6 patients were male and 6 patients were female with the median age of 66 years (range, 55 to 80 years). The locations of the tumors were as follows: cecum (n = 2), ascending colon (n = 2), descending colon (n = 1), sigmoid colon (n = 3), and rectum (n = 4). The Dukes’ stages of the tumors were A (n = 2), B (n = 5), and C (n = 5). The cancer cells of all cases had invaded through the lamina muscularis mucosae. The histological classifications of the tumors were well-differentiated adenocarcinoma (n = 4), moderately differentiated adenocarcinoma (n = 6), poorly differentiated adenocarcinoma (n = 1), and mucinous adenocarcinoma (n = 1). Both cases of tubular adenoma, with low-grade dysplasia, were from male patients. The tissue blocks of each tumor and the adjacent normal colonic mucosa were frozen immediately and preserved for DNA analysis. The resected tissues were also fixed in 10% formaldehyde and embedded in paraffin for immunohistochemical study.
Immunohistochemistry
Primary antibodies against six different α(IV) chains, recognizing the sequences of each C-terminal noncollagenous (NC1) domain [H11 for α1(IV), H21 for α2(IV), H31 for α3(IV), H43 for α4(IV), H63 for α6(IV)] or helical domain [H53 for α5(IV)], were established with the rat lymph node method.23 Immunohistochemical staining was performed on 4-μm-thick serial sections from paraffin-embedded tissue blocks according to the previous protocol.13 Briefly, for the immunostaining of the α1(IV) to α6(IV) chains, deparaffinized sections were pretreated for antigen retrieval by autoclave heating (132°C, 303 kPa) in 10 mmol/L citrate buffer (pH 3.3) for 5 minutes. These sections were blocked for endogenous peroxidase activity with 3% H2O2 in methanol for 60 minutes and then washed in phosphate-buffered saline (PBS). Thereafter, sections were immersed in 5% normal rabbit serum in PBS for 30 minutes, covered with primary rat monoclonal-specific antibodies for the α(IV) chains, and incubated overnight at 4°C. Immunoreactions were performed using a Vectastain peroxidase ABC kit (Vector Laboratories, Burlingame, CA). The antigenic sites were demonstrated by reacting the sections with a mixture of 0.05% 3,3-diaminobenzidine tetrahydrochloride (Dojin Chemicals, Tokyo, Japan) in 0.05 mol/L Tris-HCl buffer, pH 7.6, containing 0.01% H2O2 for 7 minutes. The nuclei were stained with hematoxylin.
In Situ Hybridization
In situ hybridization was performed on 5-μm serial sections of paraffin-embedded tissue blocks. We used the following chain-specific cDNA probes: MS2, a 1.4-kb EcoRI fragment containing a downstream part of the collagenous domain of the α2(IV) collagen gene, and TM27, a 1.8-kb EcoRI fragment containing the proximal part of the collagenous domain of the α6(IV) gene. Each probe was labeled with 35S-thymidine 5′-[α-thio]triphosphate by nick-translation to the specific activity of 2 to 4 × 108 cpm/μg DNA. The hybridization procedures used in this study were almost as those described previously.8 Briefly, deparaffinized sections were treated with Pronase E and acetylated with acetic anhydride. The slides were washed, dehydrated in ethanol, and dried in air. The treated sections were processed for in situ hybridization at 45°C for 18 hours in a mixture containing the 35S-labbeled cDNA probe (1 μg/ml), yeast tRNA (500 μg/ml), salmon sperm DNA (80 μg/ml), 50% formamide, 10 mmol/L Tris-HCl, 0.15 mol/L NaCl, 1 mmol/L ethylenediaminetetraacetic acid, 1× Denhardt’s mixture, and 10% dextran sulfate. After hybridization, the washed and dried slides were dipped into Kodak (Rochester, NY) NTB-2 emulation and exposed for 7 days at 4°C. The sections were counterstained with hematoxylin.
Cell Culture
Human colon cancer cell lines (Caco-2, HCT 116, SW480, DLD-1, LoVo, COLO 201, HT-29, and WiDr) and a human foreskin fibroblast cell line (Hs68) were obtained from American Type Culture Collection (Manassas, VA). Cell cultures were grown in recommended medium with 10% fetal bovine serum and incubated in 5% CO2 at 37°C. For the immunohistochemical analysis of the expression of the α(IV) chains, cell lines were cultured in Matrigel (a solubilized BM matrix extracted from the Engelbreth-Holm-Swarm mouse tumor; BD Biosciences, Bedford, MA).24 They were incubated at 37°C until the cell suspension gelled, and cell culture media was added. The gels containing the cultured cells were incubated for 2 weeks and fixed with 10% formaldehyde and embedded in paraffin for the immunohistochemical examination.
5-Aza-dC Treatment
DLD-1, HT29, and WiDr, RT-negative cell lines, were treated with the demethylating agent 5-aza-deoxycytidine (5-Aza-dC) (Sigma-Aldrich, St. Louis, MO), as described previously.25 The cell cultures were plated at a density of 1 × 105 per 10-cm2 dish, and incubated in the medium without 5-Aza-dC for 24 hours. Thereafter, 5-Aza-dC was added at a concentration of 1 μmol/L and 5 μmol/L and incubated for 1 week.
Reverse Transcriptase-Polymerase Chain Reaction (RT-PCR) for Type IV Collagen α Chains
Total RNA was extracted from 2 × 106 cells of each cell line with TRIzol reagent (Life Technologies, Inc., Grand Island, NY) following the manufacturer’s instructions. Five μg each of total RNA was reverse-transcribed to cDNA with Super Script First Strand System (Invitrogen, Carlsbad, CA). Primers were designed to detect the expression of mRNA of α(IV) chains. The chain-specific sequences were selected for the primer design from the NC1 domain coding regions: α1(IV) sense 5′-CAGCCAGACCATTCAGATCC-3′, anti-sense 5′-GGCGTAGGCTTCTTGAACAT-3′, α2(IV) sense 5′-GCATGAACAAACTCTGGAGTG-3′, anti-sense 5′-CTTGATCTCGTCCTCGGCCA-3′, α5(IV) sense 5′-AGAGCA-TCCAGCCATTCATT-3′, anti-sense 5′-TTCAGCGTTTCTGACTGAGG-3′, α6(IV) sense 5′-CAACGAGGTGTGCC-ACTATG-3′, and anti-sense 5′-TTCAGCGTTTCAGACA-CAGG-3′. GAPDH was used as the RT-PCR control to verify the quality and quantity of the template cDNAs: sense 5′-TGAACGGGAAGCTCACTGG-3′ and anti-sense 5′-TCCACCACCCTGTTGCTGTA-3′. The PCRs were performed with Accuprime Taq polymerase (Invitrogen). The PCR products were visualized on 2% agarose gels stained with ethidium bromide.
Bisulfite Sequencing
Genomic DNAs from cultured cells were extracted from 2 × 106 cells with the use of TRIzol reagent. The genomic DNAs from frozen tissues were exacted from each 50 mg of tissue with an ISOTISSUE kit (Nippon Gene, Toyama, Japan). Ten μg of EcoRI-treated DNA was modified by sodium bisulfite treatment as described previously.26 The primers for the modified sequence of the common promoter region of COL4A5 and COL4A6 were designed as follows: sense 5′-TGAGGTTTGGGTGAAGAGAAAG-3′ and anti-sense 5′-TCCCAATATTTTCACATCTTC-3′. The PCR reaction was performed for the amplification of the promoter region. The PCR product was cloned with a TOPO TA cloning kit (Invitrogen) according to the instruction manual. Ten colonies of transformed competent cells were picked, and incubated in 5 ml of LB medium with 50 μg/ml of ampicillin for 16 hours. The plasmid from each clone was extracted with QIAMP DNA mini-kit (Qiagen, Valencia, CA). Cycle sequencing was performed with a Bigdye terminator kit (Applied Biosystems, Foster City, CA), after which the DNA was sequenced on an ABI310 automated sequencer (Applied Biosystems).
Luciferase Assays
The 5′-flanking fragment of the COL4A6 gene was used for generating reporter construct to evaluate the promoter activity. The fragments from −192 and +65 (0 indicates the transcription start site of COL4A6 gene) and from −574 and +65 were amplified from genomic DNA of Caco-2 cells and was cloned into pGL3-Basic Vector (Promega, Madison, WI). The PCR primers for amplification of the promoter region were designed to yield restriction ends for ligation: kpnI and sacI (−192Luc sense 5′-GAGGTACCGACTGAGCACCATGAGTCC-3′, −574Luc sense 5′-GAGGGTACCGAGA-GGGACAGTGAGGCTTG-3′, and anti-sense 5′-CTCTCGAGTGTGAGCAGCTGGAAGGTAA-3′). The reporter construct was methylated in vitro with SssI methylase (New England Biolabs, Beverly, MA), which methylated all cytosine residues within the CpG sites. Each microgram of plasmid was treated with 3 U of SssI in the presence of 160 μmol/L S-adenosylmethionine at 37°C for 3 hours. Caco-2 cells were cultured in 4-cm2 dishes and transfected with 1.6 μg of reporter constructs with 2.0 μl of Lipofectamine 2000 (Invitrogen). As an internal control, 0.003 μg of phRL-TK vector (Promega), which expresses Renilla luciferase, was transfected in each reaction. After a 48-hour incubation, the luciferase activities were measured with Dual-Luciferase Reporter Assay system (Promega) according to the instruction manual.
Statistical Analysis
Data were expressed as the mean ± SE. Comparison of values with and without correspondence between the two groups was performed using the Wilcoxon test and the Mann-Whitney U-test. Category data were compared using the χ2 test. Values of P < 0.05 were considered statistically significant.
Results
Expression of the α(IV) Chains in the Colorectal Tumor and Normal Tissues
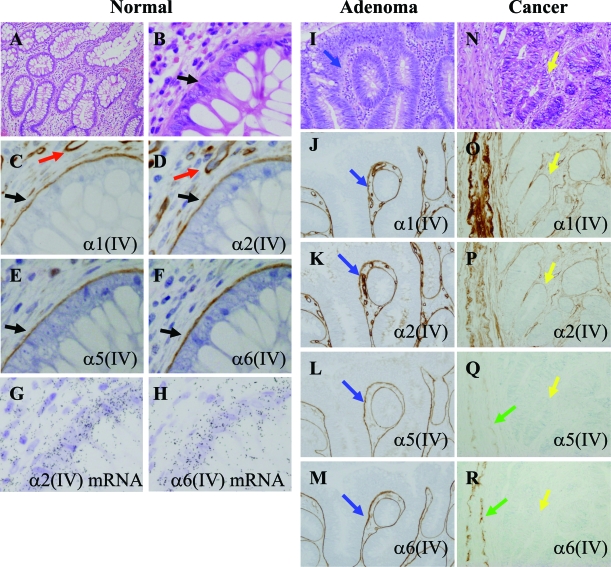
We performed the immunohistochemical study for the sections of 12 colorectal cancers, 2 cases of tubular adenoma and their adjacent normal colonic mucosa. In all of the normal mucosa (Figure 1, A–H), the α1(IV)/α2(IV) chains and the α5(IV)/α6(IV) chains were stained in a continuous linear pattern in the BM of the surface epithelium and crypts (Figure 1, C–F; black arrows). As shown in the previous report,13 the α3(IV)/α4(IV) chains were expressed only in the BM of apical surface of epithelium (data not shown). The α1(IV)/α2(IV) chains were also expressed in the BM of the capillary endothelium (Figure 1, C and D; red arrows) and smooth muscle cells of the lamina muscularis mucosae, the muscularis propria, and the large blood vessels. On the other hand, the α5(IV)/α6(IV) chains were weakly expressed only in the smooth muscle cells (Figure 1, Q and R; green arrows). In tubular adenoma (Figure 1, I–M), the α1(IV)/α2(IV) chains and the α5(IV)/α6(IV) chains were stained in continuously linear pattern in the BM of the adenoma glands (Figure 1, J–M; blue arrows). In the invasive cancer (Figure 1, N–R), the α1(IV)/α2(IV) chains were stained in the boundary between the gland of cancer and the extracellular matrix. The staining for the α1(IV)/α2(IV) chains were variably discontinuous (Figure 1, O and P; yellow arrows). However, the staining for the α5(IV)/α6(IV) chains around the cancer cell nests was completely negative (Figure 1, Q and R; yellow arrows). In the invasive areas of the submucosa and the muscularis propria, no expression of the α5(IV)/α6(IV) chains was seen around the cancer cell nests in all of the 12 cases.
Figure 1.
Immunohistochemical localizations of the α(IV) chains and their mRNA expressions in the normal colonic epithelium and colon tumors. A–H: Serial sections of the normal colonic epithelium. α1(IV), α2(IV), α5(IV), and α6(IV) chains are evident in the BM zones of colonic epithelium (black arrows). Red arrows indicate the α1(IV) and α2(IV) chains of the capillary BM. G: α2(IV) mRNA expression is evident both in the colonic epithelium and stromal cells including capillaries. H: α6(IV) mRNA expression is evident in the colonic epithelium. I–M: Serial sections of tubular adenoma with low-grade dysplasia. All of the glands indicate adenoma. α1(IV), α2(IV), α5(IV), and α6(IV) chains are evident in the BM zones of adenoma (blue arrows). N–R: Serial sections of invasive cancer. All of the glands indicate adenocarcinoma. α1(IV) and α2(IV) chains are discontinuously stained in the BM of the cancer cell nests (yellow arrows). Green arrows indicate the staining of the BM of the smooth muscle cells in an arteriole as the internal positive controls. A, B, I, N: H&E stain. C, J, O: Immunostaining for α1(IV) chain. D, K, P: Immunostaining for α2(IV) chain. E, L, Q: Immunostaining for α5(IV) chain. F, M, R: Immunostaining for α6(IV) chain. G: α2(IV) mRNA by in situ hybridization. H: α6 (IV) mRNA by in situ hybridization. Original magnifications: ×100 (A, I–R); ×400 (B–H).
To identify the origin of α(IV) chains of epithelial BM, in situ hybridization was performed on the resected normal colonic mucosa. The signal for α2(IV) chain mRNA was evident both in the colonic epithelial cells and the mesenchymal cells (Figure 1G). On the other hand, the signal for α6(IV) chain mRNA was localized predominantly in the colonic epithelial cells (Figure 1H). These results support that α6(IV) chains of the glandular BM are produced by colonic epithelium.
Expression of mRNA for α(IV) Chains in Colon Cancer Cell Lines
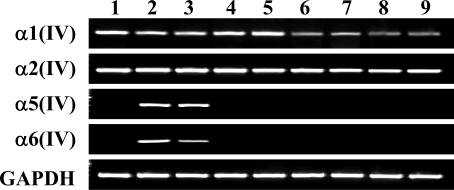
The result of the RT-PCR for the type IV collagen α chains is shown in Figure 2. Caco-2 is known as a colon cancer cell line that shows enterocyte-like differentiation.27 This cell line is useful as a model of enterocytic differentiation.28 Caco-2 cells showed obvious expression of the α1(IV), α2(IV), α5(IV), and α6(IV) chains. The specificity of the RT-PCR was confirmed with the direct sequence of the PCR product (data not shown.). HCT 116 also expressed these four α(IV) chains. However, SW480, DLD-1, LoVo, COLO 201, HT-29, and WiDr expressed the α1(IV) and α2(IV) chains but did not express the α5(IV) and α6(IV) chains. Thus, the expression of the α5(IV) and α6(IV) chains is specifically lost in six of eight colon cancer cell lines at the mRNA level. On the other hand, the human fibroblast cell line Hs68 expressed the α1(IV) and α2(IV) chains but did not express the α5(IV) and α6(IV) chains.
Figure 2.
RT-PCR for the NC1 domains of the α1(IV), α2(IV), α5(IV), and α6(IV) chains in the cell lines. Lane 1: Hs68, a human foreskin fibroblast; lane 2: Caco-2; lane 3: HCT116; lane 4: SW 480; lane 5: DLD-1; lane 6: LoVo; lane 7: COLO 201; lane 8: HT-29; lane 9: WiDr, human colon cancer cell lines. GAPDH was used as the RT-PCR control to verify the quality and quantity of the template cDNAs. The size of the RT-PCR products coincided with the expected length of the designed primers [α1(IV), 258 bp; α2(IV), 233 bp; α5(IV), 337 bp; α6(IV), 429 bp; GAPDH, 306 bp].
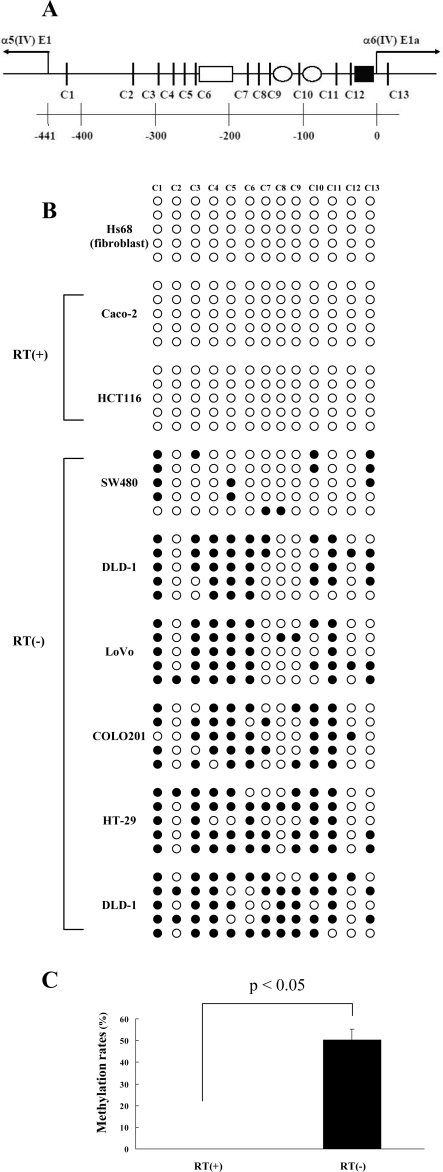
Methylation Profiles of the Common Promoter Region of COL4A5 and COL4A6 in Colon Cancer Cell Lines
The genes of the α5(IV) (COL4A5) and α6(IV) (COL4A6) chains have been found on the X chromosome at segment q22 and have been arranged in a head-to-head manner.3 The intergenic region between the transcript start sites of COL4A5 and COL4A6 is 441 bp, as illustrated in Figure 3A. We found 12 CpG sites in this region and designed the promoter to include these CpG sites and one site downstream from the transcription start site of COL4A6. To examine the methylation profiles of one fibroblast cell line and eight colon cancer cell lines, we sequenced 10 clones of each cell line after bisulfite modification. The cytosines of the CpG sites that were not modified into thymine with bisulfite treatment were considered to be methylated. The methylation profiles of the promoter region of COL4A5 and COL4A6 in one fibroblast cell line (Hs68) and eight colon cancer cell lines are shown in Figure 3B. Caco-2 and HCT116, which expressed mRNAs for the α5(IV) and α6(IV) chains, showed no methylation of the CpG sites in the promoter region. On the other hand, SW480, DLD-1, LoVo, COLO 201, HT-29, and WiDr, which lacked the expression of the mRNA for the α5(IV) and α6(IV) chains, showed promoter methylation. The methylation rate of CpG sites in the promoter of RT-PCR-positive colon cancer cell lines [RT(+): Caco-2 and HCT 116] was 0%,and that of the RT(−) cell lines (SW 480, DLD-1, LoVo, COLO 201, HT-29, and WiDr) was 50.0%. The methylation rate was significantly higher in RT(−) colon cancer cell lines than RT(+) cell lines (Figure 3C, P < 0.05). Fibroblast cell line Hs68 did not show methylation of the promoter region (Figure 3B). This indicates that the loss of expression of the α5(IV) and α6(IV) chains in this cell line is attributable to another mechanism except for the promoter hypermethylation.
Figure 3.
A: Genomic map of the sequence between exon 1a of COL4A6 and exon 1 of COL4A5. Vertical lines and C numbers show the CpG sites. The bottom numbers indicate the position (bp) from the α6(IV) transcription start site. Well-known potential interaction sites with transcriptional factors are indicated as: open box, CTC box; open circles, CCAAT boxes; and filled square, AP1 site. B: Methylation profiles in the promoter region of COL4A5 and COL4A6 of one fibroblast cell line (Hs68) and eight colon cancer cell lines. The white circles show the unmethylated, and the black circles show the methylated. The upper C numbers coincide with the CpG sites shown in A. The representative five clones for each cell line are shown in this figure. C: Methylation rates of CpG sites in the promoter region of COL4A5/COL4A6. The incidence of the methylation was significantly higher in the RT-PCR-negative colon cancer cell lines [RT(−): SW480, Lovo, COLO201, DLD-1, HT-29, and WiDr] than the RT(+) cell lines (Caco-2, HCT116) (P < 0.05, Mann-Whitney’s U-test).
Restoration of the α5(IV) and α6(IV) Chain Expression with the 5-Aza-dC Treatment in Colon Cancer Cell Lines
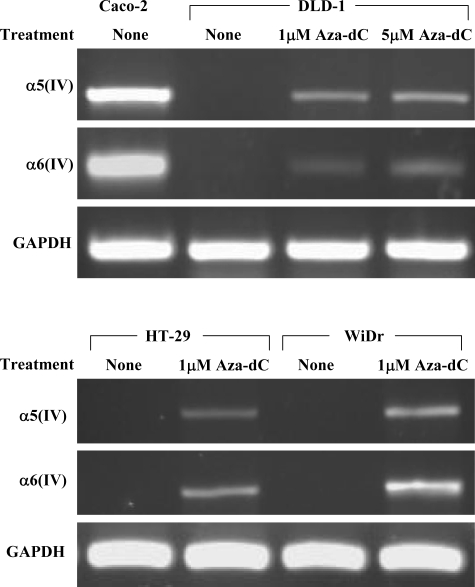
The effect of the demethylating agent 5-Aza-dC was examined with the RT-PCR for the α5(IV) and α6(IV) chains, as shown in Figure 4. DLD-1 revealed the recovery of the expression of the α5(IV) and α6(IV) chains with 1 μmol/L 5-Aza-dC. HT-29 and WiDr also showed the recovery of the expression of the α5(IV) and α6(IV) chains by 5-Aza-dC. The demethylation of the CpG sites in the promoter was confirmed with bisulfite sequencing. DLD-1 showed the decrease of methylation of the CpG sites in a dose-dependent manner, and the other two cell lines also showed the same effects (data not shown). The possible role of the promoter hypermethylation for silencing the expression of the α5(IV) and α6(IV) chains at the transcriptional level was demonstrated in vitro.
Figure 4.
RT-PCR for the NC1 domain of the α5(IV) and α6(IV) chains with and without being treated with the demethylating agent, 5-Aza-dC in the RT-PCR-negative colon cancer cell lines (DLD-1, HT-29, and WiDr). Caco-2 was used as a positive control. The demethylation effects were evident in the RT-PCR-negative cell lines, shown by the re-expression of the α5(IV)/α6(IV) mRNAs.
Expression of the α5(IV) and α6(IV) Chains in Colon Cancer Cell Lines Cultured in Matrigel
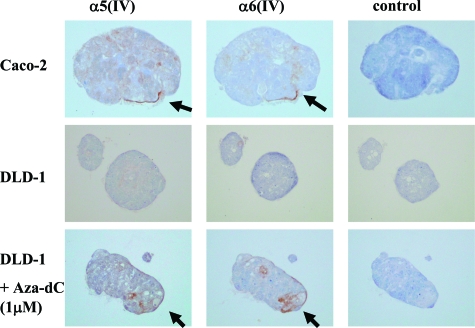
The three-dimensional culture of cells in Matrigel to evaluate the expression of the α(IV) chains is shown in Figure 5. Caco-2, which expressed the mRNAs for the α5(IV)/α6(IV) chains, formed a duct-like structure and showed the linear immunostaining pattern of the α5(IV) and α6(IV) chains around the cell clusters. DLD-1, which lacked the expression of the mRNAs, formed cell clusters without obvious polarity and showed no immunostaining of the α5(IV)/α6(IV) chains around the cell clusters. However, after treatment with 5-Aza-dC, DLD-1 expressed the α5(IV)/α6(IV) chains around the cell clusters as determined immunohistochemically.
Figure 5.
Immunohistochemical studies of the α5(IV) and α6(IV) chains in the colon cancer cell lines (Caco-2, DLD-1) cultured in Matrigel (200×). The right lane is the result without antibodies for the α(IV) chains as a negative control. Caco-2 showed partially linear immunostaining of the α5(IV) and α6(IV) chains in the boundary between the cell cluster and Matrigel (arrows). DLD-1 showed no immunostaining for the α5(IV) and α6(IV) chains. However, because of the treatment with 5-Aza-dC, the expression of the α5(IV) and α6(IV) chains were observed around the cell cluster (arrows).
Decrease of the Promoter Activity of the 5′-Flanking Region of the COL4A6 Gene with the Methylation of the Reporter Construct
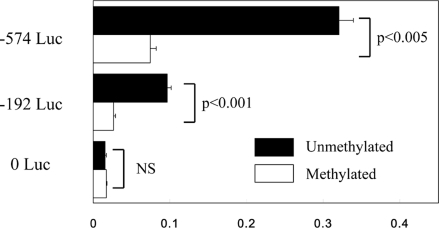
Caco-2 cells, which expressed the α5(IV)/α6(IV) chains, were transfected with plasmid constructs carrying the promoter region adjacent to the E1a of COL4A6. Three types of plasmids, −574 Luc, −192 Luc, and control vector without promoter (0 Luc), were tested. The entire length of intergenic region of COL4A5 and COL4A6 were included in −574 Luc. The shorter plasmid −192 Luc included the minimal promoter region for bidirectional transcription that was reported previously.29 The results of the luciferase assay are shown in Figure 6. The activities in −574 Luc and −192 Luc transfectants were 21.3-times and 6.3-times as potent as that of the control vector without promoter. With the SssI methylase treatment of the constructs, the activities of the promoters were significantly decreased both in −574 Luc (P < 0.005) and −192 Luc (P < 0.001). The activity in the control vector (0 Luc) transfectant was not affected by the SssI treatment. The results revealed that the promoter region of COL4A5 and COL4A6 could be inactivated by its hypermethylation.
Figure 6.
Promoter activities of the 5′-flanking region of the COL4A6 gene in Caco-2 cells. The columns represent the relative activities to the Renilla luciferase activities of phRL-TK, which was transfected together with each plasmid. The values are the average of the activities in three samples for each experiment. The error bar represents the SE. The black and white columns represent the activities in the transfectants of the constructs without and with the treatment of SssI CpG methylase.
Methylation Profiles in the Common Promoter Region of COL4A5 and COL4A6 in the Tissue Samples
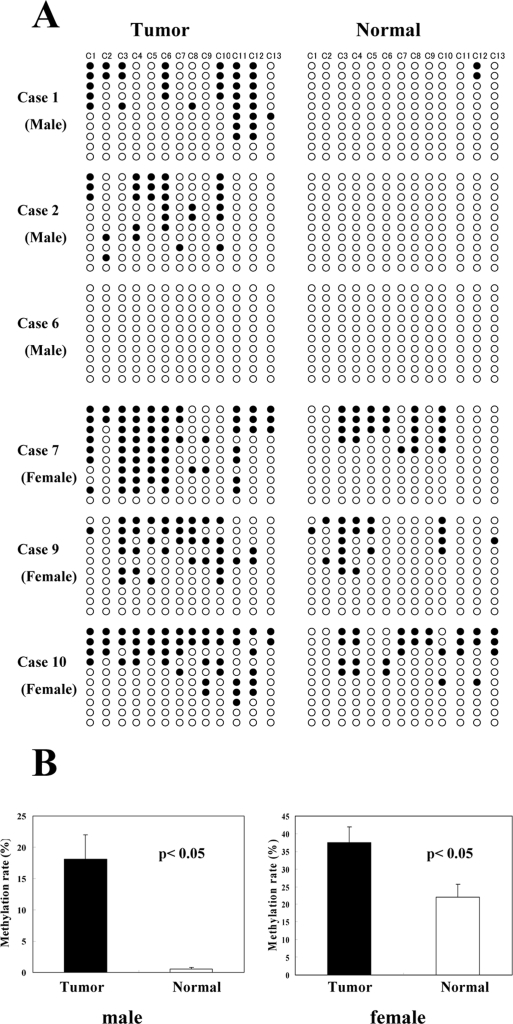
Twelve cases of colorectal adenocarcinoma and two cases of adenoma were analyzed and compared along with the matched normal epithelium. Representative cases of colorectal adenocarcinomas are shown in Figure 7A. In the female cases, almost half of the clones showed a certain level of methylation, even in the normal epithelium. This seems to be related to X chromosome inactivation. The methylation in the normal epithelium of male cases was very rare. The average methylation rates were significantly higher in cancer than in the normal epithelium in both sexes (Figure 7B). However, there were cases of males in which no methylation was detected (Figure 7A, case 6), and cases of females in which no obvious difference between the tumor and the normal epithelium were seen (Figure 7A, case 10). In such cases, nevertheless, the immunohistochemical expressions of the α5(IV)/α6(IV) chains were absent, similar to the methylated cases. These results suggested that the expression of the α5(IV)/α6(IV) chains were lost by other unknown mechanisms except for the hypermethylation of the promoter. Additionally, the methylation rates of individual cases showed no significant relationship with the clinicopathological factors (age, tumor location, Duke’s classification, histological classification). The two cases of male colonic adenoma showed no methylation (data not shown).
Figure 7.
Methylation profiles in the promoter region of COL4A5 and COL4A6 in the colon cancer tissues and the adjacent normal tissues. A: Six representative cases (three males and three females) are shown. B: The methylation rates of the colon cancer tissues and adjacent normal tissues. The data were separated into two groups according to sex because there was physiological methylation in the female X chromosome. The columns represent the average methylation rates of each of six cancer tissues and the normal mucosa. The error bar represents the SE. In both sexes, there are significant differences between the cancer and the normal mucosa (P < 0.05).
Discussion
The cellular origin of type IV collagen of epithelial BM, from epithelial cells or mesenchymal cells, is still un-determined. Several immunohistochemical studies in normal human tissues have shown tissue-specific ex-pressions of the α3(IV), α4(IV), α5(IV), and α6(IV) chains.6,8,10–13,30 They showed that the type of α(IV) chains expressed in the BMs depended on the type of the epithelium. In the present study by using in situ hybridization, the localization of the mRNA of α6(IV) chain was evident in the colonic epithelial cells (Figure 1H). Caco-2, a colon cancer cell line showing enterocyte-like differentiation, expressed both the α1(IV)/α2(IV) and the α5(IV)/α6(IV) chains at the transcriptional level. On the other hand, the fibroblast cell line Hs68 expressed only α1(IV)/α2(IV)chains. Additionally, Caco-2 produced the α5(IV)/α6(IV) chains in the boundary between the epithelial cell clusters and Matrigel in the condition without mesenchymal cells in the three-dimensional culture system, as shown in Figure 5. Consequently, we propose that the epithelial BM-specific α(IV) chains [α3(IV) to α6(IV)] are produced predominantly by their epithelial cells.
In many types of cancer, the epithelial BM-specific α(IV) chains are lost in the early stage of cancer cell invasion.8,10–13,31,32 In the present study, we showed the transcriptional down-regulation of the α5(IV)/α6(IV) chains in several colon cancer cell lines. During the first step of cancer cell invasion and metastasis, cancer cells have to break through the epithelial BM. In this regard, the degradation potency of the BM by the matrix metalloproteinases produced by cancer cells has been the main focus. Many studies described the up-regulation of matrix metalloproteinases and other proteinases and the down-regulation of the tissue inhibitors of metalloproteinase in cancer cells. Except for the degradation mechanism, our results suggest that the normal production and organization of the epithelial BM might be interrupted during carcinogenesis.
The regulatory mechanism of the expression of the α5(IV)/α6(IV) chains is not well understood. Several studies investigated the mechanisms for the tissue-specific expression of these molecules. Segal and colleagues29 demonstrated that the binding of bidirectional activator promoted the coupled expression of the α5(IV) and α6(IV) chains, and the single expression of the α5(IV) chain in renal glomerular cells was regulated by another transcription factor. Sund and colleagues33 revealed that the growth factors activated the promoter region differentially among the cell types. However, no reports have hitherto explained the loss of the expression of the α5(IV)/α6(IV) chains during cancer cell invasion.
Many types of genes are reported to be silenced due to the abnormal methylation of their promoter regions in cancer. Laminin-5, one of the components of the BM, was also reported to be down-regulated by the hypermethylation of its promoter in several types of cancer.34,35 In this study, we examined the methylation in the characteristic bidirectional promoter region between COL4A5 and COL4A6. The promoter region did not satisfy the criteria of the CpG island.36,37 However, several genes without CpG islands in the promoter region are also reported to be silenced by the methylation of the CpG sites.34,38 In our study, the loss of expression of the α5(IV)/α6(IV) chains was closely correlated to the hypermethylation of the promoter in colon cancer cell lines, and the luciferase assay showed the methylation of the promoter region was able to decrease the promoter activity.
Additionally, we showed that the promoter region was methylated in the normal colonic epithelium of females (Figure 7). This phenomenon seems to be attributable to X chromosome inactivation. The hypermethylation of the CpG islands on the inactivated female X chromosome is widely known.39 However, not all X chromosome genes are inactivated; there are several genes that escape X chromosome inactivation.40 Ke and Collins41 described that the major reason of escaping X inactivation was the lack of sensitivity to the promoter hypermethylation. On the other hand, Guo and colleagues42 reported a case of severe Alport syndrome in a woman in which the X chromosome without mutation was predominantly inactivated. Consequently, it could be said that the gene of the α5(IV)/α6(IV) chains is inactivated in one X chromosome of the female, and the inactivation is related to the hypermethylation of the promoter region.
Accordingly, we must analyze the silencing of the promoter region divided by sex. In DLD-1, LoVo, and COLO 201, originating from male tissue, and the colorectal cancer tissues of male cases, the expression might be silenced by the methylation of the promoter region on only one X chromosome. In HT-29 and WiDr, female cell lines, and the tissues in female cases, the promoter region on one X chromosome might be premethylated with the X inactivation, and the expression of the α5(IV)/α6(IV) chains was silenced because the other X chromosome was methylated related to the malignant processes.
The questions of what rate of methylation in the promoter was sufficient or which CpG sites of the promoter were responsible for silencing the transcription were not determined in this study. In SW 480, a colon cancer cell line from male tissues, the methylation rate of the CpG sites in the promoter was only 14%, but the transcription of α5(IV)/α6(IV) chains were lost. Additionally, there were cases in which the promoters were not methylated, but the expressions of the α5(IV)/α6(IV) chains were lost based on immunohistochemistry. In such cases, other molecular mechanisms for the loss of expression of the α5(IV)/α6(IV) chains; mutation, loss of the transcription factor, changes of the signaling related to growth factors or cell adhesion, were suggested. Additionally, the loss of the other components of the BM, such as the α1(IV)/α2(IV) chains and laminin-5, might cause the loss of the normal deposition of the α5(IV)/α6(IV) chains in the cancer tissues. It would appear that the mechanism of the loss of the tissue-specific α(IV) chains during cancer cell invasion is complex and many factors are involved in these events.
The possible roles of the α(IV) chains for preventing cancer cell invasion, growth, and angiogenesis have been reported. Petitclerc and colleagues15 described the inhibitory effect of recombinant NC1 domains of the α2(IV), α3(IV), and α6(IV) chains for angiogenesis and tumor growth. The anti-tumor effects of the recombinant α2(IV) chain (Canstatin)17,43 and α3(IV) chain (Tumstatin)16,44 have been well investigated. However, the biological activity of the α6(IV) chain has not been fully researched. Because the specific loss of the α5(IV)/α6(IV) chains reported to occur in many types of cancers,8,10,12,13,31,32 the further research as to the biological role of the α6(IV) chain for cancer prevention is expected.
In conclusion, the expression of the α5(IV)/α6(IV) chains are down-regulated in colorectal cancer. Furthermore, the hypermethylation of their promoter region is one of the events that is responsible for the loss of expression of the α5(IV)/α6(IV) chains and remodeling of BM during the cancer cell invasion.
Footnotes
Address reprint requests to Ken-ichi Iyama, M.D., Ph.D., Department of Surgical Pathology, Kumamoto University School of Medicine, 1-1-1 Honjo, Kumamoto, 860-8556 Japan. E-mail: iyama@kaiju.medic.kumamoto-u.ac.jp.
References
- Solomon E, Hall V, Kurkinen M. The human α2(IV) collagen gene, COL4A2, is syntenic with the α1(IV) gene, COL4A1, on chromosome 13. Ann Hum Genet. 1987;51:125–127. doi: 10.1111/j.1469-1809.1987.tb01053.x. [DOI] [PubMed] [Google Scholar]
- Mariyama M, Zheng K, Yang-Feng TL, Reeders ST. Colocalization of the genes for the α3(IV) and α4(IV) chains of type IV collagen to chromosome 2 bands q35–q37. Genomics. 1992;13:809–813. doi: 10.1016/0888-7543(92)90157-n. [DOI] [PubMed] [Google Scholar]
- Sugimoto M, Oohashi T, Ninomiya Y. The genes COL4A5 and COL4A6, coding for basement membrane collagen chains α5(IV) and α6(IV), are located head-to-head in close proximity on human chromosome Xq22 and COL4A6 is transcribed from two alternative promoters. Proc Natl Acad Sci USA. 1994;91:11679–11683. doi: 10.1073/pnas.91.24.11679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Timpl R. Structure and biological activity of basement membrane proteins. Eur J Biochem. 1989;180:487–502. doi: 10.1111/j.1432-1033.1989.tb14673.x. [DOI] [PubMed] [Google Scholar]
- Mariyama M, Leinonen A, Mochizuki T, Tryggvason K, Reeders ST. Complete primary structure of the human α3(IV) collagen chain. Coexpression of the α3(IV) and α4(IV) collagen chains in human tissues. J Biol Chem. 1994;269:23013–23017. [PubMed] [Google Scholar]
- Ninomiya Y, Kagawa M, Iyama K, Naito I, Kishiro Y, Seyer JM, Sugimoto M, Oohashi T, Sado Y. Differential expression of two basement membrane collagen genes, COL4A6 and COL4A5, demonstrated by immunofluorescence staining using peptide-specific monoclonal antibodies. J Cell Biol. 1995;130:1219–1229. doi: 10.1083/jcb.130.5.1219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sado Y, Kagawa M, Naito I, Ueki Y, Seki T, Momota R, Oohashi T, Ninomiya Y. Organization and expression of basement membrane collagen IV genes and their roles in human disorders. J Biochem (Tokyo) 1998;123:767–776. doi: 10.1093/oxfordjournals.jbchem.a022003. [DOI] [PubMed] [Google Scholar]
- Nakano S, Iyama K, Ogawa M, Yoshioka H, Sado Y, Oohashi T, Ninomiya Y. Differential tissular expression and localization of type IV collagen α1(IV), α2(IV), α5(IV), and α6(IV) chains and their mRNA in normal breast and in benign and malignant breast tumors. Lab Invest. 1999;79:281–292. [PubMed] [Google Scholar]
- Simoneau A, Herring-Gillam FE, Vachon PH, Perreault N, Basora N, Bouatrouss Y, Pageot LP, Zhou J, Beaulieu JF. Identification, distribution, and tissular origin of the α5(IV) and α6(IV) collagen chains in the developing human intestine. Dev Dyn. 1998;212:437–447. doi: 10.1002/(SICI)1097-0177(199807)212:3<437::AID-AJA11>3.0.CO;2-Y. [DOI] [PubMed] [Google Scholar]
- Tanaka K, Iyama K, Kitaoka M, Ninomiya Y, Oohashi T, Sado Y, Ono T. Differential expression of α1(IV), α2(IV), α5(IV) and α6(IV) collagen chains in the basement membrane of basal cell carcinoma. Histochem J. 1997;29:563–570. doi: 10.1023/a:1026428010104. [DOI] [PubMed] [Google Scholar]
- Nakano KY, Iyama KI, Mori T, Yoshioka M, Hiraoka T, Sado Y, Ninomiya Y. Loss of alveolar basement membrane type IV collagen α3, α4, and α5 chains in bronchioloalveolar carcinoma of the lung. J Pathol. 2001;194:420–427. doi: 10.1002/path.928. [DOI] [PubMed] [Google Scholar]
- Misumi S, Iyama K, Honda Y, Kitano T, Sado Y, Ninomiya Y, Shinohara M. Differential expression of basement membrane type-IV collagen α1, α2, α5 and α6 chains among the histological subtypes of adenoid cystic carcinoma. Virchows Arch. 2004;445:54–62. doi: 10.1007/s00428-004-1015-3. [DOI] [PubMed] [Google Scholar]
- Hiki Y, Iyama K, Tsuruta J, Egami H, Kamio T, Suko S, Naito I, Sado Y, Ninomiya Y, Ogawa M. Differential distribution of basement membrane type IV collagen α1(IV), α2(IV), α5(IV) and α6(IV) chains in colorectal epithelial tumors. Pathol Int. 2002;52:224–233. doi: 10.1046/j.1440-1827.2002.01341.x. [DOI] [PubMed] [Google Scholar]
- Oka Y, Naito I, Manabe K, Sado Y, Matsushima H, Ninomiya Y, Mizuno M, Tsuji T. Distribution of collagen type IV α1-6 chains in human normal colorectum and colorectal cancer demonstrated by immunofluorescence staining using chain-specific epitope-defined monoclonal antibodies. J Gastroenterol Hepatol. 2002;17:980–986. doi: 10.1046/j.1440-1746.2002.02789.x. [DOI] [PubMed] [Google Scholar]
- Petitclerc E, Boutaud A, Prestayko A, Xu J, Sado Y, Ninomiya Y, Sarras MP, Hudson BG, Brooks PC. New functions for non-collagenous domains of human collagen type IV. Novel integrin ligands inhibiting angiogenesis and tumor growth in vivo. J Biol Chem. 2000;275:8051–8061. doi: 10.1074/jbc.275.11.8051. [DOI] [PubMed] [Google Scholar]
- Maeshima Y, Colorado PC, Torre A, Holthaus KA, Grunkemeyer JA, Ericksen MB, Hopfer H, Xiao Y, Stillman IE, Kalluri R. Distinct antitumor properties of a type IV collagen domain derived from basement membrane. J Biol Chem. 2000;275:21340–21348. doi: 10.1074/jbc.M001956200. [DOI] [PubMed] [Google Scholar]
- Kamphaus GD, Colorado PC, Panka DJ, Hopfer H, Ramchandran R, Torre A, Maeshima Y, Mier JW, Sukhatme VP, Kalluri R. Canstatin, a novel matrix-derived inhibitor of angiogenesis and tumor growth. J Biol Chem. 2000;275:1209–1215. doi: 10.1074/jbc.275.2.1209. [DOI] [PubMed] [Google Scholar]
- Martinella-Catusse C, Polette M, Noel A, Gilles C, Dehan P, Munaut C, Colige A, Volders L, Monboisse JC, Foidart JM, Birembaut P. Down-regulation of MT1-MMP expression by the α3 chain of type IV collagen inhibits bronchial tumor cell line invasion. Lab Invest. 2001;81:167–175. doi: 10.1038/labinvest.3780224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pasco S, Ramont L, Venteo L, Pluot M, Maquart FX, Monboisse JC. In vivo overexpression of tumstatin domains by tumor cells inhibits their invasive properties in a mouse melanoma model. Exp Cell Res. 2004;301:251–265. doi: 10.1016/j.yexcr.2004.07.036. [DOI] [PubMed] [Google Scholar]
- Herman JG, Baylin SB. Gene silencing in cancer in association with promoter hypermethylation. N Engl J Med. 2003;349:2042–2054. doi: 10.1056/NEJMra023075. [DOI] [PubMed] [Google Scholar]
- van Rijnsoever M, Grieu F, Elsaleh H, Joseph D, Iacopetta B. Characterisation of colorectal cancers showing hypermethylation at multiple CpG islands. Gut. 2002;51:797–802. doi: 10.1136/gut.51.6.797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burbelo PD, Horikoshi S, Yamada Y. DNA methylation and collagen IV gene expression in F9 teratocarcinoma cells. J Biol Chem. 1990;265:4839–4843. [PubMed] [Google Scholar]
- Sado Y, Kagawa M, Kishiro Y, Sugihara K, Naito I, Seyer JM, Sugimoto M, Oohashi T, Ninomiya Y. Establishment by the rat lymph node method of epitope-defined monoclonal antibodies recognizing the six different α chains of human type IV collagen. Histochem Cell Biol. 1995;104:267–275. doi: 10.1007/BF01464322. [DOI] [PubMed] [Google Scholar]
- Petersen OW, Rønnov-Jessen L, Howlett AR, Bissell MJ. Interaction with basement membrane serves to rapidly distinguish growth and differentiation pattern of normal and malignant human breast epithelial cells. Proc Natl Acad Sci USA. 1992;89:9064–9068. doi: 10.1073/pnas.89.19.9064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang X, Phillips DL, Ferguson AT, Nelson WG, Herman JG, Davidson NE. Synergistic activation of functional estrogen receptor (ER)-α by DNA methyltransferase and histone deacetylase inhibition in human ER-α-negative breast cancer cells. Cancer Res. 2001;61:7025–7029. [PubMed] [Google Scholar]
- Hibi K, Taguchi M, Nakayama H, Takase T, Kasai Y, Ito K, Akiyama S, Nakao A. Molecular detection of p16 promoter methylation in the serum of patients with esophageal squamous cell carcinoma. Clin Cancer Res. 2001;7:3135–3138. [PubMed] [Google Scholar]
- Grasset E, Pinto M, Dussaulx E, Zweibaum A, Desjeux JF. Epithelial properties of human colonic carcinoma cell line Caco-2: electrical parameters. Am J Physiol. 1984;247:C260–C267. doi: 10.1152/ajpcell.1984.247.3.C260. [DOI] [PubMed] [Google Scholar]
- Zhang X, Cromwell JW, Kunjummen BD, Yee D, Garcia-Aguilar J. The alpha2 and alpha3 integrins are required for morphologic differentiation of an intestinal epithelial cell line. Surgery. 2003;133:429–437. doi: 10.1067/msy.2003.107. [DOI] [PubMed] [Google Scholar]
- Segal Y, Zhuang L, Rondeau E, Sraer JD, Zhou J. Regulation of the paired type IV collagen genes COL4A5 and COL4A6. Role of the proximal promoter region. J Biol Chem. 2001;276:11791–11797. doi: 10.1074/jbc.M007477200. [DOI] [PubMed] [Google Scholar]
- Kiyofuji MA, Iyama K, Kitaoka M, Sado Y, Ninomiya Y, Ueda S. Quantitative analysis of type IV collagen α chains in the basement membrane of human urogenital epithelium. Histochem J. 2002;34:479–486. doi: 10.1023/a:1024753705556. [DOI] [PubMed] [Google Scholar]
- Kadono G, Ishihara T, Yamaguchi T, Kato K, Kondo F, Naito I, Sado Y, Saisho H. Immunohistochemical localization of type IV collagen α chains in the basement membrane of the pancreatic duct in human normal pancreas and pancreatic diseases. Pancreas. 2004;29:61–66. doi: 10.1097/00006676-200407000-00057. [DOI] [PubMed] [Google Scholar]
- Dehan P, Waltregny D, Beschin A, Noel A, Castronovo V, Tryggvason K, De Leval J, Foidart JM. Loss of type IV collagen α5 and α6 chains in human invasive prostate carcinomas. Am J Pathol. 1997;151:1097–1104. [PMC free article] [PubMed] [Google Scholar]
- Sund M, Maeshima Y, Kalluri R. Bifunctional promoter of type IV collagen COL4A5 and COL4A6 genes regulates the expression of α5 and α6 chains in a distinct cell-specific fashion. Biochem J. 2005;387:755–761. doi: 10.1042/BJ20041870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sathyanarayana UG, Toyooka S, Padar A, Takahashi T, Brambilla E, Minna JD, Gazdar AF. Epigenetic inactivation of laminin-5-encoding genes in lung cancers. Clin Cancer Res. 2003;9:2665–2672. [PubMed] [Google Scholar]
- Sathyanarayana UG, Padar A, Huang CX, Suzuki M, Shigematsu H, Bekele BN, Gazdar AF. Aberrant promoter methylation and silencing of laminin-5-encoding genes in breast carcinoma. Clin Cancer Res. 2003;9:6389–6394. [PubMed] [Google Scholar]
- Takai D, Jones PA. Comprehensive analysis of CpG islands in human chromosomes 21 and 22. Proc Natl Acad Sci USA. 2002;99:3740–3745. doi: 10.1073/pnas.052410099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bird A. DNA methylation patterns and epigenetic memory. Genes Dev. 2002;16:6–21. doi: 10.1101/gad.947102. [DOI] [PubMed] [Google Scholar]
- Cho JH, Kimura H, Minami T, Ohgane J, Hattori N, Tanaka S, Shiota K. DNA methylation regulates placental lactogen I gene expression. Endocrinology. 2001;142:3389–3396. doi: 10.1210/endo.142.8.8347. [DOI] [PubMed] [Google Scholar]
- Monk M. Methylation and the X chromosome. Bioessays. 1986;4:204–208. doi: 10.1002/bies.950040505. [DOI] [PubMed] [Google Scholar]
- Carrel L, Cottle AA, Goglin KC, Willard HF. A first-generation X-inactivation profile of the human X chromosome. Proc Natl Acad Sci USA. 1999;96:14440–14444. doi: 10.1073/pnas.96.25.14440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ke X, Collins A. CpG islands in human X-inactivation. Ann Hum Genet. 2003;67:242–249. doi: 10.1046/j.1469-1809.2003.00038.x. [DOI] [PubMed] [Google Scholar]
- Guo C, Van Damme B, Vanrenterghem Y, Devriendt K, Cassiman JJ, Marynen P. Severe Alport phenotype in a woman with two missense mutations in the same COL4A5 gene and preponderant inactivation of the X chromosome carrying the normal allele. J Clin Invest. 1995;95:1832–1837. doi: 10.1172/JCI117862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He GA, Luo JX, Zhang TY, Hu ZS, Wang FY. The C-terminal domain of canstatin suppresses in vivo tumor growth associated with proliferation of endothelial cells. Biochem Biophys Res Commun. 2004;318:354–360. doi: 10.1016/j.bbrc.2004.04.038. [DOI] [PubMed] [Google Scholar]
- Maeshima Y, Sudhakar A, Lively JC, Ueki K, Kharbanda S, Kahn CR, Sonenberg N, Hynes RO, Kalluri R. Tumstatin, an endothelial cell-specific inhibitor of protein synthesis. Science. 2002;295:140–143. doi: 10.1126/science.1065298. [DOI] [PubMed] [Google Scholar]