Abstract
The immune system recognizes diverse melanoma antigens. However, tumors can evade the immune response, therefore growing and progressing. It has been reported that galectin-3 and galectin-1 can induce apoptosis of activated lymphocytes. However, there is strong evidence indicating that the regulation of galectins function in the human tumor microenvironment is a complex process that is influenced by diverse biological circumstances. Here, we have investigated 33 biopsies (eight primary and 25 metastases) from 24 melanoma patients (15–72 years old) and describe the correlation between the expression of galectin-3 or galectin-1 and the level of apoptosis of tumor-associated lymphocytes using immunohistochemistry and an in situ nick translation assay. The range of galectin-3-positive tumor cells varied between 0% and 93% and that of galectin-1-positive tumor cells varied between 5% and 97%. In addition, 23 ± 27% of tumor-associated lymphocytes were apoptotic. Although our results show a correlation between galectin-3 expression and apoptosis of tumor-associated lymphocytes, we could not find such correlation with galectin-1. Considering the complex process of cancer immunoediting, various interacting factors must be considered.
Malignant melanoma is the cancer with the fastest growing incidence,1 and the survival of patients with visceral metastases is often less than 1 year. The use of chemotherapy in this disease is quite deceiving, because mostly short-lived partial responses are obtained. Because melanoma is an immunogenic tumor, the use of immunotherapy has attracted considerable interest in recent years. It is well known that melanoma cells elicit a specific effector activity by generating T-cell clones that recognize melanoma-associated antigens in a major histocompatibility complex context.2–4 However, human melanoma cells have also developed survival strategies that allow their growth and invasion in a hostile microenvironment to be monitored by the immune system. Thus, the tumor relies on several mechanisms of immune escape such as down-regulation of major histocompatibility complex context molecules, insufficient antigen expression,5 and production of local immunosuppressive factors, such as Fas ligand6,7 or interleukin-10.8 We and others have recently demonstrated in experimental melanoma that galectin-1 (gal-1) may also contribute to tumor immune escape.9
Galectins are carbohydrate-binding proteins that share a constant recognition domain for β-galactosides and are involved in cell adhesion, migration, differentiation, angiogenesis, proliferation, mRNA splicing, and apo-ptosis.10–12 We and others have shown a pro-apoptotic activity for secreted soluble gal-1 and galectin-3 (gal-3) on T lymphocytes.9,13–15 However, sensitivity to galectin-mediated lymphocyte apoptosis in human melanoma may be regulated by several factors, such as cytoplasmic anti-apoptotic gal-3,16,17 expression of proteases that cleave gal-3 in the tumor microenvironment,18,19 and the absence of adequate glycan ligands in the surface of target cells.20
Because any chance of success for immunotherapy appears to require that cytotoxic viable CD8+ and/or CD4+ T lymphocytes enter the tumor and lyse tumor cells, we have investigated the expression of gal-3 and gal-1 in human melanoma biopsies. We provide here evidence that gal-3 expression correlates with T-cell apoptosis. The presence of other local or systemic pro-apoptotic factors may influence apoptosis at the tumor site.
Materials and Methods
Patients and Melanoma Biopsies
Samples were obtained from melanoma patients who were enrolled in several clinical trials authorized by the Institutional Review Board of the Instituto Alexander Fleming and who gave informed consent. A total of 33 biopsies from 24 patients (15 to 72 years old) were studied. In seven patients the primary tumors were unknown. Eight biopsies corresponded to primary tumors, 13 to lymph node metastasis, 10 to cutaneous metastasis, and 2 to lung metastasis. According to the histopathological reports, all tumors were epithelioid cell melanoma. The patients are listed below in Table 1. In three patients both primary tumors and metastases could be analyzed. Two patients (NB and GZ) had Stage IIB melanoma, and therefore only the primary tumors were analyzed.
Table 1.
Patient Population and Biopsies: Ages, Gender, and Clinical Stages of Patients
| Patient | Age | Gender | Biopsy | Stage | M%ISNT | M%gal-1 | M%gal-3 |
|---|---|---|---|---|---|---|---|
| AS | 33 | F | LNM | IV | 54 ± 25 | 41 ± 22 | 40 ± 32 |
| 34 | F | CM | IV | 32 ± 23 | 57 ± 23 | 67 ± 23 | |
| CS | 27 | F | LNM | IV | 0 | 6 ± 6 | 19 ± 21 |
| FM | 38 | F | PT | III | 77 ± 20 | 30 ± 26 | 49 ± 23 |
| GQ | 36 | F | LNM | IV | 32 ± 27 | 62 ± 35 | 37 ± 38 |
| GZ | 32 | M | PT | IIB | 13 ± 15 | 26 ± 35 | 11 ± 5 |
| HD | 70 | M | CM | IV | 20 ± 16 | 5 ± 3 | 12 ± 18 |
| HDS | 20 | M | LNM | IV | 16 ± 9 | 34 ± 17 | 4 ± 5 |
| HJ | 67 | F | LNM | III | 26 ± 15 | 55 ± 36 | 47 ± 31 |
| HS | 68 | M | LNM | IV | 2 ± 3 | 18 ± 16 | 15 ± 10 |
| IL | 29 | M | PT | III | 5 ± 4 | 13 ± 12 | 43 ± 20 |
| 32 | M | CM | III | 48 ± 36 | 29 ± 29 | 84 ± 19 | |
| MAF | 15 | F | LNM | III | 33 ± 25 | 6 ± 12 | 59 ± 40 |
| MB | 21 | M | PT | IV | 0 | 56 ± 27 | 42 ± 10 |
| 23 | M | LM | IV | 5 ± 9 | 55 ± 15 | 14 ± 21 | |
| MF | 48 | F | PT | III | 26 ± 20 | 15 ± 26 | 76 ± 23 |
| MFE | 33 | M | LNM | III | 8 ± 12 | 42 ± 22 | 48 ± 21 |
| MFR | 48 | F | LNM | III | 1 ± 2 | 11 ± 13 | 0 |
| MG | 33 | F | LNM | III | 16 ± 26 | 64 ± 38 | 78 ± 28 |
| DM | 72 | F | CM | III | 5 ± 9 | 70 ± 18 | 17 ± 15 |
| 72 | F | CM | III | 34 ± 27 | 37 ± 26 | 93 ± 9 | |
| NB | 45 | F | PT | IIB | 0 | 9 ± 7 | 11 ± 8 |
| ED | 60 | F | LNM | III | 59 ± 32 | 41 ± 36 | 40 ± 36 |
| OL | 54 | M | PT | IV | 33 ± 18 | 16 ± 20 | 18 ± 14 |
| 55 | M | CM | IV | 32 ± 42 | 68 ± 21 | 12 ± 11 | |
| 56 | M | CM | IV | 4 ± 7 | 33 ± 22 | 70 ± 21 | |
| PB | 24 | M | PT | IV | 8 ± 14 | 77 ± 9 | 78 ± 10 |
| MCRC | 45 | F | LNM | III | 22 ± 27 | 23 ± 23 | 24 ± 25 |
| 45 | F | CM | III | 41 ± 27 | 56 ± 25 | 85 ± 9 | |
| 45 | F | CM | III | 16 ± 14 | 27 ± 11 | 67 ± 21 | |
| ASS | 54 | F | CM | IV | 14 ± 27 | 17 ± 18 | 58 ± 39 |
| 54 | F | LM | IV | 6 ± 7 | 97 ± 4 | 17 ± 15 | |
| SVN | 54 | F | LNM | III | 58 ± 20 | 60 ± 23 | 28 ± 30 |
The percentages and standard deviations of positive ISNT lymphocytes and gal-1- and gal-3-positive tumor cells were determined as described in Materials and Methods. M, mean percentage of 10 fields of each biopsy; LNM, lymph node metastasis; CM, cutaneous metastasis; PT, primary tumor; LM, lung metastasis.
Specimens were fixed in 10% neutral buffered formalin and embedded in paraffin. Consecutive 4-μm tissue sections were cut, placed onto silane-coated microscope slides, and processed for histopathological examination, as well as for in situ nick translation (ISNT) and immunohistochemistry (IHC).
Immunohistochemistry
Tissue sections were deparaffinized, rehydrated, and placed in a pressure cooker with citrate buffer, pH 6.0, for 1 minute after reaching boiling temperature to retrieve antigenic sites masked by formalin fixation. The tissue sections were then placed in 3% hydrogen peroxide for 5 minutes to inactivate endogenous peroxidase, blocked for 20 minutes with normal horse serum (Vectastain Elite ABC Kit; Vector Laboratories, Burlingame, CA), and subsequently incubated overnight at 4°C with 1:200 gal-1 monoclonal mouse anti-human antibody or 1:200 gal-3 monoclonal mouse anti-human antibody (clones 25C1 and 9C4, respectively; Novocastra Laboratories Ltd., Newcastle, UK). As a negative control stain, IgG1-κ from murine myeloma (clone MOPC 21; Sigma-Aldrich Inc., St. Louis, MO) was used. The slides were then treated with biotinylated secondary antibody for 30 minutes at room temperature, followed by avidin- biotin complex reagent for 1 hour and peroxidase substrate (VECTOR NovaRed Substrate Kit; Vector Laboratories, Burlingame, CA) for 8 minutes. Counterstaining was performed with 10% hematoxylin. To detect gal-1-sensitive activated lymphocytes, a monoclonal mouse anti-human CD45RO antibody (clone UCHL1; DakoCytomation, Glostrup, Denmark) at 1:50 was used, and subsequent steps were as above.
For Ki-67 staining, once tissue sections were blocked for endogenous peroxidase activity, primary monoclonal mouse anti-human Ki-67 antibody (clone MIB-1; DakoCytomation) was incubated for 10 minutes at room temperature. The sections were then treated with biotinylated secondary antibody (Universal LSAB2 Kit; DakoCytomation) for 10 minutes, followed by 10-minute incubation with streptavidin-horseradish peroxidase and 3,3′-diaminobenzidine solution for another 10 minutes at room temperature. Tissue sections were counterstained with hematoxylin.
A polyclonal antibody for human cleaved poly-(ADP-ribose) polymerase (Cell Signaling Technology Inc., Beverly, MA) was used following the recommended manufacturer’s protocol.
In Situ Nick Translation
To identify nuclei with DNA strand breaks as a marker of apoptosis, ISNT was performed according to the protocol described by Koji et al21 with modifications. Tissue sections were deparaffinized with xylene and rehydrated in serial ethanol solutions. After extensive washing with phosphate-buffered saline, the sections were treated with 1 μg/ml proteinase K (Fermentas Life Sciences, Vilnius, Lithuania) in phosphate-buffered saline at 37°C for 15 minutes, washed with phosphate-buffered saline, and immersed in 50 mmol/L Tris-HCl, pH 7.5. Afterward, DNA synthesis was performed on the slides for 3 hours at 37°C in 100 μl of medium containing 50 mmol/L Tris-HCl, pH 7.5, 10 mmol/L MgCl2, 0.1 mmol/L dithiothreitol, 50 μg/ml bovine serum albumin, 100 U/ml DNA polymerase I (New England Biolabs Inc., Beverly, MA) and 20 μmol/L each of dATP, dGTP, and dCTP (Invitrogen, Carlsbad, CA) and biotin-11-dUTP (Fermentas Life Sciences). dTTP (Invitrogen) was used as a negative control instead of biotin-11-dUTP. After repeated washes, sections were placed in 3% hydrogen peroxide in methanol for 5 minutes to inactivate endogenous peroxidase, washed with phosphate-buffered saline, and then incubated for 30 minutes with avidin-biotin complex reagent (Vectastain Elite ABC Kit; Vector Laboratories) at room temperature. The horseradish peroxidase-labeled sites were visualized by incubating for 2 minutes with peroxidase substrate (VECTOR NovaRed Substrate Kit, Vector Laboratories). Counterstaining was performed in 10% hematoxylin. All materials and reagents were sterile or autoclaved to avoid nuclease contamination and thus undesirable breaks on DNA strands.
Western Blots
Extracts were prepared from tissues of melanoma biopsies that were preserved at −80°C. Tissues were thawed, pulverized with liquid nitrogen, treated for 30 minutes at 4°C with lysis buffer (50 mmol/L Tris-HCl, pH 7.5, 1% Nonidet P-40, 150 mmol/L NaCl, 5 mmol/L ethylenediamine tetraacetic acid, and 1 mmol/L phenylmethylsulfonyl fluoride) and centrifuged at 4°C during 30 minutes at 10,000 × g. The supernatant was aliquoted and frozen at −20°C. The protein concentration was measured according to Bradford.22
The extracts were run in a 15% polyacrylamide gel electrophoresis gradient after seeding 30 μg of protein per lane. The gel was then transferred to a nitrocellulose membrane (0.45-μm pore; Sigma). The membranes were blocked with 3% bovine skim milk (Molico, Argentina) and then incubated overnight at 4°C with 1:100 diluted anti-gal-3 or anti-gal-1 monoclonal antibodies. As a loading control, a monoclonal antibody against the constitutively expressed glyceraldehyde-3-phosphate dehydrogenase was also incubated (clone 6C5; Ambion Inc., Austin, TX). As standards, 2.5 μg of recombinant gal-1 and gal-3 (PeproTech, Rocky Hill, NJ) was used. After several washings, filters were incubated with alkaline phosphatase-conjugated goat Ig anti-mouse IgG (Jackson ImmunoResearch, West Grove, PA) and visualized with nitro blue tetrazolium-5 and bromo-4-chloro-3-indolyl phosphate (Promega Corp., Madison, WI).
IHC Quantitative Staining Analysis
Homologous areas of serial sections were searched and analyzed for gal-3, gal-1, and ISNT staining using histological landmarks, in which both lymphocytes and melanoma cells were present. The data were obtained by evaluating the percentage of positive cells in at least ten ×400 random fields using an optical light microscope (Olympus Microscope System BX40; Olympus, Tokyo, Japan). Apoptotic cell morphology was confirmed at ×1000 magnification.
Statistical Analysis on IHC
The data were analyzed by calculating the nonparametric Spearman correlation and Wilcoxon rank sum test, using Statistix software version 8.0 (Analytical Software, Tallahassee, FL). P < 0.05 was considered statistically significant. To associate ISNT(+) lymphocytes with gal-3(+) or gal-1(+) neoplastic cells, the Spearman correlation test was used in a total of 323 homologous fields.
The 323 fields were classified as gal3(+)/gal1(−), gal3(−)/gal1(+), gal3(+)/gal1(+), and gal3(−)/gal1(−) and further analyzed with the Wilcoxon rank sum test for significant differences on the level of ISNT(+) tumor-associated lymphocytes. A field was considered positive for gal-1 or gal-3 when ≥5% of the cells were stained.
Apoptosis Assay
A mechanically dissociated cutaneous melanoma metastasis preserved in liquid nitrogen was thawed in serum-free lymphocyte medium (AIMV; Gibco, Grand Island, NY) medium and incubated for 2 hours at 37°C and 5% CO2. An aliquot was incubated with monoclonal mouse anti-human CD3-PerCP antibody (clone SK7; BD Biosciences, San Jose, CA) to trace T lymphocytes. In a forward scatter/side scatter plot, the CD3(+) population was gated for further cell sorting. The selected gate was used to sort the remaining 1 ×106 nonincubated cells with a FACSCalibur (BD Biosciences). A total of 2 × 105 sorted cells was then incubated with 20 μmol/L recombinant gal-3 (kindly provided by Dr. M.T. Elola, Fundación 105 Instituto Leloir, Buenos Aires, Argentina) in 200 μl of RPMI 1640 medium in the presence or absence of 100 mmol/L lactose for 5 hours at 37°C and 5% CO2. Apoptotic cells were measured by annexin-V binding and propidium iodide permeability (BD Pharmingen, Basel, Switzerland) and analyzed using a FACSCalibur and CellQuest software (BD Biosciences).
Because the presence of apoptotic tumor-associated lymphocytes (TALs) in the biopsy caused by the freeze-thaw process and/or pro-apoptotic stimuli from tumor microenvironment was certain, fresh specific gp-100 cytotoxic T lymphocyte to line (G154: KTWGQYWQV, gift from Dr. C. Yee, Fred Hutchinson Cancer Research Center, Seattle, WA) was used to set zero. The fold increase of annexin-V positivity rate was determined as follows: percentage of annexin-V-positive cells treated with gal-3 alone/percentage of annexin-V-positive nontreated cells.
Results
Expression of gal-3 and gal-1 in Human Melanoma Biopsies
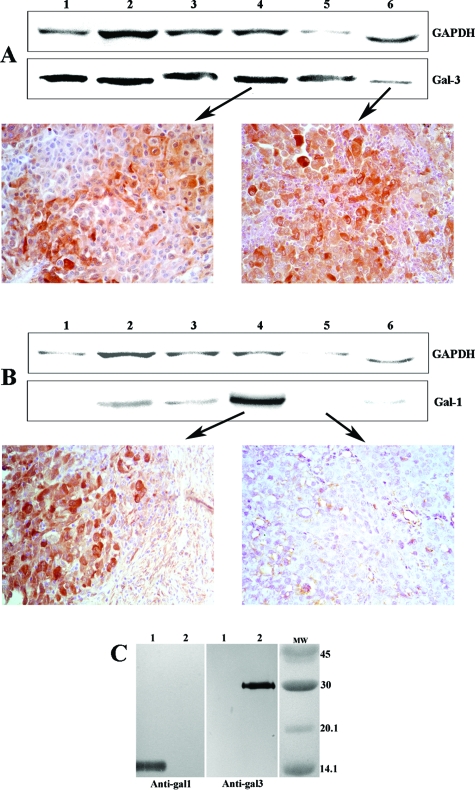
The characteristics of the patient population analyzed in this study are indicated in Table 1. There were 15 women and nine men, with ages ranging from 15 to 72 years (44 ± 16 years). To determine whether gal-3 and gal-1 were expressed in human melanoma, IHC determination was performed separately in consecutive sections of 33 biopsies. Positive staining of both galectins was detected in the cytoplasm and occasionally (7/33) in the nucleus. Membrane expression of gal-3 and gal-1 was observed in 6/33 and 14/33 samples, respectively. The expression of galectins was heterogeneous even in the same biopsy; the range of gal-3-positive cells varied between 0% and 93%, and the range of gal-1-positive cells varied between 5% and 97% (Table 1). For this reason, and considering that proteins used for Western blot also derived from normal cells negative for these galectins, the protein amount as determined by Western blots did not always match the IHC positivity (Figure 1, A and B). We also confirmed the specificity and absence of cross-reactivity between anti-human gal-3 and anti-human gal-1 monoclonal antibodies, which was done by Western blots using recombinant gal-3 and gal-1 as standards (Figure 1C).
Figure 1.
Correlation between expression of gal-1 and gal-3 as determined by IHC and Western blots. IHC of different biopsies and their corresponding Western blots were performed as described in Materials and Methods. Lanes represent patients as follows: lane 1, HS; lane 2, HJ; lane 3, MFE; lane 4, AS; lane 5, CS; and lane 6, MG. A: Anti-glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and anti-gal-3 (Gal-3). B: Anti-glyceraldehyde-3-phosphate dehydrogenase and anti-gal-1. C: Antibodies anti-gal-1 and anti-gal-3 react specifically with recombinant gal-1 and gal-3. A total of 2.5 μg of recombinant gal-1 (lane 1) and gal-3 (lane 2) was electrophoresed, and Western blots with monoclonal antibodies anti-gal-1 (left) or anti-gal-3 (right) were performed as described in Materials and Methods. Original magnification of IHC: ×200.
As shown in Table 2, no significant differences in the expression of gal-3 and gal-1 were found when all of the data were analyzed (323 fields). Only in primary tumors there was a significant difference between the expression of gal-1 and gal-3 (Wilcoxon rank sum test P = 0.008). Skin and lung metastasis expressed greater percentages of gal-3 and gal-1 than lymph node metastasis (Wilcoxon rank sum test, P < 0.005).
Table 2.
Mean Percentage and Standard Deviation of Total ISNT(+) Lymphocytes and gal-1 or gal-3 Expression in Neoplastic Cells
| Biopsies | Fields examined | % ISNT (+) TALs | % gal-1(+) NC | % gal-3(+) NC |
|---|---|---|---|---|
| Primary melanoma | 78 | 20 ± 28 | 30 ± 30 | 42 ± 30 |
| Lymph node metastasis | 130 | 23 ± 27 | 35 ± 32 | 33 ± 34 |
| Cutaneous and lung metastasis | 115 | 20 ± 26 | 49 ± 31 | 47 ± 37 |
| Total sample | 323 | 21 ± 27 | 39 ± 32 | 40 ± 34 |
ISNT, in situ nick translation; TALs, tumor-associated lymphocytes; NC, neoplastic cells.
Validation of in Situ Nick Translation as an Apoptotic Marker
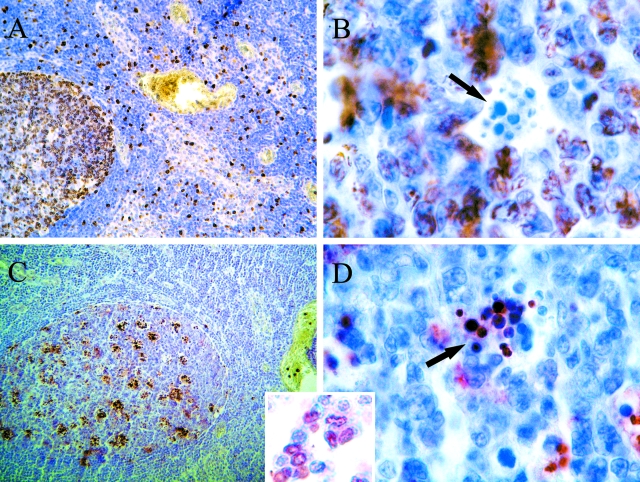
ISNT has become a common histological technique to analyze the occurrence of DNA single strand breaks in apoptosis. Despite the evidence of ISNT as a selective apoptotic marker,23,24 and as single strand breaks have also been implicated in the regulation of cell proliferation,21 we performed IHC for the cell proliferating antigen Ki-67 on a lymph node with follicular hyperplasia and six melanoma biopsies, to discard false-positive staining.25 Discrimination of false-positive staining is more straightforward in lymph nodes with follicular hyperplasia, because apoptosis and proliferation are common features and their histoarchitecture allows differentiation of them. As an example, a lymph node with follicular hyperplasia stained with ISNT and Ki-67 is shown in Figure 2. In a germinal center many lymphocytes stained with Ki-67 (Figure 2A) but not with ISNT (Figure 2C). Apoptotic bodies were Ki67(−)/ISNT(+) as shown in Figure 2 (B and D). In addition to this, apoptotic morphology supported ISNT technique in those areas in which ISNT-positive lymphocytes were found (Figure 2, inset). Distribution of ISNT(+) cells and Ki-67(+) cells was not coincident. Furthermore, IHC for cleaved-poly-(ADP-ribose) polymerase, another apoptosis marker, was performed on 12 of 33 cases, including a lymph node with follicular hyperplasia, and the staining distribution pattern was quite similar to that of ISNT (data not shown).
Figure 2.
Validation of the ISNT technique. Reactive follicular hyperplasia of a non-metastatic lymph node. A and B: Ki-67 immunohistochemistry at 100× and 1000×, respectively. C and D: biotin-11-dUTP labeled in situ nick translation at 100× and 1000×, respectively. The arrows indicate Ki-67-negative (B) and ISNT-positive (D) apoptotic bodies. Inset: Apoptotic morphology is seen in ISNT-positive lymphocytes, while ISNT-negative lymphocytes show conserved nuclei.
Correlation between gal-3 and gal-1 Expression and Apoptosis of TALs
Because a large proportion of our samples were metastatic lymph nodes, we prefer to use the term TALs rather than tumor-infiltrating lymphocytes. ISNT was performed on consecutive sections to those used for galectins determination. The mean ± SD of ISNT(+) TALs in the whole sample was 21 ± 27% (Table 2). No significant differences of these values were observed when the biopsies were grouped as primary tumors, lymph node metastases, and skin or lung metastases.
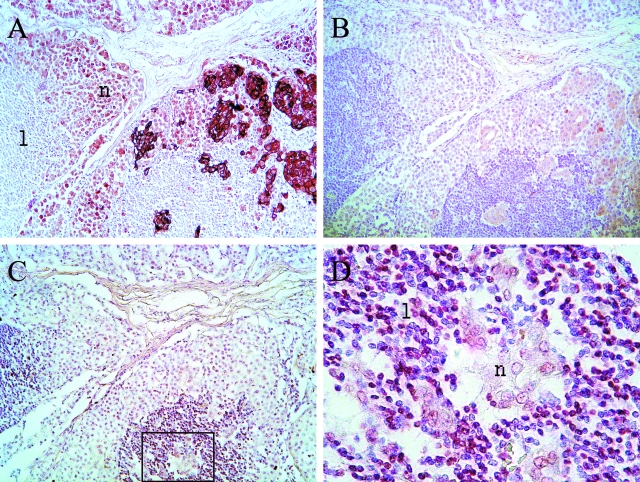
To evaluate whether gal-3 and/or gal-1 are correlated with apoptotic TALs in melanoma biopsies, IHC for gal-3, gal-1, and ISNT were analyzed. Correlation analysis was more straightforward in primary tumors and in cutaneous or visceral metastasis, because it may be assumed that lymphocytes are present there as a response to the tumor. The situation is more complex in lymphoid organs, where lymphocytes are the normal residents and tumor cells are the invading elements. Therefore, in lymph node metastases, only lymphocytes that were completely surrounded by tumor cells were analyzed. As an example of the analysis performed, a lymph node infiltrated by melanoma cells is shown in Figure 3, presenting strong, heterogeneous gal-3 staining (Figure 3A) and weak, heterogeneous gal-1 staining (Figure 3B). Enclosed lymphocytes are ISNT(+) at a high percentage (Figure 3, C and D).
Figure 3.
Gal-3, gal-1, and ISNT in a metastatic lymph node corresponding to patient MAF. A: gal-3 IHC at 100×. B: gal-1 IHC at 100×. C and D: In Situ Nick Translation at 100× and 1000×, respectively. n, neoplastic cells; l, lymphocytes.
The percentages and standard deviations of ISNT(+) TALs and neoplastic cells expressing gal-3 and gal-1 were obtained from 323 homologous fields. Standard deviations showed heterogeneity between the 33 biopsies and still within the same sample (Table 1). Due to this, and considering the absence of evidence for long distance apoptotic effect of galectins, the statistical analysis was performed by evaluating discrete fields where lymphocytes encounter neoplastic cells.
To correlate gal-3 expression with apoptosis, the 323 pairs of data were analyzed using the nonparametric Spearman test, and a positive correlation was found with a high significance (P = 0.001). To correlate gal-1 expression and apoptosis, data were treated equally, and no correlation was found (P = 0.9).
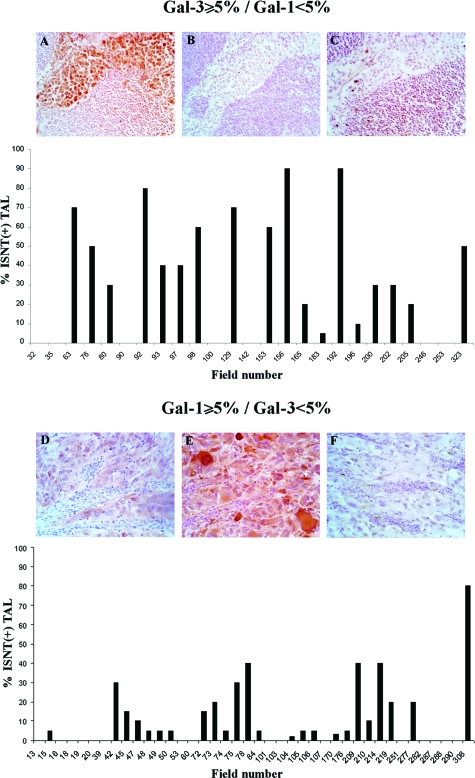
To discriminate more precisely the effect of gal-3 and gal-1 on lymphocyte apoptosis, the fields were classified by their level of galectins expression as follows: gal3(+)/gal1(−); gal3(−)/gal1(+) (Figure 4) and gal3(+)/gal1(+);gal3(−)/gal1(−) (Supplemental Figure 1, A and B, see http://ajp.amjpathol.org), and additional statistical analysis was performed.
Figure 4.
Correlation between gal-3 or gal-1 expression and lymphocytes apoptosis. Lymphocytes apoptosis was determined in areas with expression of gal-3 but not gal-1 (above) and in areas with expression of gal-1 but not gal-3 (below), as described in Materials and Methods. IHC for gal-3 (A and D), IHC for gal-1 (B and E), and ISNT (C and F). Original magnification: ×200.
A significant difference on the percentage of ISNT(+) TALs was found between gal-3-positive fields and gal-3-negative fields (Wilcoxon test P = 0.01). When the Spearman test was applied to gal-3(+) fields a positive correlation with apoptotic lymphocytes was found (P = 0.03). On the contrary, no differences were observed in apoptosis of lymphocytes when gal-1 was present or absent (Wilcoxon test), and no correlation was observed (Spearman test, P = 0.46) between gal-1 expression and apoptotic lymphocytes on these selected gal-1(+) areas.
Furthermore, we have analyzed the differential apoptotic effect in “pure areas” of gal-3 [gal3(+)/gal1(−)] or gal-1 [gal3(−)/gal1(+)] expression (Figure 4, A and B), and significant differences were found in the level of apoptotic lymphocytes (P = 0.005, Wilcoxon rank sum test).
Because lymphocytes may be sensitive to gal-1 apoptotic stimuli through the interaction with CD45RO, we determined the presence of CD45RO(+) lymphocytes within ISNT(−)/gal-1(+) areas and found them to be positive for this receptor (data not shown). Illustrating the complexity of the interrelationship between the interaction of pro-apoptotic factors and lymphocyte apoptosis, we observed several fields in which apoptotic lymphocytes were detected despite the absence of gal-3- and gal-1-expressing tumor cells (data not shown).
Gal-3 Induces Apoptosis of TALs
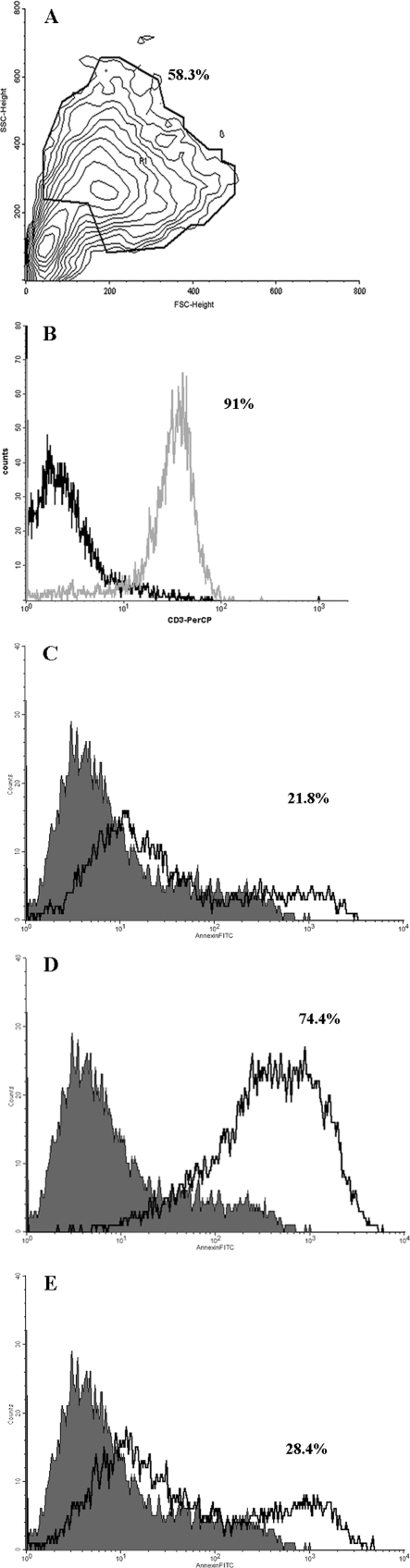
To confirm our findings with in vitro data, we performed a 5-hour assay with TALs and recombinant gal-3 in the presence or absence of lactose. A mechanically dissociated cutaneous metastasis with abundant lymphocyte infiltration was chosen, and T cells were traced by flow cytometry with an anti-CD3 antibody. Gated cells were 91% CD3(+) (Figure 5B). Using the selected region, a total of 1 × 106 cells were sorted on a forward-side scatter plot (Figure 5A) and incubated with recombinant gal-3 in the presence or absence of lactose (see Materials and Methods). The treatment of sorted TALs with 20 μmol/L recombinant gal-3 induced a 3.4-fold increase of annexin-V-positive cells. As shown in Figure 5C, nontreated TALs were 21.8% annexin-V-positive, whereas TALs treated with gal-3 were 74.4% positive (Figure 5D). Figure 5E shows no significant increase in annexin-V binding when cells were incubated in the presence of gal-3 and lactose. Lactose treatment alone had no effect on cell apoptosis (data not shown).
Figure 5.
Gal-3-mediated apoptosis of tumor-associated lymphocytes from a cutaneous metastasis. TALs were traced and sorted in a forward/side scatter plot (A) according to CD3 positivity (B). Annexin-V binding of sorted nontreated cells is 21.8% (C). The sorted cells treated with 20 μmol/L recombinant gal-3 show a 74.4% annexin-V binding (D), which is inhibited in the presence of 100 mmol/L lactose (E).
Discussion
The tumor surveillance concept, initially introduced by Sir Macfarlane Burnet in 1950 and then largely abandoned, has been recently revisited and rebaptised as “tumor immunoediting,” which proposes that the organism is initially able to cope with incipient tumors,26 although in some cases neoplastic cells eventually escape immune destruction allowing tumor growth and progression. This escape may be achieved either by hiding target antigens, ie, down-regulating HLA-I molecules, or through the inactivation of attacking lymphocytes. There are several mechanisms involved in this process of “tumor counterattack,” and galectins expression by tumor cells is one of them. We and others have shown that melanoma B16 clones in which gal-1 expression was knocked down by transfection with gal-1 antisense DNA9 were rejected by the host. However, this evidence, obtained in experimental systems, has not yet been demonstrated in the human setting, even when we have previously shown that human metastatic melanoma expresses gal-1.9 Although a functional correlation between galectin expression and lymphocyte apoptosis in vivo in humans is not possible through the analysis of biopsies, it might be inferred from circumstantial evidence. Therefore, we have performed a detailed analysis of the correlation between expression of gal-3, gal-1, and TAL apoptosis in primary and metastatic melanoma. We have found that gal-3 expressed by tumor cells correlates with TAL apoptosis. To contrast our findings with in vitro data, TALs from cryopreserved cutaneous metastasis from a melanoma patient were treated with recombinant gal-3 with or without lactose to ascertain the apoptotic effect of gal-3. Annexin-V binding was augmented 3.4-fold in gal-3-treated cells, and no increased annexin-V binding was observed in the gal-3/lactose sample. These results demonstrate that gal-3 mediates TAL apoptosis.
It should be noted that the variability of the data obtained by immunohistochemistry, as shown in Tables 1 and 2, is most probably due to differential expression of galectins by tumor cells within each biopsy (Figure 3), an event that remains to be understood. This same variability could partially explain the heterogeneity in the ISNT data. Validation of the ISNT procedure was done by performing serial determinations in normal hyperplastic lymph nodes. The high reproducibility of the ISNT values (data not shown) discarded the possibility that the variability of TAL apoptosis in tumor samples was due to the methodology used. Taking into consideration the heterogeneity in galectins and ISNT positivity within tumors, we decided to perform the statistical analysis by evaluating discrete fields, rather than focusing on the mean percentages of the different tumors analyzed.
Although histochemistry data support a pro-apoptotic role previously described for gal-3,14 there were gal-3(+) areas in which no apoptotic TALs were found. There is vast evidence demonstrating that regulation of galectins function is a complex process that depends on a variety of microenvironmental factors produced by cancer and immune cells, as well as matrix components. For instance, metalloproteinases −2, −9, and −13, which are up-regulated in cancer cells,19,27 modify gal-3 by cleaving the Ala62-Tyr63 bond.18 This would result in the loss of its apoptotic effect14 when gal-3 is released into the extracellular medium. On the other hand, recent studies have demonstrated an anti-apoptotic role of cytoplasmic gal-3 in human cancer cell lines16,17; this activity is regulated by its phosphorylation.17,28 Gal-3 was also described to be pro-apoptotic when it resides in the nucleus29; therefore, opposite effects have been described for intracellular gal-3. Moreover, a cross-talk model has been suggested in which apoptosis induced by secreted soluble gal-3 is blocked by cytoplasmic gal-3.14
How to explain that only a minority of human melanoma areas show positive correlation between gal-1 expres-sion and TAL apoptosis? There is extensive evidence13,15,30,31 for the apoptotic role of gal-1 in thymocytes and activated lymphocytes expressing the required glycosyltransferases that modulate the modification of CD45 core 2-O-glycans.32 However, the lack of correlation found in this report was not due to the absence of sensitive lymphocytes, because most TALs were CD45RO(+), suggesting that they were indeed resistant to the gal-1 apoptosis signal. He and Baum33 have recently demonstrated that stromal cells may synthesize gal-1 that deposits in tumor stroma and kills T cells at a 10-fold lower concentration (45 μg/ml) than the concentration required when gal-1 is in soluble form. Because epithelioid cell melanoma tumors tend to grow as solid sheets with scarce stroma formation, this could explain why gal-1 production by tumor cells is in most fields incapable of T-cell killing. More studies are necessary to elucidate this issue. These results point to the presence of other factors associated to the T-cell apoptosis pathway or to the functional status of gal-1 itself and/or to microenvironment interactions.
We have also found ISNT(+)/gal-1(−)/gal-3(−) areas, suggesting that other factors could also be inducing TAL apoptosis. It has been reported that Fas ligand expression in melanoma enable cancer cells to deliver death signals to activated Fas(+) lymphocytes,6,7 APO2L has also been involved in apoptosis of lymphocytes,34 and interleukin-10 expressed by melanoma cells or by abnormal CD4−/CD8− tumor-infiltrating lymphocytes is a putative immunosuppressive factor.8,35 In the whole, only ∼23% of the TALs were found to be apoptotic, independent of the cause. The viability of most TALs would explain why they may be expanded from tumor biopsies and give rise to CD8+/CD4+ populations that, when reinfused to the patient, are able to induce clinical regressions.5 There is a dialogue between tumor cells and the immune system that remains unclear. Galectins could be implicated in the apoptotic process of TALs, but this activity might be regulated by other defined and yet not defined microenvironmental components, the elucidation of which will be helpful in understanding tumor escape mechanisms, giving rise to further therapeutic approaches.
Supplementary Material
Acknowledgments
We thank Marcela Castro of the Pathology Department of the Instituto Alexander Fleming for her technical assistance and Sofia Silva for the grammatical revision of the manuscript.
Footnotes
Address reprint requests to José Mordoh, M.D., Head, Laboratory of Cancerology, Fundación Instituto Leloir, Address: Patricias Argentinas 435 (C1405BWE) Ciudad de Buenos Aires, Argentina. E-mail: jmordoh@leloir.org.ar.
Supported by grants from the Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), the Agencia Nacional de Promoción Científica y Tecnológica, the University of Buenos Aires, the Fundación Sales, the Fundación para la Investigación y Prevención del Cáncer, the Fundación Mosoteguy, and the Fundación María Calderón de la Barca, Argentina. J.M. is a research investigator of the CONICET and M.R.Z. is a fellow of the same Institution. D.F. is a fellow of the Agencia Nacional de Promoción Científica y Tecnológica, and M.B. and A.I.B. are fellows of the Fundación Sales.
M.R.Z. and D.F. contributed equally to this work.
Supplemental material for this article can be found on http://ajp.amjpathol.org.
References
- Greenlee RT, Murray T, Bolden S, Wingo PA. Cancer statistics 2000. CA-Cancer J Clin. 2000;50:7–33. doi: 10.3322/canjclin.50.1.7. [DOI] [PubMed] [Google Scholar]
- Ferradini L, Mackensen A, Genevee C, Bosq J, Duvillard P, Avril MF, Hercend T. Analysis of T cell receptor variability in tumor-infiltrating lymphocytes from a human regressive melanoma: evidence for in situ T cell clonal expansion. J Clin Invest. 1993;91:1183–1190. doi: 10.1172/JCI116278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- thor Straten P, Guldberg P, Gronbaeck K, Hansen MR, Kirkin AF, Seremet T, Zeuthen J, Becker JC. In situ T cell responses against melanoma comprise high numbers of locally expanded T cell clonotypes. J Immunol. 1999;163:443–447. [PubMed] [Google Scholar]
- Dudley ME, Wunderlich JR, Shelton TE, Even J, Rosenberg SA. Generation of tumor-infiltrating lymphocyte cultures for use in adoptive transfer therapy for melanoma patients. J Immunother. 2003;26:332–342. doi: 10.1097/00002371-200307000-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenberg SA, Dudley ME. Cancer regression in patients with metastatic melanoma after the transfer of autologous antitumor lymphocytes. Proc Natl Acad Sci USA. 2004;101(Suppl 2):14639–14645. doi: 10.1073/pnas.0405730101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hahne M, Rimoldi D, Schroter M, Romero P, Schreier M, French LE, Schneider P, Bornand T, Fontana A, Lienard D, Cerottini J, Tschopp J. Melanoma cell expression of Fas(Apo-1/CD95) ligand: implications for tumor immune escape. Science. 1996;274:1363–1366. doi: 10.1126/science.274.5291.1363. [DOI] [PubMed] [Google Scholar]
- Andreola G, Rivoltini L, Castelli C, Huber V, Perego P, Deho P, Squarcina P, Accornero P, Lozupone F, Lugini L, Stringaro A, Molinari A, Arancia G, Gentile M, Parmiani G, Fais S. Induction of lymphocyte apoptosis by tumor cell secretion of FasL-bearing microvesicles. J Exp Med. 2002;195:1303–1316. doi: 10.1084/jem.20011624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerlini G, Tun-Kyi A, Dudli C, Burg G, Pimpinelli N, Nestle FO. Metastatic melanoma secreted IL-10 down-regulates CD1 molecules on dendritic cells in metastatic tumor lesions. Am J Pathol. 2004;165:1853–1863. doi: 10.1016/S0002-9440(10)63238-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rubinstein N, Alvarez M, Zwirner NW, Toscano MA, Ilaguerri JM, Bravo A, Mordoh J, Fainboim L, Podhajcer OL, Rabinovich GA. Targeted inhibition of galectin-1 gene expression in tumor cells results in heightened T cell-mediated rejection; a potential mechanism of tumor-immune privilege. Cancer Cell. 2004;5:241–251. doi: 10.1016/s1535-6108(04)00024-8. [DOI] [PubMed] [Google Scholar]
- Nangia-Makker P, Honjo Y, Sarvis R, Akahani S, Hogan V, Pienta KJ, Raz A. Galectin-3 induces endothelial cell morphogenesis and angiogenesis. Am J Pathol. 2000;156:899–909. doi: 10.1016/S0002-9440(10)64959-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rabinovich GA, Baum LG, Tinari N, Paganelli R, Natoli C, Liu FT, Iacobelli S. Galectins and their ligands: amplifiers, silencers or tuners of the inflammatory response? Trends Immunol. 2002;23:313–320. doi: 10.1016/s1471-4906(02)02232-9. [DOI] [PubMed] [Google Scholar]
- Liu F-T, Rabinovich GA. Galectins as modulators of tumour progression. Nat Rev Cancer. 2005;5:29–41. doi: 10.1038/nrc1527. [DOI] [PubMed] [Google Scholar]
- Perillo NL, Pace KE, Seilhamer JJ, Baum LG. Apoptosis of T cells mediated by galectin-1. Nature. 1995;378:736–739. doi: 10.1038/378736a0. [DOI] [PubMed] [Google Scholar]
- Fukumori T, Takenaka Y, Yoshii T, Choi Kim HR, Hogan V, Inohara H, Kagawa S, Raz A. CD29 and CD7 mediate galectin-3-induced type II T-cell apoptosis. Cancer Res. 2003;63:8302–8311. [PubMed] [Google Scholar]
- Rabinovich GA, Daly G, Dreja H, Tailor H, Riera CM, Hirabayashi J, Chernajovsky Y. Recombinant galectin-1 and its genetic delivery suppress collagen-induced arthritis via T cell apoptosis. J Exp Med. 1999;190:385–398. doi: 10.1084/jem.190.3.385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akahani S, Nangia-Makker P, Inohara H, Kim HR, Raz A. Galectin-3: a novel antiapoptotic molecule with a functional BH1 (NWGR) domain of Bcl-2 family. Cancer Res. 1997;57:5272–5276. [PubMed] [Google Scholar]
- Takenaka Y, Fukumori T, Yoshii T, Oka N, Inohara H, Kim HR, Bresalier RS, Raz A. Nuclear export of phosphorylated galectin-3 regulates its antiapoptotic activity in response to chemotherapeutic drugs. Mol Cell Biol. 2004;24:4395–4406. doi: 10.1128/MCB.24.10.4395-4406.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ochieng J, Green B, Evans S, James O, Warfield P. Modulation of the biological functions of galectin-3 by matrix metalloproteinases. Biochim Biophys Acta. 1998;1379:97–106. doi: 10.1016/s0304-4165(97)00086-x. [DOI] [PubMed] [Google Scholar]
- Hofmann UB, Eggert AA, Blass K, Brocker EB, Becker JC. Expression of matrix metalloproteinases in the microenvironment of spontaneous and experimental melanoma metastases reflects the requirements for tumor formation. Cancer Res. 2003;63:8221–8225. [PubMed] [Google Scholar]
- Hernandez JD, Baum LG. Ah, sweet mystery of death! Galectins and control of cell fate. Glycobiology. 2002;12:127R–136R. doi: 10.1093/glycob/cwf081. [DOI] [PubMed] [Google Scholar]
- Koji T. Nonradioactive in situ nick translation: a useful molecular histochemical tool to detect single-stranded DNA breaks. Acta Histochem Cytochem. 1996;29:71–79. [Google Scholar]
- Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248–254. doi: 10.1006/abio.1976.9999. [DOI] [PubMed] [Google Scholar]
- Kockx MM, De Meyer GR, Bortier H, de Meyere N, Muhring J, Bakker A, Jacob W, Van Vaeck L, Herman A. Luminal foam cell accumulation is associated with smooth muscle cell death in the intimal thickening of human saphenous vein grafts. Circulation. 1996;94:1255–1262. doi: 10.1161/01.cir.94.6.1255. [DOI] [PubMed] [Google Scholar]
- Okada K, Komuta K, Hashimoto S, Matsuzaki S, Kanematsu T, Koji T. Frequency of apoptosis of tumor-infiltrating lymphocytes induced by fas counterattack in human colorectal carcinoma and its correlation with prognosis. Clin Cancer Res. 2000;6:3560–3564. [PubMed] [Google Scholar]
- Gerdes J, Lemke H, Baisch H, Wacker HH, Schwab U, Stein H. Cell cycle analysis of a cell proliferation-associated human nuclear antigen defined by the monoclonal antibody Ki-67. J Immunol. 1984;133:1710–1715. [PubMed] [Google Scholar]
- Shankaran V, Ikeda H, Bruce AT, White JM, Swanson PE, Old LJ, Schreiber RD. IFNγ and lymphocytes prevent primary tumour development and shape tumour immunogenicity. Nature. 2001;410:1107–1111. doi: 10.1038/35074122. [DOI] [PubMed] [Google Scholar]
- Guevremont M, Martel-Pelletier J, Boileau C, Liu FT, Richard M, Fernandes JC, Pelletier JP, Reboul P. Galectin-3 surface expression on human adult chondrocytes: a potential substrate for collagenase-3. Ann Rheum Dis. 2004;63:636–643. doi: 10.1136/ard.2003.007229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshii T, Fukumori T, Honjo Y, Inohara H, Kim HR, Raz A. Galectin-3 phosphorylation is required for its anti-apoptotic function and cell cycle arrest. J Biol Chem. 2002;277:6852–6857. doi: 10.1074/jbc.M107668200. [DOI] [PubMed] [Google Scholar]
- Califice S, Castronovo V, Bracke M, van den Brule F. Dual activities of galectin-3 in human prostate cancer: tumor suppression of nuclear galectin-3 vs tumor promotion of cytoplasmic galectin-3. Oncogene. 2004;23:7527–7536. doi: 10.1038/sj.onc.1207997. [DOI] [PubMed] [Google Scholar]
- Pace KE, Hahn HP, Pang M, Nguyen JT, Baum LG. CD7 delivers a pro-apoptotic signal during galectin-1-induced T cell death. J Immunol. 2000;165:2331–2334. doi: 10.4049/jimmunol.165.5.2331. [DOI] [PubMed] [Google Scholar]
- Hahn HP, Pang M, He J, Hernandez JD, Yang RY, Li LY, Wang X, Liu FT, Baum LG. Galectin-1 induces nuclear translocation of endonuclease G in caspase- and cytochrome c-independent T cell death. Cell Death Differ. 2004;11:1277–1286. doi: 10.1038/sj.cdd.4401485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nguyen JT, Evans DP, Galvan M, Pace KE, Leitenberg D, Bui TN, Baum LG. CD45 modulates galectin-1-induced T cell death: regulation by expression of core 2-O-glycans. J Immunol. 2001;167:5697–5707. doi: 10.4049/jimmunol.167.10.5697. [DOI] [PubMed] [Google Scholar]
- He J, Baum LG. Presentation of galectin-1 by extracellular matrix triggers T cell death. J Biol Chem. 2004;279:4705–4712. doi: 10.1074/jbc.M311183200. [DOI] [PubMed] [Google Scholar]
- Martinez-Lorenzo MJ, Anel A, Alava MA, Pineiro A, Naval J, Lasierra P, Larrad L. The human melanoma cell line MelJuSo secretes bioactive FasL and APO2L/TRAIL on the surface of microvesicles. Possible contribution to tumor counterattack. Exp Cell Res. 2004;295:315–329. doi: 10.1016/j.yexcr.2003.12.024. [DOI] [PubMed] [Google Scholar]
- Prins RM, Incardona F, Lau R, Lee P, Claus S, Zhang W, Black KL, Wheeler CJ. Characterization of defective CD4−CD8− T cells in murine tumors generated independent of antigen specificity. J Immunol. 2004;172:1602–1611. doi: 10.4049/jimmunol.172.3.1602. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.