Abstract
Current combination therapies cure Helicobacter pylori infection in 75 to 85% of cases. However, many treatment failures are not explained by antibiotic resistance. Our goal was to explore treatment failures under in vivo conditions by using the H. pylori Sydney strain (SS1) mouse model. Mice infected with H. pylori (SS1) were treated with monotherapies or combination therapies used in human trials. Bacterial levels and distribution of organisms within the stomach were assessed 24 h after treatment to determine clearance and location of treatment failures and 29 days after treatment to determine cure rates. Except for treatment with metronidazole, mono- and dual therapies did not cure infection but resulted in decreases in bacterial levels and differences in distribution within the stomach. In cases of treatment failure when clarithromycin was used, omeprazole and dual therapy with omeprazole and amoxicillin resulted in organisms being cleared from the antrum, but organisms remained in the antrum-body transitional zone. The triple therapies of OMC and bismuth subcitrate, metronidazole, and tetracycline were successful in eradicating infection. Except for metronidazole monotherapy and triple therapy with OAC, there was good correlation between the Sydney strain mouse model and humans with respect to the success of antimicrobial therapy. The antrum-body transitional zone was identified as a sanctuary site in treatment failure. This could result from antimicrobial agents not functioning effectively at this site or bacteria in this location expressing products that protect them against antimicrobial agents. This is the first demonstration of a possible sanctuary site as a reason for failure of therapy.
Helicobacter pylori infection in humans is a major cause of gastroduodenal diseases including gastric and duodenal ulceration (31, 40) as well as gastric cancer (4). Among the myriad of treatments used for H. pylori infection, the highest eradication rates are achieved with a proton pump inhibitor in combination with two antimicrobials—typically a nitroimidazole (metronidazole or tinidazole) and either amoxicillin or clarithromycin. However, there is still a minimum 10 to 25% failure rate with these combination therapies (7, 27, 28, 30, 37). Efforts to maximize therapeutic efficacy against H. pylori have proved difficult due to the fact that in vitro susceptibility testing correlates poorly to in vivo efficacy. For example, nearly all strains of H. pylori are sensitive to small amounts of amoxicillin in vitro, the typical MICs of which are 0.01 to 0.1 μg/ml (18). Despite the fact that these concentrations are attainable within the gastric juices and sera of humans (13), amoxicillin monotherapy is not effective in eradicating infection (22).
The reasons that in vitro testing fails to predict clinical efficacy are unknown. Although resistance to antimicrobial agents is a recognized cause of treatment failure, at least 50% of failures remain unexplained. It is possible that the complex nature of H. pylori's ecological niche contributes to poor performance of antimicrobials in vivo. The thickness of the mucus layer, the shape and motility of the stomach, and the acidic environment may all cause inadequate distribution and concentration of the drugs at the surface of the gastric mucosa.
Indeed, it has been proposed that suppression—rather than cure—of the infection occurs because antimicrobial treatment is more effective at some sites than at others. Recurrence of H. pylori infection after initial successful eradication has been reported in a number of studies and appears to mainly stem from recrudescence of the original strain (41). Such recrudescence indicates that although bacterial colonization can be suppressed to undetectable levels after treatment, a small number of organisms may have persisted in sanctuary sites which were inaccessible to antimicrobial action. There are few studies that support the concept that H. pylori organisms may escape killing in certain gastric sites in humans, as was shown for amoxicillin in the gastric fundus (5).
Identification of such sites in humans is difficult, as the number of gastric biopsy specimens that can be taken from one individual is limited. Furthermore, in routine practice, determination of treatment success is done 4 weeks after therapy has been completed. During this period, recrudescence of organisms to pretreatment levels may have already taken place. The H. pylori Sydney strain (SS1) mouse model is a well-established model that has a distribution of H. pylori colonization similar to that seen in humans (26). The aim of the present study was to use the H. pylori SS1 mouse model to test a variety of treatment regimens employed in human practice and to map the location and distribution of any organisms remaining in the stomach in order to identify possible sites of treatment failure.
MATERIALS AND METHODS
Abbreviations.
CSA, Campylobacter-selective agar; GSSA, Glaxo-selective supplement agar; BHI, brain heart infusion; SPF, specific pathogen free; CFU, colony-forming unit; MIC, minimal inhibitory concentration; LPS, lipopolysaccharide; OA, omeprazole plus amoxicillin; BMT, bismuth plus metronidazole plus tetracycline; OAC, omeprazole plus amoxicillin plus clarithromycin; OMC, omeprazole plus metronidazole plus clarithromycin.
Bacterial culture.
H. pylori SS1 was subcultured on CSA (26). Liquid cultures were used to infect the mice. Briefly, bacteria from plates were inoculated into a 1-liter flask containing 300 ml of BHI broth (Oxoid, Ltd., Basingstoke, England) containing 5% (vol/vol) sterile horse serum (Oxoid, Ltd.), and 5 mg of amphotericin B (Fungizone, E. R. Squibb & Sons, Inc., Princeton, N.J.) per ml and placed in an anaerobic jar with an anaerobic gas-generating kit (Oxoid Ltd.) without a catalyst for 36 to 48 h at 37°C with shaking at 60 rpm. Following incubation, cultures were centrifuged at 5,000 rpm for 5 min, and the cell pellets were resuspended in sterile BHI to a concentration of approximately 109 organisms per ml. Bacteria harvested from infected mice were cultured on GSSA (26), as GSSA is more selective than CSA.
Animals and treatment protocols.
SPF, 7- to 8-week-old, female C57BL/6 mice were obtained from Biological Resource Centre, Little Bay, N.S.W., Australia. Animals (n = 290) were infected three times over a 5-day period with a 0.1-ml volume containing 108 H. pylori (Sydney strain, SS1) organisms as previously described (26). Two weeks following inoculation, the mice were randomly divided into 10 groups and treated as follows: (i) no treatment, (ii) amoxicillin (Sigma, St. Louis, Mo.), (iii) colloidal bismuth subcitrate (Parke-Davis, Sydney, Australia), (iv) clarithromycin (Abbott Laboratories, Sydney, Australia), (v) metronidazole (Sigma), (vi) omeprazole (Astra Hassle, Molendal, Sweden), (vii) dual therapy of OA, (viii) triple therapy consisting of BMT (Sigma), (ix) triple therapy consisting of OAC, or (x) triple therapy consisting of OMC. Each group had 30 animals, with the exception of the omeprazole group, which had 20 animals.
The dosage and treatment schedules for each group are shown in Table 1. All treatments were administered intragastrically in a 0.1-ml volume twice daily for 7 days, with the exception of BMT, which was given once a day. For treatment regimens that included omeprazole, there was a 30- to 60-min interval between dosing with omeprazole and the other antimicrobial agents to allow a change in stomach acid pH to occur. The weight of each mouse was approximated to be 20 g, and treatments, with the exception of omeprazole, were calculated by using the standard dosages given to 70-kg adult humans per day. The different dosages of clarithromycin used in OAC and OMC were chosen according to the results of the MACH 1 study (28). Omeprazole was given at a dosage required for acid suppression in rats (24).
TABLE 1.
Treatment schedules for groups of mice
| Group (n) | Treatment | Total daily dosage |
|---|---|---|
| 1 (30) | Control | |
| 2 (30) | Amoxicillin | 28.6 mg/kg/day |
| 3 (30) | Bismuth subcitrate | 6.15 mg/kg/day |
| 4 (30) | Clarithromycin | 14.3 mg/kg/day |
| 5 (30) | Metronidazole | 22.5 mg/kg/day |
| 6 (20) | Omeprazole | 400 μmol/kg/day |
| 7 (30) | OA | Omeprazole, 400 μmol/kg/day; amoxicillin, 28.6 mg/kg/day |
| 8 (30) | BMT (9) | Bismuth, 6.15 mg/kg/day; metronidazole, 22.5 mg/kg/day; tetracycline, 50 mg/kg/day |
| 9 (30) | OAC (28) | Omeprazole, 400 μmol/kg/day; amoxicillin, 28.6 mg/kg/day; clarithromycin, 14.3 mg/kg/day |
| 10 (30) | OMC (28) | Omeprazole, 400 μmol/kg/day; metronidazole, 14.2 mg/kg/day; clarithromycin, 7.15 mg/kg/day |
With the exception of omeprazole and clarithromycin, the medications were dissolved in sterile water and stirred vigorously prior to use. The three medications composing BMT were mixed together just prior to use (9). Omeprazole was dissolved in a 5% hydroxypropyl methylcellulose solution buffered with 0.2% sodium bicarbonate and adjusted to pH 9 with sodium hydroxide to achieve the concentration shown in Table 1. The clarithromycin dose for the 30 animals was dissolved in 0.5 ml of methanol and diluted in 3.5 ml of distilled water. Throughout the study, animals were fed a diet of autoclaved commercial food pellets (Lab Feed Division, Clarkel Holding Pty. Ltd., Northbridge, Australia) and given sterile water ad libitum. All protocols were approved by the Animal Care and Ethics Committee, University of New South Wales, Sydney, Australia (ACE 97/54).
Animals were sacrificed 36 h (n = 20 per group, except omeprazole [n = 10]) and 29 days (n = 10 per group) after the cessation of treatment for assessment of bacterial colonization by rapid urease test, culture, and histology.
Histology.
One-half of each stomach was placed into 10% buffered formalin and processed in paraffin, and 4-μm sections were stained with a modified Steiner silver stain. Slides were coded and examined blind, i.e., examiners were unaware of the specific details of preparation. Colonization was assessed on a five-point scale: 0, no bacteria; 1, less than 1/3 of crypts colonized with 1 to 10 bacteria; 2, 1/3 to 2/3 of crypts colonized with 10 to 20 bacteria; 3, 2/3 of the crypts colonized with >20 bacteria; and 4, all crypts colonized with >20 bacteria. Colonization was assessed in three regions—the antrum, the body, and the transition zone between these regions (26). The antrum-body transitional zone was defined as the area where parietal cells of the body type glands disappear and antral type glands become dominant. Evaluation of colonization within the antrum-body transitional zone was performed by identifying the center point of the transitional zone and counting bacteria within a five-gland radius on both sides.
Viable counts and rapid urease test.
The other halves of the stomachs (n = 10 for each test) collected at 24 h were used either for the rapid urease test (16) or for determining viable counts of H. pylori organisms. Similarly, half-stomachs (n = 5 for each test) from the 29-day collection were used for a rapid urease test or for determination of viable counts. For viable counts, weighed stomach halves were placed in 1 ml of BHI broth and homogenized with an Ultra-Turrax for 10 to 15 s. Tenfold serial dilutions were plated onto GSSA and incubated for 5 days in a CO2 incubator set at 10% CO2 and 95% humidity. Viable counts were expressed as CFU per gram of stomach tissue.
Statistical analysis.
A Kruskal-Wallis test with posthoc analysis performed using Dunn's multiple comparison test was used to compare the viable counts from each treatment group to those of the control groups (alpha level of P < 0.05). The same tests were applied to histology data to compare colonization levels within corresponding stomach regions between the treatment groups and controls (an alpha level of P < 0.01 was used adjust for the number of tests performed). Histology data between stomach regions within a single treatment group (i.e., antrum versus antrum-body) was compared by using two-tailed Mann-Whitney U tests (with an adjusted alpha level of P < 0.01).
Definition of clearance and eradication of H. pylori infection.
Clearance of the infection was defined as the absence of bacteria 1 day after treatment. H. pylori was considered to be eradicated if no organisms were detected 29 days after treatment.
MICs of antimicrobials for H. pylori SS1.
MICs of amoxicillin, clarithromycin, bismuth subcitrate, tetracycline, metronidazole, and omeprazole for H. pylori SS1 were determined by the plate dilution method (12). Plates containing concentrations of antimicrobial compounds from 0.002 to 64 μg/ml were inoculated with approximately 104 organisms and incubated for 5 days at 10% CO2 and 95% humidity. The MIC was the lowest concentration of the antimicrobial that produced no growth on the plate in question.
RESULTS
H. pylori SS1 was tested for susceptibility to commonly used antimicrobial compounds both in vitro and in vivo.
MICs of antimicrobials.
The MICs against H. pylori SS1 for each of the antimicrobial compounds used in this study for each antimicrobial agent were as follows: metronidazole, 0.25 μg/ml; clarithromycin, 0.015 μg/ml; amoxicillin, 0.5 μg/liter; bismuth subcitrate, 32 μg/ml; tetracycline, 0.25 μg/ml; and omeprazole, 64 μg/ml.
Treatment efficacy in the mouse model.
The effect of antimicrobial treatment on colonization of the mouse stomachs by H. pylori was assessed by histology, viable counts (Table 2), and rapid urease tests. Results from the three types of assessments were similar, although viable counts were the most sensitive, detecting as few as 103 organisms per gram of stomach tissue. There was 100% correlation of the results between histological assessment and urease assay; as such, the latter results were not included. In the untreated group, levels of 106 to 107 organisms per gram of stomach tissue were observed and the colonization remained stable during the period of the experiment as has been previously reported (26).
TABLE 2.
H. pylori colonization 24 h and 29 days following antimicrobial treatments
| Treatment | % of mice infected (viable counts)a | Histological gradingb
|
||
|---|---|---|---|---|
| Antrum (range) | Antrum-body transition (range) | Body (range) | ||
| 24 h | ||||
| Control | 100 | 3 (0.5-4) | 3 (2-4) | 0 (0) |
| Amoxicillin | 100 | 2.5 (0.5-4) | 3 (1-4) | 0 (0) |
| Bismuth | 100 | 2 (0-3.5) | 2 (0.5-3) | 0 (0) |
| Clarithromycin | 100 | 0 (0-1)c | 1.5 (0-2) | 0 (0) |
| Metronidazole | 0 | 0 (0)c | 0 (0)c | 0 (0) |
| Omeprazole | 100 | 0.25 (0-0.5)c | 2 (0-3) | 2 (1-3)d |
| OA | 100 | 0 (0-1.5)c | 1 (0-2)c | 0 (0) |
| BMT | 0 | 0 (0)c | 0 (0)c | 0 (0) |
| OAC | —e | 0 (0)c | 0 (0)c | 0 (0) |
| OMC | 0 | 0 (0)c | 0 (0)c | 0 (0) |
| 29 days | ||||
| Control | 100 | 3.25 (0.5-4) | 3 (0.5-4) | 1 (0-1) |
| Amoxicillin | 100 | 3 (0.5-3.5) | 2.5 (1-3) | 0.5 (0-1) |
| Bismuth | 100 | 3 (2-4) | 3 (2-3) | 0.5 (0-1) |
| Clarithromycin | 100 | 2 (0.5-3.5) | 2 (0-4) | 0.5 (0-0.5) |
| Metronidazole | 40 | 0 (0-2)c | 0 (0-3)c | 0 (0-0.5)c |
| Omeprazole | 100 | 1.5 (0.5-3.5) | 2 (0.5-3) | 0.5 (0-1) |
| OA | 100 | 2.5 (0.5-3.5) | 2 (0.5-3) | 0 (0-0.5) |
| BMT | 20 | 0 (0-0.5)c | 0 (0-1)c | 0 (0)c |
| OAC | 100e (4 of 4) | 2 (0-3.5) | 3 (0-3.5) | 0 (0-1) |
| OMC | 0 | 0 (0)c | 0 (0)c | 0 (0)c |
For collection at 24 h, n = 10; for collection at 29 days, n = 5.
Histology results are expressed as median values(range) out of n = 20 for the collection at 24 h and n = 10 for the collection at 29 days.
Significantly lower than scores from the same stomach region in the untreated control (P < 0.001).
Significantly higher than score in body region of the untreated control (P < 0.001).
—, 10/10 samples from the collection at 24 h and 1/5 samples from the collection at 29 days were unable to be assessed due to a swarming contaminant on the plates.
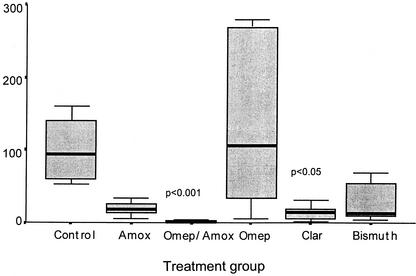
Mice treated with metronidazole monotherapy showed 100% clearance of H. pylori 36 h after treatment, with 60% of animals showing evidence of eradication at the day 29 collection. None of the other monotherapies or dual OA therapy eradicated H. pylori infection. A significant decrease in viable counts was observed 36 h after treatment in all treatments except for omeprazole monotherapy (Fig. 1). The latter is in keeping with the high MIC of omeprazole. Twenty-nine days later, colonization levels were similar to those obtained in the untreated controls (Table 2).
FIG. 1.
CFU of H. pylori obtained from an entire half of homogenized stomach 36 h after treatment with mono and dual therapies compared to the untreated control. The bar represents the median value, the box contains 50% of the data, and the bars show the range. Sample number is 10 for all groups. The P values are shown for data which differed significantly from the control group (P < 0.05).
All three triple therapies (BMT, OMC, and OAC) initially achieved clearance, with no H. pylori detected by any method (Table 2). However a swarming contaminant was found on all 10 of the viable count samples from the OAC group, so viable cell counts could not be accurately determined. Twenty-nine days after treatment, all the OMC-treated mice were devoid of H. pylori. Bacteria were detected in 1 of 10 mice by histology and in 1 of 5 mice by culture following BMT treatment. H. pylori was found in four out of the five viable count samples of OAC-treated mice, with the remaining sample containing a swarming contaminant preventing its inclusion in the data. In three of nine and in one of five mice from the OAC treatment group, clearance was observed by histology and rapid urease tests, respectively.
A total of four mice died during the study for reasons unrelated to the treatments—two within the OAC treatment group and one from the omeprazole and OA treatment groups. Three deaths were direct results of the intragastric administration of the antibiotics; the cause of death in the fourth animal is not known.
Effect of treatment on pattern of colonization.
Histological examination was used to assess the effect of drug treatment on gastric colonization patterns. In most groups, including the untreated controls, but not in the successful treatment antimicrobials (metronidazole, BMT, and OMC), the bacteria were found to primarily colonize the non-acid-secreting regions of the stomach, i.e., the antrum and the antrum-body transitional zone (Table 2). Correlation of the viable cell counts and the histology results in this trial revealed that the limit of detection of bacteria by histology was 104 organisms per gram of tissue. There was no difference in the number of bacteria colonizing the antrum compared to that of the antrum-body transitional zone in the control group at either collection.
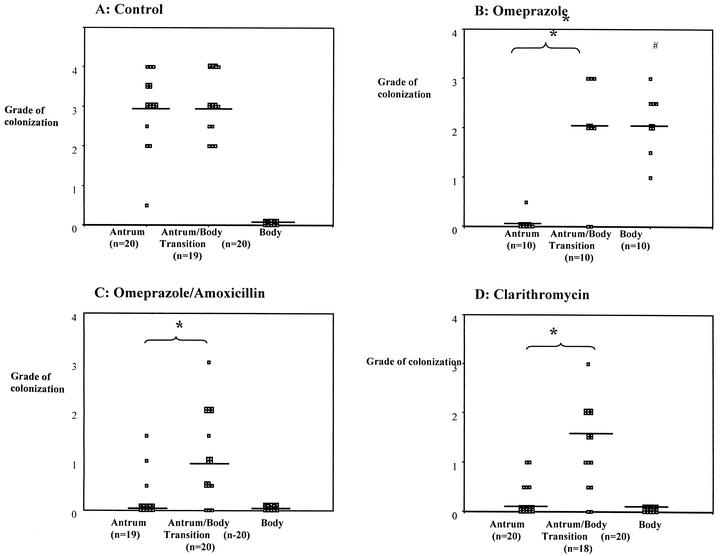
However, with three of the treatment regimens (omeprazole, OA, and clarithromycin) colonization within the antrum was significantly suppressed compared to that of the antrum-body transitional zone (Fig. 2; Table 2) at the 36-h collection. Comparison of the colonization patterns in the control mice with those animals receiving these three treatments indicated that the treatments were very successful (80%) in clearing the bacteria from the antrum, while the treatments had negligible effects on the bacteria in the antrum-body transitional zone. This decrease in bacterial levels in the antrum corresponded with significant decreases in the number of CFU in mice receiving clarithromycin and OA (Fig. 1). No decrease in CFU number was seen in the animals receiving omeprazole alone, and this corresponded with significantly increased numbers of H. pylori seen within the body mucosa 36 h after treatment. By 29 days after treatment, bacterial levels were returning to baseline levels, with a decrease in the body and an increase in the antrum.
FIG. 2.
Scatter plots showing grade (0 to 4) of H. pylori colonization in antrum, antrum-body transitional zone, and body regions of mouse stomachs 36 h after therapy with untreated controls (A), omeprazole (B), omeprazole and amoxicillin (C), and clarithromycin (D). Each data point represents a single histological section. The median value for each region is shown with a bar. *, significant difference between regions (P < 0.01). #, significant difference compared to body region of control group (P < 0.01).
DISCUSSION
Identification of sanctuary sites following antimicrobial therapy.
The results presented here show that the suppressive effects of several antimicrobial therapies on H. pylori do not occur with equal efficacy in all regions of the stomach. Specifically, we found that treatment of mice with clarithromycin, omeprazole, or a dual OA regimen was significantly more effective at reducing colonization within the antrum than within the transitional zone between the antrum and the body (Fig. 2). This result may offer an explanation for the failure of some treatment regimens. It suggests that the bacteria within this transitional zone are able to avoid being killed by antimicrobial agents. There are several possible explanations for this. In the transitional zone, organisms may be less sensitive due to special environmental conditions that may directly affect antimicrobial efficacy or there may be a regional difference in the expression of bacterial products that are protective against antimicrobial agents. Another explanation for treatment failure may be that H. pylori organisms are internalized by epithelial cells. This has only been shown in vitro by using epithelial cell lines and has not been demonstrated in in vivo systems (11). This would not explain the transitional zone as a preferential site for survival.
A major difference between the antral and body regions is the presence of acid-secreting parietal cells, which are lacking within the antrum. The number of parietal cells increases across the transitional zone between these two regions, reaching peak numbers within the body. The antimicrobial effect of many compounds, including bismuth (33), tetracycline (33), amoxicillin (14), and clarithromycin (10), is reduced under acidic conditions. In contrast, metronidazole has been found to be chemically stable under a wide range of pH values (39).
We have previously hypothesized that the gradual increase in the number of parietal cells across the transitional zone produces a pH gradient which results in the formation of microenvironments within the antrum-body border. These microenvironments may have optimal conditions for colonization by H. pylori, thereby allowing a flourishing population to develop. Alternatively, the microenvironment may be less than optimal, leading to the expression of stress proteins and thereby improving the organism's chances for survival (38). Indeed, pH has been found to regulate the production of H. pylori proteins at both the transcriptional and posttranscriptional levels (1, 21, 32). Regulation can occur over subtle variations in pH, similar to those that may occur in the transitional zone (38). This includes the induction of cagA transcription, which is significantly higher after growth at pH 6 than at pH 5 or 7 (21).
Evaluation of antimicrobial therapies with the H. pylori SS1 mouse model.
In this study, only a single strain of H. pylori was used. The bacterial strain chosen was H. pylori SS1, as this strain consistently shows high levels of bacterial colonization in the mouse and has been used worldwide by numerous investigators for assessment of bacterial colonization (25). H. pylori SS1 was responsive to all antimicrobials tested in vitro. The MICs were well within the range found for other sensitive strains (2, 3, 6, 15, 18, 34), indicating that H. pylori SS1 does not have primary resistance to any of these treatments. Furthermore, treatment regimens and dosages, extrapolated from the MACH human trials (27, 28), produced effects on infected mice similar to those seen in humans. That is, most of the monotherapies as well as the dual OA treatment were unsuccessful in eradicating H. pylori SS1 from infected mice, whereas BMT and OMC, two of the most effective triple-therapy regimens in humans, were highly successful in this study. Therefore, the Sydney strain H. pylori mouse model is suited for the evaluation of treatments against H. pylori infection used in humans and has been successfully used by others to evaluate efficacy of treatments (19, 20). Importantly this model shows a similar colonization pattern to that seen in humans, with preferential colonization of the antrum (26). In mice, however, inflammation is less pronounced than that observed in humans. The 60% eradication rate caused by metronidazole monotherapy was high in comparison to the 20% eradication rate reported in humans (17). The reduced inflammation in mice compared to humans may be one explanation for the high success rate of monotherapy with metronidazole. There are convincing data in the literature that demonstrate that metronidazole can cross the gastric mucosa (13, 39). This may improve the success rate with this medication.
Another explanation may be the sensitivity of H. pylori SS1 to a low concentration of metronidazole (MIC, 0.25 μg/liter) in contrast to the high frequency of metronidazole-resistant H. pylori strains in humans. In another study that used the same model of metronidazole monotherapy, a 25% cure rate was achieved in animals (20). The reason for the difference between the latter study and our study is unclear, but it could be related to subtle differences among mouse strains.
In our study, we calculated the dosage of antibiotics given to the animals (animals were estimated to weigh 20 g) relative to humans (the equivalent of a 70-kg person). The dose of omeprazole used is known to inhibit acid production in rats (24). Also, the doses used for the BMT triple-therapy combination have consistently resulted in very high cure rates of H. pylori infection in this model (9). We are unaware of any other pharmacokinetic data for mice for the other treatments we used, which would help guide the choice of doses. It is, however, possible that different pharmacokinetics of, for example, metronidazole in mice could explain the different results when compared to data obtained in human trials.
An important objective of our study was to discover possible areas within the stomach where H. pylori organisms may escape being killed by the antibiotics. For that reason, results of histology, culture, and rapid urease analyses were compared 36 h after the last treatment dose was given and 4 weeks later. Concerning humans, there is agreement that one must delay declaring eradication until 4 weeks have passed since a patient stopped taking treatment. This is to distinguish between mere suppression and true eradication of H. pylori organisms. We cannot exclude the possibility that, at 36 h after the last treatment, there still is some carryover effect of previously given antibiotics in mice but it seems unlikely that this could be an explanation for the observation that there are sanctuary sites within the stomach where H. pylori is capable of surviving.
Just as in human trials, the two triple therapies, BMT and OMC, were both highly successful in the eradication of H. pylori SS1. Given the high success rate of metronidazole alone, it is not surprising that these metronidazole-inclusive therapies worked well. Interpretation of the OAC treatment results was complicated by the presence of a swarming contaminant on the viable count plates. Initial performance of OAC therapy, as assessed by histology and rapid urease testing, was successful, with no bacteria detected by either method. However, 29 days after treatment, OAC was not very effective at eradicating H. pylori infection. Histology results showed only 33% of mice to be cleared of infection. By viable counts, all mice examined contained H. pylori, although the sample size included only four animals. This result was surprising, as in human trials, OAC's efficacy is similar to that of OMC (27, 28). A possible explanation is the relatively high MIC of amoxicillin (0.5 mg/liter) that we observed. Failure of OAC may reflect the poor performance of clarithromycin monotherapy and OA dual therapy in this study, both of which were less effective than has been seen for humans. Monotherapy with clarithromycin has an estimated 30% success rate in humans (35), whereas dual OA treatment eradicates H. pylori in 50 to 60% of human patients (36). However, in these trials, higher doses of amoxicillin were used than in this study. Although clarithromycin, OA, and OAC therapies failed to parallel their eradication success in humans, suppression of colonization was consistently seen for these therapies in the H. pylori SS1 mouse model (Fig. 1). Possibly higher eradication rates, similar to those observed in humans, may be achieved if dosages are further optimized.
The mouse model, as described in this trial, allowed us to assess infection by both viable counts (expressed as CFU per gram) and histology, with the former providing a 1-log advantage over histology. Monotherapy with amoxicillin, bismuth subcitrate, or clarithromycin or dual OA therapy showed suppressive effects ranging from 0.5 to 2 logs 24 h after treatment (Fig. 1). No comparable change in bacterial load was detected 24 h after omeprazole monotherapy, although the distribution of organisms at this collection was altered in a fashion similar to that observed in omeprazole-treated humans (23, 29). Namely, the number of bacteria decreased in the antrum and increased within the body of the stomach (Fig. 2). These data confirm that, as in humans, omeprazole monotherapy does not cure H. pylori infection in mice (28, 29). This shift was seen in our early H. felis mouse model studies and is due to subtle changes in the local acid environment (8, 25).
It is possible that the relatively high volume (0.1 ml) per antibiotic given in solution promotes wide distribution throughout the stomach. In one study, Atherton et al. used scintigraphy and showed that, when the combination of omeprazole and amoxicillin was given, most of the administered medications pooled in the antrum, with no detection of the drugs in the fundus (5). This is one of the few human studies that suggests as a possible explanation for treatment failure insufficient distribution of antibiotics within the stomach. However, the clinical relevance of this is uncertain, as no data were provided as to whether the lack of distribution in the fundus was associated with treatment failure in that particular region of the stomach.
This study demonstrates for the first time that the transitional zones may be the sites in the stomach where H. pylori may escape being killed by antimicrobial agents. This would explain why in many human studies apparent therapeutic success is seen initially after cessation of the treatment regimen (i.e., suppression) yet the bacteria are found again 4 weeks after therapy. In some cases, it may take even longer for the sequestered bacteria to return to normal levels (41). In our study, the area of sequestration was found to be the transitional zones. We have previously argued that the transitional zones are special areas of colonization in the stomach where the organism behaves differently (38). This area of different physiology presumably makes the organism less susceptible to antimicrobial action. Further investigation of these bacteria within the transitional zone to identify these metabolic differences may provide insights that will allow the development of novel therapies that are less likely to fail.
Acknowledgments
This work was undertaken at the University of New South Wales, Sydney, Australia.
The work was supported by a grant from the National Health and Medical Research Council of Australia.
REFERENCES
- 1.Akada, J. K., M. Shirai, H. Takeuchi, M. Tsuda, and T. Nakazawa. 2000. Identification of the urease operon in Helicobacter pylori and its control by mRNA decay in response to pH. Mol. Microbiol. 36:1071-1084. [DOI] [PubMed] [Google Scholar]
- 2.Andersen, L. P., H. Colding, and J. E. Kristiansen. 2000. Potentiation of the action of metronidazole on Helicobacter pylori by omeprazole and bismuth subcitrate. Int. J. Antimicrob. Agents 14:231-234. [DOI] [PubMed] [Google Scholar]
- 3.Ani, A. E., A. O. Malu, J. A. Onah, D. M. M. Queiroz, G. Kirschner, and G. A. Rocha. 1999. Antimicrobial susceptibility test of Helicobacter pylori isolated from Jos, Nigeria. Trans. R. Soc. Trop. Med. Hyg. 93:659-661. [DOI] [PubMed] [Google Scholar]
- 4.Asghar, R. J., and J. Parsonnet. 2001. Helicobacter pylori and risk for gastric adenocarcinoma. Semin. Gastrointest. Dis. 12:203-208. [PubMed] [Google Scholar]
- 5.Atherton, J. C., A. Cockayne, M. Balsitis, G. E. Kirk, C. J. Hawkey, and R. C. Spiller. 1995. Detection of the intragastric sites at which Helicobacter pylori evades treatment with amoxycillin and cimetidine. Gut 36:670-674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bamba, H., Y. Kondo, R. M. Wong, S. Sekine, and F. Matsuzaki. 1997. Minimum inhibitory concentration of various single agents and the effect of their combinations against Helicobacter pylori, as estimated by a fast and simple in vitro assay method. Am. J. Gastroenterol. 92:659-662. [PubMed] [Google Scholar]
- 7.Bell, G. D., K. U. Powell, S. M. Burridge, A. F. Bowden, W. Atoyebi, G. H. Bolton, P. H. Jones, and C. Brown. 1995. Rapid eradication of Helicobacter pylori infection. Aliment. Pharmacol. Ther. 9:41-46. [DOI] [PubMed] [Google Scholar]
- 8.Danon, S. J., J. L. O'Rourke, N. D. Moss, and A. Lee. 1995. The importance of local acid production in the distribution of Helicobacter felis in the mouse stomach. Gastroenterology 108:1386-1395. [DOI] [PubMed] [Google Scholar]
- 9.Dick-Hegedus, E., and A. Lee. 1991. Use of a mouse model to examine anti-Helicobacter pylori agents. Scand. J. Gastroenterol. 26:909-915. [DOI] [PubMed] [Google Scholar]
- 10.Erah, P. O., A. F. Goddard, D. A. Barrett, P. N. Shaw, and R. C. Spiller. 1997. The stability of amoxycillin, clarithromycin and metronidazole in gastric juice—relevance to the treatment of Helicobacter pylori infection. J. Antimicrob. Chemother. 39:5-12. [DOI] [PubMed] [Google Scholar]
- 11.Evans, D. G., D. J. Evans, and D. Y. Graham. 1992. Adherence and internalisation of Helicobacter pylori by HEp-2 cells. Gastroenterology 102:1557-1567. [DOI] [PubMed] [Google Scholar]
- 12.Glupczynski, Y., M. Delmee, C. Bruck, M. Labbe, V. Avesani, and A. Burette. 1988. Susceptibility of clinical isolates of Campylobacter pylori to 24 antimicrobial and anti-ulcer agents. Eur. J. Epidemiol. 4:154-157. [DOI] [PubMed] [Google Scholar]
- 13.Goddard, A. F., M. J. Jessa, D. A. Barrett, P. N. Shaw, J. P. Idstrom, C. Cederberg, and R. C. Spiller. 1996. Effect of omeprazole on the distribution of metronidazole, amoxicillin, and clarithromycin in human gastric juice. Gastroenterology 111:358-367. [DOI] [PubMed] [Google Scholar]
- 14.Grayson, M. L., G. M. Eliopoulos, M. J. Ferraro, and R. C. J. Moellering. 1989. Effect of varying pH on the susceptibility of Campylobacter pylori to antimicrobial agents. Eur. J. Clin. Microbiol. Infect. Dis. 8:888-889. [DOI] [PubMed] [Google Scholar]
- 15.Grove, D. I., G. Koutsouridis, and A. G. Cummins. 1998. Comparison of culture, histopathology and urease testing for the diagnosis of Helicobacter pylori gastritis and susceptibility to amoxycillin, clarithromycin, metronidazole and tetracycline. Pathology 30:183-187. [DOI] [PubMed] [Google Scholar]
- 16.Hazell, S. L., T. J. Borody, A. Gal, and A. Lee. 1987. Campylobacter pyloridis gastritis I: detection of urease as a marker of bacterial colonization and gastritis. Am. J. Gastroenterol. 82:292-296. [PubMed] [Google Scholar]
- 17.Hirschl, A. M., and M. Pletschette. 1989. Antibiotic treatment of Campylobacter pylori infection, p. 217-224. In B. J. Rathbone and R. V. Heatley (ed.), Campylobacter pylori and gastroduodenal disease. Blackwell Scientific Publications Ltd., Oxford, England.
- 18.Hirschl, A. M., and M. L. Rotter. 1996. Amoxicillin for the treatment of Helicobacter pylori infection. J. Gastroenterol. 31(Suppl. 9):44-47. [PubMed] [Google Scholar]
- 19.Jenks, P. J., R. L. Ferrero, J. Tankovic, J. M. Thiberge, and A. Labigne. 2000. Evaluation of nitrofurantoin combination therapy of metronidazole-sensitive and -resistant Helicobacter pylori infections in mice. Antimicrob. Agents Chemother. 44:2623-2629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jenks, P. J., A. Labigne, and R. L. Ferrero. 1999. Exposure to metronidazole in vivo readily induces resistance in Helicobacter pylori and reduces the efficacy of eradication therapy in mice. Antimicrob. Agents Chemother. 43:777-781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Karita, M., M. K. R. Tummuru, H. P. Wirth, and M. J. Blaser. 1996. Effect of growth phase and acid shock on Helicobacter pylori cagA expression. Infect. Immun. 64:4501-4507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kuipers, E. J., J. G. Kusters, and W. A. Deboer. 1997. Current therapies of Helicobacter pylori infection. Bailliere's Clin. Infect. Dis. 4:395-412. [Google Scholar]
- 23.Kuipers, E. J., A. M. Uyterlinde, A. S. Pena, H. Hazenberg, E. Bloemena, J. Lindeman, E. C. Klinkenbergknol, and S. Meuwissen. 1995. Increase of Helicobacter pylori-associated corpus gastritis during acid suppressive therapy: implications for long-term safety. Am. J. Gastroenterol. 90:1401-1406. [PubMed] [Google Scholar]
- 24.Larsson, H., E. Carlsson, B. Ryberg, J. Fryklund, and B. Wallmark. 1988. Rat parietal cell function after prolonged inhibition of gastric acid secretion. Am. J. Physiol. 254:G33-G39. [DOI] [PubMed] [Google Scholar]
- 25.Lee, A., M. F. Dixon, S. J. Danon, E. Kuipers, F. Megraud, H. Larsson, and B. Mellegard. 1995. Local acid production and Helicobacter pylori: a unifying hypothesis of gastroduodenal disease. Eur. J. Gastroenterol. Hepatol. 7:461-465. [PubMed] [Google Scholar]
- 26.Lee, A., J. O'Rourke, M. C. De Ungria, B. Robertson, G. Daskalopoulos, and M. F. Dixon. 1997. A standardized mouse model of Helicobacter pylori infection—introducing the Sydney strain. Gastroenterology 112:1386-1397. [DOI] [PubMed] [Google Scholar]
- 27.Lind, T., F. Megraud, P. Unge, E. Bayerdorffer, C. O'Morain, R. Spiller, S. V. van Zanten, K. D. Bardhan, M. Hellblom, M. Wrangstadh, L. Zeijlon, and C. Cederberg. 1999. The MACH2 study: role of omeprazole in eradication of Helicobacter pylori with 1-week triple therapies. Gastroenterology 116:248-253. [DOI] [PubMed] [Google Scholar]
- 28.Lind, T., S. Veldhuyzen van Zanten, P. Unge, R. Spiller, E. Bayerdorffer, C. O'Morain, K. Dev Bardhan, et al. 1996. Eradication of Helicobacter pylori using one-week triple therapies combining omeprazole with two antimicrobials: the MACH I study. Helicobacter 1:138-144. [DOI] [PubMed] [Google Scholar]
- 29.Logan, R. P. H., M. M. Walker, J. J. Misiewicz, P. A. Gummett, Q. N. Karim, and J. H. Baron. 1995. Changes in the intragastric distribution of Helicobacter pylori during treatment with omeprazole. Gut 36:12-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Malfertheiner, P., F. Megraud, C. O'Morain, A. P. S. Hungin, R. Jones, A. Axon, D. Y. Graham, G. Tytgat, M. Asaka, F. Bazzoli, B. Birkner, J. Bures, A. Burette, P. Bytzer, L. Castro, A. Culhane, W. de Boer, J. De Korwin, E. De Koster, N. de Wit, M. Deltenre, J. Dent, F. Di Mario, B. Dragosics, M. Farkkila, et al. 2002. Current concepts in the management of Helicobacter pylori infection: the Maastricht 2-2000 consensus report. Aliment. Pharmacol. Ther. 16:167-180. [DOI] [PubMed] [Google Scholar]
- 31.Marshall, B. J., and J. R. Warren. 1984. Unidentified curved bacilli in the stomach of patients with gastritis and peptic ulceration. Lancet i:1311-1315. [DOI] [PubMed] [Google Scholar]
- 32.McGowan, C. C., A. Necheva, S. A. Thompson, T. L. Cover, and M. J. Blaser. 1998. Acid-induced expression of an LPS-associated gene in Helicobacter pylori. Mol. Microbiol. 30:19-31. [DOI] [PubMed] [Google Scholar]
- 33.Megraud, F. 1995. Rationale for the choice of antibiotics for the eradication of Helicobacter pylori. Eur. J. Gastroenterol. Hepatol. 7(Suppl. 1):S49-S54. [PubMed] [Google Scholar]
- 34.Mirshahi, F., G. Fowler, A. Patel, and G. Shaw. 1998. Omeprazole may exert both a bacteriostatic and a bacteriocidal effect on the growth of Helicobacter pylori (NCTC 11637) in vitro by inhibiting bacterial urease activity. J. Clin. Pathol. 51:220-224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Peterson, W. L., D. Y. Graham, B. Marshall, M. J. Blaser, R. M. Genta, P. D. Klein, C. W. Stratton, J. Drnec, P. Prokocimer, and N. Siepman. 1993. Clarithromycin as monotherapy for eradication of Helicobacter pylori—a randomized, double-blind trial. Am. J. Gastroenterol. 88:1860-1864. [PubMed] [Google Scholar]
- 36.Treiber, G. 1996. The influence of drug dosage on Helicobacter pylori eradication: a cost-effectiveness analysis. Am. J. Gastroenterol. 91:246-257. [PubMed] [Google Scholar]
- 37.Unge, P. 1997. What other regimens are under investigation to treat Helicobacter pylori infection. Gastroenterology 113(Suppl. 6S):S131-S148. [DOI] [PubMed] [Google Scholar]
- 38.Veldhuyzen van Zanten, S. J. O., M. F. Dixon, and A. Lee. 1999. The gastric transitional zones: neglected links between gastroduodenal pathology and Helicobacter ecology. Gastroenterology 116:1217-1229. [DOI] [PubMed] [Google Scholar]
- 39.Veldhuyzen van Zanten, S. J. V., P. T., Pollak, H. Kapoor, and P. K. Yeung. 1996. Effect of omeprazole on movement of intravenously administered metronidazole into gastric juice and its significance in treatment of Helicobacter pylori. Dig. Dis. Sci. 41:1845-1852. [DOI] [PubMed] [Google Scholar]
- 40.Velduyzen van Zanten, S. J. O., and P. M. Sherman. 1994. Helicobacter pylori infection as a cause of gastritis, duodenal ulcer, gastric cancer and nonulcer dyspepsia: a systematic overview. Can. Med. Assoc. J. 150:177-185. [PMC free article] [PubMed] [Google Scholar]
- 41.Xia, H. X., N. J. Talley, C. T. Keane, and C. A. O'Morain. 1997. Recurrence of Helicobacter pylori infection after successful eradication: nature and possible causes. Dig. Dis. Sci. 42:1821-1834. [DOI] [PubMed] [Google Scholar]