Abstract
Hair follicle morphogenesis and cycling were examined in transgenic mice that overexpress the bone morphogenetic protein (BMP) inhibitor Noggin under the control of the neuron-specific enolase promoter. The Noggin transgene was misexpressed in the proximal portion of the hair follicle, primarily the matrix cells, apart from the usual expression in neurons. Transgene expression appeared only after induction of both the primary (tylotrich) and secondary (nontylotrich) pelage hair follicles had already occurred, thus allowing examination of the role of BMP signaling in follicles that had been induced normally in the presence of BMPs. The overexpression of Noggin in these animals resulted in a dramatic loss of hair postnatally. There was an apparently normal, but shortened period of postnatal hair follicle morphogenesis, followed by premature initiation of hair follicle cycling via entry into the first catagen transformation. This resulted in a complete loss of hair shafts from the nontylotrich hair follicles in these mice while the tylotrich hair follicles were normal. The onset of anagen of the first postnatal hair follicle cycle was also accelerated in the transgenic mice. Our results show that BMP signaling is specifically required for proper proliferation and differentiation during late morphogenesis of nontylotrich hair follicles and that inhibition of this signaling pathway may be one of the triggers for the onset of catagen when the follicles are in anagen and the onset of anagen when the follicles are in telogen. Ectopic sebocyte differentiation was another hallmark of the phenotype of these transgenic mice suggesting that BMP signaling may be an important determinant of lineage selection by common progenitor cells in the skin. BMPs likely promote a hair follicle-type differentiation pathway of keratinocytes while suppressing the sebaceous differentiation pathway of skin epithelium.
Hair follicle development involves a highly coordinated series of bidirectional epithelial-mesenchymal interactions. Because hair follicle morphogenesis is significantly affected by the sensory and autonomic innervation as well as the pigment-producing melanocytes, this miniorgan has been viewed as an epithelial-mesenchymal-neuroectodermal unit.1,2 Hair follicle induction occurs with the appearance of a thickening of the embryonic ectoderm called the ectodermal placode as a result of an initial mesodermal signal. The placode signals condensation of the underlying mesoderm to form the future dermal papilla.3,4 A second signal from the condensed mesoderm then induces proliferation of the overlying ectodermal placode allowing it to grow downwards eventually surrounding the mesenchymal condensation. The latter forms the dermal papilla, the permanent mesenchymalcomponent of the hair follicle. The basic structure of the mature hair follicle then forms by systematic differentiation of the proliferating keratinocytes, producing the outer root sheath (ORS), which is contiguous with the basal layer of the epidermis, three concentric cylinders constituting the inner root sheath (IRS) and the medulla, cortex, and cuticle of the hair shaft at the center. The sebaceous gland soon develops as an appendage to the upper part of the hair follicle and secretes lipids (sebum) at the hair canal near the skin surface.
Adult mouse fur consists of different hair types characterized by their specific morphology and their induction times during embryogenesis.5–7 The first hair follicles induced at approximately embryonic day 14.5 in the mouse are called the primary or tylotrich (guard) hair follicles, characterized by a large hair bulb, a long straight hair, and two sebaceous glands. This type constitutes 5 to 10% of all adult hairs. The thinner and shorter hairs having one sebaceous gland are known as the secondary or nontylotrich (awl and zigzag) hair follicles and are induced between embryonic day 16.6 to postnatal day 0.5.
There is a continuous cycle of keratinocyte proliferation, differentiation, cell death, and quiescence that continues all throughout the life of the hair follicle.3,4 After growth initiation of a telogen (quiescent) hair follicle, there is continuous keratinocyte proliferation and epithelial down-growth into the subcutis. This period of active hair growth is known as anagen and is the time when the matrix cells proliferate and differentiate into various components of the IRS and the hair shaft. The proliferative capacity of the matrix cells in anagen is limited and is followed by a rapid phase of hair follicle involution as a result of apoptosis in the lower third of the follicle, a phase known as catagen.8–10 During this hair follicle involution phase, the dermal papilla maintains contact with the basement membrane that separates the hair follicle epithelium from the mesenchyme. This drags the dermal papilla up to the dermis at the base of the follicle, which finally remains in a relative period of quiescence known as the telogen. A small protrusion of the permanent epithelial portion of the follicle known as the bulge is the stem cell compartment of the hair follicle, the sebaceous gland, and the interfollicular epidermis.11–13 Cells emerge from this compartment to regenerate the lower two-thirds of the follicle at the start of each new hair cycle and signals from the closely positioned dermal papilla (and possibly also some fibroblasts of the connective tissue sheath) are required for this rejuvenation.14,15
Several signaling pathways have been implicated in the induction and the proceeding of various stages of the hair follicle cycle16–18 including tumor necrosis factor, fibroblast growth factor, Wnt, and sonic hedgehog (Shh) signaling pathways.19–27 Another major group of hair growth-modulating factors belongs to the transforming growth factor (TGF)-β superfamily including TGF-β1 to TGF-β3, various bone morphogenetic proteins (BMPs), and growth differentiation factors, such as inhibins and activins.28–34 Particularly important are the BMPs and various secreted inhibitors of the BMPs including Noggin, Chordin, Follistatin, and the DAN family. Bmp2 and Bmp4, members of the Drosophila decapentaplegic (dpp) family of the BMPs, are expressed in the hair shaft precursors, ie, the precortical matrix, while Bmp4 is also expressed in the dermal papilla.33,35 Bmp7 is expressed in the IRS, ORS, and the dermal papilla.36 Mice deficient in Noggin, an inhibitor of BMP2, BMP4, and BMP7, have reduced hair follicle induction,30 particularly of the nontylotrich follicles,32 demonstrating that inhibition of BMP signaling is crucial for hair follicle morphogenesis. BMPs also regulate hair follicle cycling. Ectopic expression of Bmp4 in the ORS of hair and whisker follicles inhibits hair matrix proliferation and accelerates the differentiation program in the ORS37 whereas application of the inhibitor Noggin to telogen skin accelerates anagen development via up-regulation of Shh transcripts.31
Noggin inhibits the function of BMP2, BMP4, and BMP7, 38 all of which are expressed in the hair follicle. We generated transgenic mice overexpressing Noggin under control of the neuron-specific enolase (NSE) promoter with the intention of examining the role of BMP signaling in the nervous system. However the Noggin transgene is also misexpressed in these animals in the hair matrix cells and possibly neuroendocrine cells including melanocytes of the hair follicles, apart from the usual expression in neurons. A dramatic loss of hair was observed postnatally at approximately day 10 in these transgenic mice. This hair loss is associated with premature onset of catagen, specifically of the nontylotrich hair follicles, after normal but abrogated hair follicle morphogenesis. This results in a complete loss of hair shafts from the nontylotrich follicles in these mice while the tylotrich hair follicles remain normal. After the premature catagen development there is an accelerated anagen initiation, preceded by a shortened telogen phase. Our results show that BMP signaling is specifically required for proper proliferation and differentiation during late hair follicle morphogenesis and that inhibition of this signaling pathway may be one of the triggers for the onset of catagen when the follicles are in anagen and the onset of anagen when the follicles are in telogen.
Materials and Methods
Generation of NSE-Noggin Transgenic Mice
E19 whole brain RNA was used to amplify the Noggin cDNA by polymerase chain reaction. This was then subcloned into a bicistronic vector containing green fluorescent protein (EGFP) behind an internal ribosomal entry site (IRES) (pIRES-EGFP; Clontech, Palo Alto, CA) to make the pNoggin-IRES-EGFP plasmid. After verifying the Noggin sequence, the Noggin-IRES-EGFP fragment was further subcloned downstream of the 1.8-kb rat NSE promoter39 and upstream of an SV40 polyadenylation signal, at the SalI site of the pNSE-Ex4 plasmid. The NSE-promoter construct also contained the initial noncoding exon and the first intron of the rat Nse gene, making the NSE-Noggin-IRES-EGFP construct. The noncoding exon and the intron increase transgene expression (P. Danielson, personal communication). This plasmid was further linearized by the SalI enzyme and the NSE-Noggin-IRES-EGFP fragment was injected into the male pronucleus of FVB one-cell stage embryos in the transgenic facility of the Albert Einstein College of Medicine, Bronx, NY. Two transgenic founder mice had similar hair phenotype. The transgenic mice were identified by Southern blot analysis with a Noggin-specific probe and were further screened by polymerase chain reaction with transgene-specific primers against the IRES and EGFP sections. The primers used were: 5′IRES, 5′ AGCCCGGTGTGCGTTTG 3′ and 3′GFP 598, 5′ TGAGCAAGGGCGAGGAG 3′.
Protein Blot Analysis
Transgene expression was verified by protein blot analysis of lysates prepared from the whole brain or back skin of adult transgenic and wild-type mice for Noggin using a rat monoclonal antibody, RP57-16 against Noggin (a gift from Regeneron Pharmaceuticals, Tarrytown, NY). The lysates were Dounce-homogenized by a polytron homogenizer at 4°C in lysis buffer [150 mmol/L NaCl, 50 mmol/L N-(2-hydroxyethyl) piperazine-N′-(2-ethane sulfonic acid) (HEPES) pH 7.2, 1 mmol/L ethylenediaminetetraacetic acid, 1 mmol/L ethylene glycol-bis(α-aminoethyl ether)-N,N,N′,N′-tetraacetic acid, 1 mmol/L dithiothreitol, 0.1% polyoxyethylenesorbitan monolaurate (Tween-20), 0.1 mmol/L phenylmethyl sulfonyl fluoride, 2.5 μg/ml leupeptin, and 0.1 mmol/L sodium orthovanadate]. Lysates were centrifuged at 10,000 × g for 10 minutes and protein concentrations were determined using a modified Bradford assay protocol. A polyclonal antibody to anti-guanine nucleotide dissociation inhibitor, a gift from Dr. Perry Bickel, Washington University, St. Louis, MO, was used to verify equal loading.
Quantitative Histomorphometry
Skin from a defined region of the mouse back was taken and fixed overnight in 4% paraformaldehyde in phosphate buffer (0.1 mol/L, pH 7.2). After a graded series of dehydration in alcohol and clearing in xylene, the back skin was embedded in paraffin to obtain longitudinal hair follicle sections. Five-μm sections were then cut in a microtome and routinely stained with hematoxylin and eosin (H&E). Hair follicle morphogenesis was evaluated by light microscopy. Briefly, in each of the postnatal days evaluated, hair follicles in each classifiable stage of morphogenesis were defined on the basis of accepted morphological criteria,40 counted, and the percentage of hair follicles in each stage was calculated. The percentages were then multiplied by a different factor for each hair cycle stage (stage 1, 1×; stage 2, 2×; and so forth) and the results were added for each individual animal, giving the hair follicle morphogenesis score. Three animals of each genotype were evaluated at each time point. The mean and SEM were calculated for each genotype and statistically compared using the Student’s t-test.
Analysis of Apoptosis
Apoptotic cells were detected by the terminal deoxynucleotidyl transferase (TdT)-mediated nick-end nucleotide labeling (TUNEL) technique using the Apoptag Red kit (Serological Corp., Norcross, GA).
Immunohistochemistry
Immunohistochemistry was performed on either 5-μm paraffin sections after antigen retrieval with DAKO antigen retrieval solution (DAKO, Carpinteria, CA), pH 6.0, or cryosections of freshly frozen back skin tissue cut perpendicular to the surface so that the hair follicles are in longitudinal orientation. The various primary antibodies used were against GFP (rabbit polyclonal; Molecular Probes, Eugene, OR), Ki67 (rabbit polyclonal, Novocastra), trichohyalin (mouse monoclonal, AE15),41 keratin 14, loricrin, and filagrin (rabbit polyclonals; Babco, Richmond, CA). Appropriate secondary antibodies conjugated to Alexa Fluor 488, Alexa Fluor 568 (Molecular Probes) or Cy5 (Jackson Immunoresearch, West Grove, PA) were used for detection. Controls included omission of secondary antibody and use of irrelevant antibodies. Where indicated, confocal microscopy was done using a Bio-Rad MRC 2000 laser-scanning confocal microscope with Kr/Ar laser for excitation at 488 and 568 nm and a diode laser for excitation at 633 nm using a ×60 objective and 1.4 numerical aperture Nikon infinity-corrected optics. Immunohistochemistry analyses were done on at least two mice of each genotype.
Staining of Sebocytes with Oil Red O
Ten-μm frozen sections of skin of postnatal day 19 mice were fixed in 10% formalin, washed with distilled water followed by two changes of propylene glycol of 5 minutes duration. The sections were then stained with Oil Red O for 7 minutes, rinsed with 85% propylene glycol for 3 minutes, rinsed in distilled water, and then counterstained with hematoxylin.
Multiphoton Microscopy
Multiphoton microscopy was performed on freshly dissected back skin from 2- to 5-day-old mice or back skin of anesthetized mice to visualize GFP fluorescence from the expressed transgene. A 10 W Millenium Xs laser (Spectra Physics) was used to run a Radiance 2000 multiphoton system (Bio-Rad) that gives an output of 850 mW at 960 nm. For GFP fluorescence, 960 nm is the optimal imaging wavelength. The images were collected using Bio-Rad Lasersharp 2000 software at 50 lines/second. Images were processed using NIH Image 1.61/ppc and Adobe Photoshop.
Results
NSE-Noggin Transgenic Mice Lose Hair Shafts from Nontylotrich Hair Follicles after Initial Development of an Apparently Normal Hair Coat
We generated transgenic mice misexpressing Noggin under the control of the NSE promoter. Green fluorescence protein (EGFP) was also expressed as part of a bicistronic transcript under the same promoter to follow transgene expression. Two lines of transgenic mice with a hair follicle phenotype were identified and homozygous transgenic lines with an enhanced phenotype were generated (Figure 1). The NSE-Noggin transgenic mice are indistinguishable from their wild-type littermates in the early postnatal period. However, transgenic mice apparently lose their hair beyond postnatal day 10. The phenotype is more severe in the homozygous transgenic line suggesting that the hair follicle effects are transgene dose-dependant (Figure 2A). The transgenic mice specifically lose the zigzag hair suggesting that the effect of misexpressed Noggin is restricted to the nontylotrich hair follicles (Figure 1). In addition to the expected expression of the transgene in neurons of the brain of these mice (data not shown), there was robust misexpression of the transgene in virtually all hair follicles, ie, both tylotrich and nontylotrich hair follicles (Figure 2). This was demonstrated by multiphoton microscopy of live GFP fluorescence (Figure 2C), by immunohistochemistry detecting transgenic GFP in skin sections (Figure 2, A and B), and by Western blot analysis of Noggin protein in transgenic back skin lysates, in which homozygous animals showed even higher levels of expression than heterozygous mice (Figure 1). The NSE promoter has been shown to drive expression of transgene as early as postnatal day 16 in the brain.39 The earliest that we studied transgene expression by confocal microscopy was at postnatal day 2 when GFP fluorescence was detectable only in the proximal part of hair follicles (data not shown).
Figure 1.
Misexpression of Noggin in the skin of NSE-Noggin mice results in complete lack of zigzag hair. A: Transgene construct injected in Fvb one-cell stage embryos misexpressing Noggin and EGFP by a bicistronic cassette under the NSE promoter. B: Southern blot of genomic DNA with a Noggin-specific DNA probe identifies a 1-kb transgene band in the NSE-Noggin transgenic mice. C: Western blot analysis of whole brain lysates from 1-month-old wild-type (Wt) and homozygous (hom) transgenic mice shows that Noggin protein is overexpressed in the brain. Similar analysis of back skin lysates of 13-day-old mice shows that Noggin protein is overexpressed in the skin of the transgenic mice. Note that homozygotes (hom) have higher levels of expression than heterozygotes (het). Guanine nucleotide dissociation inhibitor is used as a loading control. D: Adult NSE-Noggin transgenic mice have significantly reduced pelage hair, with the phenotype being more severe in the homozygous transgenic compared to the heterozygous. E: NSE-Noggin transgenic mice completely lack the zigzag hair, marked by the arrows in the wild-type hair spread from a defined area in the dorsal neck region.
Figure 2.
Misexpression of the transgene in NSE-Noggin transgenic mice. A: Immunohistochemistry with an antibody against GFP shows that the transgene is misexpressed in the precortical matrix of hair follicles of the back skin of 9-day-old NSE-Noggin mice (B) compared to the wild type (A). C: Multiphoton image of freshly dissected back skin from a 5-day-old transgenic mouse reveals live GFP fluorescence in the proximal portion of the hair follicles.
Inhibition of BMP Signaling by Noggin Misexpression in the Hair Follicle Abrogates Morphogenesis and Accelerates Postnatal Cycling of Nontylotrich Hair Follicles
To characterize the hair loss in transgenic mice, histology and histomorphometry were performed on sections of mouse back skin. At birth (Figure 3A), ie, postnatal day 1, hair follicles, comprising both tylotrich and nontylotrich follicles, were in stages 1 to 5 of morphogenesis, according to the classification scheme previously published.4 Hair follicles in stage 5 of morphogenesis showed a well-differentiated IRS, a developing bulge, and sebaceous glands. The dermal papilla was completely enclosed by keratinocytes and was located at the border between the dermal and the hypodermal mesenchyme. There was no statistical difference in terms of the hair follicle morphogenesis score that was evaluated at postnatal day 1 in the transgenic versus the wild-type mice. This indicates that early hair follicle morphogenesis is unaltered in the NSE-Noggin transgenic mice, described herein. Further, the number of hair follicles, evaluated per 100-μm length of epidermis, did not differ significantly in the transgenic compared to the wild type at this time. Hair follicles in stage 8 of morphogenesis were first noticed on postnatal day 5 in both transgenic and wild-type skin and no morphological abnormalities were detected at this time (Figure 4, a and b). On postnatal day 7 multiple hair shafts were observed that penetrated the epidermis in wild-type and transgenic animals but no significant difference was noticed with respect to the hair follicle morphogenesis score (data not shown). Finally, at postnatal day 9, all hair follicles, ie, tylotrich and nontylotrich hair follicles, had reached stage 8 of morphogenesis in both wild-type and transgenic mice (Figure 3B). At postnatal day 10, ie, after hair follicles have more or less completed their morphogenesis, the first hair follicle abnormalities in transgenic animals were noticed. These included alterations in the structure of the ORS (Figure 3B). There was massive keratinocyte apoptosis by postnatal day 11, occurring within the ORS of most nontylotrich hair follicles. These follicles showed morphological criteria of catagen instead of being in morphogenesis stage 8 (Figure 4, c and d). The changes mentioned above only affected the nontylotrich hair follicles, whereas the tylotrich hair follicles remained in morphogenesis stage 8 in all transgenic animals. By postnatal day 15, all of the nontylotrich hair follicles were in the telogen stage while the tylotrich hair follicles were at the end of their morphogenesis (Figure 4, e and f). There was premature onset of the first postnatal anagen in the nontylotrich hair follicles in the transgenic skin at postnatal day 18 while the tylotrich hair follicles underwent catagen, as did all of the follicles of the wild type on postnatal days 18 and 19 (Figure 4, g and h). Because there is premature onset of anagen without a significant amount of time spent in the resting telogen phase, the club hair is not normally generated in the nontylotrich hair follicles. Finally at postnatal day 29 most hair follicles (both tylotrich and nontylotrich) were in the telogen stage in the wild-type animals, and only a few very early anagen stage follicles were observed. In contrast, nontylotrich hair follicles had reached anagen VI already in transgenic animals (Figure 4, i and j). At this time period, the remaining coat in the transgenic mice consisted of very sparse, thin, straight hairs. The appearance was consistent with a loss of the nontylotrich hair follicles and a preservation of the tylotrich hair follicles. The quantitation of the number of hair follicles in different stages of hair follicle morphogenesis on postnatal days 7, 17, and 21 underlines the tremendous differences in hair follicle morphogenesis and the first postnatal hair cycle between transgenic and wild-type mice (Figure 5). At postnatal day 7, no differences between wild-type and transgenic mice were detectable. At postnatal day 17, there was a premature catagen development in transgenic mice, in which hair follicles had already reached the telogen stage. At postnatal day 21, these hair follicles in transgenic mice already showed anagen development, whereas hair follicles in wild-type animals were still in telogen.
Figure 3.
Hair follicle morphogenesis is normal until postnatal day 10. A: Hair follicles are in similar stages of morphogenesis at birth (postnatal day 1). H&E staining of back skin reveals all follicles in morphogenesis stages 1 to 5 of anagen and there is no difference in hair follicle morphogenesis score in the transgenic and control mice. B: Hair follicles are in morphogenesis stage 8 in both genotypes, and hair shafts penetrate the epidermis (a, b). The first abnormalities appear in the transgenic skin at postnatal day 10 (c, d). There are irregularities in the follicular architecture (arrows).
Figure 4.
Analysis of postnatal hair follicle development and cycling shows that inhibition of BMP signaling by Noggin misexpression in hair follicles results in premature catagen onset and early onset of the first anagen phase. At postnatal day 5, both the transgenic hair follicles (b) and wild-type hair follicles (a) are in advanced stages of morphogenesis. By 11 days, when all wild-type follicles (c) are in morphogenesis stage 8, most of the transgenic follicles are regressing. By 15 days, when the wild-type follicles (e) are near the end of morphogenesis, the transgenic follicles have almost completely regressed. Only tylotrich follicles are found in morphogenesis stage 8 (arrowhead in f). At postnatal day 18, all wild-type follicles have started their catagen phase (g), while the transgenic nontylotrich follicles have started the first anagen (arrowheads in h). Simultaneously, tylotrich hair follicles of transgenic mice begin catagen development. At postnatal day 29, the wild-type follicles reveal telogen hair follicles with normal club hair formation (arrows, i), while nontylotrich hair follicles of transgenic mice are in late stages of anagen development (j).
Figure 5.
Percentages of hair follicles in different stages of morphogenesis and the first hair cycle. M5, morphogenesis stage 5; M6, morphogenesis stage 6; M7, morphogenesis stage 7; M8, morphogenesis stage 8; C1, early catagen (stage II to III); C2, mid-catagen (stage IV to VI); C3, late catagen (stage VII to VIII); T, telogen; A1, early anagen (stage I to II); A2, mid-anagen (stage III to V); A3/C, late anagen (stage VI)/very early catagen (stage I). Shown are evaluations at P7, P17, and P21. Three mice of each genotype were evaluated at each time point. At P7, no differences between wild-type and transgenic mice are detectable. At P17, there is a premature catagen development in transgenic mice, in which hair follicles have already reached the telogen stage. At P21, these hair follicles in transgenic mice already show anagen development, whereas hair follicles in wild-type animals are still in telogen.
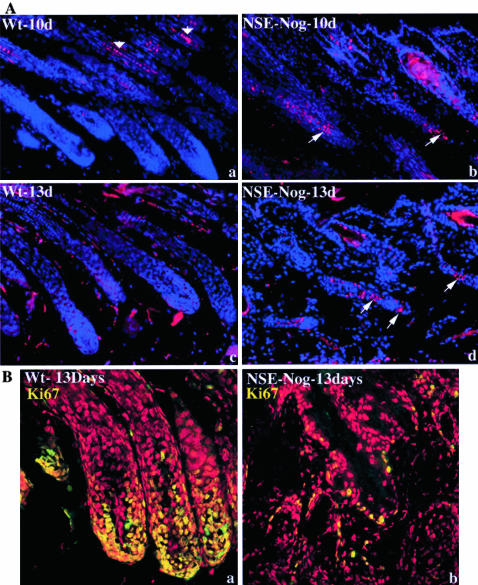
Noggin Misexpression in Transgenic Mice Induces Premature Onset of Catagen in Nontylotrich Hair Follicles
To support our morphological findings, which are suggesting premature termination of nontylotrich hair follicle morphogenesis, we performed TUNEL staining on skin sections, aiming at detecting apoptotic keratinocytes during catagen development.9 The earliest onset of TUNEL-positive apoptotic cells in the proximal part of nontylotrich hair follicles occurred on postnatal day 10 (Figure 6, a and b). The occurrence of apoptosis within the ORS was still detectable on postnatal day 13 (Figure 6, c and d), verifying that there was premature onset of catagen in nontylotrich hair follicles of the transgenic mice. The fact that the nontylotrich hair follicles of transgenic mice indeed were no longer in the proliferating anagen phase of the hair cycle was also supported by the morphology of the proximal hair follicle that lacked a matrix region with the typical abundance of Ki67-immunoreactive keratinocytes around the dermal papilla (Figure 6B, a and b).
Figure 6.
A: Premature catagen of NSE-Noggin hair follicles. TUNEL labeling of mouse back skin sections shows that apoptosis occurs in the proximal portion of the hair follicles in transgenic mice, starting at postnatal day 10. Note that there is no apoptosis in the anagen hair follicles of the wild-type animals (a). B: Reduced proliferation of NSE-Noggin hair follicles by postnatal day 13. Ki67-immunoreactive proliferating cells are abundant in the hair bulb of anagen hair follicles of the wild-type (a) while there are scattered proliferating cells in the transgenic (b) hair follicles.
Noggin Misexpression in Transgenic Mice Leads to Ectopic Differentiation of Sebocytes
An interesting feature of the transgenic hair follicles was the differentiation of ORS keratinocytes into sebocytes, which was observed in the proximal part of the hair follicle, ie, far away from the isthmus region, which is the usual location of sebaceous gland development (Figure 7C). To better delineate the extent of ectopic sebocyte differentiation, postnatal day 19 back skin was stained with Oil Red O to specifically stain the fat-laden sebocytes and there was significant increase of sebocyte differentiation in the NSE-Noggin transgenic animals compared to the wild type (Figure 7, A and B).
Figure 7.
Increased and ectopic sebocyte differentiation in NSE-Noggin skin. A and B: Increased sebocyte staining by Oil Red O in P19 transgenic skin (B) compared to the wild-type littermate (A). C: Abnormal differentiation of proximal ORS keratinocytes into sebocytes (arrowhead). H&E stain. Original magnifications, ×20.
Inhibition of BMP Signaling in the Matrix Region of Hair Follicles Resulted in a Block in Differentiation of the Nontylotrich Hair Follicles
To further define the hair follicle abnormalities in transgenic mice we looked into various markers of proliferation and differentiation in the hair follicle and the interfollicular epidermis. Keratin 14 is one of the markers of basal keratinocytes in the ORS and the basal layer of the interfollicular epidermis. At postnatal day 10, the thickness of the basal ORS layer that expressed keratin 14 was irregular in transgenic mice, compared to the very regular thickness in wild-type animals. In transgenic mice, keratin 14 immunoreactivity extended further down toward the hair bulb. By contrast, trichohyalin, which marks granules in the IRS and the medulla of the hair follicle and hence is a marker for differentiation of the hair matrix,42 was absent in all of the nontylotrich hair follicles in the transgenic animals at postnatal day 13 (Figure 8, a and b). Keratin 14 expression was normal in the interfollicular epidermis, as was expression of loricrin and filagrin, markers of terminal epidermal keratinocyte differentiation (Figure 8; c to f).
Figure 8.
Misexpression of Noggin in the hair follicles results in impaired differentiation of hair. Double immunostaining of 13-day-old mouse back skin shows that there is absence of hair differentiation marker trichohyalin (AE15) in the catagen transgenic hair follicles (arrows in b) while there is normal AE15 immunoreactivity in the anagen wild-type hair follicle (arrows in a). In contrast, there is more K14 immunoreactivity in the transgenic (arrowheads in b) compared to the wild-type (a). There is no change of other differentiation markers of the skin like loricrin and filagrin in the transgenic (d, f) compared to the wild-type (c, e). Color coding denotes secondary antibodies.
Discussion
We have generated a mouse model useful for the study of the role of BMP signaling in hair follicle morphogenesis and postnatal hair follicle cycling. BMP signaling is inhibited in these animals in the matrix region of the postnatal hair follicle by misexpression of Noggin. Previous studies have shown that BMP2, BMP4, and BMP receptor 1A (BMPR1A) are expressed in the keratinocytes of the hair follicle bulb as well as the mesenchymal dermal papilla and that the inhibitor Noggin is expressed in the dermal papilla and the connective tissue sheath of the hair follicle.30,33,43 During development BMP signaling is a negative regulator of induction of the nontylotrich hair follicles,32 and the inhibition of BMP signaling is required for embryonic hair follicle and feather bud induction.30 However transgene expression in the NSE-Noggin transgenic mice appears only after the induction of both the primary and secondary follicles have already occurred, so there is no effect on the induction of these follicles in this transgenic model. This allowed examination of the role of BMP signaling in postnatal hair follicle morphogenesis and cycling.
We found that BMP signaling is essential for the continuation of the later stages of hair follicle morphogenesis. Inhibition of BMP signaling during the later part of hair follicle morphogenesis (after the follicles have reached morphogenesis stage 8), resulted in the premature onset of catagen, ie, involution of nontylotrich hair follicles without proper differentiation of a hair shaft. A similar phenotype was observed in mice that misexpress Noggin in hair matrix cells under control of the Msx2 promoter.33 It was suggested in that study that BMP signaling is required for differentiation of proliferating keratinocytes to the hair matrix, but a detailed hair follicle cycle analysis was not performed in that mouse model. We also found that BMP signaling may be required for the proper differentiation of the hair matrix cells postnatally but that it is specific to the nontylotrich hair follicles. It is noteworthy that during induction of hair follicles at embryonic stages, inhibition of BMP signaling is required for induction of only the secondary nontylotrich hair follicles but not the primary tylotrich follicles.32 Signaling pathways other than the BMPs have been implicated in the induction of primary hair follicles. For example, the tumor necrosis factor receptor homolog Edar is required for the induction of primary hair follicles44 and β-catenin represents an important downstream component of Edar signaling in this process.45 Consistent with these observations ectopic expression of Noggin in the skin by the keratin 14 (K14) promoter increases the number and the size of nontylotrich hair follicles.46 Similarly, preliminary observations made on mice that overexpress noggin under the control of a keratin 5 promoter, 47 reportedly show acceleration of hair follicle morphogenesis and formation of enlarged hair follicles with increased proliferative activity in the hair bulb. On the contrary, there is no change in hair follicle number in the NSE-Noggin mice. Moreover, Noggin misexpression in the postnatal hair matrix region terminates prematurely the proliferative anagen phase and heralds the apoptotic catagen phase of nontylotrich hair follicles. The differences between the findings in K14- or K5-driven Noggin mice and NSE-driven Noggin misexpressing mice may reflect differences in the timing of expression of transgenic noggin. Both K14 and K5 are expressed early in embryogenesis and BMP signaling presumably is inhibited during the time of hair follicle induction. However in our NSE-noggin mice transgenic misexpression of Noggin occurs only after the normal induction of all hair follicles. We propose that whereas inhibition of BMP signaling is needed for induction of nontylotrich hair follicles, hair follicle differentiation and continuation of proper anagen in the postnatal period are dependent on BMP signaling. Our observations also suggest the cycling of primary tylotrich follicles in the postnatal period is regulated by pathways other than BMP signaling. In support of our observations, ectopic expression of Bmp4 in the hair follicle is known to inhibit hair matrix proliferation with an accelerated differentiation program in the ORS37 and conditional disruption of BMP receptor IA gene disrupts differentiation of progenitor cells of the IRS and the hair shaft.48 The specific temporal and spatial expression pattern of Noggin in the NSE-Noggin mice allows examination of the role of BMP signaling in the adult independent of possible developmental abnormalities. The predominant observation in our mouse model of Noggin misexpression in postnatal nontylotrich hair follicles is the premature catagen initiation. The molecular mechanism of catagen initiation is poorly understood. TGF-β1 has been implicated in catagen initiation,29 and the expression of the TGF-β isoforms TGF-β1 and TGF-β3 and their type I and type II receptors are maximal during anagen-catagen transition in the murine skin.49,50 The hairless (hr) gene product, which is now known to be a novel nuclear receptor co-repressor51 regulates hair follicle catagen transformation.52 The various neurotrophins, including nerve growth factor (NGF), neurotrophin-3 (NT3), brain-derived neurotrophic factor (BDNF), and neurotrophin-4 (NT4) have also been shown to regulate the catagen phase of murine hair cycle probably through activation of the p75 neurotrophin receptor.53–55 Inhibition of BMP signaling could regulate catagen initiation indirectly through any of the above-mentioned signaling pathways.
Subsequent to the premature catagen induction, NSE-Noggin transgenic mice exhibit a truncated resting phase and a premature entry into the first postnatal anagen phase. The loss of hair shafts from nontylotrich hair follicles is likely because of defective anchoring of club hairs, which has been shown to occur if telogen is shortened, eg, in mice that lack the lysosomal cysteine protease cathepsin L.56
It is possible that the improper formation of the hair shaft is simply the result of an incomplete hair follicle morphogenesis. Conversely, the induction of premature apoptosis in our mouse model may also be secondary to the block in differentiation of hair shaft of the nontylotrich hair follicles. Various signal transduction pathways have been implicated in the differentiation of hair follicle.17,57,58 Hair shaft differentiation is particularly dependent on Wnt signaling.19,45,59 It is noteworthy that whereas inhibition of BMP signaling by Noggin cooperates with Wnt signaling to induce hair follicles,60 activation of BMP signaling through BMPR IA is essential for differentiation of the hair shaft together with the Wnt signaling.48 Recently BMP signaling through the BMPRIA receptor has been found to be important for the differentiation of the IRS and hence necessary for hair follicle cycling and hair shaft differentiation.61 BMP signaling through BMPRIA has been proposed to prime hair shaft progenitor cells to respond to Wnt signaling. However it is also possible that these two signaling pathways cooperate directly at the level of transcription factors in this process. Whatever the case, misexpression of Noggin in the appropriate temporal and spatial setting in the NSE-Noggin mice interrupts this crosstalk by inhibiting BMP signaling and hence blocking differentiation of specifically nontylotrich follicles. The deregulated catagen may be a direct consequence of this process. The initiation of resting hair follicle down-growth during the onset of first postnatal anagen may be a recapitulation of the processes of embryonic hair follicle induction in which inhibition of BMP signaling by Noggin is essential. Hence in our mouse model as well as other similar experiments of misexpression of Noggin in telogen skin,31 there is premature onset of anagen.
Various studies have implicated the role of Wnt signaling in sebocyte differentiation in the skin.62,63 One of the downstream targets of Wnt signaling, c-myc, promotes the differentiation of epidermal stem cells into sebaceous glands.64 In light of these observations, it is interesting to note that inhibition of BMP signaling in the NSE-Noggin transgenic mice increased the differentiation of sebocytes. It was surprising to find sebocytes in the ectopic location such as the ORS of the proximal part of the hair follicle. Hence it is likely that inhibition of BMP signaling in the proximal part of the hair follicle by misexpressed Noggin diverts the normal lineage differentiation of common progenitor cells to the sebocyte lineage rather than hair follicle-type differentiation of hair shaft. Signals that regulate sebocyte development are still poorly understood,65,66 but this effect is potentially mediated by the Hedgehog (Shh) pathway because BMP suppression by noggin leads to its activation,31 and Shh signaling increases both size and number of sebaceous glands, including formation of ectopic sebocytes.67
In summary we have used misexpression of Noggin in hair follicles to inhibit BMP signaling during postnatal hair follicle cycling and shown that this signaling pathway specifically regulates cycling of the nontylotrich hair follicles. It is required for the proper differentiation of the proliferating keratinocytes to hair shaft. Inhibition of BMP signaling accelerates the onset of catagen as well as the following anagen. The striking phenotypic consequences of noggin overexpression observed in the current study encourage one to investigate more systematically the role of other molecules that belong to the growing family of BMP antagonists, such as chordin and ectodin, as integrators of BMP signaling eg, with the Shh and FGF signaling pathways in pilosebaceous biology.65
Acknowledgments
We thank Aris Economedes of Regeneron Pharmaceuticals for providing the Noggin antibody, Robert Lavker for advice, and Jeffrey Wyckoff for help with confocal microscopy.
Footnotes
Address reprint requests to John Kessler M.D., 303 East Chicago Ave., Chicago, IL 60611-3008. E-mail: jakessler@northwestern.edu.
Supported by the National Institutes of Health (grants NS 20778 and NS 34758 to J.A.K., R01CA70896 and RO1CA75503 to R.G.P.), the Deutsche Forschungsgemeinschaft (Pa 345/8-3 to R.P.), and the German Ministry for Education and Research (to R.P.).
Current address of U.G.: Jacobi Medical Center, Albert Einstein College of Medicine, Bronx, NY.
References
- Paus R, Peters EM, Eichmuller S, Botchkarev VA. Neural mechanisms of hair growth control. J Invest Dermatol Symp Proc. 1997;2:61–68. doi: 10.1038/jidsymp.1997.13. [DOI] [PubMed] [Google Scholar]
- Botchkarev VA, Eichmuller S, Johansson O, Paus R. Hair cycle-dependent plasticity of skin and hair follicle innervation in normal murine skin. J Comp Neurol. 1997;386:379–395. doi: 10.1002/(sici)1096-9861(19970929)386:3<379::aid-cne4>3.0.co;2-z. [DOI] [PubMed] [Google Scholar]
- Hardy MH. The secret life of the hair follicle. Trends Genet. 1992;8:55–61. doi: 10.1016/0168-9525(92)90350-d. [DOI] [PubMed] [Google Scholar]
- Paus R, Muller-Rover S, Van Der Veen C, Maurer M, Eichmuller S, Ling G, Hofmann U, Foitzik K, Mecklenburg L, Handjiski B. A comprehensive guide for the recognition and classification of distinct stages of hair follicle morphogenesis. J Invest Dermatol. 1999;113:523–532. doi: 10.1046/j.1523-1747.1999.00740.x. [DOI] [PubMed] [Google Scholar]
- Vielkind U, Hardy MH. Changing patterns of cell adhesion molecules during mouse pelage hair follicle development. 2. Follicle morphogenesis in the hair mutants, Tabby and downy. Acta Anat. 1996;157:183–194. doi: 10.1159/000147880. [DOI] [PubMed] [Google Scholar]
- Philpott MP, PR. Principles of hair follicle morphogenesis. Molecular Basis of Epithelial Appendage Morphogenesis. Chuong CM, editor. Landes Bioscience Publishers,; 1998:pp 75–103. [Google Scholar]
- Paus R, Cotsarelis G. The biology of hair follicles. N Engl J Med. 1999;341:491–497. doi: 10.1056/NEJM199908123410706. [DOI] [PubMed] [Google Scholar]
- Weedon D, Strutton G. Apoptosis as the mechanism of the involution of hair follicles in catagen transformation. Acta Derm Venereol. 1981;61:335–339. [PubMed] [Google Scholar]
- Lindner G, Botchkarev VA, Botchkareva NV, Ling G, van der Veen C, Paus R. Analysis of apoptosis during hair follicle regression (catagen). Am J Pathol. 1997;151:1601–1617. [PMC free article] [PubMed] [Google Scholar]
- Stenn KS, Combates NJ, Eilertsen KJ, Gordon JS, Pardinas JR, Parimoo S, Prouty SM. Hair follicle growth controls. Dermatol Clin. 1996;14:543–558. doi: 10.1016/s0733-8635(05)70383-1. [DOI] [PubMed] [Google Scholar]
- Cotsarelis G, Sun TT, Lavker RM. Label-retaining cells reside in the bulge area of pilosebaceous unit: implications for follicular stem cells, hair cycle, and skin carcinogenesis. Cell. 1990;61:1329–1337. doi: 10.1016/0092-8674(90)90696-c. [DOI] [PubMed] [Google Scholar]
- Taylor G, Lehrer MS, Jensen PJ, Sun TT, Lavker RM. Involvement of follicular stem cells in forming not only the follicle but also the epidermis. Cell. 2000;102:451–461. doi: 10.1016/s0092-8674(00)00050-7. [DOI] [PubMed] [Google Scholar]
- Oshima H, Roshat R, Kedzia C, Kobayashi K, Barrandon Y. Morphogenesis and renewal of hair follicles from adult multipotent stem cells. Cell. 2001;104:233–245. doi: 10.1016/s0092-8674(01)00208-2. [DOI] [PubMed] [Google Scholar]
- Oliver RF. The induction of hair follicle formation in the adult hooded rat by vibrissa dermal papillae. J Embryol Exp Morphol. 1970;23:219–236. [PubMed] [Google Scholar]
- Jahoda CA, Horne KA, Oliver RF. Induction of hair growth by implantation of cultured dermal papilla cells. Nature. 1984;311:560–562. doi: 10.1038/311560a0. [DOI] [PubMed] [Google Scholar]
- Stenn KS, Paus R. Controls of hair follicle cycling. Physiol Rev. 2001;81:449–494. doi: 10.1152/physrev.2001.81.1.449. [DOI] [PubMed] [Google Scholar]
- Millar SE. Molecular mechanisms regulating hair follicle development. J Invest Dermatol. 2002;118:216–225. doi: 10.1046/j.0022-202x.2001.01670.x. [DOI] [PubMed] [Google Scholar]
- Fuchs E, Merill BJ, Jamora C, DasGupta R. At the roots of a never-ending cycle. Dev Cell. 2001;1:13–25. doi: 10.1016/s1534-5807(01)00022-3. [DOI] [PubMed] [Google Scholar]
- Gat U, DasGupta R, Degenstein L, Fuchs E. De novo hair follicle morphogenesis and hair tumors in mice expressing a truncated beta-catenin in skin. Cell. 1998;95:605–614. doi: 10.1016/s0092-8674(00)81631-1. [DOI] [PubMed] [Google Scholar]
- Headon DJ, Overbeek PA. Involvement of a novel Tnf receptor homologue in hair follicle induction. Nat Genet. 1999;22:370–374. doi: 10.1038/11943. [DOI] [PubMed] [Google Scholar]
- Widelitz RB, Jiang TX, Noveen A, Chen CW, Chuong CM. FGF induces new feather buds from developing avian skin. J Invest Dermatol. 1996;107:797–803. doi: 10.1111/1523-1747.ep12330553. [DOI] [PubMed] [Google Scholar]
- St-Jacques B. Sonic hedgehog signaling is essential for hair development. Curr Biol. 1998;8:1058–1068. doi: 10.1016/s0960-9822(98)70443-9. [DOI] [PubMed] [Google Scholar]
- Chiang C, Swan RZ, Grachtchouk M, Bolinger M, Litingtung Y, Robertson EK, Cooper MK, Gaffield W, Westphal H, Beachy PA, Dlugosz AA. Essential role for sonic hedgehog during hair follicle morphogenesis. Dev Biol. 1999;205:1–9. doi: 10.1006/dbio.1998.9103. [DOI] [PubMed] [Google Scholar]
- Oro AE, Higgins KM, Hu Z, Bonifas JM, Epstein EH, Jr, Scott MP. Basal cell carcinomas in mice overexpressing sonic hedgehog. Science. 1997;276:817–821. doi: 10.1126/science.276.5313.817. [DOI] [PubMed] [Google Scholar]
- Finch PW, Rubin JS, Miki T, Ron D, Aaronson SA. Human KGF is FGF-related with properties of a paracrine effector of epithelial cell growth. Science. 1989;245:752–755. doi: 10.1126/science.2475908. [DOI] [PubMed] [Google Scholar]
- Zhou P, Byrne C, Jacobs J, Fuchs E. Lymphoid enhancer factor 1 directs hair follicle patterning and epithelial cell fate. Genes Dev. 1995;9:700–713. doi: 10.1101/gad.9.6.700. [DOI] [PubMed] [Google Scholar]
- DasGupta R, Fuchs E. Multiple roles for activated LEF/TCF transcription complexes during hair follicle development and differentiation. Development. 1999;126:4557–4568. doi: 10.1242/dev.126.20.4557. [DOI] [PubMed] [Google Scholar]
- Foitzik K, Paus R, Doetschman T, Dotto GP. The TGF-beta2 isoform is both a required and sufficient inducer of murine hair follicle morphogenesis. Dev Biol. 1999;212:278–289. doi: 10.1006/dbio.1999.9325. [DOI] [PubMed] [Google Scholar]
- Foitzik K, Lindner G, Mueller-Roever S, Maurer M, Botchkareva N, Botchkarev V, Handjiski B, Metz M, Hibino T, Soma T, Dotto GP, Paus R. Control of murine hair follicle regression (catagen) by TGF-beta1 in vivo. EMBO J. 2000;14:752–760. doi: 10.1096/fasebj.14.5.752. [DOI] [PubMed] [Google Scholar]
- Botchkarev VA, Botchkareva NV, Roth W, Nakamura M, Chen LH, Herzog W, Lindner G, McMahon JA, Peters C, Lauster R, McMahon AP, Paus R. Noggin is a mesenchymally derived stimulator of hair-follicle induction. Nat Cell Biol. 1999;1:158–164. doi: 10.1038/11078. [DOI] [PubMed] [Google Scholar]
- Botchkarev VA, Botchkareva NV, Nakamura M, Huber O, Funa K, Lauster R, Paus R, Gilchrest BA. Noggin is required for induction of the hair follicle growth phase in postnatal skin. EMBO J. 2001;15:2205–2214. doi: 10.1096/fj.01-0207com. [DOI] [PubMed] [Google Scholar]
- Botchkarev VA, Botchkareva NV, Sharov AA, Funa K, Huber O, Gilchrest BA. Modulation of BMP signaling by noggin is required for induction of the secondary (nontylotrich) hair follicles. J Invest Dermatol. 2002;118:3–10. doi: 10.1046/j.1523-1747.2002.01645.x. [DOI] [PubMed] [Google Scholar]
- Kulessa H, Turk G, Hogan BL. Inhibition of BMP signaling affects growth and differentiation in the anagen hair follicle. EMBO J. 2000;19:6664–6674. doi: 10.1093/emboj/19.24.6664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakamura M, Matzuk MM, Gerstmayer B, Bosio A, Lauster R, Miyachi Y, Werner S, Paus R. Control of pelage hair follicle development and cycling by complex interactions between follistatin and activin. FASEB J. 2003;17:497–499. doi: 10.1096/fj.02-0247fje. [DOI] [PubMed] [Google Scholar]
- Wilson N, Hynd PI, Powell BC. The role of BMP-2 and BMP-4 in follicle initiation and the murine hair cycle. Exp Dermatol. 1999;8:367–368. [PubMed] [Google Scholar]
- Takahashi H, Ikeda T. Transcripts for two members of the transforming growth factor-beta superfamily BMP-3 and BMP-7 are expressed in developing rat embryos. Dev Dyn. 1996;207:439–449. doi: 10.1002/(SICI)1097-0177(199612)207:4<439::AID-AJA8>3.0.CO;2-I. [DOI] [PubMed] [Google Scholar]
- Blessing M, Nanney LB, King LE, Jones CM, Hogan BL. Transgenic mice as a model to study the role of TGF-beta-related molecules in hair follicles. Genes Dev. 1993;7:204–215. doi: 10.1101/gad.7.2.204. [DOI] [PubMed] [Google Scholar]
- Zimmerman LB, DeJesus-Escobar JM, Harland RM. The Spemann organizer signal noggin binds and inactivates bone morphogenetic protein 4. Cell. 1996;86:599–606. doi: 10.1016/s0092-8674(00)80133-6. [DOI] [PubMed] [Google Scholar]
- Forss-Petter S, Danielson PE, Catsicas S, Battenberg E, Price J, Nerenberg M, Sutcliffe JG. Transgenic mice expressing beta-galactosidase in mature neurons under neuron-specific enolase promoter control. Neuron. 1990;5:187–197. doi: 10.1016/0896-6273(90)90308-3. [DOI] [PubMed] [Google Scholar]
- Muller-Rover S, Handjiski B, van der Veen C, Eichmuller S, Foitzik K, McKay IA, Stenn KS, Paus R. A comprehensive guide for the accurate classification of murine hair follicles in distinct hair cycle stages. J Invest Dermatol. 2001;117:3–15. doi: 10.1046/j.0022-202x.2001.01377.x. [DOI] [PubMed] [Google Scholar]
- O’Guin WM, Sun TT, Manabe M. Interaction of trichohyalin with intermediate filaments: three immunologically defined stages of trichohyalin maturation. J Invest Dermatol. 1992;98:24–32. doi: 10.1111/1523-1747.ep12494172. [DOI] [PubMed] [Google Scholar]
- O’Guin WM, Manabe M. The role of trichohyalin in hair follicle differentiation and its expression in nonfollicular epithelia. Ann NY Acad Sci. 1991;642:51–62. doi: 10.1111/j.1749-6632.1991.tb24380.x. [DOI] [PubMed] [Google Scholar]
- Zhang J, Tan X, Contag CH, Lu Y, Guo D, Harris SE, Feng JQ. Dissection of promoter control modules that direct Bmp4 expression in the epithelium-derived components of hair follicles. Biochem Biophys Res Commun. 2002;293:1412–1419. doi: 10.1016/S0006-291X(02)00416-3. [DOI] [PubMed] [Google Scholar]
- Headon DJ, Overbeek PA. Involvement of a novel Tnf receptor homologue in hair follicle induction. Nat Genet. 1999;22:370–374. doi: 10.1038/11943. [DOI] [PubMed] [Google Scholar]
- Huelsken J, Vogel R, Erdmann B, Cotsarelis G, Birchmeier W. Beta-catenin controls hair follicle morphogenesis and stem cell differentiation in the skin. Cell. 2001;105:533–545. doi: 10.1016/s0092-8674(01)00336-1. [DOI] [PubMed] [Google Scholar]
- Pikus M, Wong WP, Lin J, Wang X, Jiang TX, Chuong CM. Morpho-regulation of ectodermal organs: integument pathology and phenotypic variations in K14-Noggin engineered mice through modulation of bone morphogenetic protein pathway. Am J Pathol. 2004;164:1099–1114. doi: 10.1016/S0002-9440(10)63197-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharov A, Weiner L, Sharova R, Siebenhaar F, Brisette J, Botchkarev V. Multiple functions of noggin in cutaneous development: leads and lessons from noggin transgeneic mice. J Invest Dermatol. 2003;121 Abstract 0828. [Google Scholar]
- Kobielak K, Pasolli HA, Alonso L, Polak L, Fuchs E. Defining BMP functions in the hair follicle by conditional ablation of BMP receptor IA. J Cell Biol. 2003;163:609–623. doi: 10.1083/jcb.200309042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seiberg M, Marthinuss J, Stenn KS. Changes in expression of apoptosis-associated genes in skin mark early catagen. J Invest Dermatol. 1995;104:78–82. doi: 10.1111/1523-1747.ep12613555. [DOI] [PubMed] [Google Scholar]
- Paus R, Foitzik K, Welker P, Bulfone-Paus S, Eichmuller S. Transforming growth factor-beta receptor type I and type II expression during murine hair follicle development and cycling. J Invest Dermatol. 1997;109:518–526. doi: 10.1111/1523-1747.ep12336635. [DOI] [PubMed] [Google Scholar]
- Potter GB, Beaudoin GMR, DeRenzo CL, Zarach JM, Chen SH, Thompson CC. The hairless gene mutated in congenital hair loss disorders encodes a novel nuclear receptor corepressor. Genes Dev. 2001;15:2687–2701. doi: 10.1101/gad.916701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Panteleyev AA, Botchkareva NV, Sundberg JP, Christiano AM, Paus R. The role of the hairless (hr) gene in the regulation of hair follicle catagen transformation. Am J Pathol. 1999;155:159–171. doi: 10.1016/S0002-9440(10)65110-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Botchkarev VA, Welker P, Albers KM, Botchkareva NV, Metz M, Lewin GR, Bulfone-Paus S, Peters EM, Lindner G, Paus R. A new role for neurotrophin-3: involvement in the regulation of hair follicle regression (catagen). Am J Pathol. 1998;153:785–799. doi: 10.1016/S0002-9440(10)65621-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Botchkarev VA, Botchkareva NV, Welker P, Metz M, Lewin GR, Subramaniam A, Bulfone-Paus S, Hagen E, Braun A, Lommatzsch M, Renz H, Paus AR. A new role for neurotrophins: involvement of brain-derived neurotrophic factor and neurotrophin-4 in hair cycle control. EMBO J. 1999;13:395–410. doi: 10.1096/fasebj.13.2.395. [DOI] [PubMed] [Google Scholar]
- Botchkarev VA, Botchkareva NV, Albers KM, Chen LH, Welker P, Paus R. A role for p75 neurotrophin receptor in the control of apoptosis-driven hair follicle regression. EMBO J. 2000;14:1931–1942. doi: 10.1096/fj.99-0930com. [DOI] [PubMed] [Google Scholar]
- Tobin DJ, Foitzik K, Reinheckel T, Mecklenburg L, Botchkarev VA, Peters C, Paus R. The lysosomal protease cathepsin L is an important regulator of keratinocyte and melanocyte differentiation during hair follicle morphogenesis and cycling. Am J Pathol. 2002;160:1807–1821. doi: 10.1016/S0002-9440(10)61127-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuchs E, Merrill BJ, Jamora C, DasGupta R. At the roots of a never-ending cycle. Dev Cell. 2001;1:13–25. doi: 10.1016/s1534-5807(01)00022-3. [DOI] [PubMed] [Google Scholar]
- Alonso L, Fuchs E. Stem cells in the skin: waste not, Wnt not. Genes Dev. 2003;17:1189–1200. doi: 10.1101/gad.1086903. [DOI] [PubMed] [Google Scholar]
- Andl T, Reddy ST, Gaddapara T, Millar SE. WNT signals are required for the initiation of hair follicle development. Dev Cell. 2002;2:643–653. doi: 10.1016/s1534-5807(02)00167-3. [DOI] [PubMed] [Google Scholar]
- Jamora C, DasGupta R, Kocieniewski P, Fuchs E. Links between signal transduction, transcription and adhesion in epithelial bud development. Nature. 2003;422:317–322. doi: 10.1038/nature01458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yuhki M, Yamada M, Kawano M, Iwasato T, Itohara S, Yoshida H, Ogawa M, Mishina Y. BMPR1A signaling is necessary for hair follicle cycling and hair shaft differentiation in mice. Development. 2004;131:1825–1833. doi: 10.1242/dev.01079. [DOI] [PubMed] [Google Scholar]
- Niemann C, Owens DM, Hulsken J, Birchmeier W, Watt FM. Expression of DeltaNLef1 in mouse epidermis results in differentiation of hair follicles into squamous epidermal cysts and formation of skin tumours. Development. 2002;129:95–109. doi: 10.1242/dev.129.1.95. [DOI] [PubMed] [Google Scholar]
- Niemann C, Unden AB, Lyle S, Zouboulis CC, Toftgard R, Watt FM. Indian hedgehog and beta-catenin signaling: role in the sebaceous lineage of normal and neoplastic mammalian epidermis. Proc Natl Acad Sci USA. 2003;100(Suppl 1):S11873–S11880. doi: 10.1073/pnas.1834202100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koster MI, Huntzinger KA, Roop DR. Epidermal differentiation: transgenic/knockout mouse models reveal genes involved in stem cell fate decisions and commitment to differentiation. J Invest Dermatol Symp Proc. 2002;7:41–45. doi: 10.1046/j.1523-1747.2002.19639.x. [DOI] [PubMed] [Google Scholar]
- Porter RM, Jahoda CA, Lunny DP, Henderson G, Ross J, McLean IW, Whittock NV, Wilson NJ, Reichelt J, Magin TM, Lane EB. Defolliculated (dfl): a dominant mouse mutation leading to poor sebaceous gland differentiation and total elimination of pelage follicles. J Invest Dermatol. 2002;119:32–37. doi: 10.1046/j.1523-1747.2002.01806.x. [DOI] [PubMed] [Google Scholar]
- Cui CY, Durmowicz M, Ottolenghi C, Hashimoto T, Griggs B, Srivastava AK, Schlessinger D. Inducible mEDA-A1 transgene mediates sebaceous gland hyperplasia and differential formation of two types of mouse hair follicles. Hum Mol Genet. 2003;12:2931–2940. doi: 10.1093/hmg/ddg325. [DOI] [PubMed] [Google Scholar]
- Allen M, Grachtchouk M, Sheng H, Grachtchouk V, Wang A, Wei L, Liu J, Ramirez A, Metzger D, Chambon P, Jorcano J, Dlugosz AA. Hedgehog signaling regulates sebaceous gland development. Am J Pathol. 2003;163:2173–2178. doi: 10.1016/S0002-9440(10)63574-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laurikkala J, Kassai Y, Pakkasjarvi L, Thesleff I, Itoh N. Identification of a secreted BMP antagonist, ectodin, integrating BMP, FGF, and SHH signals from the tooth enamel knot. Dev Biol. 2003;264:91–105. doi: 10.1016/j.ydbio.2003.08.011. [DOI] [PubMed] [Google Scholar]