Abstract
Tissue plasminogen activator (tPA) is absent from normal human pancreas and is expressed in 95% of human pancreatic adenocarcinomas. We have analyzed the expression of components of the tPA system in murine pancreatic tumors and the role of tPA in neoplastic progression. Transgenic mice expressing T antigen and c-myc under the control of the elastase promoter (Ela1-TAg and Ela1-myc, respectively) were used. tPA was undetectable in normal pancreas, acinar dysplasia, ductal complexes, and in all acinar tumors. By contrast, it was consistently detected in Ela1-myc tumors showing ductal differentiation. Crossing transgenic Ela1-myc with tPA−/− mice had no effect on the proportion of ductal tumors, indicating that tPA is not involved in the acinar-to-ductal transition. Ela1-myc:tPA−/− mice showed an increased survival in comparison to control mice. All ductal tumors, and none of the acinar tumors, overexpressed the tPA receptor annexin A2, suggesting its participation in the effects mediated by tPA. Our findings indicate that murine and human pancreatic ductal tumors share molecular alterations in the tPA system that may play a role in tumor progression.
Exocrine pancreatic cancer is the fifth leading cause of death from malignant disease in Western society and it is one of the most aggressive human tumors.1–5 More than 90% of exocrine tumors are classified as “ductal adenocarcinomas” on the basis of their microscopic appearance.6,7 Except for duodenopancreatectomy with radical intention, there is no curative treatment and the 2-year survival of patients with a 2-cm tumor is approximately 20%.1 The reasons for these biological features are not known. They may be related to the anatomical location of the gland, the genetic alterations involved in tumor development, or other epigenetic factors, including the stromal reaction associated with the tumor.3 New approaches to improve the prevention, diagnosis, and treatment of this disease are necessary to decrease mortality and an improved knowledge of its biology should contribute to these aims.
There has been extensive debate as to the cell of origin of ductal adenocarcinomas,4,8,9 in part as a consequence of the plasticity of the pancreatic epithelium. Genetic data support the notion that most tumors indeed arise from cells in the ducts2,10,11 though the contribution of acinar-to-ductal transdifferentiation cannot be completely ruled out, at least in some cases.9,12,13
In the last few years, several studies have analyzed gene expression patterns associated with pancreas cancer development to provide strategies for improved diagnosis and/or treatment.14–18 Using subtractive hybridization, we found that cultured human pancreas cancer cells overexpress the tissue-type plasminogen activator (tPA)14 and subsequently showed that high levels of tPA are detected in 94% of pancreatic tumors. Blockade of tPA using neutralizing antibodies or chemical inhibitors leads to reduced in vitro tumor invasion.19
The plasminogen system plays a critical role in intravascular thrombolysis as well as in other biological processes that require cellular migration, such as angiogenesis, inflammatory reactions, tissue remodeling, and tumor progression.20–23 There are two types of plasminogen activators that catalyze plasmin generation from plasminogen: tissue-type and urokinase-type (uPA). Activation of plasminogen to plasmin results in progressive degradation of fibrin and other extracellular matrix components and may also lead to activation of metalloproteases, latent growth factors, and proteolysis of membrane glycoproteins.21,22,24–27 All these processes may contribute to tumor development and metastasis. There is extensive evidence supporting the notion that the uPA system, including its receptor and plasminogen activator inhibitor PAI-1, can contribute to tumorigenesis in a variety of tissue types28 but there is less evidence for such a role regarding tPA and annexin A2 (Anx A2), a putative tPA receptor.29–31 We have proposed that, in the pancreas, the tPA system plays an important role in tumor development and/or progression whereas the uPA system may play a more dominant role in pancreatitis.19 More recent studies have shown that tPA stimulates cell proliferation and angiogenesis in exocrine pancreatic tumors.32
One strategy to facilitate progress in the identification of the genes/molecules that are crucial in tumor progression, and in the analysis of their role in these processes, is the use of genetically modified mice. Transgenic mice harboring mutated oncogenes and tumor suppressor genes have proven useful to show that tumorigenic pathways in mice and humans are largely conserved.33 Several mouse models are currently available which recapitulate important aspects of human pancreatic cancer. Most of them target transgenes to acinar cells taking advantage of the well-characterized promoter/enhancers of genes coding for acinar enzymes. Tumors arising in these mice display mainly acinar characteristics. As a consequence, they may not faithfully reproduce the ductal phenotype of human pancreatic cancer.4,34 However, pancreatic epithelial cells display a tremendous plasticity, with metaplastic interconversion between acinar, ductal, and islet lineages playing substantial roles in pathological situations.4,9 Acinar cells from normal exocrine pancreas transdifferentiate in vitro to acquire a phenotype, as well as functional properties, of ductal cells.35 Similarly, mice overexpressing transforming growth factor-α (TGF-α) under the control of the metallothionein/elastase promoter display acinar-ductal metaplasia, with ductal complexes similar to those observed in the pancreas of patients with chronic pancreatitis, and occasionally develop ductal adenocarcinomas at advanced age.34,36,37
In an attempt to explore the role of tPA in pancreatic tumorigenesis, we have taken advantage of two well-established transgenic mouse models: Ela1-Tag(1–127) and Ela1-myc. In these mice, transgenes are targeted to acinar cells using the Elastase-1 enhancer/promoter. Ela1-Tag(1–127) mice, here designated as Ela1-TAg, develop multi-focal acinar cell dysplasia with areas of progression to acinar cell carcinomas.38 By contrast, Ela1-myc transgenic mice develop acinar cell carcinomas that, in approximately 50% of cases, evolve to display areas of ductal differentiation at late stages of tumor progression. The latter are particularly remarkable because they resemble human ductal adenocarcinomas in their morphology, expression of differentiation markers, and extensive desmoplasia.39
In this work, we have analyzed the expression of components of the tPA system in normal pancreas and in tumors from these mice and have found that tPA is selectively expressed in late-stage Ela1-myc tumors displaying ductal differentiation. To determine the functional role of tPA in tumor progression, tPA-deficient mice were mated to Ela1-myc mice. Histological analysis of tumors indicated that tPA is not required for the progression of acinar to ductal tumors. By contrast, Ela1-myc:tPA−/− mice displayed a modest but significant increase in survival in comparison to Ela1-myc or Ela1-myc:tPA+/− control mice. This effect was associated with reduced microvessel density in ductal tumors from Ela1-myc:tPA−/− mice, although a contribution of the genetic background to this effect cannot be completely ruled out. In addition, we have found that Anx A2, a tPA receptor that greatly enhances its catalytic activity,30,31,40 is also selectively overexpressed in tumors with a ductal phenotype, suggesting the coordinated participation of tPA and Anx A2 in tumor progression. Conservation of this signaling axis in human and mouse tumors supports the usefulness of the latter as models for the identification of novel therapeutic strategies for human pancreatic cancer.
Materials and Methods
Transgenic and Knockout Mice
Founder pairs of Ela1-TAg (C57Bl/6 genetic background) and Ela1-myc (C57Bl/6 genetic background) transgenic mice were obtained from Drs. M.J. Tevethia (Department of Microbiology, Pennsylvania State University College of Medicine, Hershey, PA) and E. Sandgren (Department of Pathobiological Sciences, School of Veterinary Medicine, University of Wisconsin-Madison, Madison, WI), respectively. Animals were housed and fed as previously described.38,39 Male transgenic mice were mated with C57Bl/6 females and the offspring screened for the presence of the transgene using PCR. The following primers were used: GCA TCC CAG AAG CCT CCA AAG and GAA TCT TTG CAG CTA ATG GAC C for Ela1-TAg mice and CAC CGC CTA CAT CCT GTC CAT TCA AGC and TTA GGA CAA GGC TGG TGG GCA CTG for Ela1-myc. PCR conditions were as follows: after 5 minutes at 95°C, 40 cycles of denaturation at 94°C for 1 minute, annealing at 60°C for 1 minute, and extension at 72°C for 1 minute were carried out, followed by a final extension at 72°C for 10 minutes. Mouse strains overexpressing TGF-α from the metallothionein (MT-TGF-α) promoter, or the type 2 cholecystokinin receptor (CCK2) from elastase promoter (Ela1-CCK2), have been reported elsewhere.36,41 Expression analyses carried out using these strains were performed on pancreatic tissue from adult mice.
Homozygous knockout mice for tPA (tPA−/− in 75% C57Bl/6, 25% 129SV/SL background) were a kind gift of Dr. P. Carmeliet (Center for Transgene Technology and Gene Therapy, Katholieke Universiteit Leuven, Leuven, Belgium). The basic features of their phenotype have been reported.42 Mice were bred and genotyped by PCR according to described procedures (Dr. V. Attenburrow, Center for Transgene Technology and Gene Therapy, Katholieke Universiteit Leuven, Leuven, Belgium).
Pancreatic Duct Ligation
C57Bl/6 and tPA−/− mice (n = 3 for each) were subjected to duct ligation as previously reported.43 Briefly, mice were anesthetized with a mixture of ketamine (80 mg/kg) and xylazine (20 mg/kg). The peritoneal cavity was explored through a midline laparotomy and the stomach, pancreas, and spleen were mobilized. After ligation of the main pancreatic duct, the viscera were replaced in anatomical position, and the incision was closed. Animals were sacrificed 7 days later and the pancreas was processed for histological analysis.
Analysis of Tumor Development in Transgenic and Hybrid Transgenic-Knockout Mice
Ela1-myc or Ela1-TAg mice were mated to tPA−/− mice to generate Ela1-myc (or Ela1-TAg):tPA+/− F1 hybrid progeny. Subsequently, the F1 hybrid progeny was mated to tPA−/− mice to generate Ela1-myc (or Ela1-TAg):tPA−/− or tPA+/− F2 hybrid progeny. Mice were housed and regularly followed according to procedures established and approved by the Institutional Animal Experimentation Committee. For some experiments, Ela1-TAg and Ela1-myc transgenic mice were sacrificed at defined time points (1, 2, 3, 4, and 5 months of age), an autopsy was performed, and the pancreas was resected and processed for histological analysis. Tumors from transgenic-knockout hybrid mice (Ela1-myc:tPA−/− or Ela1-TAg:tPA−/−) were collected at defined periods of time as indicated above. For the survival study, tumors were resected from animals sacrificed when they showed obvious signs of health deterioration (ie, wasting or abdominal distension) or at the time of death. All animal procedures were approved by the Institutional Animal Experimentation Committee.
Tissue Samples and Histopathological Analysis
Resected tumors were measured and their macroscopic appearance was carefully registered (ie, size, vascularization, consistence, presence of additional macroscopic masses). For histology, samples were fixed in buffered formalin for 24 hours and embedded in paraffin. For immunohistochemical assays using antibodies detecting von Willebrand factor (vWF) and Ki-67, tissues were fixed with fresh 4% paraformaldehyde for 24 hours and embedded in paraffin. Blocks from tumors arising in Ela1-CCK2 mice were obtained as described elsewhere.41 Five-μm sections from all blocks were stained with hematoxylin and eosin (H&E) and scored blindly at ×10 to 20 magnification by a pathologist with extensive experience in pancreatic diseases (J.M.C.).
Immunohistochemistry
tPA was detected using a rabbit antiserum raised against murine tPA that was kindly provided by Dr. L. Moons (Center for Transgene Technology and Gene Therapy, Flanders Institute for Biotechnology, Leuven, Belgium) at a 1:300 dilution. Anx A2 was detected using an antiserum obtained in our laboratory by immunizing rabbits with purified recombinant human Anx A2. The antiserum specifically detected a 36-kd molecular species in lysates from Panc-1 cells, in agreement with the reported molecular mass of Anx A2 (data not shown). To identify endothelial cells in tissue sections, rabbit polyclonal antibody against vWF (NeoMarkers, Fremont, CA) was used at 1:80 dilution; proliferating cells were identified using polyclonal rabbit anti-Ki-67 antibody NCL-Ki67p (Novocastra, Newcastle on Tyne, United Kingdom) at a 1:1500 dilution.
Immunohistochemical analyses were performed using 5-μm sections of paraffin-embedded tissue blocks. Briefly, antigen retrieval was performed by immersing slides in 10 mmol/L citrate (pH 7.3) at 120°C for 1 minute in an autoclave. A Tech-Mate 500 automated immunostainer (Ventana Medical System, Tucson, AZ) was used. Primary antibodies were added for 30 minutes. As secondary antibody, the Envision+ anti-rabbit reagent was applied (Dako, Glostrup, Denmark). Reactions were developed using diaminobenzidine as chromogenic substrate. Sections were counter-stained with hematoxylin, dehydrated, and mounted. As negative controls, tissues were incubated with non-immune (Dako) or pre-immune rabbit serum. The specificity of the antisera used in immunohistochemical assays is described below. Details on methods for detection of vWF have been published elsewhere.44
To obtain semi-quantitative data on the expression of markers of angiogenesis and cell proliferation in Ela1-myc tumors arising in mice with a tPA wild-type or −/− genotype, tissue sections were incubated with antibodies detecting vWF and Ki-67, respectively, as described above. An independent investigator recorded digitalized images of each tumor displaying predominantly acinar or predominantly ductal differentiation. Tumor areas selected for analysis were classified as “ductal”, “mixed with ductal predominance”, “mixed with acinar predominance”, and “acinar” by a pathologist who was blind to the origin of the tumors (J.M.C.). An independent investigator who was also blind to the origin of the tumor images counted the number of vessels (vWF) and the proportion of proliferating cells (Ki-67), either on a computer screen or on a printed microphotograph.
Statistical Analysis
To assess the independence of two categorical variables, the χ2 test was applied. When 20% of cells had expected counts of less than five, Fisher’s exact test was used. To assess the independence of non-normally distributed variables, such as microvessel density or proliferation rate, and the mouse genotypes, the Mann-Whitney test was applied. Survival curves computing the mean time to death or sacrifice were estimated using the Kaplan-Meier method.45,46 Log-rank and Breslow tests47 were applied to compare survival of Ela1-myc (n = 107) control transgenic mice with that of the Ela1-myc:tPA+/− F1 (n = 31), tPA+/− F2 (n = 8), and tPA−/− F2 (n = 41) hybrid progeny. Survival analysis of Ela1-myc, Ela1-myc:tPA+/− F1 and Ela1-myc:tPA+/− F2 mice showed no significant differences. Ela1-myc:tPA−/− mice were compared to either Ela1-myc or Ela1-myc:tPA+/− F1 (survival analysis) or with Ela1-myc (vessel density analysis). Results were considered significant at the two-sided p of 0.05 level. Statistical analyses were performed using version 9.0 SPSS statistical package (SPSS Inc., Chicago, IL, 1999).
Results
tPA Is Undetectable in Normal Murine Exocrine Pancreas and Is Selectively Expressed in Murine Pancreatic Tumors Displaying a Ductal Phenotype
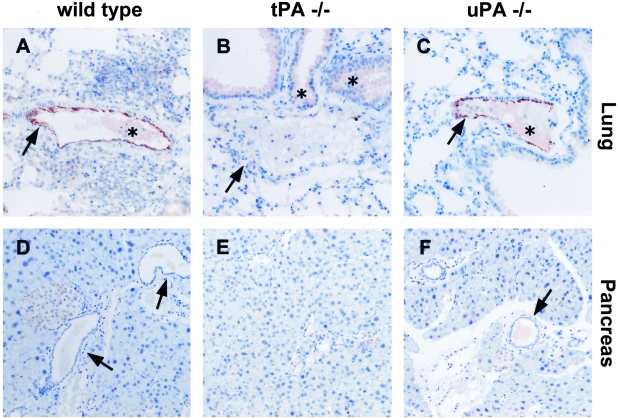
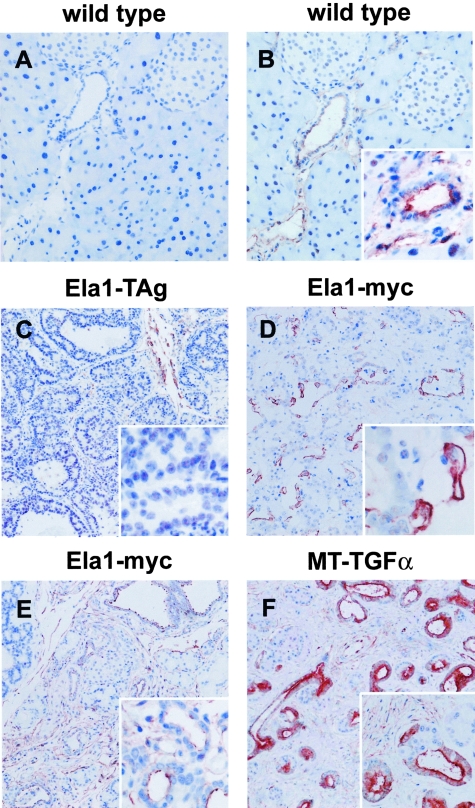
Expression of tPA in murine tissues has been poorly characterized because most of the antibodies available have been raised against human tPA and do not recognize the mouse protein. We have optimized a technique to detect murine tPA in sections from paraffin-embedded tissues using a highly specific polyclonal antibody raised against murine tPA. To demonstrate the specificity of the antiserum, we used normal lung and pancreas from wild-type, tPA−/−, and uPA−/− mice as controls (Figure 1). In normal lung, strong staining of endothelial cells was observed in wild-type and uPA−/− mice (Figure 1, A and C, arrows). By contrast, no staining was detected in pulmonary vessels from tPA−/− mice (Figure 1B, arrow). In normal pancreas, tPA was undetectable in acinar and ductal cells (Figure 1, D and F, arrows), indicating that tPA is absent from all exocrine pancreatic cells.
Figure 1-4244.
Immunostaining for tPA in normal lung and pancreas tissues from wild-type (A and D), tPA −/− (B, E) and uPA −/− mice (C and F). Anti-tPA antibodies show a strong reactivity with vascular endothelial cells in the lung of wild-type (A) and uPA−/− (C) mice but not in cells from tPA−/− (B) mice. In the pancreas, tPA is undetectable in normal acinar and ductal cells (D and F, arrows). Asterisk indicates non-specific staining. Original magnification, ×200 (A–F).
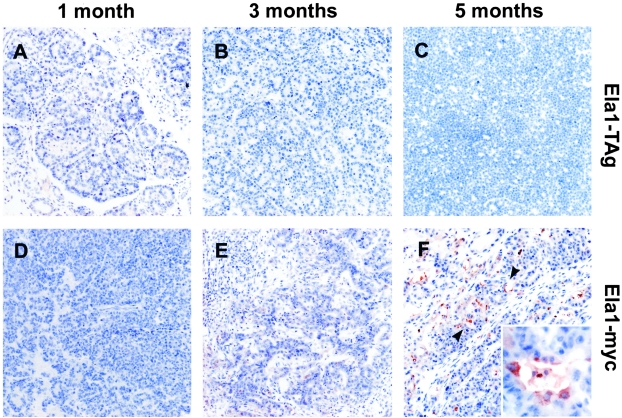
The expression of tPA in pancreatic tumors from Ela1-TAg and Ela1-myc transgenic mice at different stages of progression was analyzed using immunohistochemistry. Figure 2 shows the reactivity of anti-tPA antiserum with pancreas from 1-, 3-, and 5-month-old mice. As described previously, Ela1-TAg transgenic mice develop many neoplastic nodules embedded in a dysplastic pancreas. Histological analysis of these nodules revealed acinar cell dysplasia progressing into carcinomas (Figure 2, compare A and B, early stages, with C, late stage). Expression of tPA was completely absent in the non-neoplastic pancreatic epithelium. Similarly, tPA was not detected in any of the Ela1-TAg acinar tumors examined (0 of 14). The pancreas from young Ela1-myc mice displayed acinar cell hyperplasia and dysplasia (Figure 2D). Older mice developed acinar tumors, similar to those from Ela1-TAg mice (Figure 2E), and occasional progression to poorly differentiated carcinomas. As previously described,39 approximately half of the tumors from older mice displayed areas of ductal differentiation, associated with extensive desmoplasia, adjacent to areas of acinar differentiation (Figure 2F). tPA was undetectable in exocrine cells in areas displaying acinar cell dysplasia and in tumors showing an acinar (0 of 6) or undifferentiated (0 of 2) phenotype. In 6 tumors, areas of ductal differentiation were identified adjacent to areas of acinar differentiation (Figure 2F, arrowheads). tPA was detected in tumor cells in all 6 cases in areas showing ductal differentiation whereas it was always undetectable in areas of acinar differentiation (Figure 2F). The differential expression of tPA in tumor areas with an acinar versus ductal morphology was statistically significant (P = 0.002). tPA-expressing tumors were more common in mice ≥ 3 months age (6 of 13) than in mice < 3 months age (0 of 8) (P = 0.046).
Figure 2-4244.
Histological analysis and tPA expression in tumors from Ela1-TAg and Ela1-myc transgenic mice. Sections of paraffin-embedded pancreatic tissue from 1-month (A and D), 3-month (B and E), and 5-month-old (C and F) mice were analyzed for tPA expression as described in Materials and Methods. tPA expression is restricted to ductal cells in tumors from late stages of Ela1-myc mice (F, arrowheads and inset). There was no reactivity with dysplastic acini (A and D) nor with acinar tumors (B, C, E, and F) arising in both mouse strains. Inset of F shows tPA expression in tumor cells displaying ductal differentiation. Original magnification; ×100 (A–F), ×400 (F, inset).
To confirm and extend these findings, we analyzed tPA expression in the pancreas of Ela1-CCK2 transgenic mice.41 Overexpression of CCK2/gastrin receptor in the exocrine pancreas stimulates pancreatic growth, acinar cell hypertrophy and, in a small proportion of cases, the progression to pancreatic cancer.48 Immunohistochemical analysis showed that tPA was undetectable in acinar tumors from Ela1-CCK2 mice, further supporting the notion that tPA expression is selectively expressed in ductal tumors (data not shown).
tPA Expression Is Selectively Associated with the Neoplastic Phenotype and Is Not Required for Ductal Metaplasia
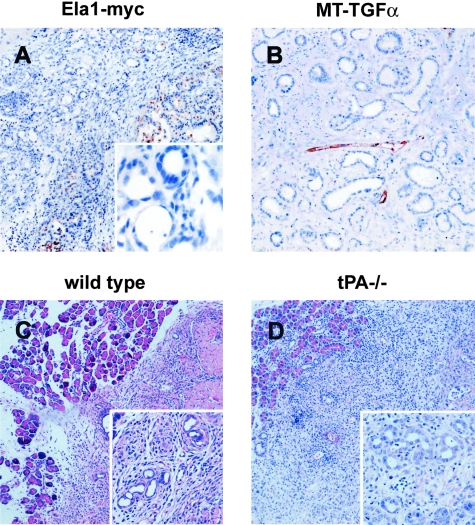
To determine whether tPA expression was associated with ductal metaplasia as well as with the neoplastic phenotype, ductal complexes characteristic of obstructive chronic pancreatitis were examined in the pancreas of Ela1-TAg (data not shown) and Ela1-myc mice (Figure 3). Metaplastic ducts consistently lacked tPA expression (Figure 3A and inset) as did normal ductal cells (Figure 1). A massive acinar-ductal transdifferentiation has been described in the pancreas of MT-TGF-α mice.36 In this strain, non-neoplastic ductal complexes consistently lacked tPA expression (n = 7 mice) (Figure 3B). This expression pattern is similar to that previously described in the human pancreas.19 To confirm and extend these findings, we analyzed the effect of pancreatic duct ligation in C57Bl/6 and tPA−/− mice. Duct complexes occurred to a similar extent in both mouse strains (Figure 3, C and D), indicating that tPA is not required for ductal metaplasia.
Figure 3-4244.
tPA is not expressed in metaplastic ducts and is not required for acinar-ductal metaplasia. A and B: Immunohistochemical analysis of the expression of tPA in non-neoplastic ductal complexes. Sections of paraffin-embeded pancreatic tissues containing non-neoplastic ductal complexes were used to examine tPA (A and B) expression in pancreatic tissue from Ela1-myc (A) and MT-TGF-α mice (B). tPA was undetectable in ductal complexes (A and B, left) but it was present in tumor cells (A, right) and endothelial cells (B). Inset in A shows the lack of tPA expression in ductal complexes from Ela1-myc pancreatic tissue using higher magnification. C and D: Pancreatic duct ligation in wild-type and tPA−/− mice. Metaplastic ducts were observed in mice of both genotypes (C and D), indicating that tPA is not required for acinar-ductal metaplasia. Insets show the ductal complexes appearing after duct ligation. Original magnification; ×100 (A–D), ×400 (inset, B), ×200 (insets, C and D).
Progression of Pancreatic Tumors Induced by the Ela1-myc Transgene in the Absence of tPA (Ela1-myc:tPA−/−)
To analyze the role of tPA in the progression of pancreatic tumors, we mated Ela1-myc transgenic mice to tPA-deficient mice and analyzed tumor progression and survival in animals with a tPA+/− or tPA−/− genotype expressing the transgene. Table 1 summarizes the histological features of tumors arising as a result of the expression of the Ela1-myc transgene in mice with a wild-type, tPA+/−, and tPA−/− genotype. Representative findings are shown in Figure 4. There was no significant difference in the proportion of mice with tumors displaying areas of ductal differentiation; the latter tended to increase with age, regardless of the tPA genotype. These data support the notion that tPA does not play a role in acinar-ductal transition (see above). Tumors showing extensive apoptotic figures were more common in the control Ela1-myc.
Table 1.
Histology of Pancreatic Carcinomas from Ela1-myc and Ela1-myc tPA−/− Transgenic Mice
| Histological pattern (acinar cell carcinoma) | Ela1-myc (n = 20) | Ela1-myc:tPA−/− (n = 20) |
|---|---|---|
| Well differentiated | 5% | 10.5% |
| Moderately differentiated with little or no apoptosis | 5% | 15.8% |
| Poor-moderately differentiated with apoptosis | 35% | 10.5% |
| With ductal differentiation | 55% | 63.2% |
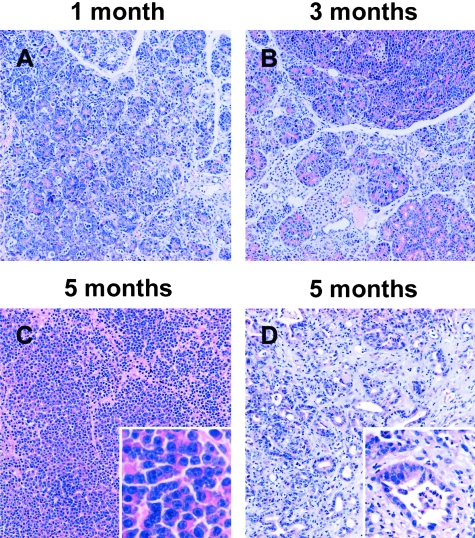
Figure 4-4244.
Histological analysis of Ela1-myc tPA−/− transgenic mice-derived tumors. Sections of paraffin-embeded pancreatic tissue from Ela1-myc tPA−/− transgenic mice were stained with hematoxylin and eosin. The pancreas from young mice shows widespread acinar dysplasia (A and B) whereas tumors in 5- month-old mice display areas of acinar (C) as well as ductal (D) differentiation. C and D and the corresponding insets display different areas from the same tumor. Original magnification; ×100 (A–D), inset, ×400 (C) and ×200 (D).
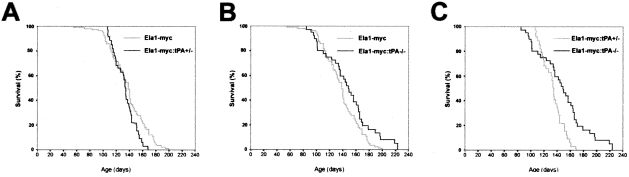
Kaplan-Meier survival curves were generated to examine tumor progression in Ela1-myc with a wild-type (n = 107), tPA+/− (n = 31), and tPA−/− (n = 41) genotype. Median survival of Ela1-myc tPA+/+, tPA+/− and tPA−/− mice was 139 ± 2.3, 134 ± 1.6, and 149 ± 6.7 days, respectively. The survival curves of Ela1-myc wild-type and Ela1-myc:tPA+/− mice showed no significant differences (Figure 5A). By contrast, the survival curve of Ela1-myc:tPA−/− mice diverged from that of wild-type (Figure 5B) or tPA+/− (Figure 5C) mice after 4 months of age: 4% of Ela1-myc and 0% of Ela1-myc:tPA+/− versus 15% of Ela1-myc:tPA−/− mice lived more than 6 months (Figure 5 and Table 2). The comparison of differences in both curves did not reach statistical significance using Breslow test but was clearly significant (P = 0.02, Figure 5B or P = 0.005, Figure 5C) using the log-rank test. This statistical is more appropriate because the Breslow test weights heavily early events, whereas the log-rank test allows comparisons of the final portion of the curves.46,47 These findings are consistent with the fact that tPA was undetectable in acinar tumors, occurring in the initial phase of tumor development, but was reproducibly expressed in ductal tumors appearing at a later age.
Figure 5-4244.
Effect of tPA null mutation on survival of Ela1-myc transgenic mice. Survival of Ela1-myc wild-type transgenic mice versus Ela1-myc:tPA+/− (A) or Ela1-myc:tPA−/− (B) is compared. C: Survival curve of Ela1-myc:tPA+/− mice versus Ela1-myc:tPA−/−. Of note, there is a statistically significant increase in survival of Ela1-myc tPA−/− mice (P = 0.02 when compared to Ela1-myc or P = 0.005 when compared to Ela1-myc:tPA+/−) which depends on effects taking place after 120 days of follow-up, when acinar-to-ductal metaplasia and tPA expression occur in these tumors.
Table 2.
Survival Analysis of Ela1-myc, Ela1-myc:tPA+/−, and Ela1-myc:tPA−/− Mice
| Genotype | n | Survival* (days) | Survival† (4 months) | Survival† (5 months) | Survival† (6 months) |
|---|---|---|---|---|---|
| Ela1-myc | 107 | 139 | 70% | 26% | 4% |
| Ela1-myc:tPA+/− | 31 | 134 | 71% | 19.3% | 0% |
| Ela1-myc:tPA−/− | 41 | 149 | 73% | 46% | 15% |
, median;
, proportion of animals alive.
To get insight into the mechanisms responsible for the increase in survival observed in Ela1-myc:tPA−/− mice, we examined vascular and nerve invasion, vessel density, and cell proliferation in tumors from Ela1-myc and Ela1-myc:tPA−/− mice. There were no significant differences in the prevalence of vascular (2 of 18 versus 3 of 14) and nerve (1 of 18 versus 3 of 14) invasion, regardless of the histological differentiation. The assessment of nodal involvement was limited by the fact that large tumors might have erased small nodes. When considering only invasion in areas where a node could be recognized, the frequency of nodal involvement was not significantly different in both groups of tumors (2 of 4 nodes among 18 tumors from Ela1-myc mice and 5 of 6 nodes among 14 tumors from Ela1-myc:tPA−/− mice). Microvessel density was analyzed using immunohistochemistry with antibodies detecting vWF: in acinar areas, no differences in density were found in relationship to the tPA genotype; by contrast, the vessel density observed in ductal areas was higher in tumors arising in Ela1-myc wild-type mice in comparison to Ela1-myc:tPA−/− (14.5 ± 3 versus 12 ± 2.3, P = 0.04) (Table 3). Proliferating cells were identified using antibodies detecting Ki-67; the proportion of proliferating cells was higher in tumors arising in tPA wild-type than in those from tPA−/− mice, regardless of the histology of the tumor (Table 3). However, these differences did not reach statistical significance.
Table 3.
Analysis of Markers of Angiogenesis and Cell Proliferation in Tumors from Ela1-myc and Ela1-myc:tPA−/− Mice
| Histology | von Willebrand factor* |
Ki-67* |
||||
|---|---|---|---|---|---|---|
| Ela1-myc | Ela1-myc: tPA−/− | p† | Ela1-myc | Ela1-myc: tPA−/− | p† | |
| Ductal + mixed/ductal | 14.5 ± 3.1 (11)‡ | 12.1 ± 2.3 (9) | 0.04 | 33.4 ± 12.5 (5) | 22.3 ± 14.3 (4) | 0.29 |
| Acinar + mixed/acinar | 8.1 ± 4.2 (13) | 6.3 ± 2.8 (10) | 0.28 | 37.9 ± 21.8 (6) | 30.7 ± 19.1 (6) | 0.31 |
, vWF, results expressed as vessels/field; Ki-67, results expressed as % immunoreactive cells (mean ± standard deviation).
, p values correspond to the comparison of data from tPA+/+ and tPA−/− mice for a given parameter using Mann-Whitney test.
, brackets indicate the total number of tumors analyzed.
Similar analyses were performed by mating Ela1-TAg transgenic mice and tPA−/− mice. In this case, survival of Ela1-TAg mice was not affected by the tPA gene status (data not shown). These results might have been expected because Ela1-TAg tumors consistently lack tPA expression (Figure 2).
Altogether, these findings suggest that tPA deficiency plays a protective role in the progression of ductal carcinomas in Ela1-myc mice and that these effects may be mediated through reduced angiogenesis.
Annexin A2 Expression in Murine Exocrine Pancreas Tumors
Anx A2 has been shown to be a tPA receptor in endothelial cells31,49,50 and we have proposed that it may also act as a tPA receptor in human pancreatic ductal adenocarcinomas19 (Peiró S, Corominas JM, Aguilar S, Ampurdanés C, Navarro P, Real EX, submitted for publication). Therefore, we also investigated Anx A2 expression in normal mouse pancreas and in Ela1-TAg and Ela1-myc tumors. Anx A2 was undetectable in normal acinar cells and was expressed at low levels in normal ductal cells, mesenchymal cells, and vascular endothelium (Figure 6, A and B). Anx A2 was undetectable in all 12 tumors showing acinar differentiation analyzed, regardless of the transgene that caused them (Figure 6C). By contrast, it was strongly expressed in ductal adenocarcinomas in Ela1-myc mice (6 of 6, P < 0.001) (Figure 6D). Anx A2 was found in the apical membrane of normal cells whereas it showed a non-polarized distribution in tumor cells. Expression of Anx A2 was similar in tumors displaying ductal differentiation arising in Ela1-myc:tPA−/− and in Ela1-myc mice (data not shown), indicating that the expression of Anx A2 expression is independent from that of tPA.
Figure 6-4244.
Immunohistochemical analysis of Anx A2 expression in tumors and non-neoplastic ductal complexes. A–D: Anx A2 expression in tumors from Ela1-TAg and Ela1-myc transgenic mice. Sections of paraffin-embeded pancreatic tissues were analyzed for Anx A2 expression as described in Materials and Methods. Normal pancreatic tissue from wild-type mice (A and B); acinar tumor from a 5-month-old Ela1-TAg mouse (C); ductal component in a mixed acinar-ductal carcinoma from a 5-month-old Ela1-myc mouse (D). Pre-immune serum (A); anti-Anx A2 serum (B–D). Anx A2 shows an apical expression in normal ductal cells (inset, B) but it is undetectable in normal acinar cells (B) and in acinar tumors (C and inset). By contrast, tumor cells with a ductal phenotype show an increased and unpolarized expression of Anx A2 (D, also see inset). Anx A2 is also detected in stromal cells (B and C). E and F: Anx A2 expression in non-neoplastic ductal complexes from Ela1-myc (E) and MT-TGF-α (F). Anx A2 was expressed in non-neoplastic ductal complexes in both mouse models (insets). Original magnification; ×200 (A, B, inset in F), ×100 (C–F), ×400 (insets in B–E).
We also analyzed Anx A2 expression in non-neoplastic ductal complexes in the pancreas from Ela1-TAg (data not shown), Ela1-myc (Figure 6E), and MT-TGF-α mice (Figure 6F). Anx A2 was strongly expressed in these ductal complexes, but not in acinar cells (Figure 6F), indicating that, in contrast to tPA, Anx A2 up-regulation is not exclusively associated with the neoplastic phenotype.
Discussion
In this work we have extended our prior finding that the tPA system plays a role in the in vitro phenotype of human exocrine pancreatic tumors by using transgenic mouse models of pancreatic cancer. The results presented here indicate that the expression of components of the tPA system is conserved in human and murine pancreatic tumors and that tPA is not required for acinar-to-ductal transdifferentiation; they also suggest that the activation of a functional tPA circuit in ductal pancreatic tumors participates in tumor progression.
tPA expression was found to be strongly associated with the neoplastic phenotype using cultured human pancreatic cells as well as human tissues.19 Similarly, tPA was undetectable in normal murine exocrine pancreas epithelium as well as non-neoplastic ductal complexes but it was expressed in all Ela1-myc tumors displaying a ductal phenotype analyzed. By contrast, it was not detected in any of the acinar tumors studied originating in three different mouse strains in which different transgenes were targeted to acinar cells. This pattern of expression is similar to that described in human tumors, thus validating the use of these transgenic mouse strains for the pre-clinical evaluation of drugs targeting the tPA-Anx A2 system.
The molecular mechanisms leading to the activation of the expression of tPA in ductal pancreatic tumors are not known. We have previously reported that tPA expression in pancreatic cell lines and tumors was associated with K-ras mutations,19 one of the most common genetic alterations associated with this tumor.51 Unlike human exocrine pancreas cancers, tumors arising in Ela1-TAg and Ela1-myc mice have been found to be K-ras wild-type regardless of their histological appearance.52 It is, however, possible that ras proteins are activated in response to extracellular signals in these tumors,53 rather than through point mutation, or that other molecular events downstream of ras are active in tumors with a ductal phenotype. In support of this notion, the ERK MAP kinase pathway has been shown to be active in ductal complexes lacking K-ras mutations in Ela-TGF-α mice.34 Furthermore, recent data suggest different roles for Ras genes in oncogenesis in mice and men.54 Wild-type p53, but not mutant p53 proteins, has been reported to repress the activity of the tPA promoter and play a role in the regulation of tPA expression.55 The Ela1-TAg mouse strain used here develops tumors that express a wild-type conformation of p53.38 However, more work is necessary to establish the role of p53 as no firm relationship between p53 mutations and tPA expression has been reported in human cancers.
The histology of the tumors induced by the Ela1-myc transgene was not modified by the lack of tPA. MMP-7 has been found to be crucial for the development of ductal complexes in response to main duct ligation.56 Using this strategy, we have not found differences in ductal complex appearance between wild-type and tPA−/− mice. These results indicate that tPA is not required for acinar-to-ductal metaplasia.
Cross-breeding of Ela1-myc and tPA knockout mice showed that tPA deficiency is associated with a modest but significant increase in survival, suggesting that tPA plays a role in murine pancreas cancer progression. Several studies have shown that the genetic background can modulate the effects of transgenes or null alleles in mouse models of cancer.57–59 In our study, Ela1-myc:tPA−/− mice were compared with either Ela1-myc or Ela1-myc:tPA+/− F1, which present quite similar but not identical genetic background. To better understand the effects of genetic background in our studies, survival curves between Ela1-myc, Ela1-myc:tPA+/− F1 and Ela1-myc:tPA+/− F2 (whose genetic background is identical to that of Ela1-myc:tPA−/− mice) were compared and no significant differences were found. These data, together with the fact that tPA genotype had no effect on the survival of Ela1-TAg mice, support the notion that the survival increase observed in Ela1-myc:tPA−/− could be attributed to the absence of tPA rather than to differences in genetic background. However, the involvement of genetic effects due to allelic variation cannot be completely ruled out. The slight increase in survival and the lack of long-term survivors in Ela1-myc:tPA−/− (ie, > 12 months) can be attributed to two reasons: 1) Ela1-myc mice develop very aggressive tumors and inactivation of a single protease is not sufficient to completely prevent tumor progression; 2) tPA is expressed only in tumors showing ductal differentiation, a phenotype that occurs only at late stage of tumor progression. Furthermore, areas of acinar differentiation lacking tPA expression, likely unaffected by the absence of tPA, are generally also present.
tPA is thought to act by binding to cellular receptors and recent evidences from our group indicate that Anx A2 is a tPA receptor in pancreas cancer cells (Peiró S, Corominas JM, Aguilar S, Ampurdanis C, Navarro P, Real FX, submitted for publication). Interestingly, Anx A2 was found to be expressed in tumors with a ductal phenotype but not in those with an acinar phenotype. These results suggest that a functional circuit is activated in ductal tumors leading to an increased tPA catalytic activity in neoplastic cells. Our recent work also suggests that Anx A2 participates in tPA-mediated intracellular signaling though the precise mechanisms by which it does so are not known (Peiró S, Corominas JM, Aguilar S, Ampurdanis C, Navarro P, Real FX, submitted for publication). Anx A2 has been reported to be overexpressed in a small number of exocrine pancreatic tumors in men60 and hamster.61 Unlike tPA, which was expressed exclusively in neoplastic ductal cells, Anx A2 was also overexpressed in non-neoplastic ductal complexes, suggesting that different molecular events participate in the activation of these two genes in ductal cells. The loss of polarity of Anx A2, found in cultured human pancreatic cells19 and confirmed here in tumors from Ela1-myc mice, supports the notion that Anx A2 could participate in the localization of tPA proteolytic activity to the basal membrane, where it could contribute to the degradation of matrix components. The elucidation of the role of Anx A2 in pancreatic tumorigenesis will be facilitated by the study of mice deficient in the gene coding for this protein62 and by the structural analysis of tPA/Anx A2 interaction.63
The mechanisms by which tPA contributes to tumor progression are not yet well-defined. Immunohistochemical analyses suggest that, in areas displaying ductal differentiation, there is a significantly lower number of vessels/field in tumors from Ela1-myc:tPA−/− mice than in those from Ela1-myc wild-type mice. By contrast, this difference was not apparent in areas displaying acinar differentiation. Due to the experimental design of our studies, a contribution of the differences in genetic background between both genotypes cannot be completely ruled out. However, the fact that vessel reduction was observed specifically in ductal areas, where tPA is expressed in control mice, and not in acinar areas, suggests a relationship to the lack of tPA. In this regard, a role for tPA in angiogenesis stimulation has previously been proposed using xenografts of human pancreas cancer cells in nude mice.32 Our results suggest that the increase in survival observed in Ela1-myc:tPA−/− mice could be a consequence of the inhibition of tumor angiogenesis by the lack of tPA. Tumors with acinar or ductal differentiation showed a lower proliferation rate in the absence of tPA. While this difference did not reach statistical significance, it is possible that the survival effect observed may also be mediated, in part, by changes in cell proliferation. This possibility is strengthened by the fact that when only the rare tumors containing exclusively ductal differentiation were considered, there was a marked decrease in the proportion of Ki-67-positive cells in tumors lacking tPA (57.3 ± 20.4 versus 29.4 ± 21). Such an effect was not observed in purely acinar tumors. Díaz et al32 have reported that tPA can stimulate the in vitro proliferation of pancreatic cells as well as tumor growth in vivo, using xenografts of human tumor cells. In addition, inhibition of tPA expression using an antisense strategy led to reduced in vivo tumor growth cells. These investigators have proposed that plasmin generation is important for tPA-mediated in vitro invasiveness but not for its mitogenic activity. In addition, previous work using chemical inhibitors and neutralizing antibodies supported the notion that the proteolytic activity of tPA played a role in in vitro tumor invasion.19 Introducing the Ela1-myc transgene in a plasminogen −/− genotype might be useful to determine the role of plasmin in tPA effects described in this work.
The findings reported here extend the evidence on the involvement of tPA in exocrine pancreas cancer progression and demonstrate that these effects are not mediated by modulation of the acinar-to-ductal transdifferentiation process.
Acknowledgments
We thank M. Bautista, T. Lobato, and C. Ampurdanés for excellent technical assistance, S. Gómez and the staff of the Animal Room for help with mouse care, Drs. E. Sandgren, J. Tevethia, and P. Carmeliet for kindly providing genetically modified mice, Dr. S. Montaner for valuable help with the study of markers of angiogenesis, and Drs. X. Mayol and G. Gil for critical reading of the manuscript and other valuable contributions.
Footnotes
Address reprint requests to Pilar Navarro, Unitat de Biologia Cellular i Molecular, Institut Municipal d’Investigació Mèdica, Dr. Aiguader, 80, 08003-Barcelona, Spain. E-mail: pnavarro@imim.es.
Supported by grants from Instituto de Salud Carlos III (00/0462), Biomed Program (BMH4-CT98.3085 and QLG-CT-2002–01196), Dirección General de Enseñanza Superior e Investigación Científica (PM97–0077), Plan Nacional de I+D (SAF2001–0420) and CIRIT (Generalitat de Catalunya) (SGR-00245, SGR-00410, and ITT-CTP98–1).
F.X.R. and P.N. made equal contributions to this work.
References
- American Gastroenterological Association Medical Position Statement Epidemiology, diagnosis, and treatment of pancreatic ductal adenocarcinoma. Gastroenterology. 1999;117:1463–1484. doi: 10.1016/s0016-5085(99)70297-0. [DOI] [PubMed] [Google Scholar]
- Jaffee EM, Hruban RH, Canto M, Kern SE. Focus on pancreas cancer. Cancer Cell. 2002;2:25–28. doi: 10.1016/s1535-6108(02)00093-4. [DOI] [PubMed] [Google Scholar]
- Real FX. A “catastrophic hypothesis” for pancreas cancer progression. Gastroenterology. 2003;124:1958–1964. doi: 10.1016/s0016-5085(03)00389-5. [DOI] [PubMed] [Google Scholar]
- Bardeesy N, Sharpless NE, DePinho RA, Merlino G. The genetics of pancreatic adenocarcinoma: a roadmap for a mouse model. Semin Cancer Biol. 2001;11:201–218. doi: 10.1006/scbi.2000.0371. [DOI] [PubMed] [Google Scholar]
- Evans DB, Abbruzzese JL, Willet CG. De Vita VH, Hellman S, Rosenberg S, editors. Lippincott Williams & Wilkins,; Philadelphia: Cancer of the pancreas Cancer Principles and Practice of Oncology. 2001:pp 1126–1161. [Google Scholar]
- Klöppel G, Fitzgerald PJ. The exocrine pancreas: Biology, Pathobiology, and Diseases. Go VLW, Brooks FP, Di Magno EP, Gardner JD, Lebenthal E, Scheele GA, editors. Raven Press,; New York: 1986:pp 649–674. [Google Scholar]
- Longnecker DS. Experimental models of exocrine pancreatic tumors. Go VLW, Brooks FP, Di Magno EP, Gardner JD, Lebenthal E, Scheele GA, editors. Raven Press,; New York: The Exocrine Pancreas Biology: Biology, Pathobiology and Diseases. 1986:pp 443–458. [Google Scholar]
- Schmid RM. Acinar-to-ductal metaplasia in pancreatic cancer development. J Clin Invest. 2002;109:1403–1404. doi: 10.1172/JCI15889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Real FX. The cell biology of pancreatic cancer: an overview. Neoptolemos J, Lemoine NR, editors. Blackwell Science Press,; Oxford: Pancreatic Cancer. 1995:pp 3–17. [Google Scholar]
- Kloppel G, Longnecker DS. Hyperplastic and metaplastic changes in pancreatic ducts: nomenclature and preneoplastic potential. Ann NY Acad Sci. 1999;880:66–73. doi: 10.1111/j.1749-6632.1999.tb09510.x. [DOI] [PubMed] [Google Scholar]
- Hruban RH, Wilentz RE, Kern SE. Genetic progression in the pancreatic ducts. Am J Pathol. 2000;156:1821–1825. doi: 10.1016/S0002-9440(10)65054-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scarpelli DG, Rao MS, Reddy JK. Are acinar cells involved in the pathogenesis of ductal adenocarcinoma of the pancreas? Cancer Cells. 1991;3:275–277. [PubMed] [Google Scholar]
- Adell T, Gomez-Cuadrado A, Skoudy A, Pettengill OS, Longnecker DS, Real FX. Role of the basic helix-loop-helix transcription factor p48 in the differentiation phenotype of exocrine pancreas cancer cells. Cell Growth Differ. 2000;11:137–147. [PubMed] [Google Scholar]
- Paciucci R, Berrozpe G, Tora M, Navarro E, Garcia de Herreros A, Real FX. Isolation of tissue-type plasminogen activator, cathepsin H, and non-specific cross-reacting antigen from SK-PC-1 pancreas cancer cells using subtractive hybridization. FEBS Lett. 1996;385:72–76. doi: 10.1016/0014-5793(96)00352-3. [DOI] [PubMed] [Google Scholar]
- Gress TM, Muller-Pillasch F, Geng M, Zimmerhackl F, Zehetner G, Friess H, Buchler M, Adler G, Lehrach H. A pancreatic cancer-specific expression profile. Oncogene. 1996;13:1819–1830. [PubMed] [Google Scholar]
- Crnogorac-Jurcevic T, Efthimiou E, Nielsen T, Loader J, Terris B, Stamp G, Baron A, Scarpa A, Lemoine NR. Expression profiling of microdissected pancreatic adenocarcinomas. Oncogene. 2002;21:4587–4594. doi: 10.1038/sj.onc.1205570. [DOI] [PubMed] [Google Scholar]
- Ryu B, Jones J, Blades NJ, Parmigiani G, Hollingsworth MA, Hruban RH, Kern SE. Relationships and differentially expressed genes among pancreatic cancers examined by large-scale serial analysis of gene expression. Cancer Res. 2002;62:819–826. [PubMed] [Google Scholar]
- Han H, Bearss DJ, Browne LW, Calaluce R, Nagle RB, Von Hoff DD. Identification of differentially expressed genes in pancreatic cancer cells using cDNA microarray. Cancer Res. 2002;62:2890–2896. [PubMed] [Google Scholar]
- Paciucci R, Tora M, Diaz VM, Real FX. The plasminogen activator system in pancreas cancer: role of t-PA in the invasive potential in vitro. Oncogene. 1998;16:625–633. doi: 10.1038/sj.onc.1201564. [DOI] [PubMed] [Google Scholar]
- Collen D, Lijnen HR. Molecular basis of fibrinolysis, as relevant for thrombolytic therapy. Thromb Haemost. 1995;74:167–171. [PubMed] [Google Scholar]
- Dano K, Andreasen PA, Grondahl-Hansen J, Kristensen P, Nielsen LS, Skriver L. Plasminogen activators, tissue degradation, and cancer. Adv Cancer Res. 1985;44:139–266. doi: 10.1016/s0065-230x(08)60028-7. [DOI] [PubMed] [Google Scholar]
- DeClerck YA, Imren S, Montgomery AM, Mueller BM, Reisfeld RA, Laug WE. Proteases and protease inhibitors in tumor progression. Adv Exp Med Biol. 1997;425:89–97. doi: 10.1007/978-1-4615-5391-5_9. [DOI] [PubMed] [Google Scholar]
- Liotta LA, Rao CN, Barsky SH. Tumor invasion and the extracellular matrix. Lab Invest. 1983;49:636–649. [PubMed] [Google Scholar]
- Carmeliet P, Moons L, Lijnen R, Baes M, Lemaitre V, Tipping P, Drew A, Eeckhout Y, Shapiro S, Lupu F, Collen D. Urokinase-generated plasmin activates matrix metalloproteinases during aneurysm formation. Nat Genet. 1997;17:439–444. doi: 10.1038/ng1297-439. [DOI] [PubMed] [Google Scholar]
- Mars WM, Zarnegar R, Michalopoulos GK. Activation of hepatocyte growth factor by the plasminogen activators uPA and tPA. Am J Pathol. 1993;143:949–958. [PMC free article] [PubMed] [Google Scholar]
- Houck KA, Leung DW, Rowland AM, Winer J, Ferrara N. Dual regulation of vascular endothelial growth factor bioavailability by genetic and proteolytic mechanisms. J Biol Chem. 1992;267:26031–26037. [PubMed] [Google Scholar]
- Saksela O, Rifkin DB. Cell-associated plasminogen activation: regulation and physiological functions. Annu Rev Cell Biol. 1988;4:93–126. doi: 10.1146/annurev.cb.04.110188.000521. [DOI] [PubMed] [Google Scholar]
- Ossowski L, Aguirre-Ghiso JA. Urokinase receptor and integrin partnership: coordination of signaling for cell adhesion, migration and growth. Curr Opin Cell Biol. 2000;12:613–620. doi: 10.1016/s0955-0674(00)00140-x. [DOI] [PubMed] [Google Scholar]
- Hajjar KA, Jacovina AT, Chacko J. An endothelial cell receptor for plasminogen/tissue plasminogen activator: I Identity with annexin II. J Biol Chem. 1994;269:21191–21197. [PubMed] [Google Scholar]
- Cesarman GM, Guevara CA, Hajjar KA. An endothelial cell receptor for plasminogen/tissue plasminogen activator (t-PA): II Annexin II-mediated enhancement of t-PA-dependent plasminogen activation. J Biol Chem. 1994;269:21198–21203. [PubMed] [Google Scholar]
- Kassam G, Choi KS, Ghuman J, Kang HM, Fitzpatrick SL, Zackson T, Zackson S, Toba M, Shinomiya A, Waisman DM. The role of annexin II tetramer in the activation of plasminogen. J Biol Chem. 1998;273:4790–4799. doi: 10.1074/jbc.273.8.4790. [DOI] [PubMed] [Google Scholar]
- Diaz VM, Planaguma J, Thomson TM, Reventos J, Paciucci R. Tissue plasminogen activator is required for the growth, invasion, and angiogenesis of pancreatic tumor cells. Gastroenterology. 2002;122:806–819. doi: 10.1053/gast.2002.31885. [DOI] [PubMed] [Google Scholar]
- Balmain A. Cancer as a complex genetic trait: tumor susceptibility in humans and mouse models. Cell. 2002;108:145–152. doi: 10.1016/s0092-8674(02)00622-0. [DOI] [PubMed] [Google Scholar]
- Wagner M, Greten FR, Weber CK, Koschnick S, Mattfeldt T, Deppert W, Kern H, Adler G, Schmid RM. A murine tumor progression model for pancreatic cancer recapitulating the genetic alterations of the human disease. Genes Dev. 2001;15:286–293. doi: 10.1101/gad.184701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vila MR, Lloreta J, Real FX. Normal human pancreas cultures display functional ductal characteristics. Lab Invest. 1994;71:423–431. [PubMed] [Google Scholar]
- Sandgren EP, Luetteke NC, Palmiter RD, Brinster RL, Lee DC. Overexpression of TGF-α in transgenic mice: induction of epithelial hyperplasia, pancreatic metaplasia, and carcinoma of the breast. Cell. 1990;61:1121–1135. doi: 10.1016/0092-8674(90)90075-p. [DOI] [PubMed] [Google Scholar]
- Song SY, Gannon M, Washington MK, Scoggins CR, Meszoely IM, Goldenring JR, Marino CR, Sandgren EP, Coffey RJ, Jr, Wright CV, Leach SD. Expansion of Pdx1-expressing pancreatic epithelium and islet neogenesis in transgenic mice overexpressing transforming growth factor-α. Gastroenterology. 1999;117:1416–1426. doi: 10.1016/s0016-5085(99)70292-1. [DOI] [PubMed] [Google Scholar]
- Tevethia MJ, Bonneau RH, Griffith JW, Mylin L. A simian virus 40 large T-antigen segment containing amino acids 1 to 127 and expressed under the control of the rat elastase-1 promoter produces pancreatic acinar carcinomas in transgenic mice. J Virol. 1997;71:8157–8166. doi: 10.1128/jvi.71.11.8157-8166.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sandgren EP, Quaife CJ, Paulovich AG, Palmiter RD, Brinster RL. Pancreatic tumor pathogenesis reflects the causative genetic lesion. Proc Natl Acad Sci USA. 1991;88:93–97. doi: 10.1073/pnas.88.1.93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hajjar KA, Krishnan S. Annexin II: a mediator of the plasmin/plasminogen activator system. Trends Cardiovasc Med. 1999;9:128–138. doi: 10.1016/s1050-1738(99)00020-1. [DOI] [PubMed] [Google Scholar]
- Saillan-Barreau C, Clerc P, Adato M, Escrieut C, Vaysse N, Fourmy D, Dufresne M. Transgenic CCK-B/gastrin receptor mediates murine exocrine pancreatic secretion. Gastroenterology. 1998;115:988–996. doi: 10.1016/s0016-5085(98)70271-9. [DOI] [PubMed] [Google Scholar]
- Carmeliet P, Schoonjans L, Kieckens L, Ream B, Degen J, Bronson R, De Vos R, van den Oord JJ, Collen D, Mulligan RC. Physiological consequences of loss of plasminogen activator gene function in mice. Nature. 1994;368:419–424. doi: 10.1038/368419a0. [DOI] [PubMed] [Google Scholar]
- Scoggins CR, Meszoely IM, Wada M, Means AL, Yang L, Leach SD. p53-dependent acinar cell apoptosis triggers epithelial proliferation in duct-ligated murine pancreas. Am J Physiol. 2000;279:G827–G836. doi: 10.1152/ajpgi.2000.279.4.G827. [DOI] [PubMed] [Google Scholar]
- Montaner S, Sodhi A, Molinolo A, Bugge TH, Sawai ET, He Y, Li Y, Ray PE, Gutkind JS. Endothelial infection with KSHV genes in vivo reveals that vGPCR initiates Kaposi’s sarcomagenesis and can promote the tumorigenic potential of viral latent genes. Cancer Cell. 2003;3:23–36. doi: 10.1016/s1535-6108(02)00237-4. [DOI] [PubMed] [Google Scholar]
- Kaplan EL, Meier P. Nonparametric estimation from incomplete observations. J Am Stat Assoc. 1958;53:457–481. [Google Scholar]
- Peto G. The calculation and interpretation of survival curves. Buyse M, Staquet M, Sylvester R, editors. Oxford University Press,; Oxford: Cancer Clinical Trials, Methods and Practice. 1984:pp 361–380. [Google Scholar]
- Breslow N. Comparison of survival curves. Buyse M, Staquet M, Sylvester R, editors. Oxford University Press,; Oxford: Cancer Clinical Trials, Methods and Practice. 1984:pp 381–406. [Google Scholar]
- Clerc P, Leung-Theung-Long S, Wang TC, Dockray GJ, Bouisson M, Delisle MB, Vaysse N, Pradayrol L, Fourmy D, Dufresne M. Expression of CCK2 receptors in the murine pancreas: proliferation, transdifferentiation of acinar cells, and neoplasia. Gastroenterology. 2002;122:428–437. doi: 10.1053/gast.2002.30984. [DOI] [PubMed] [Google Scholar]
- Hajjar KA. Cellular receptors in the regulation of plasmin generation. Thromb Haemost. 1995;74:294–301. [PubMed] [Google Scholar]
- Redlitz A, Plow EF. Receptors for plasminogen and t-PA: an update. Baillieres Clin Haematol. 1995;8:313–327. doi: 10.1016/s0950-3536(05)80270-7. [DOI] [PubMed] [Google Scholar]
- Hruban RH, Yeo CJ, Kern SE. Pancreatic cancer. Scriver CR, Beaudet AL, Sly WS, Valle D, editors. McGraw-Hill,; New York: The Metabolic and Molecular Basis of Inherited Disease. 2001:pp 1077–1090. [Google Scholar]
- Schaeffer BK, Terhune PG, Longnecker DS. Pancreatic carcinomas of acinar and mixed acinar/ductal phenotypes in Ela-1-myc transgenic mice do not contain c-K-ras mutations. Am J Pathol. 1994;145:696–701. [PMC free article] [PubMed] [Google Scholar]
- Seufferlein T, Van Lint J, Liptay S, Adler G, Schmid RM. Transforming growth factor-α activates Ha-Ras in human pancreatic cancer cells with Ki-ras mutations. Gastroenterology. 1999;116:1441–1452. doi: 10.1016/s0016-5085(99)70509-3. [DOI] [PubMed] [Google Scholar]
- Hamad NM, Elconin JH, Karnoub AE, Bai W, Rich JN, Abraham RT, Der CJ, Counter CM. Distinct requirements for Ras oncogenesis in human versus mouse cells. Genes Dev. 2002;16:2045–2057. doi: 10.1101/gad.993902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kunz C, Pebler S, Otte J, von der Ahe D. Differential regulation of plasminogen activator and inhibitor gene transcription by the tumor suppressor p53. Nucleic Acids Res. 1995;23:3710–3717. doi: 10.1093/nar/23.18.3710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crawford HC, Scoggins CR, Washington MK, Matrisian LM, Leach SD. Matrix metalloproteinase-7 is expressed by pancreatic cancer precursors and regulates acinar-to-ductal metaplasia in exocrine pancreas. J Clin Invest. 2002;109:1437–1444. doi: 10.1172/JCI15051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moennikes O, Buchmann A, Ott T, Willecke1 K, Schwarz M. The effect of connexin32 null mutation on hepatocarcinogenesis in different mouse strains. Carcinogenesis. 1999;20:1379–1382. doi: 10.1093/carcin/20.7.1379. [DOI] [PubMed] [Google Scholar]
- Dietrich WF, Lander ES, Smith JS, Moser AR, Gould KA, Luongo C, Borenstein N, Dove W. Genetic identification of Mom-1, a major modifier locus affecting Min-induced intestinal neoplasia in the mouse. Cell. 1993;75:631–639. doi: 10.1016/0092-8674(93)90484-8. [DOI] [PubMed] [Google Scholar]
- Sigmund CD. Viewpoint: are studies in genetically altered mice out of control? Arterioscler Thromb Vasc Biol. 2000;20:1425–1429. doi: 10.1161/01.atv.20.6.1425. [DOI] [PubMed] [Google Scholar]
- Vishwanatha JK, Chiang Y, Kumble KD, Hollingsworth MA, Pour PM. Enhanced expression of annexin II in human pancreatic carcinoma cells and primary pancreatic cancers. Carcinogenesis. 1993;14:2575–2579. doi: 10.1093/carcin/14.12.2575. [DOI] [PubMed] [Google Scholar]
- Kumble KD, Hirota M, Pour PM, Vishwanatha JK. Enhanced levels of annexins in pancreatic carcinoma cells of Syrian hamsters and their intrapancreatic allografts. Cancer Res. 1992;52:163–167. [PubMed] [Google Scholar]
- Ling Q, Jacovina AT, Deora A, Febbraio M, Simantov R, Silverstein RL, Hempstead B, Mark WH, Hajjar KA. Annexin II regulates fibrin homeostasis and neoangiogenesis in vivo. J Clin Invest. 2004;113:38–48. doi: 10.1172/JCI200419684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roda O, Valero ML, Peiro S, Andreu D, Real FX, Navarro P. New insights into the tPA-annexin A2 interaction: is annexin A2 Cys8 the sole requirement for this association? J Biol Chem. 2003;278:5702–5709. doi: 10.1074/jbc.M207605200. [DOI] [PubMed] [Google Scholar]