Abstract
CD1 molecules are expressed by antigen-presenting cells such as dendritic cells and mediate primary immune responses to lipids and glycolipids which have been shown to be expressed by various tumors. Glycolipids are expressed by melanoma cells but, despite their immunogenicity, no efficient spontaneous immune responses are elicited. As IL-10 has previously been shown to down-regulate CD1a on dendritic cells and is known to be expressed by various melanoma cell lines, we investigated if melanoma-derived IL-10 could down-regulate CD1 molecule expression on dendritic cells as a possible way to circumvent immune recognition. We found that CD1a, CD1b, CD1c, and CD1d were significantly down-regulated on dendritic cells in metastatic (n = 10) but not in primary melanoma lesions (n = 10). We further detected significantly higher IL-10 protein levels in metastatic than in primary melanomas. Moreover, supernatants from metastatic melanomas were significantly more effective in down-regulating CD1 molecules on dendritic cells than supernatants from primary melanoma cultures. This effect was blocked using a neutralizing IL-10 antibody in a dose dependent manner. Our findings suggest that metastatic but not primary melanomas can down-regulate CD1 molecules on infiltrating dendritic cells by secreting IL-10 which may represent a novel way to escape the immune response directed against the tumor.
CD1 molecules are cell surface glycoproteins and represent a non-classical, non-polymorphic antigen-presenting system which is distantly related to major histocompatibility complex (MHC) class I and class II molecules. While MHC molecules form complexes with peptides, the CD1 system presents lipids and glycolipids of self and foreign origin.1–7 Two groups of CD1 molecules can be distinguished in humans based on sequence homologies, function and expression patterns: CD1a, CD1b, and CD1c are preferentially expressed by antigen-presenting cells (APC) including dendritic cells (DC) and constitute group 1. Group 2 consists exclusively of CD1d which is expressed on cells of the hematopoietic lineage.8 DC are professional antigen-presenting cells which express high levels of MHC molecules, the classical antigen-presenting system, but also the entire repertoire of CD1 molecules, thus being able to initiate immune responses to lipid/glycolipid antigens.9–11
In contrast to MHC-restricted T cells, CD1-restricted T cells recognize foreign lipids which are highly expressed on the surface of bacteria and eukaryotic parasites. Several studies have demonstrated that CD1-restricted T cells play an important role in the immune responses directed against Mycobacterium tuberculosis by mediating cytotoxicity against CD1-positive target cells presenting mycobacterial glycolipids.12–14 In addition, CD1 molecules have been shown to be involved in immune responses directed against tumors which express neuraminic acid containing glycolipids called gangliosides.15–18 Immunotherapy approaches using ganglioside vaccines were therefore used in several cancers, including melanoma.19–24
Since IL-10 has been shown to down-regulate CD1a on monocyte-derived DC12 and is produced by melanoma cells,25,26 we hypothesized that melanoma may use CD1 down-regulation on APC as a way to impede an immune response directed against the tumor by compromising glycolipid presentation to CD1-restricted T cells. Here, we show down-regulation of CD1 molecules in metastatic but not primary melanoma. We further demonstrate a key role of melanoma secreted IL-10 in mediating this effect.
Materials and Methods
Cell Cultures
Freshly isolated melanoma cells were isolated as previously described and cultured until they reached confluence (first passage).27 Cells were then detached using trypsin and subcultured (second passage). Melanoma cells were grown in 75-cm2 culture flasks (Becton Dickinson, Basel, Switzerland) as monolayer cultures in complete RPMI containing 5 mmol/L glutamine, 1 mmol/L sodium pyruvate, 1% penicillin/streptomycin, and 10% heat-inactivated fetal calf serum (FCS) (all from Gibco, Basel, Switzerland).
Monocyte-derived DC were generated from peripheral blood mononuclear cells (PBMC) isolated from buffy coats by density gradient centrifugation using Ficoll-Paque (Amersham Pharmacia Biotech, Uppsala, Sweden) as previously described with minor modifications.28,29 Briefly, 50 × 106 PBMC were incubated in 10-cm Petri dishes (Becton Dickinson) in complete RPMI at 37°C and 5% CO2 for 45 minutes. Non-adherent cells were removed by washing twice with phosphate-buffered saline (PBS) and the adherent cells were further cultured in complete RPMI containing 800 IU/ml GM-CSF (Leucomax, Sandoz-Wander Pharma SA, Bern, Switzerland) and 1000 IU/ml IL-4 (R&D Systems, Abingdon, United Kingdom). Fresh medium supplemented with 800 IU/ml GM-CSF and 1000 IU/ml IL-4 was added after 3 and 5 days. The purity of monocytes was assessed by flow cytometry by measuring the percentage of CD14+ cells using a monoclonal antibody (Becton Dickinson) and ranged from 79% to 94% (mean 84.5%) of total cells in culture. Cells were analyzed at different maturation days by flow cytometry as indicated.
Immunohistochemistry
Representative 7-μm serial cryostat sections from tissue samples of normal skin (n = 10), primary (n = 10), and metastatic (n = 10) melanoma lesions were examined for the expression of CD1 molecules, HLA-DR, and IL-10 using the alkaline phosphatase anti-alkaline phosphatase (APAAP) technique as previously described.30 Each sample was obtained from a different patient. The numbers of CD1a, b, c, d- and HLA-DR-positive DC in serial sections was assessed by calculating the mean cell number counted in five different fields (original magnification, ×400). Thereafter, the ratio between CD1- and HLA-DR-positive cells was calculated to correct for the density of infiltrating dendritic cells. DC were defined as large mononuclear cells with dendrites being strongly HLA-DR positive as previously described.14,30
The antibodies used were against CD1a (IgG2a) (Dako, Zug, Switzerland), CD1b (BCD1b3.1.6, IgG1) kindly provided by Dr. S. A. Porcelli (Harvard Medical School, Boston, MA, USA), CD1c (IgG1) (Immunotech, Marseilles, France), CD1d (IgG1) (Biosource International, Camarillo, CA, USA), CD1d (51.1.3, IgG2b), kindly provided by Dr. S. P. Balk (Harvard Medical School), HLA-DR (Becton Dickinson) (all at 10 μg/ml), and IL-10 (5 μg/ml) (R&D Systems). Sections were counterstained using hematoxylin.
Immunofluorescence
Cryosections were fixed in 2% paraformaldehyde (Sigma, Buchs, Switzerland) and then washed with PBS, pH 7.4. Sections were pre-incubated with normal rabbit serum (Dako). Slides were thereafter incubated with an antibody against any of the CD1 molecules at 10 μg/ml for 2 hours at room temperature in the dark. After washing with PBS, sections were incubated with a fluorescein isothiocynate (FITC)-conjugated rabbit anti-mouse antibody (dilution 1:30) (Dako). After washing with PBS, slides were incubated with normal mouse serum (5% in PBS) for 20 minutes and thereafter incubated with a phycoerythrin (PE)-conjugated antibody against HLA-DR (dilution 1:50) for 2 hours at room temperature in the dark. All steps were performed at room temperature. For examination of CD1/HLA-DR co-expression, confocal microscopy was performed using a Leica DM-IRBE microscope (Leica, Glattbrugg, Switzerland).
Flow Cytometry Analysis
105 monocyte-derived DC or DC isolated from the tumor were double-stained with PE-conjugated antibodies against CD1a (IgG2a) (Dako), CD1b (IgG1) (Becton Dickinson), CD1c (IgG1) (Immunotech), CD1d (51.1.3, IgG2b) kindly provided by Dr. S. P. Balk (Harvard Medical School), and HLA-DR (FITC-conjugated) (Becton Dickinson). In some experiments DC were also stained with the following PE or FITC-conjugated monoclonal antibodies: anti-CD11a (IgG2a), anti-CD11b (IgG1), anti-CD11c (IgG1), anti-CD14 (IgG2a), anti-CD80 (IgG1), anti-CD83 (IgG1), and anti-CD86 (IgG1) (all from Becton Dickinson). Each antibody was diluted in PBS containing 2% FCS at a concentration of 10 μg/ml and cells were stained at 4°C for 30 minutes in the dark. The corresponding isotype-matched antibodies were used as negative controls (all from Becton Dickinson). Flow cytometry was performed using a FACSCalibur, and data were subsequently analyzed using CellQuest (both from Becton Dickinson).
Neutralization of IL-10 in Cell Culture Supernatants
Supernatants were prepared by culturing 106 melanoma cells or normal fibroblasts in 25-cm2 flasks (Becton Dickinson) with 10 ml of complete RPMI. Supernatants were collected after 24 hours, centrifuged to remove cells and debris, and then stored at −80°C as previously described.31
Cell culture supernatants from primary and metastatic melanoma cell lines, as well as from melanoma cells 12 hours after isolation from the tumor were pre-incubated for 1 hour at 4°C with 0, 0.1, 1, and 10 μg/ml of a monoclonal neutralizing anti-IL-10 antibody (R&D Systems). An irrelevant antibody of the same isotype (IgG2b) (R&D Systems) was used as negative control at 10 μg/ml.
IL-10 Detection in Cell Culture Supernatants
Cell culture supernatants were prepared by culturing 106 melanoma cells or normal fibroblasts in 25-cm2 flasks with 10 ml of complete RPMI. IL-10 in the supernatants was measured using a Quantikine HS Human IL-10 ELISA kit from R&D Systems following the manufacturer’s guidelines. The detection limit was less than 3.9 pg/ml.
Statistical Analysis
Data were statistically analyzed using the Student’s t-test and results were considered significant when P < 0.05.
Results
CD1 Expression Is Down-Regulated in Metastatic Compared to Primary Melanoma in Vivo
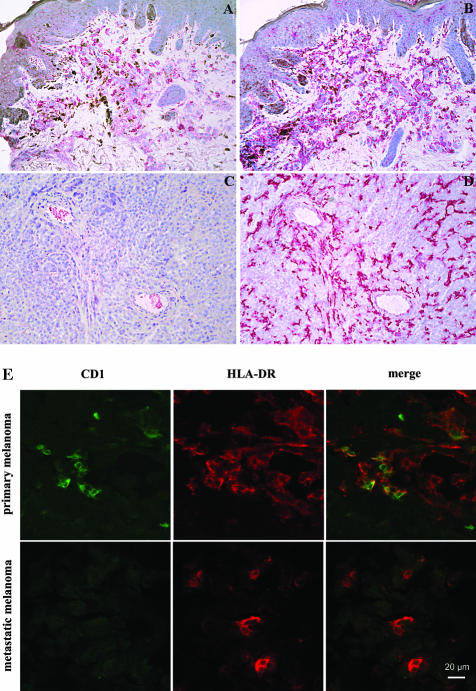
Expression of CD1 molecules and HLA-DR was studied by immunohistochemical staining of serial cryosections of primary (n = 10) and metastatic (n = 10) melanoma biopsies as well as normal skin samples (n = 10) from healthy donors. Each sample was obtained from a different donor. In normal skin, CD1a-positive epidermal Langerhans cells and some scattered CD1a, b, c, d-positive dermal DC were observed mainly in the papillary dermis and in a perivascular localization as previously described (data not shown).32 Primary melanoma lesions showed numerous large dendritic mononuclear cells staining positive for CD1 a, b, c, d located in the dermal intra- and peritumoral infiltrate. Figure 1A shows CD1c and is representative for CD1 molecule expression in primary melanoma sections examined. Staining of serial primary melanoma sections showed HLA-DR expression by large mononuclear dendritic cells (Figure 1B). In metastatic melanoma sections, CD1a, b, c, d expression by DC was consistently reduced and often undetectable. Figure 1C shows a representative CD1c staining of metastatic melanoma which is representative for CD1 molecule expression in metastatic lesions. In contrast, HLA-DR was still strongly expressed by large mononuclear cells with dendritic processes (Figure 1D).
Figure 1.
In situ expression of CD1 molecules and HLA-DR in primary and metastatic melanoma. Immunohistochemistry was performed to study in vivo CD1 expression in primary (A and B) and metastatic (C and D) melanoma lesions using the APAAP technique. Sections were counterstained with hematoxylin. Both A and C show CD1c expression. HLA-DR expression in primary and metastatic lesions is shown in B and D. Pictures shown are representative stainings. Original magnification, ×200. E: Confocal microscopy shows co-expression of CD1 molecules and HLA-DR in primary but not metastatic melanoma. To demonstrate co-expression of CD1 molecules and HLA-DR on melanoma cells as well as CD1 down-regulation in metastatic melanoma lesions, confocal microscopy was performed. Pictures show CD1b (green), HLA-DR (red), and CD1b/HLA-DR co-expression (yellow) and are representative stainings. Top row, primary melanoma; bottom row, metastatic melanoma. The horizontal bar indicates 20 μm.
To further support these data, cell suspensions from metastatic melanoma lesions and a primary melanoma sample were prepared. Flow cytometry analysis showed that the percentages of CD1a, b, c, d/HLA-DRbright double-positive cells in the primary melanoma was higher than in the metastatic melanoma tested (data not shown).
To further demonstrate in situ CD1 down-regulation on HLA-DR-positive cells in metastatic but not in primary melanoma, confocal microscopy was performed. Figure 1E shows representative CD1b/HLA-DR double-immunofluorescence stainings of primary and metastatic melanoma cryosections. CD1b-positive (green) and CD1b/HLA-DR double-positive cells (yellow) could be detected in primary melanomas (top row) whereas CD1b was down-regulated in metastatic melanoma lesions (bottom row). HLA-DR expression levels were comparable in both primary and metastatic melanoma (red). These data support the immunohistochemical results showing that CD1 molecules, but not HLA-DR, are down-regulated in metastatic melanoma lesions compared to primary melanoma.
To quantify CD1a, b, c, d and HLA-DR down-regulation in primary (n = 10) and metastatic (n = 10) melanoma lesions, the numbers of CD1 and HLA-DR expressing cells in serial sections were assessed by counting the positive cells in five different microscopic fields (original magnification, ×400). The mean values were thereafter calculated as previously described.14 To correct for the density of infiltrating DC, the ratios between CD1a, b, c- and HLA-DR-positive cells were thereafter calculated for each sample. A significantly lower mean number of CD1a, b, c, d-positive cells relative to HLA-DR-positive cells was observed in metastatic compared to primary lesions. The CD1/HLA-DR ratios in metastatic compared to primary melanomas were as follows: for CD1a, 0.02 ± 0.03 versus 0.4 ± 0.1 (P < 0.001); for CD1b, 0.04 ± 0.05 versus 0.3 ± 0.1 (P < 0.001); for CD1c, 0.1 ± 0.1 versus 0.5 ± 0.2 (P < 0.001); and for CD1d, 0.1 ± 0.1 versus 0.2 ± 0.05 (P < 0.001) (Figure 2). The mean numbers of HLA-DR-positive cells in primary (35.1 ± 5.4) and metastatic (31 ± 4.5) melanoma did not differ significantly (P = 0.335) (data not shown).
Figure 2.
To assess the number of cells expressing CD1a, b, c, d in melanoma lesions, primary (n = 10) and metastatic (n = 10) melanoma serial cryosections were stained using the respective antibodies and the APAAP technique. The number of CD1a, b, c and HLA-DR-positive cells was thereafter counted in five different microscopic fields (original magnification, ×400) and the mean values were calculated. Results are given as the ratio between CD1a, b, c- and HLA-DR-positive cells to correct for the density of infiltrating DC. *, P < 0.05, in relation to the primary melanoma group.
Overexpression of IL-10 in Metastatic Melanoma
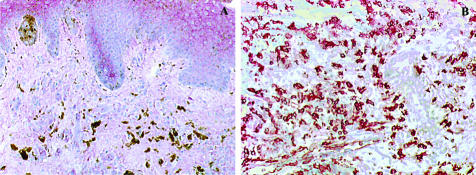
IL-10 is believed to be involved in the suppression of an anti-tumor immune response and could be detected at high levels in the blood of metastatic melanoma patients compared to primary melanoma patients and healthy individuals.26,33 To study IL-10 expression in melanoma lesions in situ, immunohistochemistry was performed on primary as well as metastatic melanoma cryosections. Faint IL-10 staining was detected in suprabasal keratinocytes but not in melanoma cells of primary melanoma sections (n = 10). In contrast, metastatic lesions (n = 10) showed strong IL-10 staining in most of the melanoma cells. Figure 3, A and B, show representative IL-10 staining in primary and metastatic melanoma lesions, respectively. Some melanin-producing melanoma cells can be seen in Figure A (brown color).
Figure 3.
IL-10 expression in primary and metastatic melanoma lesions. To study in situ IL-10 expression, cryosections of primary (A) and metastatic (B) tumor lesions were stained using a monoclonal IL-10 antibody and the APAAP technique. Pictures shown are representative stainings. Original magnification, ×200.
To further compare IL-10 protein secretion by primary and metastatic melanoma cells, we performed enzyme-linked immunosorbent assays (ELISA) using supernatants from primary (n = 3) and metastatic (n = 21) melanoma cell lines, as well as from fibroblast cultures (n = 3). Metastatic melanoma cell lines were divided into first passage metastatic melanoma cell cultures (n = 3), short-term (two passages, n = 7), and long-term cultures (> two passages, n = 11). Primary melanoma and fibroblast cultures were in continuous culture for two passages. The highest IL-10 concentrations could be detected in metastatic melanoma supernatants of first passage cultures (91 to 306 pg/ml, mean 199.6 ± 87.8 pg/ml) and were significantly higher (P < 0.05) than in second passage metastatic melanoma cultures (0.1 to 31.6 pg/ml, mean 14.6 ± 11.2 pg/ml), and in long-term metastatic melanoma cultures (0 to 0.6 pg/ml, mean 0.1 ± 0.2 pg/ml, Figure 4). No IL-10 could be detected in the supernatants of primary melanoma cell lines. The difference in IL-10 levels between short-term primary and both short-term and long-term metastatic melanoma cell line supernatants were statistically significant (P < 0.05). The significant decrease of IL-10 from passage one to passage two (P < 0.05) and from passage two to long-term cultures (P < 0.05) suggests that cultured melanoma cells gradually loose the capacity to produce IL-10. Only low amounts of IL-10 were found in the supernatants from fibroblast cell cultures (0 to 0.2 pg/ml, mean 0.1 ± 0.1 pg/ml) (Figure 4).
Figure 4.
High IL-10 concentrations can be detected in supernatants from metastatic melanoma cultures. To measure IL-10 concentrations in supernatants from primary as well as metastatic melanoma cell cultures (1, 2, and >2 passages) and normal fibroblasts, 106 cells in 25-cm2 culture flasks were cultured in 10 ml of complete RPMI for 24 hours. Supernatants were thereafter centrifuged to remove debris and the IL-10 concentration was assessed by ELISA. MM, metastatic melanoma; PM, primary melanoma; P, passage. *, P < 0.05, in relation to first passage metastatic melanoma IL-10 concentrations in supernatants; **, P < 0.05, in relation to second passage metastatic melanoma IL-10 concentrations in supernatants.
Dose-Dependent Down-Regulation of CD1 Molecules on Monocyte-Derived Dendritic Cells by IL-10
Dendritic cells in the blood of tumor patients have been shown to be reduced in numbers and function.34 To rule out the possibility that monocyte-derived DC from patients suffering from metastatic melanoma have inherently altered CD1 expression, monocyte-derived DC were generated from PBMC of melanoma patients (n = 3) as well as healthy individuals (n = 3) and CD1 expression was compared after 5 days using flow cytometry. No significant difference in CD1a, b and c expression levels could be observed (data not shown). Down-regulation of CD1d molecules by IL-10 treatment was not assessed since FCS-cultured monocyte-derived DC bear little or no CD1d, as previously described.32 It has previously been demonstrated that IL-10 added at the beginning of the culture inhibits expression of CD1 molecules on GMCSF/IL-4-treated monocytes and that IL-10 down-regulates CD1a on monocyte-derived DC.12,35 We therefore studied whether IL-10 can down-regulate CD1a, CD1b, CD1c expression on our monocyte-derived DC preparations. After 3 days in culture, DC exhibited the features of immature DC: the majority of cells was in suspension and showed a dendritic phenotype. Flow cytometry analysis showed high expression levels of HLA-DR (98.3 ± 0.6% of cells; mean fluorescence intensity (MFI), 326.4 ± 35.1), CD11a (97.7 ± 1.3%; MFI, 214.2 ± 31.1), CD11b (95.5 ± 1%; MFI, 69.9 ± 12.1) and CD11c (98.8 ± 0.5%; MFI, 156.8 ± 26.5). CD80 and CD86 expression could be detected on 23.8 ± 9.7% and 48.6 ± 11.5% of DC, respectively. Only 29.4 ± 2.5% of DC expressed CD14, which was a 4.7-fold decrease compared to fresh monocytes (MFI 124.1 ± 18.0 versus 588.3 ± 23.1, respectively). A certain degree of spontaneous maturation was observed on 5.7 ± 2.3% of cells that expressed CD83 molecules. At this time point the majority of the cells stained positive for CD1 molecules: CD1a, 87.8 ± 6.8%; CD1b, 88.0 ± 5.3%; and CD1c, 95.7 ± 2.2% (data not shown).
Cells were further cultured for 48 and 72 hours in the presence of 800 IU/ml GM-CSF and 1000 IU/ml IL-4 with or without IL-10 (1, 10, and 100 ng/ml). Flow cytometry analysis of DC cultured in the absence of IL-10 showed that the MFI of HLA-DR (469.7 ± 12.9), CD11a (288.9 ± 8.4), CD11b (81.0 ± 9.8), and CD11c (210.6 ± 11.1) was further up-regulated after 72 hours although no significant changes in the percentage of positive cells was observed. Similarly, CD80 and CD86 expression levels were further increased and could be detected on 33.5 ± 9.5% and 77.8 ± 4.9% of DC, respectively. In contrast, CD14 was no longer detectable.
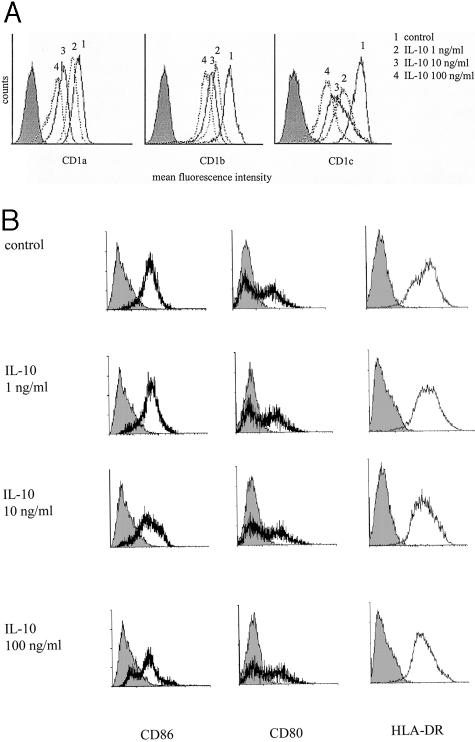
A dose-dependent down-regulation of CD1a, b, c expression on IL-10-treated, monocyte-derived DC could be observed after 48 hours (data not shown) and was even stronger after 72 hours (Figure 5A). Relative to DC cultured in the absence of IL-10, the decrease of the MFI of IL-10-treated, monocyte-derived DC using 1, 10, and 100 ng/ml was as follows: CD1a: 15% (P = 0.260), 48% (P < 0.05), 80% (P < 0.01); CD1b: 34% (P < 0.01), 39% (P < 0.01), 64% (P < 0.01), and CD1c: 32% (P = 0.080), 46% (P < 0.01), 68% (P < 0.01).
Figure 5.
IL-10 down-regulates CD1 expression on monocyte-derived DC in a dose-dependent manner. Monocyte-derived DC were cultured for 3 days in the presence of IL-4 (1000 IU/ml) and GM-CSF (800 IU/ml). Monocyte-derived DC were thereafter cultured for another 72 hours in GM-CSF and IL-4 in the presence of various IL-10 concentrations (1, 10, and 100 ng/ml). A: Analysis of CD1a, b, c and CD86, CD80, and (B) HLA-DR expression by flow cytometry. Monocyte-derived DC cultured in presence of GM-CSF and IL-4 without IL-10 were used as positive control. Results are representative for three independent experiments. Filled histograms represent isotype controls.
Previous studies have shown that IL-10 treatment also affects co-stimulatory molecule expression by DC.36–38 We confirmed that IL-10-treated DC exhibited lower CD86 expression compared to untreated DC (Figure 5B). Compared to DC cultured for 72 hours in the absence of IL-10, the decrease of CD86 MFI of IL-10-treated, monocyte-derived DC using 1, 10, 100 ng/ml was as follows: 17% (P = 0.660), 56% (P < 0.05), and 63% (P < 0.024). In our hands CD80 and HLA-DR expression were not significantly affected by IL-10 treatment as previously described.35,36 At the highest IL-10 concentration a slight decrease of the HLA-DR MFI was observed compared to untreated DC (447.3 ± 10.2), which was not statistically significant. Similarly, no effect on CD14, CD11a, CD11b, and CD11c expression levels could be observed (data not shown).
Melanoma Cell Supernatant Down-Regulates CD1 Expression on Monocyte-Derived Dendritic Cells
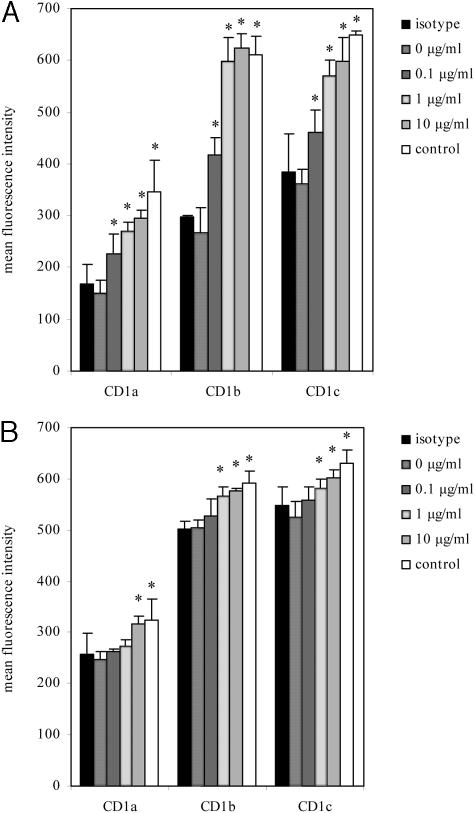
To study whether metastatic melanoma supernatant has the ability to down-regulate CD1 molecules, monocyte-derived DC were cultured in the presence of various dilutions of melanoma cell supernatants. After cultivation of monocyte-derived DC in the presence of 800 IU/ml GM-CSF and 1000 IU/ml IL-4 for 3 days, cells were incubated for another 3 days in the presence of different dilutions (10%, 50%, and 100%) of supernatants from first passage metastatic melanoma cultures. GM-CSF and IL-4 concentrations were thereafter adjusted to the initial concentrations (800 IU/ml GM-CSF and 1000 IU/ml IL-4). DC cultured in the absence of melanoma supernatants were used as negative controls. CD1a, b, c expression levels were analyzed by flow cytometry. Applying different dilutions of melanoma supernatants resulted in a dose-dependent decrease of CD1a, b, and c. Moreover, CD1 expression levels were also dependent on the IL-10 levels detected in the different first passage metastatic melanoma supernatants. Upon replacing 10%, 50%, and 100% of the DC culture medium with supernatant from the culture which showed the highest IL-10 concentration (306 pg/ml, cell culture 000630, see Figure 4), the decrease of the MFI of CD1 molecules relative to the negative control was as follows: CD1a: 37%, 57%, 81%; CD1b: 40%, 55%, 63%; and CD1c: 28%, 47%, 58%. CD1 down-regulation was significant for CD1a, b, c at every concentration (P < 0.05) (Figure 6A). When using supernatant from a first passage metastatic melanoma culture with a lower IL-10 concentration (91 pg/ml, cell culture 000515, see Figure 4), the decrease of the MFI was not as pronounced, but still evident: CD1a: 20%, 36%, 48%; CD1b: 13%, 32%, 41%; CD1c: 5%, 22%, 60%. CD1a, b, c down-regulation was significant at 50% and 100% supernatant concentration (P < 0.05) (Figure 6A). In addition, we also measured CD86, CD80, and HLA-DR expression using supernatant from cell line 000630. No significant differences in HLA-DR expression levels could be detected in supernatant-treated DC at any dilution compared to control DC. In contrast, CD86 and CD80 expression was significantly down-regulated by using 50% and 100% supernatant concentration: the decrease of the MFI using 50% and 100% of supernatant was 23 and 29% for CD86 and 18% and 24% for CD80 (P < 0.05) (Figure 6B).
Figure 6.
Effects of melanoma supernatants on CD1, CD86, and HLA-DR expression on monocyte-derived DC. A: Supernatants from first passage metastatic melanoma cultures with high (cell line 000630, 306 pg/ml IL-10) and low (cell line 000515, 91 pg/ml IL-10) IL-10 concentrations were used to study the effects of different supernatant dilutions on CD1 expression on GM-CSF/IL-4-generated DC. After 3 days, 0%, 10%, 50%, and 100% of the medium was replaced with melanoma supernatants from the melanoma cell culture indicated and incubated for another 3 days. CD1a, b, and c expression was thereafter assessed by flow cytometry. B: CD86, CD80, and HLA-DR expression on DC after replacement of 0%, 10%, 50%, and 100% of DC culture medium with melanoma supernatant (cell line 000630). C: Supernatants from different melanoma and fibroblast cell cultures were used to study the effects on CD1 expression on DC using flow cytometry. Fifty percent of the DC culture medium was replaced with first passage metastatic melanoma (culture 000630), second passage metastatic melanoma (culture 000621), primary melanoma (culture 990220), and fibroblast cell culture supernatants. CD1 expression was thereafter measured using flow cytometry. Results are shown as the mean MFI of three independent experiments. Vertical bars represent the standard deviations of the means. *, P < 0.05, in relation to monocyte-derived DC cultured in the absence of cell culture supernatant. MM, metastatic melanoma; PM, primary melanoma; fibro, fibroblasts; P, passage.
We next compared the ability of different melanoma culture supernatants from different melanoma cell lines to down-regulate CD1 expression. When replacing 50% of DC culture medium with supernatants from first and second passage metastatic melanoma cultures as well as second passage primary melanoma and fibroblast cultures, a significant CD1a, b, c down-regulation could be observed on DC cultured in the presence of first and second passage metastatic melanoma supernatants compared to DC cultured in the absence of supernatant (P < 0.05). The reduction relative to untreated DC was CD1a, 56.3%; CD1b, 56.1%; and CD1c, 44.2% for first passage melanoma cells and for second passage melanoma cells: CD1a, 45.4%; CD1b, 37.3%; and CD1c, 32.9%. No significant down-regulation could be observed when culturing DC in the presence of primary melanoma or fibroblast supernatants (Figure 6C).
CD1 Down-Regulation by Melanoma Cell Culture Supernatant is IL-10 Mediated
To show that down-regulation of CD1 molecules on monocyte-derived DC is IL-10 mediated, supernatants from first passage metastatic melanoma cell cultures were pre-incubated with a neutralizing anti-IL-10 antibody (0, 0.1, 1, and 10 μg/ml) or the corresponding isotype control (IgG2b, 10 μg/ml) for 1 hour at 4°C. Thereafter, monocyte-derived DC were incubated in the presence of 50% antibody-treated melanoma supernatant, 800 IU/ml GM-CSF, and 1000 IU/ml IL-4 for another 3 days. Monocyte-derived DC cultured in the absence of melanoma supernatant were used as negative control. Figure 7A shows that neutralization of IL-10 in cell culture supernatants with high IL-10 concentrations (cell line 000630, 306 pg/ml IL-10) significantly reduced CD1a, b, and c down-regulation in a dose-dependent manner compared to untreated and isotype-treated control supernatants (P < 0.05). No significant inhibition of CD1a, b, c down-regulation could be observed in samples incubated with isotype-treated supernatants compared to untreated supernatants. Ten μg/ml neutralizing antibody inhibited CD1a, b, c down-regulation, resulting in CD1 expression levels which were not significantly different from cells cultured in the absence of metastatic melanoma supernatant. When using melanoma cell culture supernatant with a lower IL-10 concentration (cell line 000622, 26.9 pg/ml IL-10), untreated and isotype-treated supernatants induced a lower CD1 down-regulation compared to the respective supernatants with higher IL-10 concentrations. However, when using 10 μg/ml neutralizing antibody the reduction of CD1 down-regulation relative to untreated supernatant was still significant (P < 0.05) (Figure 7B).
Figure 7.
CD1 down-regulation on monocyte-derived DC is IL-10 mediated. Monocyte-derived DC were generated in the presence of IL-4 (1000 IU/ml) and GM-CSF (800 IU/ml). After 3 days, 50% of the culture medium was replaced with different melanoma cell culture supernatants containing various IL-10 concentrations (A: Cell culture 000630, 306 pg/ml IL-10. B: Cell culture 000622, 26.9 pg/ml IL-10), which have been pre-incubated with a range of concentrations of an IL-10-neutralizing antibody (0, 0.1, 1, and 10 μg/ml) or a corresponding isotype-matched control antibody (IgG2b, 10 μg/ml). DC cultured in the absence of melanoma supernatant were used as control. CD1a, b, and c expression was measured thereafter by flow cytometry. Results are shown as the mean MFI of three independent experiments. *, P < 0.05, in relation to monocyte-derived DC cultured in the presence of 50% cell culture supernatant in the absence of an antibody.
Discussion
We show that CD1 molecules are down-regulated on dendritic cells in metastatic melanoma compared to primary melanoma lesions. This effect was mediated by IL-10, highly expressed in metastatic but not in primary melanoma lesions.
There is an increasing body of evidence showing that the presentation of non-peptide antigens such as lipids and glycolipids by CD1-positive DC to T cells plays an important role in the immune responses directed against cancer and microorganisms.3,13–16 Gangliosides are a family of ubiquitous membrane-associated acidic glycosphingolipids and can be found in most cell types, especially in the central nervous system.39 Ganglioside composition has been demonstrated to be altered in many tumors, including melanoma. Normal melanocytes express solely GM3 whereas melanoma cells overexpress various gangliosides such as GD3 and GM3, which have been shown to play a role in tumor growth and metastasis.40–44
CD1-mediated presentation of mycobacterial lipids and glycolipids to CD1-restricted T cells plays a central role in the immunity to Mycobacterium leprae and Mycobacterium tuberculosis.10,45,46 In leprosy, CD1 expression has been demonstrated to strongly correlate with the immune response directed against M. leprae and the clinical course of the disease. Strong induction of CD1a, b, and c expression on DC has been shown in leprosy skin lesions of the tuberculoid form and in granulomas with reversal reactions which are both associated with an active cellular immunity directed against M. leprae whereas no CD1 induction could be observed in the lepromatous form which lacks an effective cell-mediated immunity to the pathogen.14 APC from individuals infected with M. tuberculosis contain mycobacteria and present mycobacterial lipid antigens bound to CD1 molecules to CD1-restricted T cells which lyse the infected APC, resulting in the death of the bacteria. However, M. tuberculosis has been shown to be able to down-regulate CD1 on the surface of their host cells, circumventing immune recognition and thus leading to the persistence of intracellular infection.13,47 It has therefore been suggested that the unsatisfactory results in the treatment of tuberculosis obtained with BCG vaccination may be due to mycobacterium-induced impairment of the CD1 system.48
Similar to the role of CD1 molecule down-regulation in the inefficient immune response toward mycobacteria, we hypothesized that CD1 down-regulation might play a role in escaping the immune system in melanoma, as tumor expressed gangliosides have been shown to be immunogenic but do not elicit a clinically effective immune response.49–52 When CD1 expression was analyzed in melanoma lesions, we found significantly higher CD1a, b, c, d expression in primary compared to metastatic lesions in situ (Figures 1 and 2).
Reduced numbers and function of DC have been shown in the peripheral blood of cancer patients.34 Moreover, accumulation of immature macrophages, DC, and myeloid cells in the peripheral blood and subsequent inhibition of antigen-specific T cell responses have been demonstrated in tumor patients.53 To study whether monocyte-derived DC generated from the peripheral blood of melanoma patients have altered CD1 expression, we compared CD1 expression by monocyte-derived DC of melanoma patients to healthy individuals. No significant difference in CD1 expression was found. We therefore concluded that CD1 down-regulation on tumor-infiltrating DC was caused by local factors. Melanoma cells produce various soluble factors such as VEGF, TGF-β, and IL-10 which have been shown to have immunosuppressive effects.54–56 A particular relevant soluble factor is IL-10, since high IL-10 levels have been shown in the tumor tissue and the blood of metastatic melanoma patients compared to patients with primary melanoma lesions and healthy individuals.26,33 In addition, high tumor-derived IL-10 levels in progressing but not in regressing melanoma lesions after chemoimmunotherapy have been shown to induce tolerance in tumor-infiltrating DC.57 Furthermore, IL-10 has been shown to down-regulate CD1a on monocyte-derived DC in vitro.12 We therefore examined IL-10 levels in melanoma cells of primary and metastatic melanomas and found significant IL-10 overexpression in metastatic melanoma lesions compared to primary melanomas both in situ and in cell cultures (Figures 3 and 4). Even though we could not detect IL-10-producing primary melanoma cells in vivo, we cannot exclude the possibility of rare IL-10-secreting tumor cells in these primary lesions. If such cells would exist and IL-10-mediated immune escape is a valid mechanism in vivo, these cells might give rise to metastases and therefore be an important therapeutic target.
When monocyte-derived DC were cultured in the presence of melanoma cell culture supernatants derived from primary and metastatic tumors, a significant down-regulation of CD1a, b, c could be shown in the presence of metastatic, but not of primary, melanoma cell culture supernatants which could be blocked using an IL-10 neutralizing antibody, demonstrating that the effect was IL-10 mediated (Figures 5 to 7). Moreover, supernatants from metastatic melanoma cell lines containing high IL-10 levels could induce a stronger CD1a, b, c down-regulation than supernatant containing low IL-10 concentrations. Taken together, these data suggest an important role for IL-10 in the down-regulation of CD1 on DC in metastatic melanoma lesions. Although IL-10-mediated CD1 down-regulation has been shown to be involved in the immune escape of mycobacteria, this is the first report demonstrating a similar mechanism of potential relevance for an anti-cancer immune response.
IL-10 is a well-documented immunosuppressive cytokine being capable of reducing the expression of co-stimulatory and antigen-presenting molecules35,58,59 and therefore inhibiting the antigen-presenting capacity of DC.60,61 In addition, IL-10-treated DC can induce anergy in melanoma-specific CD8 T cells.62 Previous reports have also shown that IL-10 affects the expression of CD1 molecules on monocytes. Monocytes cultured in presence of GM-CSF, IL-4, and IL-10 give rise to CD1-negative DC35 and monocyte-derived DC cultured in the presence of IL-10 down-regulate CD1a.12 Here we extend these findings by showing that IL-10 can decrease the expression of CD1a, CD1b, and CD1c on monocyte-derived DC in a dose-dependent manner. However, IL-10 could not abolish CD1 expression completely, suggesting that DC are less sensitive to the effects of IL-10 than monocytes. Additional factors secreted by melanoma cells may also contribute to CD1 down-regulation. For example, VEGF has previously been shown to be secreted by various melanoma cell lines55 and has been demonstrated to induce CD1a down-regulation on monocyte-derived dendritic cells.55,63 VEGF has also been shown to down-regulate CD80 in monocyte-derived DC which might explain our observation that CD80 was down-regulated by melanoma supernatant but not by IL-10.64 Moreover, decreased CD1d surface expression levels in murine tumors in response to various cytokines, including IL-10 and interferon γ, have been shown to inhibit tumor cell lysis by NK cells.65
The central role of NF-κB in mediating the effects of IL-10 in immature DC has been elucidated recently. Reduced DNA binding and nuclear translocation of NF-κB in IL-10-pretreated DC, on stimulation with lipopolysaccharide or anti-CD40, have been shown to be mediated by inhibition of NF-κB through suppression of IκB activity. Moreover, Akt phosphorylation was blocked by IL-10, and inhibition of PI3-kinase resulted in suppression of Akt, IκB, and NF-κB activity. This suggests that IL-10 inhibits NF-κB through suppression of the PI3-kinase pathway which, in turn, leads to inhibition of IκB activity.66 In contrast, no phosphorylation of ERK2, SAPK/JNK, and p38 could be detected in immature DC on stimulation with IL-10.67
The gangliosides GD2, GD3, and GM2 have been shown to be expressed on the surface of melanoma cells and to be immunogenic in both mice and humans and have therefore been used as targets for active immunization with cancer vaccines resulting in an IgG and IgM response.50–52,68 Pre-existing anti-GM2 antibodies before vaccination or vaccination-induced anti-GM2 antibodies have been associated with a favorable diagnosis.52 Despite their immunogenicity, IgG and IgM autoantibody titers against these gangliosides in unvaccinated melanoma patients are either low or not detectable. Moreover, the incidence of spontaneous GM2 antibody production seems to be similar in unvaccinated stage III melanoma patients and healthy patients (5% to 10%), suggesting that growth of melanoma in skin and lymph nodes is not a strong stimulus for GM2 antibody production.49–52,69 This raises the question why no increased IgG and IgM titers can be found in patients with advanced melanoma before ganglioside vaccination. While several immune escape mechanisms including MHC loss or TAP deficiency have been reported (reviewed in70), IL-10-mediated down-regulation of CD1 molecules on the surface of tumor-infiltrating APC may offer an explanation for the lacking humoral immune response against tumor-associated glycolipid antigens observed in unvaccinated metastatic melanoma patients.
Taken together, our data show that melanoma-secreted IL-10 mediates down-regulation of CD1 molecules on dendritic cells. These data suggest that metastatic melanoma cells may use CD1 down-regulation as a way to escape the immune recognition and destruction.
Acknowledgments
We thank Dr. Mathias Höchli and Dr. Anne Greet Bittermann from the Laboratory of Electron Microscopy at the University of Zurich for their excellent technical assistance with the confocal microscope.
Footnotes
Address reprint requests to Frank O. Nestle, M.D., Department of Dermatology, University of Zurich Medical School, Gloriastrasse 31, CH-8091 Zurich, Switzerland. E-mail: nestle@derm.unizh.ch.
Supported by Ente Cassa di Risparmio di Firenze (G.G.) and the Swiss Cancer League and Robert Wenner Prize Foundation (F.O.N.).
G.G. and A.T.K. contributed equally to this work.
References
- Knowles RW, Bodmer WF. A monoclonal antibody recognizing a human thymus leukemia-like antigen associated with beta 2-microglobulin. Eur J Immunol. 1982;12:676–681. doi: 10.1002/eji.1830120810. [DOI] [PubMed] [Google Scholar]
- Terhorst C, van Agthoven A, LeClair K, Snow P, Reinherz E, Schlossman S. Biochemical studies of the human thymocyte cell-surface antigens T6, T9, and T10. Cell. 1981;23:771–780. doi: 10.1016/0092-8674(81)90441-4. [DOI] [PubMed] [Google Scholar]
- Grant EP, Degano M, Rosat JP, Stenger S, Modlin RL, Wilson IA, Porcelli SA, Brenner MB. Molecular recognition of lipid antigens by T cell receptors. J Exp Med. 1999;189:195–205. doi: 10.1084/jem.189.1.195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Porcelli SA, Segelke BW, Sugita M, Wilson IA, Brenner MB. The CD1 family of lipid antigen-presenting molecules. Immunol Today. 1998;19:362–368. doi: 10.1016/s0167-5699(98)01289-4. [DOI] [PubMed] [Google Scholar]
- Blumberg RS, Gerdes D, Chott A, Porcelli SA, Balk SP. Structure and function of the CD1 family of MHC-like cell surface proteins. Immunol Rev. 1995;147:5–29. doi: 10.1111/j.1600-065x.1995.tb00085.x. [DOI] [PubMed] [Google Scholar]
- Balk SP, Bleicher PA, Terhorst C. Isolation and characterization of a cDNA and gene coding for a fourth CD1 molecule. Proc Natl Acad Sci USA. 1989;86:252–256. doi: 10.1073/pnas.86.1.252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosat JP, Grant EP, Beckman EM, Dascher CC, Sieling PA, Frederique D, Modlin RL, Porcelli SA, Furlong ST, Brenner MB. CD1-restricted microbial lipid antigen-specific recognition found in the CD8+ alpha beta T cell pool. J Immunol. 1999;162:366–371. [PubMed] [Google Scholar]
- Dutronc Y, Porcelli SA. The CD1 family and T cell recognition of lipid antigens. Tissue Antigens. 2002;60:337–353. doi: 10.1034/j.1399-0039.2002.600501.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Banchereau J, Steinman RM. Dendritic cells and the control of immunity. Nature. 1998;392:245–252. doi: 10.1038/32588. [DOI] [PubMed] [Google Scholar]
- Porcelli SA, Modlin RL. The CD1 system: antigen-presenting molecules for T cell recognition of lipids and glycolipids. Annu Rev Immunol. 1999;17:297–329. doi: 10.1146/annurev.immunol.17.1.297. [DOI] [PubMed] [Google Scholar]
- Cella M, Sallusto F, Lanzavecchia A. Origin, maturation, and antigen-presenting function of dendritic cells. Curr Opin Immunol. 1997;9:10–16. doi: 10.1016/s0952-7915(97)80153-7. [DOI] [PubMed] [Google Scholar]
- Fortsch D, Rollinghoff M, Stenger S. IL-10 converts human dendritic cells into macrophage-like cells with increased antibacterial activity against virulent Mycobacterium tuberculosis. J Immunol. 2000;165:978–987. doi: 10.4049/jimmunol.165.2.978. [DOI] [PubMed] [Google Scholar]
- Stenger S, Modlin RL. T cell-mediated immunity to Mycobacterium tuberculosis. Curr Opin Microbiol. 1999;2:89–93. doi: 10.1016/s1369-5274(99)80015-0. [DOI] [PubMed] [Google Scholar]
- Sieling PA, Jullien D, Dahlem M, Tedder TF, Rea TH, Modlin RL, Porcelli SA. CD1 expression by dendritic cells in human leprosy lesions: correlation with effective host immunity. J Immunol. 1999;162:1851–1858. [PubMed] [Google Scholar]
- Toura I, Kawano T, Akutsu Y, Nakayama T, Ochiai T, Taniguchi M. Cutting edge: inhibition of experimental tumor metastasis by dendritic cells pulsed with alpha-galactosylceramide. J Immunol. 1999;163:2387–2391. [PubMed] [Google Scholar]
- Kawano T, Cui J, Koezuka Y, Toura I, Kaneko Y, Sato H, Kondo E, Harada M, Koseki H, Nakayama T, Tanaka Y, Taniguchi M. Natural killer-like nonspecific tumor cell lysis mediated by specific ligand-activated Valpha14 NKT cells. Proc Natl Acad Sci USA. 1998;95:5690–5693. doi: 10.1073/pnas.95.10.5690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi T, Johnson TD, Nishinaka Y, Morton DL, Irie RF. IgM anti-ganglioside antibodies induced by melanoma cell vaccine correlate with survival of melanoma patients. J Invest Dermatol. 1999;112:205–209. doi: 10.1046/j.1523-1747.1999.00493.x. [DOI] [PubMed] [Google Scholar]
- Livingston P. Ganglioside vaccines with emphasis on GM2. Semin Oncol. 1998;25:636–645. [PubMed] [Google Scholar]
- Livingston PO, Ragupathi G. Carbohydrate vaccines that induce antibodies against cancer: 2. Previous experience and future plans. Cancer Immunol Immunother. 1997;45:10–19. doi: 10.1007/s002620050395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carr A, Rodriguez E, Arango Mdel C, Camacho R, Osorio M, Gabri M, Carrillo G, Valdes Z, Bebelagua Y, Perez R, Fernandez LE. Immunotherapy of advanced breast cancer with a heterophilic ganglioside (NeuGcGM3) cancer vaccine. J Clin Oncol. 2003;21:1015–1021. doi: 10.1200/JCO.2003.02.124. [DOI] [PubMed] [Google Scholar]
- Fredman P, Hedberg K, Brezicka T. Gangliosides as therapeutic targets for cancer. BioDrugs. 2003;17:155–167. doi: 10.2165/00063030-200317030-00002. [DOI] [PubMed] [Google Scholar]
- Irie RF, Matsuki T, Morton DL. Human monoclonal antibody to ganglioside GM2 for melanoma treatment. Lancet. 1989;1:786–787. doi: 10.1016/s0140-6736(89)92606-8. [DOI] [PubMed] [Google Scholar]
- Tai T, Cahan LD, Tsuchida T, Saxton RE, Irie RF, Morton DL. Immunogenicity of melanoma-associated gangliosides in cancer patients. Int J Cancer. 1985;35:607–612. doi: 10.1002/ijc.2910350507. [DOI] [PubMed] [Google Scholar]
- Kirkwood JM, Mascari RA, Edington HD, Rabkin MS, Day RS, Whiteside TL, Vlock DR, Shipe-Spotloe JM. Analysis of therapeutic and immunologic effects of R(24) anti-GD3 monoclonal antibody in 37 patients with metastatic melanoma. Cancer. 2000;88:2693–2702. [PubMed] [Google Scholar]
- Dummer W, Bastian BC, Ernst N, Schanzle C, Schwaaf A, Brocker EB. Interleukin-10 production in malignant melanoma: preferential detection of IL-10-secreting tumor cells in metastatic lesions. Int J Cancer. 1996;66:607–610. doi: 10.1002/(SICI)1097-0215(19960529)66:5<607::AID-IJC4>3.0.CO;2-X. [DOI] [PubMed] [Google Scholar]
- Nemunaitis J, Fong T, Shabe P, Martineau D, Ando D. Comparison of serum interleukin-10 (IL-10) levels between normal volunteers and patients with advanced melanoma. Cancer Invest. 2001;19:239–247. doi: 10.1081/cnv-100102550. [DOI] [PubMed] [Google Scholar]
- Yue FY, Dummer R, Geertsen R, Hofbauer G, Laine E, Manolio S, Burg G. Interleukin-10 is a growth factor for human melanoma cells and down-regulates HLA class-I, HLA class-II, and ICAM-1 molecules. Int J Cancer. 1997;71:630–637. doi: 10.1002/(sici)1097-0215(19970516)71:4<630::aid-ijc20>3.0.co;2-e. [DOI] [PubMed] [Google Scholar]
- Jonuleit H, Kuhn U, Muller G, Steinbrink K, Paragnik L, Schmitt E, Knop J, Enk AH. Pro-inflammatory cytokines and prostaglandins induce maturation of potent immunostimulatory dendritic cells under fetal calf serum-free conditions. Eur J Immunol. 1997;27:3135–3142. doi: 10.1002/eji.1830271209. [DOI] [PubMed] [Google Scholar]
- Nestle FO, Alijagic S, Gilliet M, Sun Y, Grabbe S, Dummer R, Burg G, Schadendorf D. Vaccination of melanoma patients with peptide- or tumor lysate-pulsed dendritic cells. Nat Med. 1998;4:328–332. doi: 10.1038/nm0398-328. [DOI] [PubMed] [Google Scholar]
- Nestle FO, Burg G, Fah J, Wrone-Smith T, Nickoloff BJ. Human sunlight-induced basal cell carcinoma-associated dendritic cells are deficient in T cell co-stimulatory molecules and are impaired as antigen-presenting cells. Am J Pathol. 1997;150:641–651. [PMC free article] [PubMed] [Google Scholar]
- Kiertscher SM, Luo J, Dubinett SM, Roth MD. Tumors promote altered maturation and early apoptosis of monocyte-derived dendritic cells. J Immunol. 2000;164:1269–1276. doi: 10.4049/jimmunol.164.3.1269. [DOI] [PubMed] [Google Scholar]
- Gerlini G, Hefti HP, Kleinhans M, Nickoloff BJ, Burg G, Nestle FO. Cd1d is expressed on dermal dendritic cells and monocyte-derived dendritic cells. J Invest Dermatol. 2001;117:576–582. doi: 10.1046/j.0022-202x.2001.01458.x. [DOI] [PubMed] [Google Scholar]
- Dummer W, Becker JC, Schwaaf A, Leverkus M, Moll T, Brocker EB. Elevated serum levels of interleukin-10 in patients with metastatic malignant melanoma. Melanoma Res. 1995;5:67–68. doi: 10.1097/00008390-199502000-00008. [DOI] [PubMed] [Google Scholar]
- Almand B, Resser JR, Lindman B, Nadaf S, Clark JI, Kwon ED, Carbone DP, Gabrilovich DI. Clinical significance of defective dendritic cell differentiation in cancer. Clin Cancer Res. 2000;6:1755–1766. [PubMed] [Google Scholar]
- Thomssen H, Kahan M, Londei M. Differential effects of interleukin-10 on the expression of HLA class II and CD1 molecules induced by granulocyte/macrophage colony-stimulating factor/interleukin-4. Eur J Immunol. 1995;25:2465–2470. doi: 10.1002/eji.1830250909. [DOI] [PubMed] [Google Scholar]
- Buelens C, Willems F, Delvaux A, Pierard G, Delville JP, Velu T, Goldman M. Interleukin-10 differentially regulates B7–1 (CD80) and B7–2 (CD86) expression on human peripheral blood dendritic cells. Eur J Immunol. 1995;25:2668–2672. doi: 10.1002/eji.1830250940. [DOI] [PubMed] [Google Scholar]
- Brown RD, Pope B, Murray A, Esdale W, Sze DM, Gibson J, Ho PJ, Hart D, Joshua D. Dendritic cells from patients with myeloma are numerically normal but functionally defective as they fail to up-regulate CD80 (B7–1) expression after huCD40LT stimulation because of inhibition by transforming growth factor-beta1 and interleukin-10. Blood. 2001;98:2992–2998. doi: 10.1182/blood.v98.10.2992. [DOI] [PubMed] [Google Scholar]
- Corinti S, Albanesi C, la Sala A, Pastore S, Girolomoni G. Regulatory activity of autocrine IL-10 on dendritic cell functions. J Immunol. 2001;166:4312–4318. doi: 10.4049/jimmunol.166.7.4312. [DOI] [PubMed] [Google Scholar]
- Sandhoff K, van Echten G. Ganglioside metabolism: enzymology, topology, and regulation. Prog Brain Res. 1994;101:17–29. doi: 10.1016/s0079-6123(08)61937-8. [DOI] [PubMed] [Google Scholar]
- Portoukalian J, Zwingelstein G, Dore JF. Lipid composition of human malignant melanoma tumors at various levels of malignant growth. Eur J Biochem. 1979;94:19–23. doi: 10.1111/j.1432-1033.1979.tb12866.x. [DOI] [PubMed] [Google Scholar]
- Tsuchida T, Saxton RE, Morton DL, Irie RF. Gangliosides of human melanoma. J Natl Cancer Inst. 1987;78:45–54. doi: 10.1093/jnci/78.1.45. [DOI] [PubMed] [Google Scholar]
- Shurin GV, Shurin MR, Bykovskaia S, Shogan J, Lotze MT, Barksdale EM., Jr Neuroblastoma-derived gangliosides inhibit dendritic cell generation and function. Cancer Res. 2001;61:363–369. [PubMed] [Google Scholar]
- Grayson G, Ladisch S. Immunosuppression by human gangliosides: II. Carbohydrate structure and inhibition of human NK activity. Cell Immunol. 1992;139:18–29. doi: 10.1016/0008-8749(92)90096-8. [DOI] [PubMed] [Google Scholar]
- Birkle S, Zeng G, Gao L, Yu RK, Aubry J. Role of tumor-associated gangliosides in cancer progression. Biochimie. 2003;85:455–463. doi: 10.1016/s0300-9084(03)00006-3. [DOI] [PubMed] [Google Scholar]
- Beckman EM, Porcelli SA, Morita CT, Behar SM, Furlong ST, Brenner MB. Recognition of a lipid antigen by CD1-restricted alpha beta+ T cells. Nature. 1994;372:691–694. doi: 10.1038/372691a0. [DOI] [PubMed] [Google Scholar]
- Porcelli S, Morita CT, Brenner MB. CD1b restricts the response of human CD4–8-T lymphocytes to a microbial antigen. Nature. 1992;360:593–597. doi: 10.1038/360593a0. [DOI] [PubMed] [Google Scholar]
- Stenger S, Mazzaccaro RJ, Uyemura K, Cho S, Barnes PF, Rosat JP, Sette A, Brenner MB, Porcelli SA, Bloom BR, Modlin RL. Differential effects of cytolytic T cell subsets on intracellular infection. Science. 1997;276:1684–1687. doi: 10.1126/science.276.5319.1684. [DOI] [PubMed] [Google Scholar]
- Prete SP, Giuliani A, Iona E, Fattorini L, Orefici G, Franzese O, Bonmassar E, Graziani G. Bacillus Calmette-Guerin down-regulates CD1b induction by granulocyte-macrophage colony stimulating factor in human peripheral blood monocytes. J Chemother. 2001;13:52–58. doi: 10.1179/joc.2001.13.1.52. [DOI] [PubMed] [Google Scholar]
- Houghton AN, Taormina MC, Ikeda H, Watanabe T, Oettgen HF, Old LJ. Serological survey of normal humans for natural antibody to cell surface antigens of melanoma. Proc Natl Acad Sci USA. 1980;77:4260–4264. doi: 10.1073/pnas.77.7.4260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Livingston PO, Natoli EJ, Calves MJ, Stockert E, Oettgen HF, Old LJ. Vaccines containing purified GM2 ganglioside elicit GM2 antibodies in melanoma patients. Proc Natl Acad Sci USA. 1987;84:2911–2915. doi: 10.1073/pnas.84.9.2911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Portoukalian J, Carrel S, Dore JF, Rumke P. Humoral immune response in disease-free advanced melanoma patients after vaccination with melanoma-associated gangliosides: EORTC Cooperative Melanoma Group. Int J Cancer. 1991;49:893–899. doi: 10.1002/ijc.2910490616. [DOI] [PubMed] [Google Scholar]
- Livingston PO, Wong GY, Adluri S, Tao Y, Padavan M, Parente R, Hanlon C, Calves MJ, Helling F, Ritter G. Improved survival in stage III melanoma patients with GM2 antibodies: a randomized trial of adjuvant vaccination with GM2 ganglioside. J Clin Oncol. 1994;12:1036–1044. doi: 10.1200/JCO.1994.12.5.1036. [DOI] [PubMed] [Google Scholar]
- Almand B, Clark JI, Nikitina E, van Beynen J, English NR, Knight SC, Carbone DP, Gabrilovich DI. Increased production of immature myeloid cells in cancer patients: a mechanism of immunosuppression in cancer. J Immunol. 2001;166:678–689. doi: 10.4049/jimmunol.166.1.678. [DOI] [PubMed] [Google Scholar]
- Chen Q, Daniel V, Maher DW, Hersey P. Production of IL-10 by melanoma cells: examination of its role in immunosuppression mediated by melanoma. Int J Cancer. 1994;56:755–760. doi: 10.1002/ijc.2910560524. [DOI] [PubMed] [Google Scholar]
- Westphal JR, Van’t Hullenaar R, Peek R, Willems RW, Crickard K, Crickard U, Askaa J, Clemmensen I, Ruiter DJ, De Waal RM. Angiogenic balance in human melanoma: expression of VEGF, bFGF, IL-8, PDGF, and angiostatin in relation to vascular density of xenografts in vivo. Int J Cancer. 2000;86:768–776. doi: 10.1002/(sici)1097-0215(20000615)86:6<768::aid-ijc3>3.0.co;2-e. [DOI] [PubMed] [Google Scholar]
- Krasagakis K, Tholke D, Farthmann B, Eberle J, Mansmann U, Orfanos CE. Elevated plasma levels of transforming growth factor (TGF)-beta1 and TGF-beta2 in patients with disseminated malignant melanoma. Br J Cancer. 1998;77:1492–1494. doi: 10.1038/bjc.1998.245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Enk AH, Jonuleit H, Saloga J, Knop J. Dendritic cells as mediators of tumor-induced tolerance in metastatic melanoma. Int J Cancer. 1997;73:309–316. doi: 10.1002/(sici)1097-0215(19971104)73:3<309::aid-ijc1>3.0.co;2-3. [DOI] [PubMed] [Google Scholar]
- Koch F, Stanzl U, Jennewein P, Janke K, Heufler C, Kampgen E, Romani N, Schuler G. High level IL-12 production by murine dendritic cells: upregulation via MHC class II and CD40 molecules and downregulation by IL-4 and IL-10. J Exp Med. 1996;184:741–746. doi: 10.1084/jem.184.2.741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang CH, Furue M, Tamaki K. B7–1 expression of Langerhans cells is up-regulated by proinflammatory cytokines, and is down-regulated by interferon-gamma or by interleukin-10. Eur J Immunol. 1995;25:394–398. doi: 10.1002/eji.1830250213. [DOI] [PubMed] [Google Scholar]
- Enk AH, Angeloni VL, Udey MC, Katz SI. Inhibition of Langerhans cell antigen-presenting function by IL-10: a role for IL-10 in induction of tolerance. J Immunol. 1993;151:2390–2398. [PubMed] [Google Scholar]
- Caux C, Massacrier C, Vanbervliet B, Barthelemy C, Liu YJ, Banchereau J. Interleukin 10 inhibits T cell alloreaction induced by human dendritic cells. Int Immunol. 1994;6:1177–1185. doi: 10.1093/intimm/6.8.1177. [DOI] [PubMed] [Google Scholar]
- Steinbrink K, Jonuleit H, Muller G, Schuler G, Knop J, Enk AH. Interleukin-10-treated human dendritic cells induce a melanoma-antigen-specific anergy in CD8(+) T cells resulting in a failure to lyse tumor cells. Blood. 1999;93:1634–1642. [PubMed] [Google Scholar]
- Fernandez Pujol B, Lucibello FC, Zuzarte M, Lutjens P, Muller R, Havemann K. Dendritic cells derived from peripheral monocytes express endothelial markers and in the presence of angiogenic growth factors differentiate into endothelial-like cells. Eur J Cell Biol. 2001;80:99–110. doi: 10.1078/0171-9335-00136. [DOI] [PubMed] [Google Scholar]
- Takahashi A, Kono K, Ichihara F, Sugai H, Fujii H, Matsumoto Y. Vascular endothelial growth factor inhibits maturation of dendritic cells induced by lipopolysaccharide, but not by proinflammatory cytokines. Cancer Immunol Immunother. 2004;53:543–550. doi: 10.1007/s00262-003-0466-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fiedler T, Walter W, Reichert TE, Maeurer MJ. Regulation of CD1d expression by murine tumor cells: escape from immunosurveillance or alternate target molecules? Int J Cancer. 2002;98:389–397. doi: 10.1002/ijc.10141. [DOI] [PubMed] [Google Scholar]
- Bhattacharyya S, Sen P, Wallet M, Long B, Baldwin AS, Tisch R. Immunoregulation of dendritic cells by IL-10 is mediated through suppression of the PI3K/Akt pathway and I{kappa}B kinase activity. Blood. 2004 doi: 10.1182/blood-2003-12-4302. [DOI] [PubMed] [Google Scholar]
- Sato K, Nagayama H, Tadokoro K, Juji T, Takahashi TA. Extracellular signal-regulated kinase, stress-activated protein kinase/c-Jun N-terminal kinase, and p38mapk are involved in IL-10-mediated selective repression of TNF-alpha-induced activation and maturation of human peripheral blood monocyte-derived dendritic cells. J Immunol. 1999;162:3865–3872. [PubMed] [Google Scholar]
- Livingston PO, Ritter G, Srivastava P, Padavan M, Calves MJ, Oettgen HF, Old LJ. Characterization of IgG and IgM antibodies induced in melanoma patients by immunization with purified GM2 ganglioside. Cancer Res. 1989;49:7045–7050. [PubMed] [Google Scholar]
- Portoukalian J, Zwingelstein G, Dore JF, Bourgoin JJ. Studies of a ganglioside fraction extracted from human malignant melanoma. Biochimie. 1976;58:1285–1287. doi: 10.1016/s0300-9084(76)80130-7. [DOI] [PubMed] [Google Scholar]
- Geertsen R, Hofbauer G, Kamarashev J, Yue FY, Dummer R. Immune escape mechanisms in malignant melanoma. Int J Mol Med. 1999;3:49–57. doi: 10.3892/ijmm.3.1.49. [DOI] [PubMed] [Google Scholar]