Abstract
Objective
The authors tested the hypothesis that p42/44- (ERK-1/2) and/or p38-mitogen-activated protein kinases (MAPK) are in vivo regulatory elements in the platelet-activating factor (PAF) activated signaling cascade that stimulates microvascular hyperpermeability.
Methods
FITC-dextran 70 was used as the macromolecular tracer for microvascular permeability in the mouse mesenteric fat tissue. Interstitial integrated optical intensity (IOI) was used as an index of permeability.
Results
An application of 10−7 MPAF increased IOI from 23.1 ± 3.6 to 70.8 ± 7.4 (mean ± SEM). Inhibition of ERK-1/2 with 3 μM and 30 μM AG126 reduced IOI to 32.3 ± 2.5. Similarly, inhibition of p38-MAPK with 6 nM, 60 nM and 600 nM SB203580 lowered IOI to 29.1 ± 2.4.
Conclusions
The results demonstrate that ERK-1/2 and p38MAPK participate in the signaling cascade that regulates PAF-induced microvascular hyperpermeability in vivo.
Keywords: ERK-1/2, microvascular permeability, p38MAPK, p42/44MAPK, platelet-activating factor
Hyperpermeability is an important hallmark of the inflammatory reaction that characterizes vascular disease and wound healing. However, the signaling pathways that regulate microvascular permeability are only partially understood (1). We and other investigators have documented in vivo and in vitro that endothelial nitric oxide synthase (eNOS, through production of nitric oxide [NO]), protein kinase C (PKC), PKB/akt, and phospholipase C (PLC) are elements of the signaling cascade triggered by platelet-activating factor (PAF), vascular endothelial growth factor (VEGF), and other pro-inflammatory agonists (2-18,40).
We have shown in vitro that p42/44 (ERK-1/2) and p38MAPK (mitogen-activated protein kinases) mediate VEGF-induced hyperpermeability in human umbilical vein endothelial cells (HUVEC) (2,3,19). The purpose of this study was to test the hypothesis that p42/44 and/or p38 MAPK regulates the PAF-stimulated signaling cascade, leading to microvascular hyperpermeability in vivo.
MATERIALS AND METHODS
Animals
Thirty-five male wild-type C57BL-6J mice (Japan Biological Material Center, Saitama, Japan), 10–15 weeks old, 25–35 g in body weight, were used in this study. The study was approved by the Animal Care and Use Committee at the University of Tokyo, School of Medicine, and complied with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Intravital Microscopy and Digital Image Analysis
A U-LH 100HG microscope (Olympus Optical CO, LTD, Japan) was used for intravital microscopy. The image was transferred through a CCD camera C6790-81 (Hamamatsu Photonics, Japan) to a computer with digital image analysis software “U4469-01 Argus/Hisca Software” (Hamamatsu Photonics) for analysis.
Microvascular permeability was measured as interstitial integrated optical intensity (IOI) (20,21), which was calculated by integrating gray-scale values of 512 × 512-pixel windows at each selected area. Five areas in the fat tissue interphase of the mesentery were randomly selected. The same areas were recorded and analyzed after the experimental interventions. Net IOI, used in the evaluation of permeability changes, was defined as the difference between experimental IOI value and average of baseline IOI value.
Surgical Technique
Under general anesthesia (50–60 mg/kg of sodium pentobarbital, IP), the anterior neck and abdomen were prepared with an electronic shaver. The mouse was placed on a heating pad (temperature was maintained at 37°C) under the dissecting microscope in the supine position, and the forelegs, hind legs, and jaws were mildly stretched to facilitate the operation. Tracheotomy was performed with an Intramedic polyethylene tube PE-50 (Becton Dickinson, Japan) to ensure clear airway passage. The left jugular vein was cannulated with PE-10 for the administration of supplemental doses of anesthetic.
After full midline incision, the mouse was placed in the prone position on the saline-filled mesentery chamber with the mesentery gently splayed to fit in the chamber. The mesenteric preparation was super-perfused with bicarbonate buffer (adjusted to pH 7.4 and continuously equilibrated with 95% nitrogen/5% carbon dioxide).
Experimental Design
A period of 60 min was allowed for stabilization. Subsequently, FITC-dx 70 was administered intravenously 15 min before baseline measurements. Pretreatment with topical application of MAPK inhibitors (AG126 or SB203580) was initiated 10 min before PAF application and maintained throughout the experiment. PAF was topically applied to the mesentery fat tissue for 3 min. The intravital microscopic image was captured by a TV camera and transferred to the computer. Microvascular transport was measured using a computer-assisted digital image analysis system showing the image and the value of IOI. Baseline data was acquired every 10 min three times and experimental data was recorded every 5 min for 60 min.
Chemicals and Agents
All chemicals were purchased from Sigma-Aldrich (St Louis, MO) unless otherwise noted. PAF (dl-α-phosphatidylcholine, β-acetyl-γ-o-hexadecyl) at 10−7 M was used as a pro-inflammatory agonist. Preliminary data demonstrated that 10−7 M PAF is an effective dose to induce permeability changes in the mouse mesentery. This concentration of PAF is in agreement with our earlier data in the hamster cheek pouch (22). FITC-dx 70 (fluorescein isothiocyanatedextran 70 kDa) served as the macromolecular tracer for microvascular permeability. AG126 (Tyrphostin AG 126, 3-hydroxy-4-nitrobenzylidene) at 3 and 30 μM was used to inhibit ERK-1/2. SB203580 [4-(4-fluorophenyl)-2-(4-methylsulfinyl-phenyl)-5-(4-pyridyl)-1h-imidazole] at 6, 60, and 600 nM was used to selectively block p38MAPK.
The composition of bicarbonate buffer (pH 7.4) was (in mM) 131.9 NaCl, 4.7 KCl, 2.0 CaCl2, 1.2 MgSO4, and 18.0 NaHCO3. PAF, AG126, and SB203580 were dissolved in dimethyl sulfoxide (DMSO) to stock concentrations. Immediately before topical application, a PAF aliquot was diluted to 10−7 M with bicarbonate buffer containing 1.5% bovine serum albumin. FITC-dextran 70 was injected initially intravenously as a bolus (100 mg/kg), and then infused continuously throughout the experiment at the rate of 0.15 mg/kg/min via a jugular vein. The tracer solution was prepared by diluting FITC-dextran 70 in 0.9% NaCl. Bovine serum albumin (BSA; 1.5% solution) was also dissolved in 0.9% NaCl.
Statistical Analysis
IOI data are presented as means ± SEM for each time-point. Time-related changes in IOI and differences of IOI between the control group and experimental group with MAPK inhibitors were analyzed with one-way ANOVA (SPSS), followed by Tukey's test. Statistical significance was accepted at p < .05.
RESULTS
PAF-Induced Hyperpermeability
Topical application of 10−7 MPAF induced large and significant changes in microvascular permeability relative to control, as indicated by analysis of IOI (Figure 1). An increase in permeability was detected in the first sample (5 min) after PAF and remained elevated for the remainder of the experiment. PAF at 10−7 M increased IOI from 23.1 ± 3.6 to 70.8 ± 7.4 (mean ± SEM).
Figure 1.
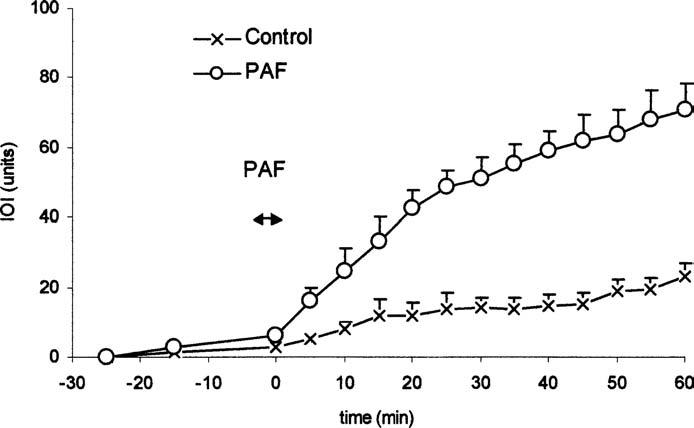
PAF (10−7 M) increases microvascular permeability in mesenteric tissue–fat interphase. The panel shows the time course of microvascular permeability as assessed by changes in net IOI. PAF significantly increased IOI relative to control. Data are means ± SEM (n = 5 for control and PAF experiments; p < .05 at t = 5 min; p < .001 thereafter).
ERK-1/2 and p38MAPK Regulate PAF-Induced Microvascular Permeability
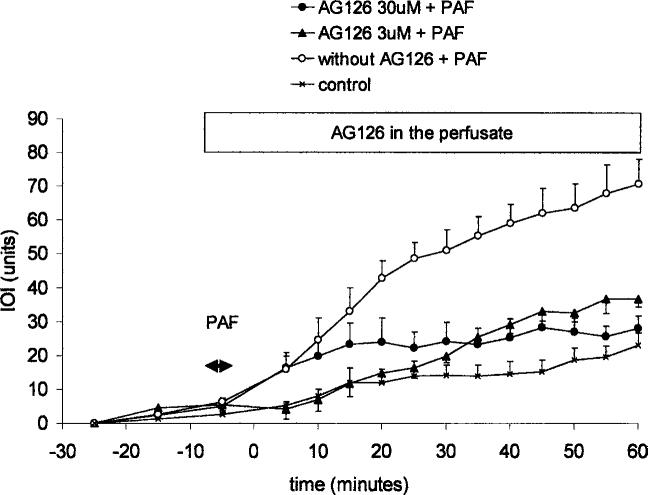
The selective ERK-1/2 inhibitor AG126 at 3 and 30 μM significantly attenuated the microvascular hyperpermeability induced by 10−7 M PAF to 32.3 ± 2.5 (Figure 2). There was no significant difference in the degree of attenuation between AG126 at 3 and 30 μM. AG126 at 0.3 μM had no inhibitory effect. The statistical analysis of net IOI of PAF-induced permeability changes with or without p42/44 MAPK inhibitor yielded the following values for significance: no AG vs. AG 3 μM: p = .003; no AG vs. AG 30 μM: p = .001; AG 3 μM vs. AG 30 μM: p = .792
Figure 2.
Inhibition of p42/p44 MAPK, with AG126, reduces PAF-induced hyperpermeability. The panel displays the time course of IOI as an index of microvascular permeability to FITC-dextran 70. Inhibitor was applied topically via superfusate, starting 10 min before topical application of 10−7 M PAF and continued throughout the remainder of the experiment. AG126 significantly inhibited PAF-induced hyperpermeability. Data are means ± SEM (n = 5 for PAF and each AG126 concentration; p < .01 at t ≥ 20 min).
SB203580, a selective inhibitor of p38MAPK, significantly reduced the increase in IOI induced by PAF at 10−7 M to 29.1 ± 2.4 (Figure 3). All concentrations of SB203580, 6, 60, and 600 nM, produced similar reductions in PAF-stimulated hyperpermeability (p > .05), while 0.6 nM SB203580 had no effect. The statistical analysis of net IOI of PAF-induced permeability changes with or without SB203580 (p38 MAPK inhibitor) yielded the following values for significance: no SB vs. SB 6 nM: p = .006; no SB vs. SB 60 nM: p = .011; no SB vs. SB 600 nM: p = .009; SB 60 nM vs. 6nM: P = .953; SB 600 nM vs. 6 μM: p = .826; SB 60 nM vs. 600 nM: p = .821.
Figure 3.
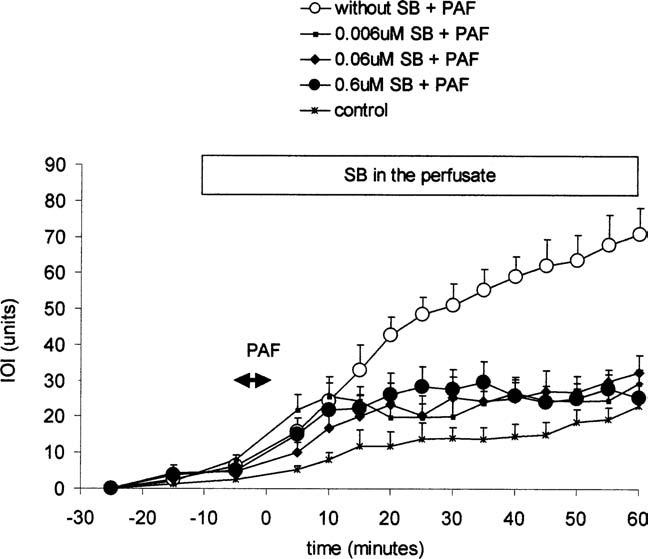
Inhibition of p38-MAPK, with SB203580, reduces PAF-induced hyperpermeability. The panel displays the time course of IOI as an index of microvascular permeability to FITC-dextran 70. Inhibitor was applied topically via superfusate, starting 10 min before topical application of 10−7 M PAF and continued throughout the remainder of the experiment. SB203580 significantly inhibited PAF-induced hyperpermeability. Data are means ± SEM (n = 5 for PAF and each SB203580 concentration; p < .01 at t ≥ 20 min).
DISCUSSION
Our experiments demonstrate in vivo that ERK-1/2 and p38 MAPK are operational elements of the signaling pathways that regulate PAF-stimulated microvascular hyperpermeability. The conclusion rests on evidence from other laboratories supporting the concept that AG126 (23) and SB203580 (24,25) are specific inhibitors of ERK-1/2 and p38MAPK, respectively. Our observations and conclusions are consistent with our previous in vitro study where inhibition of either ERK-1/2 or p38MAPK significantly reduced VEGF-induced hyperpermeability in HUVEC (2,3). These studies, in vivo and in vitro, provide strong evidence that both ERK-1/2 and p38 MAPK are involved in the signal transduction regulating microvascular permeability.
The basic elements of the signaling pathways initiating the control of microvascular permeability have been identified through work from several laboratories, including our own. Elements that have been confirmed in vitro and in vivo include specific receptors (usually G-protein coupled receptors), PLC, PKB/akt, PKC, eNOS, ERK-1/2, and p38MAPK (1-12,14,15,17-19,40). The precise sequence of signaling still remains to be elucidated. Whether all these elements work in series or some are arranged in parallel is an open question. While there is consensus that PKB/akt phosphorylates eNOS at serine 1179, causing release of NO (18), there are presently two opposing views regarding the sequence of biochemical events involving MAPK and eNOS. One view is that the activity of eNOS releases NO, which increases the production of cGMP by soluble guanylyl cyclase and activates the cGMP-dependent kinase, PKG. In turn, PKG activates MAPK. Thus, MAPK may be considered a signal element downstream of eNOS. The opposing view is that MAPK are upstream of eNOS. According to this view, MAPK are activated by an agonist (such as bradykinin) and phosphorylate eNOS (26). Whether MAPK are upstream or downstream of eNOS or represent a parallel pathway as well as the nature of possible cross-talks (2) in the regulation of microvascular permeability remains unresolved. However, it is noteworthy that inhibition of ERK-1/2 decreases baseline permeability in HUVEC (19). In this regard, ERK-1/2 shares this capability with PLC (27) and myosin light-chain kinase (28). Even though this regulatory capacity is difficult to demonstrate in vivo (7), it is plausible and reasonable to envision a signaling pathway that can lower baseline microvascular permeability. MAPK seem to be a signaling element involved in maintenance of baseline microvascular integrity.
In addition to controlling microvascular permeability, the MAPK superfamily has roles in cellular differentiation and proliferation (29), apoptosis (30-33), and expression of the proinflammatory phenotype in innate immune cells, including monocytes, endothelial cells, and neutrophils (34-39). The answer to the crucial question regarding how the MAPK pathway manages and codes the signaling information to precisely deliver signals to the downstream molecules and to finally elicit specific biological responses remains open to active investigation.
Footnotes
This work was supported in part by NIH grant 5RO1 HL70634. The authors are grateful to Hiroyuki Koyama, Masatake Katsu, and Taketo Saito for helpful discussions and Atsuko Onozuka for her kind help.
REFERENCES
- 1.Yuan SY. Signal transduction pathways in enhanced microvascular permeability. Microcirculation. 2000;7:395–403. [PubMed] [Google Scholar]
- 2.Breslin JW, Pappas PJ, Cerveira JJ, Hobson RW, II, Durán WN. VEGF increases endothelial permeability by separate signaling pathways involving ERK-1/2 and nitric oxide. Am J Physiol: Heart Circ Physiol. 2003;284:H92–H100. doi: 10.1152/ajpheart.00330.2002. [DOI] [PubMed] [Google Scholar]
- 3.Lal BK, Varma S, Pappas PJ, Hobson RW, II, Durán WN. VEGF increases permeability of the endothelial cell monolayer by activation of PKB/akt, endothelial nitric-oxide synthase, and MAP kinase pathways. Microvasc Res. 2001;62:252–262. doi: 10.1006/mvre.2001.2338. [DOI] [PubMed] [Google Scholar]
- 4.Ramírez MM, Quardt SM, Kim D, Oshiro H, Minnicozzi M, Durán WN. Platelet activating factor modulates microvascular permeability through nitric oxide synthesis. Microvasc Res. 1995;50:223–234. doi: 10.1006/mvre.1995.1055. [DOI] [PubMed] [Google Scholar]
- 5.Kobayashi I, Kim D, Hobson RW, II, Durán WN. Platelet-activating factor modulates microvascular transport by stimulation of protein kinase C. Am J Physiol. 1994;266:H1214–H1220. doi: 10.1152/ajpheart.1994.266.3.H1214. Heart Circ. Physiol 35. [DOI] [PubMed] [Google Scholar]
- 6.Ramírez MM, Kim DD, Durán WN. Protein kinase C modulates microvascular permeability through nitric oxide synthase. Am J Physiol. 1996;271:H1702–H1705. doi: 10.1152/ajpheart.1996.271.4.H1702. Heart Circ Physiol 40. [DOI] [PubMed] [Google Scholar]
- 7.Kim DD, Ramírez MM, Durán WN. Platelet-activating factor modulates microvascular dynamics through phospholipase C in the hamster cheek pouch. Microvasc Res. 2000;59:7–13. doi: 10.1006/mvre.1999.2195. [DOI] [PubMed] [Google Scholar]
- 8.Durán WN, Dillon PK. Acute microcircula-tory effects of platelet-activating factor. J Lipid Mediat. 1990;2(Suppl):S215–S227. [PubMed] [Google Scholar]
- 9.Huang QB, Wu M, Meininger C, Kelly K, Yuan Y. Neutrophil-dependent augmentation of PAF-induced vasoconstriction and albumin flux in coronary arterioles. Am J Physiol. 1998;275:H1138–H1147. doi: 10.1152/ajpheart.1998.275.4.H1138. Heart Circ Physiol 44. [DOI] [PubMed] [Google Scholar]
- 10.Ohishi S. Evaluation of time course and interrelationship of inflammatory mediators in experimental inflammatory reaction. Yakugaku Zasshi. 2000;120:455–462. [PubMed] [Google Scholar]
- 11.Durán WN, Seyama A, Yoshimura K, González DR, Jara PI, Figueroa XF, Boric MP. Stimulation of NO production and of eNOS phosphorylation in the microcirculation in vivo. Microvasc Res. 2000;60:104–111. doi: 10.1006/mvre.2000.2250. [DOI] [PubMed] [Google Scholar]
- 12.Fukumura D, Gohongi T, Kadambi A, Izumi Y, Ang J, Yun CO, Buerk DG, Huang PL, Jain RK. Predominant role of endothelial nitric oxide synthase in vascular endothelial growth factor-induced angio-genesis and vascular permeability. Proc Natl Acad Sci USA. 2001;98:2604–2609. doi: 10.1073/pnas.041359198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ignarro LJ, Cirino G, Casini A, Napoli C. Nitric oxide as a signaling molecule in the vascular system: an overview. J Cardiovasc Pharmacol. 1999;34:879–886. doi: 10.1097/00005344-199912000-00016. [DOI] [PubMed] [Google Scholar]
- 14.Jeon SY, Kim EA, Ma YW, Kim JP, Jung TG, Hwang EG. Nitric oxide mediates platelet activating factor-induced microvascular leakage in rat airways. Ann Otol Rhinol Laryngol. 2001;110:83–86. doi: 10.1177/000348940111000115. [DOI] [PubMed] [Google Scholar]
- 15.Mayhan WG. Role of nitric oxide in modulating permeability of hamster cheek pouch in response to adenosine 5′-diphosphate and bradykinin. Inflammation. 1992;16:295–305. doi: 10.1007/BF00917622. [DOI] [PubMed] [Google Scholar]
- 16.Parenti A, Morbidelli L, Cui XL, Douglas JG, Hood JD, Granger HJ, Ledda F, Ziche M. Nitric oxide is an upstream signal of vascular endothelial growth factor-induced extracellular signal-regulated kinase 1/2 activation in postcapillary endothelium. J Biol Chem. 1998;273:4220–4226. doi: 10.1074/jbc.273.7.4220. [DOI] [PubMed] [Google Scholar]
- 17.Murray MA, Heistad DD, Mayhan WG. Role of protein kinase C in bradykinin-induced increases in microvascular permeability. Circ Res. 1991;68:1340–1348. doi: 10.1161/01.res.68.5.1340. [DOI] [PubMed] [Google Scholar]
- 18.Fulton D, Gratton JP, McCabe TJ, Fontana J, Fujio Y, Walsh K, Franke TF, Papapetropoulos A, Sessa WC. Regulation of endothelium-derived nitric oxide production by the protein kinase Akt. Nature. 1999;399:597–601. doi: 10.1038/21218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Varma S, Breslin JW, Lal BK, Pappas PJ, Hobson RW, II, Durán WN. p42/44MAPK regulates baseline permeability and cGMP-induced hyperpermeability in endothelial cells. Microvasc Res. 2002;63:172–178. doi: 10.1006/mvre.2001.2381. [DOI] [PubMed] [Google Scholar]
- 20.Bekker AY, Ritter AB, Durán WN. Analysis of microvascular permeability to macromolecules by video-image digital processing. Microvasc Res. 1989;38:200–216. doi: 10.1016/0026-2862(89)90028-9. [DOI] [PubMed] [Google Scholar]
- 21.Kim D, Armenante PM, Durán WN. Transient analysis of macromolecular transport across microvascular wall and into interstitium. Am J Physiol. 1993;265:H993–H999. doi: 10.1152/ajpheart.1993.265.3.H993. [DOI] [PubMed] [Google Scholar]
- 22.Dillon PK, Durán WN. Effect of platelet-activating factor on microvascular permselectivity: dose-response relations and pathways of action in the hamster cheek pouch microcirculation. Circ Res. 1988;62:732–740. doi: 10.1161/01.res.62.4.732. [DOI] [PubMed] [Google Scholar]
- 23.Novogrodsky A, Vanichkin A, Patya M, Gazit A, Osherov N, Levitzki A. Prevention of lipopolysaccharide-induced lethal toxicity by tyrosine kinase inhibitors. Science. 1994;264:1319–1322. doi: 10.1126/science.8191285. [DOI] [PubMed] [Google Scholar]
- 24.Cuenda A, Rouse J, Doza YN, Meier R, Cohen P, Gallagher TF, Young PR, Lee JC. SB 203580 is a specific inhibitor of a MAP kinase homologue which is stimulated by cellular stresses and interleukin-1. FEBS Lett. 1995;364:229–233. doi: 10.1016/0014-5793(95)00357-f. [DOI] [PubMed] [Google Scholar]
- 25.Lee JC, Laydon JT, McDonnell PC, Gallagher TF, Kumar S, Green D, McNulty D, Blumenthal MJ, Heys JR, Landvatter SW, Strickler JE, McLaughlin MM, Siemens IR, Fisher SM, Livi GP, White JR, Adams JL, Young PR. A protein kinase involved in the regulation of inflammatory cytokine biosynthesis. Nature. 1994;372:739–746. doi: 10.1038/372739a0. [DOI] [PubMed] [Google Scholar]
- 26.Bernier SG, Haldar S, Michel T. Bradykinin-regulated interactions of the mitogen-activated protein kinase pathway with the endothelial nitric-oxide synthase. J Biol Chem. 2000;275:30707–30715. doi: 10.1074/jbc.M005116200. [DOI] [PubMed] [Google Scholar]
- 27.Yuan Y, Granger HJ, Zawieja DC, DeFily DV, Chilian WM. Histamine increases venular permeability via a phospholipase C-NO synthase-guanylate cascade. Am J Physiol. 1993;264:H1734–H1739. doi: 10.1152/ajpheart.1993.264.5.H1734. [DOI] [PubMed] [Google Scholar]
- 28.Yuan Y, Huang QB, Mac Wu H. Myosin light chain phosphorylation: modulation of basal and agonist-stimulated venular permeability. Am J Physiol. 1997;272:H1437–H1443. doi: 10.1152/ajpheart.1997.272.3.H1437. [DOI] [PubMed] [Google Scholar]
- 29.Kolch W. Meaningful relationships: the regulation of the Ras/Raf/MEK/ERK pathway by protein interactions. Biochem J. 2000;351(Pt 2):289–305. [PMC free article] [PubMed] [Google Scholar]
- 30.Harada J, Sugimoto M. An inhibitor of p38 and JNK MAP kinases prevents activation of caspase and apoptosis of cultured cerebellar granule neurons. Jpn J Pharmacol. 1999;79:369–378. doi: 10.1254/jjp.79.369. [DOI] [PubMed] [Google Scholar]
- 31.Noguchi K, Yamana H, Kitanaka C, Mochizuki T, Kokubu A, Kuchino Y. Differential role of the JNK and p38 MAPK pathway in c-Myc- and s-Myc-mediated apoptosis. Biochem Biophys Res Commun. 2000;267:221–227. doi: 10.1006/bbrc.1999.1952. [DOI] [PubMed] [Google Scholar]
- 32.Assefa Z, Vantieghem A, Garmyn M, Declercq W, Vandenabeele P, Vandenheede JR, Bouillon R, Merlevede W, Agostinis P. p38 mitogen-activated protein kinase regulates a novel, caspase-independent pathway for the mitochondrial cytochrome c release in ultraviolet B radiation-induced apoptosis. J Biol Chem. 2000;275:21416–21421. doi: 10.1074/jbc.M002634200. [DOI] [PubMed] [Google Scholar]
- 33.Harper SJ, LoGrasso P. Signalling for survival and death in neurones: the role of stress-activated kinases, JNK and p38. Cell Signal. 2001;13:299–310. doi: 10.1016/s0898-6568(01)00148-6. [DOI] [PubMed] [Google Scholar]
- 34.Arbabi S, Rosengart MR, Garcia I, Jelacic S, Maier RV. Priming interleukin 8 production: role of platelet-activating factor and p38. Arch Surg. 1999;134:1348–1353. doi: 10.1001/archsurg.134.12.1348. [DOI] [PubMed] [Google Scholar]
- 35.Nick JA, Avdi NJ, Gerwins P, Johnson GL, Worthen GS. Activation of a p38 mitogen-activated protein kinase in human neutrophils by lipopolysaccharide. J Immunol. 1996;156:4867–4875. [PubMed] [Google Scholar]
- 36.Nick JA, Avdi NJ, Young SK, Knall C, Gerwins P, Johnson GL, Worthen GS. Common and distinct intracellular signaling pathways in human neutrophils utilized by platelet activating factor and FMLP. J Clin Invest. 1997;99:975–986. doi: 10.1172/JCI119263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Marshall LA, Hansbury MJ, Bolognese BJ, Gum RJ, Young PR, Mayer RJ. Inhibitors of the p38 mitogen-activated kinase modulate IL-4 induction of low affinity IgE receptor (CD23) in human monocytes. J Immunol. 1998;161:6005–6013. [PubMed] [Google Scholar]
- 38.Ridley SH, Sarsfield SJ, Lee JC, Bigg HF, Cawston TE, Taylor DJ, Dewitt DL, Saklatvala J. Actions of IL-1 are selectively controlled by p38 mitogen-activated protein kinase: regulation of prostaglandin H synthase-2, metalloproteinases, and IL-6 at different levels. J Immunol. 1997;158:3165–3173. [PubMed] [Google Scholar]
- 39.Yin T, Sandhu G, Wolfgang CD, Burrier A, Webb RL, Rigel DF, Hai T, Whelan J. Tissue-specific pattern of stress kinase activation in ischemic/reperfused heart and kidney. J Biol Chem. 1997;272:19943–19950. doi: 10.1074/jbc.272.32.19943. [DOI] [PubMed] [Google Scholar]
- 40.Aramoto H, Breslin JW, Pappas PJ, Hobson RW, II, Durán WN. Vascular endothelial growth factor stimulates differential signaling pathways in the in vivo microcirculation. Am J Physiol: Heart & Circ Physiol. 2004;287:H1590–H1598. doi: 10.1152/ajpheart.00767.2003. [DOI] [PubMed] [Google Scholar]