Abstract
Recombinant adeno-associated virus vectors based on serotype 2 (rAAV2) can direct transgene expression in the central nervous system (CNS), but it is not known how other rAAV serotypes perform as CNS gene transfer vectors. Serotypes 4 and 5 are distinct from rAAV2 and from each other in their capsid regions, suggesting that they may direct binding and entry into different cell types. In this study, we examined the tropisms and transduction efficiencies of β-galactosidase-encoding vectors made from rAAV4 and rAAV5 compared with similarly designed rAAV2-based vectors. Injection of rAAV5 β-galactosidase (βgal) or rAAV4βgal into the lateral ventricle resulted in stable transduction of ependymal cells, with approximately 10-fold more positive cells than in mice injected with rAAV2βgal. Major differences between the three vectors were revealed upon striatal injections. Intrastriatal injection of rAAV4βgal resulted again in striking ependyma-specific expression of transgene, with a notable absence of transduced cells in the parenchyma. rAAV2βgal and rAAV5βgal intrastriatal injections led to β-gal-positive parenchymal cells, but, unlike rAAV2βgal, rAAV5βgal transduced both neurons and astrocytes. The number of transgene-positive cells in rAAV5βgal-injected brains was 130 and 5,000 times higher than in rAAV2βgal-injected brains at 3 and 15 wk, respectively. Moreover, transgene-positive cells were widely dispersed throughout the injected hemisphere in rAAV5βgal-transduced animals. Together, our data provide in vivo support for earlier in vitro work, suggesting that rAAV4 and rAAV5 gain cell entry by means of receptors distinct from rAAV2. These differences could be exploited to improve gene therapy for CNS disorders.
Gene transfer to the central nervous system (CNS) of mammals has been accomplished with various vectors, including recombinant adeno-associated virus type 2 (AAV2). AAV's are dependoviruses, whose small genomes are easily manipulated to produce recombinant viral vectors (1). Vectors derived from AAV2 have been shown to direct long-term expression of transgenes in multiple tissues, including the CNS (2–8). AAV is not known to be the etiologic agent of any disease in humans, and recombinant forms that are devoid of viral coding regions elicit little if any damage or inflammatory response following intracerebral injection (3, 8–12).
Recombinant AAV2 (rAAV2)-based vectors appear to preferentially infect neurons when injected into the CNS (3, 8, 12, 13). Studies using fluorescently labeled rAAV2 showed rapid binding and uptake into neurons following injection into the adult rat hippocampus or inferior colliculus, supporting a receptor-based neuronal tropism (14). Although the receptor responsible for transduction of neurons is not known, in vitro studies using nonneuronal cell lines show that rAAV2 transduction depends on cell surface heparan sulfate proteoglycans (HSPG), and is inhibited by soluble heparin (15). HSPG are prevalent on neuronal cell surfaces (16–18). Such HSPG may be responsible for the observed rapid, high-affinity binding and focal neuronal transduction after intracerebral injection of rAAV2.
There are six primary isolates of AAV (AAV1–6). Only AAV2 has been studied as a recombinant vector for gene transfer to the brain. Their genomes are organized similarly, with inverted terminal repeats (ITRs) flanking rep- and cap-encoding regions (19, 20). AAV2 and AAV4 ITRs are nearly identical, such that AAV4 genomes can be packaged by AAV2 and vice versa (19). AAV5 is not capable of packaging AAV2 DNA (20, 21). Extensive heterogeneity exists within the proposed surface-exposed regions of the AAV2, -4, and -5 capsid structures, suggesting that each serotype may have a distinct mechanism of uptake. Data from several studies support this notion (19–21). First, binding competition experiments demonstrate that neither AAV4- or AAV5-mediated transduction is inhibited by soluble heparin (ref. 20; and J.Z., B.L.D., and J.A.C., unpublished observations). Second, transduction efficiencies for AAV4 and -5 differ from AAV2 in several cell lines (19, 20). Third, unlike AAV2, AAV4 transduction is trypsin-insensitive (19). Finally, in cotransduction experiments, the viruses do not competitively inhibit AAV2 transduction (19).
Given that AAV5, -4, and -2 likely mediate infection through attachment to different cellular receptors, we set out to determine the transduction efficiencies and tropisms of recombinant vectors derived from AAV4 and AAV5 in mammalian brain, in comparison to AAV2. Furthermore, because receptor specificities may affect particle diffusion through the extracellular spaces, we also compared the distribution of transduction by rAAV2, rAAV4, and rAAV5.
Materials and Methods
Vector Production.
rAAV vectors based on AAV2, AAV4, or AAV5 were prepared by using high efficiency electroporation and adenovirus infection as described previously (22). All three vectors contained a nucleus-targeted Escherichia coli β-galactosidase (βgal) gene with expression driven off the Rous sarcoma virus long terminal repeat (RSV) promoter. The expression cassette was flanked by AAV2 ITR sequences for rAAV2βgal particles and rAAV4βgal particles. The expression cassette was flanked by AAV5 ITR's for rAAV5βgal particles. The number of recombinant particles were quantified by Southern dot blot, and the biological activity was tested by 5-bromo-4-chloro-3-indolyl β-d-galactoside (X-Gal) histochemical staining in a serial dilution on Cos cells. The viral titers ranged between 2 × 1011 and 2 × 1011 particles per ml, and the ratio of transducing to total particles was similar to that described previously for each of the types (19, 20, 22). The recombinant viruses used were screened for wild-type AAV contamination by PCR, and for wild-type adenovirus by a serial dilution assay using an FITC-hexon antibody (<103 replication competent adenoviruses per ml) (23).
Injections.
Six- to eight-week-old adult male C57BL/6 mice were purchased from The Jackson Laboratory and housed at the University of Iowa Animal Care facility. All animal procedures were approved by the University of Iowa Animal Care and Use Committee. Vector injections were performed as previously described (24). Briefly, mice were anesthetized and vectors were stereotactically injected into either the right lateral ventricle or the right striatum, using a 26-gauge Hamilton syringe driven by a microinjector (Micro 1; World Precision Instruments, Sarasota, FL) at 0.5 μl per minute. For ventricular injections, 10-μl volumes were injected at coordinates 0.4 mm rostral and 1.0 mm lateral to bregma, and at a 2-mm depth. For striatal injections, 5-μl volumes were injected at coordinates 0.4 mm rostral and 2 mm lateral to bregma, and at a 3-mm depth. The doses of vector injected into the striatum, given as particle doses, were as follows: rAAV2βgal, 4 × 109 (n = 5); rAAV4βgal, 2 × 109 (n = 4) or 8 × 109 (n = 3); rAAV5βgal, 1.5 × 1010 (n = 6) or 3 × 1010 (n = 2). For injections into the ventricle, the doses were as follows: rAAV2βgal, 1 × 109 (n = 3) or 2 × 109 (n = 2); rAAV4βgal, 4 × 109 (n = 8); rAAV5βgal, 3 × 1010 (n = 4). A minimum of two independent experiments was done for each vector and injection site.
Histochemistry.
Three or fifteen weeks after injection, groups of mice were perfused with 2% paraformaldehyde; the brains were removed and processed as previously described (24). Then, 10-μm-thick coronal sections were cut at 100-μm intervals, and X-Gal histochemical staining was performed to identify β-galactosidase expressing cells (25). For each mouse, the number of β-galactosidase-positive cells in every fourth section, spanning 1.3 mm of tissue rostral and 1.3 mm caudal to the injection site, were counted and summed. These sums allow quantitative comparisons among the three vectors, although they do not reflect the total number of transduced cells in vivo.
Immunofluorescent Staining.
Ten-micrometer coronal cryosections of brains harvested 15 wk after intrastriatal injection of rAAV5βgal were dual stained for β-galactosidase and either neuronal or astrocytic markers. The primary antibodies used were as follows: rabbit IgG specific for E. coli β-galactosidase (Biodesign International, Kennebunkport, ME); mouse monoclonal IgG specific for NeuN (Chemicon), which strongly stains neuronal cell nuclei with lighter staining of the cytoplasm; and a Cy5-conjugated mouse monoclonal specific for glial fibrillary acidic protein (GFAP) (Sigma), an intermediate filament of astrocytes. Secondary antibodies used were ALEXA 488 goat anti-rabbit IgG (Molecular Probes) and lissamine-rhodamine goat anti-mouse IgG (Jackson ImmunoResearch). Sections were blocked for 2 h at room temperature in PBS with 3% BSA, 10% normal goat serum, and 0.1% Triton X-100. Sections were incubated overnight with primary antibodies diluted in PBS with 3% BSA and 0.1% Triton X-100 at 4°C, then washed and incubated with secondary antibodies in PBS with 1% normal goat serum and 0.1% Triton X-100 for 2 h at room temperature. Confocal laser microscopy was performed by using ×63 and ×40 oil-immersion objectives on a Zeiss LSM 510 and associated software. Z-series images (0.3- to 1.0-μm-wide slices) were captured and analyzed for cellular colocalization of antigens. Colocalization of β-galactosidase and either NeuN or GFAP is represented in two-color merged images from single slices within the series.
Statistical Analysis.
The data was analyzed by using a three-way ANOVA with the three factors consisting of rAAVβgal type, injection location, and time interval following vector injection. A log transformation was applied to the data to normalize the data distribution and reduce heterogeneity of group variances. Bonferroni's method was applied to each set of comparisons to adjust for the number of mice injected and to the P values for each set of comparisons. A Bonferroni-adjusted P value < 0.05 was considered statistically significant.
Results
Quantification of Transduced Cells.
We evaluated the efficiency of transduction of rAAV2, rAAV4, and rAAV5 in the brain using recombinant viruses expressing the β-galactosidase reporter gene under control of the Rous sarcoma virus (RSV) promoter (rAAV2βgal, rAAV4βgal, and rAAV5βgal, respectively). Groups of mice received either 10 μl of vector in the right lateral ventricle, or 5 μl of vector into the right striatum, at the particle doses stated in Materials and Methods. Three or fifteen weeks later, the brains were harvested and transgene-positive cells in cryosections spanning 2.6 mm rostral-caudal were quantified (Fig. 1).
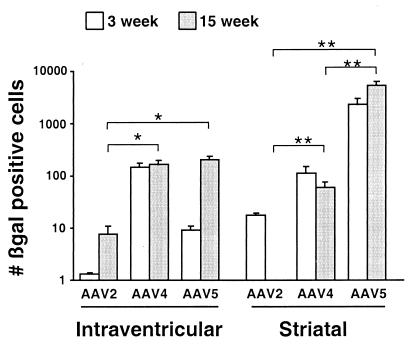
Figure 1.
The number of β-galactosidase-positive cells after intracerebral injection. Animals were injected with rAAV2βgal, rAAV4βgal, or rAAV5βgal into the ventricle or striatum of mice, and brains taken at the times indicated. Blocks (2.6 mm, surrounding the injection site) were sectioned, processed for β-galactosidase histochemistry, and transgene-positive cells were counted. Data represent mean ± SEM. *, P < 0.05; **, P < 0.005.
Three weeks after intraventricular injection, the number of rAAV4βgal-transduced cells was approximately 100- and 10-fold greater than for rAAV2βgal and rAAV5βgal, respectively. rAAV2βgal yielded the poorest results, with the rare positive cell observed. After 15 wk, the number of rAAV5βgal-transduced cells was increased compared with the 3-wk time point, reaching numbers similar to that of rAAV4βgal. This increase in rAAV5βgal expression nearly reached statistical significance (P = 0.055). The number of transduced cells also tended to increase from 3 to 15 wk for rAAV2βgal, but remained significantly lower than for the rAAV4βgal and rAAV5βgal vectors (P = 0.007 and 0.019, respectively).
After striatal injections, strikingly greater numbers of transgene-expressing cells were detected after injection of rAAV5βgal compared with both rAAV2βgal and rAAV4βgal (for both, P < 0.0001). In turn, rAAV4βgal transduced more cells than rAAV2βgal by 15 wk (P = 0.001). Comparison of the 3- and 15-wk time points showed complete loss of rAAV2βgal-mediated transgene expression, but stable expression after rAAV4βgal injection. In contrast, there was a trend toward increased numbers of β-galactosidase-expressing cells from 3 to 15 wk after rAAV5βgal injections.
Regional Distribution of Transduced Cells.
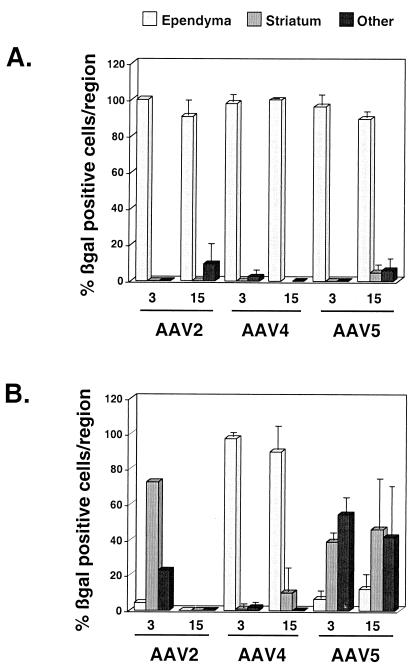
To analyze potential regional tropisms, β-galactosidase-positive cells were categorized into ependyma/choroid, striatum, or other (septal area, corpus callosum, neocortex, and fornix) regions. Fig. 2 illustrates the distribution of transduced cells for each vector after intraventricular or intrastriatal injections. After intraventricular injections, transgene-expressing cells were localized predominantly to the ependyma for all rAAVβgal types at both 3 and 15 wk (Fig. 2A). Striatal injections yielded several interesting results (Fig. 2B). First, rAAV2βgal and rAAV5βgal vectors mediated transduction in multiple regions. Second, this data again demonstrates the global loss in rAAV2βgal-transduced cells in all cerebral regions from 3 to 15 wk. Finally, and perhaps most surprisingly, rAAV4βgal injection into the striatum resulted in almost exclusive ependymal cell transduction.
Figure 2.
The distribution of β-galactosidase positive cells in brains of mice at 3 or 15 wk after injection of rAAV2βgal, rAAV4βgal, or rAAV5βgal. β-galactosidase-positive cells within the ependymal, striatal, or “other” (septal and fornix regions, corpus callosum, and neocortex) regions, from sections encompassing the injection site, were counted and that value represented as a percent of the total number of transgene positive cells. (A) Data from sections obtained 3 or 15 wk after intraventricular injection. (B) Data from sections obtained 3 or 15 wk after injection into the striatum. Data represent means ± SEM.
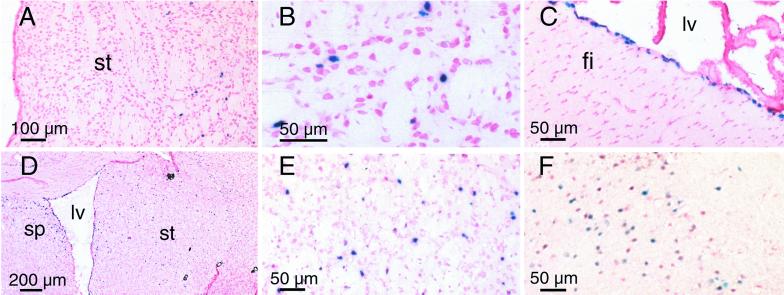
The patterns of transduction observed after striatal injections of the three vectors are illustrated in Fig. 3, which shows representative images of X-Gal-stained sections. Few blue-stained nuclei were evident in the striatum of rAAV2βgal-injected mice (injected dose = 4 × 109 particles), and only at the 3-wk time point (Fig. 3 A and B), while rAAV4βgal (injected dose = 4 × 109 particles) selectively transduced the ependyma (Fig. 3C). rAAV5βgal injections (1.5 × 1010 particles) resulted in diffuse transduction in multiple cerebral regions, including the striatum (Fig. 3 D and E), septal region (Fig. 3D), and neocortex (Fig. 3F). Although the particle dose for rAAV5βgal was only ≈4-fold greater, the relative spread of cells transduced by rAAV5βgal was extensive; β-galactosidase-expressing cells were detected 4.0 mm in the rostral-caudal, 3.5 mm dorsal-ventral, and 3.2 mm laterally, to encompass much of the injected hemisphere and portions of the medial region.
Figure 3.
β-galactosidase histochemistry for transgene-positive cells after striatal injection of rAAV vectors. (A and B) Representative photomicrograph of sections from mice injected with rAAV2βgal. (B) A magnified photograph of the transgene positive striatal region seen in A. (C) Demonstration of distinctive ependymal-specific staining for β-galactosidase in sections from animals injected with rAAV4βgal. (D–F). Photomicrographs illustrating the extensive distribution of transgene-positive cells after rAAV5βgal injection. (E) Magnification of the striatal region in D. (F) Transgene positive cells in the cortex distant from the injection site. The photomicrographs are representative of at least three independent experiments. fi, fimbria region; lv, lateral ventricle; sp, medial septal region; st, striatum.
Characterization of rAAV5βgal-Transduced Cells.
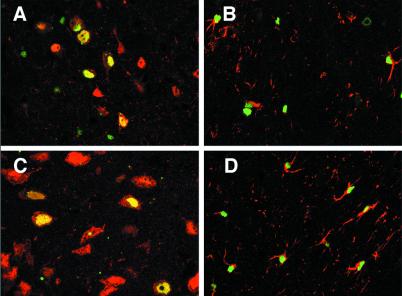
Previous studies have characterized the cell types transduced after parenchymal injection of rAAV2βgal under control of the CMV immediate early enhancer/promoter to be predominantly neurons, with an occasional transgene-expressing astrocyte (3, 12, 13). To determine which cell types were transduced by rAAV5βgal, we immunofluorescently stained representative sections of brains harvested 15 wk after intrastriatal injection. Confocal microscopy was performed to assess colocalization of β-galactosidase and representative markers. Sections were dual stained for β-galactosidase and either GFAP (astrocyte marker) or NeuN (neuron marker). In the striatum, many transgene-expressing cells stained positive for NeuN, indicating substantial neuronal cell transduction (Fig. 4A). Transduced astrocytes were also evident in the striatum, with GFAP-positive cell processes enveloping β-galactosidase-positive nuclei (Fig. 4B). Analyses of cells transduced in regions outside the striatum revealed that transgene-expressing cells in the cortex were also a mix of neurons and astrocytes (not shown), while those in the septal area were predominantly neurons (Fig. 4C). In addition, although rAAV5βgal-transduced cells were noticeably more concentrated in gray-matter areas, a minor proportion of transgene-positive nuclei were evident in the corpus callosum, sometimes far-removed from the injection site. In these instances, GFAP immunoreactivity identified these cells as astrocytes (Fig. 4D).
Figure 4.
Identification of transduced cells after intrastriatal injection of rAAV5βgal. Fifteen weeks after injection of rAAV5βgal, coronal brain sections were dual stained for β-galactosidase (green nuclei) and NeuN (neuronal-specific, red nuclei and light red cytoplasm), or for β-galactosidase and GFAP (astrocyte-specific, red cell processes). Confocal microscopy image analysis was performed, and representative two-color-merged images of single z-series slices are shown. In the striatum, both transduced neurons (yellow cell nuclei in A) and transduced astrocytes (B) were detected. In the medial septal region, transduction appeared to be restricted to neurons (C), whereas in the corpus callosum, the transduced cells were GFAP-positive astrocytes (D). Images were captured by using a ×40 (A, B, and D) or ×63 (C) oil-immersion objective.
Discussion
In this study, we assessed CNS cell transduction with rAAV2, rAAV4, and rAAV5 vectors carrying an RSV-β-galactosidase expression cassette after intracerebral injections into the lateral ventricle or the striatum. After intraventricular injections, all three vectors transduced primarily ependymal cells. Our results with rAAV2βgal were similar to prior reports showing that transduced cells were few, and restricted to the ependyma/choroid plexus (3, 10). Ependymal cell transduction was more impressive with rAAV4βgal and rAAV5βgal vectors. Because our rAAV2βgal and rAAV4βgal particles contain identical DNA sequences, differences in transduction efficiencies between these two vectors must be attributed to variations in their capsids. This implies that the rAAV4 capsid mediates more efficient entry into ependymal cells than rAAV2. rAAV5 capsid is also distinct and may likewise target ependyma more efficiently than rAAV2. Differences in the ITR region of rAAV5βgal may additionally influence expression. Interestingly, for rAAV5βgal, the number of β-galactosidase-positive ependymal cells increased significantly after 3 wk, reaching levels similar to rAAV4βgal at 15 wk. A slower rate of rAAV5βgal ITR-dependent second-strand synthesis could explain this delay in transgene expression.
We noted very limited transduction outside of the ependyma following ventricular injection of any of the vectors in our study. Using a neuronal-specific promoter, Wu et al. (26) observed transgene expression in the hypothalamus after rAAV2βgal delivery to rat ventricle. It is probable then that in our study, rAAV2βgal viruses were similarly able to transduce nonependymal cells, but that RSV promoter activity was restricted to ependymal cells.
After intrastriatal injections, we observed distinct regional patterns of transduction for all three vectors. With rAAV4βgal, numerous positive cells lined the ventricles, with very few transgene-expressing cells in the parenchyma. In contrast, rAAV2βgal and rAAV5βgal vectors transduced predominantly parenchymal cells, and unlike rAAV2βgal (3, 12, 13), rAAV5βgal transduced a significant proportion of astrocytes as well as neurons. Moreover, rAAV5βgal transduced a greater number of cells, over a larger volume of tissue compared with rAAV2βgal.
rAAV2 is considered to have a wide host range, with ability to infect multiple tissues and cell types in vitro and in vivo. HSPG has been reported as a primary receptor for AAV2 attachment, while human fibroblast growth factor receptor-1 and αVβ5 integrin have been reported to be coreceptors necessary for transduction (15, 27–29). Also, a 150-kDa protein has been identified on the surface of AAV2-permissive cells (30). Whether requirements for these receptors hold true for rAAV2βgal infectivity of CNS cells remains to be determined. With respect to AAV4 and AAV5, little information is available regarding the cellular proteins necessary for binding and uptake. When compared with AAV2, heterogeneities in the capsid-encoding regions, heparin-insensitive transduction, and differential abilities to transduce cell lines in vitro together strongly implicate different receptor requirements for cell entry (19, 20, 31). The inability of rAAV4βgal to transduce parenchymal cells and its selective ependymal transduction following intrastriatal injection clearly demonstrate an altered tropism for rAAV4βgal and indicate that, in the CNS, AAV4 uses a receptor and/or coreceptor unique to ependymal cells. Interestingly, ependyma was equally transduced when rAAV4βgal was applied either intraventricularly or intraparenchymally. One explanation is that the receptor is present on both apical and basal surfaces. Alternatively the receptor is localized on the basal surface, and the vector is able to diffuse away from the needle tract and dissect through the subependymal plane to gain basal access. Experiments are underway to delineate these possibilities.
The enhanced parenchymal cell transduction we observed for rAAV5βgal compared with rAAV2βgal, as well as the diffuse and widespread pattern of transduction mediated by rAAV5βgal may similarly reflect distinct receptor requirements. In the rat brain, rAAV2βgal particles have been shown to preferentially bind neurons, but not glial cells, within minutes of injection, and be transported within 30 min to neuronal cell nuclei (14). Considering that neuronal subtypes in adult rodent brain express integral membrane HSPG, such as syndecans (16, 17) and glypican-1 (18), it is conceivable that AAV2 binds strongly to and enters neurons surrounding the injection site. Moreover, AAV2 particles may become sequestered in extracellular HSPG in a way that limits vector diffusion and reduces transduction efficiency. An ability of rAAV5βgal to travel in a less-restricted fashion may explain our observations of widespread transduction in comparison to rAAV2βgal.
It appears that other investigators have achieved greater rAAV2-mediated transduction efficiencies in the striatum of rodent brain than in our study (7, 10, 12). As those studies used the CMV enhancer/promoter, it is probable that the RSV-driven transcription is not as strong as that from CMV in rAAV2-transduced CNS. These results suggest that rAAV5 and rAAV4 vectors containing the CMV enhancer/promoter could further increase the number of transgene-positive cells detectable after cerebral injection.
Following intrastriatal injection, we observed loss of rAAV2βgal-transduced cells over time consistent with observations of others (3, 10, 12, 13, 32, 33). This may be a consequence of RSV promoter shut-off, similar to what has been proposed for CMV-driven transgenes in the CNS (3, 10, 33) and/or may represent loss of episomal transgenes (32). In contrast, transgene expression after rAAV5βgal injection was stable over the time-course of our study. rAAV5 could target to cell subsets better able to sustain RSVp-driven transcription, or there could be positive influences of the AAV5 ITRS on either genome stability or RSV promoter activity.
In summary, our experiments explored the use of rAAV4 and rAAV5 as vectors for gene transfer to the CNS. rAAV4βgal demonstrated a distinct affinity for transduction of the ependyma, a desirable characteristic when attempting to restrict gene delivery to ependymal cells. Secretion of a transgene product from transduced ependymal cells into the cerebral spinal fluid could be a useful protein delivery mechanism, as has been shown in prior studies using recombinant adenoviruses (34, 35). In addition, rAAV4 has the potential to transfer genes to ependymal resident stem cells (36), which could lead to transgene expression in differentiated populations of progeny cells. rAAV5βgal transduced large numbers of cells, with lasting expression in both neuronal and glial types. More importantly, rAAV5βgal exhibited an extensive transduction volume. Vector diffusion is an extremely valuable feature for gene therapy of CNS diseases exhibiting widespread pathology, such as the neurodegenerative aspect of the lysosomal storage diseases. This characteristic coupled to persistent expression could reduce the need for multiple injection sites and repeat injections and their obvious associated risks.
Acknowledgments
We thank Christine McLennan for secretarial assistance and M. Bridget Zimmerman and the University of Iowa Biostatistics Consulting Center for expert statistical analysis. This work was supported by the National Institutes of Health (HD 33531) and the Roy J. Carver Trust (to B.L.D. and J.Z.).
Abbreviations
- CNS
central nervous system
- AAV
adeno-associated virus
- r
recombinant
- HSPG
heparan sulfate proteoglycan
- ITR
inverted terminal repeat
- βgal
β-galactosidase
- X-Gal
5-bromo-4-chloro-3-indolyl β-d-galactoside
- GFAP
glial fibrillary acidic protein
- RSV
Rous sarcoma virus
Footnotes
Article published online before print: Proc. Natl. Acad. Sci. USA, 10.1073/pnas.050581197.
Article and publication date are at www.pnas.org/cgi/doi/10.1073/pnas.050581197
References
- 1.Berns K I. In: Virology. Fields B N, Knipe D M, editors. New York: Raven; 1990. pp. 1743–1763. [Google Scholar]
- 2.Zhang L, Wang D, Fischer H, Fan P-D, Widdicombe J H, Kan Y W, Dong J-Y. Proc Natl Acad Sci USA. 1998;95:10158–10163. doi: 10.1073/pnas.95.17.10158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.McCown T J, Xiao X, Li J, Breese G R, Samulski R J. Brain Res. 1996;713:99–107. doi: 10.1016/0006-8993(95)01488-8. [DOI] [PubMed] [Google Scholar]
- 4.Herzog R W, Hagstrom J N, Kung S-H, Tai S J, Wilson J M, Fisher K J, High K A. Proc Natl Acad Sci USA. 1997;94:5804–5809. doi: 10.1073/pnas.94.11.5804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Snyder R O, Miao C H, Patijn G A, Spratt S K, Danos O, Nagy D, Gown A M, Winther B, Meuse L, Cohen L K, et al. Nat Genet. 1997;16:270–276. doi: 10.1038/ng0797-270. [DOI] [PubMed] [Google Scholar]
- 6.Flotte T R, Afione S A, Conrad C, McGrath S A, Solow R, Oka H, Zeitlin P L, Guggino W B, Carter B J. Proc Natl Acad Sci USA. 1993;90:10613–10617. doi: 10.1073/pnas.90.22.10613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fan D-S, Ogawa M, Fujimoto K-I, Ikeguchi K, Ogasawara Y, Urabe M, Nishizawa M, Nakano I, Yoshida M, Nagatsu I, et al. Hum Gene Ther. 1998;9:2527–2535. doi: 10.1089/hum.1998.9.17-2527. [DOI] [PubMed] [Google Scholar]
- 8.During M J, Samulski R J, Elsworth J D, Kaplitt M G, Leone P, Xiao X, Li J, Freese A, Taylor J R, Roth R H, et al. Gene Ther. 1998;5:820–827. doi: 10.1038/sj.gt.3300650. [DOI] [PubMed] [Google Scholar]
- 9.Blömer U, Naldini L, Kafri T, Trono D, Verma I M, Gage F H. J Virol. 1997;71:6641–6649. doi: 10.1128/jvi.71.9.6641-6649.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lo W D, Qu G, Sferra T J, Clark R, Chen R, Johnson P R. Hum Gene Ther. 1999;10:201–213. doi: 10.1089/10430349950018995. [DOI] [PubMed] [Google Scholar]
- 11.Skorupa A F, Fisher K J, Wilson J M, Parente M K, Wolfe J H. Exp Neurol. 1999;160:17–27. doi: 10.1006/exnr.1999.7176. [DOI] [PubMed] [Google Scholar]
- 12.Mandel R J, Rendahl K G, Spratt S K, Snyder R O, Cohen L K, Leff S E. J Neurosci. 1998;18:4271–4284. doi: 10.1523/JNEUROSCI.18-11-04271.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kaplitt M G, Leone P, Samulski R J, Xiao X, Pfaff D W, O'Malley K L, During M J. Nat Genet. 1994;8:148–154. doi: 10.1038/ng1094-148. [DOI] [PubMed] [Google Scholar]
- 14.Bartlett J S, Samulski R J, McCown T J. Hum Gene Ther. 1998;9(8):1181–1186. doi: 10.1089/hum.1998.9.8-1181. [DOI] [PubMed] [Google Scholar]
- 15.Summerford C, Samulski R J. J Virol. 1998;72(2):1438–1445. doi: 10.1128/jvi.72.2.1438-1445.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hsueh Y-P, Yang F-C, Kharazia V, Naisbitt S, Cohen A R, Weinberg R J, Sheng M. J Cell Biol. 1998;142:139–151. doi: 10.1083/jcb.142.1.139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hsueh Y-P, Sheng M. J Neurosci. 1999;19:7415–7425. doi: 10.1523/JNEUROSCI.19-17-07415.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Liang Y, Annan R S, Carr S A, Popp S, Mevissen M, Margolis R K, Margolis R U. J Biol Chem. 1999;274:17885–17892. doi: 10.1074/jbc.274.25.17885. [DOI] [PubMed] [Google Scholar]
- 19.Chiorini J A, Yang L, Liu Y, Safer B, Kotin R M. J Virol. 1997;71:6823–6833. doi: 10.1128/jvi.71.9.6823-6833.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chiorini J A, Kim F, Yang L, Kotin R M. J Virol. 1999;73:1309–1319. doi: 10.1128/jvi.73.2.1309-1319.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chiorini J A, Afione S, Kotin R M. J Virol. 1999;73:4293–4298. doi: 10.1128/jvi.73.5.4293-4298.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Chiorini J A, Wendtner C M, Urcelay E, Safer B, Hallek M, Kotin R M. Hum Gene Ther. 1995;6:1531–1541. doi: 10.1089/hum.1995.6.12-1531. [DOI] [PubMed] [Google Scholar]
- 23.Hehir K M, Armentano D, Cardoza L M, Choquette T L, Berthelette P B, White G A, Couture L A, Everton M B, Keegan J, Martin J M, et al. J Virol. 1996;70:8459–8467. doi: 10.1128/jvi.70.12.8459-8467.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ghodsi A, Stein C, Derksen T, Yang G, Anderson R D, Davidson B L. Hum Gene Ther. 1998;9:2331–2340. doi: 10.1089/hum.1998.9.16-2331. [DOI] [PubMed] [Google Scholar]
- 25.Davidson B L, Doran S E, Shewach D S, Latta J M, Hartman J W, Roessler B J. Exp Neurol. 1994;125:258–267. doi: 10.1006/exnr.1994.1028. [DOI] [PubMed] [Google Scholar]
- 26.Wu P, Phillips M I, Bui J, Terwilliger E F. J Virol. 1998;72:5919–5926. doi: 10.1128/jvi.72.7.5919-5926.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Qing K, Mah C, Hansen J, Zhou S, Dwarki V, Srivastava A. Nat Med. 1999;5:71–77. doi: 10.1038/4758. [DOI] [PubMed] [Google Scholar]
- 28.Summerford C, Bartlett J S, Samulski R J. Nat Med. 1999;5:78–82. doi: 10.1038/4768. [DOI] [PubMed] [Google Scholar]
- 29.Qiu J, Brown K E. Virology. 1999;264:436–440. doi: 10.1006/viro.1999.0010. [DOI] [PubMed] [Google Scholar]
- 30.Mizukami H, Young N S, Brown K E. Virology. 1996;217:124–130. doi: 10.1006/viro.1996.0099. [DOI] [PubMed] [Google Scholar]
- 31.Bantel-Schaal U, Delius H, Schmidt R, zur Hausen H. J Virol. 1999;73:939–947. doi: 10.1128/jvi.73.2.939-947.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Doll R F, Crandall J E, Dyer C A, Aucoin J M, Smith F I. Gene Ther. 1996;3:437–447. [PubMed] [Google Scholar]
- 33.Klein R L, Meyer E M, Peel A L, Zolotukhin S, Meyers C, Muzyczka N, King M A. Exp Neurol. 1998;150:183–194. doi: 10.1006/exnr.1997.6736. [DOI] [PubMed] [Google Scholar]
- 34.Bajocchi G, Feldman S H, Crystal R G, Mastrangeli A. Nat Genet. 1993;3:229–234. doi: 10.1038/ng0393-229. [DOI] [PubMed] [Google Scholar]
- 35.Ghodsi A, Stein C, Derksen T, Martins I, Anderson R D, Davidson B L. Exp Neurol. 1999;160:109–116. doi: 10.1006/exnr.1999.7205. [DOI] [PubMed] [Google Scholar]
- 36.Johansson C B, Momma S, Clarke D L, Risling M, Lendahl U, Frisen J. Cell. 1999;96:25–34. doi: 10.1016/s0092-8674(00)80956-3. [DOI] [PubMed] [Google Scholar]