Abstract
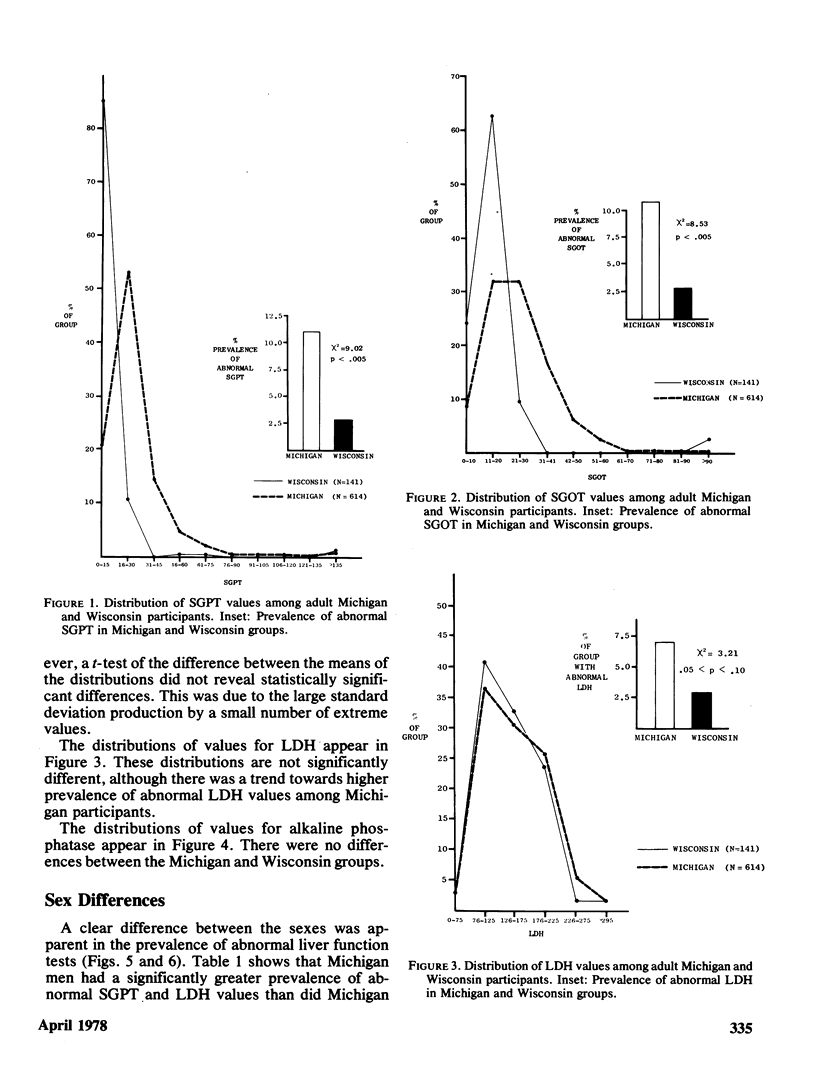
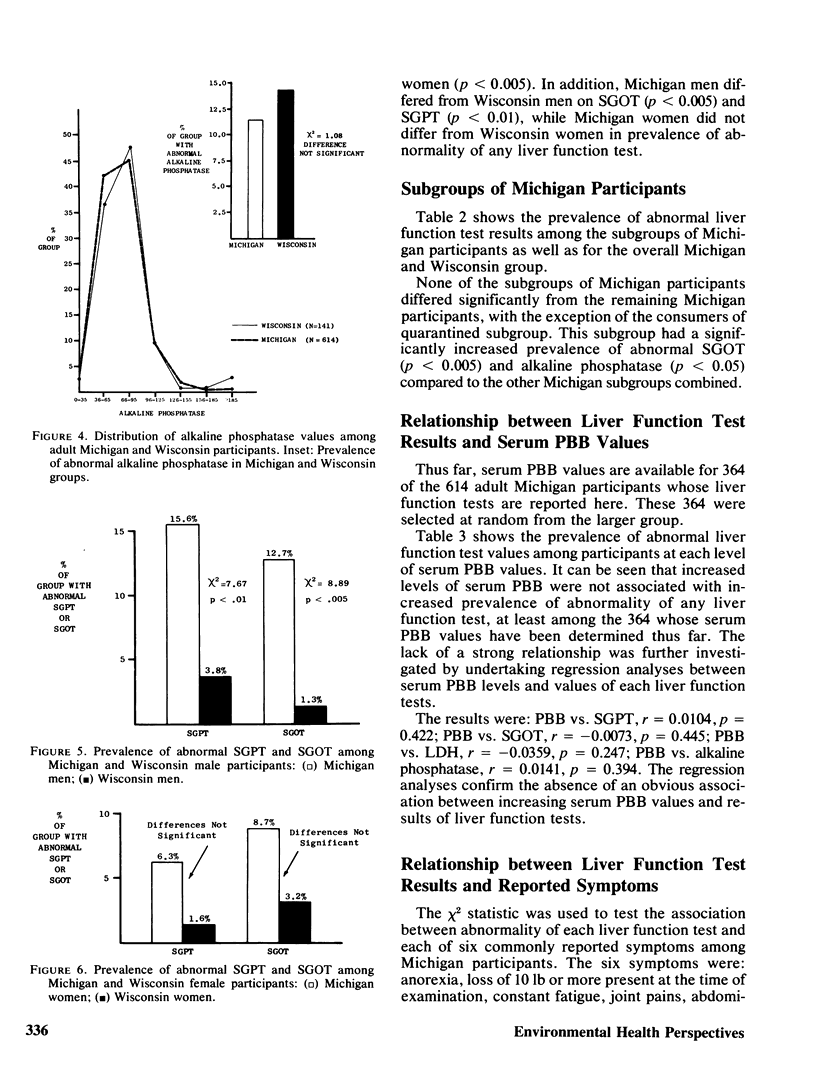
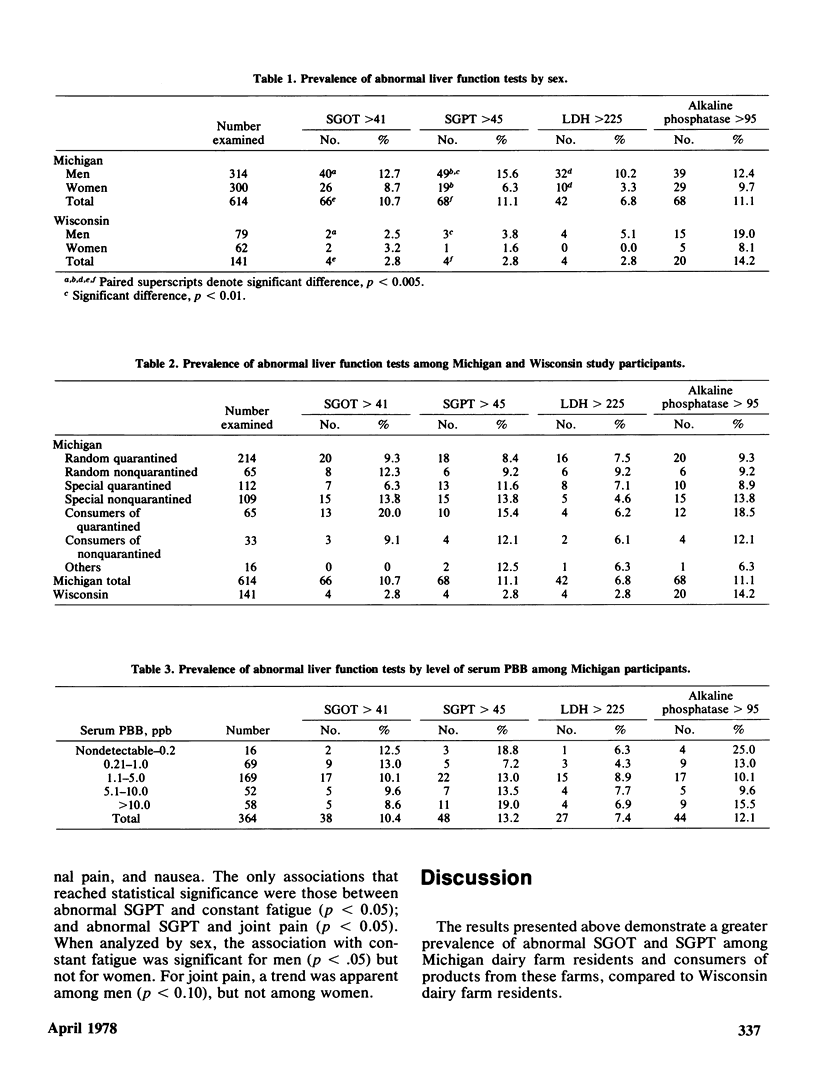
Serum activity of SGOT, SGPT, LDH, and alkaline phosphatase was measured in 614 Michigan adults exposed to PBB and 141 Wisconsin adults not so exposed. The Michigan group had higher prevalence of abnormal SGOT (p less than 0.005) and SGPT (p less than 0.005). A clear sex difference was observed. Michigan men had a higher prevalence of abnormal SGPT (p less than 0.005) and LDH (p less than 0.005) than Michigan women, and a higher prevalence than Wisconsin men of abnormal SGOT (p less than 0.005) and SGPT (p less than 0.01). These differences could not be ascribed to differing patterns of alcohol consumption, laboratory error, or choice of criteria for normality/abnormality. Seven Michigan subgroups were defined on the basis of the criteria by which they had been selected to participate. The two subgroups who were essentially self-invited did not differ from the remaining five randomly selected subgroups combined in prevalence of these abnormal liver function tests. Based on 364 serum PBB analyses thus far analyzed of the 614 Michigan participants, no obvious relationship between serum PBB values and liver function tests was observed. However, this is a tentative conclusion that will be further evaluated when remaining serum PBB analyses are completed. The greater prevalence of abnormal SGPT and SGOT among Michigan dairy farm residents compared to the Wisconsin dairy farm residents is tentatively ascribed to the former group's exposure to PBB.
Full text
PDF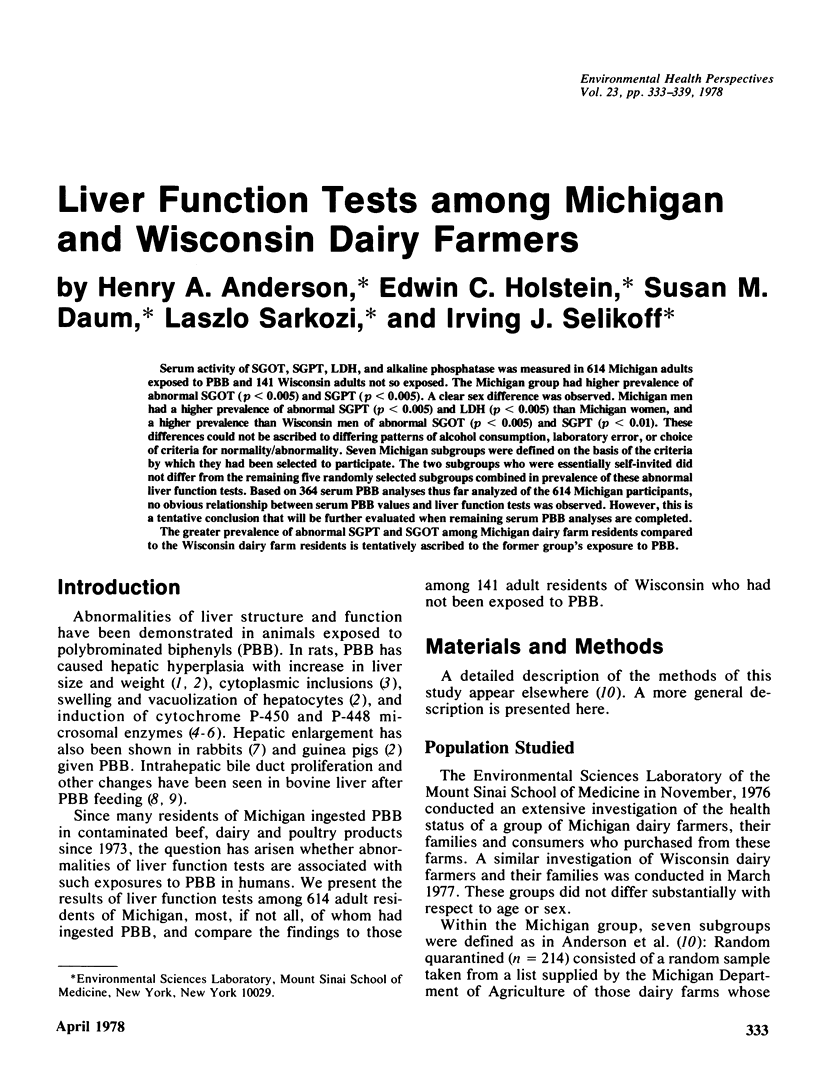



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anderson H. A., Lilis R., Selikoff I. J., Rosenman K. D., Valciukas J. A., Freedman S. Unanticipated prevalence of symptoms among dairy farmers in Michigan and Wisconsin. Environ Health Perspect. 1978 Apr;23:217–226. doi: 10.1289/ehp.7823217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dent J. G. Characteristics of cytochrome P-450 and mixed function oxidase enzymes following treatment with PBBs. Environ Health Perspect. 1978 Apr;23:301–307. doi: 10.1289/ehp.7823301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gutenmann W. H., Lisk D. J. Tissue storage and excretion in milk of polybrominated biphenyls in ruminants. J Agric Food Chem. 1975 Sep-Oct;23(5):1005–1007. doi: 10.1021/jf60201a008. [DOI] [PubMed] [Google Scholar]
- Jackson T. F., Halbert F. L. A toxic syndrome associated with the feeding of polybrominated biphenyl-contaminated protein concentrate to dairy cattle. J Am Vet Med Assoc. 1974 Sep 1;165(5):437–439. [PubMed] [Google Scholar]
- Kimbrough R. D., Burse V. W., Liddle J. A. Persistent liver lesions in rats after a single oral dose of polybrominated biphenyls (firemaster FF-1) and concomitant PBB tissue levels. Environ Health Perspect. 1978 Apr;23:265–273. doi: 10.1289/ehp.7823265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee K. P., Hebert R. R., Sherman H., Aftosmis J. G., Waritz R. S. Octabromobiphemyl-induced ultrastructural changes in rat liver. Arch Environ Health. 1975 Sep;30(9):465–471. doi: 10.1080/00039896.1975.10666752. [DOI] [PubMed] [Google Scholar]
- Lee K. P., Herbert R. R., Sherman H., Aftosmis J. G., Waritz R. S. Bromine tissue residue and hepatotoxic effects of octabromobiphenyl in rats. Toxicol Appl Pharmacol. 1975 Oct;34(1):115–127. doi: 10.1016/0041-008x(75)90180-5. [DOI] [PubMed] [Google Scholar]
- Sleight S. D., Sanger V. L. Pathologic features of polybrominated biphenyl toxicosis in the rat and guinea pig. J Am Vet Med Assoc. 1976 Dec 1;169(11):1231–1235. [PubMed] [Google Scholar]


