Abstract
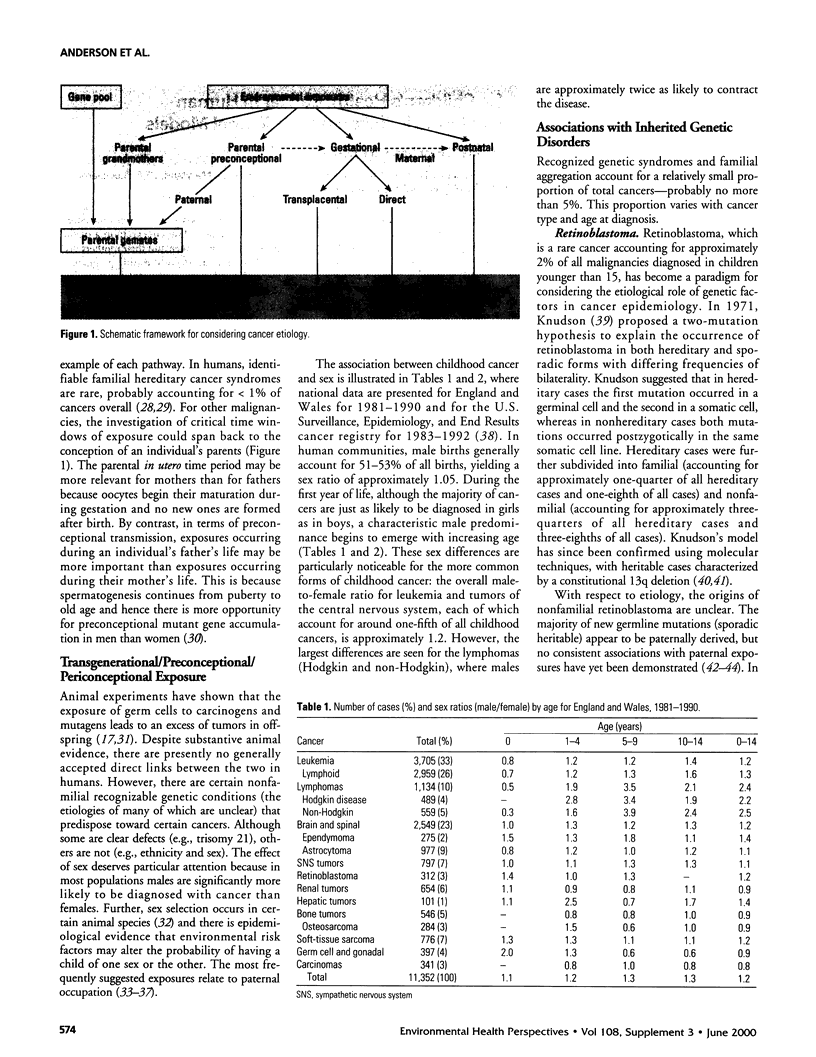
In humans, cancer may be caused by genetics and environmental exposures; however, in the majority of instances the identification of the critical time window of exposure is problematic. The evidence for exposures occurring during the preconceptional period that have an association with childhood or adulthood cancers is equivocal. Agents definitely related to cancer in children, and adulthood if exposure occurs in utero, include: maternal exposure to ionizing radiation during pregnancy and childhood leukemia and certain other cancers, and maternal use of diethylstilbestrol during pregnancy and clear-cell adenocarcinoma of the vagina of their daughters. The list of environmental exposures that occur during the perinatal/postnatal period with potential to increase the risk of cancer is lengthening, but evidence available to date is inconsistent and inconclusive. In animal models, preconceptional carcinogenesis has been demonstrated for a variety of types of radiation and chemicals, with demonstrated sensitivity for all stages from fetal gonocytes to postmeiotic germ cells. Transplacental and neonatal carcinogenesis show marked ontogenetic stage specificity in some cases. Mechanistic factors include the number of cells at risk, the rate of cell division, the development of differentiated characteristics including the ability to activate and detoxify carcinogens, the presence of stem cells, and possibly others. Usefulness for human risk estimation would be strengthened by the study of these factors in more than one species, and by a focus on specific human risk issues.
Full text
PDF















Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- AGER E. A., SCHUMAN L. M., WALLACE H. M., ROSENFIELD A. B., GULLEN W. H. AN EPIDEMIOLOGICAL STUDY OF CHILDHOOD LEUKEMIA. J Chronic Dis. 1965 Feb;18:113–132. doi: 10.1016/0021-9681(65)90096-2. [DOI] [PubMed] [Google Scholar]
- Alexandrov V. A. Blastomogenic effect of dimethylnitrosamine on pregnant rats and their offspring. Nature. 1968 Apr 20;218(5138):280–281. doi: 10.1038/218280a0. [DOI] [PubMed] [Google Scholar]
- Althoff J., Grandjean C., Pour P. Transplacental effect of nitrosamines in Syrian hamsters. IV. Metabolites of dipropyl- and dibutylnitrosamine. Z Krebsforsch Klin Onkol Cancer Res Clin Oncol. 1977 Nov 18;90(2):119–126. doi: 10.1007/BF00285318. [DOI] [PubMed] [Google Scholar]
- Althoff J., Pour P., Grandjean C., Eagen M. Transplacental effects of nitrosamines in Syrian hamsters: I. Dibutylnitrosamine and nitrosohexamethyleneimine. Z Krebsforsch Klin Onkol Cancer Res Clin Oncol. 1976 May 3;86(1):69–75. doi: 10.1007/BF00304935. [DOI] [PubMed] [Google Scholar]
- Anderson L. M., Hagiwara A., Kovatch R. M., Rehm S., Rice J. M. Transplacental initiation of liver, lung, neurogenic, and connective tissue tumors by N-nitroso compounds in mice. Fundam Appl Toxicol. 1989 Apr;12(3):604–620. doi: 10.1016/0272-0590(89)90033-x. [DOI] [PubMed] [Google Scholar]
- Anderson L. M., Hecht S. S., Dixon D. E., Dove L. F., Kovatch R. M., Amin S., Hoffmann D., Rice J. M. Evaluation of the transplacental tumorigenicity of the tobacco-specific carcinogen 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone in mice. Cancer Res. 1989 Jul 15;49(14):3770–3775. [PubMed] [Google Scholar]
- Anderson L. M., Hecht S. S., Kovatch R. M., Amin S., Hoffmann D., Rice J. M. Tumorigenicity of the tobacco-specific carcinogen 4-(methyl-nitrosamino)-1-(3-pyridyl)-1-butanone in infant mice. Cancer Lett. 1991 Jul 4;58(3):177–181. doi: 10.1016/0304-3835(91)90097-2. [DOI] [PubMed] [Google Scholar]
- Anderson L. M., Jones A. B., Riggs C. W., Kovatch R. M. Modification of transplacental tumorigenesis by 3-methylcholanthrene in mice by genotype at the Ah locus and pretreatment with beta-naphthoflavone. Cancer Res. 1989 Apr 1;49(7):1676–1681. [PubMed] [Google Scholar]
- Anderson L. M., Jones A. B., Riggs C. W., Ohshima M. Fetal mouse susceptibility to transplacental lung and liver carcinogenesis by 3-methylcholanthrene: positive correlation with responsiveness to inducers of aromatic hydrocarbon metabolism. Carcinogenesis. 1985 Sep;6(9):1389–1393. doi: 10.1093/carcin/6.9.1389. [DOI] [PubMed] [Google Scholar]
- Anderson L. M., Ruskie S., Carter J., Pittinger S., Kovatch R. M., Riggs C. W. Fetal mouse susceptibility to transplacental carcinogenesis: differential influence of Ah receptor phenotype on effects of 3-methylcholanthrene, 12-dimethylbenz[a]anthracene, and benzo[a]pyrene. Pharmacogenetics. 1995 Dec;5(6):364–372. doi: 10.1097/00008571-199512000-00005. [DOI] [PubMed] [Google Scholar]
- Anisimov V. N., Gvardina O. E. N-nitrosomethylurea-induced carcinogenesis in the progeny of male rats of different ages. Mutat Res. 1995 Feb;316(3):139–145. doi: 10.1016/0921-8734(95)90007-1. [DOI] [PubMed] [Google Scholar]
- Ansell P., Bull D., Roman E. Childhood leukaemia and intramuscular vitamin K: findings from a case-control study. BMJ. 1996 Jul 27;313(7051):204–205. doi: 10.1136/bmj.313.7051.204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arfellini G., Grilli S., Prodi G. In vivo DNA repair after N-methyl-N-nitrosourea administration to rats of different ages. Z Krebsforsch Klin Onkol Cancer Res Clin Oncol. 1978 May 31;91(2):157–164. doi: 10.1007/BF00284022. [DOI] [PubMed] [Google Scholar]
- Auerbach A. D. Fanconi anemia and leukemia: tracking the genes. Leukemia. 1992;6 (Suppl 1):1–4. [PubMed] [Google Scholar]
- Babosa M., Baki M., Bodrogi I., Gundy S. A study of children, fathered by men treated for testicular cancer, conceived before, during, and after chemotherapy. Med Pediatr Oncol. 1994;22(1):33–38. doi: 10.1002/mpo.2950220107. [DOI] [PubMed] [Google Scholar]
- Bader J. L., Miller R. W. Neurofibromatosis and childhood leukemia. J Pediatr. 1978 Jun;92(6):925–929. doi: 10.1016/s0022-3476(78)80362-x. [DOI] [PubMed] [Google Scholar]
- Ballesta F., Queralt R., Gómez D., Solsona E., Guitart M., Ezquerra M., Moreno J., Oliva R. Parental origin and meiotic stage of non-disjunction in 139 cases of trisomy 21. Ann Genet. 1999;42(1):11–15. [PubMed] [Google Scholar]
- Bearer C. F. How are children different from adults? Environ Health Perspect. 1995 Sep;103 (Suppl 6):7–12. doi: 10.1289/ehp.95103s67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beebe L. E., Kim Y. E., Amin S., Riggs C. W., Kovatch R. M., Anderson L. M. Comparison of transplacental and neonatal initiation of mouse lung and liver tumors by N-nitrosodimethylamine (NDMA) and 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK) and promotability by a polychlorinated biphenyls mixture (Aroclor 1254). Carcinogenesis. 1993 Aug;14(8):1545–1548. doi: 10.1093/carcin/14.8.1545. [DOI] [PubMed] [Google Scholar]
- Benjamin S. A., Saunders W. J., Angleton G. M., Lee A. C. Radiation carcinogenesis in dogs irradiated during prenatal and postnatal development. J Radiat Res. 1991 Dec;32 (Suppl 2):86–103. doi: 10.1269/jrr.32.supplement2_86. [DOI] [PubMed] [Google Scholar]
- Berleur M. P., Cordier S. The role of chemical, physical, or viral exposures and health factors in neurocarcinogenesis: implications for epidemiologic studies of brain tumors. Cancer Causes Control. 1995 May;6(3):240–256. doi: 10.1007/BF00051796. [DOI] [PubMed] [Google Scholar]
- Bhatia S., Neglia J. P. Epidemiology of childhood acute myelogenous leukemia. J Pediatr Hematol Oncol. 1995 May;17(2):94–100. doi: 10.1097/00043426-199505000-00002. [DOI] [PubMed] [Google Scholar]
- Bialkowski K., Bialkowska A., Anderson L. M., Kasprzak K. S. Higher activity of 8-oxo-2'-deoxyguanosine 5'-triphosphate pyrophosphohydrolase (8-oxo-dGTPase) coincides with lower background levels of 8-oxo-2'-deoxyguanosine in DNA of fetal compared with maternal mouse organs. Free Radic Biol Med. 1999 Jul;27(1-2):90–94. doi: 10.1016/s0891-5849(99)00040-4. [DOI] [PubMed] [Google Scholar]
- Births in Great Britain resulting from assisted conception, 1978-87. MRC Working Party on Children Conceived by In Vitro Fertilisation. BMJ. 1990 May 12;300(6734):1229–1233. doi: 10.1136/bmj.300.6734.1229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blatt J., Olshan A. F., Lee P. A., Ross J. L. Neuroblastoma and related tumors in Turner's syndrome. J Pediatr. 1997 Nov;131(5):666–670. doi: 10.1016/s0022-3476(97)70090-8. [DOI] [PubMed] [Google Scholar]
- Blot W. J., Henderson B. E., Boice J. D., Jr Childhood cancer in relation to cured meat intake: review of the epidemiological evidence. Nutr Cancer. 1999;34(1):111–118. doi: 10.1207/S15327914NC340115. [DOI] [PubMed] [Google Scholar]
- Boylan E. S., Calhoon R. E. Mammary tumorigenesis in the rat following prenatal exposure to diethylstilbestrol and postnatal treatment with 7,12-dimethylbenz[a]anthracene. J Toxicol Environ Health. 1979 Nov;5(6):1059–1071. doi: 10.1080/15287397909529814. [DOI] [PubMed] [Google Scholar]
- Branstetter D. G., Moseley P. P. Effect of lung development on the histological pattern of lung tumors induced by ethylnitrosourea in the C3HeB/FeJ mouse. Exp Lung Res. 1991 Mar-Apr;17(2):169–179. doi: 10.3109/01902149109064409. [DOI] [PubMed] [Google Scholar]
- Branstetter D. G., Stoner G. D., Budd C., Conran P. B., Goldblatt P. J. Effect of gestational development of lung tumor size and morphology in the mouse. Cancer Res. 1988 Jan 15;48(2):379–386. [PubMed] [Google Scholar]
- Branstetter D. G., Stoner G. D., Budd C., Conran P. B., Goldblatt P. J. Relationship between in utero development of the mouse liver and tumor development following transplacental exposure to ethylnitrosourea. Cancer Res. 1989 Jul 1;49(13):3620–3626. [PubMed] [Google Scholar]
- Branstetter D. G., Stoner G. D., Schut H. A., Senitzer D., Conran P. B., Goldblatt P. J. Ethylnitrosourea-induced transplacental carcinogenesis in the mouse: tumor response, DNA binding, and adduct formation. Cancer Res. 1987 Jan 15;47(2):348–352. [PubMed] [Google Scholar]
- Bunin G. R., Meadows A. T., Emanuel B. S., Buckley J. D., Woods W. G., Hammond G. D. Pre- and postconception factors associated with sporadic heritable and nonheritable retinoblastoma. Cancer Res. 1989 Oct 15;49(20):5730–5735. [PubMed] [Google Scholar]
- Bunin G. R., Petrakova A., Meadows A. T., Emanuel B. S., Buckley J. D., Woods W. G., Hammond G. D. Occupations of parents of children with retinoblastoma: a report from the Children's Cancer Study Group. Cancer Res. 1990 Nov 15;50(22):7129–7133. [PubMed] [Google Scholar]
- Börzsönyi M., Pintér A., Surján A., Farkas I. Transplacental induction of lymphomas in Swiss mice by carbendazim and sodium nitrite. Int J Cancer. 1976 Jun 15;17(6):742–747. doi: 10.1002/ijc.2910170609. [DOI] [PubMed] [Google Scholar]
- Cahill D. F., Wright J. F., Godbold J. H., Ward J. M., Laskey J. W., Tompkins E. A. Neoplastic and life-span effects of chronic exposure to tritium. II. Rats exposed in utero. J Natl Cancer Inst. 1975 Nov;55(5):1165–1169. doi: 10.1093/jnci/55.5.1165. [DOI] [PubMed] [Google Scholar]
- Call K. M., Glaser T., Ito C. Y., Buckler A. J., Pelletier J., Haber D. A., Rose E. A., Kral A., Yeger H., Lewis W. H. Isolation and characterization of a zinc finger polypeptide gene at the human chromosome 11 Wilms' tumor locus. Cell. 1990 Feb 9;60(3):509–520. doi: 10.1016/0092-8674(90)90601-a. [DOI] [PubMed] [Google Scholar]
- Cattanach B. M., Papworth D., Patrick G., Goodhead D. T., Hacker T., Cobb L., Whitehill E. Investigation of lung tumour induction in C3H/HeH mice, with and without tumour promotion with urethane, following paternal X-irradiation. Mutat Res. 1998 Jul 17;403(1-2):1–12. doi: 10.1016/s0027-5107(97)00322-9. [DOI] [PubMed] [Google Scholar]
- Cattanach B. M., Patrick G., Papworth D., Goodhead D. T., Hacker T., Cobb L., Whitehill E. Investigation of lung tumour induction in BALB/cJ mice following paternal X-irradiation. Int J Radiat Biol. 1995 May;67(5):607–615. doi: 10.1080/09553009514550721. [DOI] [PubMed] [Google Scholar]
- Chandley A. C. On the parental origin of de novo mutation in man. J Med Genet. 1991 Apr;28(4):217–223. doi: 10.1136/jmg.28.4.217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen B. P., Berman J. J., Ching W. M., Rice J. M. DNA breakage by methyl methanesulfonate and its repair in brain and liver cells cultured from fetal rat and mouse. Chem Biol Interact. 1983 Apr-May;44(1-2):63–77. doi: 10.1016/0009-2797(83)90130-8. [DOI] [PubMed] [Google Scholar]
- Chen T. T., Heidelberger C. Quantitative studies on the malignant transformation of mouse prostate cells by carcinogenic hydrocarbons in vitro. Int J Cancer. 1969 Mar 15;4(2):166–178. doi: 10.1002/ijc.2910040207. [DOI] [PubMed] [Google Scholar]
- Clutton-Brock T. H., Iason G. R. Sex ratio variation in mammals. Q Rev Biol. 1986 Sep;61(3):339–374. doi: 10.1086/415033. [DOI] [PubMed] [Google Scholar]
- Cnattingius S., Zack M. M., Ekbom A., Gunnarskog J., Kreuger A., Linet M., Adami H. O. Prenatal and neonatal risk factors for childhood lymphatic leukemia. J Natl Cancer Inst. 1995 Jun 21;87(12):908–914. doi: 10.1093/jnci/87.12.908. [DOI] [PubMed] [Google Scholar]
- Cnattingius S., Zack M., Ekbom A., Gunnarskog J., Linet M., Adami H. O. Prenatal and neonatal risk factors for childhood myeloid leukemia. Cancer Epidemiol Biomarkers Prev. 1995 Jul-Aug;4(5):441–445. [PubMed] [Google Scholar]
- Coccia P., Salmona M., Diomede L., Citti L., Mariani L., Romano M. Liver DNA alkylation after a single carcinogenic dose of dimethylnitrosamine to newborn and adult CFW Swiss mice. Chem Biol Interact. 1988;68(3-4):259–271. doi: 10.1016/0009-2797(88)90020-8. [DOI] [PubMed] [Google Scholar]
- Coppes M. J., Haber D. A., Grundy P. E. Genetic events in the development of Wilms' tumor. N Engl J Med. 1994 Sep 1;331(9):586–590. doi: 10.1056/NEJM199409013310906. [DOI] [PubMed] [Google Scholar]
- Coppes M. J., Liefers G. J., Higuchi M., Zinn A. B., Balfe J. W., Williams B. R. Inherited WT1 mutation in Denys-Drash syndrome. Cancer Res. 1992 Nov 1;52(21):6125–6128. [PubMed] [Google Scholar]
- Cosgrove G. E., Selby P. B., Upton A. C., Mitchell T. J., Steele M. H., Russell W. L. Lifespan and autopsy findings in the first-generation offspring of X-irradiated male mice. Mutat Res. 1993 Sep;319(1):71–79. doi: 10.1016/0165-1218(93)90032-9. [DOI] [PubMed] [Google Scholar]
- Craddock V. M., Henderson A. R., Gash S. Repair and replication of DNA in rat brain and liver during foetal and post-natal development, in relation to nitroso-alkyl-urea induced carcinogenesis. J Cancer Res Clin Oncol. 1984;108(1):30–35. doi: 10.1007/BF00390970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DAM H., DYGGVE H., LARSEN H., PLUM P. The relation of vitamin K deficiency to hemorrhagic disease of the newborn. Adv Pediatr. 1952 Jan;5:129–153. [PubMed] [Google Scholar]
- Daher A., Varin M., Lamontagne Y., Oth D. Effect of pre-conceptional external or internal irradiation of N5 male mice and the risk of leukemia in their offspring. Carcinogenesis. 1998 Sep;19(9):1553–1558. doi: 10.1093/carcin/19.9.1553. [DOI] [PubMed] [Google Scholar]
- Di Majo V., Coppola M., Rebessi S., Covelli V. Age-related susceptibility of mouse liver to induction of tumors by neutrons. Radiat Res. 1990 Nov;124(2):227–234. [PubMed] [Google Scholar]
- Dickinson H. O., Parker L., Binks K., Wakeford R., Smith J. The sex ratio of children in relation to paternal preconceptional radiation dose: a study in Cumbria, northern England. J Epidemiol Community Health. 1996 Dec;50(6):645–652. doi: 10.1136/jech.50.6.645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dickman P. W., Hakulinen T., Luostarinen T., Pukkala E., Sankila R., Söderman B., Teppo L. Survival of cancer patients in Finland 1955-1994. Acta Oncol. 1999;38 (Suppl 12):1–103. doi: 10.1080/028418699432996. [DOI] [PubMed] [Google Scholar]
- Diwan B. A., Kasprzak K. S., Rice J. M. Transplacental carcinogenic effects of nickel(II) acetate in the renal cortex, renal pelvis and adenohypophysis in F344/NCr rats. Carcinogenesis. 1992 Aug;13(8):1351–1357. doi: 10.1093/carcin/13.8.1351. [DOI] [PubMed] [Google Scholar]
- Diwan B. A., Meier H. Strain- and age-dependent transplacental carcinogenesis by 1-ethyl-1-nitrosourea in inbred strains of mice. Cancer Res. 1974 Apr;34(4):764–770. [PubMed] [Google Scholar]
- Diwan B. A., Meier H. Transplacental carcinogenic effects of diethylnitrosamine in mice. Naturwissenschaften. 1976 Oct;63(10):487–488. doi: 10.1007/BF00624591. [DOI] [PubMed] [Google Scholar]
- Diwan B. A., Rehm S., Rice J. M. Age- and dose-dependent transplacental carcinogenesis by N-nitrosoethylurea in Syrian golden hamsters. J Cancer Res Clin Oncol. 1996;122(11):643–652. doi: 10.1007/BF01209026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diwan B. A., Rice J. M. Effect of stage of development on frequency and pathogenesis of kidney tumors induced in Noble (Nb) rats exposed prenatally or neonatally to N-nitrosoethylurea. Carcinogenesis. 1995 Sep;16(9):2023–2028. doi: 10.1093/carcin/16.9.2023. [DOI] [PubMed] [Google Scholar]
- Diwan B. A., Riggs C. W., Logsdon D., Haines D. C., Olivero O. A., Rice J. M., Yuspa S. H., Poirier M. C., Anderson L. M. Multiorgan transplacental and neonatal carcinogenicity of 3'-azido-3'-deoxythymidine in mice. Toxicol Appl Pharmacol. 1999 Nov 15;161(1):82–99. doi: 10.1006/taap.1999.8782. [DOI] [PubMed] [Google Scholar]
- Dockerty J. D., Skegg D. C., Elwood J. M., Herbison G. P., Becroft D. M., Lewis M. E. Infections, vaccinations, and the risk of childhood leukaemia. Br J Cancer. 1999 Jul;80(9):1483–1489. doi: 10.1038/sj.bjc.6690548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Donovan P. J., Smith G. T. Cell sensitivity to transplacental mutagenesis by N-ethyl-N-nitrosourea is greatest during early gestation in the Syrian hamster. Mutat Res. 1999 Jun 1;427(1):47–58. doi: 10.1016/S0027-5107(99)00085-8. [DOI] [PubMed] [Google Scholar]
- Doyle P., Bunch K. J., Beral V., Draper G. J. Cancer incidence in children conceived with assisted reproduction technology. Lancet. 1998 Aug 8;352(9126):452–453. doi: 10.1016/s0140-6736(05)79186-8. [DOI] [PubMed] [Google Scholar]
- Doyle P. The outcome of multiple pregnancy. Hum Reprod. 1996 Dec;11 (Suppl 4):110–120. doi: 10.1093/humrep/11.suppl_4.110. [DOI] [PubMed] [Google Scholar]
- Draper G. J., Kroll M. E., Stiller C. A. Childhood cancer. Cancer Surv. 1994;19-20:493–517. [PubMed] [Google Scholar]
- Draper G. J., Little M. P., Sorahan T., Kinlen L. J., Bunch K. J., Conquest A. J., Kendall G. M., Kneale G. W., Lancashire R. J., Muirhead C. R. Cancer in the offspring of radiation workers: a record linkage study. BMJ. 1997 Nov 8;315(7117):1181–1188. [PMC free article] [PubMed] [Google Scholar]
- Easton D., Peto J. The contribution of inherited predisposition to cancer incidence. Cancer Surv. 1990;9(3):395–416. [PubMed] [Google Scholar]
- Edwards A. J., Anderson D., Brinkworth M. H., Myers B., Parry J. M. An investigation of male-mediated F1 effects in mice treated acutely and sub-chronically with urethane. Teratog Carcinog Mutagen. 1999;19(2):87–103. [PubMed] [Google Scholar]
- Ekbom A., Trichopoulos D., Adami H. O., Hsieh C. C., Lan S. J. Evidence of prenatal influences on breast cancer risk. Lancet. 1992 Oct 24;340(8826):1015–1018. doi: 10.1016/0140-6736(92)93019-j. [DOI] [PubMed] [Google Scholar]
- Ekelund H., Finnström O., Gunnarskog J., Källén B., Larsson Y. Administration of vitamin K to newborn infants and childhood cancer. BMJ. 1993 Jul 10;307(6896):89–91. doi: 10.1136/bmj.307.6896.89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ernst H., Emura M., Bellmann B., Seinsch D., Mohr U. Failure to transmit diethylnitrosamine tumorigenicity from transplacentally exposed F1 generation Syrian hamsters to the respiratory tract of F2 and F3 generations. Cancer Res. 1987 Oct 1;47(19):5112–5115. [PubMed] [Google Scholar]
- Fear N. T., Roman E., Reeves G., Pannett B. Childhood cancer and paternal employment in agriculture: the role of pesticides. Br J Cancer. 1998 Mar;77(5):825–829. doi: 10.1038/bjc.1998.134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feinberg A. P. Genomic imprinting and gene activation in cancer. Nat Genet. 1993 Jun;4(2):110–113. doi: 10.1038/ng0693-110. [DOI] [PubMed] [Google Scholar]
- Ford A. M., Pombo-de-Oliveira M. S., McCarthy K. P., MacLean J. M., Carrico K. C., Vincent R. F., Greaves M. Monoclonal origin of concordant T-cell malignancy in identical twins. Blood. 1997 Jan 1;89(1):281–285. [PubMed] [Google Scholar]
- Ford A. M., Ridge S. A., Cabrera M. E., Mahmoud H., Steel C. M., Chan L. C., Greaves M. In utero rearrangements in the trithorax-related oncogene in infant leukaemias. Nature. 1993 May 27;363(6427):358–360. doi: 10.1038/363358a0. [DOI] [PubMed] [Google Scholar]
- Fox R. R., Diwan B. A., Meier H. Transplacental induction of primary renal tumors in rabbits treated with 1-ethyl-1-nitrosourea. J Natl Cancer Inst. 1975 Jun;54(6):1439–1448. doi: 10.1093/jnci/54.6.1439. [DOI] [PubMed] [Google Scholar]
- Fox R. R., Meier H., Pottathil R., Bedigian H. G. Transplacental teratogenic and carcinogenic effects in rabbits chronically treated with N-ethyl-N-nitrosourea. J Natl Cancer Inst. 1980 Sep;65(3):607–614. [PubMed] [Google Scholar]
- Francis A. J., Anderson D., Evans J. G., Jenkinson P. C., Godbert P. Tumours and malformations in the adult offspring of cyclophosphamide-treated and control male rats--preliminary communication. Mutat Res. 1990 Apr;229(2):239–246. doi: 10.1016/0027-5107(90)90097-n. [DOI] [PubMed] [Google Scholar]
- Fujii K. Evaluation of the newborn mouse model for chemical tumorigenesis. Carcinogenesis. 1991 Aug;12(8):1409–1415. doi: 10.1093/carcin/12.8.1409. [DOI] [PubMed] [Google Scholar]
- GILES D., HEWITT D., STEWART A., WEBB J. Malignant disease in childhood and diagnostic irradiation in utero. Lancet. 1956 Sep 1;271(6940):447–447. doi: 10.1016/s0140-6736(56)91923-7. [DOI] [PubMed] [Google Scholar]
- Gardner M. J. Paternal occupations of children with leukemia. BMJ. 1992 Sep 19;305(6855):715–715. doi: 10.1136/bmj.305.6855.715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gardner M. J., Snee M. P., Hall A. J., Powell C. A., Downes S., Terrell J. D. Results of case-control study of leukaemia and lymphoma among young people near Sellafield nuclear plant in West Cumbria. BMJ. 1990 Feb 17;300(6722):423–429. doi: 10.1136/bmj.300.6722.423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibson R. E., D'Ambrosio S. M. Differing levels of excision repair in human fetal dermis and brain cells. Photochem Photobiol. 1982 Feb;35(2):181–185. doi: 10.1111/j.1751-1097.1982.tb03829.x. [DOI] [PubMed] [Google Scholar]
- Gilman E. A., Wilson L. M., Kneale G. W., Waterhouse J. A. Childhood cancers and their association with pregnancy drugs and illnesses. Paediatr Perinat Epidemiol. 1989 Jan;3(1):66–94. doi: 10.1111/j.1365-3016.1989.tb00371.x. [DOI] [PubMed] [Google Scholar]
- Giusti R. M., Iwamoto K., Hatch E. E. Diethylstilbestrol revisited: a review of the long-term health effects. Ann Intern Med. 1995 May 15;122(10):778–788. doi: 10.7326/0003-4819-122-10-199505150-00008. [DOI] [PubMed] [Google Scholar]
- Goerttler K., Loehrke H., Hesse B., Milz A., Schweizer J. Diaplacental initiation of NMRI mice with 7,12-dimethylbenz[a]anthracene during gestation days 6--20 and postnatal treatment of the F1-generation with the phorbol ester 12-O-tetradecanoylphorbol-13-acetate: tumor incidence in organs other than the skin. Carcinogenesis. 1981;2(11):1087–1094. doi: 10.1093/carcin/2.11.1087. [DOI] [PubMed] [Google Scholar]
- Goerttler K., Loehrke H., Schweizer J., Hesse B. Two-stage skin carcinogenesis by systemic initiation of pregnant mice with 7,12-dimethylbenz(a)anthracene during gestation days 6-20 and postnatal promotion of the F 1-generation with the phorbol ester 12-tetradecanoylphorbol-13-acetate. J Cancer Res Clin Oncol. 1980;98(3):267–275. doi: 10.1007/BF00410789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Golding J., Greenwood R., Birmingham K., Mott M. Childhood cancer, intramuscular vitamin K, and pethidine given during labour. BMJ. 1992 Aug 8;305(6849):341–346. doi: 10.1136/bmj.305.6849.341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodrich D. W., Lee W. H. The molecular genetics of retinoblastoma. Cancer Surv. 1990;9(3):529–554. [PubMed] [Google Scholar]
- Goth R., Rajewsky M. F. Persistence of O6-ethylguanine in rat-brain DNA: correlation with nervous system-specific carcinogenesis by ethylnitrosourea. Proc Natl Acad Sci U S A. 1974 Mar;71(3):639–643. doi: 10.1073/pnas.71.3.639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greaves M. F. Aetiology of acute leukaemia. Lancet. 1997 Feb 1;349(9048):344–349. doi: 10.1016/s0140-6736(96)09412-3. [DOI] [PubMed] [Google Scholar]
- Hawkins M. M., Draper G. J., Smith R. A. Cancer among 1,348 offspring of survivors of childhood cancer. Int J Cancer. 1989 Jun 15;43(6):975–978. doi: 10.1002/ijc.2910430604. [DOI] [PubMed] [Google Scholar]
- Hawkins M. M. Is there evidence of a therapy-related increase in germ cell mutation among childhood cancer survivors? J Natl Cancer Inst. 1991 Nov 20;83(22):1643–1650. doi: 10.1093/jnci/83.22.1643. [DOI] [PubMed] [Google Scholar]
- Hedenskog M., Sjögren M., Cederberg H., Rannug U. Induction of germline-length mutations at the minisatellites PC-1 and PC-2 in male mice exposed to polychlorinated biphenyls and diesel exhaust emissions. Environ Mol Mutagen. 1997;30(3):254–259. [PubMed] [Google Scholar]
- Hemminki E., Gissler M., Toukomaa H. Exposure to female hormone drugs during pregnancy: effect on malformations and cancer. Br J Cancer. 1999 Jun;80(7):1092–1097. doi: 10.1038/sj.bjc.6690469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herbst A. L., Ulfelder H., Poskanzer D. C. Adenocarcinoma of the vagina. Association of maternal stilbestrol therapy with tumor appearance in young women. N Engl J Med. 1971 Apr 15;284(15):878–881. doi: 10.1056/NEJM197104222841604. [DOI] [PubMed] [Google Scholar]
- Hsieh C. C., Tzonou A., Zavitsanos X., Kaklamani E., Lan S. J., Trichopoulos D. Age at first establishment of chronic hepatitis B virus infection and hepatocellular carcinoma risk. A birth order study. Am J Epidemiol. 1992 Nov 1;136(9):1115–1121. doi: 10.1093/oxfordjournals.aje.a116577. [DOI] [PubMed] [Google Scholar]
- Huff V., Meadows A., Riccardi V. M., Strong L. C., Saunders G. F. Parental origin of de novo constitutional deletions of chromosomal band 11p13. Am J Hum Genet. 1990 Jul;47(1):155–160. [PMC free article] [PubMed] [Google Scholar]
- Ivankovic S., Druckrey H. Transplacentare Erzeugung maligner Tumoren des Nervensystems. I. Athyl-nitroso-harnstoff (ANH) an BD IX-Ratten. Z Krebsforsch. 1968;71(4):320–360. [PubMed] [Google Scholar]
- Ivankovic S., Schmähl D., Zeller W. J. N-Demethylierung des Carcinogens Dimethylnitrosamin durch embryonales menschliches Gewebe. Z Krebsforsch Klin Onkol Cancer Res Clin Oncol. 1974;81(3-4):269–272. doi: 10.1007/BF00305029. [DOI] [PubMed] [Google Scholar]
- Iwasaki T., Hashimoto N., Endoh D., Imanisi T., Itakura C., Sato F. Life span and tumours in the first-generation offspring of the gamma-irradiated male mouse. Int J Radiat Biol. 1996 Apr;69(4):487–492. doi: 10.1080/095530096145788. [DOI] [PubMed] [Google Scholar]
- Jannetti R. A., Anderson L. M. Dimethylnitrosamine demethylase activity in fetal, suckling, and maternal mouse liver and its transplacental and transmammary induction by polychlorinated biphenyls. J Natl Cancer Inst. 1981 Aug;67(2):461–466. [PubMed] [Google Scholar]
- Jull J. W., Streeter D. J., Sutherland L. The mechanism of induction of ovarian tumors in the mouse by 7,12-dimethylbenz-[alpha]anthracene. I. Effect of steroid hormones and carcinogen concentration in vivo. J Natl Cancer Inst. 1966 Oct;37(4):409–420. [PubMed] [Google Scholar]
- Jurgelski W., Jr, Hudson P., Falk H. L. Tissue differentiation and susceptibility to embryonal tumor induction by ethylnitrosourea in the opossum. Natl Cancer Inst Monogr. 1979 May;(51):123–158. [PubMed] [Google Scholar]
- Kakunaga T. The role of cell division in the malignant transformation of mouse cells treated with 3-methylcholanthrene. Cancer Res. 1975 Jul;35(7):1637–1642. [PubMed] [Google Scholar]
- Kalter H., Mandybur T. I., Ormsby I., Warkany J. Dose-related reduction by prenatal x-irradiation of the transplacental neurocarcinogenicity of ethylnitrosourea in rats. Cancer Res. 1980 Nov;40(11):3973–3976. [PubMed] [Google Scholar]
- Kato M. V., Ishizaki K., Shimizu T., Ejima Y., Tanooka H., Takayama J., Kaneko A., Toguchida J., Sasaki M. S. Parental origin of germ-line and somatic mutations in the retinoblastoma gene. Hum Genet. 1994 Jul;94(1):31–38. doi: 10.1007/BF02272838. [DOI] [PubMed] [Google Scholar]
- Kattan J., Tournade M. F., Culine S., Terrier-Lacombe M. J., Droz J. P. Adult Wilms' tumour: review of 22 cases. Eur J Cancer. 1994;30A(12):1778–1782. doi: 10.1016/0959-8049(94)00315-v. [DOI] [PubMed] [Google Scholar]
- Kauffman S. L. Susceptibility of fetal lung to transplacental 1-ethyl-1-nitrosourea: its relation to epithelial proliferation. J Natl Cancer Inst. 1976 Oct;57(4):821–825. doi: 10.1093/jnci/57.4.821. [DOI] [PubMed] [Google Scholar]
- Kingston J. E., Hawkins M. M., Draper G. J., Marsden H. B., Kinnier Wilson L. M. Patterns of multiple primary tumours in patients treated for cancer during childhood. Br J Cancer. 1987 Sep;56(3):331–338. doi: 10.1038/bjc.1987.199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klebanoff M. A., Read J. S., Mills J. L., Shiono P. H. The risk of childhood cancer after neonatal exposure to vitamin K. N Engl J Med. 1993 Sep 23;329(13):905–908. doi: 10.1056/NEJM199309233291301. [DOI] [PubMed] [Google Scholar]
- Kleihues P., Cooper H. K., Buecheler J., Kolar G. F., Diessner H. Mechanism of perinatal tumor induction by neuro-oncogenic alkylnitrosoureas and dialkylaryltriazenes. Natl Cancer Inst Monogr. 1979 May;(51):227–231. [PubMed] [Google Scholar]
- Kleihues P., Rajewsky M. F. Chemical neuro-oncogenesis: role of structural DNA modifications, DNA repair and neural target cell population. Prog Exp Tumor Res. 1984;27:1–16. doi: 10.1159/000408218. [DOI] [PubMed] [Google Scholar]
- Knudson A. G., Jr Mutation and cancer: statistical study of retinoblastoma. Proc Natl Acad Sci U S A. 1971 Apr;68(4):820–823. doi: 10.1073/pnas.68.4.820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knudson A. G., Jr, Strong L. C. Mutation and cancer: a model for Wilms' tumor of the kidney. J Natl Cancer Inst. 1972 Feb;48(2):313–324. [PubMed] [Google Scholar]
- Kobayashi N., Matsui I., Tanimura M., Nagahara N., Akatsuka J., Hirayama T., Sato K. Childhood neuroectodermal tumours and malignant lymphoma after maternal ovulation induction. Lancet. 1991 Oct 12;338(8772):955–955. doi: 10.1016/0140-6736(91)91829-j. [DOI] [PubMed] [Google Scholar]
- Kristensen P., Andersen A., Irgens L. M., Bye A. S., Sundheim L. Cancer in offspring of parents engaged in agricultural activities in Norway: incidence and risk factors in the farm environment. Int J Cancer. 1996 Jan 3;65(1):39–50. doi: 10.1002/(SICI)1097-0215(19960103)65:1<39::AID-IJC8>3.0.CO;2-2. [DOI] [PubMed] [Google Scholar]
- Likhachev A. J., Alekandrov V. A., Anisimov V. N., Bespalov V. G., Korsakov M. V., Ovsyannikov A. I., Popovich I. G., Napalkov N. P., Tomatis L. Persistence of methylated purines in the DNA of various rat fetal and maternal tissues and carcinogenesis in the offspring following a single transplacental dose of N-methyl-N-nitrosourea. Int J Cancer. 1983 Jun 15;31(6):779–784. doi: 10.1002/ijc.2910310618. [DOI] [PubMed] [Google Scholar]
- Lu L. J., Anderson L. M., Jones A. B., Moskal T. J., Salazar J. J., Hokanson J. A., Rice J. M. Persistence, gestation stage-dependent formation and interrelationship of benzo[a]pyrene-induced DNA adducts in mothers, placentae and fetuses of Erythrocebus patas monkeys. Carcinogenesis. 1993 Sep;14(9):1805–1813. doi: 10.1093/carcin/14.9.1805. [DOI] [PubMed] [Google Scholar]
- Lu L. J., Wang M. Y. Modulation of benzo[a]pyrene-induced covalent DNA modifications in adult and fetal mouse tissues by gestation stage. Carcinogenesis. 1990 Aug;11(8):1367–1372. doi: 10.1093/carcin/11.8.1367. [DOI] [PubMed] [Google Scholar]
- Lyster W. R. Altered sex ratio in children of divers. Lancet. 1982 Jul 17;2(8290):152–152. doi: 10.1016/s0140-6736(82)91113-8. [DOI] [PubMed] [Google Scholar]
- MIRVISH S., CIVIDALLI G., BERENBLUM I. SLOW ELIMINATION OF URETHAN IN RELATION TO ITS HIGH CARCINOGENICITY IN NEWBORN MICE. Proc Soc Exp Biol Med. 1964 Jun;116:265–268. doi: 10.3181/00379727-116-29220. [DOI] [PubMed] [Google Scholar]
- Mann J. R., Dodd H. E., Draper G. J., Waterhouse J. A., Birch J. M., Cartwright R. A., Hartley A. L., McKinney P. A., Stiller C. A. Congenital abnormalities in children with cancer and their relatives: results from a case-control study (IRESCC). Br J Cancer. 1993 Aug;68(2):357–363. doi: 10.1038/bjc.1993.340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsunaga E. Genetics of Wilms' tumor. Hum Genet. 1981;57(3):231–246. doi: 10.1007/BF00278936. [DOI] [PubMed] [Google Scholar]
- McKinney P. A., Juszczak E., Findlay E., Smith K. Case-control study of childhood leukaemia and cancer in Scotland: findings for neonatal intramuscular vitamin K. BMJ. 1998 Jan 17;316(7126):173–177. doi: 10.1136/bmj.316.7126.173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLaughlin J. R., King W. D., Anderson T. W., Clarke E. A., Ashmore J. P. Paternal radiation exposure and leukaemia in offspring: the Ontario case-control study. BMJ. 1993 Oct 16;307(6910):959–966. doi: 10.1136/bmj.307.6910.959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mertens A. C., Wen W., Davies S. M., Steinbuch M., Buckley J. D., Potter J. D., Robison L. L. Congenital abnormalities in children with acute leukemia: a report from the Children's Cancer Group. J Pediatr. 1998 Nov;133(5):617–623. doi: 10.1016/s0022-3476(98)70100-3. [DOI] [PubMed] [Google Scholar]
- Michels K. B., Trichopoulos D., Robins J. M., Rosner B. A., Manson J. E., Hunter D. J., Colditz G. A., Hankinson S. E., Speizer F. E., Willett W. C. Birthweight as a risk factor for breast cancer. Lancet. 1996 Dec 7;348(9041):1542–1546. doi: 10.1016/S0140-6736(96)03102-9. [DOI] [PubMed] [Google Scholar]
- Mili F., Khoury M. J., Flanders W. D., Greenberg R. S. Risk of childhood cancer for infants with birth defects. I. A record-linkage study, Atlanta, Georgia, 1968-1988. Am J Epidemiol. 1993 Mar 15;137(6):629–638. doi: 10.1093/oxfordjournals.aje.a116720. [DOI] [PubMed] [Google Scholar]
- Mili F., Lynch C. F., Khoury M. J., Flanders W. D., Edmonds L. D. Risk of childhood cancer for infants with birth defects. II. A record-linkage study, Iowa, 1983-1989. Am J Epidemiol. 1993 Mar 15;137(6):639–644. doi: 10.1093/oxfordjournals.aje.a116721. [DOI] [PubMed] [Google Scholar]
- Miller R. W. Special susceptibility of the child to certain radiation-induced cancers. Environ Health Perspect. 1995 Sep;103 (Suppl 6):41–44. doi: 10.1289/ehp.95103s641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller R. W., Young J. L., Jr, Novakovic B. Childhood cancer. Cancer. 1995 Jan 1;75(1 Suppl):395–405. doi: 10.1002/1097-0142(19950101)75:1+<395::aid-cncr2820751321>3.0.co;2-w. [DOI] [PubMed] [Google Scholar]
- Mohr U., Dasenbrock C., Tillmann T., Kohler M., Kamino K., Hagemann G., Morawietz G., Campo E., Cazorla M., Fernandez P. Possible carcinogenic effects of X-rays in a transgenerational study with CBA mice. Carcinogenesis. 1999 Feb;20(2):325–332. doi: 10.1093/carcin/20.2.325. [DOI] [PubMed] [Google Scholar]
- Mohr U., Emura M., Aufderheide M., Riebe M., Ernst H. Possible role of genetic predisposition in multigeneration carcinogenesis. IARC Sci Publ. 1989;(96):93–103. [PubMed] [Google Scholar]
- Mohr U., Emura M., Kamino K., Steinmann J., Kohler M., Morawietz G., Dasenbrock C., Tomatis L. Increased risk of cancer in the descendants of Syrian hamsters exposed prenatally to diethylnitrosamine (DEN). Int J Cancer. 1995 Sep 27;63(1):86–91. doi: 10.1002/ijc.2910630116. [DOI] [PubMed] [Google Scholar]
- Mohr U., Reznik-Schüller H., Emura M. Tissue differentiation as a prerequisite for transplacental carcinogenesis in the hamster respiratory system, with specific respect to the trachea. Natl Cancer Inst Monogr. 1979 May;(51):117–122. [PubMed] [Google Scholar]
- Mohr U., Reznik-Schüller H., Reznik G., Hilfrich J. Transplacental effects of diethylnitrosamine in Syrian hamsters as related to different days of administration during pregnancy. J Natl Cancer Inst. 1975 Sep;55(3):681–683. doi: 10.1093/jnci/55.3.681. [DOI] [PubMed] [Google Scholar]
- Mole R. H. Childhood cancer after prenatal exposure to diagnostic X-ray examinations in Britain. Br J Cancer. 1990 Jul;62(1):152–168. doi: 10.1038/bjc.1990.249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morris E. P., Burns F. J., Albert R. E. Transplacental skin tumor initiation of Ha/ICR mice at different fetal ages. Cancer Res. 1983 Sep;43(9):4271–4274. [PubMed] [Google Scholar]
- Müller R., Rajewsky M. F. Elimination of O6-ethylguanine from the DNA of brain, liver, and other rat tissues exposed to ethylnitrosourea at different stages of prenatal development. Cancer Res. 1983 Jun;43(6):2897–2904. [PubMed] [Google Scholar]
- Naito M., Naito Y., Ito A. Effect of age at treatment on the incidence and location of neurogenic tumors induced in Wistar rats by a single dose of N-ethyl-N-nitrosourea. Gan. 1981 Aug;72(4):569–577. [PubMed] [Google Scholar]
- Narod S. A., Stiller C., Lenoir G. M. An estimate of the heritable fraction of childhood cancer. Br J Cancer. 1991 Jun;63(6):993–999. doi: 10.1038/bjc.1991.216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newbold R. R., Bullock B. C., McLachlan J. A. Uterine adenocarcinoma in mice following developmental treatment with estrogens: a model for hormonal carcinogenesis. Cancer Res. 1990 Dec 1;50(23):7677–7681. [PubMed] [Google Scholar]
- Newbold R. R., Hanson R. B., Jefferson W. N., Bullock B. C., Haseman J., McLachlan J. A. Increased tumors but uncompromised fertility in the female descendants of mice exposed developmentally to diethylstilbestrol. Carcinogenesis. 1998 Sep;19(9):1655–1663. doi: 10.1093/carcin/19.9.1655. [DOI] [PubMed] [Google Scholar]
- Newbold R. R., McLachlan J. A. Vaginal adenosis and adenocarcinoma in mice exposed prenatally or neonatally to diethylstilbestrol. Cancer Res. 1982 May;42(5):2003–2011. [PubMed] [Google Scholar]
- Niederreither K., Harbers M., Chambon P., Dollé P. Expression of T:G mismatch-specific thymidine-DNA glycosylase and DNA methyl transferase genes during development and tumorigenesis. Oncogene. 1998 Sep 24;17(12):1577–1585. doi: 10.1038/sj.onc.1202072. [DOI] [PubMed] [Google Scholar]
- Nomura T. An analysis of the changing urethan response of the developing mouse embryo in relation to mortality, malformation, and neoplasm. Cancer Res. 1974 Sep;34(9):2217–2231. [PubMed] [Google Scholar]
- Nomura T. Induction of persistent hypersensitivity to lung tumorigenesis by in utero X-radiation in mice. Environ Mutagen. 1984;6(1):33–40. doi: 10.1002/em.2860060105. [DOI] [PubMed] [Google Scholar]
- Nomura T. Letter: Transmission of tumors and malformations to the next generation of mice subsequent to urethan treatment. Cancer Res. 1975 Jan;35(1):264–266. [PubMed] [Google Scholar]
- Nomura T., Okamoto E., Tateishi N., Kimura S., Isa Y. Tumor induction in the progeny of mice receiving 4-nitroquinoline 1-oxide and N-methyl-N-nitrosourethan during pregnancy or lactation. Cancer Res. 1974 Dec;34(12):3373–3378. [PubMed] [Google Scholar]
- Nomura T., Okamoto E. Transplacental carcinogenesis by urethan in mice: teratogenesis and carcinogenesis in relation to organogenesis. Gan. 1972 Dec;63(6):731–742. [PubMed] [Google Scholar]
- Nomura T. Parental exposure to x rays and chemicals induces heritable tumours and anomalies in mice. Nature. 1982 Apr 8;296(5857):575–577. doi: 10.1038/296575a0. [DOI] [PubMed] [Google Scholar]
- Nomura T. Paternal exposure to radiation and offspring cancer in mice: reanalysis and new evidence. J Radiat Res. 1991 Dec;32 (Suppl 2):64–72. doi: 10.1269/jrr.32.supplement2_64. [DOI] [PubMed] [Google Scholar]
- Nomura T. Sensitivity of a lung cell in the developing mouse embryo to tumor induction by urethan. Cancer Res. 1974 Dec;34(12):3363–3372. [PubMed] [Google Scholar]
- Nomura T., Takebe H., Okamoto E. Long retention of urethan transferred into newborn mice transplacentally, as a possible cause of high carcinogenesis. Gan. 1973 Feb;64(1):29–40. [PubMed] [Google Scholar]
- Nomura T. X-ray-induced germ-line mutation leading to tumors. Its manifestation in mice given urethane post-natally. Mutat Res. 1983 Jul;121(1):59–65. doi: 10.1016/0165-7992(83)90087-8. [DOI] [PubMed] [Google Scholar]
- Olsen J. H., Boice J. D., Jr, Fraumeni J. F., Jr Cancer in children of epileptic mothers and the possible relation to maternal anticonvulsant therapy. Br J Cancer. 1990 Dec;62(6):996–999. doi: 10.1038/bjc.1990.424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olsen J. H., Hertz H., Blinkenberg K., Verder H. Vitamin K regimens and incidence of childhood cancer in Denmark. BMJ. 1994 Apr 2;308(6933):895–896. doi: 10.1136/bmj.308.6933.895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parker L., Cole M., Craft A. W., Hey E. N. Neonatal vitamin K administration and childhood cancer in the north of England: retrospective case-control study. BMJ. 1998 Jan 17;316(7126):189–193. doi: 10.1136/bmj.316.7126.189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Passmore S. J., Draper G., Brownbill P., Kroll M. Case-control studies of relation between childhood cancer and neonatal vitamin K administration. BMJ. 1998 Jan 17;316(7126):178–184. doi: 10.1136/bmj.316.7126.178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelletier J., Bruening W., Li F. P., Haber D. A., Glaser T., Housman D. E. WT1 mutations contribute to abnormal genital system development and hereditary Wilms' tumour. Nature. 1991 Oct 3;353(6343):431–434. doi: 10.1038/353431a0. [DOI] [PubMed] [Google Scholar]
- Pour P. M. Transplacental induction of gonadal tumors in rats by a nitrosamine. Cancer Res. 1986 Aug;46(8):4135–4138. [PubMed] [Google Scholar]
- Ragge N. K. Clinical and genetic patterns of neurofibromatosis 1 and 2. Br J Ophthalmol. 1993 Oct;77(10):662–672. doi: 10.1136/bjo.77.10.662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rehm S., Ward J. M., Anderson L. M., Riggs C. W., Rice J. M. Transplacental induction of mouse lung tumors: stage of fetal organogenesis in relation to frequency, morphology, size, and neoplastic progression of N-nitrosoethylurea-induced tumors. Toxicol Pathol. 1991;19(1):35–46. doi: 10.1177/019262339101900105. [DOI] [PubMed] [Google Scholar]
- Rice J. M., Rehm S., Donovan P. J., Perantoni A. O. Comparative transplacental carcinogenesis by directly acting and metabolism-dependent alkylating agents in rodents and nonhuman primates. IARC Sci Publ. 1989;(96):17–34. [PubMed] [Google Scholar]
- Roman E., Ansell P., Bull D. Leukaemia and non-Hodgkin's lymphoma in children and young adults: are prenatal and neonatal factors important determinants of disease? Br J Cancer. 1997;76(3):406–415. doi: 10.1038/bjc.1997.399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roman E., Doyle P., Maconochie N., Davies G., Smith P. G., Beral V. Cancer in children of nuclear industry employees: report on children aged under 25 years from nuclear industry family study. BMJ. 1999 May 29;318(7196):1443–1450. doi: 10.1136/bmj.318.7196.1443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roman E., Watson A., Beral V., Buckle S., Bull D., Baker K., Ryder H., Barton C. Case-control study of leukaemia and non-Hodgkin's lymphoma among children aged 0-4 years living in west Berkshire and north Hampshire health districts. BMJ. 1993 Mar 6;306(6878):615–621. doi: 10.1136/bmj.306.6878.615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ross J. A., Perentesis J. P., Robison L. L., Davies S. M. Big babies and infant leukemia: a role for insulin-like growth factor-1? Cancer Causes Control. 1996 Sep;7(5):553–559. doi: 10.1007/BF00051889. [DOI] [PubMed] [Google Scholar]
- Ross J. A., Potter J. D., Robison L. L. Infant leukemia, topoisomerase II inhibitors, and the MLL gene. J Natl Cancer Inst. 1994 Nov 16;86(22):1678–1680. doi: 10.1093/jnci/86.22.1678. [DOI] [PubMed] [Google Scholar]
- Rowland M., Drumm B. Clinical significance of Helicobacter infection in children. Br Med Bull. 1998;54(1):95–103. doi: 10.1093/oxfordjournals.bmb.a011685. [DOI] [PubMed] [Google Scholar]
- Rudel R. Predicting health effects of exposures to compounds with estrogenic activity: methodological issues. Environ Health Perspect. 1997 Apr;105 (Suppl 3):655–663. doi: 10.1289/ehp.97105s3655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- STEWART A., WEBB J., HEWITT D. A survey of childhood malignancies. Br Med J. 1958 Jun 28;1(5086):1495–1508. doi: 10.1136/bmj.1.5086.1495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sasaki S. Influence of the age of mice at exposure to radiation on life-shortening and carcinogenesis. J Radiat Res. 1991 Dec;32 (Suppl 2):73–85. doi: 10.1269/jrr.32.supplement2_73. [DOI] [PubMed] [Google Scholar]
- Sasaki S., Kasuga T., Sato F., Kawashima N. Induction of hepatocellular tumor by x-ray irradiation at perinatal stage of mice. Gan. 1978 Jun;69(3):451–452. [PubMed] [Google Scholar]
- Sasaki S., Kasuga T., Sato F., Kawashima N. Late effects of fetal mice x-irradiated at middle or late intrauterine stage. Gan. 1978 Apr;69(2):167–177. [PubMed] [Google Scholar]
- Schmahl W., Geber E., Lehmacher W. Diaplacental carcinogenic effects of 5-azacytidine in NMRI-mice. Cancer Lett. 1985 May;27(1):81–90. doi: 10.1016/0304-3835(85)90011-4. [DOI] [PubMed] [Google Scholar]
- Schmahl W., Kriegel H. Ovary tumors in NMRI mice subjected to fractionated X-irradiation during fetal development. J Cancer Res Clin Oncol. 1980;98(1):65–74. doi: 10.1007/BF00413178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seizinger B. R. NF1: a prevalent cause of tumorigenesis in human cancers? Nat Genet. 1993 Feb;3(2):97–99. doi: 10.1038/ng0293-97. [DOI] [PubMed] [Google Scholar]
- Sharpe C. R., Franco E. L. Etiology of Wilms' tumor. Epidemiol Rev. 1995;17(2):415–432. doi: 10.1093/oxfordjournals.epirev.a036201. [DOI] [PubMed] [Google Scholar]
- Sharpe C. R., Franco E. L., de Camargo B., Lopes L. F., Barreto J. H., Johnsson R. R., Mauad M. A. Parental exposures to pesticides and risk of Wilms' tumor in Brazil. Am J Epidemiol. 1995 Feb 1;141(3):210–217. doi: 10.1093/oxfordjournals.aje.a117422. [DOI] [PubMed] [Google Scholar]
- Shoemaker A. R., Moser A. R., Dove W. F. N-ethyl-N-nitrosourea treatment of multiple intestinal neoplasia (Min) mice: age-related effects on the formation of intestinal adenomas, cystic crypts, and epidermoid cysts. Cancer Res. 1995 Oct 1;55(19):4479–4485. [PubMed] [Google Scholar]
- Sorahan T., Lancashire R. J., Hultén M. A., Peck I., Stewart A. M. Childhood cancer and parental use of tobacco: deaths from 1953 to 1955. Br J Cancer. 1997;75(1):134–138. doi: 10.1038/bjc.1997.22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sorahan T., Lancashire R., Prior P., Peck I., Stewart A. Childhood cancer and parental use of alcohol and tobacco. Ann Epidemiol. 1995 Sep;5(5):354–359. doi: 10.1016/1047-2797(95)00032-3. [DOI] [PubMed] [Google Scholar]
- Stavrou D., Hänichen T. Oncogene Wirkung von Athylnitrosoharnstoff beim kaninchen während der pränatalen Periode. Z Krebsforsch Klin Onkol Cancer Res Clin Oncol. 1975 Oct 27;84(2):207–215. doi: 10.1007/BF00304046. [DOI] [PubMed] [Google Scholar]
- Stiller C. A., Allen M. B., Eatock E. M. Childhood cancer in Britain: the National Registry of Childhood Tumours and incidence rates 1978-1987. Eur J Cancer. 1995 Nov;31A(12):2028–2034. doi: 10.1016/0959-8049(95)00428-9. [DOI] [PubMed] [Google Scholar]
- Stiller C. A., Chessells J. M., Fitchett M. Neurofibromatosis and childhood leukaemia/lymphoma: a population-based UKCCSG study. Br J Cancer. 1994 Nov;70(5):969–972. doi: 10.1038/bjc.1994.431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stiller C. A. Population based survival rates for childhood cancer in Britain, 1980-91. BMJ. 1994 Dec 17;309(6969):1612–1616. doi: 10.1136/bmj.309.6969.1612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swerdlow A. J., Huttly S. R., Smith P. G. Prenatal and familial associations of testicular cancer. Br J Cancer. 1987 May;55(5):571–577. doi: 10.1038/bjc.1987.116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi T., Watanabe H., Dohi K., Ito A. 252Cf relative biological effectiveness and inheritable effect of fission neutrons in mouse liver tumorigenesis. Cancer Res. 1992 Apr 1;52(7):1948–1953. [PubMed] [Google Scholar]
- Tomatis L., Cabral J. R., Likhachev A. J., Ponomarkov V. Increased cancer incidence in the progeny of male rats exposed to ethylnitrosourea before mating. Int J Cancer. 1981 Oct 15;28(4):475–478. doi: 10.1002/ijc.2910280413. [DOI] [PubMed] [Google Scholar]
- Tomatis L., Goodall C. M. The occurrence of tumours in F1, F2, and F3 descendants of pregnant mice injected with 7,12-dimethylbenz(a)anthracene. Int J Cancer. 1969 Mar 15;4(2):219–225. doi: 10.1002/ijc.2910040212. [DOI] [PubMed] [Google Scholar]
- Tomatis L., Narod S., Yamasaki H. Transgeneration transmission of carcinogenic risk. Carcinogenesis. 1992 Feb;13(2):145–151. doi: 10.1093/carcin/13.2.145. [DOI] [PubMed] [Google Scholar]
- Tomatis L. Overview of perinatal and multigeneration carcinogenesis. IARC Sci Publ. 1989;(96):1–15. [PubMed] [Google Scholar]
- Tomatis L. Transgeneration carcinogenesis: a review of the experimental and epidemiological evidence. Jpn J Cancer Res. 1994 May;85(5):443–454. doi: 10.1111/j.1349-7006.1994.tb02378.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomatis L., Turusov V. S., Cardis E., Cabral J. P. Tumour incidence in the progeny of male rats exposed to ethylnitrosourea before mating. Mutat Res. 1990 Apr;229(2):231–237. doi: 10.1016/0027-5107(90)90096-m. [DOI] [PubMed] [Google Scholar]
- Toth B. A critical review of experiments in chemical carcinogensis using newborn animals. Cancer Res. 1968 Apr;28(4):727–738. [PubMed] [Google Scholar]
- Truhan A. P. Sun protection in childhood. Clin Pediatr (Phila) 1991 Dec;30(12):676–681. doi: 10.1177/000992289103001205. [DOI] [PubMed] [Google Scholar]
- Turusov V. S., Trukhanova L. S., Parfenov YuD, Tomatis L. Occurrence of tumours in the descendants of CBA male mice prenatally treated with diethylstilbestrol. Int J Cancer. 1992 Jan 2;50(1):131–135. doi: 10.1002/ijc.2910500126. [DOI] [PubMed] [Google Scholar]
- UPTON A. C., ODELL T. T., Jr, SNIFFEN E. P. Influence of age at time of irradiation on induction of leukemia and ovarian tumors in RF mice. Proc Soc Exp Biol Med. 1960 Aug-Sep;104:769–772. doi: 10.3181/00379727-104-25982. [DOI] [PubMed] [Google Scholar]
- Urquhart J. D., Black R. J., Muirhead M. J., Sharp L., Maxwell M., Eden O. B., Jones D. A. Case-control study of leukaemia and non-Hodgkin's lymphoma in children in Caithness near the Dounreay nuclear installation. BMJ. 1991 Mar 23;302(6778):687–692. doi: 10.1136/bmj.302.6778.687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vesselinovitch S. D., Koka M., Rao K. V., Mihailovich N., Rice J. M. Prenatal multicarcinogenesis by ethylnitrosourea in mice. Cancer Res. 1977 Jun;37(6):1822–1828. [PubMed] [Google Scholar]
- Vesselinovitch S. D., Rao K. V., Mihailovich N. Neoplastic response of mouse tissues during perinatal age periods and its significance in chemical carcinogenesis. Natl Cancer Inst Monogr. 1979 May;(51):239–250. [PubMed] [Google Scholar]
- Vessey M. P. Epidemiological studies of the effects of diethylstilboestrol. IARC Sci Publ. 1989;(96):335–348. [PubMed] [Google Scholar]
- Vorobtsova I. E., Aliyakparova L. M., Anisimov V. N. Promotion of skin tumors by 12-O-tetradecanoylphorbol-13-acetate in two generations of descendants of male mice exposed to X-ray irradiation. Mutat Res. 1993 Jun;287(2):207–216. doi: 10.1016/0027-5107(93)90013-6. [DOI] [PubMed] [Google Scholar]
- Vorobtsova I. E., Kitaev E. M. Urethane-induced lung adenomas in the first-generation progeny of irradiated male mice. Carcinogenesis. 1988 Nov;9(11):1931–1934. doi: 10.1093/carcin/9.11.1931. [DOI] [PubMed] [Google Scholar]
- Wakeford R. The risk of childhood cancer from intrauterine and preconceptional exposure to ionizing radiation. Environ Health Perspect. 1995 Nov;103(11):1018–1025. doi: 10.1289/ehp.951031018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walinder G., Sjödén A. M. Late effects of irradiation on the thyroid gland in mice. 3. Comparison between irradiation of foetuses and adults. Acta Radiol Ther Phys Biol. 1973 Jun;12(3):201–208. doi: 10.3109/02841867309130395. [DOI] [PubMed] [Google Scholar]
- Walker B. E., Kurth L. A. Multi-generational carcinogenesis from diethylstilbestrol investigated by blastocyst transfers in mice. Int J Cancer. 1995 Apr 10;61(2):249–252. doi: 10.1002/ijc.2910610218. [DOI] [PubMed] [Google Scholar]
- Walker B. E., Kurth L. A. Pituitary tumors in mice exposed prenatally to diethylstilbestrol. Cancer Res. 1993 Apr 1;53(7):1546–1549. [PubMed] [Google Scholar]
- Wanderås E. H., Grotmol T., Fosså S. D., Tretli S. Maternal health and pre- and perinatal characteristics in the etiology of testicular cancer: a prospective population- and register-based study on Norwegian males born between 1967 and 1995. Cancer Causes Control. 1998 Oct;9(5):475–486. doi: 10.1023/a:1008857702380. [DOI] [PubMed] [Google Scholar]
- Watanabe H., Takahashi T., Lee J. Y., Ohtaki M., Roy G., Ando Y., Yamada K., Gotoh T., Kurisu K., Fujimoto N. Influence of paternal (252) Cf neutron exposure on abnormal sperm, embryonal lethality, and liver tumorigenesis in the F(1) offspring of mice. Jpn J Cancer Res. 1996 Jan;87(1):51–57. doi: 10.1111/j.1349-7006.1996.tb00199.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wechsler W., Rice J. M., Vesselinovitch S. D. Transplacental and neonatal induction of neurogenic tumors in mice: comparison with related species and with human pediatric neoplasms. Natl Cancer Inst Monogr. 1979 May;(51):219–226. [PubMed] [Google Scholar]
- Westergaard T., Andersen P. K., Pedersen J. B., Olsen J. H., Frisch M., Sørensen H. T., Wohlfahrt J., Melbye M. Birth characteristics, sibling patterns, and acute leukemia risk in childhood: a population-based cohort study. J Natl Cancer Inst. 1997 Jul 2;89(13):939–947. doi: 10.1093/jnci/89.13.939. [DOI] [PubMed] [Google Scholar]
- White L., Giri N., Vowels M. R., Lancaster P. A. Neuroectodermal tumours in children born after assisted conception. Lancet. 1990 Dec 22;336(8730):1577–1577. doi: 10.1016/0140-6736(90)93350-x. [DOI] [PubMed] [Google Scholar]
- Wiestler O. D., Kleihues P., Rice J. M., Ivankovic S. DNA methylation in maternal, fetal and neonatal rat tissues following perinatal administration of procarbazine. J Cancer Res Clin Oncol. 1984;108(1):56–59. doi: 10.1007/BF00390973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson T. M., Rivkees S. A., Deutsch W. A., Kelley M. R. Differential expression of the apurinic / apyrimidinic endonuclease (APE/ref-1) multifunctional DNA base excision repair gene during fetal development and in adult rat brain and testis. Mutat Res. 1996 Apr 2;362(3):237–248. doi: 10.1016/0921-8777(95)00053-4. [DOI] [PubMed] [Google Scholar]
- Windham G. C., Bjerkedal T., Langmark F. A population-based study of cancer incidence in twins and in children with congenital malformations or low birth weight, Norway, 1967-1980. Am J Epidemiol. 1985 Jan;121(1):49–56. doi: 10.1093/oxfordjournals.aje.a113982. [DOI] [PubMed] [Google Scholar]
- Wyatt R., Wilson A. M. Children of anaesthetists. Br Med J. 1973 Mar 17;1(5854):675–675. doi: 10.1136/bmj.1.5854.675-a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang P., Grufferman S., Khoury M. J., Schwartz A. G., Kowalski J., Ruymann F. B., Maurer H. M. Association of childhood rhabdomyosarcoma with neurofibromatosis type I and birth defects. Genet Epidemiol. 1995;12(5):467–474. doi: 10.1002/gepi.1370120504. [DOI] [PubMed] [Google Scholar]
- Yu W., Sipowicz M. A., Haines D. C., Birely L., Diwan B. A., Riggs C. W., Kasprzak K. S., Anderson L. M. Preconception urethane or chromium(III) treatment of male mice: multiple neoplastic and non-neoplastic changes in offspring. Toxicol Appl Pharmacol. 1999 Jul 15;158(2):161–176. doi: 10.1006/taap.1999.8692. [DOI] [PubMed] [Google Scholar]
- Zack M., Adami H. O., Ericson A. Maternal and perinatal risk factors for childhood leukemia. Cancer Res. 1991 Jul 15;51(14):3696–3701. [PubMed] [Google Scholar]
- van Steensel-Moll H. A., Valkenburg H. A., Vandenbroucke J. P., van Zanen G. E. Are maternal fertility problems related to childhood leukaemia? Int J Epidemiol. 1985 Dec;14(4):555–559. doi: 10.1093/ije/14.4.555. [DOI] [PubMed] [Google Scholar]
- van den Heuvel R., Gerber G. B., Leppens H., Vander Plaetse F., Schoeters G. E. Long-term effects on tumour incidence and survival from 241Am exposure of the BALB/c mouse in utero and during adulthood. Int J Radiat Biol. 1995 Dec;68(6):679–686. doi: 10.1080/09553009514551691. [DOI] [PubMed] [Google Scholar]
- von Kries R., Göbel U., Hachmeister A., Kaletsch U., Michaelis J. Vitamin K and childhood cancer: a population based case-control study in Lower Saxony, Germany. BMJ. 1996 Jul 27;313(7051):199–203. doi: 10.1136/bmj.313.7051.199. [DOI] [PMC free article] [PubMed] [Google Scholar]




