Abstract
Leukemia-specific AML1/ETO transcripts are detectable in most patients with t(8;21) acute myelogenous leukemia (AML) in long-term remission. To understand the inconsistency between the clinical cure and the presence of “residual disease” at a molecular level, we separated and identified the cells expressing AML1/ETO by phenotype and function. Here we demonstrate that AML1/ETO transcripts are present in a fraction of stem cells, monocytes, and B cells in remission marrow, and in a fraction of B cells in leukemic marrow, but not in T cells. AML1/ETO transcripts also were demonstrated in a fraction of colony-forming cells of erythroid, granulocyte-macrophage, and/or megakaryocyte lineages in both leukemic and remission marrow. These data strongly suggest that the acquisition of the t(8;21) occurs at the level of stem cells capable of differentiating into B cells as well as all myeloid lineages, and that a fraction of the AML1/ETO-expressing stem cells undergo additional oncogenic event(s) that ultimately leads to transformation into AML.
The 8;21 translocation, t(8;21)(q22;q22), is one of the most frequent chromosomal abnormalities in acute myelogenous leukemia (AML) (1). It involves AML1 at 21q22 and ETO at 8q22, resulting in the expression of AML1/ETO chimeric transcripts in leukemic cells (2–4). Recent studies have shown, using sensitive reverse transcriptase–PCR (RT-PCR) assays that AML1/ETO transcripts remain detectable in most t(8;21) AML patients who maintain hematological and cytogenetic long-term remission after chemotherapy, autologous bone marrow (BM) transplantation, or autologous mobilized blood transplantation (MBT) (5–10).
It remains unclear whether or not the persistence of AML1/ETO in remission indicates minimal residual disease that may cause relapse. To understand this issue, one needs to know whether only malignant cells express AML1/ETO, and what the function of these transcripts in AML1/ETO-expressing cells is during remission. The long-term persistence of AML1/ETO expression suggests that at least a subset of the cells with t(8;21) in remission would be capable of self-renewal; in normal marrow, this is a property largely restricted to hematopoietic stem cells (HSC). We have reported that AML1/ETO transcripts could be found in a small fraction of myeloid progenitors in t(8;21) AML remission marrow, including burst-forming unit-erythroid (BFU-E), colony-forming unit-megakaryocytes (CFU-Meg), and CFU-granulocyte/macrophages (CFU-GM) (10). This finding raises an interesting possibility that t(8;21) might involve HSC and their descendant myeloid progeny with normal differentiation activity, which contribute to normal hematopoiesis in remission, giving rise to mature hematopoietic cells that possess a limited lifespan. In this case, the t(8;21)+ progenitors themselves should not cause AML. Alternatively, because it has not yet been proven that these t(8;21)+ progenitors give rise to mature blood cells in vivo, the residual leukemia clones may be latent in vivo, but differentiate into mature myeloid cells in vitro in response to exogenous cytokines. This is not unlikely because t(8;21) AML usually presents AML-M2 (by French-American-British classification) that is characterized by the differentiating tendency of leukemic blasts (1).
To clarify the origin of AML1/ETO transcripts, we surveyed AML1/ETO mRNA expression in rigorously purified HSC, progenitors, and mature hematopoietic cells of various lineages. Our study demonstrates that acquisition of t(8;21) occurs in progenitors or HSC, that the translocation is a necessary, but not sufficient, precondition for transformation, and that these t(8;21)+ progenitors contribute to B lymphopoiesis as well as myelopoiesis throughout the clinical course of t(8;21) AML.
Materials and Methods
Patients.
The clinical characteristics of patients are shown in Table 1. This study included BM mononuclear cells from six cases with t(8;21) AML in leukemic phase (cases 1–4, 5a, and 6a), and 13 cases in remission (cases 5b, 6b, 12, 13, 15–18, 20, and 22–25). An additional eight patients (cases 7–11, 14, 19, and 21), whose samples were analyzed only in our previous study (10) and who maintained remission at the time of this report, were entered in this study. Remission was achieved by the protocols at the Hiroshima Red Cross Hospital (11) or in the Fukuoka Bone Marrow Transplantation Group (12). Six patients (cases 5–10) were further treated with auto-MBT (12), and the remaining cases received intensification chemotherapy (11). All patients studied remained in remission at the time of this report. The median remission duration at the time of sampling was 37 and 32 months, and the median disease-free survival was 98 and 97 months in the patient groups treated with chemotherapy and auto-MBT, respectively. Informed consent was obtained from all patients.
Table 1.
Characteristics of t(8;21) AML patients
| Case no. | Age/ sex | FAB | Therapy | Remission duration at sampling, Mo | Total remission duration, Mo | AML1/ETO+ colonies, % |
|---|---|---|---|---|---|---|
| 1 | 31/M | M2 | — | at Dx | — | ND |
| 2 | 48/M | M2 | — | at Dx | — | 59.7 |
| 3 | 46/F | M2 | — | at Dx | — | 75.0 |
| 4 | 45/M | M2 | — | at Dx | — | 57.1 |
| 5a | 49/M | M2 | — | at Dx | — | 66.7 |
| 5b | Auto-MBT | 13 | +18 | 2.8 | ||
| 6a | 20/M | M2 | — | at Dx | — | 61.9 |
| 6b | Auto-MBT | 13 | +18 | 2.1 | ||
| 7a | 47/M | M2 | Auto-MBT | 15 | +99 | 2.6 |
| 7b | 27 | +87 | 0.8 | |||
| 8a | 33/F | M2 | Auto-MBT | 49 | +131 | 2.5 |
| 8b | 60 | +120 | 0 | |||
| 9 | 31/M | M2 | Auto-MBT | 37 | +97 | 2.2 |
| 10 | 21/F | M2 | Auto-MBT | 39 | +99 | 1.6 |
| 11 | 37/F | M2 | Ch-Tx | 12 | +72 | 2.9 |
| 12 | 32/M | M2 | Ch-Tx | 18 | +78 | 3.1 |
| 13 | 62/M | M2 | Ch-Tx | 18 | +78 | 3.4 |
| 14 | 57/M | M2 | Ch-Tx | 28 | +88 | 4.7 |
| 15 | 28/F | M2 | Ch-Tx | 30 | +90 | 2.5 |
| 16 | 45/M | M2 | Ch-Tx | 34 | +94 | 2.7 |
| 17 | 41/F | M1 | Ch-Tx | 34 | +94 | 3.0 |
| 18 | 52/F | M2 | Ch-Tx | 37 | +97 | 1.9 |
| 19 | 35/F | M2 | Ch-Tx | 45 | +105 | 2.0 |
| 20 | 19/M | M1 | Ch-Tx | 49 | +109 | 1.6 |
| 21 | 53/F | M2 | Ch-Tx | 50 | +110 | 1.6 |
| 22 | 20/M | M2 | Ch-Tx | 80 | +140 | 1.7 |
| 23 | 52/M | M2 | Ch-Tx | 88 | +148 | 0.8 |
| 24 | 31/M | M2 | Ch-Tx | 124 | +184 | 0.7 |
| 25 | 54/M | M2 | Ch-Tx | 150 | +210 | 0.8 |
FAB: Classification of AML according to the French-American-British classification; auto-MBT: autologous MBT; Ch-Tx: chemotherapy; at Dx: samples of blastic BM cells were collected at diagnosis; Mo: months; ND: not done. Total percentages of AML1/ETO+ colonies included CFU-GM, BFU-E, CFU-Meg, and CFU-Mix. Detailed data are shown in Table 4 and in our previous report (10).
Cell Purification by Triple-Sorting by Using a Five-Color Flow Cytometer.
BM cells were cryopreserved by using hydroxyethyl starch and DMSO as a cryoprotective agent at −80°C (13). For sorting HSC and progenitor populations, BM cells were stained with Cy5-phycoerythrin (PE)-conjugated lineage (Lin) mixture (anti-CD3, -CD4, -CD8, -CD13, -CD16, -CD19, -CD20, -CD33, and-glycophorin A) (Caltag, South San Francisco, CA), fluorescein-5-isothiocyanate (FITC)-conjugated CD10 (Ancell Bayport, MN), PE-conjugated Thy-1 (PharMingen), Texas red (TR)-conjugated anti-CD38, and allophycocyanin (APC)-conjugated anti-CD34 antibodies. For sorting monocytes, B cells, and T cells, BM cells were stained with Cy5-PE-conjugated CD3 or CD20, FITC-conjugated anti-IgM (Ancell), anti-T cell antigen receptor (TCR) αβ (Caltag), or anti-CD13, PE-conjugated anti-CD3 or-CD20, TR-conjugated anti-CD14, and APC-conjugated anti-CD34 antibodies. CD34+Thy-1+Lin-CD10-CD38-/lo HSC, CD34+Thy1-Lin-CD10+CD38+ common lymphoid progenitors (CLP) (14, 15), CD34-CD20+IgM+CD3-CD14- B cells, CD34-CD3+TCRαβ+CD20-CD14- T cells, and CD34-CD13+CD14+CD3-CD20- monocytes were sorted by a highly modified triple laser (488-nm argon laser, 599-nm dye laser, and UV laser) fluorescence activated cell sorter (FACS: FACS Vantage, Becton Dickinson) (16). The five-color sort using both positive and negative gates in multiple channels usually gives rise to cells of more than 98% of purity, avoiding cosorting cells stained in a nonspecific manner. The sorted cells were subjected to two additional rounds of sorting using the same gate to eliminate contaminating cells and doublets (17). The sample line was washed out by 75% ethanol followed by saline between each round of sorting to eliminate remaining cells.
In Vitro Hematopoietic Progenitor Assay.
Clonogenic progenitor assays were performed by using the methylcellulose culture system as reported (10, 15). Human cytokines such as steel factor (20 ng/ml), IL-3 (30 ng/ml), IL-6 (10 ng/ml), erythropoietin (2 units/ml), and thrombopoietin (20 ng/ml) (R & D Systems) were added at the start of the culture. An automatic cell deposition unit system (Becton Dickinson) (15) was used to deposit single cells onto 96-well plates.
RT-PCR Analysis.
Total RNA was extracted from purified cells and colonies by the acid guanidine/phenol/chloroform method. The detailed method for RT-PCR and the primer sequences were described elsewhere (9, 10). Each PCR product was confirmed to be the expected sequences by testing the length of fragments after digesting with DNA restriction enzyme ApaI or by Southern blot analysis using the probe spanning the AML1/ETO junction (10). A t(8;21) AML cell line, Kasumi-1 (18), was used as a positive control. The nested RT-PCR assay we used could detect AML1/ETO as well as myeloperoxidase (MPO) genes in a single Kasumi-1 cell among 107 T cell lines (9, 10). When we started sorting from a positive control sample that was a mixture of 90% Kasumi-1 and 10% normal BM cells, AML1/ETO or MPO mRNA was not detected in ≥100,000 triple-sorted T or B cells, indicating that those triple-sorted cells were free from Kasumi-1 contaminants. Based on these data, all RT-PCR analyses were applied only for triple-sorted populations throughout this study. At the end of each experiment, triple-sorted 30,000 T cells from the positive control sample were collected and were verified not to be contaminated with Kasumi-1 cells.
Transformation of Purified B Cells by Epstein–Barr Virus (EBV).
Triple-sorted B cells were cultured with supernatant from the EBV-producing cell line (19). The transformed B cells were cultured for more than 3 months, and the EBV-transformed B cell lines were established.
Results
Analysis of Remission and Leukemic BM in t(8,21) AML for HSC and Progenitor Cells.
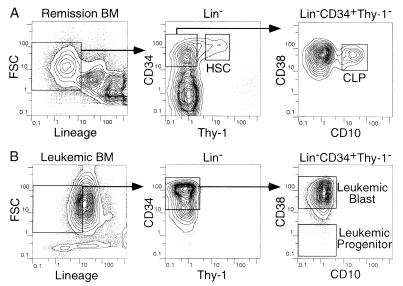
In remission BM, a minority of the CD34+ fraction contained Thy-1+ stem cells (20) (Fig. 1A). The most primitive subset of these stem cells is CD38-/lo (21). Another fraction of BM is CD34+Thy-1- cells that include separable CLP (CD10+CD38+) (Fig. 1A); these cells possess lymphoidrestricted differentiation potential (14). Primitive HSC (CD34+Thy-1+CD38-/lo) and CLP (CD34+Thy1-CD10+CD38+) were depleted in leukemic BM in all six cases analyzed (Fig. 1B). Instead, the leukemic BM contained a small fraction of primitive leukemic progenitors (CD34+Thy-1-CD38-/lo) that contain self-renewing leukemic stem cells (22) (Fig. 1B). The purified HSC, CLP, and primitive leukemic progenitors as well as mature monocytes, B cells, and T cells were subjected to further analyses.
Figure 1.
HSC and CLP in remission and leukemic BM. Five-color flow cytometric analyses of BM cells in remission (A: case 5b) and in leukemic phase (B: case 5a). BM cells were first gated by the negative expression of lineage-related antigens. (A) The CD34+Thy-1+CD38-/lo HSC and CD34+Thy-1-CD10+CD38+ CLP were sorted from remission BM. (B) In leukemic BM, HSC and CLP were absent, whereas primitive CD34+CD38-/lo leukemic progenitors and CD34+CD38+ leukemic blasts existed. FSC, forward light scatter.
AML1/ETO Is Expressed in HSC, CLP, Monocytes, and B Cells, but Not in T Cells in Remission.
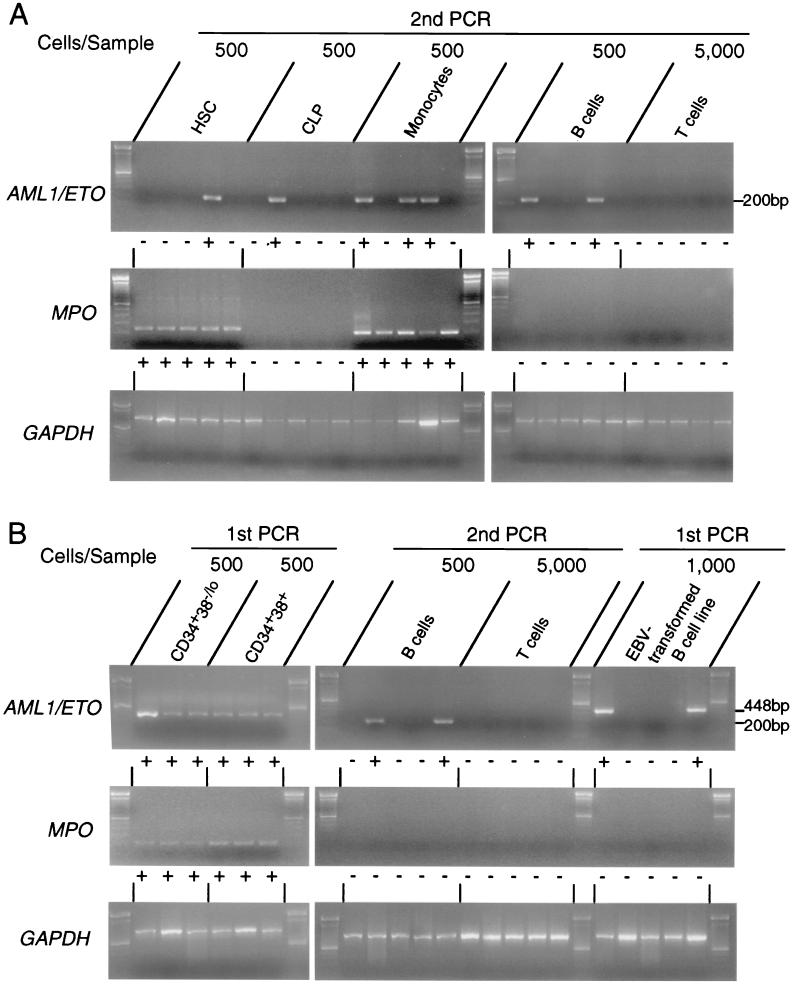
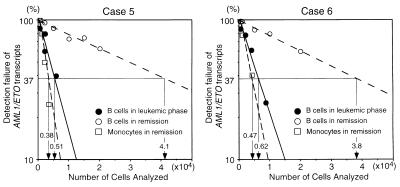
In all patients in remission, AML1/ETO transcripts could be found if 105 BM cells were analyzed by RT-PCR (Table 2). To identify BM subpopulations that express AML1/ETO transcripts, 500 HSC, 500 CLP, 500 mature B cells, 500 monocytes, or 5,000 mature T cells from the remission BM of 13 cases were subjected to RT-PCR analysis. AML1/ETO transcripts were detectable in HSC (in seven of 13 cases studied), CLP (in two of 13 cases studied), monocytes (in all 10 cases studied), and B cells (in 10 of 12 cases studied) (Table 2). The AML1/ETO+ B cell population did not express MPO mRNA, indicating that the sorted B cells were not contaminated with myelomonocytic cells (Fig. 2). The frequency of AML1/ETO+ cells was estimated at limit dilution, where Poisson statistics would allow determination of the average number of cells that contains a single AML1/ETO+ cell (15). In this analysis, the frequency of AML1/ETO+ cells was 1 in 3,000–4,800 (median 4,500) monocytes in five cases studied (cases 5b, 6b, 12, 20, and 22), and 1 in 20,000–45,000 (median 36,000) mature B cells in six cases studied (cases 5b, 6b, 20, and 22–24). Typical limit dilution tests in cases 5 and 6 are shown in Fig. 3. In contrast, in all cases, AML1/ETO transcripts were undetectable in mature T cells.
Table 2.
AML1/ETO mRNA in FACS-purified cells in remission
| Case | AML1/ETO mRNA (105BM cells) | No. of AML1/ETO+ samples/no. of samples tested
|
||||
|---|---|---|---|---|---|---|
| HSC (500 cells) | CLP (500 cells) | Monocytes (500 cells) | B cells (500 cells) | T cells (5,000 cells) | ||
| 5b | + | 0/3 | 0/8 | 4/29 | 4/205 | 0/24 |
| 6b | + | 0/8 | 0/3 | 3/32 | 3/52 | 0/20 |
| 12 | + | 1/12 | 0/23 | 2/11 | 1/82 | 0/25 |
| 13 | + | 1/20 | 0/5 | ND | ND | 0/10 |
| 15 | + | 3/26 | 0/8 | 1/7 | 2/81 | 0/6 |
| 16 | + | 1/20 | 0/30 | 2/9 | 2/37 | 0/13 |
| 17 | + | 0/4 | 0/2 | ND | 1/64 | 0/2 |
| 18 | + | 0/3 | 0/2 | 1/3 | 0/16 | 0/5 |
| 20 | + | 1/9 | 0/7 | 2/17 | 2/140 | 0/6 |
| 22 | + | 1/27 | 1/30 | 3/13 | 3/149 | 0/6 |
| 23 | + | 2/17 | 1/8 | 2/10 | 6/230 | 0/10 |
| 24 | + | 0/11 | 0/4 | 3/16 | 2/41 | 0/9 |
| 25 | + | 0/12 | 0/3 | ND | 0/26 | 0/9 |
| Total | 10/172 (5.8) | 2/133 (1.5) | 23/147 (15.6) | 26/1123 (2.3) | 0/145 (0) | |
Each population was purified by a five-color FACS sorting and pooled either 500 cells (HSC, CLP, monocytes, and B cells) or 5,000 cells (T cells) were analyzed by RT-PCR for AML1/ETO mRNA. Representative data are shown in Fig. 2A. ND, not done.
Figure 2.
Detection of AML1/ETO+ cells in triple-sorted populations from remission or leukemic BM. (A) RT-PCR analysis on purified cells from remission BM. Five hundred HSC, CLP, monocytes, and B cells and 5,000 T cells were triple-sorted and subjected to RT-PCR analysis. Representative data in case 22, who had maintained remission for 80 months at the time of sampling, are shown. The AML1/ETO transcript was sometimes detectable in HSC, CLP, monocytes, and B cells, but not in T cells after the second round of PCR amplification; + and − under each lane depict positive and negative result of PCR, respectively. Data of all cases are summarized in Table 2. Note that MPO gene is not expressed in AML1/ETO+ pooled B cells and CLP, which confirms that the samples do not contain myelomonocytic cells. (B) RT-PCR analysis on purified cells from leukemic BM. Representative data in case 5a are shown. All 500 pooled CD34+CD38-/lo and CD34+CD38+ cells expressed AML1/ETO mRNA, which is detectable by the first round of PCR, and AML1/ETO+ B cells were found at a higher frequency compared with those in remission BM as summarized in Table 3. The last five right lanes show results of PCR analysis in two AML1/ETO+ and three AML1/ETO- EBV-transformed B cell lines established from case 1. Note that in these AML1/ETO+ B cell lines, AML1/ETO transcripts were detectable by the first round of PCR amplification. GAPDH, glyceraldehyde-3-phosphate dehydrogenase.
Figure 3.
Frequency of AML1/ETO+ cells in B cells and monocytes estimated by limit dilution analyses. The percent of samples negative by RT-PCR for AML1/ETO transcripts is plotted on the y axis versus the number of cells per sample tested. According to the Poisson statistics, the frequency of AML1/ETO+ cells can be estimated as numbers of cells in samples that show 37% of detection failures (arrows). The frequency of AML1/ETO+ cells was estimated to be 1 in 41,000 (case 5) and 38,000 (case 6) mature B cells in remission marrow (○), and 1 in 5,100 (case 5) and 6,200 (case 6) mature B cells in leukemic marrow (●), indicating that leukemic BM contains t(8;21)+ B cells at ≈10-fold higher frequency, compared with remission BM. AML1/ETO+ cell was estimated to be in 1 in 3,800 (case 5) and 4,700 (case 6) monocytes in remission marrow (□).
AML1/ETO-Expressing Cells in Leukemic Phase of t(8;21) AML Include B Cells, but Not T Cells.
We analyzed the expression of AML1/ETO in purified T and B cells from leukemic BM in five cases (cases 1, 3–6) (Table 3). In all 500 pooled CD34+Thy-1-CD38+ leukemic blasts and primitive CD34+Thy-1-CD38-/lo leukemic progenitors, we detected AML1/ETO transcripts. Strikingly, AML1/ETO+ mature B cells were detectable in all cases and existed at a higher frequency (1 in 5,000 to 6,200 cells) in leukemic phase compared with remission (Fig. 3). However, mature T cells did not express AML1/ETO in four cases studied.
Table 3.
AML1/ETO mRNA in FACS-purified cells in leukemic phase
| Case | No. of AML1/ETO+ samples/no. of samples tested
|
|||
|---|---|---|---|---|
| CD34+CD38−/lo (500 cells) | CD34+CD38+ (500 cells) | B cells (500 cells) | T cells (5,000 cells) | |
| 1 | 11/11 | 5/5 | 4/14 | 0/10 |
| 3 | 64/64 | 5/5 | 2/2 | ND |
| 4 | 10/10 | 5/5 | 3/15 | 0/9 |
| 5a | 4/4 | 5/5 | 5/75 | 0/20 |
| 6a | 30/30 | 5/5 | 5/40 | 0/21 |
| Total | 119/119 (100) | 25/25 (100) | 19/146 (13) | 0/60 (0) |
Pooled 500 CD34+ (Thy-1−)CD38−/lo cells, CD34+(Thy-1−) CD38+ cells and B cells or 5,000 T cells were analyzed by RT-PCR for AML1/ETO mRNA. Representative data are shown in Fig. 2B.
We used EBV to transform the sorted B cells in cases 1 and 5a. Among 143 B cell lines established from ≈3,345 samples, each containing 1,000 B cells, four EBV-transformed B cell lines were found to express AML1/ETO (Fig. 2). Based on these data, AML1/ETO+ B cells are present at the earliest times tested after diagnosis of t(8;21) AML.
AML1/ETO Is Expressed in Various Myeloid Colony-Forming Progenitors in Both the Leukemic and the Remission Phases.
We analyzed the expression of AML1/ETO in myeloid colony-forming progenitors in remission BM from five cases (cases 5b, 6b, 15, 16, and 18), in addition to cases reported in our previous study (10). The plating efficiency of single cell culture of purified HSC in methylcellulose was 53.7%. As shown in Table 4, a small, but significant, percent of single cell-derived colonies of various myeloid lineages expressed AML1/ETO in all cases.
Table 4.
AML1/ETO mRNA in single-cell-derived myeloid colonies in remission and leukemic phase
| Phase | Case | No. of AML1/ETO+ colonies/no. of colonies tested
|
|||||
|---|---|---|---|---|---|---|---|
| CFU-L | CFU-GM | BFU-E | CFU-Meg | CFU-Mix | Total | ||
| Remission | 5 | 0/0 | 2/58 | 1/30 | 0/28 | 1/28 | 4/144 (2.8) |
| 6 | 0/0 | 2/76 | 1/28 | 0/20 | 0/20 | 3/144 (2.1) | |
| 15 | 0/0 | 2/70 | 0/30 | 1/24 | 0/20 | 3/144 (2.1) | |
| 16 | 0/0 | 3/62 | 0/37 | 0/26 | 1/24 | 4/149 (2.7) | |
| 18 | 0/0 | 4/94 | 2/38 | 0/34 | 0/26 | 6/192 (3.1) | |
| Total | 0/0 | 13/360 (3.6) | 4/163 (2.5) | 1/132 (0.8) | 2/118 (1.7) | 20/773 (2.6) | |
| Leukemic | 2 | 14/14 | 31/48 | 8/17 | 0/0 | 1/2 | 40/67 (59.7) |
| 3 | 4/4 | 3/3 | 2/4 | 1/1 | 0/0 | 6/8 (75.0) | |
| 4 | 6/6 | 7/12 | 1/2 | 0/0 | 0/0 | 8/14 (57.1) | |
| 5 | 3/3 | 1/2 | 1/1 | 0/0 | 0/0 | 2/3 (66.7) | |
| 6 | 12/12 | 24/37 | 2/5 | 0/0 | 0/0 | 26/42 (61.9) | |
| Total | 39/39 (100) | 66/102 (64.7) | 14/29 (48.2) | 1/1 (100) | 1/2 (50) | 82/134 (61.2) | |
FACS-purified 288 single CD34+Thy-1+CD38−/lo cells in remission marrow and 1,440 CD34+Thy-1−CD38−/lo cells in leukemic marrow were cultured in methylcellulose, and all colonies formed were picked on day 14 for RT-PCR analysis. Representative data are shown in Fig. 4.
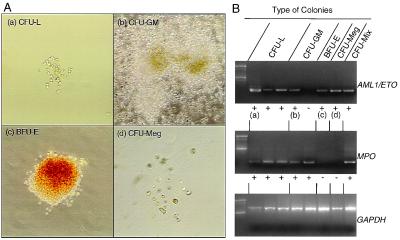
We also cultured the CD34+Thy-1-CD38+ leukemic blast and CD34+Thy-1-CD38-/lo leukemic progenitor fractions sorted from leukemic BM. Almost 0.5–1% of the CD34+Thy-1-CD38+ cells could form colonies, all of which were composed of leukemic blasts (CFU-leukemia: CFU-L). In contrast, more primitive CD34+Thy-1-CD38-/lo cells formed colonies at higher frequencies (2.4%) in single-cell cultures, and these single cell-derived colonies included 77% of differentiated myeloid colonies such as CFU-GM, CFU-Meg, and BFU-E (Table 4, Fig. 4). All CFU-L expressed AML1/ETO. Strikingly, up to 60% of CFU-GM, CFU-Meg, BFU-E, and CFU-Mix from the CD34+Thy-1-CD38-/lo fraction expressed AML1/ETO (Table 4, Fig. 4). The morphology of the AML1/ETO+ myeloid colonies and the composition of the cells were indistinguishable from colonies that lack AML1/ETO transcripts (data not shown). Accordingly, the t(8;21)+ myeloid progenitors capable of differentiating into mature myeloid cells exist from the onset of t(8;21) AML.
Figure 4.
AML1/ETO+ myeloid progenitors in the CD34+CD38-/lo fraction of leukemic BM. (A) Morphology of AML1/ETO+ myeloid colonies and AML1/ETO+ leukemic blast colonies derived from single AML1/ETO+ progenitors. These AML1/ETO+ colonies included colonies composed of CFU-L (a), CFU-GM (b), BFU-E (c), and CFU-Meg (d). (B) RT-PCR analysis of cells picked from single cell-derived colonies. a–d correspond to a–d in A. Note that erythrocyte and megakaryocyte colonies did not express MPO gene, which confirms that these colonies did not contain myelomonocytic components. The frequency of these AML1/ETO+ myeloid progenitors was up to 60% in total myeloid colonies as shown in Table 4. GAPDH, glyceraldehyde-3-phosphate dehydrogenase.
Gradual Decrease in AML1/ETO+Myeloid Progenitors During Remission.
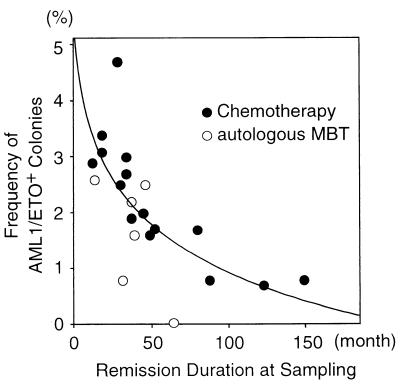
We analyzed the relationship between percentages of AML1/ETO+ myeloid progenitors and remission duration by a Spearman rank correlation analysis; the results shown in Table 1 summarize all of our data here and elsewhere (10). The frequencies of AML1/ETO+ myeloid progenitors decrease along with remission duration in both patient groups treated with autologous MBT and chemotherapy (Fig. 5). In 15 patients treated with chemotherapy who had maintained complete remission for more than 5 years after the sampling, the frequency of AML1/ETO+ progenitors and the duration of remission were inversely correlated (P = 0.0006, r = −0.907).
Figure 5.
Gradual decrease in frequency of AML1/ETO+ progenitors along with remission duration. Twenty-one remission marrow samples from 19 patients who had maintained complete remission >5 years after the sampling were analyzed. Fifteen cases were treated with chemotherapy (●) and four cases were treated with autologous MBT (○). The frequency of AML1/ETO+ myeloid progenitors and remission duration at the time of sampling appeared to be inversely correlated by a Spearman rank correlation analysis (P = 0.0006, r = −0.907 in patients treated with chemotherapy; P = 0.2774, r = −0.486 in patients treated with autologous MBT; P = 0.0007, r = −0.751 in total patients). The correlation curve shows the result in patients treated with chemotherapy.
Discussion
We demonstrate that t(8;21)-specific AML1/ETO mRNA is expressed in mature monocytes and B cells as well as HSC, lymphoid progenitors, and myeloid progenitors during remission. This strongly suggests that t(8;21)+ HSC in remission that are capable of self-renewal differentiate into mature blood cells in vivo.
Fialkow and coworkers (23–25) reported that hematopoiesis in remission might be supported by HSC that share a clonal origin with AML, given the observation that X-chromosome inactivation patterns of glucose-6-phosphate dehydrogenase gene in colony-forming progenitors, mature granulocytes, and B cells in remission frequently were skewed toward those in AML blasts. The “clonal remission” in AML patients suggested that dominant “preleukemic” stem cells capable of normal differentiation may exist, and that additional mutational event(s) may transform these preleukemic stem cells into true leukemic stem cells as a result of a multistep leukemogenesis (26, 27). On the other hand, remission hematopoiesis in t(8;21) AML determined by conventional X-chromosome inactivation analyses is usually polyclonal (10, 28). We have shown that, although a major population of AML1/ETO- myeloid progenitors in remission inactivated either allele of the X-chromosome-linked phosphoglycerate kinase (PGK) gene, t(8;21) AML blasts and AML1/ETO+ myeloid progenitors in remission are of clonal origin because both inactivated the identical allele of PGK gene (10). We demonstrate here that t(8;21) could occur at a stem cell level as a primary event of leukemogenesis. Because the percentages of AML1/ETO-expressing myeloid progenitors and B cells in leukemic phase were significantly higher than those in remission, the t(8;21)+ HSC might be dominant before or at the onset of t(8;21) AML. After treatment, the t(8;21)+ HSC become a minor population, and their frequency gradually declined in remission. These data strongly suggest that additional oncogenic event(s) might be required for the t(8;21)+ HSC to become t(8;21)+ leukemic stem cells.
To interpret “residual diseases” at a molecular level, both the specificity and the sensitivity of assays are important. The residual BCR/ABL in chronic myelogenous leukemia and PML/RARα in t (15, 17) AML occasionally become undetectable in patients maintaining remission, but this does not necessarily indicate their permanent cure (29, 30). In childhood acute lymphoblastic leukemia, persistence of leukemia-specific T cell antigen receptor rearrangements in long-term remission have been reported (31). Our study shows that t(8;21)-specific AML1/ETO does not necessarily mark leukemic cells. Therefore, it is important to know whether the residual cells found in various hematological malignancies are progeny of malignant or nonmalignant stem cells with leukemia-specific genes.
This study also provides quantitative information of residual cells in t(8;21) AML remission; the residual AML1/ETO+ cells originate from a small number of t(8;21)+ HSC, and these decline along with remission duration in patients who had been clinically cured. The gradual fade-out of t(8;21)+ HSC could be caused by host immune surveillance during remission or natural productive lifespan of these clones. Thus, eradication of acute leukemia clones, but not of t(8;21)+ HSC might be required to cure t(8;21) AML patients.
It is of interest that AML1/ETO mRNA was undetectable in T cells throughout the patients' clinical course, and the frequency of AML1/ETO+ cells in B cells was estimated to be ≈10-fold less than that in monocytes. Although it is necessary to confirm the absence of AML1/ETO chimeric DNA in T cells, AML1/ETO may inhibit lymphoid rather than myeloid differentiation (32). Interestingly, cell populations involved in BCR/ABL are similar in chronic myelogenous leukemia patients; BCR/ABL is detectable in all myeloid cells, ≈20% of the mature B cell (33), and rarely in T cells (34–36). It is possible that in normal adults, the demands for lymphoid (especially T cell) regeneration of these long-lived cells may be rare, compared with regeneration of short-lived myeloid cells.
Thus, the formation and expression of the AML1/ETO chimeric gene is not sufficient for leukemic transformation in t(8;21) AML. The AML1/ETO fusion protein may play an important and early step in the process that can lead to leukemic transformation, possibly through suppressing normal AML1 function in a dominant negative manner (37, 38) to cause myelodysplasia (38) or through inhibiting apoptotic cell death of hematopoietic progenitors (39, 40). Although t(8;21)+ HSC are not leukemic, some of their downstream progeny might achieve additional mutation(s) to become dominant before leukemic transformation and to expand to obliterate the niches that support both normal HSC and lymphoid progenitors. Furthermore, because virtually all leukemias with t(8;21) exhibit AML, these additional mutational events might preferentially involve differentiation machinery of the myeloid lineage. Further investigations on the function of the AML1/ETO fusion protein and on additional gene alterations in t(8;21)+ HSC or progenitors are required to understand the cooperative mechanisms among multiple oncogenic events that ultimately lead to the occurrence of t(8;21) AML.
Acknowledgments
We are indebted to Drs. Taiichi Kyo (Hiroshima Red Cross Hospital, Hiroshima, Japan), Mine Harada and Yoshiyuki Niho (Kyushu University, Fukuoka, Japan) for providing or organizing patient samples, and Dr. Nanao Kamada (Hiroshima University, Hiroshima, Japan) for supplying the Kasumi-1 cell line. This work was supported partly by a grant from the Uehara Memorial Foundation to T.M., the Jose Carreras International Leukemia Society (1997) to K.A., and U.S. Public Health Service Grant CA42551 to I.L.W.
Abbreviations
- AML
acute myelogenous leukemia
- HSC
hematopoietic stem cells
- MBT
mobilized blood transplantation
- RT-PCR
reverse transcriptase–PCR
- BFU-E
burst-forming unit-erythroid
- CFU-Meg
colony-forming unit-megakaryocytes
- CFU-GM
colony-forming unit granulocyte/macrophages
- BM
bone marrow
- Lin
lineage
- CLP
common lymphoid progenitors
- MPO
myeloperoxidase
- EBV
Epstein–Barr virus
References
- 1.Hagemeijer A, Garson O M, Kondo K. Cancer Genet Cytogenet. 1984;11:284–287. doi: 10.1016/s0165-4608(84)80007-2. [DOI] [PubMed] [Google Scholar]
- 2.Miyoshi H, Shimizu K, Kozu T, Maseki N, Kaneko Y, Ohki M. Proc Natl Acad Sci USA. 1991;88:10431–10434. doi: 10.1073/pnas.88.23.10431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Shimizu K, Miyoshi H, Kozu T, Nagata J, Enomoto K, Maseki N, Kaneko Y, Ohki M. Cancer Res. 1992;52:6945–6948. [PubMed] [Google Scholar]
- 4.Erickson P, Gao J, Chang K S, Look T, Whisenant E, Raimondi S, Lasher R, Trujillo J, Rowley J, Drabkin H. Blood. 1992;80:1825–1831. [PubMed] [Google Scholar]
- 5.Nucifora G, Larson R A, Rowley J D. Blood. 1993;82:712–715. [PubMed] [Google Scholar]
- 6.Kusec R, Laczika K, Knobl P, Friedl J, Greinix H, Kahls P, Linkesch W, Schwarzinger I, Mitterbauer G, Purtscher B, et al. Leukemia. 1994;8:735–739. [PubMed] [Google Scholar]
- 7.Downing J R. Br J Haematol. 1999;106:296–308. doi: 10.1046/j.1365-2141.1999.01377.x. [DOI] [PubMed] [Google Scholar]
- 8.Liu Yin J A, Tobal K. Br J Haematol. 1999;106:578–590. doi: 10.1046/j.1365-2141.1999.01522.x. [DOI] [PubMed] [Google Scholar]
- 9.Miyamoto T, Nagafuji K, Harada M, Eto T, Fujisaki T, Kubota A, Akashi K, Mizuno S, Takenaka K, Kanaji T, et al. Br J Haematol. 1995;91:132–138. doi: 10.1111/j.1365-2141.1995.tb05258.x. [DOI] [PubMed] [Google Scholar]
- 10.Miyamoto T, Nagafuji K, Akashi K, Harada M, Kyo T, Akashi T, Takenaka K, Mizuno S, Gondo H, Okamura T, et al. Blood. 1996;87:4789–4796. [PubMed] [Google Scholar]
- 11.Kyo T, Dohy H. Acta Hematol Jpn. 1988;51:1641–1652. [PubMed] [Google Scholar]
- 12.Harada M, Akashi K, Hayashi S, Eto T, Takamatsu Y, Teshima T, Hirota Y, Taniguchi S, Nagafuji K, Mizuno S, et al. Int J Hematol. 1997;66:297–301. doi: 10.1016/s0925-5710(97)00037-6. [DOI] [PubMed] [Google Scholar]
- 13.Makino S, Harada M, Akashi K, Taniguchi S, Shibuya T, Inaba S, Niho Y. Bone Marrow Transplant. 1991;8:239–244. [PubMed] [Google Scholar]
- 14.Galy A, Travis M, Cen D, Chen B. Immunity. 1995;3:459–473. doi: 10.1016/1074-7613(95)90175-2. [DOI] [PubMed] [Google Scholar]
- 15.Kondo M, Weissman I L, Akashi K. Cell. 1997;91:661–672. doi: 10.1016/s0092-8674(00)80453-5. [DOI] [PubMed] [Google Scholar]
- 16.Akashi K, Kondo M, von Freeden-Jeffry U, Murray R, Weissman I L. Cell. 1997;89:1033–1041. doi: 10.1016/s0092-8674(00)80291-3. [DOI] [PubMed] [Google Scholar]
- 17.Akashi K, Weissman I L. Immunity. 1996;5:147–161. doi: 10.1016/s1074-7613(00)80491-4. [DOI] [PubMed] [Google Scholar]
- 18.Asou H, Tashiro S, Hamamoto K, Otsuji A, Kita K, Kamada N. Blood. 1991;77:2031–2036. [PubMed] [Google Scholar]
- 19.Koizumi S, Fujiwara S, Kikuta H, Okano M, Imai S, Mizuno F, Osato T. Virology. 1986;150:161–169. doi: 10.1016/0042-6822(86)90275-8. [DOI] [PubMed] [Google Scholar]
- 20.Baum C M, Weissman I L, Tsukamoto A S, Buckle A M, Peault B. Proc Natl Acad Sci USA. 1992;89:2804–2808. doi: 10.1073/pnas.89.7.2804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Uchida N, Sutton R E, Friera A M, He D, Reitsma M J, Chang W C, Veres G, Scollay R, Weissman I L. Proc Natl Acad Sci USA. 1998;95:11939–11944. doi: 10.1073/pnas.95.20.11939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bonnet D, Dick J E. Nat Med. 1997;3:730–737. doi: 10.1038/nm0797-730. [DOI] [PubMed] [Google Scholar]
- 23.Fialkow P J, Singer J W, Adamson J W, Vaidya K, Dow L W, Ochs J, Moohr J W. Blood. 1981;57:1068–1073. [PubMed] [Google Scholar]
- 24.Ferraris A M, Raskind W H, Bjornson B H, Jacobson R J, Singer J W, Fialkow P J. Blood. 1985;66:342–344. [PubMed] [Google Scholar]
- 25.Fialkow P J, Singer J W, Raskind W H, Adamson J W, Jacobson R J, Bernstein I D, Dow L W, Najfeld V, Veith R. N Engl J Med. 1987;317:468–473. doi: 10.1056/NEJM198708203170802. [DOI] [PubMed] [Google Scholar]
- 26.Fialkow P J, Martin P J, Najfeld V, Penfold G K, Jacobson R J, Hansen J A. Blood. 1981;58:158–163. [PubMed] [Google Scholar]
- 27.Fialkow P J, Janssen J W, Bartram C R. Blood. 1991;77:1415–1417. [PubMed] [Google Scholar]
- 28.Guerrasio A, Rosso C, Martinelli G, Lo Coco F, Pampinella M, Santoro A, Lanza C, Allione B, Resegotti L, Saglio G. Br J Haematol. 1995;90:364–368. doi: 10.1111/j.1365-2141.1995.tb05160.x. [DOI] [PubMed] [Google Scholar]
- 29.Seale J R, Varma S, Swirsky D M, Pandolfi P P, Goldman J M, Cross N C. Br J Haematol. 1996;95:95–101. doi: 10.1046/j.1365-2141.1996.d01-1881.x. [DOI] [PubMed] [Google Scholar]
- 30.Faderl S, Talpaz M, Kantarjian H M, Estrov Z. Blood. 1999;93:2755–2759. [PubMed] [Google Scholar]
- 31.Roberts W M, Estrov Z, Ouspenskaia M V, Johnston D A, McClain K L, Zipf T F. N Engl J Med. 1997;336:317–323. doi: 10.1056/NEJM199701303360501. [DOI] [PubMed] [Google Scholar]
- 32.Westendorf J J, Yamamoto C M, Lenny N, Downing J R, Selsted M E, Hiebert S W. Mol Cell Biol. 1998;18:322–333. doi: 10.1128/mcb.18.1.322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.al-Amin A, Lennartz K, Runde V, Seeber S, Opalka B, Kloke O. Cancer Genet Cytogenet. 1998;104:45–47. doi: 10.1016/s0165-4608(97)00435-4. [DOI] [PubMed] [Google Scholar]
- 34.Akashi K, Mizuno S, Harada M, Kimura N, Kinjyo M, Shibuya T, Shimoda K, Takeshita M, Okamura S, Matsumoto I, et al. Exp Hematol. 1993;21:743–748. [PubMed] [Google Scholar]
- 35.Barr R D, Fialkow P J. N Engl J Med. 1973;289:307–309. doi: 10.1056/NEJM197308092890608. [DOI] [PubMed] [Google Scholar]
- 36.Jonas D, Lubbert M, Kawasaki E S, Henke M, Bross K J, Mertelsmann R, Herrmann F. Blood. 1992;79:1017–1023. [PubMed] [Google Scholar]
- 37.Yergeau D A, Hetherington C J, Wang Q, Zhang P, Sharpe A H, Binder M, Marin-Padilla M, Tenen D G, Speck N A, Zhang D E. Nat Genet. 1997;15:303–306. doi: 10.1038/ng0397-303. [DOI] [PubMed] [Google Scholar]
- 38.Okuda T, Cai Z, Yang S, Lenny N, Lyu C J, van Deursen J M, Harada H, Downing J R. Blood. 1998;91:3134–3143. [PubMed] [Google Scholar]
- 39.Klampfer L, Zhang J, Zelenetz A O, Uchida H, Nimer S D. Proc Natl Acad Sci USA. 1996;93:14059–14064. doi: 10.1073/pnas.93.24.14059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Traver D, Akashi K, Weissman I L, Lagasse E. Immunity. 1998;9:47–57. doi: 10.1016/s1074-7613(00)80587-7. [DOI] [PubMed] [Google Scholar]