Abstract
Patients with primary biliary cirrhosis develop progressive ductopenia associated with the production of antimitochondrial antibodies that react with a protein aberrantly expressed on biliary epithelial cells and peri-hepatic lymph nodes. Although no specific microbe has been identified, it is thought that an infectious agent triggers this autoimmune liver disease in genetically predisposed individuals. Previous serologic studies have provided evidence to suggest a viral association with primary biliary cirrhosis. Here we describe the identification of viral particles in biliary epithelium by electron microscopy and the cloning of exogenous retroviral nucleotide sequences from patients with primary biliary cirrhosis. The putative agent is referred to as the human betaretrovirus because it shares close homology with the murine mammary tumor virus and a human retrovirus cloned from breast cancer tissue. In vivo, we have found that the majority of patients with primary biliary cirrhosis have both RT-PCR and immunohistochemistry evidence of human betaretrovirus infection in lymph nodes. Moreover, the viral proteins colocalize to cells demonstrating aberrant autoantigen expression. In vitro, we have found that lymph node homogenates from patients with primary biliary cirrhosis can induce autoantigen expression in normal biliary epithelial cells in coculture. Normal biliary epithelial cells also develop the phenotypic manifestation of primary biliary cirrhosis when cocultivated in serial passage with supernatants containing the human betaretrovirus or the murine mammary tumor virus, providing a model to test Koch's postulates in vitro.
Patients with primary biliary cirrhosis (PBC) develop progressive granulomatous destruction of small intrahepatic bile ducts that often leads to cirrhosis and a requirement for liver transplantation (1). PBC is considered a classical autoimmune disease because of the strong association with other autoimmune disorders, such as Sjögren's syndrome, and the presence of antimitochondrial antibodies (AMA) reactive with the dihydrolipoamide acetyltransferase component of pyruvate dehydrogenase complex (PDC)-E2 (2). In healthy subjects, this complex is located on the mitochondrial inner membrane of all nucleated cells, but in patients with PBC, proteins resembling the autoantigens are present on the plasma membrane of interlobular and proliferating bile duct cells as well as peri-hepatic lymph node macrophages (3). Although an increased and aberrant expression of the AMA reactive protein is seen in cells targeted by the disease process, the transcriptional activity of PDC-E2 in PBC patient's biliary epithelium cells (BEC) is not increased (4).
PBC predominantly occurs in women and has a higher frequency in related and unrelated family members, indicative of both environmental and genetic influences (5). PBC recurs after transplantation with all of the clinical manifestations including the detection of AMA in serum (6), the aberrant expression of the AMA reactive protein on BEC (7), and histologic evidence of disease in up to 45% of patients. In addition, greater immunosuppressive therapy used for transplant recipients is associated with earlier and more aggressive recurrence (1). Accordingly, we have been working on the hypothesis that PBC occurs in susceptible individuals as a result of an infectious agent or environmental trigger and have recently shown that a transmissible factor can promote the aberrant expression of the PDC-E2 like protein in normal BEC, in vitro (8).
In our initial attempts to identify an infectious agent, we performed representational difference analysis using the liver of a patient with PBC. We isolated several human endogenous retrovirus sequences (L.X. and A.M., unpublished data) but found no evidence that these or other human endogenous retroviruses functioned as infectious agents in human autoimmune diseases (9, 10). However, a report documenting the isolation a transmissible retrovirus from patients with Sjögren's syndrome (11), known as the human intracisternal A-type particle (HIAP), provided an impetus to study whether a similar etiology was applicable to PBC. To assess whether PBC patients had serologic evidence of an unrecognized retrovirus infection, we used HIAP proteins to show that 50% of patients with PBC were Western blot positive. This study brought up the possibility that patients with PBC had serologic reactivity to a virus antigenically related to HIAP, and once we derived electron microscopy evidence for viral infection, we directly addressed the hypothesis for a retroviral trigger for PBC. Here, we report evidence for infection with an agent related to the murine mammary tumor virus (MMTV) in PBC patients and show that this agent is associated with the PBC phenotype in vivo and in vitro with aberrant expression of PDC-E2.
Methods
BEC cDNA Library Construction. BEC were extracted from hepatectomy specimens from three patients with PBC and two healthy livers and cultured for 10 days (12). Total RNA was extracted from ≈9.5 × 106 PBC BEC and 25 × 106 normal BEC, reverse transcribed and cloned into λ Uni-ZAP XR cDNA vectors by using a cDNA Gigapack Cloning kit (Stratagene). The PBC BEC cDNA library had an estimated amplified titer of 7.7 × 108 and the normal BEC cDNA library of 8.9 × 108. Both libraries were mass excised by using the helper phage Ex-Assist (Stratagene) as per the manufacturer's instructions.
Virus Cloning and RT-PCR. The cloning of a retroviral pol sequence was performed by using mass excised cDNA library as template for degenerate PCR using a modification of methods described by Shih (13), with primers (5′-IIIIADRTCATCCATRT-3′ and 5′-TGGAAIGTIYTRCCMCARGG-3′) by touchdown PCR: 15 cycles at 94°C for 20 sec, at 58°C with a reduction of 1°C per cycle for 20 sec, and 72°C for 20 sec; followed by 20 cycles of 94°C for 20 sec, 46°C for 20 sec, and 72°C for 20 sec. The PCR product was cloned with the Zero Background cloning kit (Invitrogen), and eight clones were sequenced (GenBank accession nos. AY083214, AF491121–AF491126, and AF491943).
For the RT-PCR of the human betaretrovirus LTR region, total RNA was extracted from frozen tissue by using Trizol (GIBCO/BRL, Grand Island, NY) generating cDNA with random primers. Nested PCR was performed for the human betaretrovirus LTR by using 5′-AGAAATGGTTGAACTCCCGAGAG-3′ and 5′-GGCTTGTAAGAGGAAGTTGGCTG-3′ as external primers, and 5′-TTGTTTCCCACCAAGGACGAC-3′ and 5′-GAAGGTCGAGTTCTCCGAATCG-3′ as internal primers, producing a 216-bp product. Both reactions were performed in 2.5 mM Mg2+ at 94°C, 52°C, 72°C, 20 sec for each temperature in the first PCR and 10 sec each in the second PCR. Nucleic acid extraction, PCR reagent mix, and evaluation of PCR products were performed in three separate laboratories to avoid PCR contamination.
Immunohistochemistry. Frozen lymph node and liver tissues were incubated with 20% normal rabbit serum (NRS) in TBS (pH 7.4) for 20 min, followed by incubation with anti-MMTV p27CA monoclonal antibody (14) diluted 1:5 in TBS containing 10% NRS for 1 h. The samples incubated for 1 h with alkaline phosphatase-labeled anti-mouse antibodies and developed with Fast Red substrate.
For confocal microscopy, tissues were incubated with 1:5 diluted AMA (c150 murine monoclonal AMA reactive to PDCE2; ref. 15) and developed with Fast Red. The tissues were dual labeled with anti-p27CA by using a FITC-conjugated rabbit anti-mouse antibody for 1 h. Control studies were performed with omission of the anti-p27CA antibody to ensure that the FITC-labeled rabbit anti-mouse antibody was not reacting with the Fast Red-labeled c150 AMA.
Coculture Studies. BEC were isolated by established methods (12), plated in six-well plates (Iwaki Glass, Tokyo) at 2–3 × 105 cells per ml and cultured for 24 h in growth medium. The BEC were incubated with lymph node homogenate diluted in human growth factor-free culture medium (0.1 g/ml) for 18–20 h and cultured for a further 5 days, after which the medium was removed, the cells lysed in 0.01% Triton X-100/0.02% EDTA, and both lysates and medium (“conditioned medium”) were stored at -70°C. BEC were incubated with conditioned medium for 18 h, which was replaced with growth medium, and the BEC were incubated for a further 5 days. Conditioned medium was divided into two aliquots and processed by γ irradiation for 16 h with 30 kGy or maintained at room temperature overnight without irradiation and used for the coculture studies. BEC were also incubated with supernatants from MMTV and other control virus producing cell lines in geographically separate laboratory to avoid viral contamination of the human lymph node cocultures and viral cocultures.
BEC were evaluated by immunofluorescence, immunoelectron microscopy and Western blot for PDC-E2 production. Western blots of BEC lysates were performed with anti-PDC E2 affinity purified AMA and anti-CK-19 (anti-cytokeratin 19, DAKO, Ely, UK) as a control to quantify PDC-E2 production (8). These results were expressed as an AMA index: the ratio of the affinity-purified AMA immunoreactivity to BEC after coculture compared with nontreated BEC with levels normalized to CK-19. The effect of the irradiation was expressed as a ratio of the PDC-E2 expression of the irradiated conditioned medium treated cells relative to the cells treated with nonirradiated conditioned medium (see supporting information, which is published on the PNAS web site, www.pnas.org). BEC were also assessed before and after coculture for evidence of viral infection by immunofluorescence with anti-p27CA and RT-PCR.
Supernatants were pooled into three batches from PBC cocultures and three batches from control experiments and evaluated for evidence of virus by negative stain electron microscopy and reverse transcriptase activity by using the Silver methodology (16). These conditioned media were concentrated by ultracentrifugation over a 60% sucrose cushion of at 100,000 × g (27,000 rpm on TST41 rotor) for 4 h at 4°C, and linear sucrose gradients were performed on reverse transcriptase positive samples (if sufficient quantity was available) by layering samples over a 20–60% linear sucrose gradient followed by ultracentrifugation at 100,000 × g for 16 h at 4°C. The density of 25 dripped 500-μl fractions was determined, and each fraction was processed for virus RT-PCR as well as reverse transcriptase activity. A purified sample of FIV was run in parallel as a control for reverse transcriptase activity.
The institutional internal review and ethics boards at all institutions gave permission to perform these clinical studies.
Results
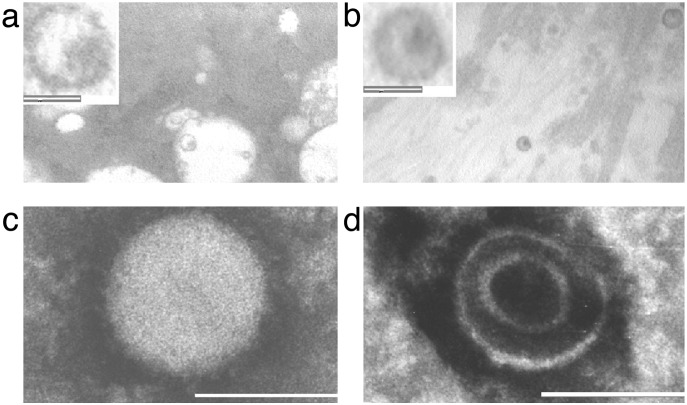
Visualization of Virus-Like Particles in PBC Biliary Epithelium Cells. To find out whether a microbial agent could be directly visualized in samples from patients with PBC, electron microscopy studies were performed by using coded samples of BEC freshly isolated from hepatectomy specimens. Approximately 200–400 BEC were reviewed per patient from three PBC and five control subjects, and virus-like particles were detected in all three of the PBC patients' BEC in ≈1:100 cells. The structures were observed in the extracellular space of BEC, consistent in size and morphology with mature retroviruses; the diameter of the particles varied from 100 to 120 nm, and each had a definable envelope and an electron dense core (Fig. 1). In examination of all five samples of BEC from patients with other liver diseases, just one similar appearing particle was seen.
Fig. 1.
Electron microscopy studies reveal virus-like particles in vivo and in vitro in samples from patients with PBC. (a and b) Freshly isolated biliary epithelial cells from a PBC patient showing virus-like particles in the extracellular space with a distinct envelope and electrondense core (Inset shows particle at ×5 magnification; white bar represents 100 nm). (c and d) Similar particles ranging in size from 110 to 120 nm and showing features consistent with B-type particles (25) were observed in negatively stained pellets from PBC-conditioned media in BEC supernatants. The negative stain has penetrated the structure in d to reveal the nucleoprotein core.
Cloning Exogenous Betaretrovirus Sequences from Patients with PBC. Because the electron microscopy studies suggested a low viral burden, two cDNA libraries were constructed by using BEC extracted from three PBC patients and two normal livers (12, 13, 17). To address the hypothesis that PBC patients' BEC harbored a retrovirus, a PCR cloning methodology with degenerative primers complementary to the highly conserved regions of the retroviral pol gene was used with the mass excised library cDNA as a template (13). A 125-bp PCR product was obtained from the PBC but not the normal BEC cDNA library, and eight clones derived from the PCR product were sequenced. All clones shared 97% homology with each other, and the heterogeneity of nucleotide variability between the eight different clones provided reassurance against a potential single source PCR contamination (GenBank accession nos. AY083214, AF491121-AF491126, and AF491943). blastn searches revealed the highest score with 97% homology to HUMREVTRAC, “human reverse transcriptase gene,” a sequence isolated from human peripheral blood mononuclear cells by using a similar methodology (13). The search also revealed a 95–97% nucleotide sequence homology with multiple MMTV pol genes and 95% similarity with a retroviral pol gene cloned from human breast cancer tissue (18). The blastx search revealed 100% identity with the MMTV Pol protein (see supporting information).
As the PBC pol nucleotide sequence was nearly identical to MMTV, we used a PCR walking methodology with primers complementary to MMTV (L.X. and A.M., unpublished data, see also supporting information) to clone additional viral sequences and derived in total 9,690 bp of the proviral genome (GenBank accession nos. AF513913–AF513923) from a PBC patient's peri-hepatic lymph node DNA. blastn searches showed that the PBC clones shared a variable 91–99% identity with the human proviral sequences isolated from breast cancer tissues as well as the multiple strains of MMTV. The genome organization of the PBC viral sequence is the same as MMTV containing a 5′ and 3′ LTR as well as gag, pro, pol, env, and sag genes with five complete major ORFs encoding the Gag, protease, reverse transcriptase, envelope, and superantigen proteins (L.X. and A.M., unpublished data).
A blastp analysis of the 695-aa sequence translated from the PBC env gene was performed to determine the relatedness of the putative PBC virus with other retroviruses. More than 90% identity was observed with the viral envelope proteins from various subtypes of MMTV and the human breast cancer sequence, with a distant relatedness (<30% identity) with other betaretroviruses such as the ovine pulmonary adenocarcinoma virus and the human endogenous retrovirus K (see supporting information). The proteins identified in the blastp search were subjected to a clustalw phylogenetic analysis; a variable 92–97% amino acid sequence identity was observed with the PBC sequence and other MMTV and human breast cancer proteins, but the human sequences did not cluster together and therefore could not be distinguished as a separate entity from MMTV (see supporting information). Because the human sequence is nearly identical to the murine betaretrovirus, MMTV, we refer to this putative PBC related agent as the human betaretrovirus in accordance with the International Committee on Taxonomy of Viruses (19).
Southern blot and nested PCR studies were performed to assess whether the human betaretrovirus is endogenously encoded in the human genome. When a 4-kb PBC betaretrovirus probe was used, no Southern blot signal was observed to hybridize with DNA from PBMC of five PBC patients and five liver disease controls or hepatic DNA from 12 PBC and eight control subjects. Nested PCR identified proviral sequences in hepatic DNA from 2 of 12 PBC after the second round of amplification and none of eight control patients (see supporting information). Furthermore, blastn searches restricted to Homo sapiens with the PBC betaretroviral sequences showed no comparable homology with human genomic nucleotide sequences >60 bp. In total, these studies suggest that it is highly unlikely that the human betaretrovirus is a human endogenous retrovirus.
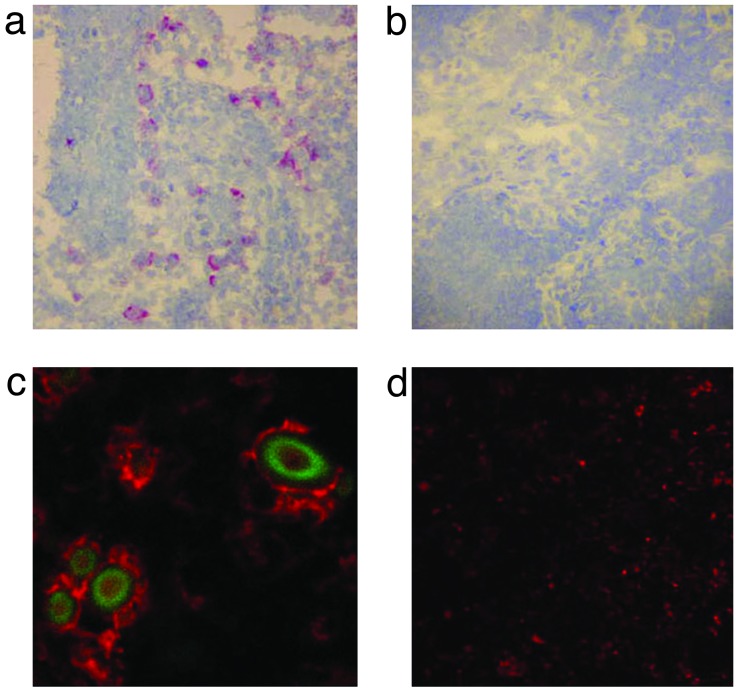
Evidence for Human Betaretrovirus Infection in Vivo. We had previously found that peri-hepatic lymph nodes from PBC harbored a transmissible agent capable inducing the mitochondrial autoantigen expression in normal BEC (8). Accordingly, we assessed lymph node and liver samples from patients undergoing liver transplantation for evidence of betaretrovirus infection by using RT-PCR and immunohistochemistry. When an anti-MMTV p27CA monoclonal antibody was used, no reactivity was observed in liver samples from 12 PBC patients and 12 control subjects. In contrast, in immunochemistry studies using peri-hepatic lymph node samples, 9 of 12 PBC patients had >5% of peri-sinusoidal cells reactive to anti-p27ca as compared with 1 of 11 controls (P < 0.003). Of note, the viral proteins were observed in a similar distribution to the aberrant PDC-E2 expression previously observed in lymph nodes by AMA immunochemistry (20). Accordingly, we performed dual staining for virus and autoantigen and observed viral proteins only in cells with aberrant PDC-E2 expression (Fig. 2). Dual detection of virus and autoantigen was observed in two lymph nodes from three PBC patients and none of the three control lymph node samples (Table 1).
Fig. 2.
Immunohistochemistry studies show viral capsid protein in PBC patient's peri-portal lymph nodes in the same distribution as aberrant autoantigen expression. (a) A PBC lymph node displays a peri-sinusoidal distribution of anti-p27CA reactivity where macrophage/monocyte cells are located. (b) Anti-p27CA reactivity is absent from a control lymph node from a patient with primary sclerosing cholangitis (PSC). (c and d) Aberrant expression of PDC-E2 in the PBC lymph node but not in the PSC lymph node control. Plasma membrane localization of mitochondrial antigen can be seen by fast red stain of AMA in c, but only mitochondrial staining can be seen in d. Cellular colocalization of FITC-stained (green) anti-p27CA in the cytoplasm is observed with aberrant AMA staining in c. (Original magnification: ×100 for a and b and ×400 for c and d.)
Table 1. Increased prevalence of human betaretrovirus infection in patients with PBC.
|
RT-PCR (%)
|
|||
|---|---|---|---|
| Hepatic | Lymph node | Serum | |
| Blood donors | ND | ND | 1/22 (5) |
| Liver disease controls | 2/29 (7) | 3/15 (20) | 2/26 (8) |
| PBC | 14/49 (29)* | 11/15 (73)† | 6/27 (22) |
ND, not determined
P < 0.025 for PBC vs. liver disease controls
P < 0.01 for PBC vs. liver disease controls
The RT-PCR studies also provided evidence to suggest that the peri-hepatic lymph nodes provide the predominant reservoir for human betaretrovirus infection. Betaretroviral cDNA was detected in lymph node samples from 73% of patients with PBC patients as compared with 20% of control subjects with other liver diseases (P < 0.01). In contrast, 29% of PBC patients had detectable betaretroviral RNA in the liver as compared with 7% of liver disease controls (P < 0.025), and viral sequences were infrequently detected in serum. The low frequency of viral detection by RT-PCR in the liver of patients with PBC is consistent with the electron microscopy and immunohistochemistry studies where evidence of virus was rarely encountered in the liver. Thus, it is conceivable that like MMTV, the human betaretrovirus predominantly replicates in dividing lymphocytes and is sparsely represented in hepatic tissue (21).
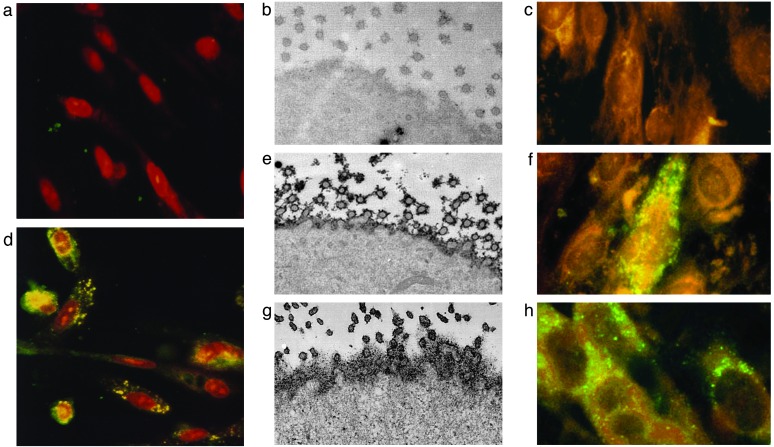
Induction of the PBC Phenotype in Vitro by Betaretrovirus. To test the hypothesis that PBC patients harbor an infectious agent, we cocultured BEC extracted from normal liver with homogenized peri-hepatic lymph nodes from liver transplant recipients for 24 h and then replaced the culture medium. After 7 days in culture, we found that normal BEC incubated with PBC lymph nodes demonstrated increased PDC-E2 by immunoblot as well as AMA immunostaining of plasma membrane (Fig. 3), the phenotypic manifestation of PBC (8, 22, 23). The PBC lymph node treated BEC had a >2-fold increase in AMA index (the ratio of PDC-E2 in treated and untreated BEC), but little change was observed in BEC incubated with liver disease control and normal donor lymph nodes (P = 0.001, Table 2 Group a). A progressive increase with time in AMA reactivity was observed after coculture: no cells were AMA reactive before day 3, whereas 20% of BEC were reactive at day 5 and 70% at day 7.
Fig. 3.
Normal BEC develop the phenotypic manifestation of PBC when incubated with PBC patient's lymph node homogenates. (a–c) Before coculture, studies in normal BEC show no AMA staining by immunofluorescence (a), no cell surface expression of PDC-E2 by immunoelectron microscopy (b), and no evidence of viral proteins by immunofluorescence (c). (d–g) After coculture with PBC patient's lymph node homogenates, the BEC develop aberrant PDC-E2 expression after 7 days in culture (d), with cell surface AMA reactivity on BEC (e) that is similar to that seen in PBC BEC (g), and evidence of cytoplasmic localization of betaretrovirus capsid protein (f). (h) Normal BEC incubated with supernatant from MMTV-producing MM5MT cells also show a similar punctate, cytoplasmic signal from the anti-p27CA immunofluorescence.
Table 2. Induction of PBC phenotype in normal BEC by coculture with PBC lymph node homogenates and pure MMTV isolates.
|
Coculture inoculum with normal BEC
|
||||
|---|---|---|---|---|
| Group | Description | No. of experiments | AMA index* (range) | P value† |
| a | PBC lymph node | 17 | 2.38 (1.2-6.4) | 0.01 |
| Control lymph node | 12 | 0.89 (0.31-1.44) | NS | |
| Healthy lymph node | 10 | 0.78 (0.42-1.64) | NS | |
| b | 5 day supernatant: PBC conditioned media | 4 | 2.19 (1.07-3.14) | 0.04 |
| 5 day supernatant: control conditioned media | 4 | 1.06 (0.93-1.21) | NS | |
| c | 5 day supernatant: PBC conditioned media | 13 | 3.0 (0.4-6.4) | 0.028 |
| Irradiated 5 day supernatant: PBC conditioned media‡ | 4 | 0.96 (0.67-1.24) | NS | |
| d | MMTV | 7 | 2.1 (1.4-2.8) | 0.04 |
| Herpes simplex virus 1 | 3 | 0.99 | NS | |
| Adenovirus 2 | 3 | 0.98 | NS | |
| Coxsackie B4 | 3 | 1.14 | NS | |
AMA induction index was calculated as the ratio of affinity-purified AMA immunoreactivity to BEC after coculture compared to nontreated BEC with levels normalized to CK-19
Calculated by two-tailed Wilcoxon rank test; P value > 0.05 considered not significant (NS)
Irradiated with 30 kGy for 16 h before coculture
The conditioned media (derived from the supernatants taken after 5 days after coculture) were incubated with fresh BEC and only the media derived from PBC patients induced the AMA index (P = 0.04), whereas the conditioned media from control experiments induced little change (Table 2, group b). The PBC conditioned media were subsequently irradiated for 16 h with 30 kGy, which we had found to abolish the infectious activity of 3 control viruses, herpes simplex virus 1 (HSV-1), Coxsackie B4 (CB4), and adenovirus 2 (AV2). After incubation with normal BEC, the PBC supernatants treated with γ irradiation failed to induce PDC-E2 expression, whereas those handled in a similar fashion without irradiation had retained their ability to increase PDC-E2 in BEC (P < 0.03, Table 2, group c), providing evidence that the transmissible factor contained radiation sensitive nucleic acid.
Pooled conditioned media from three PBC and three control lymph node cocultures were examined for evidence of virus by electron microscopy, reverse transcriptase activity and RT-PCR for the human betaretrovirus. In the electron microscopy experiments, negatively stained pellets were examined in a blinded fashion and virus-like particles, consistent in size and morphology to those previously seen in vivo were observed in one of the three PBC-conditioned media and in none of three control-conditioned media (Fig. 1). The particles were 110–120 nm in size, spherical in shape with an eccentric, dense nuclear core (Fig. 1), and thus comparable in morphology to the prototype B-type particle MMTV (24).
When the sensitive Silver assay was used, all three of the conditioned media from PBC cocultures and one of three conditioned media from controls had evidence of reverse transcriptase activity. To determine the buoyant density of particle associated reverse transcriptase activity and human betaretrovirus RNA, linear sucrose gradient studies were performed on two of the PBC samples. Only reverse transcriptase positive fractions form PBC supernatants had RT-PCR evidence of human betaretrovirus. In the first PBC gradient, eight fractions ranging from 1.15 to 1.17 g/ml had reverse transcriptase activity and by RT-PCR, three of the same fractions also had demonstrable human betaretrovirus cDNA (see supporting information). In the second PBC gradient, four fractions ranging from 1.15 to 1.165 g/ml had demonstrable reverse transcriptase activity and three of these fractions had human betaretrovirus cDNA by RT-PCR. None of the three control samples had demonstrable human betaretrovirus RNA by RT-PCR and in one control supernatant, a single fraction was found to be reverse transcriptase positive at 1.24 g/ml where nuclear cores from endogenous retroviruses may be observed. Thus PBC conditioned media contained particles consistent with a B-type morphology as well as particle associated reverse transcriptase activity with betaretrovirus RNA sequences at a buoyant density where enveloped retroviruses usually co-sediment.
As our cloning, in vivo and in vitro studies suggested that the human betaretrovirus shares a common biology with MMTV, we hypothesized that MMTV may be able to induce the PBC phenotype in normal BEC. Accordingly, supernatants from the MMTV producing cell line, MM5MT and from cell lines infected with three control viruses, herpes simplex virus 1 (HSV-1), Coxsackie B4 (CB4), and adenovirus 2 (AV2) were cocultured with normal BEC. Both HSV-1 and AV2 were cytopathic for the BEC, CB4 replicated without evidence for cell lysis and none of the control viruses impacted on the AMA index (Table 2d). In contrast, the MM5MT supernatants induced the AMA index 2-fold in the BEC in a similar fashion to PBC lymph nodes and the PBC conditioned supernatants (P < 0.05, Table 2d).
RT-PCR and immunofluorescence studies were performed in the BEC before and after coculture to assess for evidence of betaretrovirus infection. By RT-PCR, we found no evidence of betaretrovirus in BEC before coculture with lymph nodes and after 7 days, the human betaretrovirus was detected in 7 of 10 BEC derived from PBC lymph node cocultures as compared with 2 of 15 BEC incubated with control lymph node (P < 0.01). When immunofluorescence with FITC-labeled anti-p27CA was used, three of three BEC cocultivated with PBC lymph node homogenates and BEC cocultured with MM5MT supernatants demonstrated green punctate, cytoplasmic signal, whereas untreated BEC and 2 BEC samples cocultured with control lymph nodes showed no reactivity (Fig. 3).
Discussion
We have identified virus-like particles with a B-type morphology from PBC patients; these structures have reverse transcriptase activity, the hydrodynamic properties of enveloped retroviruses and contain betaretrovirus RNA sequences. Although this is the first report demonstrating evidence for betaretrovirus infection in patients with PBC, our findings are concordant with other investigators who have provided immunochemical, serologic, molecular hybridization and electron microscopy evidence of a human betaretrovirus infection in patients with breast cancer and healthy subjects (13, 18, 25–28). Full-length proviral betaretroviral sequences have been cloned from one PBC patients' peri-portal lymph node and two human breast cancer samples and fluorescent in situ hybridization studies have shown betaretrovirus integration in human chromosomes from metastatic breast cancer cells (ref. 18 and unpublished data). However, the human betaretrovirus has not been isolated in culture to date.
The low abundance of the human betaretrovirus nucleic acid in liver, lymph node and breast tissues, high level of sequence homology between isolates, maintenance of ORFs, lack of betaretrovirus sequences in the human genome databases and our lack of detection of betaretrovirus proviral sequences in healthy subjects DNA suggests that the human betaretrovirus is not an endogenous retrovirus. Our inability to detect human betaretrovirus in multiple tissues and the variance of sequences from PCR products in vivo and in vitro suggests that the cloning and RT-PCR prevalence data are unlikely to be related to PCR contamination. No previous studies on human or animal retroviruses had been performed in any of the laboratories before cloning the human betaretrovirus. Also, it should be noted that the variance of individual clones derived from PBC patients (0.3–10.3%), suggests that the PCR products were not amplified from a point source contamination (L.X. and A.M., unpublished data).
The majority of patients with PBC had evidence of betaretrovirus infection in peri-portal lymph nodes. Although human BEC are either permissive or semipermissive for betaretroviral replication, it remains to be determined whether the human betaretrovirus is hepatotrophic as the immunochemistry studies only identified virus in PBC patients' lymph nodes but not liver. In mice, MMTV has limited hepatotrophism and replicates in the gut associated lymphoid tissue before passage in lymphocytes to breast and salivary epithelium (21). Likewise, the human betaretrovirus appears to be preferentially located in lymphoid tissues with a limited viral abundance in the liver, where the end organ damage occurs in patients with PBC. The biology of MMTV infection may also explain the female preponderance seen in patients with PBC as viral replication is regulated in part by a progesterone responsive glucocorticoid regulatory element in the promoter region of the MMTV LTR (29, 30).
Evidence for betaretrovirus infection was also observed in the control populations studied. As PBC clusters in families and occurs in ≈1:50,000 individuals in Europe and North America (1), it is likely that betaretroviral infection is far more common than the development of PBC, which probably only manifests in genetically predisposed individuals. The viral burden observed in the peri-hepatic lymph nodes was one of the notable differences between PBC and control subjects, which appeared far greater in PBC patients by immunochemistry. This suggests that once infected the development of PBC may be related to the extent of viral replication and the ability of the virus to access the liver. Accordingly, if a causal association between betaretrovirus infection and PBC can be established and if normal BEC are susceptible to betaretroviral infection, as reported in this study, the genetic component of the disease may be partially related to limiting the viral replication in lymphoid tissue and preventing spread to the biliary epithelium.
We have shown that normal BEC incubated with PBC patient's lymph nodes develop plasma membrane PDC-E2 expression. This phenotypic manifestation of PBC was associated with human betaretrovirus in BEC, viral particles in pooled supernatants and was induced in a specific fashion by pure MMTV isolates. It has been suggested that the aberrant expression of PDC-E2 in PBC patients is related to the transcellular transport of secretory IgA and PDC-E2 immune complexes from BEC into bile (2). However, this hypothesis is not consistent with our coculture model of PBC as a 2-fold increase in PDC-E2 was repeatedly observed after coculture but no IgA was detected in supernatants (A.K. and J.N., unpublished data, see also supporting information). In addition, once the initial lymph node homogenates were removed after 24 h in coculture, 5 days elapsed before the development of aberrant PDC-E2 expression in BEC; the process was transmissible in serial conditioned media and was abrogated by gamma irradiation. These data are consistent with an infectious process rather than immune complex uptake into cells. The induction of AMA index by MMTV in human BEC also supports the role of an infectious process in autoantigen expression. This was a surprising result, considering the limited capacity of MMTV replication in murine cells and previous difficulties infecting human cells (21).
Our studies lend credence to the hypothesis that PBC has a retroviral trigger but cannot answer the question of whether the virus is present in all patients. We have found evidence of betaretrovirus infection in the majority of patients associated with a limited frequency of detection in the liver; possibly related to low hepatic viral burden and limited sensitivity of methods devised for viral detection. Serologic studies may provide additional data concerning the prevalence of betaretrovirus infection. Moreover, controlled trials with anti-retroviral therapy may provide further support for an etiologic role of retrovirus infection in PBC. An interim analysis of pilot studies using combination anti-retroviral therapy with Lamivudine and Zidovudine showed that PBC patients unresponsive to standard ursodeoxycholic acid therapy had significant reductions in serum liver function tests and a proportion of patients normalized hepatic biochemistry studies (9). A multicenter, randomized controlled trial is now underway and will be conducted as a proof of principal to determine whether antiviral therapy positively impacts on the disease process.
These studies do not directly address a causal relationship between microbe and disease but they support the numerous reports documenting the presence of a betaretrovirus infection in humans (13, 18, 25–28). In this regard, we have devised a coculture model that can be used to test Koch's postulates in vitro by using either pure isolates or transfected DNA of the human betaretrovirus. Further work is required to confirm that this virus is indeed pathogenic in humans and does trigger PBC.
Supplementary Material
Acknowledgments
We thank M. E. Gershwin, T. Golovkina, J. Palmer, G. Loss, and M. Sakalian for supplying reagents and Robert Garry and David Griffiths for providing technical advice. This work was supported by the American Liver Foundation, National Institutes of Health Grant KO8 AI01467, the Hepatitis Research Fund (Alton Ochsner Medical Foundation), the British Liver Trust, and the PBC Foundation, U.K.
Abbreviations: AMA, antimitochondrial antibodies; BEC, biliary epithelium cells; MMTV, murine mammary tumor virus; PBC, primary biliary cirrhosis; PDC, pyruvate dehydrogenase complex.
Data deposition: The sequences reported in this paper have been deposited in the GenBank database (accession nos. AY083214, AF491121–AF491126, AF491943, and AF513913–AF513923).
References
- 1.Neuberger, J. (1997) Lancet 350 875-879. [DOI] [PubMed] [Google Scholar]
- 2.Nishio, A., Keeffe, E. B. & Gershwin, M. E. (2002) Semin. Liver Dis. 22 211-314. [DOI] [PubMed] [Google Scholar]
- 3.Joplin, R. & Gershwin, M. E. (1997) Semin. Liver Dis. 17 97-103. [DOI] [PubMed] [Google Scholar]
- 4.Harada, K., Van de Water, J., Leung, P. S., Coppel, R. L., Nakanuma, Y. & Gershwin, M. E. (1997) Hepatology 25 27-32. [DOI] [PubMed] [Google Scholar]
- 5.Haydon, G. & Neuberger, J. (2000) Gut 47 586-588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Neuberger, J. M., Portmann, B., MacDougall, B., Calne, R. & Williams, R. (1982) N. Engl. J. Med. 306 1-4. [DOI] [PubMed] [Google Scholar]
- 7.Van-de-Water, J., Gerson, L. B., Ferrell, L. D., Lake, J. R., Coppel, R. L., Batts, K. P., Wiesner, R. H. & Gershwin, M. E. (1996) Hepatology 24 1079-1084. [DOI] [PubMed] [Google Scholar]
- 8.Sadamoto, T., Joplin, R., Keogh, A., Mason, A., Carman, W. & Neuberger, J. (1998) Lancet 352 1595-1596. [DOI] [PubMed] [Google Scholar]
- 9.Mason, A. & Nair, S. (2002) Curr. Gastroenterol. Rep. 4 45-51. [DOI] [PubMed] [Google Scholar]
- 10.Lan, M. S., Mason, A., Chen, Q. Y., Vargas, A., Rao, J., Gomez, R., Chalew, S., Garry, R. & Maclaren, N. K. (1998) Cell 95 14-16. [DOI] [PubMed] [Google Scholar]
- 11.Mason, A., Xu, L., Guo, L., Munoz, S., Jaspen, J. B., Bryer-Ash, M., Coa, Y., Sander, D. M., Shoenfeld, Y., Ahmed, A., et al. (1998) Lancet 351 1620-1624. [DOI] [PubMed] [Google Scholar]
- 12.Joplin, R. (1994) Gut 35 875-878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Shih, A., Misra, R. & Rush, M. G. (1989) J. Virol. 63 64-75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Purdy, A., Case, L., Duvall, M., Overstrom-Coleman, M., Monnier, N., Chervonsky, A. & Golovkina, T. (2003) J. Exp. Med. 197 233-243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cha, S., Leung, P. S. C., Gershwin, M. E., Fletcher, M. P., Ansari, A. A. & Coppel, R. L. (1993) Proc. Natl. Acad. Sci. USA 90 2527-2531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Silver, J., Maudru, T., Fujita, K. & Repaske, R. (1993) Nucleic Acids Res. 21 3593-3594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Joplin, R., Hishida, T., Tsubouchi, H., Diakuhara, Y., Ayres, R., Neuberger, J. M. & Strain, A. J. (1992) J. Clin. Invest. 90 1284-1289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Liu, B., Wang, Y., Melana, S. M., Pelisson, I., Najfeld, V., Holland, J. F. & Pogo, B. G. (2001) Cancer Res. 61 1754-1759. [PubMed] [Google Scholar]
- 19.Hunter, E., Casey, J., Hahn, B., Hayami, M., Korber, B., Kurth, R., Neil, J., Rethwilm, A., Sonigo, P. & Stoye, J. (1999) Seventh Report of the International Committee on Taxonomy of Viruses (Academic, London).
- 20.Joplin, R., Lindsay, J. G., Hubscher, S. G., Johnson, G. D., Shaw, J. C., Strain, A. J. & Neuberger, J. M. (1991) Hepatology 14 442-447. [PubMed] [Google Scholar]
- 21.Ross, S. (1997) Adv. Pharmacol. 39 21-46. [DOI] [PubMed] [Google Scholar]
- 22.Joplin, R., Lindsay, J. G., Johnson, G. D., Strain, A. & Neuberger, J. (1992) Lancet 339 93-94. [DOI] [PubMed] [Google Scholar]
- 23.Joplin, R. E., Wallace, L. L., Lindsay, J. G., Palmer, J. M., Yeaman, S. J. & Neuberger, J. M. (1997) Gastroenterology 113 1727-1733. [DOI] [PubMed] [Google Scholar]
- 24.Vogt, V. M. (1997) Retroviral Virions and Genomes (Cold Spring Harbor Lab. Press, Plainview, NY). [PubMed]
- 25.Moore, D. H., Charney, J., Kramarsky, B., Lasfargues, E. Y., Sarkar, N. H., Brennan, M. J., Burrows, J. H., Sirsat, S. M., Paymaster, J. C. & Vaidya, A. B. (1971) Nature 229 611-615. [DOI] [PubMed] [Google Scholar]
- 26.Pogo, B. G. & Holland, J. F. (1997) Biol. Trace Elem. Res. 56 131-142. [DOI] [PubMed] [Google Scholar]
- 27.Wang, Y., Holland, J. F., Bleiweiss, I. J., Melana, S., Liu, X., Pelisson, I., Cantarella, A., Stellrecht, K., Mani, S. & Pogo, B. G. (1995) Cancer Res. 55 5173-5179. [PubMed] [Google Scholar]
- 28.Wang, Y., Go, V., Holland, J. F., Melana, S. M. & Pogo, B. G. (1998) Clin. Cancer Res. 4 2565-2568. [PubMed] [Google Scholar]
- 29.Shyamala, G. & Dickson, C. (1976) Nature 262 107-112. [DOI] [PubMed] [Google Scholar]
- 30.Scheidereit, C., von der Ahe, D., Cato, A. C., Wenz, M., Suske, G., Carlson, C., Bosshard, H., Westphal, H. M. & Beato, M. (1989) Endocr. Res. 15 417-440. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.